Introduction
Skeletal muscle atrophy can result from various
diseases such as sepsis, cachexia, diabetes and chronic obstructive
pulmonary disease, and is referred to as pathological atrophy when
it occurs in these contexts (1).
Increased levels of endotoxins in the bloodstream are a prevalent
cause of pathological atrophy, with skeletal muscle inflammation
being a key known contributing component (2,3).
Lipopolysaccharide (LPS) is a representative stimulus that elicits
a potent inflammatory response, leading to skeletal muscle
inflammation during sepsis progression (4). Hence, clarifying the processes that
govern LPS-induced inflammation in muscle is imperative.
LPS-induced sepsis is associated with muscle
atrophy. Sepsis is a life-threatening condition and in severe
cases, when the immune response is not properly regulated, an
excessive immune response occurs, causing organ dysfunction and
failure (5). The entrance of LPS
into cells activates the immune system, releasing large amounts of
inflammatory cytokines (such as TNF-α, IL-1β and IL-6). These
cytokines activate muscle protein degradation pathways
(ubiquitin-proteasome system) and inhibit protein synthesis,
causing muscle wasting (6). The
association between sepsis and muscle atrophy has clinical
significance. Therefore, understanding the mechanisms of
LPS-induced sepsis and muscle atrophy is important to maintain
muscle health and develop therapeutic strategies.
Pyroptosis, a mechanism regulating cell death as an
inflammatory response, is key to the development and course of
sepsis (7). The activation of the
NLR family pyrin domain containing 3 (NLRP3) inflammasome initiates
pyroptosis. The NLRP3 inflammasome is formed when NLRP3 receives
external stimuli and activates caspase-1, which causes the
pore-forming protein gasdermin-D (GSDMD) to become cleaved. The
cleaved GSDMD creates pores in the cell membrane that release an
overabundance of proinflammatory cytokines (such as IL-1β and
IL-18), causing cell death (8,9). LPS
influx is commonly recognized as triggering pyroptosis, which
serves a notable role in muscle proteolysis (10). However, to the best of our
knowledge, the manner in which pyroptosis mechanisms act in the
healing process following inflammation-induced muscle atrophy is
unknown.
Veronicastrum sibiricum (L.) Pennell is a
perennial herb species that grows in northern Korea, China, Japan,
Manchuria and Siberia (11).
Furthermore, Veronicastrum sibiricum (L.) Pennell is a
medicinal herb that has been previously used to treat arthritis,
diarrhea and rheumatism (12).
Veronicastrum sibiricum (L.) Pennell has been suggested to
exhibit antioxidant activity by scavenging reactive oxygen species
(ROS) and anti-inflammatory activity by inhibiting nitric oxide
production; however, the precise mechanism of action through which
Veronicastrum sibiricum (L.) Pennell functions remains
unclear (13). Additionally, to
the best of our knowledge, no prior research has been carried out
on the impact of Veronicastrum sibiricum on muscle. There is
also a dearth of research on the mechanism through which
Veronicastrum sibiricum Pennell extract counteracts
LPS-induced muscle atrophy to increase muscular strength.
Therefore, the present study employed an inflammatory muscle
atrophy model to investigate the mechanism of action of
Veronicastrum sibiricum seed extract (VSE) and its impact on
the pyroptosis pathway.
Materials and methods
Preparation of VSE
The VSE (cat. no. KPM012-022) used in the present
study was obtained from the Natural Product Central Bank at the
Korea Research Institute of Bioscience and Biotechnology. The plant
was collected in 2001 from Baegam-myeon, Cheoin, Yongin Gyeonggi,
Korea. To create the extract, 74 g dried and powdered seeds were
added to 1 liter of 99.9% methyl alcohol (high-performance liquid
chromatography grade) and extracted at room temperature through 30
cycles of ultrasonication (40 KHz; 1,500 W) for 15 min using an
ultrasonic extractor (SDN-900H; Sungdong Ultrasonic Co., Ltd.),
then left to stand for 120 min. The extract was filtered
(Qualitative Filter No. 100; HYUNDAI MICRO Co., Ltd.) and dried
under reduced pressure. A total of 6.53 g VSE was obtained.
Cell culture and differentiation
Murine C2C12 myoblast cells were obtained from
American Type Culture Collection. The cells were grown at 37°C with
5% CO2 in DMEM (Gibco; Thermo Fisher Scientific, Inc.)
with fetal bovine serum (Gibco; Thermo Fisher Scientific, Inc.) at
10% (v/v) and penicillin-streptomycin (Gibco; Thermo Fisher
Scientific, Inc.) at 1% (v/v). To induce myoblast differentiation
into C2C12 myotubes, once cells reached 90% confluency, the medium
was switched to DMEM containing horse serum (Gibco; Thermo Fisher
Scientific, Inc.) at 2% (v/v). The differentiation media were
changed every 2 days and the cells were cultivated for 4 days.
Giemsa staining
C2C12 myoblasts were seeded at a concentration of
1×106 cells/ml in 6-well plates and differentiated into
myotubes over 4 days. Subsequently, the existing medium was removed
by aspiration, and the myotubes were rinsed twice with PBS and
fixed in 4% paraformaldehyde for 10 min at room temperature. Giemsa
solution, diluted 1:10 in distilled water and filtered through a
0.2-µm filter, was applied to fixed cells and left for 40 min at
room temperature. After removing the Giemsa solution by aspiration,
cells were rinsed three times with PBS, and then imaged under a
bright-field microscope (Leica Microsystems GmbH) and analyzed with
ImageJ (National Institutes of Health).
Cell viability assay
Cell viability was measured using a Cell Counting
Kit-8 (CCK-8; Dojindo Laboratories, Inc.) according to the
manufacturer's instructions. After differentiating C2C12 myoblasts
into myotubes for 4 days in 96-well plates, the cells were treated
with VSE at concentrations of 0 (control), 1.25, 2.5, 5 and 10
µg/ml for 48 h at 37°C. Given the lack of previous studies on VSE
in muscle models, cytotoxicity testing was conducted to determine
appropriate concentrations for subsequent experiments. After
treatment, 10 µl CCK-8 solution was added to each well, and the
cells were incubated for an additional 2 h. Absorbance was measured
at 450 nm using a SPECTROstar microplate reader (BMG Labtech
GmbH).
Reverse transcription-quantitative PCR
(RT-qPCR) analysis
Total RNA was obtained from cells using RNAiso Plus
Reagent (Takara Bio, Inc.). After measuring the mRNA yield, cDNA
synthesis was carried out using ReverTra Ace™ qPCR RT Master Mix
(cat. no. FSQ-201; Toyobo Co., Ltd.) according to the
manufacturer's instructions. The reaction conditions were as
follows: 37°C for 5 min for genomic DNA removal, followed by 37°C
for 15 min for reverse transcription and 98°C for 5 min for enzyme
inactivation. RT-qPCR was performed using a Cycler iQ™ Real-Time
PCR Detection System (Bio-Rad Laboratories, Inc.) and SYBR Green
master mix (Toyobo Co., Ltd.) with the appropriate primers
(Table I) and the template cDNA.
The thermocycling conditions were as follows: Initial denaturation
at 95°C for 1 min, followed by 40 cycles of 95°C for 15 sec and
60°C for 60 sec. A melt curve analysis was conducted to verify
amplification specificity. Analysis of relative gene expression
data using RT-qPCR and the 2−ΔΔCq method (14).
 | Table I.Primer sequences for reverse
transcription-quantitative PCR. |
Table I.
Primer sequences for reverse
transcription-quantitative PCR.
|
| Primer sequence
(5′-3′) |
|---|
|
|
|
|---|
| Gene | Forward | Reverse |
|---|
| MuRF1 |
TACCAAGCCTGTGGTCATCCTG |
ACGGAAACGACCTCCAGACATG |
|
Atrogin-1 |
AAGGCTGTTGGAGCTGATAGCA |
CACCCACATGTTAATGTTGCCC |
| TNF-α |
TGGAACTGGCAGAAGAGGCACT |
AGAGGCTCAGACATAGGCACCG |
| IL-1β |
TGTGAAATGCCACCTTTTGA |
GGTCAAAGGTTTGGAAGCAG |
| NLRP3 |
TGGACCTCTGCCGAAACTGA |
CTTGAGGTGACACGTGAGGA |
| β-actin |
CGTGCGTGACATCAAAGAGAA |
GCTCGTTGCCAATAGTGATGA |
ELISA
Differentiated myotubes were pretreated with VSE at
concentrations of 0 (control), 1.25, 2.5 and 5 µg/ml for 3 h at
room temperature and then co-treated with LPS (500 ng/ml) for 24 h
at 37°C. Supernatants were acquired, and secretion of the
proinflammatory cytokines TNF-α (cat. no. MTA00B; R&D systems,
Inc.) and IL-1β was analyzed using ELISA kits (cat. no. DY401;
R&D Systems, Inc.) according to the manufacturer's
guidelines.
Protein extraction and western blot
analysis
To acquire lysates from myotubes in vitro,
cells were treated with 1% protease and 1% phosphatase inhibitors
in RIPA buffer (Biosesang) at room temperature for 30 min. To
acquire lysates in vivo, gastrocnemius (GAS) samples were
lysed using PRO-PREPTM (Intron Biotechnology, Inc.) at
room temperature for 30 min. After Bradford assay quantification of
protein concentrations, 30 µg of protein were separated by SDS-PAGE
using 10% polyacrylamide gels. Subsequently, the proteins were
transferred to a 0.2-µm nitrocellulose membrane and the membrane
was blocked in 5% skimmed milk in Tris-buffered saline containing
0.1% Tween 20 (TBST) for 1 h at room temperature. Each membrane was
incubated with one of 18 primary antibodies (Table II) on a shaker overnight at 4°C to
facilitate primary antibody binding. The membrane was then rinsed
with TBST three times, and the membrane was incubated with
secondary antibody (rabbit and mouse) at room temperature for 1 h.
After rinsing three times with TBST, protein bands were detected
using an enhanced chemiluminescence detection kit (Cytiva). For
semi-quantitative protein expression analysis, membrane images were
processed using ImageJ version 1.54d (National Institutes of
Health).
 | Table II.Primary antibodies used for western
blotting. |
Table II.
Primary antibodies used for western
blotting.
| Antibody | Origin | Cat. no. | Manufacturer | Dilution |
|---|
| Secondary
antibody | Mouse
monoclonal | 7076 | Cell Signaling
Technology, Inc. | 1:2,000 |
| Secondary
antibody | Rabbit
monoclonal | 7074 | Cell Signaling
Technology, Inc. | 1:3,000 |
| MyHC | Mouse
monoclonal | MA5-35613 | Invitrogen; Thermo
Fisher Scientific, Inc. | 1:1,000 |
| MuRF1 | Rabbit
monoclonal | 4305 | Cell Signaling
Technology, Inc. | 1:1,000 |
| MaFbx | Rabbit
monoclonal | 30919 | Cell Signaling
Technology, Inc. | 1:1,000 |
| Akt | Rabbit
monoclonal | 9272 | Cell Signaling
Technology, Inc. | 1:1,000 |
| p-Akt | Rabbit
monoclonal | 9271 | Cell Signaling
Technology, Inc. | 1:1,000 |
| Foxo3a | Rabbit
monoclonal | 2497 | Cell Signaling
Technology, Inc. | 1:1,000 |
| p-Foxo3a | Rabbit
monoclonal | 5538 | Cell Signaling
Technology, Inc. | 1:1,000 |
| PGC-1α | Rabbit
monoclonal | ab191838 | Abcam | 1:1,000 |
| OXPHOS | Mouse
monoclonal | ab110413 | Abcam | 1:1,000 |
| Nrf2 | Rabbit
monoclonal | 12721 | Cell Signaling
Technology, Inc. | 1:1,000 |
| HO-1 | Mouse
monoclonal | sc-136960 | Santa Cruz
Biotechnology, Inc. | 1:500 |
| Keap1 | Mouse
monoclonal | sc-365626 | Santa Cruz
Biotechnology, Inc. | 1:500 |
| NLRP3 | Rabbit
monoclonal | 15101 | Cell Signaling
Technology, Inc. | 1:1,000 |
| Caspase-1 | Rabbit
monoclonal | 83383 | Cell Signaling
Technology, Inc. | 1:1,000 |
| Cleaved
caspase-1 | Rabbit
monoclonal | 89222 | Cell Signaling
Technology, Inc. | 1:1,000 |
| GSDMD | Rabbit
monoclonal | 39754 | Cell Signaling
Technology, Inc. | 1:1,000 |
| Cleaved GSDMD | Rabbit
monoclonal | 34677 | Cell Signaling
Technology, Inc. | 1:1,000 |
| β-actin | Mouse
monoclonal | sc-47778 | Santa Cruz
Biotechnology, Inc. | 1:500 |
Immunofluorescence assay
Myotubes were treated with either 500 ng/ml LPS
alone or co-treated with 1.25, 2.5 or 5 µg/ml VSE for 24 h at 37°C
and then stained with MitoTracker Orange (Invitrogen; Thermo Fisher
Scientific, Inc.) for 30 min at 37°C. To target mitochondrial ROS
(mtROS), cells were stained with MitoSOX (Invitrogen; Thermo Fisher
Scientific, Inc.) dissolved in anhydrous N,N-dimethylformamide
(MilliporeSigma) for 30 min at 37°C. The mitochondria were observed
at 554/576 nm, and the generation of mtROS was observed at 488/510
nm, under a fluorescence microscope. Images were recorded using the
LAS X version 3.7.2.22383 software (Leica Microsystems GmbH).
To assess the mitochondrial membrane potential
(Δψm), cells were incubated with JC-1 dye (Invitrogen; Thermo
Fisher Scientific, Inc.) at a final concentration of 5 µM at 37°C
in the dark for 30 min according to the manufacturer's protocol.
JC-1 fluorescence was detected at 514/529 nm for green fluorescence
and 514/590 nm for red fluorescence using a fluorescence microscope
(Leica Microsystems GmbH). All fluorescence intensities were
quantified using ImageJ version 1.54d software (National Institutes
of Health).
Animal experiments
A total of 6 male C57BL/6J mice aged 5 weeks
(average body weight, 23 g) were purchased from Hyochang Science
and underwent an acclimation period of 1 week. The mice were housed
in an appropriately controlled environment at 25–30°C with 50–60%
relative humidity and a 12-h light/dark cycle. No restrictions were
applied on access to food and water. To validate the effects of VSE
in the sepsis-induced sarcopenia model, mice were divided into four
groups to ensure comparable average body weights across groups with
six mice per group: i) Control (saline); ii) LPS + saline (1 mg/kg
LPS for 24 h); iii) LPS + low VSE (1 mg/kg LPS and 2.5 mg/kg/day
VSE); and iv) LPS + high VSE (1 mg/kg LPS and 5 mg/kg/day VSE) for
7 days. LPS was dissolved in sterile water and VSE was dissolved in
saline solution; all groups, including the control group, were
orally administered the same volume of saline (100 µl/mouse) either
alone or containing VSE daily for 7 days, followed by
administration of 1 mg/kg LPS in 100 µl sterile water by
intraperitoneal injection on day 8 to induce an inflammatory
response in vivo. Body weight was measured on days 0, 2, 4,
6, 8 (before LPS treatment) and 9 (before sacrifice) to monitor the
effects of VSE and LPS over time. Daily body weight measurement was
avoided to minimize animal handling stress. The control group was
administered sterile saline (100 µl; intraperitoneally), equivalent
in volume and route to the LPS-treated group.
Grip strength tests were carried out 18 h after LPS
administration. The mice were then fasted from food only, with free
access to water, for 24 h in order to reduce biological variability
and ensure consistency in downstream measurements. Grip strength
was measured 18 h after LPS administration, based on the results of
a pilot study (Fig. S1). In this
study, C57BL/6J mice were intraperitoneally injected with LPS (1
mg/kg), and physiological and molecular parameters were assessed at
18 and 24 h post-injection. Body and skeletal muscle weights
(gastrocnemius, tibialis anterior, extensor digitorum longus and
soleus) were slightly decreased at both time points, although this
was not significant. Serum TNF-α levels were significantly elevated
at 18 h, and mRNA expression levels of MuRF1 and Atrogin-1 were
markedly increased at 18 h compared with those in the control group
(Fig. S1). Based on these
results, 18 h was selected as the optimal time point for evaluating
early-stage systemic inflammation and muscle atrophy. These
findings support the appropriateness of the 18 h time point for
grip strength measurement. Blood samples were collected and mice
were sacrificed 6 h after the grip strength test. The animal
experiments were approved by the Animal Ethics Committee of
Kyungpook National University (approval no. KNU 2024-0511; Daegu,
South Korea) and were carried out in accordance with the
institution's ethical guidelines. The sample size (n=6 per group)
was selected based on previous studies using murine models of
inflammation-induced muscle atrophy, where similar group sizes
yielded statistically valid and biologically relevant results
(15–17). Additionally, the group size was
determined in consideration of ethical guidelines, statistical
feasibility and animal welfare. At the end of the experiment, mice
were deeply anesthetized with isoflurane (4–5% in oxygen), followed
by cervical dislocation. Death was confirmed by the absence of a
heartbeat and corneal reflex. Immediately after sacrifice, the GAS
muscles were carefully dissected, rinsed in ice-cold PBS to remove
residual blood, blotted dry, snap-frozen in liquid nitrogen, and
stored at −80°C until analysis. For protein extraction, tissues
were lysed in PRO-PREP™ protein extraction solution (Intron
Biotechnology, Inc.) on ice, homogenized using a tissue grinder,
and centrifuged at 13,000 × g for 20 min at 4°C. The supernatants
were collected and used for subsequent western blotting.
Grip strength test
Grip strengths of the mice were measured using a
grip strength test device (Grip Strength Meter for Mice and Rats;
Ugo Basile SRL), with five repetitions per group. After acquiring
the results, the data were standardized to body weight and
expressed as the mean.
H&E staining
GAS muscle tissues were obtained from each group.
The tissues were fixed in 10% neutral buffered formalin at room
temperature for 24 h, dehydrated and embedded in paraffin.
Paraffin-embedded tissues were sectioned at a thickness of 5 µm,
deparaffinized in xylene and rehydrated through a graded ethanol
series followed by distilled water. The sections were then stained
with hematoxylin for 5–10 min at room temperature, rinsed in water
and counterstained with eosin for 1–3 min at room temperature. All
staining procedures were performed using a standard protocol by a
commercial pathology service (18). After staining, muscle tissue
sections were imaged using a bright-light microscope, and muscle
fiber organization was analyzed using ImageJ 1.54d (National
Institutes of Health).
Statistical analysis
In vitro experiments were conducted with
three independent biological replicates, and in vivo
experiments were performed with six mice per group (n=6), and all
data are presented as the mean ± SD. Statistical analyses were
carried out using GraphPad Prism 9.4.1 (Dotmatics). One-way ANOVA
was used to compare differences among multiple groups, followed by
Tukey's post hoc test for pairwise comparisons. P<0.05 was
considered to indicate a statistically significant difference.
Results
VSE attenuates inflammation-induced
muscle atrophy in C2C12 myotubes
Inflammation is one of the main elements influencing
skeletal muscle atrophy. Inflammation was induced in muscles using
LPS, an effective proinflammatory agent (6,19).
Muscle atrophy results from activation of the ubiquitin system,
which is associated with muscle metabolism (20). Controlling skeletal muscle atrophy
specifically requires regulation of the expression of the
muscle-specific E3 ubiquitin ligases MuRF1 and muscle atrophy F-box
protein (MaFbx) and their upstream mechanism, the Akt/Foxo3a
pathway (21). Fig. 1 shows the effects of VSE on cell
viability and muscle atrophy-related factors. As Fig. 1A illustrates, VSE exerted no toxic
effects on C2C12 myotubes at concentrations ≤5 µg/ml. Meanwhile,
cell viability significantly increased with VSE treatment
(114–131%) compared with the non-treatment group (100%). Cell
viability slightly decreased between 5 and 10 µg/ml (Fig. 1A); however, the viability at 10
µg/ml was still increased compared with the control, suggesting no
cytotoxicity at concentrations >5 µg/ml. The treatment
concentrations were set as 1.25, 2.5 and 5 µg/ml in subsequent
experiments.
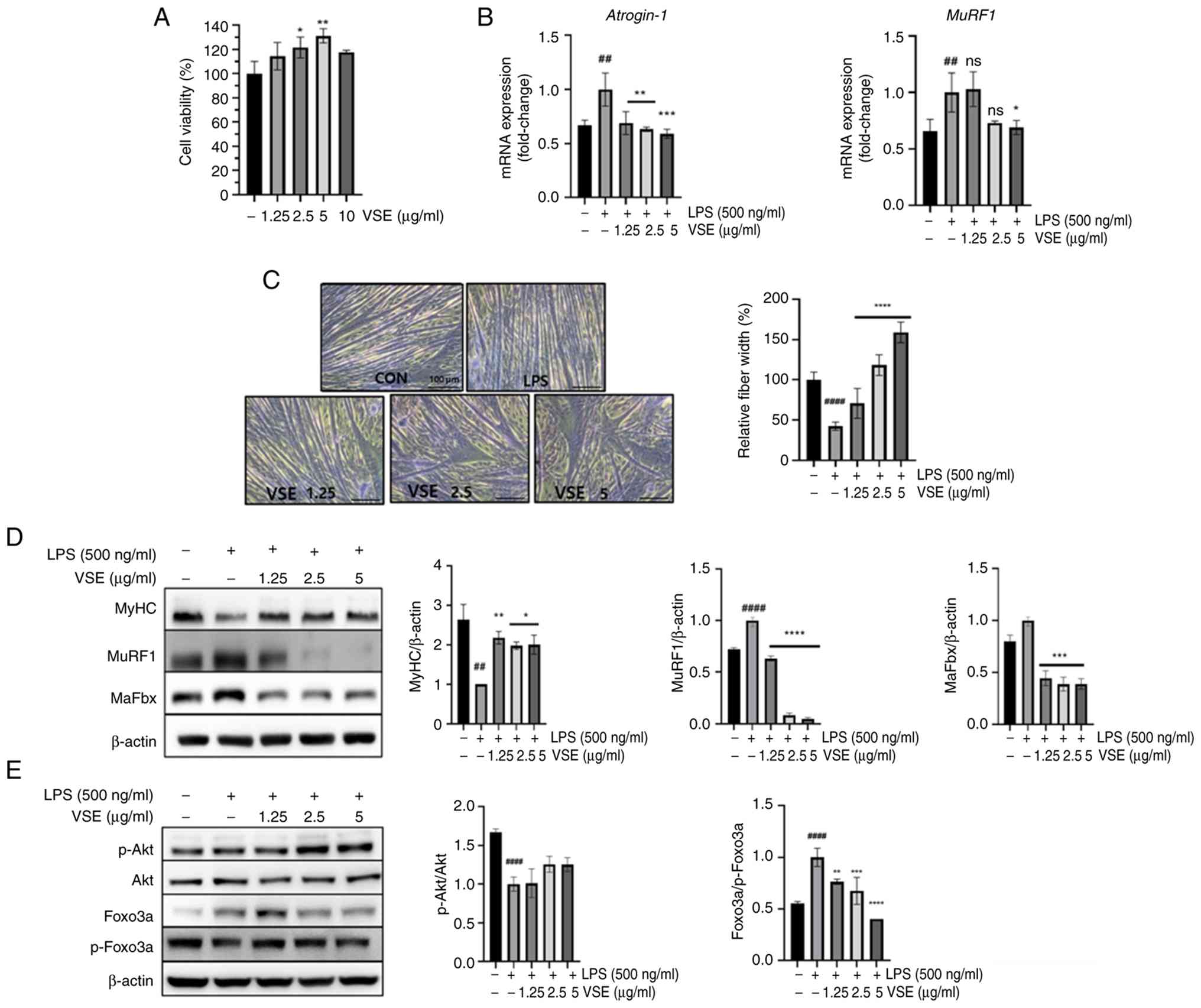 | Figure 1.Effects of VSE on
inflammation-induced muscle atrophy in C2C12 myotubes. All
experiments were carried out after C2C12 myoblasts had
differentiated into myotubes for 4 days. C2C12 myotubes were
pretreated with 0 (LPS only), 1.25, 2.5 or 5 µg/ml VSE for 3 h and
then co-treated with 500 ng/ml LPS for (B, D and E) 24 h and (C) 48
h. Controls received no treatments. (A) C2C12 myotubes were exposed
to 0–10 µg/ml VSE for 48 h to assess cytotoxicity. (B) mRNA levels
of muscle-specific E3 ubiquitin ligases MuRF1 and
atrogin-1 were measured using reverse
transcription-quantitative PCR. (C) Giemsa-stained images of C2C12
myotubes showing changes in relative fiber width after treatment.
CON, untreated control; LPS, 500 ng/ml; 1.25, 2.5 and 5,
co-treatment with LPS (500 ng/ml) and VSE at the indicated
concentrations (µg/ml). Analysis was performed using ImageJ
(National Institutes of Health). Scale bar, 100 µm. Western blot
analysis of (D) MuRF1, MaFbx and MyHC protein levels, and (E)
phosphorylated Akt and Foxo3a, including membrane images and
semi-quantitative analysis using ImageJ. All data are presented as
the mean ± SD of triplicate results and statistical analysis was
carried out using one-way ANOVA with Tukey's post hoc test.
##P<0.01 and ####P<0.0001 vs. control
group; *P<0.05, **P<0.01,
***P<0.001, ****P<0.0001 vs.
LPS-treated group. MuRF1, muscle-specific RING finger protein 1;
MaFbx, muscle atrophy F-box protein; MyHC, myosin heavy chain; CON,
control; LPS, lipopolysaccharide; VSE, Veronicastrum
sibiricum seed extract; p-, phosphorylated; ns, not
significant. |
VSE treatment at 5 µg/ml reduced the upregulation of
MuRF1 and atrogin-1 mRNA expression caused by LPS
Meanwhile, an analogous pattern in expression was observed for
protein expression levels of MuRF1 and MaFbx. Moreover, VSE
treatment significantly attenuated the LPS-induced decrease in MyHC
protein expression (Fig. 1D).
Giemsa staining revealed that VSE treatment restored the thinned
myotube morphology caused by LPS exposure, indicating an
alleviating effect on LPS-induced atrophy (Fig. 1C). Furthermore, LPS significantly
reduced the phosphorylation of both Akt and Foxo3a, leading to the
activation of muscle atrophy-related genes such as MuRF1 and
MaFbx. By contrast, VSE treatment increased the protein
levels of MyHC, phosphorylated Akt and Foxo3a (Fig. 1E). These findings indicated that
VSE alleviated inflammation-induced skeletal muscle atrophy by
modulating the Akt/Foxo3a signaling pathway and subsequently
suppressing the expression of atrophy-related genes.
VSE mitigates pyroptosis by
suppressing NLRP inflammasome activation
Pyroptosis, which is activated under various
inflammatory states, including LPS-induced inflammation, is
modulated by the NLRP inflammasome, with NLRP3 initiating
inflammasome expression (22,23).
Thus, the effect of VSE on NLRP inflammasome activation via the
NLRP3 pathway was investigated (Fig.
2). When administered alone, LPS increased the mRNA levels of
NLRP3, which were downregulated by VSE treatment to levels
similar to those observed in the control group (Fig. 2A). Additionally, the protein
expression levels of pyroptosis-related factors were investigated,
including those of NLRP3, caspase-1, cleaved-caspase-1, GSDMD and
cleaved-GSDMD. The ratios of cleaved to total caspase-1 and GSDMD
were significantly increased in the LPS group, indicating
activation of pyroptosis. VSE treatment effectively suppressed
these ratios, suggesting inhibition of LPS-induced pyroptotic
signaling. A significant reduction in cleaved caspase-1 levels was
observed only for 5 µg/ml VSE (Fig.
2B). These results demonstrated that VSE may reduce NLRP3
inflammasome-mediated pyroptosis.
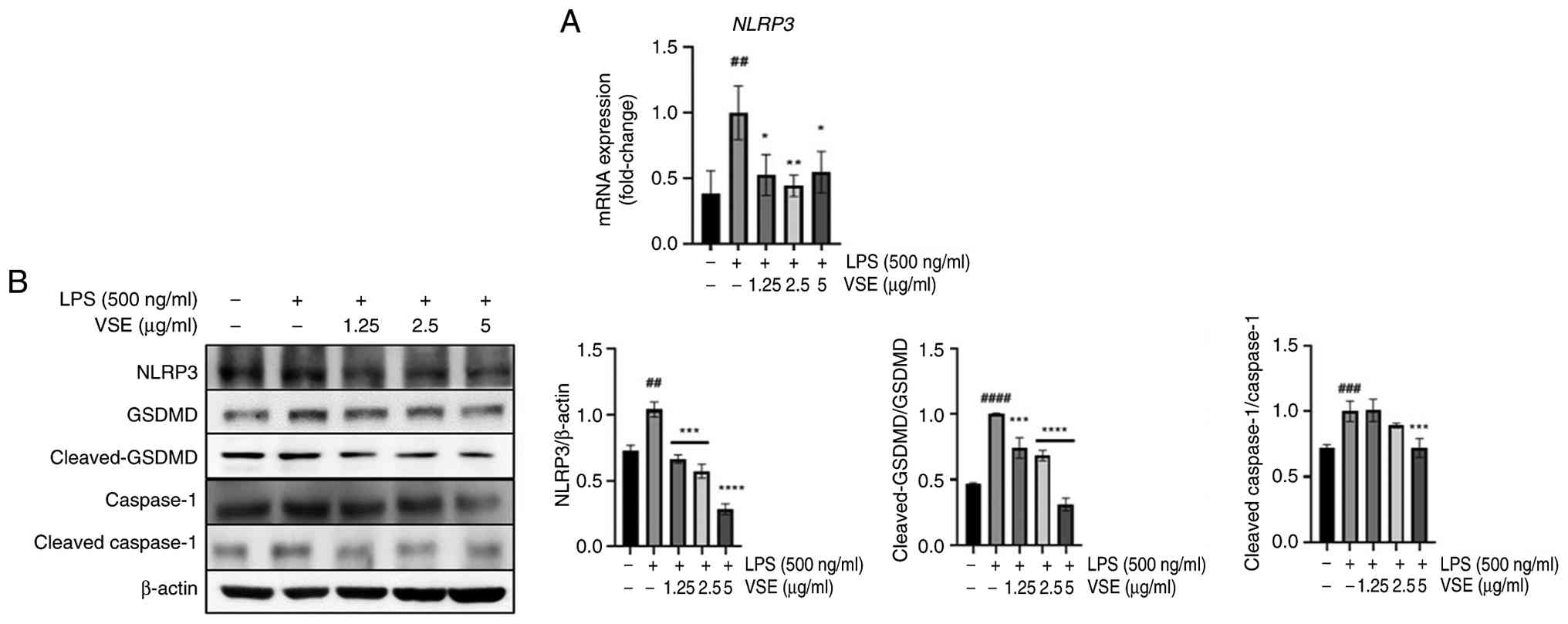 | Figure 2.VSE downregulates the NLRP3
inflammasome. C2C12 myotubes were pretreated with VSE at 0 (LPS
only), 1.25, 2.5 and 5 µg/ml for 3 h and then co-treated with 500
ng/ml LPS for 24 h. Controls received no treatments. (A) mRNA
expression levels of NLRP3, the NLRP3 inflammasome
initiator, were analyzed using reverse transcription-quantitative
PCR. (B) Protein expression levels of factors associated with the
NLPR3 pathway, a major pyroptosis pathway, were analyzed using
western blotting and ImageJ. All data are presented as the mean ±
SD of triplicate results and statistical analysis was carried out
using one-way ANOVA with Tukey's post hoc test.
####P<0.0001, ###P<0.001,
##P<0.01 vs. control; *P<0.05, **P<0.01,
***P<0.001, ****P<0.0001 vs. LPS treatment. NLRP3, NLR family
pyrin domain containing 3; GSDMD, gasdermin-D; LPS,
lipopolysaccharide; VSE, Veronicastrum sibiricum seed
extract. |
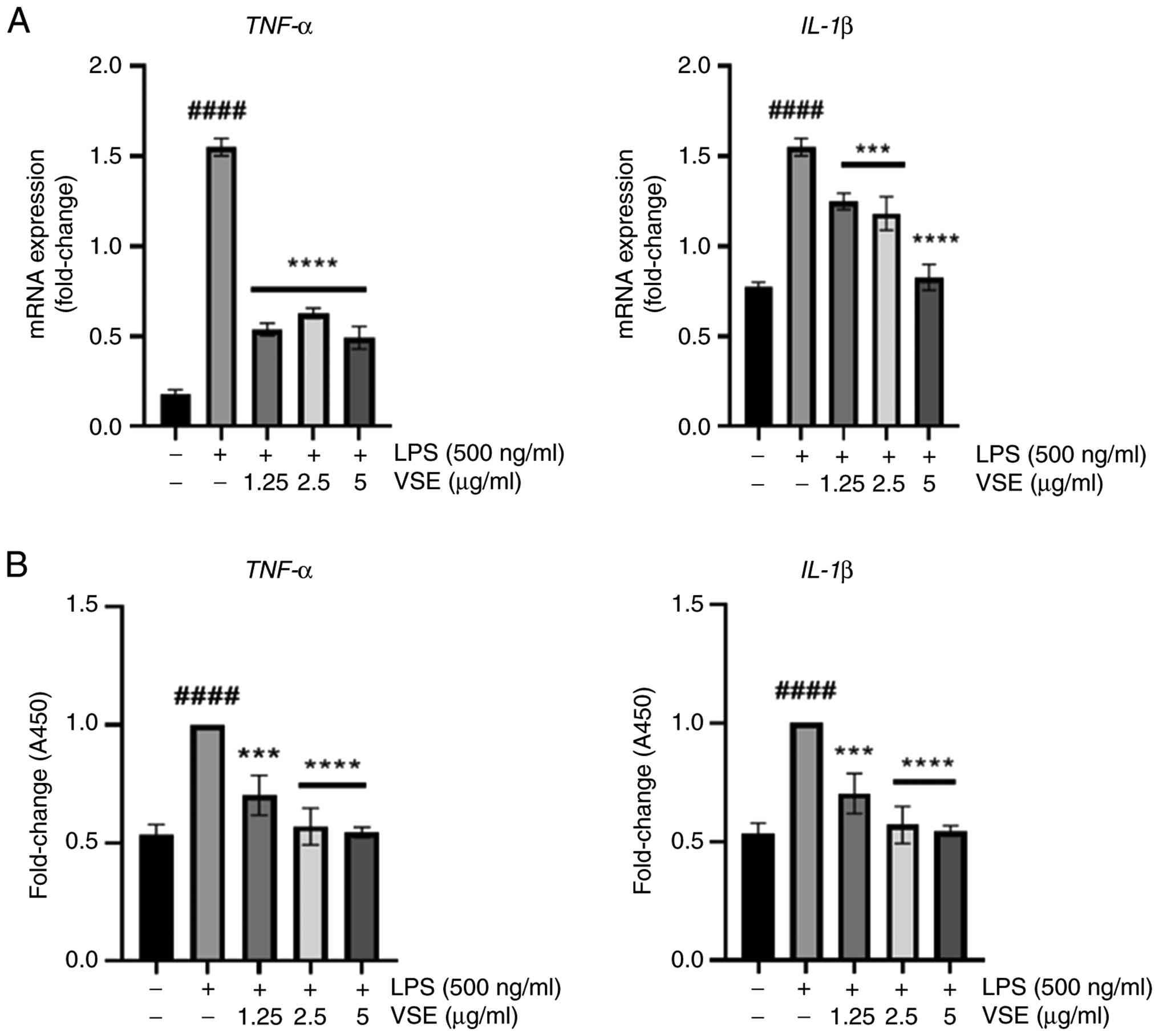
VSE exerts anti-inflammatory effects
during pyroptosis
Release of IL-1β, a proinflammatory cytokine, is
increased during pyroptosis, leading to the initiation of several
cellular processes, including inflammation (24), and the release of IL-1β through
cell membrane pores created by cleaved-GSDMD is considered to be a
key factor in pyroptosis (25). To
ascertain the effect of VSE on LPS-induced inflammatory responses,
VSE-mediated release of IL-1β and TNF-α was observed. The secretion
of both cytokines was evaluated at the mRNA and protein levels.
When administered alone, LPS substantially increased the levels of
IL-1β and TNF-α; however, VSE treatment decreased the LPS-induced
upregulation at all concentrations (Fig. 3A and B). These findings suggested
that the anti-inflammatory effects exerted by VSE include modifying
the release of proinflammatory cytokines.
VSE alleviates oxidative stress during
pyroptosis
Pyroptosis and the generation of ROS are mutually
dependent, functioning as cause and effect. Specifically,
regulation of mtROS is a key process in pyroptosis (26). The expression levels of factors
associated with the nuclear factor erythroid 2-related factor 2
(Nrf2)/heme oxygenase-1 (HO-1) signaling pathway were assessed to
examine the potential alleviating effects of VSE on oxidative
stress. VSE treatment at 5 µg/ml significantly upregulated the
Nrf2/HO-1 signaling pathway (Fig.
4A). Although statistical significance was not assessed,
treatment with 5 µg/ml VSE appeared to increase HO-1, Nrf2 and
PGC-1α expression when compared with the untreated control.
Furthermore, the impact of VSE on mtROS release was assessed. To
quantify this impact using immunofluorescence staining, mitotracker
Orange staining, which labels mitochondria, was combined with
mitoSOX green staining, specifically targeting mtROS. The
LPS-induced generation of mtROS was decreased by VSE treatment
(Fig. 4B), suggesting that VSE may
influence pyroptosis by regulating mtROS levels.
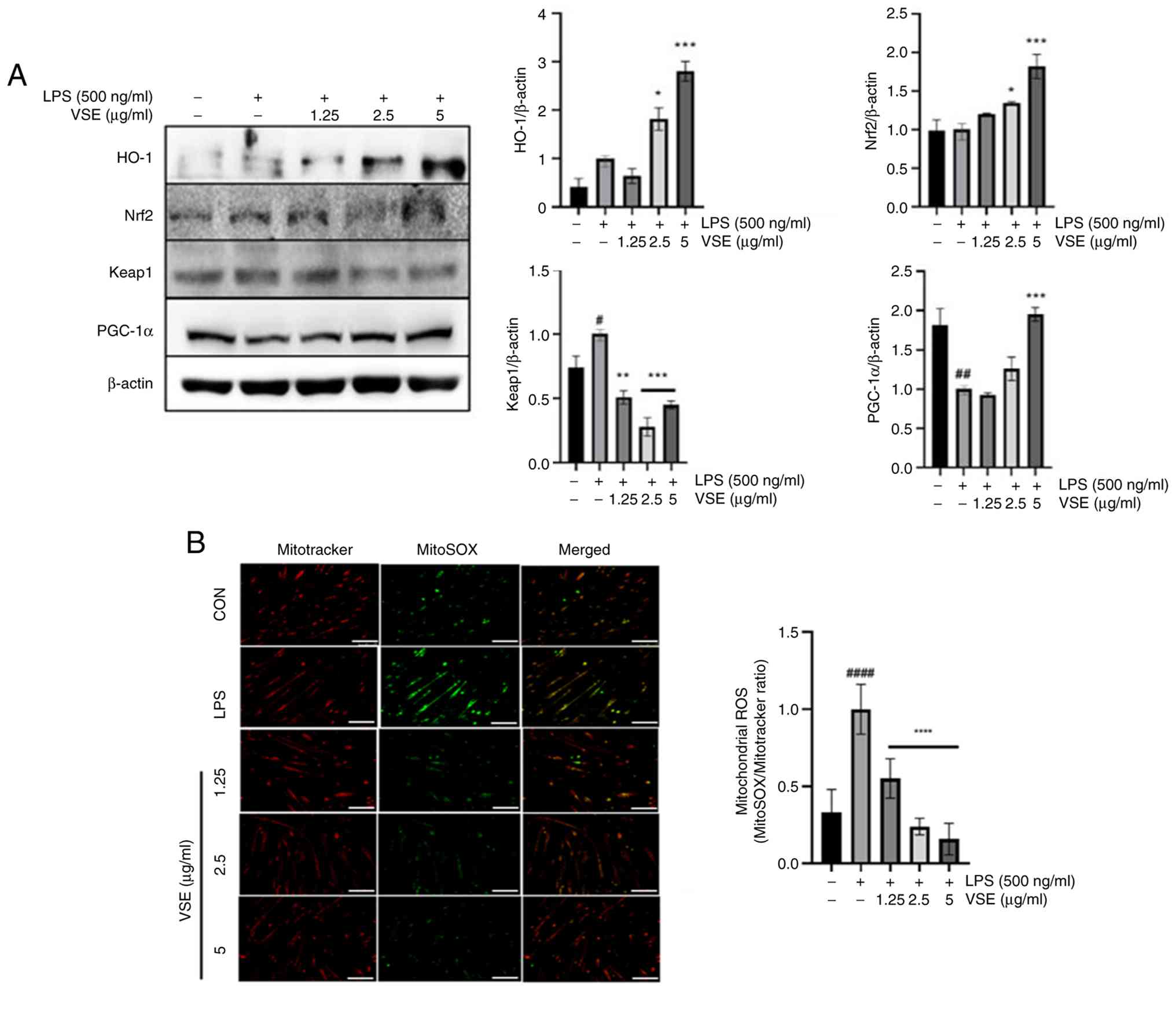 | Figure 4.VSE exerts antioxidant effects in
C2C12 myotubes. C2C12 myotubes were pretreated with 0 (LPS only),
1.25, 2.5 and 5 µg/ml VSE for 3 h and then co-treated with 500
ng/ml LPS for 24 h. (A) Protein expression levels of
antioxidant-related factors HO-1, Nrf2 and Keap1, and mitochondrial
metabolism-related factor PGC-1α were analyzed using western
blotting. (B) Images (left) were obtained under a fluorescence
microscope to investigate the generation of mitochondrial ROS using
ImageJ (right). Scale bar, 100 µm. All data are presented as the
mean ± SD of triplicate results, and statistical analysis was
carried out using one-way ANOVA with Tukey's post hoc test.
#P<0.05, ##P<0.01,
####P<0.0001 vs. control; *P<0.05, **P<0.01,
***P<0.001, ****P<0.0001 vs. LPS treatment. LPS,
lipopolysaccharide; VSE, Veronicastrum sibiricum seed
extract; ROS, reactive oxygen species; HO-1, heme oxygenase-1;
Nrf2, nuclear factor erythroid 2-related factor 2; Keap1, kelch
like ECH associated protein 1; PGC-1α, peroxisome
proliferator-activated receptor γ coactivator 1α; CON, control. |
VSE restores mitochondrial function
during pyroptosis progression
The NLPR3 protein translocates to the mitochondria
in response to stimuli such as LPS, which induces mitochondrial
fragmentation. Healthy mitochondria are required to suppress NLPR3
inflammasome activation (27,28).
Additionally, muscle atrophy is largely influenced by the
degradation of mitochondrial functions, including oxidative
phosphorylation, aerobic respiration and Δψm (12).
The present study aimed to investigate the effects
of VSE treatment on mitochondrial functions, which are known to be
negatively impacted following LPS stimulation (29). Therefore, the expression levels of
oxidative phosphorylation system (OXPHOS) complexes I–V were
examined. While LPS stimulation significantly reduced the
expression of most OXPHOS complexes, complex IV remained unchanged.
VSE treatment, however, dose-dependently restored the expression of
complexes I–III and V, with complexes IV and V showing particularly
notable increases at higher VSE concentrations (Fig. 5A). These findings suggest that VSE
helps maintain mitochondrial metabolism at near-normal levels.
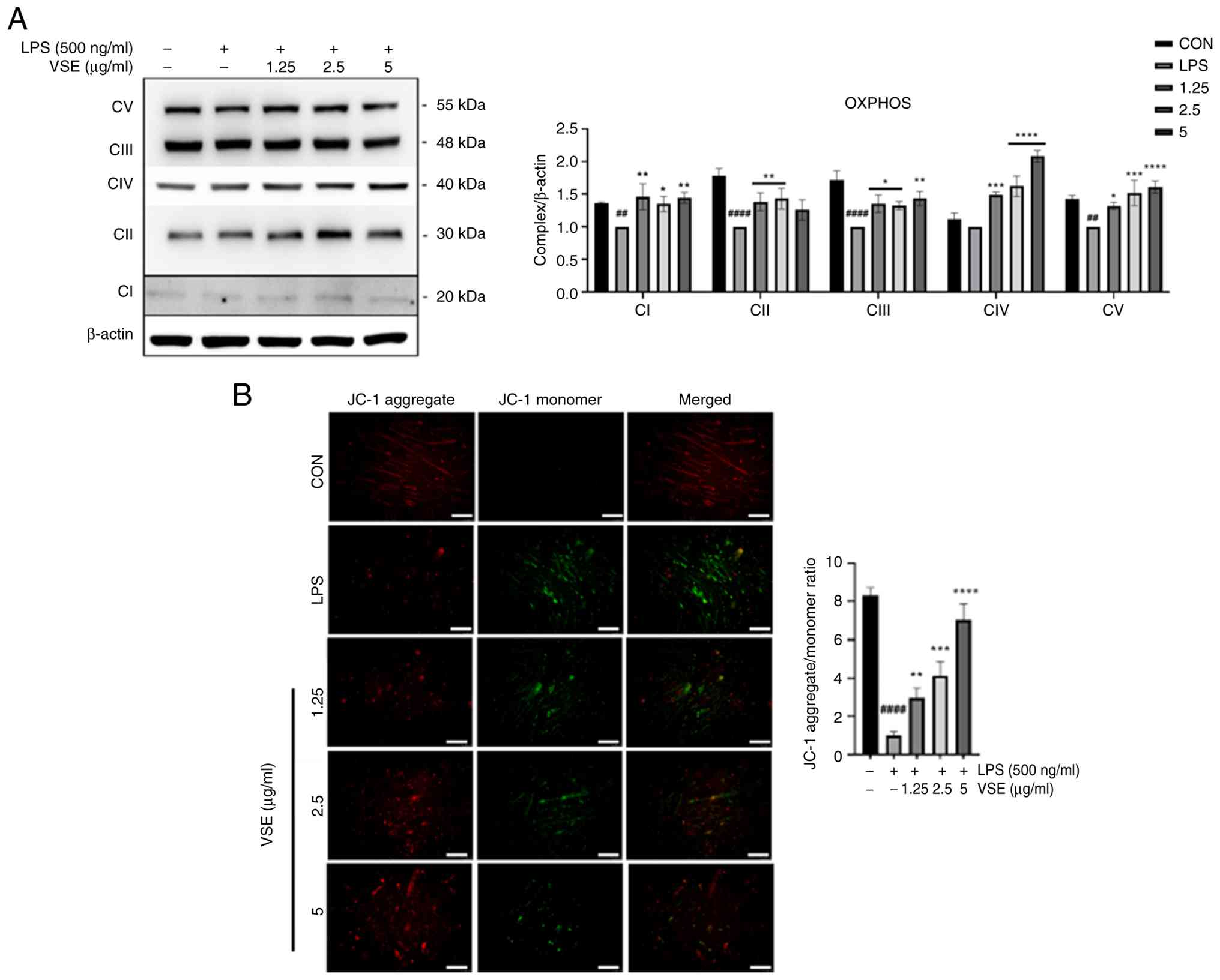 | Figure 5.VSE exerts protective effects on
mitochondrial function, reducing functional impairment under
inflammatory conditions. C2C12 myotubes were pretreated with 0 (LPS
only), 1.25, 2.5 and 5 µg/ml VSE for 3 h and then co-treated with
500 ng/ml LPS for 24 h. (A) Protein expression levels of the
mitochondrial dynamics-related OXPHOS genes, including CI-CV, were
analyzed using western blotting. (B) JC-1 staining was carried out
to assess the mitochondrial membrane potential. Representative
images of JC-1 fluorescence (red/green) were analyzed using ImageJ
(National Institutes of Health). Scale bar, 25 µm. All data are
presented as the mean ± SD of triplicate results and statistical
analysis was carried out using one-way ANOVA with Tukey's post hoc
test. ##P<0.01, ####P<0.0001 vs.
control; *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001
vs. LPS treatment. LPS, lipopolysaccharide; VSE, Veronicastrum
sibiricum seed extract; CI-CV, complexes I–V; OXPHOS, oxidative
phosphorylation system; CON, control. |
Additionally, JC-1 staining was performed to assess
changes in Δψm. In healthy mitochondria, JC-1 forms aggregates
emitting red fluorescence, whereas in depolarized mitochondria it
exists as monomers emitting green fluorescence. LPS treatment
reduced red fluorescence and significantly decreased the red/green
fluorescence ratio, indicating mitochondrial depolarization. By
contrast, VSE treatment restored Δψm in a dose-dependent manner, as
evidenced by the increasing red/green ratio (Fig. 5B).
Collectively, these results indicate that VSE
preserves mitochondrial function at both the molecular (OXPHOS
protein expression) and functional (Δψm) levels under inflammatory
conditions, potentially alleviating muscle atrophy induced by
mitochondrial dysfunction.
VSE improves muscle atrophy in a
sepsis-induced sarcopenia mouse model
Sepsis-induced sarcopenia is a disease in which
sepsis causes a loss of muscle mass and strength; this condition is
considered an important model for studying the effects of systemic
inflammatory responses on muscle (30). Therefore, the potential preventive
impact of VSE treatment on muscle atrophy was investigated in a
sepsis-induced sarcopenia mouse model, since in vitro
analysis revealed that VSE treatment reduced pyroptosis and muscle
atrophy under LPS-induced inflammatory conditions. To this end, 2.5
and 5 mg/kg/day VSE was orally administered daily to 5-week-old
male C57BL/6J mice (n=6). On the 8th day, 1 mg/kg LPS was
administered intraperitoneally. Body weight was measured at
specific time points (days 0, 2, 4, 6, 8 and 9). The decrease in
body weight observed in all groups after day 8 can be partially
attributed to pre-sacrifice fasting, while the greater reduction in
the LPS group may represent LPS-induced catabolic effects. Although
not statistically analyzed, VSE treatment appeared to partially
restore body weight compared with the LPS group (Fig. 6A). The decrease in body weight and
TA tissue weight induced by LPS was slightly restored by VSE
treatment. In particular, the reduction in tissue weight caused by
LPS in the tibialis anterior muscle and the recovery caused by VSE
were both significant (Fig. 6B).
GAS muscle tissue in the LPS-treated group comprised very narrow
fibers, and LPS treatment reduced the muscle surface area
significantly. Tissue patterns were severely disrupted by LPS
treatment, as evidenced by a significant reduction in muscle fiber
surface area. However, VSE administration (2.5 and 5 mg/kg/day)
markedly restored the tissue morphology, with the muscle fiber
surface area returning to levels comparable to the control group.
These improvements were confirmed by quantitative analysis using
ImageJ (Fig. 6C). LPS treatment
significantly weakened the grip strength of mice; however, the grip
strength was significantly improved by 5 µg/ml VSE treatment
(Fig. 6D). These results suggested
that VSE treatment may prevent sepsis-induced sarcopenia by
improving muscle strength.
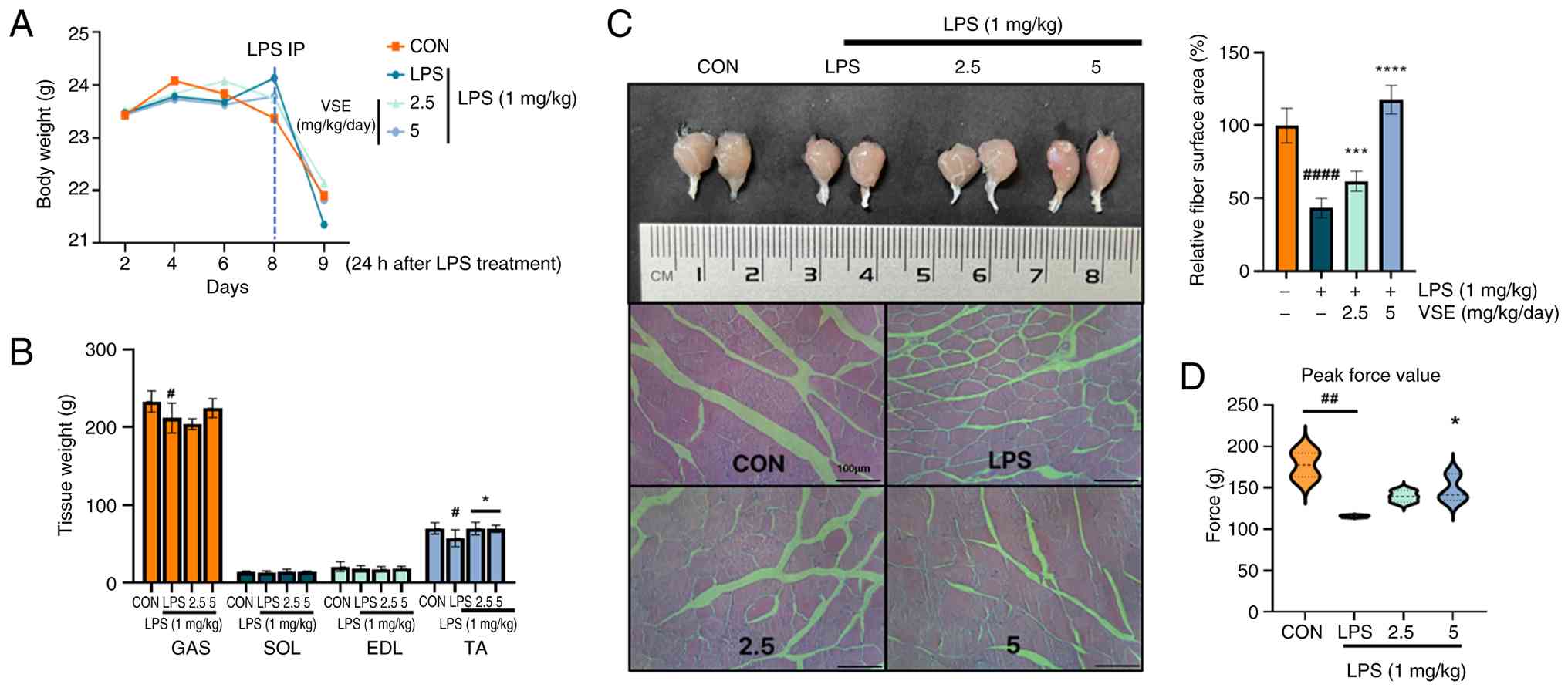 | Figure 6.VSE improves muscle strength through
muscle fiber recovery. (A) Body weight (n=6). (B) Muscle tissue
weight of the GAS, SOL, EDL and TA muscles (n=6). (C) Images
obtained after H&E staining of GAS muscle tissue. Each H&E
staining image was imaged under a microscope, and the fiber surface
area of the muscle tissue was quantitatively analyzed using ImageJ
(National Institutes of Health). Scale bar, 100 µm. (D) Grip
strength measurements (n=5 per group). All data are presented as
the mean ± SD of triplicate results and statistical analysis was
carried out using one-way ANOVA followed by Tukey's post hoc test
in GraphPad Prism 10. #P<0.05,
##P<0.01, ####P<0.0001 vs. control;
*P<0.05, ***P<0.001, ****P<0.0001 vs. LPS treatment. LPS,
lipopolysaccharide; IP, intraperitoneal; VSE, Veronicastrum
sibiricum seed extract; GAS, gastrocnemius; SOL, soleus; EDL,
extensor digitorum longus; TA, tibialis anterior; CON, control. |
VSE relieves pyroptosis in a
sepsis-induced sarcopenia mouse model
NLRP3 inflammasome activation during sepsis induces
muscle cell pyroptosis, a key factor in accelerating muscle fiber
damage and muscle strength loss (31). Pyroptosis may also promote
sarcopenia by modulating the inflammatory microenvironment within
muscles (32). Similar to the
in vitro experiments, the regulatory effects of VSE
treatment on the NLRP3 inflammasome were investigated in
vivo. The protein expression levels of the proinflammatory
cytokine TNF-α were reduced in the serum of mice following VSE
administration (Fig. 7A), which
also reduced the mRNA expression levels of muscle degradation
factor MuRF1 in GAS tissues at 5 µg/ml of VSE (Fig. 7B). Additionally, the protein
expression levels of the muscle protein degradation factors MuRF1
and MaFbx and the NLPR3 pathway-related factors NLRP3, GSDMD,
cleaved-GSDMD, caspase-1 and cleaved-caspase-1 in GAS tissues were
analyzed. While LPS treatment decreased myosin heavy chain (MyHC)
expression, the treatment generally increased the expression levels
of muscle protein degradation factors and NLRP3 pathway-related
factors. Furthermore, VSE treatment restored the expression of MyHC
and MafBx, and reduced the expression of MuRF1, NLRP3,
cleaved-GSDMD and cleaved-caspase-1 to levels comparable with or
lower than those observed in the LPS group, suggesting a protective
effect against muscle atrophy and pyroptotic cell death (Fig. 7C). These results indicate that VSE
may effectively counteract inflammatory muscle damage in
vivo.
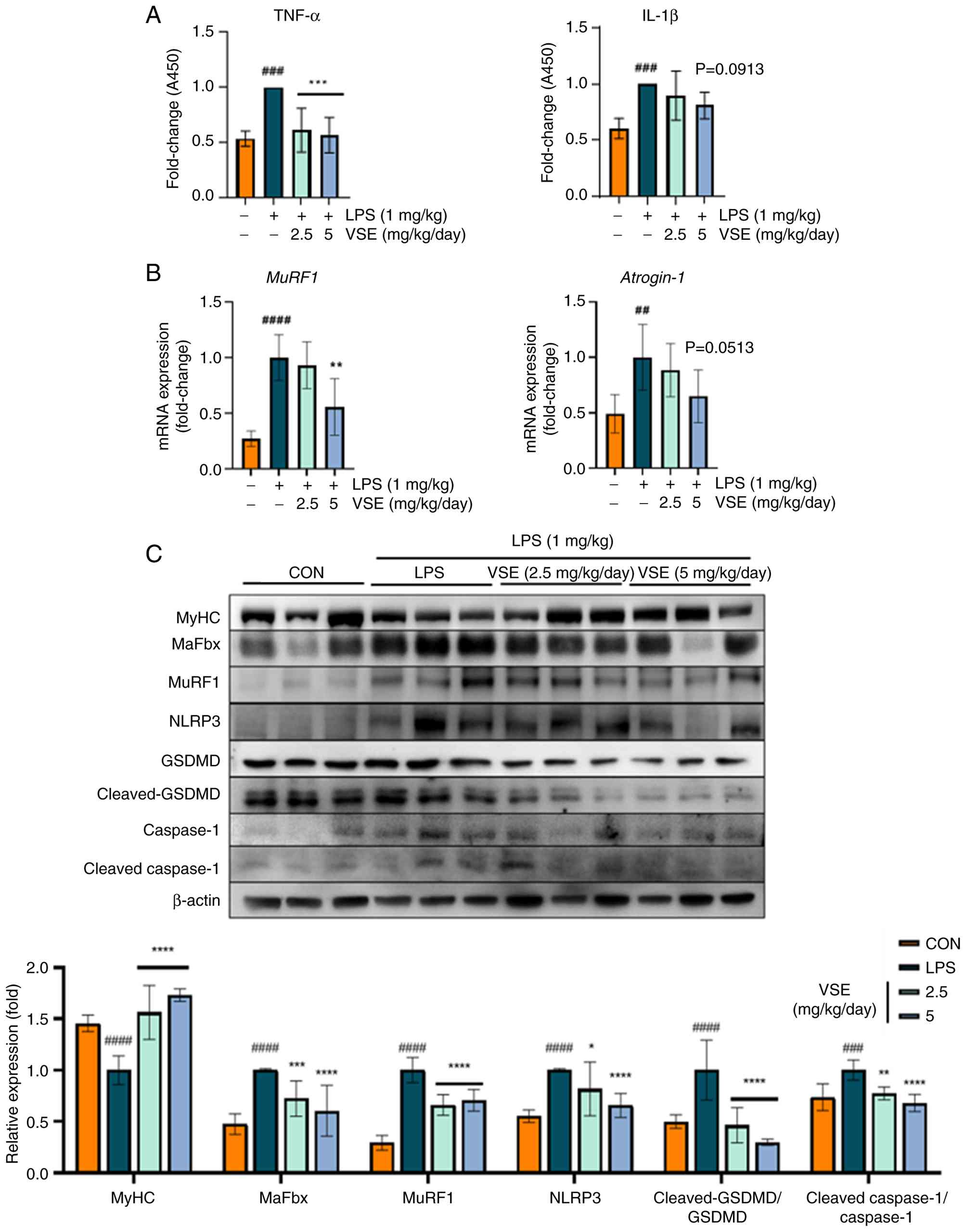 | Figure 7.VSE exhibits protective effects
against pyroptosis. (A) Mouse serum was obtained 24 h after
intraperitoneal LPS administration, diluted to 1:50, and the
protein expression levels of proinflammatory cytokines TNF-α and
IL-1β were analyzed using ELISAs. (B) GAS muscle tissue was
obtained, the mRNA was extracted and the mRNA expression levels of
muscle atrophy-related genes MuRF1 and atrogin-1 were
analyzed using reverse transcription-quantitative PCR. (C) GAS
muscle tissue was obtained, proteins were extracted utilizing
PRO-PREP™, and the protein expression levels of muscle
atrophy-related and NLRP3 pathway-related proteins were analyzed
using western blotting (n=6 per group). All data are presented as
the mean ± SD of triplicate results and statistical analysis was
carried out using one-way ANOVA followed by Tukey's post hoc test
in GraphPad Prism 10. ##P<0.01,
###P<0.001, ####P<0.0001 vs. control;
*P<0.05, **P<0.01, ***P<0.001, ****P<0.0001 vs. LPS
treatment. LPS, lipopolysaccharide; VSE, Veronicastrum
sibiricum seed extract; GAS, gastrocnemius; MuRF1,
muscle-specific RING finger protein 1; MaFbx, muscle atrophy F-box
protein; MyHC, myosin heavy chain; NLRP3, NLR family pyrin domain
containing 3; GSDMD, gasdermin-D; CON, control; A450, absorbance at
450 nm. |
Discussion
LPS is an endotoxin, and LPS signaling is key for
the pathogenesis of acute and chronic inflammatory diseases
(33). In severe cases, this
inflammation can lead to inflammatory myopathy and sepsis, which is
characterized by skeletal muscle dysfunction due to elevations in
inflammatory mediators (34). To
address muscle atrophy caused by inflammation, controlling the
release of inflammatory mediators by balancing anabolic and
catabolic effects is potentially important (33). Anti-inflammatory medications, such
as non-steroidal anti-inflammatory drugs, are actively prescribed
to address inflammatory muscle atrophy; however, the prolonged use
of these drugs can have adverse effects (35). One potential treatment approach is
the development of natural compounds with low toxicity that
specifically target inflammatory reactions (36). Ryu et al (37) revealed that Sargassum
Serratifolium ethanol extract suppressed inflammation-induced
muscle atrophy by exerting anti-inflammatory activity. Furthermore,
Lee et al (38) suggested
that curcumin from Curcuma longa L. Zingiberaceae had
protective effects on sarcopenia by alleviating the inflammatory
condition. Therefore, the present study aimed to investigate the
potential clinical application of VSE treatment considering VSE is
a phytochemical and therefore has potential in muscle wasting
disorders (39).
The activation of the ubiquitin-proteasome system
during an inflammatory state serves a role in muscular atrophy
(1). The ubiquitin-proteasome
process is activated by the interaction of three enzymes, E1, E2
and E3, and is controlled by the Akt/Foxo3a pathway, which promotes
its upstream mechanism (40).
Therefore, it is important to identify how the expression of E3
ligases involved in muscle atrophy is regulated. In LPS-induced
inflammatory conditions, VSE treatment downregulated the protein
expression levels of MaFbx and MuRF1, which are transcriptionally
regulated by the Akt/Foxo3a signaling pathway. To elucidate the
underlying mechanism, the upstream Akt/Foxo3a pathway was examined.
Specifically, VSE increased the phosphorylation of Akt (p-Akt) and
Foxo3a (p-Foxo3a), thereby suppressing the nuclear activity of
Foxo3a and reducing downstream E3 ligase expression. The initiation
of the NLRP3 inflammasome is regarded to be a key element in the
pathogenic mechanism of pyroptosis-induced muscle atrophy (41). NLRP3 inflammasome initiation has
also been demonstrated to interact with the ubiquitin-proteasome
system.
Specifically, LPS can activate both canonical and
non-canonical inflammasome pathways, with NLRP3 serving as a key
component of the canonical pathway by sensing cellular stress and
initiating caspase-1 activation (42). The NLRP3 inflammasome complex
comprises the NLRP3 protein, the apoptosis-associated speck-like
protein containing a caspase recruitment domain adaptor and
pro-caspase-1 (43). Caspase-1 is
activated following NLRP3 activation and cleaves GSDMD into cleaved
GSDMD, contributing to the maturation of IL-1β. Once GSDMD is
cleaved, it forms transmembrane pores that allow an excessive
release of IL-1β, promoting inflammatory cell death, known as
pyroptosis (44). Furthermore,
numerous immune-mediated inflammatory illnesses are caused by
increased IL-1β release (45). The
present study aimed to elucidate the efficacy of VSE treatment in
regulating factors associated with the NLRP3 pathway, including
NLRP3, caspase-1 and GSDMD. These NLRP3-associated factors were
upregulated by exposure to LPS; however, VSE treatment reduced
LPS-induced inflammation. Thus, the present study demonstrated that
VSE may prevent cell pyroptosis by blocking NLRP inflammasome
activation.
Numerous studies have reported that mitochondrial
damage is inflicted by caspase-1, an NLRP3 inflammasome-associated
factor, which then translocates NLRP3 to mitochondria (26,46).
This phenomenon increases mtROS production, thereby impairing
mitochondrial homeostasis, with high levels of damage to the inner
and outer mitochondrial membranes and mitochondrial fragmentation
as common symptoms (47). A
previous study revealed that mtROS served a key role as a stress
factor in activating the NLPR3 inflammasome (48). In addition, ATP and other NLRP3
stimulators cause an influx of Ca2+, which, in turn,
triggers the production of mtROS. By these mechanisms, NLRP3
inflammasome initiation and mitochondrial dysfunction form a
reciprocal network, creating a positive feedback loop (49). PGC-1α is a well-recognized element
that aids in restoring mitochondrial stability, alongside being a
key regulator of mitochondrial biogenesis. Increased PGC-1α
expression reduces mtROS production and helps stabilize the outer
and inner mitochondrial membranes (50). Upregulation of the expression of
the antioxidant factors Nrf2 and HO-1 has also been revealed to aid
in reducing ROS formation (51).
The reduction in mtROS and the upregulation of antioxidant factors
following VSE treatment in the present study suggest that VSE
treatment may shield mitochondrial function against NLPR3
inflammasome activators.
OXPHOS contributes to the pivotal mitochondrial role
in cellular metabolism. The primary function of this system is to
control the generation of ATP (52). OXPHOS comprises five electron
transport chain (ETC) complexes: CI-CV. Of these, CI is the main
entry point for electrons; CII is involved in the oxidation of
succinate to fumarate and transfers electrons to coenzyme Q (CoQ);
and CIII, located in the center of the ETC, distributes reduced
CoQH2 to CIV (cytochrome c), which is an oxidase
found outside the ETC. Finally, CV is responsible for ATP synthesis
(53). Therefore, the operation of
each OXPHOS complex is key to mitochondrial function. The OXPHOS
system serves a major role in acute and chronic inflammation
situations, meaning inhibition of the OXPHOS system accompanies a
decrease in cellular function (54). The present study similarly
demonstrated that mitochondrial function was impaired under
LPS-induced inflammation and that VSE treatment exerted a
protective effect on mitochondria, which may be explained by its
capacity to increase mitochondrial function.
To further validate the protective effects of VSE on
mitochondria, the Δψm was assessed using JC-1 staining, a widely
used indicator of mitochondrial functional integrity (55). Under normal conditions, JC-1
accumulates in mitochondria and forms red-fluorescent aggregates,
whereas in depolarized or dysfunctional mitochondria, JC-1 presents
in a green-fluorescent monomeric form (56). Analysis revealed that LPS treatment
reduced the red fluorescence intensity and the red/green
fluorescence ratio, indicating loss of the Δψm. By contrast, VSE
treatment preserved red fluorescence dose-dependently, suggesting
that VSE may help maintain mitochondrial membrane integrity under
inflammatory conditions. These findings further support the
hypothesis that VSE alleviates mitochondrial dysfunction through
structural (OXPHOS protein levels) and functional (membrane
potential) recovery mechanisms.
The preventive effects of VSE on inflammatory muscle
atrophy were verified by testing VSE applications in a
sepsis-induced sarcopenia mouse model. The sepsis-induced
sarcopenia model is suitable for evaluating muscle atrophy under
inflammatory conditions (57).
This muscle atrophy has clinical significance, and the sepsis model
is important in understanding the pathophysiology of sarcopenia,
whether occurring in acute or chronic inflammatory settings
(58). The sepsis-induced
sarcopenia model used in the present study was suitable for
evaluating the effects of systemic inflammatory responses on muscle
tissue. Furthermore, this model provided data that supported the
anti-inflammatory and muscle-protective effects of VSE. In
particular, VSE treatment in vivo downregulated mediators
(NLRP3, GSDMD, cleaved-GSDMD, caspase-1 and cleaved-caspase-1) in
the NLRP3 inflammasome pathway, which is known to serve a key role
in sepsis-sarcopenia and fiber damage (41), supporting a possible role in
attenuating sepsis-associated inflammatory responses and muscle
damage.
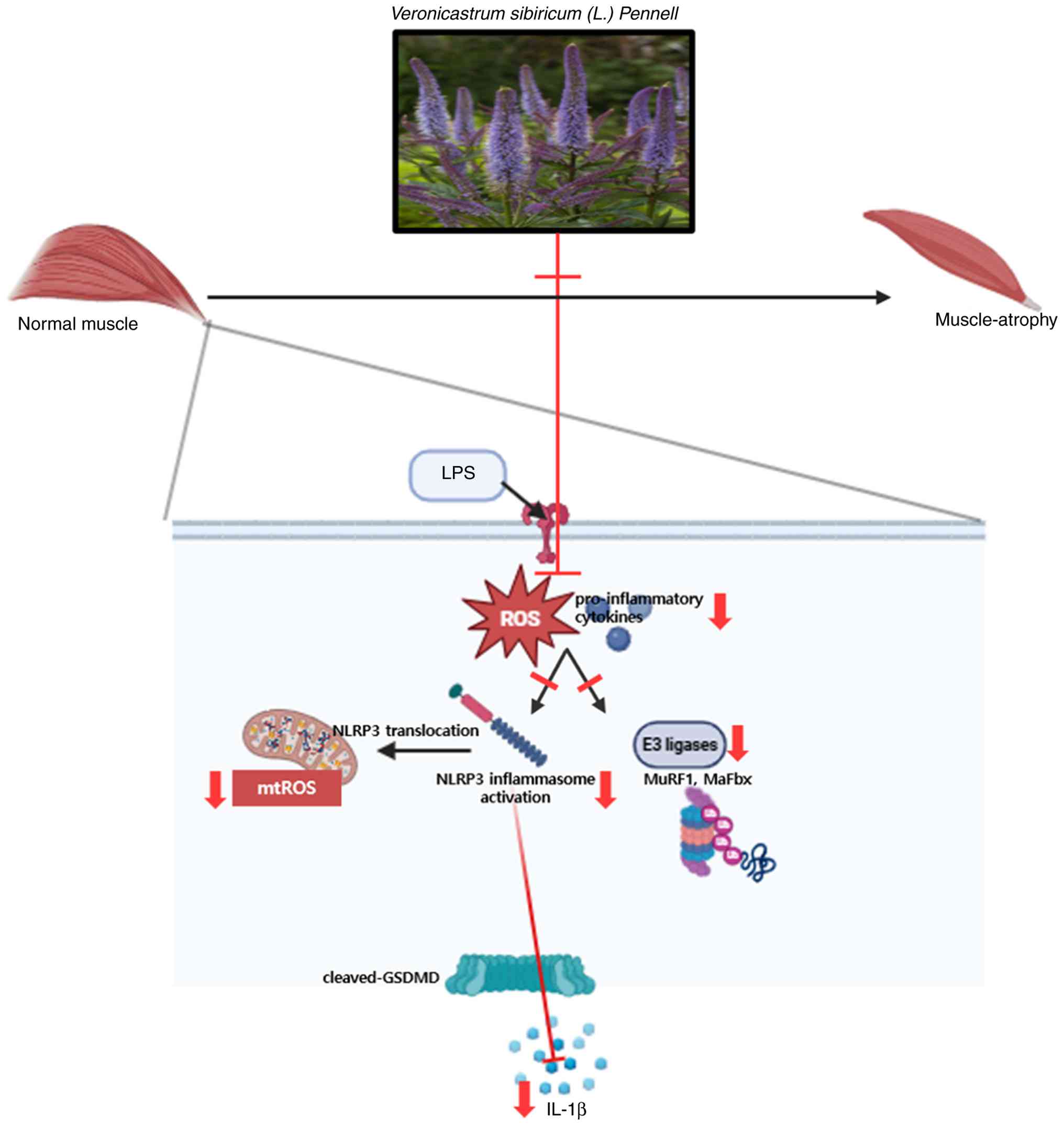
To provide an integrated overview of the proposed
mechanism, a schematic diagram is presented in Fig. 8. This illustration summarizes how
VSE ameliorates LPS-induced muscle atrophy by targeting multiple
key pathological events. LPS treatment induces excessive ROS
production, leading to mitochondrial ROS accumulation, activation
of the NLRP3 inflammasome, and the release of pro-inflammatory
cytokines such as IL-1β. These changes contribute to muscle cell
pyroptosis and promote the expression of muscle-specific E3
ubiquitin ligases, MuRF1 and MaFbx, thereby accelerating muscle
protein degradation and fiber loss.
VSE treatment attenuates these pathological
processes by suppressing mitochondrial ROS generation and
inhibiting the translocation and activation of the NLRP3
inflammasome. Additionally, VSE downregulates the expression of
MuRF1 and MaFbx, thereby preventing excessive muscle protein
degradation. Through these combined actions, VSE effectively
preserves muscle morphology and function under inflammatory
conditions, supporting its therapeutic potential against
inflammation-induced muscle atrophy.
Sepsis-induced sarcopenia, a severe manifestation of
muscle atrophy in critically ill patients, poses pronounced
clinical challenges due to its association with prolonged
hospitalization, impaired physical recovery and increased mortality
(59). The condition, often called
intensive care unit-acquired weakness, is triggered by systemic
inflammatory responses, and closely mimics the pathological
features modeled in the present study (60). Therefore, the outcomes of the
present research have potential translational relevance and provide
valuable insights into the development of inflammation-induced
sarcopenia therapeutics.
Further investigations are necessary to evaluate the
full therapeutic potential of VSE. One major limitation of the
present study is that the bioactive compounds within VSE have yet
to be identified and characterized. While prior research has
reported that plants from the Veronicastrum genus contain
biologically active phytochemicals such as terpenoids, iridoids,
flavonoids and carbohydrates, to the best of our knowledge, the
specific constituents of Veronicastrum sibiricum seeds and
their pharmacological targets remain unknown (39). Therefore, isolating and
characterizing these compounds will be essential for understanding
their mechanisms of action and standardizing VSE concentrations for
therapeutic use. In addition, further studies should explore key
physiological indicators such as the survival rate, inflammation
scores and physiological states of VSE in chronic models of
sarcopenia. Investigating potential synergistic effects with
existing therapies and evaluating the performance of VSE in human
clinical trials are also warranted to develop VSE as a novel
treatment for inflammation-induced muscle atrophy.
Supplementary Material
Supporting Data
Acknowledgements
This abstract was presented at the 2024 KFN
International Symposium and Annual Meeting: Advances in Farm to
Table Technologies for Human Health, held on October 23–25, 2024,
at ICC JEJU, Jeju Island, Korea, and was published as Abstract no.
P09-106.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
EL contributed to the design and optimization of
experimental methods, data collection and performance of in
vitro and in vivo experiments, statistical analysis and
interpretation of results, drafting of the original manuscript,
critical review and editing, validation of findings and figure
preparation. MK contributed to data collection and sample
preparation. JN contributed to the conception and design of
experiments, performed statistical analysis and supervised the
study, reviewed and edited the manuscript and managed the overall
project. EL and JN confirm the authenticity of all the raw data.
All authors have read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
All animal protocols were approved by the Ethical
Committee of Kyungpook National University (approval no.
KNU-2024-0522; Daegu, South Korea).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
LPS
|
lipopolysaccharide
|
|
NLRP3
|
NLR family pyrin domain containing
3
|
|
GSDMD
|
gasdermin-D
|
|
ROS
|
reactive oxygen species
|
|
VSE
|
Veronicastrum sibiricum seed
extract
|
|
CCK-8
|
Cell Counting Kit-8
|
|
RT-qPCR
|
reverse transcription-quantitative
PCR
|
|
GAS
|
gastrocnemius
|
|
TBST
|
Tris-buffered saline containing 0.1%
Tween 20
|
|
mtROS
|
mitochondrial reactive oxygen
species
|
|
Δψm
|
mitochondrial membrane potential
|
|
MuRF1
|
muscle-specific RING finger protein
1
|
|
MaFbx
|
muscle atrophy F-box protein
|
|
Nrf2
|
nuclear factor erythroid 2-related
factor 2
|
|
HO-1
|
heme oxygenase-1
|
|
PGC-1α
|
peroxisome proliferator-activated
receptor γ coactivator 1α
|
|
OXPHOS
|
oxidative phosphorylation system
|
|
MyHC
|
myosin heavy chain
|
|
ETC
|
electron transport chain
|
|
CoQ
|
coenzyme Q
|
References
|
1
|
Haberecht-Müller S, Krüger E and Fielitz
J: Out of control: The role of the ubiquitin proteasome system in
skeletal muscle during inflammation. Biomolecules. 11:13272021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhang J, Huang Y, Chen Y, Shen X, Pan H
and Yu W: Impact of muscle mass on survival in patients with
sepsis: A systematic review and meta-analysis. Ann Nutr Metab.
77:330–336. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Valentine RJ, Jefferson MA, Kohut ML and
Eo H: Imoxin attenuates LPS-induced inflammation and MuRF1
expression in mouse skeletal muscle. Physiol Rep. 6:e139412018.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liang H, Hussey SE, Sanchez-Avila A,
Tantiwong P and Musi N: Effect of lipopolysaccharide on
inflammation and insulin action in human muscle. PLoS One.
8:e639832013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lang CH, Frost RA and Vary TC: Regulation
of muscle protein synthesis during sepsis and inflammation. Am J
Physiol Endocrinol Metab. 293:E453–E459. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Frost RA, Nystrom GJ and Lang CH:
Lipopolysaccharide regulates proinflammatory cytokine expression in
mouse myoblasts and skeletal muscle. Am J Physiol Regul Integr Comp
Physiol. 283:R698–R709. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bergsbaken T, Fink SL and Cookson BT:
Pyroptosis: Host cell death and inflammation. Nat Rev Microbiol.
7:99–109. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang L, Jiao XF, Wu C, Li XQ, Sun HX, Shen
XY, Zhang KZ, Zhao C, Liu L, Wang M, et al: Trimetazidine
attenuates dexamethasone-induced muscle atrophy via inhibiting
NLRP3/GSDMD pathway-mediated pyroptosis. Cell Death Discov.
7:2512021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jin H, Xie W, He M, Li H, Xiao W and Li Y:
Pyroptosis and sarcopenia: Frontier perspective of disease
mechanism. Cells. 11:10782022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Park E, Choi H, Truong CS and Jun HS: The
inhibition of autophagy and pyroptosis by an ethanol extract of
Nelumbo nucifera leaf contributes to the amelioration of
dexamethasone-induced muscle atrophy. Nutrients. 15:8042023.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kim MI and Kim CY: Four new acylated
iridoid glycosides from the aerial part of Veronicastrum
sibiricum and their antioxidant response element-inducing
activity. Chem Biodivers. 15:2018. View Article : Google Scholar
|
|
12
|
Peng Y, Li J, Luo D, Zhang S, Li S, Wang
D, Wang X, Zhang Z, Wang X, Sun C, et al: Muscle atrophy induced by
overexpression of ALAS2 is related to muscle mitochondrial
dysfunction. Skelet Muscle. 11:92021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gao W, Zhang R, Jia W, Zhang J, Takaishi Y
and Duan H: Immunosuppressive diterpenes from Veronicastrum
sibiricum. Chem Pharm Bull (Tokyo). 52:136–137. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mofarrahi M, Sigala I, Guo Y, Godin R,
Davis EC, Petrof B, Sandri M, Burelle Y and Hussain SN: Autophagy
and skeletal muscles in sepsis. PLoS One. 7:e472652012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Morihara N, Hino A, Miki S, Takashima M
and Suzuki JI: Aged garlic extract suppresses inflammation in
apolipoprotein E-knockout mice. Mol Nutr Food Res. 61:17003082017.
View Article : Google Scholar
|
|
17
|
Suzuki T, Kumazoe M, Kim Y, Yamashita S,
Nakahara K, Tsukamoto S, Sasaki M, Hagihara T, Tsurudome Y, Huang
Y, et al: Green tea extract containing a highly absorbent catechin
prevents diet-induced lipid metabolism disorder. Sci Rep.
3:27492013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fischer AH, Jacobson KA, Rose J and Zeller
R: Hematoxylin and eosin staining of tissue and cell sections. CSH
Protoc. 2008.pdb.prot4986. 2008.
|
|
19
|
Ji Y, Li M, Chang M, Liu R, Qiu J, Wang K,
Deng C, Shen Y, Zhu J, Wang W, et al: Inflammation: Roles in
skeletal muscle atrophy. Antioxidants (Basel). 11:16862022.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Eddins MJ, Marblestone JG, Suresh Kumar
KG, Leach CA, Sterner DE, Mattern MR and Nicholson B: Targeting the
ubiquitin E3 ligase MuRF1 to inhibit muscle atrophy. Cell Biochem
Biophys. 60:113–118. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pang X, Zhang P, Chen X and Liu W:
Ubiquitin-proteasome pathway in skeletal muscle atrophy. Front
Physiol. 14:12895372023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhao LR, Xing RL, Wang PM, Zhang NS, Yin
SJ, Li XC and Zhang L: NLRP1 and NLRP3 inflammasomes mediate
LPS/ATP-induced pyroptosis in knee osteoarthritis. Mol Med Rep.
17:5463–5469. 2018.PubMed/NCBI
|
|
23
|
Fusco R, Siracusa R, Genovese T, Cuzzocrea
S and Di Paola R: Focus on the role of NLRP3 inflammasome in
diseases. Int J Mol Sci. 21:42232020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hsu SK, Li CY, Lin IL, Syue WJ, Chen YF,
Cheng KC, Teng YN, Lin YH, Yen CH and Chiu CC: Inflammation-related
pyroptosis, a novel programmed cell death pathway, and its
crosstalk with immune therapy in cancer treatment. Theranostics.
11:8813–8835. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li Y and Jiang Q: Uncoupled pyroptosis and
IL-1β secretion downstream of inflammasome signaling. Front
Immunol. 14:11283582023. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yu JW and Lee MS: Mitochondria and the
NLRP3 inflammasome: physiological and pathological relevance. Arch
Pharm Res. 39:1503–1518. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nicholls DG: Mitochondrial function and
dysfunction in the cell: Its relevance to aging and aging-related
disease. Int J Biochem Cell Biol. 34:1372–1381. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Luo L, Wang F, Xu X, Ma M, Kuang G, Zhang
Y, Wang D, Li W, Zhang N and Zhao K: STAT3 promotes NLRP3
inflammasome activation by mediating NLRP3 mitochondrial
translocation. Exp Mol Med. 56:1980–1990. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Park J, Min JS, Kim B, Chae UB, Yun JW,
Choi MS, Kong IK, Chang KT and Lee DS: Mitochondrial ROS govern the
LPS-induced pro-inflammatory response in microglia cells by
regulating MAPK and NF-κB pathways. Neurosci Lett. 584:191–196.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yoshihara I, Kondo Y, Okamoto K and Tanaka
H: Sepsis-associated muscle wasting: A comprehensive review from
bench to bedside. Int J Mol Sci. 24:50402023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu Y, Wang D, Li T, Yang F, Li Z, Bai X
and Wang Y: The role of NLRP3 inflammasome in inflammation-related
skeletal muscle atrophy. Front Immunol. 13:10357092022. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
McBride MJ, Foley KP, D'Souza DM, Li YE,
Lau TC, Hawke TJ and Schertzer JD: The NLRP3 inflammasome
contributes to sarcopenia and lower muscle glycolytic potential in
old mice. Am J Physiol Endocrinol Metab. 313:E222–E232. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Page MJ, Kell DB and Pretorius E: The role
of lipopolysaccharide-induced cell signalling in chronic
inflammation. Chronic Stress (Thousand Oaks).
6:247054702210763902022. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Londhe P and Guttridge DC: Inflammation
induced loss of skeletal muscle. Bone. 80:131–142. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Alturki M, Beyer I, Mets T and Bautmans I:
Impact of drugs with anti-inflammatory effects on skeletal muscle
and inflammation: A systematic literature review. Exp Gerontol.
114:33–49. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang RX, Zhou M, Ma HL, Qiao YB and Li QS:
The role of chronic inflammation in various diseases and
anti-inflammatory therapies containing natural products.
ChemMedChem. 16:1576–1592. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ryu H, Jeong HH, Kim MJ, Lee S, Jung WK
and Lee B: Modulation of macrophage transcript and secretion
profiles by Sargassum Serratifolium extract is associated
with the suppression of muscle atrophy. Sci Rep. 14:132822024.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lee DY, Chun YS, Kim JK, Lee JO, Ku SK and
Shim SM: Curcumin attenuates sarcopenia in chronic forced exercise
executed aged mice by regulating muscle degradation and protein
synthesis with antioxidant and anti-inflammatory effects. J Agric
Food Chem. 69:6214–6228. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mutinda ES, Mkala EM, Ren J, Kimutai F,
Waswa EN, Odago WO, Nanjala C, Gichua MK, Njire MM and Hu GW: A
review on the traditional uses, phytochemistry, and pharmacology of
the genus Veronicastrum (Plantaginaceae). J Ethnopharmacol.
300:1156952023. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Yu H, Zhu G, Wang D, Huang X and Han F:
PI3K/AKT/FOXO3a pathway induces muscle atrophy by
ubiquitin-proteasome system and autophagy system in COPD rat model.
Cell Biochem Biophys. 82:805–815. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Huang N, Kny M, Riediger F, Busch K,
Schmidt S, Luft FC, Slevogt H and Fielitz J: Deletion of Nlrp3
protects from inflammation-induced skeletal muscle atrophy.
Intensive Care Med Exp. 5:32017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Xu J and Núñez G: The NLRP3 inflammasome:
Activation and regulation. Trends Biochem Sci. 48:331–344. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Leu WJ, Chen JC and Guh JH: Extract from
plectranthus amboinicus inhibit maturation and release of
interleukin 1β through inhibition of NF-κB nuclear translocation
and NLRP3 inflammasome activation. Front Pharmacol. 10:5732019.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kang L, Dai J, Wang Y, Shi P, Zou Y, Pei
J, Tian Y, Zhang J, Buranasudja VC, Chen J, et al: Blocking
caspase-1/Gsdmd and caspase-3/-8/Gsdme pyroptotic pathways rescues
silicosis in mice. PLoS Genet. 18:e10105152022. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Yang Y, Wang H, Kouadir M, Song H and Shi
F: Recent advances in the mechanisms of NLRP3 inflammasome
activation and its inhibitors. Cell Death Dis. 10:1282019.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wen Y, Liu Y, Tang T, Lv L, Liu H, Ma K
and Liu B: NLRP3 inflammasome activation is involved in Ang
II-induced kidney damage via mitochondrial dysfunction. Oncotarget.
7:542902016. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Yu J, Nagasu H, Murakami T, Hoang H,
Broderick L, Hoffman HM and Horng T: Inflammasome activation leads
to caspase-1-dependent mitochondrial damage and block of mitophagy.
Proc Natl Acad Sci USA. 111:15514–15519. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Ip WK and Medzhitov R: Macrophages monitor
tissue osmolarity and induce inflammatory response through NLRP3
and NLRC4 inflammasome activation. Nat Commun. 6:69312015.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Lee GS, Subramanian N, Kim AI,
Aksentijevich I, Goldbach-Mansky R, Sacks DB, Germain RN, Kastner
DL and Chae JJ: The calcium-sensing receptor regulates the NLRP3
inflammasome through Ca2+ and cAMP. Nature. 492:123–127. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Haque PS, Kapur N, Barrett TA and Theiss
AL: Mitochondrial function and gastrointestinal diseases. Nat Rev
Gastroenterol Hepatol. 21:537–555. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wang X, Chen J, Tie H, Tian W, Zhao Y, Qin
L, Guo S, Li Q and Bao C: Eriodictyol regulated ferroptosis,
mitochondrial dysfunction, and cell viability via Nrf2/HO-1/NQO1
signaling pathway in ovarian cancer cells. J Biochem Mol Toxicol.
37:e233682023. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Putignani L, Raffa S, Pescosolido R,
Aimati L, Signore F, Torrisi MR and Grammatico P: Alteration of
expression levels of the oxidative phosphorylation system (OXPHOS)
in breast cancer cell mitochondria. Breast Cancer Res Treat.
110:439–452. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Fernandez-Vizarra E and Zeviani M:
Mitochondrial disorders of the OXPHOS system. FEBS Lett.
595:1062–1106. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Lee I and Hüttemann M: Energy crisis: The
role of oxidative phosphorylation in acute inflammation and sepsis.
Biochim Biophys Acta. 1842:1579–1586. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Smiley ST, Reers M, Mottola-Hartshorn C,
Lin M, Chen A, Smith TW, Steele GD Jr and Chen LB: Intracellular
heterogeneity in mitochondrial membrane potentials revealed by a
J-aggregate-forming lipophilic cation JC-1. Proc Natl Acad Sci USA.
88:3671–3675. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Garner DL and Thomas CA:
Organelle-specific probe JC-1 identifies membrane potential
differences in the mitochondrial function of bovine sperm. Mol
Reprod Dev. 53:222–229. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Goossens C, Weckx R, Derde S, Van
Helleputte L, Schneidereit D, Haug M, Reischl B, Friedrich O, Van
Den Bosch L, Van den Berghe G and Langouche L: Impact of prolonged
sepsis on neural and muscular components of muscle contractions in
a mouse model. J Cachexia Sarcopenia Muscle. 12:443–455. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Langhans C, Weber-Carstens S, Schmidt F,
Hamati J, Kny M, Zhu X, Wollersheim T, Koch S, Krebs M, Schulz H,
et al: Inflammation-induced acute phase response in skeletal muscle
and critical illness myopathy. PLoS One. 9:e920482014. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Cox MC, Booth M, Ghita G, Wang Z, Gardner
A, Hawkins RB, Darden DB, Leeuwenburgh C, Moldawer LL, Moore FA, et
al: The impact of sarcopenia and acute muscle mass loss on
long-term outcomes in critically ill patients with intra-abdominal
sepsis. J Cachexia Sarcopenia Muscle. 12:1203–1213. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Mitobe Y, Morishita S, Ohashi K, Sakai S,
Uchiyama M, Abeywickrama H, Yamada E, Kikuchi Y, Nitta M, Honda T,
et al: Skeletal muscle index at intensive care unit admission is a
predictor of intensive care unit-acquired weakness in patients with
sepsis. J Clin Med Res. 11:834–841. 2019. View Article : Google Scholar : PubMed/NCBI
|