Historically, lactate was dismissed as a byproduct
of glycolytic metabolism, resulting in an oversight of its vital
physiological contributions (1).
The seminal work of Otto Warburg in the 1920s revealed that
malignant cells display dysregulated glycolysis, generating surplus
lactate despite the availability of oxygen (2,3).
While traditionally viewed strictly as an endpoint of anaerobic
respiration, research has now established that lactate serves as a
crucial bridge between glycolysis and oxidative phosphorylation,
and also as a versatile signaling agent (4). Consequently, this paradigm shift has
redefined lactate from a metabolic remnant to a pivotal biological
indicator (5).
Protein post-translational modifications (PTMs) are
enzymatically mediated covalent attachments of functional groups
that occur either during or after protein biosynthesis. These
alterations markedly expand the proteomic landscape, generating a
functional diversity that extends beyond the genomic blueprint
(6). In eukaryotes, the majority
of proteins are subject to terminal modifications via diverse
mechanisms, including acetylation, lipidation and others (7). Crucially, PTMs are closely linked to
oncogenesis, governing key carcinogenic processes such as sustained
proliferation, apoptosis evasion, angiogenic induction and
metastasis (8) In 2019, Zhang
et al (9) characterized a
novel PTM termed lactylation (or lysine lactylation), which was
initially observed on histones. This process entails the covalent
bonding of a lactyl moiety, derived from lactate, to lysine
residues, thereby functioning as a mechanism for gene regulation;
specifically, lactate acts as the precursor providing the essential
substrate pool for this modification (9). Furthermore, subsequent investigations
have revealed that lactylation is not limited to histones but also
broadly affects non-histone proteins, modulating their functions
through distinct regulatory pathways (10–12).
Lactylation serves as a fundamental regulator of
essential physiological processes, including cardiac performance
(13), embryogenesis (14) and osteoblast differentiation
(15). Concurrently, this PTM is
implicated in the etiology and advancement of diverse pathologies,
ranging from inflammatory cascades (16) and renal tubular damage (17), to cerebral trauma (18) and pulmonary fibrosis (19). Crucially, lactylation profoundly
impacts the tumor microenvironment (TME). Beyond influencing
oncogenic expression, it orchestrates a complex network involving
immune modulation, metabolic reprogramming, therapeutic resistance
and autophagy. By engaging with cancer-associated genes [e.g., MYC
proto-oncogene (MYC), snail family transcriptional repressor 1 and
programmed cell death 1 ligand 1], lactylation alters
transcriptional landscapes to drive proliferation, invasiveness and
immune escape (20,21). Consequently, the present review
comprehensively summarizes the current knowledge on lactate
metabolism and lactylation in oncology, with a specific emphasis on
emerging strategies for tumor diagnosis and targeted therapy.
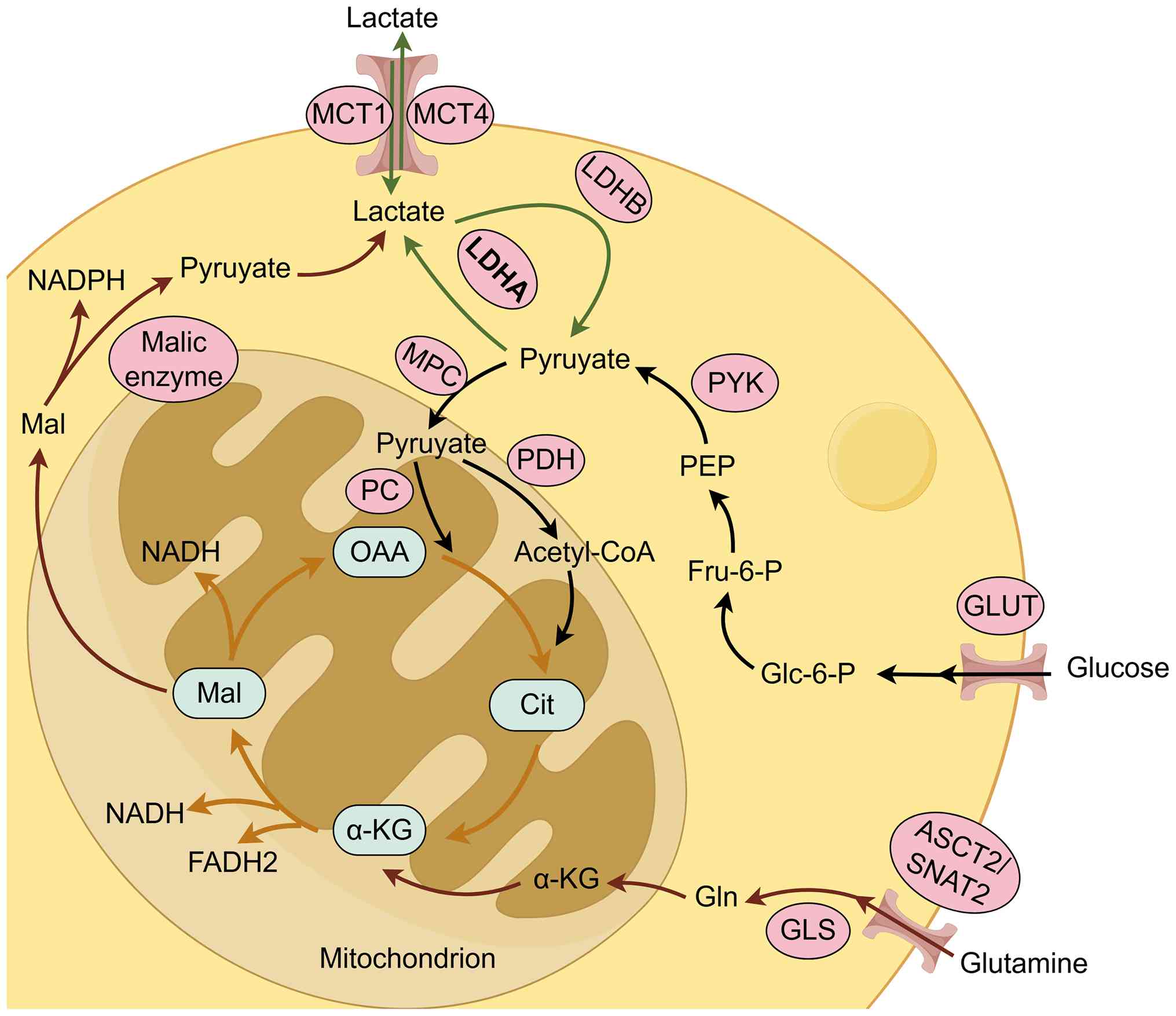
In both physiological and neoplastic contexts,
glucose acts as the principal substrate for lactate generation,
marking the end of the glycolytic cascade (Fig. 1) (22). Cellular uptake of glucose is
predominantly mediated by glucose transporter (GLUT) proteins
(23). Following internalization,
glucose is metabolized to pyruvate under normoxic conditions.
Subsequently, the bulk of this pyruvate enters the mitochondria via
the mitochondrial pyruvate carrier, or is shuttled after conversion
to undergo oxidative phosphorylation. Within the mitochondria, the
tricarboxylic acid (TCA) cycle and electron transport chain drive
the synthesis of ATP, releasing CO2 and H2O
as byproducts (24) Conversely, in
hypoxic or anaerobic environments, oxidative phosphorylation is
compromised. To maintain redox homeostasis and sustain glycolysis,
cytosolic pyruvate is reduced to lactate by lactate dehydrogenase
(LDH) (25) Structurally, LDH
exists as a tetramer formed by varying combinations of muscle
subunits (M) and heart subunits (H), yielding five distinct
isozymes: LDH-1 (4H), LDH-2 (3H1M), LDH-3 (2H2M), LDH-4 (1H3M) and
LDH-5 (4M) (26). While these
isoforms are bidirectional, LDHA (LDH-5) preferentially drives the
reduction of pyruvate to lactate, thereby markedly fueling tumor
progression, whereas LDHB (LDH-1) favors the oxidation of lactate
back to pyruvate (27,28). Beyond glucose, research has
highlighted glutaminolysis as an alternative metabolic route in
cancer cells (29). Driven by the
c-Myc proto-oncogene, glutamine enters the cytoplasm via specific
transporters, including ASCT2 and SNAT2 (30). Once inside the cell, glutaminase
(GLS) converts glutamine into glutamate, which is further processed
into α-ketoglutarate by enzymes such as glutamate dehydrogenase to
enter the TCA. Carbon derived from glutamine is then metabolized
into oxaloacetate and subsequently malate, which is exported from
the mitochondria. In the cytosol, malic enzyme converts malate to
pyruvate, generating NADPH in the process, which is ultimately
reduced to lactate by LDHA. Although this pathway typically serves
as a secondary source of lactate, glutamine becomes the dominant
carbon reservoir supporting cancer cell metabolism during glucose
starvation (31). Furthermore,
evidence suggests that under such glucose-deprived conditions,
lactate enhances cell survival primarily by sustaining
mitochondrial respiration through GLS1-mediated glutaminolysis
(32).
Driven by metabolic reprogramming, lactate
concentrations in cancer cells are markedly higher than those in
the circulation or healthy parenchyma (33). Malignant tumors exhibit upregulated
glycolytic flux even when oxygen availability is adequate and
oxidative phosphorylation remains functional. Specifically, through
aerobic glycolysis, tumor tissues generate lactate levels that are
10- to 100-fold higher than those produced via complete
mitochondrial glucose oxidation (34). Characterized as the Warburg effect,
this phenomenon describes the propensity of cancer cells to
prioritize glycolysis and lactate synthesis over oxidative
phosphorylation, despite the presence of oxygen (3). This metabolic shift is essential for
meeting neoplastic requirements for bioenergetics, redox
homeostasis and biosynthetic precursors, a process accompanied by
extensive reprogramming of metabolic enzymes and transport proteins
(35,36). Consequently, the accumulation of
substantial lactic acid stands as a definitive outcome of this
metabolic reconfiguration.
Following its efflux from glycolytic cells, lactate
enters the systemic circulation, where, under homeostatic
conditions, it functions as a vital metabolic substrate for the
myocardium, brain and skeletal musculature (37–39).
To avert the onset of acidosis caused by supraphysiological lactate
levels, rapid metabolic clearance and irreversible elimination
mediated by LDH are essential for maintaining tissue equilibrium
(40). Concurrently, the
accumulation of lactate stimulates hepatic gluconeogenesis,
allowing the liver to recycle lactate into glucose via the Cori
cycle for redistribution into the vascular system (41). In cancer, where metabolic
reprogramming drives excessive lactate generation, efflux is
predominantly facilitated by monocarboxylate transporters (MCTs),
with MCT4 serving as a pivotal mediator of this export mechanism
(42). Furthermore, in hypoxic
microenvironments, MCT1 also contributes to the extracellular
transport of lactate (43).
The TME constitutes a complex ecosystem comprising
malignant cells, stromal elements such as endothelial cells and
cancer-associated fibroblasts (CAFs), and infiltrating immune
populations. These cellular components are embedded within a
non-neoplastic extracellular matrix rich in bioactive peptides,
including cytokines, chemokines, antibodies and growth factors
(44). Upon accumulation in the
TME, lactate actively potentiates immune escape by dampening the
functional activity of immune cells and suppressing the synthesis
of inflammatory mediators (45).
Beyond its metabolic function, lactate acts simultaneously as an
energy substrate, a signaling messenger and a potent
immunosuppressive agent, thereby orchestrating diverse cellular
behaviors (46). Notably, lactate
buildup sustains a chronically acidic extracellular pH; this
localized acidic milieu is a critical driver of angiogenesis,
therapeutic resistance, immunosuppression and metastatic
dissemination (47–50). Specifically, the acidic conditions
compromise cytokine secretion by monocytes (51) and impair the cytotoxic capabilities
of natural killer (NK) and NKT cells (52,53),
ultimately fostering an environment conducive to tumor
proliferation. Furthermore, lactate stimulates the upregulation of
vascular endothelial growth factor, ensuring sufficient oxygen and
nutrient delivery to support neoplastic growth and
neovascularization (54). In
colorectal cancer (CRC), research has indicated that lactic
acidosis blunts the activity of antitumor immune cells and inhibits
the phagocytic capacity of tumor-associated macrophages (TAMs).
Concurrently, metabolic stress and nutritional deprivation favor
the differentiation of regulatory T cells (Tregs) and M2-polarized
macrophages, the latter of which promote tumor expansion and
metastasis (55). Collectively,
these mechanisms orchestrate robust tumor immune evasion (56). In summary, the synergistic effects
of TME acidification and sustained high lactate levels drive tumor
progression by disabling immune surveillance and fortifying cancer
cells against host defenses.
Mechanistic investigations have confirmed that
GPR81, a lactate receptor, is a pivotal modulator of both tumor
proliferation and metastatic progression (57). Evidence from in vitro and
in vivo models has suggested that GPR81 regulates metabolic
processes in cancer cells through autocrine loops and
simultaneously drives immune escape via paracrine mechanisms
(58,59). In breast cancer (BC), in
vitro experiments have revealed elevated GPR81 levels relative
to healthy mammary epithelium (60), and knockdown of GPR81 markedly
suppresses BC cell proliferation (61). Furthermore, activation of GPR81 by
lactate induces PD-L1 upregulation, thereby promoting immune
evasion (62). Additionally,
experimental findings have identified a signaling axis in which
lactate activates STAT3, which subsequently binds to the GPR81
promoter to induce GPR81 transcription (57). In lung cancer (LC) models,
inhibition of GPR81 signaling reduces PD-L1 protein abundance
(63). Moreover, silencing GPR81
in hypopharyngeal squamous cell carcinoma cells potentiates
cisplatin-induced apoptosis (64).
Preclinical studies have also demonstrated that antagonizing GPR81
augments the therapeutic efficacy of metformin (65,66).
From a clinical perspective, the contribution of lactate metabolism
to metastasis is corroborated by correlative analyses in patient
populations (67). Driven by the
Warburg effect, LDHA expression shows a direct linear relationship
with lactate levels, and its depletion has been observed to inhibit
metastasis in hepatocellular carcinoma (HCC) (68,69).
In human BC tissues, the potassium channel KCNK1 is markedly
upregulated and is associated with poor prognosis. Mechanistically,
KCNK1 physically interacts with LDHA to enhance lactate production,
creating a positive feedback loop that drives metastatic spread
(70). Furthermore, clinical data
have indicated that patients with cervical cancer (CC) exhibiting
high LDH levels are at a greater risk for deep stromal invasion and
nodal metastasis (71). Similarly,
in cases of brain metastasis, elevated serum LDH is inversely
associated with overall survival (OS) (72).
While the pivotal role of lactate in tumor biology
has long been recognized, the precise molecular machinery governing
its regulatory effects remained obscure until the conceptualization
of lactylation. Consequently, the identification of this
modification has notably broadened the horizon of PTM research,
offering a novel framework to decipher the molecular impact of
lactate on key physiological and pathological events within cancer
(9). Current evidence has
established lactylation as a pervasive biological phenomenon that
is intrinsically linked to the proliferation of diverse
malignancies (20). Driven by
these insights, substantial investigative efforts have been
invested in this field.
Histones are fundamental chromatin proteins that
compact the eukaryotic genome into nucleosomes and higher-order
chromosomal structures, thereby maintaining physiological stability
(73,74). PTMs of these proteins provide a
crucial regulatory layer, influencing DNA-dependent processes to
facilitate cellular differentiation and optimize gene function.
Furthermore, sequence variants within histone families enrich the
chromatin landscape, offering diverse mechanisms for signaling and
regulation. While initial reports characterized histone lactylation
broadly, a recent study by Zhang et al (75) employed chemical analysis to
differentiate three distinct isomers: Lysine L-lactylation (Kl-la),
N-ε-(carboxyethyl)-lysine (Kce) and D-lactyllysine (Kd-la).
Notably, this investigation established Kl-la as the dominant
isomer on histones, specifically induced by upregulated glycolytic
flux. This distinction is particularly relevant in oncology, given
that the Warburg effect drives tumor cells to generate L-lactate,
the specific substrate for Kl-la. Conversely, Kd-la originates from
methylglyoxal, a toxic metabolic byproduct. This suggests that
Kl-la serves as the primary functional bridge connecting metabolic
reprogramming to tumor epigenetics, whereas the specific roles of
Kce and Kd-la within the TME warrant further independent study
(75). Utilizing high-performance
liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) on
trypsinized core histones from human MCF-7 cells, Zhang et
al (9) pioneered the detection
of a specific 72.021 Da mass shift on lysine residues. The authors
further validated the widespread occurrence of histone lysine
lactylation using metabolic isotope labeling, thereby establishing
a cornerstone for Kl-la research. While this work focused on
characterizing histone lactylation as a novel epigenetic regulator
of transcription, subsequent research broadened this perspective.
Subsequently, Gaffney et al (76) detected lactylation on various
metabolic enzymes associated with glycolysis, suggesting that this
modification could exert negative feedback control on glycolytic
flux. The findings of this previous study underscored the ubiquity
of lactylation across human tissues, demonstrating that this
modification is not confined to histones but also exerts regulatory
effects on non-histone proteins.
Non-histone lactylation is increasingly recognized
as a pivotal regulator of a range of cellular processes in both
physiological and pathological contexts. The functional impact and
enzymatic dynamics of this modification are dictated by the
subcellular positioning and protein-protein interactions of the
specific substrates, employing varied mechanisms to modulate target
protein activity (77). Broadly,
lactate-driven protein modification is markedly ubiquitous across
diverse biological systems. In the context of Toxoplasma
gondii, a prevalent parasitic pathogen, investigations have
revealed that lactylated proteins are present in various
subcellular organelles. These modifications are closely linked to
vital processes such as mRNA splicing, glycolytic metabolism,
aminoacyl-transfer RNA (tRNA) biosynthesis, RNA transport and
several signaling cascades (e.g., DNA damage repair and mTOR
signaling) (78). Similarly,
proteomic analyses in yaks have identified 421, 308 and 650
lactylated proteins in cardiac, muscular and hepatic tissues,
respectively; these modifications are integral to metabolic
regulation and the maintenance of physiological homeostasis
(79). Furthermore, in the plant
kingdom, researchers have characterized 927 specific lactylation
sites across 394 proteins in wheat (80). Furthermore, mechanistic inquiries
into specific non-histone targets have yielded profound insights.
For example, the lactylation of α-myosin heavy chain has been shown
to maintain sarcomeric integrity and functionality in a mouse
model, effectively mitigating the progression of heart failure
(81). In oncology, MRE11, a key
mediator of homologous recombination (HR), has been reported to
undergo lactylation at residue K673, a modification that reduces
tumor cell susceptibility to chemotherapy. Consequently, inhibiting
MRE11 lactylation restores the sensitivity of cancer cells to
agents such as cisplatin and poly(ADP-ribose) polymerase inhibitors
(PARPi) (82).
Elucidating the precise regulatory machinery of
lactylation necessitates a thorough characterization of its
enzymatic effectors. This regulatory system is orchestrated by
three distinct classes of proteins: ‘Writers’ that deposit lactyl
moieties onto specific residues, ‘erasers’ that catalyze the
removal of these modifications, and ‘readers’ that possess
specialized domains to interpret site-specific epigenetic codes.
Drawing parallels to the successful therapeutic targeting of
acetylation enzymes in oncology (83), it is postulated that the enzymatic
regulators of lactylation hold comparable potential as precision
targets for treating cancer and other pathologies.
The bromodomain-containing protein p300,
traditionally recognized for catalyzing histone H3 acetylation to
drive transcription (84), was
established by Zhang et al (9) to be a dual-function enzyme also
capable of mediating histone lactylation. Mechanistic studies have
revealed that p300 overexpression can augment histone lactylation
in 293T cells, whereas its depletion was shown to markedly diminish
H3 lysine 18 lactylation (H3K18la) in both HCT116 and 293T cell
lines (85,86). Subsequent investigations have
broadened the functional scope of p300. For example, Minami et
al (87) demonstrated that
p300 facilitated osteoblast differentiation through lactate-induced
histone modification. In cardiovascular pathology, Dong et
al (88) reported that lipid
peroxidation accelerated atherosclerosis by triggering
endothelial-to-mesenchymal transition, a process driven by
lactate-dependent H3K18la via the p300/ASF1A complex. Furthermore,
therapeutic interventions targeting p300 have shown promise; Wang
et al (89) reported that
andrographolide alleviated aortic valve calcification by disrupting
p300-mediated histone H3 lactylation. p300 also modulated
inflammatory responses; its inhibition suppressed the lactylation
of Yin-Yang 1, thereby mitigating microglial inflammation (90). In oncology, p300 has been
characterized as the primary histone lactylation writer in
pancreatic ductal adenocarcinoma (PDAC) (91). Additionally, in intrahepatic
cholangiocarcinoma, p300 has been reported to catalyze the
lactylation of nucleolin (NCL) at lysine (K)477, a modification
that fuels tumor proliferation and invasion (92).
Recent evidence has also highlighted the role of
5-methylcytosine (m5C) in CRC progression (95,96).
The m5C methyltransferase NSUN2 is frequently upregulated and
drives oncogenesis in CRC. A critical signaling axis involves NSUN2
(as the writer) and YBX1 (as the reader) regulating ENO1, thereby
reprogramming glucose metabolism to increase lactate production.
This accumulated lactate subsequently enhances NSUN2 transcription
via H3K18la and promotes NSUN2 lactylation at K356, a modification
essential for RNA binding. This establishes a positive feedback
loop within the NSUN2/YBX1/m5C-ENO1 axis, effectively linking
metabolic shifts with epigenetic remodeling (95). Additionally, TIP60 acts as the
specific writer for NBS1 lactylation at K388, a modification that
facilitates HR repair (97). In
hepatic stellate cell activation, hexokinase 2 (HK2) serves a
requisite role in driving histone lactylation-dependent gene
expression; consequently, HK2 knockdown mitigates stellate cell
activation and liver fibrosis (98). Collectively, these findings
underscore the pivotal roles of p300, AARS1, KAT8, NSUN2, TIP60 and
HK2 as key enzymatic effectors of protein lactylation.
The superfamily of histone lysine deacetylases
(HDACs) is traditionally divided into two primary lineages: The
classical zinc-dependent HDACs and the NAD+-requiring
Sirtuin (SIRT) family (99). While
the classical zinc-dependent HDAC1-3 are characterized as robust
deacylase complexes (100), the
SIRT family generally demonstrates comparatively limited
delactylase efficacy (101).
Specific functional roles have been delineated for these enzymes;
for example, Fan et al (102) reported that HDAC2 acted as a
negative regulator of angiogenesis by diminishing H3K9 lactylation.
Concurrently, HDAC2 has been implicated as a putative eraser of
histone lactylation within PDAC cells (91). During genomic maintenance, HDAC3
serves as the specific delactylase for NBS1 during DNA repair
processes (97). Furthermore, Zu
et al (103) established
SIRT2 as a proficient eraser of histone lactylation. This is
exemplified by the regulation of METTL16, an atypical
methyltransferase: Under copper stress, METTL16 undergoes
lactylation at K229, a modification that is antagonized by SIRT2.
Collectively, current evidence positions HDAC1-3 and SIRT2 as
pivotal regulatory enzymes governing the removal of lactyl
modifications.
During PTM, the functional execution of an
epigenetic mark depends not merely on its deposition by a writer,
but crucially on its subsequent recognition by specialized effector
proteins. These proteins, termed readers, possess distinct
structural domains engineered to identify and bind to specific
site-specific modifications (104). While the characterization of
readers specific to protein lactylation has historically lagged
behind that of writers and erasers, the field has seen advances
over the past 2 years. As aforementioned, YBX1 has been identified
as a direct reader of lactylated ENO1 in CRC, serving an integral
role in the NSUN2/YBX1/m5C-ENO1 signaling axis; a discovery with
profound implications for understanding CRC pathogenesis. Hu et
al (105) used proteomic
profiling of H3K18la immunoprecipitates to identify the specific
recruitment of Brg1 during cellular reprogramming. Their analysis
demonstrated co-enrichment of H3K18la and Brg1 at the promoters of
genes governing pluripotency and epithelial junction integrity.
Furthermore, biochemical validation of the physical interaction
between Brg1 and H3K18la effectively established Brg1 as a bona
fide reader of histone lactylation (105). Despite these isolated advances,
the current landscape of lactylation reader research remains
nascent and limited in scope, underscoring an urgent need for more
comprehensive mechanistic investigations to fully map this
regulatory network.
Historically, lactyl-CoA was primarily recognized
for its industrial utility as a foundational building block in the
biosynthesis of biodegradable and biocompatible lactate-derived
copolymers (106). From a
biochemical perspective, the generation of this intermediate is a
prerequisite for protein lactylation, a process that depends on the
conversion of lactate to lactyl-CoA catalyzed by the enzyme
acyl-CoA transferase. To elucidate this mechanism, Zhang et
al (106) performed
comprehensive enzymological characterization, encompassing
screening, cloning and purification, of five candidate lactyl-CoA
synthetases, specifically evaluating the stability profiles of the
three variants that demonstrated superior specific activity.
Furthermore, the endogenous existence of this metabolite has been
definitively confirmed in vivo, particularly in mouse liver
and muscle tissues. By employing liquid
chromatography-high-resolution mass spectrometry validated against
synthetic standards, investigators have successfully verified the
ubiquitous presence of lactyl-CoA across a range of mammalian cell
lines and murine tissues (107).
The generation of lactate serves as the fundamental
nexus connecting the glycolytic pathway to protein lactylation.
Generally, the abundance of lactylation modifications mirrors the
extent of glycolytic reprogramming and is directly modulated by the
concentration of lactate byproducts generated during this metabolic
shift. In a study examining the postmortem dynamics of the broiler
chicken pectoralis major muscle over 48 h, researchers observed
distinct metabolic associations: Protein lactylation was inversely
associated with glycogen reserves, glucose levels, glycolytic
potential and pH, whereas it was positively associated with lactate
accumulation. Furthermore, lactylation levels tracked consistently
with the enzymatic activities of LDH and phosphofructokinase,
suggesting that this modification may actively participate in the
feedback regulation of glycolysis by modulating enzyme expression
and function (108). In embryonic
stem cells (ESCs), glycolytic metabolism is indispensable for
regulating the pluripotency lifecycle, from establishment through
maintenance to exit. A specific focus on Esrrb, an orphan nuclear
receptor that drives pluripotency and differentiation toward
extraembryonic endoderm stem cells, has revealed that it undergoes
lactylation at K228 and K232. This modification potentiates the
binding affinity of Esrrb for target genes, thereby reinforcing ESC
self-renewal capabilities (109).
In conclusion, the glycolytic cascade is a primary determinant of
the lactylation landscape, positioning lactate as a critical
molecular bridge linking metabolic reprogramming to
post-translational signaling.
In hypoxic microenvironments, the activation of
hypoxia-inducible factor (HIF)-1α acts as a critical driver of
malignancy, fostering tumor cell migration, invasion and metastatic
spread through the promotion of epithelial-mesenchymal transition
(110). Mechanistically, HIF-1α
functions as a master transcriptional regulator that drives
glycolytic machinery (111). By
orchestrating the expression of genes essential for glycolysis,
HIF-1α indirectly amplifies lactate generation, which subsequently
fuels protein lactylation. This regulatory axis has been
corroborated by studies utilizing PX-478, a potent HIF-1α
inhibitor; inhibition of HIF-1α was shown to abrogate the surge in
extracellular lactate levels, thereby confirming its modulatory
role in lactylation substrate availability (112,113). Beyond indirect regulation,
hypoxia may also exert direct control over lactylation-associated
enzymes. Recent findings have indicated that intracellular hypoxia
triggers mitochondrial protein lactylation via a distinct
mechanism: It induces the accumulation of mitochondrial AARS
(AARS2). Acting as a non-canonical lysine lactyltransferase, AARS2
targets and inactivates key metabolic enzymes, specifically the
pyruvate dehydrogenase complex (PDC) and carnitine
palmitoyltransferase 2 (114).
There is a distinct association between p53 status
and the expression profile of proteins governing aerobic glycolysis
in HCC cells. Functionally, the stabilization and subsequent
activation of p53 effectively downregulate genes driving aerobic
glycolysis and restricts glycolytic flux; this regulatory action
notably diminishes lactate generation, thereby impeding the
progression of HCC (115). Other
studies in HCC have demonstrated that interrupting the
p53/isocitrate dehydrogenase 1/HIF-1α signaling cascade shifts
cellular metabolism toward mitochondrial respiration while
suppressing aerobic glycolysis (116,117). This metabolic switch results in
reduced lactate output, further arresting malignant advancement
(116). Collectively, these
findings indicate that p53 exerts its regulatory influence on
lactylation primarily by indirectly modulating the glycolytic
pathway.
While histone lactylation is recognized as a
canonical epigenetic modifier, emerging proteomic landscapes have
unveiled a ubiquitous role for this modification across the
non-histone proteome, where it critically governs protein
stability, enzymatic kinetics and interactome dynamics. These
non-histone substrates primarily cluster into three functional
domains: Metabolic regulation, DNA damage response and signal
transduction, which together orchestrate complex networks that fuel
tumorigenesis.
Metabolic enzymes are primary targets of
lactylation, establishing a feed-forward mechanism that amplifies
the Warburg effect. Specific examples include the lactylation of
PDHX at K488 in HCC, which impairs complex assembly to block
oxidative phosphorylation while boosting glycolysis (118). Furthermore, aldolase A (ALDOA)
lactylation diminishes its binding to DEAD-box helicase 17 (DDX17),
a molecular event that augments the stem-like properties of liver
cancer cells (119). In
immunometabolism, the modification of pyruvate kinase M2 (PKM2) at
K62 acts as a switch for macrophage reprogramming, effectively
dampening inflammatory outputs (120).
In genomic integrity, lactylation modulates DNA
repair machinery, directly underpinning therapeutic resistance. The
HR factor MRE11 is lactylated at K673, a modification that
potentiates DNA repair efficiency, and consequently reduces cancer
cell susceptibility to cisplatin and PARPi (82). Similarly, lactylation events on
NBS1 (at K388) and XRCC1 (at K247) have been characterized as
drivers of chemo- and radioresistance across diverse malignancies
(97,121). This modification tunes critical
signaling effectors governing proliferation and immune escape. For
example, in Tregs, lactylation of Moesin at K72 amplifies TGF-β
signaling, thereby facilitating Treg differentiation and fostering
an immunosuppressive niche (122). In intrahepatic
cholangiocarcinoma, p300-mediated lactylation of NCL has been shown
to drive tumor expansion and invasion by regulating RNA splicing
(92). For a comprehensive
synthesis of these findings, the specific substrates, modification
sites and downstream consequences are systematically detailed in
Table I.
Metabolic reprogramming has been widely recognized
as a fundamental driver of tumorigenesis and malignant progression
(123). To fuel sustained
proliferation, neoplastic cells undergo profound bioenergetic
restructuring (33). Lactylation
has emerged as a pivotal epigenetic modulator, altering chromatin
topology to influence transcriptional landscapes. Crucially, this
modification creates a bidirectional regulatory axis; it not only
reshapes the transcriptional and functional profiles of key
glycolytic enzymes (124), but
also serves as a molecular bridge connecting metabolism to
epigenetics, thereby directly upregulating metabolism-associated
genes (125).
As established, LDHA functions as the central enzyme
catalyzing pyruvate-to-lactate conversion. Concurrently, GLUT3 is
integral to glucose uptake and glycolysis in cancer cells (126). Single-cell sequencing data have
revealed the notable upregulation of GLUT3 in both primary and
metastatic lesions. Notably, GLUT3 expression has been shown to be
positively associated with LDHA levels and lactylation pathways; in
gastric cancer, GLUT3 has been reported to modulate lactylation
dynamics, thereby driving disease progression (127). In esophageal carcinoma, hypoxic
conditions trigger the lactylation of Axin1 and SHMT2 proteins, a
modification that further potentiates cell proliferation,
metastasis and stemness features (128,129).
The landscape in non-small cell LC (NSCLC) appears
multifaceted. Evidence has suggested that lactate can suppress the
mRNA levels of glycolytic enzymes (HK1 and PKM) and TCA cycle
enzymes (SDHA and IDH3G); chromatin immunoprecipitation assays have
confirmed an enrichment of histone lactylation at the promoters of
HK1 and IDH3G, indicating that lactate regulates metabolism partly
through epigenetic repression (130). Conversely, hypoxia-induced
lactylation of SOX9 has been reported to amplify glycolytic
activity, thereby enhancing stemness and invasive potential in
NSCLC cells (131). Thus,
lactylation exerts complex, context-dependent effects on glycolysis
within this malignancy.
In LC models, the IGF1/IGF1R axis is critical for
metabolic regulation. Research has indicated that stability of the
IGF1R oncogene is enhanced by lactate-mediated lactylation,
establishing a positive feedback loop that further fuels glycolysis
and lactate generation (132). In
pancreatic adenocarcinoma, studies have linked elevated global
histone lactylation to poor prognosis (91,133). Mechanistically, EP300 has been
identified as the writer catalyzing lactylation at K128 of NMNAT1.
This modification supports the nuclear NAD+ salvage
pathway, thereby sustaining tumor growth (32). Furthermore, in CC, lactate has been
shown to upregulate and stabilize DCBLD1, a type I protein
containing an LCCL domain, via lactylation. This stabilization
activates the pentose phosphate pathway, ultimately promoting
proliferation and metastasis (134). Collectively, these findings
underscore protein lactylation as a critical nexus integrating
lactate metabolism, oncogenic signaling and clinical outcomes.
Functionally and structurally, the TME constitutes a
sophisticated ecosystem orchestrated by a heterogeneous array of
cell types, including endothelial cells, CAFs, mesenchymal stromal
cells and various immune cell subsets, such as macrophages,
dendritic cells, NK cells, T cells and B cells. Crucially, this
multicellular environment establishes a permissive niche that
actively supports protein lactylation. Therefore, dissecting the
crosstalk between these TME constituents and lactylation pathways,
particularly their contributions to tumorigenesis, invasive
behavior, and immune evasion, holds potential to advance the
frontiers of cancer immunotherapy.
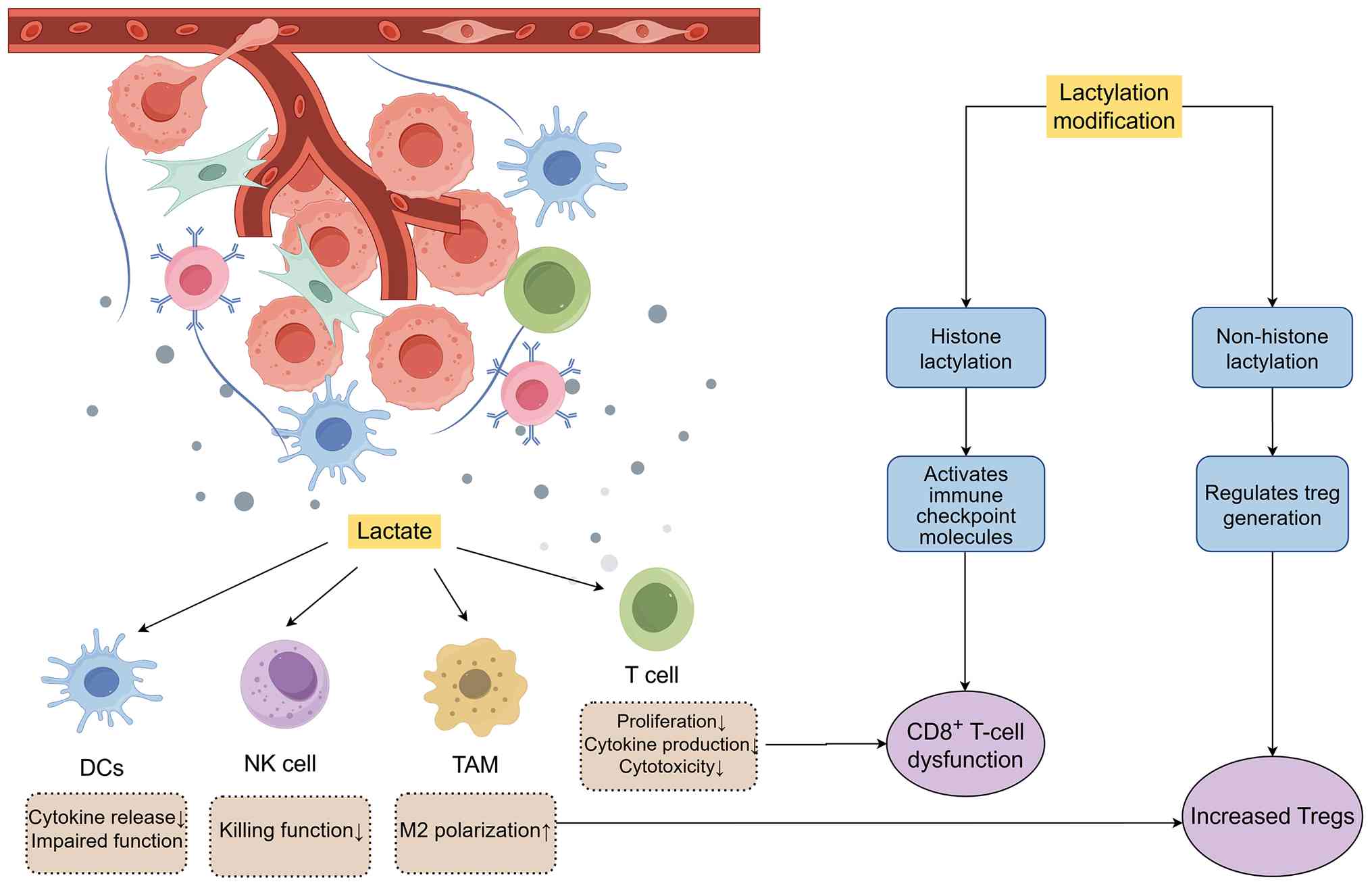
Immune populations represent pivotal constituents of
the TME; consequently, elucidating the dynamics of lactylation
within these cells is of profound scientific significance (Fig. 2) (135).
Traditionally, the immune system is conceptualized
as a defensive apparatus tasked with distinguishing self from
non-self, orchestrating innate and adaptive responses to eliminate
threats and preserve tissue homeostasis (136). Within this framework, T
lymphocytes occupy a central position (137). As the linchpins of cellular
immunotherapy and tumor surveillance, T cells are responsible for
antigen recognition, cytokine secretion, and the coordination of
broad immune responses to thwart malignant invasion. Based on their
distinct surface markers and functional profiles, these lymphocytes
are further categorized into specialized subsets (138).
Research highlights the notable sensitivity of T
lymphocytes to the lactate-rich milieu of the TME, where lactate
signaling fundamentally alters cellular signaling and therapeutic
outcomes (139). Specifically,
lactate accumulation drives histone lactylation, thereby
compromising histone transcriptional activity in CD4+
and CD8+ cells. This epigenetic suppression reduces
cytokine production and cytolytic efficacy, ultimately blunting
lymphocyte responses and favoring tumor progression (140–142).
In head and neck squamous cell carcinoma (HNSCC),
H3K9 lactylation is linked to immunotherapy resistance, activating
immune checkpoints via IL-11 and causing CD8+ T-cell
dysfunction (142). Furthermore,
GLUT10 has been implicated as a crucial GLUT for CD8+
T-cell activation. Lactate accumulates in the TME and directly
binds to GLUT10, blocking glucose uptake; therefore, disrupting
this lactate-GLUT10 interaction offers a novel therapeutic avenue
to restore CD8+ cytotoxicity (145).
The clinical efficacy of PD-1 blockade hinges on the
balance between effector CD8+ T cells and
immunosuppressive Tregs. In highly glycolytic tumors (such as
MYC-amplified or hepatic cancers), Tregs exhibit higher PD-1 levels
than effector T cells. This occurs because Tregs in low-glucose
environments actively import lactate via MCT1, thereby driving
nuclear translocation of NFAT1 and upregulating PD-1, whereas
effector expression is suppressed. Thus, a lactate-rich TME
selectively upregulates PD-1 on Tregs (146). Additionally, lactate fuels Treg
generation through the specific lactylation of Moesin at K72. This
modification strengthens the interaction between Moesin and TGF-β
receptor I and activates downstream SMAD3 signaling. Consequently,
combining anti-PD-1 therapy with LDH inhibitors yields superior
efficacy over monotherapy (122).
These findings suggest that targeting lactylation-driven Treg
differentiation constitutes a promising strategy to provide novel
options in cancer treatment.
As cornerstones of the innate immune architecture,
macrophages are defined by their marked phenotypic malleability.
This high plasticity allows them to orchestrate responses to
diverse physiological and pathological cues by transitioning
between functional states (147,148). This heterogeneity is
traditionally conceptualized as ‘polarization’, a spectrum often
simplified into a dichotomy dictated by the microenvironment: The
classically activated (M1) and alternatively activated (M2)
phenotypes, which generally exhibit antagonistic roles (149). M1 macrophages are characterized
by robust antigen-presenting capabilities, and the secretion of
proinflammatory cytokines such as IL-12, IL-23 and TNF-α (150). Conversely, the M2 phenotype is
predominantly aligned with anti-inflammatory processes, and is
strongly implicated in fostering tumor proliferation and metastasis
(55). Emerging evidence has
suggested that metabolic rewiring is not merely a consequence but a
prerequisite for this phenotypic switching (151). Specifically, the lactylation of
PKM2 at K62 has been isolated as a pivotal mechanism driving the
metabolic reprogramming essential for macrophage inflammatory
activation (120).
In the TME, TAMs display notable heterogeneity,
adapting their function in response to niche-specific signals.
While it is established that metabolic shifts accompany these
phenotypic changes, recent findings have argued that metabolic
reprogramming actively dictates the activation state and function
of macrophages (152,153). For example, in CRC, RARγ
downregulation in TAMs is associated with poor clinical outcomes.
Mechanistically, tumor-derived lactate drives H3K18 lactylation,
which transcriptionally suppresses the RARγ gene in macrophages,
thereby accelerating disease progression (154). Both bioinformatics profiling and
real-world clinical data corroborate that elevated H3K18
lactylation serves as a negative prognostic indicator (155,156). Notably, the regulation is
bidirectional: Macrophages can also modulate the lactylation
landscape. In glioblastoma (GBM) models, monocyte-derived
macrophages have been shown to actively erase histone lactylation.
This erasure facilitates the intratumoral accumulation of T cells
and suppresses tumor growth (157). Consequently, a reciprocal
regulatory axis between macrophages and lactylation collectively
shapes the trajectory of tumor initiation and development.
In addition to macrophages and T cells, high
lactate accumulation in the TME also impairs the function and
cytokine release of DCs (21). In
tumor-infiltrating myeloid cells (TIMs), the upregulation of
lactylation enzyme METTL3 induces immunosuppressive effects in CRC
(158). Lactic acid inhibits the
differentiation and antigen presentation of DCs, promoting their
transformation into tumor-associated DCs (TADCs), which results in
a reduction of IL-12 secretion and impaired immune response
(159). The transcription factor
SREBP2, influenced by lactic acid, further regulates the maturation
of DCs and their immunosuppressive functions, thereby inhibiting
anti-tumor immune responses (160).
Leveraging established gene signatures associated
with histone lactylation, a study utilized single-sample gene set
enrichment analysis to assess the cellular landscape of the TME.
The analysis highlighted a clear stratification; the
low-lactylation cohort was characterized by a robust infiltration
of NK cells and cytotoxic lymphocytes. Conversely, the
high-lactylation group exhibited a predominance of stromal
elements, specifically showing enriched populations of fibroblasts
and endothelial cells (125).
CAFs represent a prominent and diverse mesenchymal
population within the TME, serving a pivotal role in orchestrating
malignant behaviors (161). In
clear cell renal cell carcinoma (ccRCC), histone lactylation in
CAFs has been shown to drive tumor progression, with TIMP1
identified as a central downstream target of this epigenetic
modification (162). Furthermore,
in gastric cancer, the epigenetic landscape of CAFs, specifically
defined by H3K18la levels, critically affects the therapeutic
efficacy of immune checkpoint inhibitors by regulating PD-L1
abundance (163). Regarding the
vascular component, tumor endothelial cells typically remain
quiescent under physiological conditions. However, in lung
adenocarcinoma (LUAD), SLC25A29 has emerged as a vital prognostic
determinant linked to tumor staging. Mechanistic studies have
confirmed that SLC25A29 expression is epigenetically regulated by
H3K14 and H3K18 lactylation at its promoter (164,165). Functionally, downregulation of
SLC25A29 promotes endothelial proliferation and migration while
suppressing apoptosis, thereby accelerating LUAD progression
(166).
Epigenetic dysregulation is increasingly recognized
as a cornerstone of tumorigenesis. As a newly defined epigenetic
modification, lactylation orchestrates tumor development by
regulating both histone and non-histone substrates, while
simultaneously modulating metabolic plasticity (167–169). HCC represents a highly lethal
malignancy and ranks as the second leading cause of cancer
mortality globally (170). Its
pathogenesis is frequently preceded by chronic liver injury,
stemming from hepatitis B, hepatitis C or steatohepatitis, where
the risk of mortality scales exponentially with the severity of
fibrosis (171). The pathogenic
continuum from fibrosis to cirrhosis, and ultimately to HCC,
involves complex regulatory shifts (172).
Emerging evidence has implicated lactylation, a
structural analog of acetylation, as a driver of this fibrotic
progression. Notably, gene signatures marked by histone lactylation
in fibrotic tissues are concomitantly upregulated in HCC,
suggesting a direct mechanistic link between fibrotic remodeling
and hepatocarcinogenesis (173).
Furthermore, liver cancer stem cells (LCSCs) underpin the
phenotypic heterogeneity of HCC. Proteomic profiling has unveiled
that ALDOA undergoes lactylation at residues K230 and K322;
mechanistically, this modification disrupts the physical
interaction between ALDOA and DDX17, thereby potentiating DDX17
function and reinforcing the stem-like phenotype of LCSCs (119).
In BC, The Cancer Genome Atlas analysis has
highlighted the pivotal role of lactylation (174). Specifically in triple-negative BC
(TNBC), an aggressive subtype, elevated histone H4 lysine 12
lactylation (H4K12la) is inversely associated with OS (175). This poor prognosis is driven by
the H4K12la-mediated transcriptional suppression of Schlafen 5,
delineating a novel oncogenic axis (176). Similarly, in ovarian cancer (OC),
the chemokine CCL18 is frequently upregulated; this is
epigenetically driven by H3K18 lactylation, which actively fuels
tumorigenesis (177).
Uncontrolled proliferation and metastatic
dissemination represent defining hallmarks of malignancy, with the
latter accounting for ~90% of cases of cancer-related mortality
(178,179). Against this backdrop, the rapidly
expanding field of lactylation research offers novel therapeutic
paradigms for the treatment of refractory tumors. In HCC,
lactylation drives malignancy through dual epigenetic and metabolic
mechanisms. First, pyrroline-5-carboxylate reductase 1 increases
IRS1 transcriptional activity by modulating H3K18 lactylation at
its promoter, thereby increasing proliferation and metastasis
(180). Moreover, the metabolic
shift from oxidative phosphorylation to aerobic glycolysis is
cemented by inactivation of the mitochondrial PDC. Specifically,
lactylation of the PDHX component at K488 impairs PDC assembly, a
critical event that accelerates HCC progression (118).
In TNBC, elevated glycolytic flux initiates a
specific oncogenic cascade: It promotes histone lactylation at the
c-Myc promoter, leading to c-Myc upregulation. This, in turn,
transcriptionally activates the splicing factor SRSF10, ultimately
driving accelerated tumor growth (181). Similarly, in OC, bioinformatics
profiling has linked lactylation-associated gene signatures to
specific tumor subtypes and immune landscapes (156). Immunohistochemical validation has
further confirmed that elevated histone lactylation serves as a
robust indicator of poor prognosis in patients with OC (155).
Immune evasion constitutes a fundamental barrier in
oncology. In NSCLC, elevated lactylation is associated with adverse
outcomes. Mechanistically, H3K18la directly drives POM121
transcription, activating the POM121/MYC/PD-L1 signaling axis to
blunt immune surveillance. Consequently, targeting this axis by
combining glycolysis inhibitors with anti-PD-1 antibodies has
demonstrated potent antitumor efficacy (182).
The CRC landscape reveals complex roles for
lactylation in initiation, progression and therapeutic resistance
(94,183). Regarding chemoresistance, SMC4
downregulation induces a ‘diapause-like’ quiescent state. This
downregulation increases lactate production via glycolytic enzymes
while suppressing PGAM1. The resulting increase in lactate levels
drives histone lactylation to upregulate ABC transporters, thereby
conferring chemoresistance (184). Furthermore, the interplay between
the microbiome and non-histone lactylation is critical yet
underappreciated (185,186). For example, tumor-resident '
suppresses NF-κB signaling by inducing lactylation of RIG-I. This
modification drives M2 macrophage polarization and facilitates
colorectal liver metastasis. Targeting this pathway with a specific
RIG-I lactylation inhibitor has been shown to restore sensitivity
to 5-fluorouracil (187).
In ccRCC, pathogenesis is closely tied to the
inactivation of von Hippel-Lindau (VHL) (188). Loss of VHL function perturbs
histone lactylation dynamics, leading to the transcriptional
activation of PDGFRβ. This establishes a positive feedback loop
between histone lactylation and PDGFRβ signaling, presenting a
viable therapeutic target (189).
Beyond solid tumors, lactylation markedly impacts
hematological malignancies. In leukemia, histone lactylation
promotes progression by upregulating ALKBH3, which removes m1A
methylation from SP100A and consequently destabilizes the tumor
suppressor PML (190). In diffuse
large B-cell lymphoma, elevated lactate levels are closely linked
to prognosis, immune function and drug resistance (191). Similarly, in multiple myeloma
(MM), lactylation-related genes have emerged as prognostic
biomarkers (192). Addressing
resistance to proteasome inhibitors in MM, research indicates that
Mucin20 drives resistance by modulating the MET signaling pathway
through the suppression of IGF-1R lactylation (193). Despite these advances, the field
remains nascent and mechanistic landscapes await further
elucidation.
Lactate serves as the primary substrate for protein
lactylation and drives tumor progression through multiple molecular
mechanisms. Consequently, pharmacologically intercepting the
lactate axis constitutes a promising frontier in oncology (194). Current therapeutic strategies
have focused extensively on blocking lactate metabolism and
transport, with research directed toward LDHA/B and MCT1/4.
Regarding LDH inhibition, several potent compounds
targeting LDHA have emerged. For example, ML-05 has been shown to
effectively curtail cellular lactate synthesis and suppress
proliferation in melanoma models (195). Furthermore, evidence suggests
that LDHA inhibitors can function as valuable adjunctive agents in
pancreatic cancer regimens (196). While LDHB is generally considered
secondary to LDHA in tumorigenesis, it retains functional
importance in modulating autophagy, apoptosis and the immune
microenvironment (197,198). To date, the selective LDHB
inhibitor AXKO-0046 has demonstrated competitive inhibition;
however, it remains a tool for mechanistic validation in cancer
metabolism rather than a clinical therapeutic (199).
AZD3965 represents a first-in-class inhibitor
targeting MCT1 and MCT2 with selectivity over MCT3 and MCT4.
Preclinically, it suppresses hematological tumor growth, and
exhibits synergy with doxorubicin or rituximab in diffuse large
B-cell and Burkitt lymphomas (200). Crucially, the 2023 publication of
its Phase I trial (NCT01791595) provided pivotal clinical insights.
While the drug showed a favorable safety profile, its single-agent
efficacy was limited, likely due to a compensatory lactate efflux
mechanism mediated by MCT4 in solid tumors. This underscores the
necessity for patient stratification strategies, specifically
targeting MCT4-negative tumors, or the development of combination
therapies (201).
Conversely, MCT4 is essential for lactate efflux
under hypoxic conditions, and its blockade can induce lethal
intracellular lactate accumulation (202). In HNSCC, MCT4 inhibition has been
proven to attenuate invasiveness (203). A notable advancement involves
VB124, a potent MCT4 inhibitor that restricts liver tumor growth in
immunocompetent models by potentiating CD8+ T-cell
infiltration and cytotoxicity (204). These findings position VB124 as a
high-priority candidate for future clinical investigation,
particularly in synergistic combinations with immunotherapy.
Therapeutic recalcitrance represents a formidable
barrier in oncology, profoundly undermining the clinical efficacy
of antineoplastic regimens (205). Accumulating evidence implicates
protein lactylation, which affects both histone and non-histone
substrates, as a potent driver of this phenomenon, conferring
resistance to radiotherapy and chemotherapy through multiple
molecular mechanisms.
In GBM, clinical outcomes have revealed that
patients with elevated ALDH1A3 expression derive minimal benefit
from postoperative chemoradiation. To elucidate the molecular basis
of this failure, proteomic profiling of lactate-rich GBM stem cells
identified the lactylation of XRCC1 at K247 as a pivotal
determinant of radio- and chemoresistance (121). Parallel investigations in GBM
have further established that H3K9 histone lactylation drives
temozolomide resistance. Notably, the repurposed antiepileptic
agent stiripentol has been shown to effectively inhibit this
lactylation event, offering a viable combinatorial strategy for GBM
management (206).
In bladder cancer, where resistance to
platinum-based agents is common, single-cell RNA sequencing has
unveiled a strong association between cisplatin resistance and
histone H3K18la (207).
Similarly, for advanced prostate cancer, resistance to the androgen
receptor signaling inhibitor enzalutamide poses a notable
challenge. Mechanistic studies attribute this to the
NF-κB/STAT3/SLC4A4 signaling axis, which facilitates p53
lactylation, thereby driving tumor progression and drug
insensitivity (208,209).
In HCC, acquired resistance remains the primary
limiting factor for molecular targeted therapies. In
lenvatinib-resistant models, a lactylated IGF2BP3-PCK2-SAM-m6A
feedback loop has been found to sustain high expression of PCK2 and
NRF2; this upregulation bolsters the antioxidant defense system,
thereby maintaining lenvatinib resistance (210). Furthermore, although bevacizumab
is a cornerstone of CRC treatment, its efficacy is often
compromised by resistance mechanisms linked to histone lactylation.
Consequently, targeting histone lactylation in conjunction with
bevacizumab represents a novel therapeutic option for overcoming
resistance in advanced malignancies (183). For reference, detailed data
regarding these treatment associations are compiled in Table II.
Despite the substantial therapeutic potential of
targeting lactate metabolism and lactylation, translation of these
strategies into clinical practice is currently hindered by
limitations in single-agent potency and pharmacological
specificity.
Monotherapies frequently fail to achieve durable
responses, a failure attributed to tumor metabolic flexibility and
compensatory activation of alternative transport mechanisms,
exemplified by the upregulation of MCT4 following MCT1 blockade. To
overcome this, combinatorial regimens have been prioritized to
induce synergistic antitumor activity. A particularly promising
option involves coupling glycolysis or lactate inhibitors with
immunotherapeutic agents (211).
Given that lactate accumulation within the TME actively dampens
T-cell cytotoxicity, therapeutic depletion of lactate alleviates
this immunosuppression. Indeed, empirical data have demonstrated
that the concurrent administration of anti-PD-1 antibodies and LDH
inhibitors yields efficacy superior to that of either agent alone
(212). In NSCLC, combining
glycolysis inhibitors with immune checkpoint blockade has been
shown to disrupt the histone lactylation-driven POM121/MYC/PD-L1
signaling axis, effectively resensitizing resistant tumors to
anti-PD-1 therapy (182).
Conversely, the development of lactylation-specific
inhibitors is constrained by the risk of off-target toxicity. A
notable bottleneck is the functional duality of ‘writer’ enzymes;
p300, for example, functions as both a histone acetyltransferase
and a lactyltransferase (85).
Consequently, indiscriminate inhibition of p300 risks suppressing
physiological histone acetylation, which could catastrophically
disrupt essential transcriptional programs and cellular
homeostasis. Future medicinal chemistry efforts must therefore
prioritize the structural delineation of domains specific to
lactyl-CoA binding. The goal is to uncouple lactylation inhibition
from acetylation, thereby maximizing therapeutic precision while
minimizing collateral toxicity. Furthermore, recent literature
positions lactylation as a central epigenetic node integrating
multifactorial resistance mechanisms. This suggests that targeting
downstream effectors, rather than the writers themselves, may offer
a strategic bypass around compensatory metabolic adaptations
(213).
While the therapeutic potential of targeting
lactylation is evident, translating these findings from bench to
bedside faces several critical hurdles that must be addressed in
future research.
Currently, the identification of lactylation relies
heavily on HPLC-MS/MS and isotope metabolic labeling (9). While robust for research, these
methods are costly, time-consuming and impractical for routine
clinical diagnosis using tissue biopsies or blood samples. Although
immunohistochemistry has been used to detect lactylation in OC
tissues (155), standardization
of antibodies remains a challenge. Future priorities should focus
on developing highly specific, lactylation-sensitive imaging agents
and noninvasive liquid-biopsy biomarkers to enable real-time
monitoring of lactylation dynamics in patients.
Lactylation levels exhibit notable heterogeneity
not only among patients but also within the TME. For example,
single-cell analyses have revealed distinct lactylation patterns in
different cell subsets, such as macrophages and cancer stem cells
(119,120). This heterogeneity suggests that a
‘one-size-fits-all’ metabolic therapy may fail. Consequently, the
construction of patient-derived xenograft models or organoids to
evaluate individual sensitivity to lactylation inhibitors is
essential. Furthermore, gene signatures related to lactylation, as
identified in OC (156), should
be validated as predictive tools for therapeutic response.
Consistent with this, recent literature posits that deciphering the
specific interaction networks between lactylation and immune
checkpoints is critical for advancing precision oncology (214).
A major ethical and safety concern is that lactate
is not solely a tumor metabolite but a critical fuel for normal
physiological functions, including cardiac modulation (13) and skeletal muscle metabolism
(39). Broad-spectrum inhibition
of lactate production or transport (via systemic LDH or MCT
inhibition) carries a risk of severe systemic toxicity, including
muscle fatigue and cardiac dysfunction. Therefore, developing drug
delivery systems that specifically target the acidic TME or
exploiting synthetic lethality to minimize off-target effects on
healthy tissues is a prerequisite for clinical trials.
Lactate has long been regarded as the end product
of glycolysis, but it is now recognized as a multifunctional
signaling molecule that serves a central role in promoting tumor
cell metabolism and immune evasion. Lactylation, a recently
discovered PTM, directly affects the functions of both histones and
non-histones, linking metabolic reprogramming to epigenetic
regulation. Moreover, lactylation modifications, by modulating
cellular metabolic pathways, markedly influence tumor progression,
immune suppression and mechanisms of drug resistance across various
cancer types. In tumor immunology, lactylation influences the
polarization and function of immune cells, including T cells and
macrophages, thereby suppressing antitumor immune responses and
promoting tumor resistance and recurrence. In the future, in-depth
investigation of the mechanisms underlying lactylation,
identification of its regulatory factors and the development of
targeted therapeutics may provide novel directions and strategies
for cancer treatment. By combining metabolic inhibitors with
immunotherapy, it is anticipated that the efficacy of cancer
treatment can be enhanced and resistance to current therapies can
be overcome. These studies will help elucidate the complex role of
lactylation modifications in tumorigenesis and drive the
development of innovative therapeutic approaches.
Not applicable.
The present study was supported in part by the National Natural
Science Foundation of China (grant no. 62372276) and in part by the
Investigator-Initiated Basic Research Project of Beijing Luhe
Hospital (grant nos. LHYY2025-YJZJC0011 and
LHYY2025-YJZJC0012).
Not applicable.
All authors contributed to the study conception and
design. Literature search, data extraction and literature analysis
were performed by YH, WY, QH and YZ. The first draft of the
manuscript was written by CZ, XT and NC, and all authors commented
on previous versions of the manuscript. Data authentication is not
applicable. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Dart A: Tumour metabolism: Lactic acid:
Not just a waste product? Nat Rev Cancer. 16:676–677. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Koukourakis MI and Giatromanolaki A:
Warburg effect, lactate dehydrogenase, and radio/chemo-therapy
efficacy. Int J Radiat Biol. 95:408–426. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Warburg O, Wind F and Negelein E: The
metabolism of tumors in the body. J Gen Physiol. 8:519–530. 1927.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen AN, Luo Y, Yang YH, Fu JT, Geng XM,
Shi JP and Yang J: Lactylation, a novel metabolic reprogramming
code: Current status and prospects. Front Immunol. 12:6889102021.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shih CC, Lee TS, Tsuang FY, Lin PL, Cheng
YJ, Cheng HL and Wu CY: Pretreatment serum lactate level as a
prognostic biomarker in patients undergoing supratentorial primary
brain tumor resection. Oncotarget. 8:63715–63723. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang S, Osgood AO and Chatterjee A:
Uncovering post-translational modification-associated
protein-protein interactions. Curr Opin Struct Biol. 74:1023522022.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen L and Kashina A: Post-translational
modifications of the Protein Termini. Front Cell Dev Biol.
9:7195902021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pan S and Chen R: Pathological implication
of protein post-translational modifications in cancer. Mol Aspects
Med. 86:1010972022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang D, Tang Z, Huang H, Zhou G, Cui C,
Weng Y, Liu W, Kim S, Lee S, Perez-Neut M, et al: Metabolic
regulation of gene expression by histone lactylation. Nature.
574:575–580. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hu Y, He Z, Li Z, Wang Y, Wu N, Sun H,
Zhou Z, Hu Q and Cong X: Lactylation: The novel histone
modification influence on gene expression, protein function, and
disease. Clin Epigenetics. 16:722024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu W, Zhang T, Wang B and Yin H: Aerobic
glycolysis promotes NLRP3 inflammasome activation via NLRP3
lactylation. Cell Chem Biol. 33:213–226.e5. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhang Y, Wang J, Li C, Lei Z, Zhao X, Xuan
X, Sukocheva O, Tse E and Liu J: Protein lactylation in cancer and
other pathologies: Epigenetic regulation of glycolysis and its
therapeutic perspectives. Semin Cancer Biol. 118:28–43. 2026.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ghosh-Choudhary S and Finkel T:
Lactylation regulates cardiac function. Cell Res. 33:653–654. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yang D, Zheng H, Lu W, Tian X, Sun Y and
Peng H: histone lactylation is involved in mouse oocyte maturation
and embryo development. Int J Mol Sci. 25:48212024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nian F, Qian Y, Xu F, Yang M, Wang H and
Zhang Z: LDHA promotes osteoblast differentiation through histone
lactylation. Biochem Biophys Res Commun. 615:31–35. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Susser LI, Nguyen MA, Geoffrion M, Emerton
C, Ouimet M, Khacho M and Rayner KJ: Mitochondrial fragmentation
promotes inflammation resolution responses in macrophages via
histone lactylation. Mol Cell Biol. 43:531–546. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen J, Feng Q, Qiao Y, Pan S, Liang L,
Liu Y, Zhang X, Liu D and Liu Z and Liu Z: ACSF2 and lysine
lactylation contribute to renal tubule injury in diabetes.
Diabetologia. 67:1429–1443. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhou Y, Yang L, Liu X and Wang H:
Lactylation may be a novel posttranslational modification in
inflammation in neonatal hypoxic-ischemic encephalopathy. Front
Pharmacol. 13:9268022022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li J, Zeng G, Zhang Z, Wang Y, Shao M, Li
C, Lu Z, Zhao Y, Zhang F and Ding W: Urban airborne PM(2.5) induces
pulmonary fibrosis through triggering glycolysis and subsequent
modification of histone lactylation in macrophages. Ecotoxicol
Environ Saf. 273:1161622024. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yu X, Yang J, Xu J, Pan H, Wang W, Yu X
and Shi S: Histone lactylation: From tumor lactate metabolism to
epigenetic regulation. Int J Biol Sci. 20:1833–1854. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen J, Huang Z, Chen Y, Tian H, Chai P,
Shen Y, Yao Y, Xu S, Ge S and Jia R: Lactate and lactylation in
cancer. Signal Transduct Target Ther. 10:382025. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Qiao Q, Hu S and Wang X: The regulatory
roles and clinical significance of glycolysis in tumor. Cancer
Commun (Lond). 44:761–786. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yadav D, Yadav A, Bhattacharya S, Dagar A,
Kumar V and Rani R: GLUT and HK: Two primary and essential key
players in tumor glycolysis. Semin Cancer Biol. 100:17–27. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sun Q, Güven B, Wagg CS, Almeida de
Oliveira A, Silver H, Zhang L, Chen B, Wei K, Ketema EB, Karwi QG,
et al: Mitochondrial fatty acid oxidation is the major source of
cardiac adenosine triphosphate production in heart failure with
preserved ejection fraction. Cardiovasc Res. 120:360–371. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Rogatzki MJ, Ferguson BS, Goodwin ML and
Gladden LB: Lactate is always the end product of glycolysis. Front
Neurosci. 9:222015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Read JA, Winter VJ, Eszes CM, Sessions RB
and Brady RL: Structural basis for altered activity of M- and
H-isozyme forms of human lactate dehydrogenase. Proteins.
43:175–185. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Vanderlinde RE: Measurement of total
lactate dehydrogenase activity. Ann Clin Lab Sci. 15:13–31.
1985.PubMed/NCBI
|
|
28
|
Serganova I, Cohen IJ, Vemuri K, Shindo M,
Maeda M, Mane M, Moroz E, Khanin R, Satagopan J, Koutcher JA and
Blasberg R: LDH-A regulates the tumor microenvironment via
HIF-signaling and modulates the immune response. PLoS One.
13:e02039652018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Pérez-Escuredo J, Dadhich RK, Dhup S,
Cacace A, Van Hée VF, De Saedeleer CJ, Sboarina M, Rodriguez F,
Fontenille MJ, Brisson L, et al: Lactate promotes glutamine uptake
and metabolism in oxidative cancer cells. Cell Cycle. 15:72–83.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang Z, Liu R, Shuai Y, Huang Y, Jin R,
Wang X and Luo J: ASCT2 (SLC1A5)-dependent glutamine uptake is
involved in the progression of head and neck squamous cell
carcinoma. Br J Cancer. 122:82–93. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hensley CT, Wasti AT and DeBerardinis RJ:
Glutamine and cancer: Cell biology, physiology, and clinical
opportunities. J Clin Invest. 123:3678–3684. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Huang H, Wang S, Xia H, Zhao X, Chen K,
Jin G, Zhou S, Lu Z, Chen T, Yu H, et al: Lactate enhances NMNAT1
lactylation to sustain nuclear NAD(+) salvage pathway and promote
survival of pancreatic adenocarcinoma cells under glucose-deprived
conditions. Cancer Lett. 588:2168062024. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liberti MV and Locasale JW: The warburg
effect: How does it benefit cancer cells? Trends Biochem Sci.
41:211–218. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shang RZ, Qu SB and Wang DS: Reprogramming
of glucose metabolism in hepatocellular carcinoma: Progress and
prospects. World J Gastroenterol. 22:9933–9943. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hay N: Reprogramming glucose metabolism in
cancer: can it be exploited for cancer therapy? Nat Rev Cancer.
16:635–649. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Dienel GA: Brain lactate metabolism: The
discoveries and the controversies. J Cereb Blood Flow Metab.
32:1107–1138. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Dong S, Qian L, Cheng Z, Chen C, Wang K,
Hu S, Zhang X and Wu T: Lactate and myocardiac energy metabolism.
Front Physiol. 12:7150812021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Bartoloni B, Mannelli M, Gamberi T and
Fiaschi T: The multiple roles of lactate in the skeletal muscle.
Cells. 13:11772024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Igah I, Ihediwa U and Uzoigwe CE: Lactic
acidosis. N Engl J Med. 372:10772015.PubMed/NCBI
|
|
41
|
Manoj KM, Nirusimhan V, Parashar A, Edward
J and Gideon DA: Murburn precepts for lactic-acidosis, Cori cycle,
and Warburg effect: Interactive dynamics of dehydrogenases,
protons, and oxygen. J Cell Physiol. 237:1902–1922. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Bisetto S, Wright MC, Nowak RA, Lepore AC,
Khurana TS, Loro E and Philp NJ: New insights into the lactate
shuttle: Role of MCT4 in the modulation of the exercise capacity.
iScience. 22:507–518. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hong CS, Graham NA, Gu W, Espindola
Camacho C, Mah V, Maresh EL, Alavi M, Bagryanova L, Krotee PAL,
Gardner BK, et al: MCT1 modulates cancer cell pyruvate export and
growth of tumors that Co-express MCT1 and MCT4. Cell Rep.
14:1590–1601. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Anderson NM and Simon MC: The tumor
microenvironment. Curr Biol. 30:R921–R925. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Gao Y, Zhou H, Liu G, Wu J, Yuan Y and
Shang A: Tumor microenvironment: Lactic acid promotes tumor
development. J Immunol Res. 2022:31193752022. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Apostolova P and Pearce EL: Lactic acid
and lactate: Revisiting the physiological roles in the tumor
microenvironment. Trends Immunol. 43:969–977. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wang JX, Choi SYC, Niu X, Kang N, Xue H,
Killam J and Wang Y: lactic acid and an acidic tumor
microenvironment suppress anticancer immunity. Int J Mol Sci.
21:83632020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Choi JW, Jung SJ, Kasala D, Hwang JK, Hu
J, Bae YH and Yun CO: pH-sensitive oncolytic adenovirus hybrid
targeting acidic tumor microenvironment and angiogenesis. J Control
Release. 205:134–143. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Audero MM, Prevarskaya N and Fiorio Pla A:
Ca(2+) Signalling and hypoxia/acidic tumour microenvironment
interplay in tumour progression. Int J Mol Sci. 23:73772022.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Chen S, Liao C, Hu H, Liao J, Chen Z, Li
S, Zeng X, Peng B, Shen S, Li D, et al: Hypoxia-driven tumor
stromal remodeling and immunosuppressive microenvironment in
scirrhous HCC. Hepatology. 79:780–797. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Nasi A, Fekete T, Krishnamurthy A, Snowden
S, Rajnavölgyi E, Catrina AI, Wheelock CE, Vivar N and Rethi B:
Dendritic cell reprogramming by endogenously produced lactic acid.
J Immunol. 191:3090–3099. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Kumar A, Pyaram K, Yarosz EL, Hong H,
Lyssiotis CA, Giri S and Chang CH: Enhanced oxidative
phosphorylation in NKT cells is essential for their survival and
function. Proc Natl Acad Sci USA. 116:7439–7448. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Xie D, Zhu S and Bai L: Lactic acid in
tumor microenvironments causes dysfunction of NKT cells by
interfering with mTOR signaling. Sci China Life Sci. 59:1290–1296.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Hirschhaeuser F, Sattler UG and
Mueller-Klieser W: Lactate: A metabolic key player in cancer.
Cancer Res. 71:6921–6925. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Yunna C, Mengru H, Lei W and Weidong C:
Macrophage M1/M2 polarization. Eur J Pharmacol. 877:1730902020.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Nicolini A and Ferrari P: Involvement of
tumor immune microenvironment metabolic reprogramming in colorectal
cancer progression, immune escape, and response to immunotherapy.
Front Immunol. 15:13537872024. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Xie Q, Zhu Z, He Y, Zhang Z, Zhang Y, Wang
Y, Luo J, Peng T, Cheng F, Gao J, et al: A lactate-induced
Snail/STAT3 pathway drives GPR81 expression in lung cancer cells.
Biochim Biophys Acta Mol Basis Dis. 1866:1655762020. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Lundø K, Trauelsen M, Pedersen SF and
Schwartz TW: Why warburg works: Lactate controls immune evasion
through GPR81. Cell Metab. 31:666–668. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Brown TP and Ganapathy V: Lactate/GPR81
signaling and proton motive force in cancer: Role in angiogenesis,
immune escape, nutrition, and Warburg phenomenon. Pharmacol Ther.
206:1074512020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Roland CL, Arumugam T, Deng D, Liu SH,
Philip B, Gomez S, Burns WR, Ramachandran V, Wang H,
Cruz-Monserrate Z and Logsdon CD: Cell surface lactate receptor
GPR81 is crucial for cancer cell survival. Cancer Res.
74:5301–5310. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Ishihara S, Hata K, Hirose K, Okui T,
Toyosawa S, Uzawa N, Nishimura R and Yoneda T: The lactate sensor
GPR81 regulates glycolysis and tumor growth of breast cancer. Sci
Rep. 12:62612022. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Brown TP, Bhattacharjee P, Ramachandran S,
Sivaprakasam S, Ristic B, Sikder MOF and Ganapathy V: The lactate
receptor GPR81 promotes breast cancer growth via a paracrine
mechanism involving antigen-presenting cells in the tumor
microenvironment. Oncogene. 39:3292–3304. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Feng J, Yang H, Zhang Y, Wei H, Zhu Z, Zhu
B, Yang M, Cao W, Wang L and Wu Z: Tumor cell-derived lactate
induces TAZ-dependent upregulation of PD-L1 through GPR81 in human
lung cancer cells. Oncogene. 36:5829–5839. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Jia Q, Xu O, Wang J, Dong J, Ren X, Jia X
and Shan C: Effects of GPR81 silencing combined with cisplatin
stimulation on biological function in hypopharyngeal squamous cell
carcinoma. Mol Med Rep. 22:1727–1736. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Chen S, Zhou X, Yang X, Li W, Li S, Hu Z,
Ling C, Shi R, Liu J, Chen G, et al: Dual Blockade of Lactate/GPR81
and PD-1/PD-L1 pathways enhances the anti-tumor effects of
metformin. Biomolecules. 11:13732021. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Niu Y, Hu S, Zhang Y, Yang J, Zhang J, He
R, Chen L, Xu L, Zhao H, Gan B, et al: Lactate-activated
GPR81/FARP1 signaling drives insulin-independent glucose uptake and
metabolic control. Cell Res. 36:137–151. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Wang Y, Jia H, Lin H, Tan X, Du Z, Chen H,
Xu Y, Han X, Zhang J, Zhao S, et al: Metastasis-associated gene,
mag-1 improves tumour microenvironmental adaptation and potentiates
tumour metastasis. J Cell Mol Med. 16:3037–3051. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Rizwan A, Serganova I, Khanin R, Karabeber
H, Ni X, Thakur S, Zakian KL, Blasberg R and Koutcher JA:
Relationships between LDH-A, lactate, and metastases in 4T1 breast
tumors. Clin Cancer Res. 19:5158–5169. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Sheng SL, Liu JJ, Dai YH, Sun XG, Xiong XP
and Huang G: Knockdown of lactate dehydrogenase A suppresses tumor
growth and metastasis of human hepatocellular carcinoma. FEBS J.
279:3898–3910. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Hou X, Ouyang J, Tang L, Wu P, Deng X, Yan
Q, Shi L, Fan S, Fan C, Guo C, et al: KCNK1 promotes proliferation
and metastasis of breast cancer cells by activating lactate
dehydrogenase A (LDHA) and up-regulating H3K18 lactylation. PLoS
Biol. 22:e30026662024. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Huang Q, Li S, Chen X, He C, Chen Y, Huang
Y, Liu Y, Wang Y and Zheng X: Association between serum lactate
dehydrogenase and lymph node metastasis in cervical cancer. Oncol
Lett. 26:4822023. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Ippolito L, Duatti A, Iozzo M, Comito G,
Pardella E, Lorito N, Bacci M, Pranzini E, Santi A, Sandrini G, et
al: Lactate supports cell-autonomous ECM production to sustain
metastatic behavior in prostate cancer. EMBO Rep. 25:3506–3531.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Pham KN, Mamun Y and Fernandez-Lima F:
Structural heterogeneity of human histone H2A.1. J Phys Chem B.
125:4977–4986. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Taylor BC and Young NL: Combinations of
histone post-translational modifications. The Biochemical journal.
478:511–532. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Zhang D, Gao J, Zhu Z, Mao Q, Xu Z, Singh
PK, Rimayi CC, Moreno-Yruela C, Xu S, Li G, et al: Lysine
L-lactylation is the dominant lactylation isomer induced by
glycolysis. Nat Chem Biol. 21:91–99. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Gaffney DO, Jennings EQ, Anderson CC,
Marentette JO, Shi T, Schou Oxvig AM, Streeter MD, Johannsen M,
Spiegel DA, Chapman E, et al: Non-enzymatic lysine lactoylation of
glycolytic enzymes. Cell Chem Biol. 27:206–213.e6. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Gao X, Pang C, Fan Z, Wang Y, Duan Y and
Zhan H: Regulation of newly identified lysine lactylation in
cancer. Cancer Lett. 587:2166802024. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Yin D, Jiang N, Cheng C, Sang X, Feng Y,
Chen R and Chen Q: Protein Lactylation and metabolic regulation of
the zoonotic parasite toxoplasma gondii. Genomics Proteomics
Bioinformatics. 21:1163–1181. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Wu Z, Chai Z, Cai X, Wang J, Wang H, Yue
B, Zhang M, Wang J, Wang H, Zhong J and Xin J: Protein lactylation
profiles provide insights into molecular mechanisms underlying
metabolism in Yak. J Agric Food Chem. Jun 8–2024.(Epub ahead of
print).
|
|
80
|
Zhu J, Guo W and Lan Y: Global analysis of
lysine lactylation of germinated seeds in wheat. Int J Mol Sci.
24:161952023. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Zhang N, Zhang Y, Xu J, Wang P, Wu B, Lu
S, Lu X, You S, Huang X, Li M, et al: α-myosin heavy chain
lactylation maintains sarcomeric structure and function and
alleviates the development of heart failure. Cell Res. 33:679–698.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Chen Y, Wu J, Zhai L, Zhang T, Yin H, Gao
H, Zhao F, Wang Z, Yang X, Jin M, et al: Metabolic regulation of
homologous recombination repair by MRE11 lactylation. Cell.
187:294–311.e21. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Shang X, Peng Y, Wang Y, Feng Z, Li M,
Peng Z and Ren W: Profile analysis of N4-acetylcytidine (ac4C) on
mRNA of human lung adenocarcinoma and paired adjacent non-tumor
tissues. Biochim Biophys Acta Gen Subj. 1867:1304982023. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Wang X, Yang Y, Ren D, Xia Y, He W, Wu Q,
Zhang J, Liu M, Du Y, Ren C, et al: JQ1, a bromodomain inhibitor,
suppresses Th17 effectors by blocking p300-mediated acetylation of
RORγt. Br J Pharmacol. 177:2959–2973. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Zong Z, Ren J, Yang B, Zhang L and Zhou F:
Emerging roles of lysine lactyltransferases and lactylation. Nat
Cell Biol. 27:563–574. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Huang H, Zhang D, Weng Y, Delaney K, Tang
Z, Yan C, Qi S, Peng C, Cole PA, Roeder RG and Zhao Y: The
regulatory enzymes and protein substrates for the lysine
β-hydroxybutyrylation pathway. Sci Adv. 7:eabe27712021. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Minami E, Sasa K, Yamada A, Kawai R,
Yoshida H, Nakano H, Maki K and Kamijo R: Lactate-induced histone
lactylation by p300 promotes osteoblast differentiation. PLoS One.
18:e02936762023. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Dong M, Zhang Y, Chen M, Tan Y, Min J, He
X, Liu F, Gu J, Jiang H, Zheng L, et al: ASF1A-dependent
P300-mediated histone H3 lysine 18 lactylation promotes
atherosclerosis by regulating EndMT. Acta Pharm Sin B.
14:3027–3048. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Wang C, Wang S, Wang Z, Han J, Jiang N, Qu
L and Xu K: Andrographolide regulates H3 histone lactylation by
interfering with p300 to alleviate aortic valve calcification. Br J
Pharmacol. 181:1843–1856. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Huang J, Wang X, Li N, Fan W, Li X, Zhou
Q, Liu J, Li W, Zhang Z, Liu X, et al: YY1 lactylation aggravates
autoimmune uveitis by enhancing microglial functions via
inflammatory genes. Adv Sci (Weinh). 11:e23080312024. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Li F, Si W, Xia L, Yin D, Wei T, Tao M,
Cui X, Yang J, Hong T and Wei R: Positive feedback regulation
between glycolysis and histone lactylation drives oncogenesis in
pancreatic ductal adenocarcinoma. Mol Cancer. 23:902024. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Yang L, Niu K, Wang J, Shen W, Jiang R,
Liu L, Song W, Wang X, Zhang X, Zhang R, et al: Nucleolin
lactylation contributes to intrahepatic cholangiocarcinoma
pathogenesis via RNA splicing regulation of MADD. J Hepatol.
81:651–666. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Ju J, Zhang H, Lin M, Yan Z, An L, Cao Z,
Geng D, Yue J, Tang Y, Tian L, et al: The alanyl-tRNA synthetase
AARS1 moonlights as a lactyltransferase to promote YAP signaling in
gastric cancer. J Clin Invest. 134:e1745872024. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Xie B, Zhang M, Li J, Cui J, Zhang P, Liu
F, Wu Y, Deng W, Ma J, Li X, et al: KAT8-catalyzed lactylation
promotes eEF1A2-mediated protein synthesis and colorectal
carcinogenesis. Proc Natl Acad Sci USA. 121:e23141281212024.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Chen B, Deng Y, Hong Y, Fan L, Zhai X, Hu
H, Yin S, Chen Q, Xie X, Ren X, et al: Metabolic recoding of
NSUN2-mediated m(5)C modification promotes the progression of
colorectal cancer via the NSUN2/YBX1/m(5)C-ENO1 positive feedback
loop. Adv Sci (Weinh). 11:e23098402024. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Wang J, Qi C, Wang R, Cao Y, Zhou Y and Ye
G: m5C RNA modification in colorectal cancer: mechanisms and
therapeutic targets. J Transl Med. 23:9482025. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Chen H, Li Y, Li H, Chen X, Fu H, Mao D,
Chen W, Lan L, Wang C, Hu K, et al: NBS1 lactylation is required
for efficient DNA repair and chemotherapy resistance. Nature.
631:663–669. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Rho H, Terry AR, Chronis C and Hay N:
Hexokinase 2-mediated gene expression via histone lactylation is
required for hepatic stellate cell activation and liver fibrosis.
Cell Metab. 35:1406–1423.e8. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
de Ruijter AJ, van Gennip AH, Caron HN,
Kemp S and van Kuilenburg AB: Histone deacetylases (HDACs):
Characterization of the classical HDAC family. Biochem J.
370:737–749. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Moreno-Yruela C, Zhang D, Wei W, Bæk M,
Liu W, Gao J, Danková D, Nielsen AL, Bolding JE, Yang L, et al:
Class I histone deacetylases (HDAC1-3) are histone lysine
delactylases. Sci Adv. 8:eabi66962022. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Fan Z, Liu Z, Zhang N, Wei W, Cheng K, Sun
H and Hao Q: Identification of SIRT3 as an eraser of H4K16la.
iScience. 26:1077572023. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Fan W, Zeng S, Wang X, Wang G, Liao D, Li
R, He S, Li W, Huang J, Li X, et al: A feedback loop driven by H3K9
lactylation and HDAC2 in endothelial cells regulates VEGF-induced
angiogenesis. Genome Biol. 25:1652024. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Zu H, Li C, Dai C, Pan Y, Ding C, Sun H,
Zhang X, Yao X, Zang J and Mo X: SIRT2 functions as a histone
delactylase and inhibits the proliferation and migration of
neuroblastoma cells. Cell Discov. 8:542022. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Suskiewicz MJ: The logic of protein
post-translational modifications (PTMs): Chemistry, mechanisms and
evolution of protein regulation through covalent attachments.
Bioessays. 46:e23001782024. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Hu X, Huang X, Yang Y, Sun Y, Zhao Y,
Zhang Z, Qiu D, Wu Y, Wu G and Lei L: Dux activates
metabolism-lactylation-MET network during early iPSC reprogramming
with Brg1 as the histone lactylation reader. Nucleic Acids Res.
52:5529–5548. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Zhang X, Mao Y, Wang B, Cui Z, Zhang Z,
Wang Z and Chen T: Screening, expression, purification and
characterization of CoA-transferases for lactoyl-CoA generation. J
Ind Microbiol Biotechnol. 46:899–909. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Varner EL, Trefely S, Bartee D, von
Krusenstiern E, Izzo L, Bekeova C, O'Connor RS, Seifert EL, Wellen
KE, Meier JL and Snyder NW: Quantification of lactoyl-CoA
(lactyl-CoA) by liquid chromatography mass spectrometry in
mammalian cells and tissues. Open Biol. 10:2001872020. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Wang Z, Xing T, Zhang L, Zhao L and Gao F:
Dynamic changes of protein lactylation and their correlations with
the glycolytic process during the postmortem acidification of
broiler breast. Poult Sci. 103:1043542024. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Dong Q, Zhang Q, Yang X, Nai S, Du X and
Chen L: Glycolysis-Stimulated Esrrb lactylation promotes the
self-renewal and extraembryonic endoderm stem cell differentiation
of embryonic stem cells. Int J Mol Sci. 25:26922024. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Zhang M, Zhang Y, Ding Y, Huang J, Yao J,
Xie Z, Lv Y and Zuo J: Regulating the expression of HIF-1alpha or
lncRNA: Potential directions for cancer therapy. Cells.
11:28112022. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Yan P, Yang K, Xu M, Zhu M, Duan Y, Li W,
Liu L, Liang C, Li Z, Pan X, et al: CCT6A alleviates pulmonary
fibrosis by inhibiting HIF-1α-mediated lactate production. J Mol
Cell Biol. 16:mjae0212024. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Ilegems E, Bryzgalova G, Correia J,
Yesildag B, Berra E, Ruas JL, Pereira TS and Berggren PO: HIF-1α
inhibitor PX-478 preserves pancreatic β cell function in diabetes.
Sci Transl Med. 14:eaba91122022. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Yang Z, Su W, Wei X, Pan Y, Xing M, Niu L,
Feng B, Kong W, Ren X, Huang F, et al: Hypoxia inducible factor-1α
drives cancer resistance to cuproptosis. Cancer Cell.
43:937–954.e9. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Mao Y, Zhang J, Zhou Q, He X, Zheng Z, Wei
Y, Zhou K, Lin Y, Yu H, Zhang H, et al: Hypoxia induces
mitochondrial protein lactylation to limit oxidative
phosphorylation. Cell Res. 34:13–30. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Zhou X, Wu D, Mi T, Li R, Guo T and Li W:
Icaritin activates p53 and inhibits aerobic glycolysis in liver
cancer cells. Chem Biol Interact. 392:1109262024. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Zhou X, Wu D, Zhu L, Li R, Yu H and Li W:
Withaferin A inhibits liver cancer tumorigenesis by suppressing
aerobic glycolysis through the p53/IDH1/HIF-1α signaling axis. Curr
Cancer Drug Targets. 24:534–545. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Sobrido-Cameán D, Coulson B, Miller M,
Oswald MCW, Pettini T, Bailey DMD, Baines RA and Landgraf M:
Mitochondrial ROS and HIF-1α signaling mediate synaptic plasticity
in the critical period. PLoS Biol. 23:e30033382025. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Jiang Z, Xiong N, Yan R, Li ST, Liu H, Mao
Q, Sun Y, Shen S, Ye L, Gao P, et al: PDHX acetylation facilitates
tumor progression by disrupting PDC assembly and activating
lactylation mediated gene expression. Protein Cell. 16:49–63. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Feng F, Wu J, Chi Q, Wang S, Liu W, Yang
L, Song G, Pan L, Xu K and Wang C: Lactylome analysis unveils
lactylation-dependent mechanisms of stemness remodeling in the
liver cancer stem cells. Adv Sci (Weinh). 11:e24059752024.
View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Wang J, Yang P, Yu T, Gao M, Liu D, Zhang
J, Lu C, Chen X, Zhang X and Liu Y: Lactylation of PKM2 suppresses
inflammatory metabolic adaptation in pro-inflammatory macrophages.
Int J Biol Sci. 18:6210–6225. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Li G, Wang D, Zhai Y, Pan C, Zhang J, Wang
C, Huang R, Yu M, Li Y, Liu X, et al: Glycometabolic
reprogramming-induced XRCC1 lactylation confers therapeutic
resistance in ALDH1A3-overexpressing glioblastoma. Cell Metab.
36:1696–1710.e10. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Gu J, Zhou J, Chen Q, Xu X, Gao J, Li X,
Shao Q, Zhou B, Zhou H, Wei S, et al: Tumor metabolite lactate
promotes tumorigenesis by modulating MOESIN lactylation and
enhancing TGF-beta signaling in regulatory T cells. Cell Rep.
39:1109862022. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Zhang X, Song W, Gao Y, Zhang Y, Zhao Y,
Hao S and Ni T: The role of tumor metabolic reprogramming in tumor
immunity. Int J Mol Sci. 24:174222023. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Li Y, Cao Q, Hu Y, He B, Cao T, Tang Y,
Zhou XP, Lan XP and Liu SQ: Advances in the interaction of
glycolytic reprogramming with lactylation. Biomed Pharmacother.
177:1169822024. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Zhou J, Ma X, Liu X, Liu Y, Fu J, Qi Y and
Liu H: The impact of histone lactylation on the tumor
microenvironment and metabolic pathways and its potential in cancer
therapy. Genes Genomics. 46:991–1011. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Masin M, Vazquez J, Rossi S, Groeneveld S,
Samson N, Schwalie PC, Deplancke B, Frawley LE, Gouttenoire J,
Moradpour D, et al: GLUT3 is induced during epithelial-mesenchymal
transition and promotes tumor cell proliferation in non-small cell
lung cancer. Cancer Metab. 2:112014. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Yang H, Yang S, He J, Li W, Zhang A, Li N,
Zhou G and Sun B: Glucose transporter 3 (GLUT3) promotes
lactylation modifications by regulating lactate dehydrogenase A
(LDHA) in gastric cancer. Cancer Cell Int. 23:3032023. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Li Q, Lin G, Zhang K, Liu X, Li Z, Bing X,
Nie Z, Jin S, Guo J and Min X: Hypoxia exposure induces lactylation
of Axin1 protein to promote glycolysis of esophageal carcinoma
cells. Biochem Pharmacol. 226:1164152024. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Qiao Z, Li Y, Li S, Liu S and Cheng Y:
Hypoxia-induced SHMT2 protein lactylation facilitates glycolysis
and stemness of esophageal cancer cells. Mol Cell Biochem.
479:3063–3076. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Jiang J, Huang D, Jiang Y, Hou J, Tian M,
Li J, Sun L, Zhang Y, Zhang T, Li Z, et al: Lactate modulates
cellular metabolism through histone lactylation-mediated gene
expression in non-small cell lung cancer. Front Oncol.
11:6475592021. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Yan F, Teng Y, Li X, Zhong Y, Li C, Yan F
and He X: Hypoxia promotes non-small cell lung cancer cell
stemness, migration, and invasion via promoting glycolysis by
lactylation of SOX9. Cancer Biol Ther. 25:23041612024. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Zhang R, Li L and Yu J: Lactate-induced
IGF1R protein lactylation promotes proliferation and metabolic
reprogramming of lung cancer cells. Open Life Sci. 19:202208742024.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Yang J and Yu X, Xiao M, Xu H, Tan Z, Lei
Y, Guo Y, Wang W, Xu J, Shi S and Yu X: Histone lactylation-driven
feedback loop modulates cholesterol-linked immunosuppression in
pancreatic cancer. Gut. 74:1859–1872. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Meng Q, Sun H, Zhang Y, Yang X, Hao S, Liu
B, Zhou H, Xu ZX and Wang Y: Lactylation stabilizes DCBLD1
activating the pentose phosphate pathway to promote cervical cancer
progression. J Exp Clin Cancer Res. 43:362024. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Qu J, Li P and Sun Z: Histone lactylation
regulates cancer progression by reshaping the tumor
microenvironment. Front Immunol. 14:12843442023. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Pereira MVA, Galvani RG, Gonçalves-Silva
T, de Vasconcelo ZFM and Bonomo A: Tissue adaptation of CD4 T
lymphocytes in homeostasis and cancer. Front Immunol.
15:13793762024. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Khan U and Ghazanfar H: T lymphocytes and
autoimmunity. Int Rev Cell Mol Biol. 341:125–168. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Wu Y, Yuan M, Wang C, Chen Y, Zhang Y and
Zhang J: T lymphocyte cell: A pivotal player in lung cancer. Front
Immunol. 14:11027782023. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Tu VY, Ayari A and O'Connor RS: Beyond the
Lactate Paradox: How Lactate and Acidity Impact T Cell Therapies
against Cancer. Antibodies (Basel, Switzerland). 10:2021.PubMed/NCBI
|
|
140
|
Fan W, Wang X, Zeng S, Li N, Wang G, Li R,
He S, Li W, Huang J, Li X, et al: Global lactylome reveals
lactylation-dependent mechanisms underlying T(H)17 differentiation
in experimental autoimmune uveitis. Sci Adv. 9:eadh46552023.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Brescia C, Audia S, Pugliano A, Scaglione
F, Iuliano R, Trapasso F, Perrotti N, Chiarella E and Amato R:
Metabolic drives affecting Th17/Treg gene expression changes and
differentiation: Impact on immune-microenvironment regulation.
APMIS. 132:1026–1045. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Wang R, Li C, Cheng Z, Li M, Shi J, Zhang
Z, Jin S and Ma H: H3K9 lactylation in malignant cells facilitates
CD8(+) T cell dysfunction and poor immunotherapy response. Cell
Rep. 43:1146862024. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Quinn WJ III, Jiao J, TeSlaa T, Stadanlick
J, Wang Z, Wang L, Akimova T, Angelin A, Schäfer PM, Cully MD, et
al: Lactate limits T cell proliferation via the NAD(H) Redox State.
Cell Rep. 33:1085002020. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Rostamian H, Khakpoor-Koosheh M,
Jafarzadeh L, Masoumi E, Fallah-Mehrjardi K, Tavassolifar MJ, M
Pawelek J, Mirzaei HR and Hadjati J: Restricting tumor lactic acid
metabolism using dichloroacetate improves T cell functions. BMC
Cancer. 22:392022. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Liu Y, Wang F, Peng D, Zhang D, Liu L, Wei
J, Yuan J, Zhao L, Jiang H, Zhang T, et al: Activation and
antitumor immunity of CD8(+) T cells are supported by the glucose
transporter GLUT10 and disrupted by lactic acid. Sci Transl Med.
16:eadk73992024. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Kumagai S, Koyama S, Itahashi K,
Tanegashima T, Lin YT, Togashi Y, Kamada T, Irie T, Okumura G, Kono
H, et al: Lactic acid promotes PD-1 expression in regulatory T
cells in highly glycolytic tumor microenvironments. Cancer Cell.
40:201–218.e9. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Orecchioni M, Ghosheh Y, Pramod AB and Ley
K: Macrophage Polarization: Different gene signatures in M1(LPS+)
vs. Classically and M2(LPS-) vs. alternatively activated
macrophages. Front Immunol. 10:10842019. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
da Rocha RF, De Bastiani MA and Klamt F:
Bioinformatics approach to evaluate differential gene expression of
M1/M2 macrophage phenotypes and antioxidant genes in
atherosclerosis. Cell Biochem Biophys. 70:831–839. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Wang Y, Smith W, Hao D, He B and Kong L:
M1 and M2 macrophage polarization and potentially therapeutic
naturally occurring compounds. Int Immunopharmacol. 70:459–466.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Mauro A, Russo V, Di Marcantonio L,
Berardinelli P, Martelli A, Muttini A, Mattioli M and Barboni B: M1
and M2 macrophage recruitment during tendon regeneration induced by
amniotic epithelial cell allotransplantation in ovine. Res Vet Sci.
105:92–102. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Di Conza G and Ho PC: Metabolic adaptation
of macrophages in chronic diseases. Cancer Lett. 414:250–256. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Ammarah U, Pereira-Nunes A, Delfini M and
Mazzone M: From monocyte-derived macrophages to resident
macrophages-how metabolism leads their way in cancer. Mol Oncol.
18:1739–1758. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Xu B, Liu Y, Li N and Geng Q: Lactate and
lactylation in macrophage metabolic reprogramming: current progress
and outstanding issues. Front Immunol. 15:13957862024. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Li XM, Yang Y, Jiang FQ, Hu G, Wan S, Yan
WY, He XS, Xiao F, Yang XM, Guo X, et al: Histone lactylation
inhibits RARγ expression in macrophages to promote colorectal
tumorigenesis through activation of TRAF6-IL-6-STAT3 signaling.
Cell Rep. 43:1136882024. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Chao J, Chen GD, Huang ST, Gu H, Liu YY,
Luo Y, Lin Z, Chen ZZ, Li X, Zhang B, et al: High histone H3K18
lactylation level is correlated with poor prognosis in epithelial
ovarian cancer. Neoplasma. 71:319–332. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Yu L, Jing C, Zhuang S, Ji L and Jiang L:
A novel lactylation-related gene signature for effectively
distinguishing and predicting the prognosis of ovarian cancer.
Transl Cancer Res. 13:2497–2508. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
De Leo A, Ugolini A, Yu X, Scirocchi F,
Scocozza D, Peixoto B, Pace A, D'Angelo L, Liu JKC, Etame AB, et
al: Glucose-driven histone lactylation promotes the
immunosuppressive activity of monocyte-derived macrophages in
glioblastoma. Immunity. 57:1105–1123.e8. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Xiong J, He J, Zhu J, Pan J, Liao W, Ye H,
Wang H, Song Y, Du Y, Cui B, et al: Lactylation-driven
METTL3-mediated RNA m(6)A modification promotes immunosuppression
of tumor-infiltrating myeloid cells. Mol Cell. 82:1660–1677.e10.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Kumar S, Sahu N, Jawaid T, Jayasingh
Chellammal HS and Upadhyay P: Dual role of lactate in human health
and disease. Front Physiol. 16:16213582025. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Plebanek MP, Xue Y, Nguyen YV, DeVito NC,
Wang X, Holtzhausen A, Beasley GM, Theivanthiran B and Hanks BA: A
lactate-SREBP2 signaling axis drives tolerogenic dendritic cell
maturation and promotes cancer progression. Sci Immunol.
9:eadi41912024. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
LeBleu VS and Kalluri R: A peek into
cancer-associated fibroblasts: Origins, functions and translational
impact. Dis Model Mech. 11:dmm0294472018. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Kong W, He J, Zhou Q, Zhou X, Wei X, Yang
Y, Mei Y, Wang S, Zhang X, Yao B, et al: Histone
lactylation-related genes correlate with the molecular patterns and
functions of cancer-associated fibroblasts and have significant
clinical implications in clear cell renal cell carcinoma. Heliyon.
10:e335542024. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Li Z, Liang P, Chen Z, Chen Z, Jin T, He
F, Chen X and Yang K: CAF-secreted LOX promotes PD-L1 expression
via histone Lactylation and regulates tumor EMT through TGFβ/IGF1
signaling in gastric cancer. Cell Signal. 124:1114622024.
View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Lv M, Huang Y, Chen Y and Ding K:
Lactylation modification in cancer: mechanisms, functions, and
therapeutic strategies. Exp Hematol Oncol. 14:322025. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Wei Y, Guo H, Chen S and Tang XX:
Regulation of macrophage activation by lactylation in lung disease.
Front Immunol. 15:14277392024. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Zheng P, Mao Z, Luo M, Zhou L, Wang L, Liu
H, Liu W and Wei S: Comprehensive bioinformatics analysis of the
solute carrier family and preliminary exploration of SLC25A29 in
lung adenocarcinoma. Cancer Cell Int. 23:2222023. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Crispo F, Condelli V, Lepore S,
Notarangelo T, Sgambato A, Esposito F, Maddalena F and Landriscina
M: Metabolic dysregulations and epigenetics: A bidirectional
interplay that drives tumor Progression. Cells. 8:7982019.
View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Zhang X, Liang C, Wu C, Wan S and Xu L,
Wang S, Wang J, Huang X and Xu L: A rising star involved in tumour
immunity: Lactylation. J Cell Mol Med. 28:e701462024. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Chen L, Huang L, Gu Y, Cang W, Sun P and
Xiang Y: Lactate-Lactylation Hands between Metabolic Reprogramming
and Immunosuppression. Int J Mol Sci. 23:119432022. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Fu Y and Chung FL: Oxidative stress and
hepatocarcinogenesis. Hepatoma Res. 4:392018. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Berumen J, Baglieri J, Kisseleva T and
Mekeel K: Liver fibrosis: Pathophysiology and clinical
implications. WIREs Mech Dis. 13:e14992021. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Šmíd V: Liver fibrosis. Vnitr Lek.
66:61–66. 2020.(In Czech). View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Li LN, Li WW, Xiao LS and Lai WN:
Lactylation signature identifies liver fibrosis phenotypes and
traces fibrotic progression to hepatocellular carcinoma. Front
Immunol. 15:14333932024. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Deng J and Liao X: Lysine lactylation
(Kla) might be a novel therapeutic target for breast cancer. BMC
Med Genomics. 16:2832023. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Cui Z, Li Y, Lin Y, Zheng C, Luo L, Hu D,
Chen Y, Xiao Z and Sun Y: Lactylproteome analysis indicates histone
H4K12 lactylation as a novel biomarker in triple-negative breast
cancer. Front Endocrinol (Lausanne). 15:13286792024. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Li J, Chen Z, Jin M, Gu X, Wang Y, Huang
G, Zhao W and Lu C: Histone H4K12 lactylation promotes malignancy
progression in triple-negative breast cancer through SLFN5
downregulation. Cell Signal. 124:1114682024. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Sun J, Feng Q, He Y, Wang M and Wu Y:
Lactate activates CCL18 expression via H3K18 lactylation in
macrophages to promote tumorigenesis of ovarian cancer. Acta
Biochim Biophys Sin (Shanghai). 56:1373–1386. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Wang X, Li Y, Li J, Li L, Zhu H, Chen H,
Kong R, Wang G, Wang Y, Hu J and Sun B: Cell-in-cell phenomenon and
its relationship with tumor microenvironment and tumor progression:
A review. Front Cell Dev Biol. 7:3112019. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Zheng A, Zhang L, Yang J, Yin X, Zhang T,
Wu X and Ma X: Physical activity prevents tumor metastasis through
modulation of immune function. Front Pharmacol. 13:10341292022.
View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Wang H, Xu M, Zhang T, Pan J, Li C, Pan B,
Zhou L, Huang Y, Gao C, He M, et al: PYCR1 promotes liver cancer
cell growth and metastasis by regulating IRS1 expression through
lactylation modification. Clin Transl Med. 14:e700452024.
View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Pandkar MR, Sinha S, Samaiya A and Shukla
S: Oncometabolite lactate enhances breast cancer progression by
orchestrating histone lactylation-dependent c-Myc expression.
Transl Oncol. 37:1017582023. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Zhang C, Zhou L, Zhang M, Du Y, Li C, Ren
H and Zheng L: H3K18 lactylation potentiates immune escape of
non-small cell lung cancer. Cancer Res. 84:3589–3601. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Li W, Zhou C, Yu L, Hou Z, Liu H, Kong L,
Xu Y, He J, Lan J, Ou Q, et al: Tumor-derived lactate promotes
resistance to bevacizumab treatment by facilitating autophagy
enhancer protein RUBCNL expression through histone H3 lysine 18
lactylation (H3K18la) in colorectal cancer. Autophagy. 20:114–130.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Sun X, He L, Liu H, Thorne RF, Zeng T, Liu
L, Zhang B, He M, Huang Y, Li M, et al: The diapause-like
colorectal cancer cells induced by SMC4 attenuation are
characterized by low proliferation and chemotherapy insensitivity.
Cell Metab. 35:1563–1579.e8. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Huang H, Chen K, Zhu Y, Hu Z, Wang Y, Chen
J, Li Y, Li D and Wei P: A multi-dimensional approach to unravel
the intricacies of lactylation related signature for prognostic and
therapeutic insight in colorectal cancer. J Transl Med. 22:2112024.
View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Chu YD, Cheng LC, Lim SN, Lai MW, Yeh CT
and Lin WR: Aldolase B-driven lactagenesis and CEACAM6 activation
promote cell renewal and chemoresistance in colorectal cancer
through the Warburg effect. Cell Death Dis. 14:6602023. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Gu J, Xu X, Li X, Yue L, Zhu X, Chen Q,
Gao J, Takashi M, Zhao W, Zhao B, et al: Tumor-resident microbiota
contributes to colorectal cancer liver metastasis by lactylation
and immune modulation. Oncogene. 43:2389–2404. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Liu J, Chen P, Zhou J, Li H and Pan Z:
Prognostic impact of lactylation-associated gene modifications in
clear cell renal cell carcinoma: Insights into molecular landscape
and therapeutic opportunities. Environ Toxicol. 39:1360–1373. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Yang J, Luo L, Zhao C, Li X, Wang Z, Zeng
Z, Yang X, Zheng X, Jie H, Kang L, et al: A positive feedback loop
between Inactive VHL-Triggered Histone Lactylation and PDGFRβ
signaling drives clear cell renal cell carcinoma progression. Int J
Biol Sci. 18:3470–3483. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Gu X, Zhuang A, Yu J, Yang L, Ge S, Ruan
J, Jia R, Fan X and Chai P: Histone lactylation-boosted ALKBH3
potentiates tumor progression and diminished promyelocytic leukemia
protein nuclear condensates by m1A demethylation of SP100A. Nucleic
Acids Res. 52:2273–2289. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Zhu M, Xiao Q, Cai X, Chen Z, Shi Q, Sun
X, Xie X and Sun M: Predicting lymphoma prognosis using machine
learning-based genes associated with lactylation. Transl Oncol.
49:1021022024. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Sun C, Zhang W, Liu H, Ding Y, Guo J,
Xiong S, Zhai Z and Hu W: Identification of a novel
lactylation-related gene signature predicts the prognosis of
multiple myeloma and experiment verification. Sci Rep.
14:151422024. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Wang X, Shi Y, Shi H, Liu X, Liao A, Liu
Z, Orlowski RZ, Zhang R and Wang H: MUC20 regulated by
extrachromosomal circular DNA attenuates proteasome inhibitor
resistance of multiple myeloma by modulating cuproptosis. J Exp
Clin Cancer Res. 43:682024. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Byun JK: Tumor lactic acid: A potential
target for cancer therapy. Arch Pharm Res. 46:90–110. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Du M, Yu T, Zhan Q, Li H, Zou Y, Geng M,
Meng T and Xie Z: Development of a novel lactate dehydrogenase A
inhibitor with potent antitumor activity and immune activation.
Cancer Sci. 113:2974–2985. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Xian ZY, Liu JM, Chen QK, Chen HZ, Ye CJ,
Xue J, Yang HQ, Li JL, Liu XF and Kuang SJ: Inhibition of LDHA
suppresses tumor progression in prostate cancer. Tumour Biol.
36:8093–8100. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Zhang P, Wan Y, Ma J, Gong J, Zhong Z, Cui
Y, Zhang H, Da Y, Ma J, Li C, et al: Epigenetic silencing of LDHB
promotes hepatocellular carcinoma by remodeling the tumor
microenvironment. Cancer Immunol Immunother. 73:1272024. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Urbańska K and Orzechowski A:
Unappreciated Role of LDHA and LDHB to control apoptosis and
autophagy in tumor cells. Int J Mol Sci. 20:20852019. View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Shibata S, Sogabe S, Miwa M, Fujimoto T,
Takakura N, Naotsuka A, Kitamura S, Kawamoto T and Soga T:
Identification of the first highly selective inhibitor of human
lactate dehydrogenase B. Sci Rep. 11:213532021. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Curtis NJ, Mooney L, Hopcroft L,
Michopoulos F, Whalley N, Zhong H, Murray C, Logie A, Revill M,
Byth KF, et al: Pre-clinical pharmacology of AZD3965, a selective
inhibitor of MCT1: DLBCL, NHL and Burkitt's lymphoma anti-tumor
activity. Oncotarget. 8:69219–69236. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Halford S, Veal GJ, Wedge SR, Payne GS,
Bacon CM, Sloan P, Dragoni I, Heinzmann K, Potter S, Salisbury BM,
et al: A phase I Dose-escalation Study of AZD3965, an oral
monocarboxylate transporter 1 inhibitor, in patients with advanced
cancer. Clin Cancer Res. 29:1429–1439. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Goldberg FW, Kettle JG, Lamont GM, Buttar
D, Ting AKT, McGuire TM, Cook CR, Beattie D, Morentin Gutierrez P,
Kavanagh SL, et al: Discovery of clinical candidate AZD0095, a
selective inhibitor of monocarboxylate transporter 4 (MCT4) for
oncology. J Med Chem. 66:384–397. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Bisetto S, Whitaker-Menezes D, Wilski NA,
Tuluc M, Curry J, Zhan T, Snyder CM, Martinez-Outschoorn UE and
Philp NJ: Monocarboxylate transporter 4 (MCT4) knockout mice have
attenuated 4NQO induced carcinogenesis; A Role for MCT4 in driving
oral squamous cell cancer. Front Oncol. 8:3242018. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Fang Y, Liu W, Tang Z, Ji X, Zhou Y, Song
S, Tian M, Tao C, Huang R, Zhu G, et al: Monocarboxylate
transporter 4 inhibition potentiates hepatocellular carcinoma
immunotherapy through enhancing T cell infiltration and immune
attack. Hepatology. 77:109–123. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Damia G and Garattini S: The
pharmacological point of view of resistance to therapy in tumors.
Cancer Treat Rev. 40:909–916. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Yue Q, Wang Z, Shen Y, Lan Y, Zhong X, Luo
X, Yang T, Zhang M, Zuo B, Zeng T, et al: Histone H3K9 lactylation
confers temozolomide resistance in glioblastoma via LUC7L2-Mediated
MLH1 intron retention. Adv Sci (Weinh). 11:e23092902024. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Li F, Zhang H, Huang Y, Li D, Zheng Z, Xie
K, Cao C, Wang Q, Zhao X, Huang Z, et al: Single-cell transcriptome
analysis reveals the association between histone lactylation and
cisplatin resistance in bladder cancer. Drug Resist Updat.
73:1010592024. View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Chen HJ, Yu MM, Huang JC, Lan FY, Liao HH,
Xu ZH, Yu YJ, Huang YC and Chen F: SLC4A4 is a novel driver of
enzalutamide resistance in prostate cancer. Cancer Lett.
597:2170702024. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Wang Y, Chen J, Wang Y, Zhao J, Zhang J
and Wang H: Pharmacologically targeting protein lactylation to
overcome cancer drug resistance. Eur J Med Chem. 296:1179052025.
View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Lu Y, Zhu J, Zhang Y, Li W, Xiong Y, Fan
Y, Wu Y, Zhao J, Shang C, Liang H and Zhang W: Lactylation-driven
IGF2BP3-mediated serine metabolism reprogramming and RNA
m6A-modification promotes lenvatinib resistance in HCC. Adv Sci
(Weinh). 11:e24013992024. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Fang C, Peng Q, Li X, Qu X, Qiao Z, Yin B,
Xiao L, Chen Y, Dong X, Cai L, et al: Multichannel immune
nanoregulators suppress lactic acid metabolism and lactic
acid-shaped acidic microenvironment to uproot anti-tumor
immunosuppression. Adv Mater. 38:e122302026. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Wan J, Shi JH, Shi M, Huang H, Zhang Z, Li
W, Guo C, Bao R, Yu X, Han Q, et al: Lactate dehydrogenase B
facilitates disulfidptosis and exhaustion of tumour-infiltrating
CD8(+) T cells. Nat Cell Biol. 27:972–982. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Xu K, Shou Y, Liu R, Xiong H, Dai X, Bao
X, Chen X, Huang L, Sheng H, Zhang H and Lu Y: Lactylation in
cancer: Advances and opportunities for treatment resistance. Clin
Transl Med. 15:e704782025. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Chang D, Li D, Sun Y, Shi J, Zhang S and
Wang C: Targeting lactylation for cancer: Mechanisms, effects, and
therapeutic prospects. Int J Mol Sci. 26:112782025. View Article : Google Scholar : PubMed/NCBI
|