Introduction
Cancer remains one of the leading causes of
mortality worldwide and involves the dysregulation of molecular
pathways that control fundamental cellular processes, including
proliferation, apoptosis, migration and invasion. The regulation of
gene expression serves a key role in maintaining cellular
homeostasis and preventing malignant transformation. Accumulating
evidence has highlighted the key role that RNA-binding proteins
(RBPs) serve in orchestrating these regulatory networks,
particularly in cancer development and progression (1). Among RBPs, heterogeneous nuclear
ribonucleoproteins (hnRNPs) represent a particularly important
subfamily that governs RNA metabolism, including transcription,
splicing, stability and translation. The hnRNP family comprises
>20 members, each with distinct structural domains and
functional capabilities. These proteins exhibit notable versatility
in shuttling between the nuclear and cytoplasmic compartments and
participating in diverse cellular processes, ranging from chromatin
remodeling to signal transduction. Evidence suggests that the
dysregulation of hnRNP proteins markedly contributes to oncogenesis
and tumor progression in a number of cancer types (2). hnRNPK has emerged as a promising
member of this family owing to its multifaceted roles and aberrant
expression patterns in numerous malignancies, including gastric
cancer, colorectal cancer, hepatocellular carcinoma, lung cancer,
acute myeloid leukemia and bladder cancer (3). Characterized by its signature
K-homology (KH) domains, hnRNPK functions as a multifunctional
regulator that interfaces with DNA, RNA and protein components. The
ability of hnRNPK to modulate gene expression at multiple levels
serves as a key hub in the molecular networks governing cell fate
decisions. With this, the dynamic subcellular localization of
hnRNPK underscores its potential as a therapeutic target and
biomarker. Despite increasing awareness of the importance of
hnRNPK, key questions remain regarding its dual nature as both an
oncogene and a tumor suppressor in different contexts. The present
comprehensive review summarizes the current knowledge on the
structure, function and clinical importance of hnRNPK in cancer,
while identifying key areas for future investigation of novel
therapeutic strategies.
hnRNP family
Precursor mRNA processing requires the participation
of numerous proteins. At different stages of mRNA processing, these
proteins may form RNP complexes with RNA. Based on the type of
bound RNA, RNP complexes can be classified into three types,
hnRNPs, small nuclear RNP complexes and messenger RNP complexes
(2). RBPs are the primary
components involved in the formation of these complexes, sharing
similar structural features and subcellular distribution patterns,
but differing from other nuclear protein complexes.
The RNP-conserved sequence RNA-binding domain
(RNP-CS-RBD) and KH domain are the two most frequently occurring
RNA-binding domains in hnRNPs. The three-dimensional crystal
structures of these two domains differ notably, suggesting that the
RNA-binding sequences recognized are distinct (4). A stretch of ~80 amino acids, known as
the RNP-CS-RBD or RNA-recognition motif (RRM), contains two highly
conserved sequences, the RNP1 octamer and the RNP2 hexamer, which
are positioned adjacently and directly interact with RNA. Proteins
that bind to RNA typically contain 1–4 RRM. The KH domain, first
identified in hnRNPK, is primarily responsible for recognizing RNA
and single-stranded DNA (5). KH
domains are classified into two subtypes, KH1 and KH2, based on
their extended regions at the N and C termini. RBPs may contain ≤15
KH domains.
hnRNP complexes comprise ≥20 hnRNPs, with molecular
weights ranging from 32–120 kDa. Members of the hnRNP family, A-U,
are named according to their molecular weights. The hnRNP family is
widely distributed and found in plants, yeast and various
vertebrate tissues and organs. Additionally, the expression levels
vary across tissues and organs, with relatively high expression in
metabolically active tissues, such as the brain and lungs.
Expression patterns also vary across developmental stages, even
within the same tissue type (3).
hnRNPs can be divided into two groups based on their physiological
functions, namely those confined to the nucleus, such as hnRNPB and
hnRNPC and those capable of shuttling between the nucleus and
cytoplasm, including hnRNPA, D, E, I and K (5). The survival and growth of
multicellular organisms depend heavily on the presence of hnRNPs.
These proteins interact with chromatin and kinases involved in
transcription, splicing and translation and serve key roles in
chromatin remodeling and gene expression regulation. Tumorigenesis
is associated with alterations in hnRNP activity due to various
factors, including environmental and genetic influences (6). Distinct hnRNPs have unique functions
within the same tumor type. The primary mechanism by which hnRNPs
contribute to cancer is the regulation of tumor-related gene
expression. Table I (7–25)
provides a comprehensive overview of the differential expression
patterns of numerous hnRNP family members in different tumor types.
For example, hnRNPA1 is upregulated in liver and colorectal cancer,
whereas hnRNPA2/B1 shows decreased expression in breast cancer but
increased expression in pancreatic cancer, demonstrating
context-dependent roles.
 | Table I.Expression of hnRNPs in different
tumors. |
Table I.
Expression of hnRNPs in different
tumors.
| First author,
year | hnRNP type | Tumor | Expression | Methods | Mechanism | (Refs.) |
|---|
| Zhou, 2013 | hnRNPA1 | Liver cancer | Increased | RT-qPCR and
IHC | Regulates the level
of CD44v6 | (7) |
| Huang, 2024 | hnRNPA1 | Colorectal
cancers | Increased | Two-dimensional
electro-phoresis and image analysis | Promotes tumor
metastasis | (8) |
| Liu, 2020 | hnRNPA2/B1 | Breast cancer | Decreased | Tissue chips, mouse
models and publicly available data | Inhibits STAT3 and
Wnt/TCF4 signaling pathways | (9) |
| Gu, 2013 | hnRNPA2/B1 | Pancreatic
cancer | Increased | RT-qPCR | Regulates the
levels of E-cadherin and MMP-2 | (10) |
| Huang, 2017 | hnRNPA2/B1 | Gastric cancer | Increased | Proteomics
technique, western blotting, laser confocal microscope and
RT-qPCR | Interacts with
oncogenes and tumor-suppressor genes | (11) |
| Bidot, 2001 | hnRNPA2/B1 | Thyroid
carcinoma | Decreased | IHC | Loss of hnRNPA2/B1
expression appears to be a characteristic feature of thyroid
malignant lesions | (12) |
| Matsuyama,
2000 | hnRNPB1 | Esophageal
cancer | Increased | IHC | Acts as a unique
diagnostic marker with regard to association between expression
level and histopathological grading | (13) |
| Sueoka, 2005 | hnRNPB1 | Lung cancer | Increased | RT-qPCR | Plasma hnRNPB1 mRNA
is a useful non-invasive marker for detection of lung cancer | (14) |
| Tani, 2003 | hnRNPB1 |
Lymphoma/leukemia | Increased | IHC | Process of hnRNPB1
expression in ATLL differs from those in other lymphoid neoplasms
and carcinoma | (15) |
| Dos Santos,
2022 | hnRNPC | Thyroid
carcinoma | Increased | RT-qPCR | Directly affects
the expression of miR-17-92 miRNAs | (16) |
| Wu, 2018 | hnRNPC | Breast cancer | Increased | RT-qPCR | Controls the
endogenous dsRNA and the down-stream interferon response | (17) |
| Howley, 2022 | hnRNPE1 | Breast cancer | Increased | RT-qPCR | Interacts with
ARIH1 | (18) |
| Roychoudhury,
2007 | hnRNPE2 | Oral cancer | Decreased | RT-qPCR | Enhances the
resistance of cancer cells to apoptosis | (19) |
| Li, 2019 | hnRNPF | Bladder cancer | Increased | Proteomic methods
and RNA immunoprecipitation | Mediates the
stabilization of Snail1 mRNA by binding to its 3′UTR | (20) |
| Xu, 2018 | hnRNPF | Gastric cancer | Decreased | RT-qPCR | ECD prevents E3
ligase ZFP91-mediated hnRNPF ubiquitination and degradation | (21) |
| Peng, 2019 | hnRNPK | Pancreatic
cancer | Increased | RT-qPCR | SGLT2 activates the
Hippo signaling pathway through the hnRNPK-YAP1 axis | (22) |
| Peng, 2021 | hnRNPK | Colorectal
cancer | Increased | RNA pulldown, RNA
sequencing and RT-qPCR | Circ-GALNT16 could
enhance the formation of the hnRNPK-p53 complex | (23) |
| Zhang, 2024 | hnRNPM | Breast cancer | Increased | RT-qPCR | Promotes breast
cancer metastasis by activating the switch of alternative
splicing | (24) |
| Chen, 2019 | hnRNPM | Colon cancer | Increased | Proteomic and
bioinformatic analyses |
hnRNPM-IRES-mediated translation in
transforming hypoxia-induced proteome toward malignancy | (25) |
hnRNPK structure
Through alternative cleavage, hnRNPK can freely move
between the cytoplasm and nucleus to create five distinct cleaved
entities, hnRNPK A-E. The majority of the hnRNPK protein comprises
two bidirectional nuclear localization signals (NLS), three
functional regions (KH domains) and one K protein interaction (KI)
region (26). The KH region, a
tertiary structure composed of three helices and three folds
(βααββα), is involved in the binding of DNA or RNA, splicing of
mRNA and control of transcription. The proline-rich KI region
primarily serves as a binding site for sarcoma (Src) kinase family
proteins. The mRNA export process is highly selective. The nucleus
has a bilayer membrane structure and the only channel present is
the nuclear pore complex (NPC). The export of mRNA molecules
through the NPC requires energy and the formation of an
mRNA-protein complex (23). After
forming this complex with the precursor mRNA, hnRNPK transports it
into the cytoplasm through the NPC. Together with additional
proteins, such as RNA polymerase II, it undergoes splicing and
modification before dissociating from the complex and returning to
the nucleus to participate in the subsequent round of transport
(27). The molecular structure of
hnRNPK is illustrated in Fig.
1.
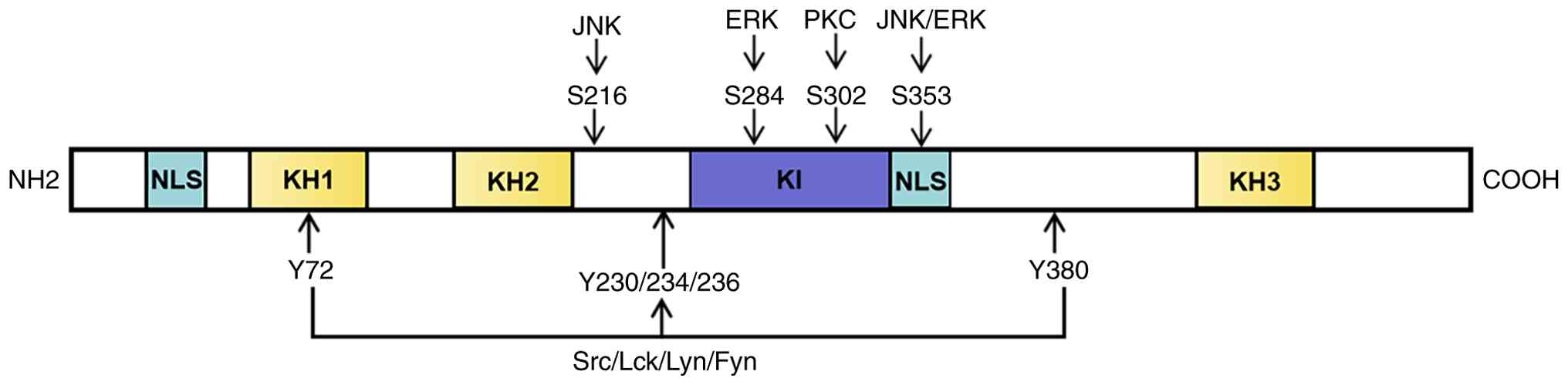 | Figure 1.Molecular structure of heterogeneous
nuclear ribonucleoprotein K. NLS, nuclear-localization signals; KH,
functional regions; KI, protein interaction region; Src, serine
family protein kinases; Lck, tyrosine kinase like protein kinase;
Lyn, non-receptor tyrosine kinase; Fyn, proto-oncogene tyrosine
protein kinase; PKC, phospholipid inositol signal pathway.
NH2, amine group; COOH, carboxyl group. |
hnRNPK function
Functional domains and molecular
interactions
With the presence of the KH, NLS and KI domains,
hnRNPK can bind to DNA, RNA and proteins with specificity. This
allows it to participate in numerous biological processes,
including the regulation of gene transcription, alternative mRNA
splicing, mRNA polyadenylation, mRNA stability, mRNA translation
and cell signal transduction.
Regulation of gene transcription
hnRNPK attaches to specific DNA locations on genes
and participates in the regulation of gene transcription by
interacting with DNA proteins. hnRNPKs serve roles in both
transcriptional activation and inhibition. hnRNPK can bind to the
promoters of numerous genes, including those of the simian
vacuolating virus 40, neuronal nicotinic acetylcholine receptor,
BRCA1, cellular Src (c-Src), cellular Myc (c-Myc) and eukaryotic
translation initiation factor 4E, to increase transcription of the
corresponding genes (28). hnRNPK
can also bind to the human thymidine kinase gene promoter to
inhibit transcription.
Regulation of mRNA variable
splicing
Gene expression is regulated by alternative splicing
of mRNA. Gene function can be enhanced by altering the splicing of
the same DNA segment to produce mRNA, which can subsequently be
translated into proteins that exhibit specific biological effects.
The most notable cell splicing regulators are hnRNPs, which are
found in precursor mRNA and contain splicing enhancement and
inhibition elements that selectively bind to cytokines, to open and
close splicing sites (22). hnRNPK
serves a key role in regulating the variable splicing of
Runt-related transcription factor 1 and synaptosome-associated
protein 25 kDa mRNA during neuronal differentiation. hnRNPK binds
to the enhancer of chicken P-tropomyosin precursor mRNA and
promotes exon splicing, specifically when hnRNPK is combined with a
splicing inhibitor to suppress the synthesis of the
apoptosis-promoting gene Bcl-x short-isoform. Subsequently, ~50% of
alternative splicing events in apoptotic genes are affected
(29).
Regulation of mRNA stability
Cells tightly regulate mRNA stability through
RNA-binding proteins and controlled degradation pathways. The
biological functions of expressed proteins and the half-life of
mRNA are closely related. mRNA stabilization can be achieved
through controlled degradation (30). Currently, four mechanisms of mRNA
decay have been recognized (31),
including the deadenylation-dependent process, endogenous
ribozyme-mediated system, nonsense-mediated pathway and non-stop
degradation pathway. Numerous cis-regions in the mRNA sequence can
be detected and bound by hnRNPs, which affect mRNA stability
primarily through the deadenylation-dependent and nonsense-mediated
decay pathways. Proreninogen mRNA is stabilized and renin
production is aided by the interaction of hnRNPK with the
3′-untranslated region (UTR) of proreninogen mRNA. To improve viral
mRNA stability, the KH domain of hnRNPK interacts with poliovirus
RNA-splicing regulatory components (32). Collagen I and III are expressed
more efficiently when hnRNPK interacts with the 3′-UTRs of their
mRNAs.
Regulation of mRNA translation
hnRNPK is key for controlling cytoplasmic mRNA
translation. The final step in the transition from reticulocytes to
mature erythrocytes is mediated by reticulocyte 15-lipoxygenase
(r15-LOX). Gradual translation of r15-LOX mRNA occurs during cell
development. The 3′-UTR of LOX mRNA contains a differentiation
control element (DICE), which serves a role in regulating this
translation (33). The interaction
between hnRNPK and DICE halts translation and hinders the assembly
of complete 80S ribosomes. hnRNPK functions as a substrate for
c-Src during erythrocyte maturation. To enable the translation of
LOX mRNA, active c-Src phosphorylates hnRNPK and disrupts its
binding to DICE. hnRNPK can bind to the 5′-UTR of the
proto-oncogene Myc in vivo and in vitro, promoting
ribosome entry and enhancing c-Myc translation. In addition to the
DNA or RNA binding involved in signal transduction, hnRNPK
influences the transcription and translation of signaling pathways
by interacting with key signaling proteins, such as Vav and c-Src
(34). The proto-oncogene Vav is a
key regulator of the B cell receptor (BCR) signaling pathway.
According to previous studies (2,6),
hnRNPK binds to the SH3 domain of the Vav protein and promotes cell
transformation through the BCR pathway. c-Src regulates the
MAPK/ERK, integrin/focal adhesion kinase and STAT signaling
pathways, amongst others, serving a role in biological processes
such as cell division and apoptosis (35). Upon hnRNPK binding to the SRC
homology 3 domain of c-Src, c-Src is activated, thereby controlling
downstream signaling molecules.
Regulation of chromatin
remodeling
Chromatin remodeling factors regulate chromatin
structure by altering the position and configuration of nucleosomes
during DNA replication and transcription. These changes affect the
binding of transcription-related proteins to DNA, thereby
controlling gene transcription (36). hnRNPK can directly interact with
DNA methyltransferase, EED (a core component of polycomb repressive
complex 2) and nuclear scaffold attachment factor B to regulate
chromatin remodeling, which, in turn, affects gene expression. The
structural framework that remains in the eukaryotic nucleus after
removal of the nuclear membrane, soluble proteins and chromatin is
known as the nuclear matrix (37).
Nuclear matrix proteins constitute the majority of these
structures, while DNA, RNA and lipids make up the remainder. The
nuclear matrix is important for chromatin remodeling, DNA
replication, gene transcription and post-transcriptional
regulation. Nuclear matrix proteins, including hnRNPK, are key in
maintaining the grid-like structure of the nuclear matrix (38). Changes in the internal structure of
the nuclear matrix can affect various biological processes,
including chromatin remodeling and gene transcription.
Interaction between hnRNPK and
non-coding RNAs (ncRNAs)
hnRNPK exhibits the ability to interact with
numerous ncRNAs and participate in the regulation of various
cellular pathways. Long ncRNAs (lncRNAs), small nucleolar RNAs and
cyclic RNAs are the three primary categories of ncRNAs (39). lncRNAs, which are >200
nucleotides in length, are key for the biological functions of
various types of cancer, including colorectal, hepatocellular,
breast and bladder cancer, serving an important role in disease
etiology and acting as primary regulators of hnRNPK. Table II illustrates how hnRNPK interacts
with cellular processes and contributes to the regulation of
protein-coding gene networks. These interactions include: i)
lncRNA-hnRNPK interactions, such as lncRNA-p21, Tcl1 upstream
neuron-associated lncRNA, lncRNA essential for naïve ESC
self-renewal 1, promoter-associated noncoding RNA of ETS1, Ewing
sarcoma-associated transcript 1, cancer susceptibility candidate
11, MYC-inducible lncRNA 2 and lncRNA91H, which regulate gene
transcription; ii) the regulation of mRNA stability and translation
by lncRNA-hnRNPK interactions, including c-Myc-upregulated lncRNA,
translation regulatory lncRNA and linc0046660; iii) the promotion
of lncRNA nuclear localization, as observed with short interspersed
nuclear element-derived nuclear RNA localization; iv) the
regulation of genes involved in X-inactive specific transcript
activity through lncRNA-hnRNPK interactions; and v) hnRNPK-mediated
alternative splicing of lncRNAs such as nuclear paraspeckle
assembly transcript 1 (40).
Table II (26,27,41–53)
summarizes the specific mechanisms underlying these interactions
and highlights the diverse roles of hnRNPK in the regulatory
networks of protein-coding genes. Notable examples include MYU
stabilizing CDK6 expression in the cytoplasm, CTHCC activating YAP1
transcription in the nucleus and CASC11 promoting the Wnt/β-catenin
pathway.
 | Table II.Mechanism of interaction between a
number of lncRNAs and hnRNPK. |
Table II.
Mechanism of interaction between a
number of lncRNAs and hnRNPK.
| First author,
year | lncRNA type | hnRNPK
location | Function | Mechanism | (Refs.) |
|---|
| Kawasaki, 2016 | MYU | Cytoplasm | Promotes cell
proliferation | Stabilizes CDK6
expression | (41) |
| Xu, 2019 | treRNA | Cytoplasm | Promotes cell
proliferation | Inhibits epithelial
cadherin | (26) |
| Xia, 2022 | CTHCC | Nucleus | Promotes cell
proliferation and invasion | Activates YAP1
transcription | (42) |
| Huarte, 2010 | LincRNA-p21 | Nucleus | Promotes cell
proliferation and inhibits p53 mediated apoptosis | Transcriptional
regulation | (43) |
| Lin, 2014 | TUNA | Nucleus | Promotes stem cell
differentiation | Activates multiple
signal paths | (44) |
| Li, 2018 | pancEts-1 | Nucleus | Promotes cell
proliferation and invasion | Activates β
Annexin | (45) |
| Xi, 2024 | ELF3-AS1 | Nucleus | Promotes cell
proliferation and invasion | Regulates the
downstream target gene, C-C motif chemokine 20 | (46) |
| Wang, 2022 | CRLM | Nucleus | Promotes metastasis
and regulating gene expression | Associated with the
chromatin regions of genes involved in cell adhesion and DNA
damage | (47) |
| Lee, 2021 | LINC00263 | Nucleus | Promotes cell
proliferation and invasion | Acts as a miR-147a
decoy and thus upregulating CAPN2 | (27) |
| Ji, 2020 | LINC01413 | Nucleus | Facilitates cell
proliferation, migration, invasion and EMT | LINC01413 as a
positive regulator through the LINC01413/hnRNP-K/TAZ1/YAP1/ZEB1
axis | (48) |
| Zhang, 2016 | CASC11 | Nucleus | Promotes cell
proliferation and invasion | Activates the Wnt/
β Annexin pathway | (49) |
| Gu, 2019 | LBCS | Nucleus | Activates androgen
receptor | LBCS interacted
directly with | (50) |
|
|
|
| signaling | hnRNPK to suppress
and |
|
|
|
|
|
| rogen receptor
translation |
|
| Gu, 2019 | lncRNA-LBCS | Nucleus | Inhibits tumor drug
resistance | Inhibits SOX2
transcription | (50) |
| Pintacuda,
2017 | Xist | Nucleus | Interacts with X
chromosome | Modifies
chromosome | (51) |
| Gao, 2018 | lncRNA 91H | Exosomes | Promotes tumor
occurrence and metastasis | Regulates the
expression of HnRNPK | (52) |
| Peng, 2020 | FAM84B-4 | Nucleus | Promotes
tumorigenesis | Lnc-FAM84B-4
regulates | (53) |
|
|
|
|
| MAPK pathway by
restraining |
|
|
|
|
|
| DUSP1
expression |
|
Biological function of hnRNPK in tumors
Overview of the roles of hnRNPK in
cancer
hnRNPK controls the expression of numerous oncogenes
and tumor suppressor genes in malignancies, as well as the
proliferation, apoptosis, migration and invasion of tumor cells.
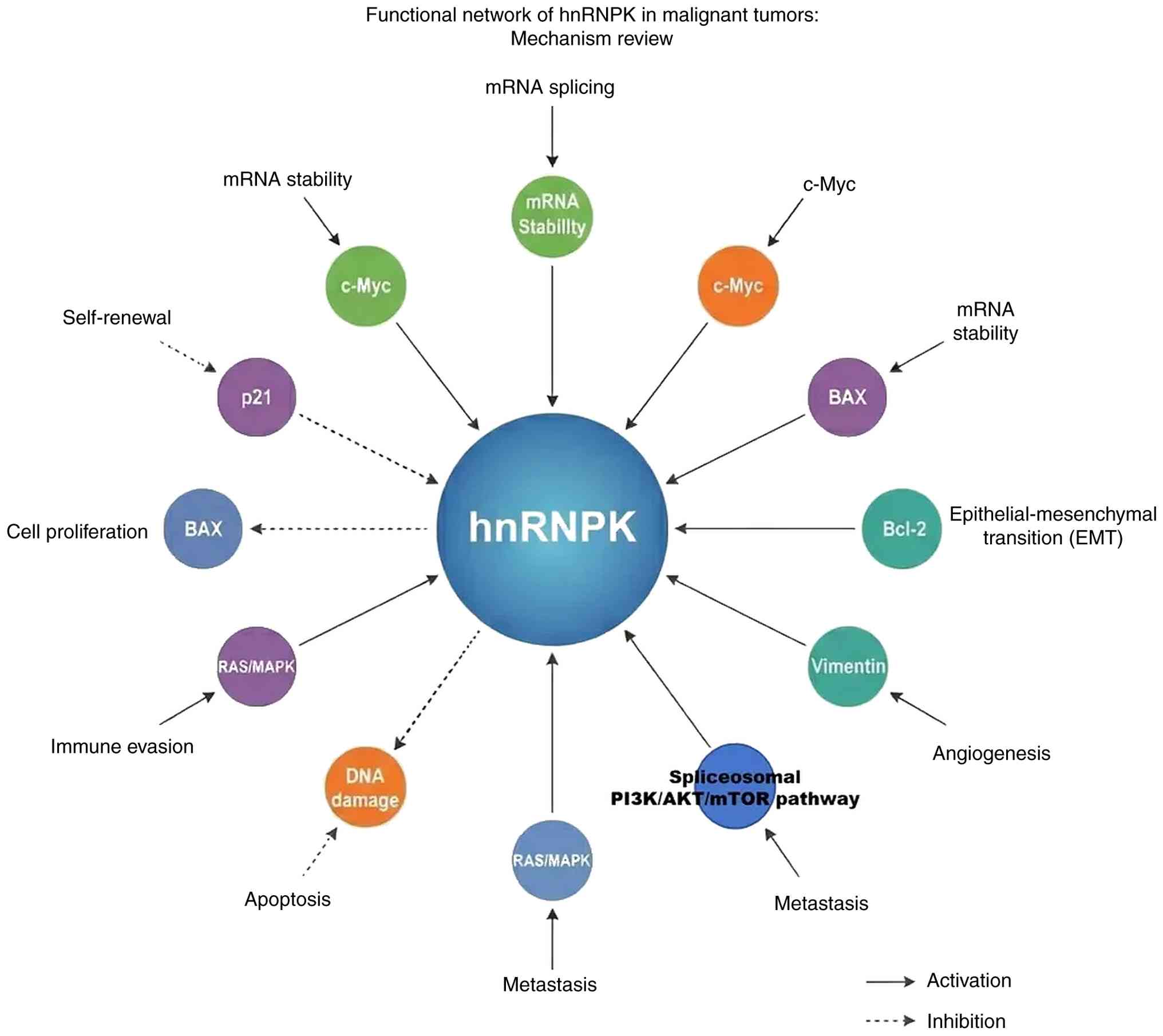
Fig. 2 presents a comprehensive
overview of the hnRNPK functional network in malignant tumors,
illustrating its multifaceted roles in cancer biology through a
number of molecular mechanisms and signaling pathways.
hnRNPK and tumor cell
proliferation
In certain malignancies, hnRNPK has been implicated
in the regulation of tumor growth. A previous study suggested that
hnRNPK controls the p53/p21/cyclin-D1 axis to suppress tumor cell
proliferation, colony formation and tumor progression in gastric
cancer cells (11). An association
has also been observed between hnRNPK expression and poor prognosis
in patients with bladder cancer (54). The primary mechanism of this
involves the regulation of cyclin-D1, a key cell cycle protein, by
hnRNPK to promote the proliferation and survival of bladder cancer
cells. The human telomerase reverse transcriptase and c-Myc genes,
which are involved in tumor cell proliferation, are also closely
associated with hnRNPK (55).
hnRNPK and tumor apoptosis
One of the key hallmarks of cancer is its ability to
evade apoptosis. hnRNPK regulates tumor apoptosis through a number
of pathways. Tumor necrosis factor-related apoptosis-inducing
ligand (TRAIL) is a novel and efficient therapeutic agent that
targets tumor cell death (56).
The NF-κB pathway, which is implicated in apoptosis, is activated
by phosphatidylinositol signaling and can induce apoptosis in
non-small-cell carcinomas. Additional research has revealed that
TRAIL treatment of H1299 cells promotes hnRNPK accumulation and
induced apoptosis. However, ERK1/2 inhibitors and ERK
phosphorylation receptor mutations were shown to reduce
TRAIL-induced cytoplasmic accumulation of hnRNPK and act as
anti-apoptotic agents (57).
hnRNP, DNA repair and tumor
formation
If cellular DNA is not repaired promptly after
exposure to ionizing radiation, chemotherapeutic agents or other
external stimuli, it can lead to chromosomal remodeling, gene loss
and genome instability, thereby promoting cancer development
(47). Aberrant hnRNPK expression
in tumor cells has been shown to impair DNA repair capacity. When
the expression of hnRNPK was downregulated in irradiated bronchial
epithelial cells, DNA strand repair was notably impeded. Proteomics
analysis has shown that hnRNPK binds to chromatin in response to
DNA damage in HeLa cells (58).
hnRNPK and tumor angiogenesis
Tumor metastasis occurs through direct extension,
hematogenous and lymphatic spread and implantation of tumor cells.
The early diffusion stage involves the adhesion plaque complex
precursor, in which hnRNPK is expressed. Disruption of hnRNPK
synthesis increases tumor cell metastasis, indicating that hnRNPK
serves a rate-limiting role in tumor growth (59). The relationship between hnRNPK and
metastasis has been established in colon, prostate and gallbladder
cancer types. Notably, tumor size typically does not exceed 2–3 mm
without blood supply and the angiogenic agents that facilitate this
include vascular endothelial growth factor (VEGF) and fibroblast
growth factor. Therefore, hnRNP proteins have been shown to
regulate angiogenic factors and under hypoxic conditions, hnRNPL
preferentially binds to the 3′-UTR of VEGF mRNA (60). Inhibition of hnRNPK expression
suppresses glioma growth and invasion, suggesting that hnRNPK
promotes tumor angiogenesis and facilitates malignant
progression.
hnRNPK and tumor cell migration and
invasion
hnRNPK is further associated with tumor metastasis.
Through the Ras-Raf-MAPK pathway, hnRNPK promotes the expression of
metastasis-related genes, such as MMP3 and MMP10, prostaglandin G/H
synthase 2 and thymosin. Ras and cholecystokinin expression further
promotes tumor spread. DAB2-interacting protein stimulates hnRNPK
nuclear accumulation, enhances MMP2 transcription and drives
colorectal cancer invasion and metastasis through the MAPK/ERK
pathway (61). hnRNPK also
promotes tumor spread in nasopharyngeal cancer by upregulating
MMP12 expression. Strozynski et al (62) found that hnRNPK was markedly
expressed in irradiated cells using two-dimensional electrophoresis
and mass spectrometry.
Targeted inhibition of hnRNPK expression can reduce
the metastatic potential of head and neck squamous cell carcinoma
cells. In small-cell renal carcinoma, cytoplasmic aggregation of
hnRNPK promotes tumor cell invasion into surrounding tissues.
hnRNPK deletion results in transcriptional inactivation of p53
target genes and defective cell cycle arrest. DNA damage-induced
hnRNPK undergoes small ubiquitin-related modifier (SUMO)-ylation,
which regulates p53 transcriptional activation (63). Additionally, methylation of
arginine residues at positions 296 and 299 inhibits the
phosphorylation of serine at position 302 by the pro-apoptotic
protein kinase C, thereby reducing apoptosis induced by DNA damage.
This suggests that hnRNPK serves a key role in anti-apoptotic
mechanisms in tumor cells. Chen et al (25) demonstrated that hnRNPK exhibits
anti-apoptotic activity by regulating downstream genes.
Specifically, hnRNPK binds to the promoter of anti-apoptotic FLICE
inhibitory protein and activates its expression. The lncRNA CASC11
interacts with hnRNPK to activate the WNT/β-catenin pathway,
ultimately contributing to colorectal cancer development (43).
hnRNPK and drug resistance of tumor
cells
hnRNPK has been shown to be associated with drug
resistance in tumor cells. After radiotherapy, hnRNPK increased in
a dose-dependent manner and accumulated in the cytoplasm of
melanoma cells with neuroblastoma RAS viral oncogene homolog
mutations, causing the cells to become radiotherapy-tolerant
(64). Mitogen-activated
extracellular signal-regulated kinase (MEK) inhibitors can
downregulate hnRNPK expression, which, when combined with
radiation, markedly increases apoptosis and promotes
radiosensitivity (65). Targeted
inhibition of hnRNPK expression was found to be consistent with the
pro-apoptotic effects of MEK inhibitors. A similar conclusion was
reached in colorectal cancer (CRC) cells. Kirsten rat sarcoma
virus-mutant CRC cells exhibited rapid upregulation of hnRNPK
following radiation therapy, which increased their tolerance to
irradiation. MEK inhibitor therapy downregulates hnRNPK, improving
sensitivity to radiation (66).
hnRNPK is highly expressed in resistant cell lines and the bone
marrow of patients with drug-resistant acute myeloid leukemia
(AML). Drug-resistant cells lose their tolerance to doxorubicin
through targeted suppression of hnRNPK expression. In addition,
hnRNPK may contribute to doxorubicin resistance by regulating
autophagy.
hnRNPK may serve as a prognostic and
chemosensitivity marker in nasopharyngeal carcinoma, as it
regulates thymidine phosphorylase expression. Cells with high
thymidine phosphorylase levels are sensitive to doxifloruridine
treatment (67).
Interferon-stimulated gene 15 (ISG15) regulates hnRNPK expression
in lung cancer cells. ISG15 downregulation induces cell cycle
arrest, allowing extended repair of cisplatin-damaged DNA,
stabilization of p53 and increased hnRNPK expression. This process
can enhance cisplatin resistance. In lung adenocarcinoma cells,
hnRNPK blocks the phosphorylation of glycogen synthase kinase-3 at
Ser9 to stabilize cellular FLICE inhibitory protein and increase
TRAIL resistance. Zhang et al found that hnRNPK increases
AML resistance to adriamycin through modulation. These findings
suggest that hnRNPK regulates tumor cell sensitivity to
chemotherapy and may serve as a marker for chemosensitivity
(68).
Tumor therapy targeting hnRNPK
In Traditional Chinese Medicine, certain therapeutic
compounds can target hnRNPK to exert antitumor effects. Further
research using bioinformatics and biochemical methods revealed that
the ethanol extract of Indian ginseng serves a role in inhibiting
tumor metastasis and angiogenesis by downregulating
metastasis-related proteins, including hnRNPK, VEGF and MMPs
(69). The ethanol extract of
Indian ginseng selectively inhibits tumor cell activity and
suppresses metastasis, invasion and angiogenesis. Gambogic acid in
Garcinia nujiangensis extract can lower hnRNPK levels by
promoting the ubiquitin-proteasome-dependent degradation of hnRNPK,
leading to cell cycle arrest and antitumor effects. The protein
hnRNPK, associated with human telomerase reverse transcriptase, is
a potential biomarker for liver cancer prognosis and may also serve
as a therapeutic target for liver cancer (70). The primary antigen target is
located at the N-terminus of hnRNPK and contains a glutamic
acid-rich domain. Therefore, hnRNPK may be used as a biomarker for
the detection of hepatocellular carcinoma associated with hepatitis
B virus (HBV). hnRNPK is a biomarker of chemoresistance in gastric
cancer (GC). Additionally, upregulation of hnRNPB1 has been
identified as a useful biomarker for the early diagnosis of lung
cancer and human squamous cell carcinomas. While these preclinical
findings are promising, it is important to note that
hnRNPK-targeted therapeutic development remains in early stages.
Unlike established targets such as HER2 or EGFR, to the best of our
knowledge, no hnRNPK-specific inhibitors have yet entered clinical
trials. The primary challenge lies in the multifunctional nature
and context-dependent role of the protein, which complicates the
development of selective therapeutic strategies. Current research
focuses primarily on understanding the mechanistic roles of hnRNPK
across different cancer types to identify optimal intervention
points.
Expression and prognostic value of hnRNPK in
malignant tumors
Although hnRNPK contributes markedly to the
occurrence and progression of specific cancer types, it still
remains unclear as to whether it functions as an oncogene or a
tumor suppressor gene. The ability of hnRNPK to regulate both
carcinogenesis and tumor-inhibitory pathways has been demonstrated
in numerous studies (49,71). Cell proliferation and apoptosis
inhibition have been linked to the upregulation and downregulation
of hnRNPK. Clinical reports present varying perspectives. hnRNPK
functions as an oncogene in gastric, colorectal, nasopharyngeal,
prostate, melanoma and oral squamous cell carcinoma (OSCC).
Upregulation of this gene was found to be positively associated
with tumor progression and a poor prognosis. Conversely, hnRNPK has
been shown to act as a tumor suppressor gene in acute myeloid
leukemia (72). Mice with
haploinsufficient hnRNPK expression are more susceptible to
lymphoma and acute myeloid leukemia. Therefore, hnRNPK cannot be
categorized solely as an oncogene or tumor suppressor gene based on
these cytological and clinical findings. The paradoxical dual
nature of hnRNPK appears to be determined by several key factors:
i) Cellular context and tissue type, where gastric epithelial cells
show tumor-suppressive responses while mesenchymal-derived cancers
exhibit oncogenic effects; ii) subcellular localization, with
nuclear hnRNPK often being protective while cytoplasmic
accumulation promotes malignancy; iii) expression levels, where
both upregulation and haploinsufficiency can promote tumorigenesis
through different mechanisms; iv) post-translational modifications
including phosphorylation, SUMOylation and methylation that
modulate the protein interactions and functions of hnRNPK; and v)
interaction partners, particularly p53 status, which fundamentally
alter the role of hnRNPK in cell fate decisions. Table III summarizes these
context-dependent factors including cancer type, hnRNPK function
(oncogene vs. tumor suppressor), expression levels, key regulatory
mechanisms and clinical outcomes (6,11,26,33,55,61,68).
 | Table III.Context-dependent dual roles of
hnRNPK in cancer. |
Table III.
Context-dependent dual roles of
hnRNPK in cancer.
| First author,
year | Cancer type | K function | Expression
level | Key mechanisms | Regulatory
factors | Clinical
outcome | (Refs.) |
|---|
| Huang, 2017 | Gastric cancer | Tumor
suppressor | Low | p53/p21/CCND1
pathway activation | p53
interaction | Poor prognosis with
low expression | (11) |
| Zhang, 2016 | Acute myeloid
leukemia | Tumor
suppressor |
Low/haploinsufficient | Cell cycle
regulation | SUMO
modifications | Increased
susceptibility in knockout mice | (68) |
| Wu, 2020 | Colorectal
cancer | Oncogene | High | MAPK/ERK activation
and MMP2 upregulation | DAB2IP
interaction | Poor prognosis with
high expression | (61) |
| Li, 2020 | Nasopharyngeal
cancer | Oncogene | High | MMP12
upregulation | Post-translational
modifications | Associated with
metastasis | (6) |
| Li, 2019 | Lung cancer | Oncogene | High | Cytoplasmic
accumulation | Subcellular
localization shift | Associated with
invasion | (55) |
| Xu, 2019 | Bladder cancer | Oncogene | High | CCND1 regulation
and ERCC4 modulation | Nuclear
localization | Poor prognosis | (26) |
| Meng, 2021 | Hepatocellular
carcinoma | Oncogene | High | c-Myc activation
through IRES | HBV
interaction | Associated with
HBV+tumors | (33) |
Chronic myeloid leukemia (CML)
CML, specifically the late acute phase, has a poor
prognosis. With this, research into hnRNPK and its family members
during the acute phase of CML is relatively common. Other
chromosomal and molecular abnormalities are also present in
patients with CML, in addition to the aberrant BCR/ABL fusion gene
(73). Together with other
defective genes, the BCR/ABL gene can influence transcription,
protein function and mRNA translation, and improperly abnormally
activate downstream signaling pathways, thereby contributing to
disease progression. The ability of hnRNPK, hnRNPE1 and hnRNPE2 to
limit the proliferation of BCR/ABL-positive cells is inhibited by
the upregulation of CML-blast crisis expression (74). The leukemogenic activity of
BCR/ABL, which can increase c-Myc gene expression, enhance CML cell
proliferation, block apoptosis and potentially promote rapid
transformation, relies heavily on hnRNPK-mediated regulation of
mRNA translation. The homologous region of the hnRNPK is the
structural motif shared by hnRNPK and hnRNPE1/E2. The structural
basis of the hnRNPK-mRNA interaction is described as follows.
MAPK/ERK1/2 can enhance hnRNPK transcription and mRNA stability in
bone marrow cells and lymphocytes expressing the BCR/ABL fusion
gene through a BCR/ABL-dependent mechanism (75). Leukemia can be induced by hnRNPK,
which inhibits cytokine-dependent colony formation in
BCR/ABL-positive cells. hnRNPK binds to MYC mRNA through the
internal ribosome entry site (IRES) and upregulates MYC expression
at both the transcriptional and translational levels, ultimately
promoting cell proliferation and inhibiting apoptosis in
hepatocellular carcinoma cells. These effects may be linked to the
dysregulation of the oncogene, MYC (76). To identify hnRNPK expression at the
protein and transcriptional levels in the bone marrow cells of
patients with CML in the chronic and acute phases, Zhu et al
(77) used western blotting and
reverse transcription-quantitative PCR techniques, finding that
hnRNPK expression varied before and after the acute phase of CML,
indicating that mRNA translation regulation may underlie changes in
hnRNPK protein levels.
Lung cancer
Although hnRNPK expression was not observed during
the aberrant proliferation of healthy alveolar and bronchiolar
epithelial cells, it was slightly upregulated in cells with
bronchial epithelial dysplasia (78). The localization of hnRNPK was also
shown to gradually shift from the nucleus to the cytoplasm in the
study by Huang et al (79)
on lung cancer cell lines, suggesting that this shift is associated
with the biological state of tumor cells. According to Li et
al (55), hnRNPK was expressed
in both the cytoplasm and nucleus of lung cancer tissues and
control lung tissues. These findings suggest that the hnRNPK
positivity rate in lung cancer tissues is higher than that in
non-cancerous lung tissues. A total of three histological subtypes
of lung cancer stained positive for hnRNPK and the positivity rates
for small cell and non-small cell lung cancers did not differ
significantly. Although hnRNPK was notably expressed in lung cancer
tissues, there was no clear association between hnRNPK expression
and the tissue type. In addition, this study found that invasive
and metastatic lung cancer tissues showed notable levels of hnRNPK
expression, which may indicate a connection between hnRNPK and
tumor aggressiveness. However, the precise role of hnRNPK in lung
cancer initiation, progression and metastasis remains elusive.
GC
In accordance with findings by Huang et al
(8), hnRNPK is a useful prognostic
marker in patients with GC. Han et al (80) discovered that hnRNPK is a
GC-related antigen, with tissue microarray analysis revealing that
hnRNPK expression was elevated in GC tissue. Patients with high
hnRNPK expression exhibited a poor prognosis, suggesting that
hnRNPK may be associated with GC occurrence, progression and
prognosis. Poor prognosis in GC is linked to low hnRNPK
transcription levels, particularly in patients with early-stage
disease without metastasis. Through the p53/p21/cyclin D1 pathway,
hnRNPK upregulation decreases tumor cell proliferation and colony
formation in vitro and tumor growth in vivo (81). hnRNPK interacts with
tumor-associated genes, including p53 and p21. High hnRNPK
expression has been observed in GC tissue. Infection with the
L-form of Helicobacter pylori may promote the expression of
hnRNPK. The expression of hnRNPK and Helicobacter pylori
L-form infection may work together to increase the risk of GC. The
degree of differentiation, lymphatic metastasis and clinical stage
of GC are associated with hnRNPK expression (82). Research has demonstrated that
hnRNPK is primarily expressed in the nucleus of human GC SGC-7901
cells, with a minor quantity expressed in the cytoplasm of in
vitro cultured gastric mucosal cell lines and GC SGC-7901 cells
(53). The expression of hnRNPK
was found to be higher in human GC SGC-7901 cells than in gastric
mucosal gastric epithelial-1 cells, both in the cytoplasm and
nucleus. hnRNPK was also marginally expressed in the nucleus of
gastric mucosal GES-1 cells, but not in the cytoplasm of these
cells. Knockdown of hnRNPK reduced the proliferation, migration and
invasion of human GC SGC-7901 cells. The cytoplasmic localization
and elevated expression of hnRNPK in SGC-7901/DDP cells indicated
that hnRNPK was associated with drug resistance in human GC
SGC-7901 cells.
Liver cancer
To demonstrate that hnRNPK is primarily expressed in
the nucleus, Meng et al (33) examined hnRNPK expression in
hepatocellular carcinoma and adjacent tissues. Findings revealed
that hnRNPK protein expression was higher in 70% of hepatocellular
carcinoma tissues than in the corresponding adjacent tissues
(83), suggesting that it promotes
liver cancer cell development. By separating the cytoplasm and
nucleus of hepatoma cells at various densities, changes were
observed in hnRNPK expression during cell proliferation. These
findings indicate that hnRNPK expression in the nucleus increases
with higher cell density, suggesting that elevated nuclear hnRNPK
expression may promote cell proliferation (84). In addition, while only 50% of
HBV-negative liver cancer tissues showed higher hnRNPK expression
than their adjacent tissues, 80% of HBV-positive liver cancer
tissues did, indicating an association between hnRNPK expression
and HBV infection. Further research is needed to determine whether
hnRNPK has a synergistic effect with HBV in promoting liver cancer
development and whether HBV upregulates hnRNPK expression in liver
cancer tissues. Harris et al (85) demonstrated that hnRNPK can interact
with the 3′-UTR of the hepatitis C virus and participate in viral
replication. The IRES, located at the 3′ end, is a key regulatory
element in viral gene translation. IRES and hnRNPK work together to
notably enhance the mRNA translation efficiency, thereby increasing
viral expression and replication, thus accelerating disease
progression.
OSCC
With regard to OSCC, researchers used
isotope-labeled relative and absolute quantitative techniques
combined with liquid chromatography-mass spectrometry and found
that hnRNPK protein expression increased with higher tumor-lymph
node-metastasis tumor staging levels and was associated with poor
prognoses. Therefore, hnRNPK has the potential to be a useful
marker for the early detection and prognostic monitoring of OSCC
(86). N6-methyladenosine (m6A)
levels in OSCC tissues were notably higher than those in adjacent
non-tumor tissues and eight m6A-modified genes, including hnRNPC,
exhibited differential expression patterns. HnRNPC alone may serve
as a standalone biomarker and therapeutic target in OSCC. Compared
with healthy oral mucosal tissues, OSCC tissues exhibit markedly
elevated levels of hnRNPL, which is primarily concentrated in
discrete nuclear regions, forming a punctate structure (87). The expression of hnRNPL was higher
in mesenchymal tissues than in epithelial tissues. A novel target
of hnRNPL, Ser/Arg-rich splicing factor 3, may be regulated by
hnRNPL at both the transcriptional and post-transcriptional
alternative splicing levels.
Tumors of the urinary system
In accordance with results from a study by
Mukhopadhyay et al (88),
while the androgen receptor (AR) can control the production of
androgen-responsive genes and the proliferation of prostate cancer
cells, hnRNPK can reduce AR expression by inhibiting the
translation of AR mRNA. Analysis of hnRNPK expression in 188
patients with bladder cancer (89)
revealed that bladder cancer tissues had markedly higher levels of
hnRNPK expression and that hnRNPK expression levels were associated
with prognosis. Additionally, research has demonstrated that hnRNPK
inhibits tumor growth in vivo by enhancing proliferation,
inhibiting apoptosis and contributing to treatment resistance in
bladder cancer cells (90). This
mechanism involves hnRNPK-mediated transcriptional regulation of
cyclin D1, excision repair cross-complementing group four, amongst
other components that influence bladder cancer activity.
After examining modifications in hnRNPK protein
functionality in patients with advanced prostate cancer,
researchers have found that reducing cholesterol levels inhibited
the release of hnRNPK protein and hnRNPK-containing exosomes from
prostate cancer cells (91).
Prostate cancer cells release exosomes to facilitate the spread of
the disease to other organs. hnRNPK helps regulate the quantity of
exosomes produced by prostate cancer cells, thereby preventing the
spread of malignancy to other parts of the body (92). Exosomes act as regulators prior to
metastasis, conditioning the microenvironment of distant tissues to
facilitate tumor cell colonization. According to Iwabuchi et al
(93), decreasing cellular
cholesterol levels may prevent hnRNPK from exiting tumor cells and
transmitting oncogenic signals.
Current status of clinical
translation
Despite extensive mechanistic studies, the clinical
translation of hnRNPK research faces several limitations. The
majority of current evidence derives from retrospective analyses of
tumor samples and correlative studies. For instance, while multiple
studies have demonstrated associations between hnRNPK expression
and prognosis across numerous cancer types (30,35),
these findings have not yet been incorporated into clinical
practice guidelines. The lack of standardized detection methods and
validated cut-off values for hnRNPK expression limits its immediate
clinical utility. Furthermore, the dual nature of hnRNPK as both
oncogene and tumor suppressor in different contexts presents unique
challenges for therapeutic targeting, requiring more sophisticated
patient stratification processes than currently available.
Conclusion
There is an association between hnRNPK and the
initiation and progression of numerous cancer types. Identifying
tumor markers is one of the primary strategies used to address key
challenges in modern cancer research and hnRNPA2/B1 has been
utilized as a marker for the early detection of lung cancer.
Current findings suggest that hnRNPK holds promise as a molecular
biomarker for related malignancies. Although research in this area
is still in its early stages, several notable issues remain.
Studies specifically focusing on hnRNPK are still limited. To
establish the groundwork for future screening of human tumor
molecular markers applicable to early clinical diagnosis,
treatment, prognosis evaluation and disease monitoring, it is
important to further elucidate the specific mechanisms by which
members of the hnRNP family, including hnRNPK, contribute to the
genesis, development and metastasis of tumors.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Basic Science (Natural
Science) Research Project for Higher Education Institutions in
Jiangsu Province (grant no. 24KJB320002) and the Qinglan Project of
Jiangsu Province's Colleges and Universities (2024) and the
Scientific and Technological Innovation Team of Jiangsu Medical
College (2024).
Availability of data and materials
Not applicable.
Authors' contributions
YZ and YQ wrote the manuscript. YQ performed the
literature search and revised the manuscript. YZ generated the
figures. YQ. Both authors read and approved the final version of
the manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Holmgaard AB, Askou AL, Jensen EG, Alsing
S, Bak RO, Mikkelsen JG and Corydon TJ: Targeted knockout of the
Vegfa gene in the retina by subretinal injection of RNP complexes
containing Cas9 protein and modified sgRNAs. Mol Ther. 29:191–207.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chaudhury A, Chander P and Howe PH:
Heterogeneous nuclear ribonucleoproteins (hnRNPs) in cellular
processes: Focus on hnRNP E1′s multifunctional regulatory roles.
RNA. 16:1449–1462. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang Z, Qiu H, He J, Liu L, Xue W, Fox A,
Tickner J and Xu J: The emerging roles of hnRNPK. J Cell Physiol.
235:1995–2008. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhao E, Li J, Xie Y, Jin W, Zhang Z, Chen
J, Zeng L, Yin G, Qian J, Wu H, et al: Cloning and identification
of a novel human RNPC3 gene that encodes a protein with two RRM
domains and is expressed in the cell nucleus. Biochem Genet.
41:315–323. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Moujalled D, Grubman A, Acevedo K, Yang S,
Ke YD, Moujalled DM, Duncan C, Caragounis A, Perera ND, Turner BJ,
et al: TDP-43 mutations causing amyotrophic lateral sclerosis are
associated with altered expression of RNA-binding protein hnRNP K
and affect the Nrf2 antioxidant pathway. Hum Mol Genet.
26:1732–1746. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li H, Liu J, Shen S, Dai D, Cheng S, Dong
X, Sun L and Guo X: Pan-cancer analysis of alternative splicing
regulator heterogeneous nuclear ribonucleoproteins (hnRNPs) family
and their prognostic potential. J Cell Mol Med. 24:11111–11119.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhou ZJ, Dai Z, Zhou SL, Fu XT, Zhao YM,
Shi YH, Zhou J and Fan J: Overexpression of HnRNP A1 promotes tumor
invasion through regulating CD44v6 and indicates poor prognosis for
hepatocellular carcinoma. Int J Cancer. 132:1080–1089. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Huang Y, Liu Y, Pu M, Zhang Y, Cao Q, Li
S, Wei Y and Hou L: SOX2 interacts with hnRNPK to modulate
alternative splicing in mouse embryonic stem cells. Cell Biosci.
14:1022024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu Y, Li H, Liu F, Gao LB, Han R, Chen C,
Ding X, Li S, Lu K, Yang L, et al: Heterogeneous nuclear
ribonucleoprotein A2/B1 is a negative regulator of human breast
cancer metastasis by maintaining the balance of multiple genes and
pathways. EBioMedicine. 51:1025832020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gu WJ and Liu HL: Induction of pancreatic
cancer cell apoptosis, invasion, migration, and enhancement of
chemotherapy sensitivity of gemcitabine, 5-FU, and oxaliplatin by
hnRNP A2/B1 siRNA. Anticancer Drugs. 24:566–576. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Huang H, Han Y, Yang X, Li M, Zhu R, Hu J,
Zhang X, Wei R, Li K and Gao R: HNRNPK inhibits gastric cancer cell
proliferation through p53/p21/CCND1 pathway. Oncotarget.
8:103364–103374. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bidot P, Morgan M, Zhukov T, Tannenbaum M
and Tockman MS: Loss of heterogeneous ribonucleoprotein A(2)/B(1)
expression in thyroid neoplasms. Endocr Pract. 7:157–161. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Matsuyama S, Goto Y, Sueoka N, Ohkura Y,
Tanaka Y, Nakachi K and Sueoka E: Heterogeneous nuclear
ribonucleoprotein B1 expressed in esophageal squamous cell
carcinomas as a new biomarker for diagnosis. Jpn J Cancer Res.
91:658–663. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sueoka E, Sueoka N, Iwanaga K, Sato A,
Suga K, Hayashi S, Nagasawa K and Nakachi K: Detection of plasma
hnRNP B1 mRNA, a new cancer biomarker, in lung cancer patients by
quantitative real-time polymerase chain reaction. Lung Cancer.
48:77–83. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tani H, Ohshima K, Haraoka S, Hamasaki M,
Kamma H, Ikeda S and Kikuchi M: Reduced expression of heterogeneous
nuclear ribonucleoprotein B1 in adult T-cell lymphoma/leukemia. Int
J Oncol. 22:529–534. 2003.PubMed/NCBI
|
|
16
|
Dos Santos MGP, Gatti da Silva GH, Nagasse
HY, Fuziwara CS, Kimura ET and Coltri PP: hnRNP A1 and hnRNP C
associate with miR-17 and miR-18 in thyroid cancer cells. FEBS Open
Bio. 12:1253–1264. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wu Y, Zhao W, Liu Y, Tan X, Li X, Zou Q,
Xiao Z, Xu H, Wang Y and Yang X: Function of HNRNPC in breast
cancer cells by controlling the dsRNA-induced interferon response.
EMBO J. 37:e990172018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Howley BV, Mohanty B, Dalton A, Grelet S,
Karam J, Dincman T and Howe PH: The ubiquitin E3 ligase ARIH1
regulates hnRNP E1 protein stability, EMT and breast cancer
progression. Oncogene. 41:1679–1690. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Roychoudhury P, Paul RR, Chowdhury R and
Chaudhuri K: HnRNP E2 is downregulated in human oral cancer cells
and the overexpression of hnRNP E2 induces apoptosis. Mol Carcinog.
46:198–207. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li F, Zhao H, Su M, Xie W, Fang Y, Du Y,
Yu Z, Hou L and Tan W: HnRNP-F regulates EMT in bladder cancer by
mediating the stabilization of Snail1 mRNA by binding to its 3′
UTR. EBioMedicine. 45:208–219. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xu SH, Zhu S, Wang Y, Huang JZ, Chen M, Wu
QX, He YT, Chen D and Yan GR: ECD promotes gastric cancer
metastasis by blocking E3 ligase ZFP91-mediated hnRNP F
ubiquitination and degradation. Cell Death Dis. 9:4792018.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Peng WZ, Liu JX, Li CF, Ma R and Jie JZ:
hnRNPK promotes gastric tumorigenesis through regulating CD44E
alternative splicing. Cancer Cell Int. 19:3352019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Peng C, Tan Y, Yang P, Jin K, Zhang C,
Peng W, Wang L, Zhou J, Chen R, Wang T, et al: Circ-GALNT16
restrains colorectal cancer progression by enhancing the
SUMOylation of hnRNPK. J Exp Clin Cancer Res. 40:2722021.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang S, Zhang B, Liao Z, Chen Y, Guo W,
Wu J, Liu H, Weng R, Su D, Chen G, et al: Hnrnpk protects against
osteoarthritis through targeting WWC1 mRNA and inhibiting Hippo
signaling pathway. Mol Ther. 32:1461–1478. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chen TM, Lai MC, Li YH, Chan YL, Wu CH,
Wang YM, Chien CW, Huang SY, Sun HS and Tsai SJ: hnRNPM induces
translation switch under hypoxia to promote colon cancer
development. EBioMedicine. 41:299–309. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Xu Y, Wu W, Han Q, Wang Y, Li C, Zhang P
and Xu H: New insights into the interplay between non-coding RNAs
and RNA-binding protein HnRNPK in regulating cellular functions.
Cells. 8:622019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lee WJ, Shin CH, Ji H, Jeong SD, Park MS,
Won HH, Pandey PR, Tsitsipatis D, Gorospe M and Kim HH:
hnRNPK-regulated LINC00263 promotes malignant phenotypes through
miR-147a/CAPN2. Cell Death Dis. 12:2902021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Choufani S, McNiven V, Cytrynbaum C,
Jangjoo M, Adam MP, Bjornsson HT, Harris J, Dyment DA, Graham GE,
Nezarati MM, et al: An HNRNPK-specific DNA methylation signature
makes sense of missense variants and expands the phenotypic
spectrum of Au-Kline syndrome. Am J Hum Genet. 109:1867–1884. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tang S, Xie Z, Wang P, Li J, Wang S, Liu
W, Li M, Wu X, Su H, Cen S, et al: LncRNA-OG promotes the
osteogenic differentiation of bone marrow-derived mesenchymal stem
cells under the regulation of hnRNPK. Stem Cells. 37:270–283. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li J, Chen Y, Xu X, Jones J, Tiwari M,
Ling J, Wang Y, Harismendy O and Sen GL: HNRNPK maintains epidermal
progenitor function through transcription of proliferation genes
and degrading differentiation promoting mRNAs. Nat Commun.
10:41982019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ren D, Sun Y, Zhang D, Li D, Liu Z, Jin X
and Wu H: SGLT2 promotes pancreatic cancer progression by
activating the Hippo signaling pathway via the hnRNPK-YAP1 axis.
Cancer Lett. 519:277–288. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bampton A, Gatt A, Humphrey J, Cappelli S,
Bhattacharya D, Foti S, Brown AL, Asi Y, Low YH, Foiani M, et al:
HnRNP K mislocalisation is a novel protein pathology of
frontotemporal lobar degeneration and ageing and leads to cryptic
splicing. Acta Neuropathol. 142:609–627. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Meng Y, Zhao Q, An L, Jiao S, Li R, Sang
Y, Liao J, Nie P, Wen F, Ju J, et al: A TNFR2-hnRNPK axis promotes
primary liver cancer development via activation of YAP signaling in
hepatic progenitor cells. Cancer Res. 81:3036–3050. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Toki N, Takahashi H, Sharma H, Valentine
MNZ, Rahman FM, Zucchelli S, Gustincich S and Carninci P: SINEUP
long non-coding RNA acts via PTBP1 and HNRNPK to promote
translational initiation assemblies. Nucleic Acids Res.
48:11626–11644. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chen Y, Wu J, Zhang S, Gao W, Liao Z, Zhou
T, Li Y, Su D, Liu H, Yang X, et al: Hnrnpk maintains chondrocytes
survival and function during growth plate development via
regulating Hif1α-glycolysis axis. Cell Death Dis. 13:8032022.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Braems E, Bercier V, Van Schoor E, Heeren
K, Beckers J, Fumagalli L, Dedeene L, Moisse M, Geudens I, Hersmus
N, et al: HNRNPK alleviates RNA toxicity by counteracting DNA
damage in C9orf72 ALS. Acta Neuropathol. 144:465–488. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sun XL, Wang ZL, Wu Q, Jin SQ, Yao J and
Cheng H: LncRNA RMST activates TAK1-mediated NF-κB signaling and
promotes activation of microglial cells via competitively binding
with hnRNPK. IUBMB Life. 71:1785–1793. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chen CC, Yang JH, Fu SL, Lin WJ and Lin
CH: Arginine methylation of hnRNPK inhibits the DDX3-hnRNPK
interaction to play an anti-apoptosis role in osteosarcoma cells.
Int J Mol Sci. 22:97642021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhou W, Jie Q, Pan T, Shi J, Jiang T,
Zhang Y, Ding N, Xu J, Ma Y and Li Y: Single-cell RNA binding
protein regulatory network analyses reveal oncogenic HNRNPK-MYC
signalling pathway in cancer. Commun Biol. 6:822023. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Xie M, Wang C, Sun Y, Mao Q, Sun S, Wu M,
Zhu J, Li W and Jiang Z: Maimendong and Qianjinweijing Tang
combined with cisplatin suppressed lung cancer through targeting
lncRNA-p21. J Ethnopharmacol. 322:1175472024. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kawasaki Y, Komiya M, Matsumura K, Negishi
L, Suda S, Okuno M, Yokota N, Osada T, Nagashima T, Hiyoshi M, et
al: MYU, a target lncRNA for Wnt/c-Myc signaling, mediates
induction of CDK6 to promote cell cycle progression. Cell Rep.
16:2554–2564. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Xia A, Yuan W, Wang Q, Xu J, Gu Y, Zhang
L, Chen C, Wang Z, Wu D, He Q, et al: The cancer-testis lncRNA
lnc-CTHCC promotes hepatocellular carcinogenesis by binding hnRNP K
and activating YAP1 transcription. Nat Cancer. 3:203–218. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Huarte M, Guttman M, Feldser D, Garber M,
Koziol MJ, Kenzelmann-Broz D, Khalil AM, Zuk O, Amit I, Rabani M,
et al: A large intergenic noncoding RNA induced by p53 mediates
global gene repression in the p53 response. Cell. 142:409–419.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lin N, Chang KY, Li Z, Gates K, Rana ZA,
Dang J, Zhang D, Han T, Yang CS, Cunningham TJ, et al: An
evolutionarily conserved long noncoding RNA TUNA controls
pluripotency and neural lineage commitment. Mol Cell. 53:1005–1019.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Li D, Wang X, Mei H, Fang E, Ye L, Song H,
Yang F, Li H, Huang K, Zheng L and Tong Q: Long noncoding RNA
pancEts-1 promotes neuroblastoma progression through
hnRNPK-mediated β-catenin stabilization. Cancer Res. 78:1169–1183.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Xi Z, Huang H, Hu J, Yu Y, Ma X, Xu M,
Ming J, Li L, Zhang H, Chen H and Huang T: LINC00571 drives
tricarboxylic acid cycle metabolism in triple-negative breast
cancer through HNRNPK/ILF2/IDH2 axis. J Exp Clin Cancer Res.
43:222024. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wang Z, Chen J, Sun F, Zhao X, Dong Y, Yu
S, Li J and Liang H: LncRNA CRLM1 inhibits apoptosis and promotes
metastasis through transcriptional regulation cooperated with
hnRNPK in colorectal cancer. Cell Biosci. 12:1202022. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Ji L, Li X, Zhou Z, Zheng Z, Jin L and
Jiang F: LINC01413/hnRNP-K/ZEB1 axis accelerates cell proliferation
and EMT in colorectal cancer via inducing YAP1/TAZ1 translocation.
Mol Ther Nucleic Acids. 19:546–561. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhang Z, Zhou C, Chang Y, Zhang Z, Hu Y,
Zhang F, Lu Y, Zheng L, Zhang W and Li X and Li X: Long non-coding
RNA CASC11 interacts with hnRNP-K and activates the WNT/β-catenin
pathway to promote growth and metastasis in colorectal cancer.
Cancer Lett. 376:62–73. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Gu P, Chen X, Xie R, Xie W, Huang L, Dong
W, Han J, Liu X, Shen J, Huang J and Lin T: A novel AR
translational regulator lncRNA LBCS inhibits castration resistance
of prostate cancer. Mol Cancer. 18:1092019. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Pintacuda G, Wei G, Roustan C, Kirmizitas
BA, Solcan N, Cerase A, Castello A, Mohammed S, Moindrot B,
Nesterova TB and Brockdorff N: hnRNPK recruits PCGF3/5-PRC1 to the
Xist RNA B-repeat to establish polycomb-mediated chromosomal
silencing. Molecular Cell. 68:955–969.e10. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Gao T, Liu X, He B, Nie Z, Zhu C, Zhang P
and Wang S: Exosomal lncRNA 91H is associated with poor development
in colorectal cancer by modifying HNRNPK expression. Cancer Cell
Int. 18:112018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Peng W, Zhang C, Peng J, Huang Y, Peng C,
Tan Y, Ji D, Zhang Y, Zhang D, Tang J, et al: Lnc-FAM84B-4 acts as
an oncogenic lncRNA by interacting with protein hnRNPK to restrain
MAPK phosphatases-DUSP1 expression. Cancer Lett. 494:94–106. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Xu W, Wu Y, Fang X, Zhang Y, Cai N, Wen J,
Liao J, Zhang B, Chen X and Chu L: SnoRD126 promotes the
proliferation of hepatocellular carcinoma cells through
transcriptional regulation of FGFR2 activation in combination with
hnRNPK. Aging (Albany NY). 13:13300–13317. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Li L, Yan S, Zhang H, Zhang M, Huang G and
Chen M: Interaction of hnRNP K with MAP 1B-LC1 promotes
TGF-β1-mediated epithelial to mesenchymal transition in lung cancer
cells. BMC Cancer. 19:8942019. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zhang M, Wu J, Zhong W, Zhao Z and He W:
DNA-methylation-induced silencing of DIO3OS drives non-small cell
lung cancer progression via activating hnRNPK-MYC-CDC25A axis. Mol
Ther Oncolytics. 23:205–219. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Xu L, Zhang T, Huang W, Liu X, Lu J, Gao
X, Zhang YF and Liu L: YAP mediates the positive regulation of
hnRNPK on the lung adenocarcinoma H1299 cell growth. Acta Biochim
Biophys Sin (Shanghai). 51:677–687. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Avar M, Heinzer D, Thackray AM, Liu Y,
Hruska-Plochan M, Sellitto S, Schaper E, Pease DP, Yin JA,
Lakkaraju AK, et al: An arrayed genome-wide perturbation screen
identifies the ribonucleoprotein Hnrnpk as rate-limiting for prion
propagation. EMBO J. 41:e1123382022. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Swiatkowska A, Dutkiewicz M, Machtel P,
Janecki DM, Kabacinska M, Żydowicz-Machtel P and Ciesiołka J:
Regulation of the p53 expression profile by hnRNP K under stress
conditions. RNA Boil. 17:1402–1415. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Mucha B, Qie S, Bajpai S, Tarallo V, Diehl
JN, Tedeschi F, Zhou G, Gao Z, Flashner S, Klein-Szanto AJ, et al:
Tumor suppressor mediated ubiquitylation of hnRNPK is a barrier to
oncogenic translation. Nat Commun. 13:66142022. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Wu L, Zhu X, Song Z, Guo M, Liang J and
Yan D: FGD5-AS1 facilitates glioblastoma progression by activation
of Wnt/β-catenin signaling via regulating miR-129-5p/HNRNPK axis.
Life Sci. 256:1179982020. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Strozynski J, Heim J, Bunbanjerdsuk S,
Wiesmann N, Zografidou L, Becker SK, Meierl AM, Gouveris H, Lüddens
H, Grus F and Brieger J: Proteomic identification of the
heterogeneous nuclear ribonucleoprotein K as irradiation responsive
protein related to migration. J Proteomics. 113:154–161. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Xia Y, Lv J, Jiang T, Li B, Li Y, He Z,
Xuan Z, Sun G, Wang S, Li Z, et al: CircFAM73A promotes the cancer
stem cell-like properties of gastric cancer through the
miR-490-3p/HMGA2 positive feedback loop and HNRNPK-mediated
β-catenin stabilization. J Exp Clin Cancer Res. 40:1032021.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Robinson H, Ruelcke JE, Lewis A, Bond CS,
Fox AH, Bharti V, Wani S, Cloonan N, Lai A, Margolin D, et al:
Caveolin-1-driven membrane remodelling regulates hnRNPK-mediated
exosomal microRNA sorting in cancer. Clin Transl Med. 11:e3812021.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Feng J, Li H, Li J, Meng P, Wang L, Liu C,
Zhao S, Sun W and Zhang Y: hnRNPK knockdown alleviates NLRP3
inflammasome priming by repressing FLIP expression in Raw264.7
macrophages. Redox Rep. 25:104–111. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Sidhu R, Gatt A, Fratta P, Lashley T and
Bampton A: HnRNP K mislocalisation in neurons of the dentate
nucleus is a novel neuropathological feature of neurodegenerative
disease and ageing. Neuropathol Appl Neurobiol. 48:e127932022.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Puvvula PK, Buczkowski S and Moon AM:
hnRNPK-derived cell-penetrating peptide inhibits cancer cell
survival. Mol Ther Oncolytics. 23:342–354. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Zhang J, Liu X, Lin Y, Li Y, Pan J, Zong
S, Li Y and Zhou Y: HnRNP K contributes to drug resistance in acute
myeloid leukemia through the regulation of autophagy. Exp Hematol.
44:850–856. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Qin W, Kong N, Wang C, Dong S, Zhai H,
Zhai X, Yang X, Ye C, Ye M, Tong W, et al: hnRNP K degrades viral
nucleocapsid protein and induces type I IFN production to inhibit
porcine epidemic diarrhea virus replication. J Virol.
96:e01555222022. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Yang X, Wen Y, Liu S, Duan L, Liu T, Tong
Z, Wang Z, Gu Y, Xi Y, Wang X, et al: LCDR regulates the integrity
of lysosomal membrane by hnRNP K-stabilized LAPTM5 transcript and
promotes cell survival. Proc Natl Acad Sci USA.
119:e21104281192022. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Zhang L, Zhang S, Fan Z, Jiang Z, Li S,
Liu A and Sun J: D816V mutation of KIT specifically induces
phosphorylation of HNRNPL and HNRNPK in COS-1 cells. Xi Bao Yu Fen
Zi Mian Yi Xue Za Zhi. 39:138–143. 2023.(In Chinese). PubMed/NCBI
|
|
72
|
Shu B, Zeng P, Kang S, Li PH, Hu D, Kuang
G, Cao J, Li X, Zhang M, An LK, et al: Syntheses and evaluation of
new Quinoline derivatives for inhibition of hnRNP K in regulating
oncogene c-myc transcription. Bioorg Chem. 85:1–17. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Liu W, Yang D, Sun C, Wang H, Zhao B, Zhou
G and Yu L: hnRNP K is a novel internal ribosomal entry
site-transacting factor that negatively regulates foot-and-mouth
disease virus translation and replication and is antagonized by
viral 3C protease. J Virol. 94:e00803–20. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Chen J, Dai L, Zheng H, Liu G, Zhao Y and
Wang J: Analysis of clinical features and genetic variant in a
neonate with Au-Kline syndrome due to a de novo variant of the
HNRNPK gene. Zhonghua Yi Xue Yi Chuan Xue Za Zhi. 40:226–229.
2023.(In Chinese). PubMed/NCBI
|
|
75
|
Malaney P, Benitez O, Zhang X and Post SM:
Assessing the role of intrinsic disorder in RNA-binding protein
function: hnRNP K as a case study. Methods. 208:59–65. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Wu HL, Li SM, Huang YC, Xia QD, Zhou P, Li
XM, Yu X, Wang SG, Ye ZQ and Hu J: Transcriptional regulation and
ubiquitination-dependent regulation of HnRNPK oncogenic function in
prostate tumorigenesis. Cancer Cell Int. 21:6412021. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Zhu HQ, Liu XL, Li R, Du QF, Zhang S, Yao
F and Liu Z: Preliminary study of proteins related to blast crisis
in chronic myeloid leukemia. Zhonghua Zhong Liu Za Zhi. 31:655–659.
2009.(In Chinese). PubMed/NCBI
|
|
78
|
Lu Y, Cheng J, Cai W, Zhuo H, Wu G and Cai
J: Inhibition of circRNA circVPS33B reduces warburg effect and
tumor growth through regulating the miR-873-5p/HNRNPK axis in
infiltrative gastric cancer. Onco Targets Ther. 14:3095–3108. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Huang WS, Xu FM, Zeng QZ, Liu XH, Gao XJ
and Liu LX: ERK1/2-mediated cytoplasmic accumulation of hnRNPK
antagonizes TRAIL-induced Apoptosis through upregulation of XIAP in
H1299 cells. Biomed Environ Sci. 30:473–481. 2017.PubMed/NCBI
|
|
80
|
Han J, Nie M, Chen C, Cheng X, Guo T,
Huangfu L, Li X, Du H, Xing X and Ji J: SDCBP-AS1 destabilizes
β-catenin by regulating ubiquitination and SUMOylation of hnRNP K
to suppress gastric tumorigenicity and metastasis. Cancer Commun
(Lond). 42:1141–1161. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Bakhmet EI, Nazarov IB, Gazizova AR,
Vorobyeva NE, Kuzmin AA, Gordeev MN, Sinenko SA, Aksenov ND,
Artamonova TO, Khodorkovskii MA, et al: HnRNP-K targets open
chromatin in mouse embryonic stem cells in concert with multiple
regulators. Stem Cells. 37:1018–1029. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Zhao W, Wang S, Qin T and Wang W: Circular
RNA (circ-0075804) promotes the proliferation of retinoblastoma via
combining heterogeneous nuclear ribonucleoprotein K (HNRNPK) to
improve the stability of E2F transcription factor 3 E2F3. J Cell
Biochem. 121:3516–3525. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Zhang J, Liu X, Yin C and Zong S:
hnRNPK/Beclin1 signaling regulates autophagy to promote imatinib
resistance in Philadelphia chromosome-positive acute lymphoblastic
leukemia cells. Exp Hematol. 108:46–54. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Yokoi S, Ito T, Sahashi K, Nakatochi M,
Nakamura R, Tohnai G, Fujioka Y, Ishigaki S, Udagawa T, Izumi Y, et
al: The SYNGAP1 3′UTR variant in ALS patients causes aberrant
SYNGAP1 splicing and dendritic spine loss by recruiting HNRNPK. J
Neurosci. 42:8881–8896. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Harris D, Zhang Z, Chaubey B and Pandey
VN: Identification of cellular factors associated with the
3′-nontranslated region of the hepatitis C virus genome. Mol Cell
Proteomics. 5:1006–1018. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Shu H, Hu J and Deng H: miR-1249-3p
accelerates the malignancy phenotype of hepatocellular carcinoma by
directly targeting HNRNPK. Mol Genet Genomic Med. 7:e008672019.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Li Y, Chen H, Li S, Li Y, Liu G, Bai J,
Luo H, Lan X and He Z: LncSSBP1 functions as a negative regulator
of IL-6 through interaction with hnRNPK in bronchial epithelial
cells infected with Talaromyces marneffei. Front Immunol.
10:29772020. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Mukhopadhyay NK, Kim J, Cinar B,
Ramachandran A, Hager MH, Di Vizio D, Adam RM, Rubin MA,
Raychaudhuri P, De Benedetti A and Freeman MR: Heterogeneous
nuclear ribonucleoprotein K is a novel regulator of androgen
receptor translation. Cancer Res. 69:2210–2218. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Cho E, Che X, Ang MJ, Cheon S, Lee J, Kim
KS, Lee CH, Lee SY, Yang HY, Moon C, et al: Peroxiredoxin 5
regulates osteogenic differentiation via interaction with hnRNPK
during bone regeneration. bioRxiv. 2022.06.09.495435. 2022.
|
|
90
|
Gray C, Kelleher B, Grimaldi L and
Mihailescu ME: A G-quadruplex structure within the 3′-untranslated
region of hnRNP K affects its translation by controlling the
accessibility of miR-1249-3p to its binding site. Biophys J. 122
(Suppl 1):360A–361A. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Miki Y, Iwabuchi E, Takagi K, Suzuki T,
Sasano H, Yaegashi N and Ito K: Co-expression of nuclear
heterogeneous nuclear ribonucleic protein K and estrogen receptor α
in endometrial cancer. Pathol Res Pract. 231:1537952022. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Li M, Yang X, Zhang G, Wang L, Zhu Z,
Zhang W, Huang H and Gao R: Heterogeneous nuclear ribonucleoprotein
K promotes the progression of lung cancer by inhibiting the
p53-dependent signaling pathway. Thorac Cancer. 13:1311–1321. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Iwabuchi E, Miki Y, Suzuki T, Hirakawa H,
Ishida T and Sasano H: Heterogeneous nuclear ribonucleoprotein K is
involved in the estrogen-signaling pathway in breast cancer. Int J
Mol Sci. 22:25812021. View Article : Google Scholar : PubMed/NCBI
|