Introduction
With rapid economic development and lifestyle
changes, obesity has become a notable global public health problem.
Since 1990, the prevalence of adult obesity has doubled worldwide,
whereas adolescent obesity has tripled (1). According to the latest data released
by the World Health Organization in 2022, >1 billion individuals
worldwide are affected by obesity, of whom ~65% are adults, 34% are
adolescents and <1% are children. This alarming prevalence
markedly increases the risk of developing insulin resistance (IR),
type II diabetes mellitus (T2DM), cardiovascular disease,
non-alcoholic fatty liver disease and various types of cancer,
including gastric, esophageal, liver, pancreatic, bladder and
breast cancer (1–3). Obesity is more than a simple
imbalance between energy intake and expenditure; rather, it arises
from complex interactions among genetic, immune and metabolic
factors. Elucidating the molecular mechanisms underlying the onset
and progression of obesity is therefore of notable importance for
both prevention and therapeutic intervention.
Adipose tissue is not merely an energy storage depot
but also an active endocrine and immune organ. In addition to
adipocytes, it harbors a variety of immune cells, including
macrophages, T lymphocytes, B lymphocytes and dendritic cells
(4–7). Under conditions of obesity,
anti-inflammatory resident immune cells decline, whereas
pro-inflammatory immune cells accumulate and become activated,
leading to chronic low-grade inflammation in adipose tissue, a key
step in the development of obesity-related metabolic disorders.
Thus, the immune cell network within the adipose tissue plays a
marked role in maintaining tissue homeostasis and metabolic
function (8,9). Over the years, the role of B cells in
obesity and its associated metabolic complications has attracted
increasing attention. B lymphocytes regulate the adipose tissue
immune microenvironment through multiple mechanisms, including
interactions with T cells and macrophages, secretion of antibodies
such as IgM and IgG, and production of cytokines, including IL-10,
TNF-α and IL-6, thereby shaping local inflammatory responses and
metabolic homeostasis (10–12).
During obesity, B cells in adipose tissue undergo numerical
expansion and aberrant activation, thereby exacerbating tissue
inflammation and IR. Understanding the heterogeneity, functional
diversity and regulatory mechanisms of adipose tissue B cells is
essential for unraveling the pathogenesis of obesity and related
metabolic diseases. The present review aimed to comprehensively
summarize the roles of B-cell subsets in adipose tissue
inflammation and metabolic dysregulation during obesity, highlight
the underlying mechanisms and discuss the therapeutic potential of
targeting B cells in obesity-associated metabolic disorders.
Overview of B cells
B-cell subsets and their
characteristics
B cells originate from hematopoietic stem cells in
the bone marrow and undergo a series of differentiation and
maturation processes before participating in immune responses
through the secretion of cytokines and antibodies. As a fundamental
component of the immune system, they play a notable role in
maintaining immune homeostasis and defending against pathogens
(13). B cells can be broadly
categorized into two major subsets, B-1 and B-2 cells, based on
their developmental origins, phenotypic characteristics and
functional properties. B-1 cells originate predominantly from fetal
liver precursors and represent one of the earliest identified
B-cell subsets. They are mainly localized in the pleural and
peritoneal cavities, with a smaller proportion residing in the
spleen and adipose tissue. In contrast, B-2 cells are primarily
derived from adult bone marrow hematopoietic stem cells and are
widely distributed in secondary lymphoid organs, including the
spleen and lymph nodes. According to CD5 expression, B-1 cells can
be further subdivided into CD5+ B-1a and CD5- B-1b cells.
Functionally, B-1a cells rapidly produce natural IgM antibodies
with broad reactivity against self and microbial antigens, thereby
contributing to early immune defense and immune homeostasis,
whereas B-1b cells mainly mediate T cell-independent adaptive
immune responses and generate long-lasting protective immunity. In
contrast, B-2 cells primarily participate in T cell-dependent
immune responses, undergo affinity maturation and class-switch
recombination, and predominantly produce high-affinity IgG
antibodies, thus playing a central role in adaptive humoral
immunity. Although natural antibodies secreted by B-1 cells exhibit
relatively low affinity, their polyreactivity and functional
diversity enable rapid recognition of a wide spectrum of antigens,
making them essential components of the early innate immune system
(14).
B-2 cells, also referred to as conventional B cells,
progress through stages including pro-B and immature B cells before
migrating to the spleen and lymph nodes. There, they further
differentiate into follicular B cells (FOB) and marginal zone B
cells (MZB) (11). B-2 cells
exhibit notable antigen specificity and participate in T
cell-dependent humoral immune responses. MZB, located within the
splenic marginal sinus, serve as a first line of defense against
blood-borne pathogens. FOB, upon antigen stimulation and activation
by T follicular helper (TFH) cells, initiate germinal center
responses and ultimately differentiate into memory B cells or
plasma cells, thereby mediating long-lasting immune protection. In
addition, B-2 cells can modulate adaptive immunity through the
production of cytokines such as IFN-γ, IL-4 and TNF-α (15). In a healthy immune system, B-1 and
B-2 cells reciprocally regulate each other through cytokine
signaling and intercellular interactions, thereby maintaining
immune homeostasis (16).
Over the years, regulatory B cells (Bregs), a subset
of B cells with immunosuppressive functions, have attracted
considerable attention. Bregs can arise from various B-cell
subsets, including immature B cells, MZB, transitional 2-marginal
zone precursor B cells, B-1b cells and plasmablast-like B cells
(17–19). Bregs maintain immune tolerance and
mitigate inflammation and metabolic dysregulation by secreting
immunosuppressive cytokines, including IL-10, IL-35 and
transforming growth factor-β (TGF-β). These cytokines regulate T
helper 1/2 (Th1/Th2) differentiation, suppress the activity of
antigen-presenting cells (APCs) and promote the proliferation of
regulatory T cells (Tregs) (20,21).
Distribution of B-cell subsets in
adipose tissue under homeostasis
In healthy white adipose tissue of both mice and
humans, B-1 cells constitute the predominant B-cell population and
play a notable role in maintaining immune homeostasis within
adipose tissue. However, precise quantitative data regarding the
ratio of B-1 to B-2 cells remain limited, largely due to technical
challenges in accurately identifying and distinguishing
tissue-resident B-cell subsets. B-1a cells produce natural IgM
antibodies and anti-inflammatory cytokines, such as IL-10, which
recognize and clear apoptotic cells, oxidized lipids and other
self-antigens within adipose tissue, thereby mitigating local
inflammatory responses (9,12,22,23).
Although B-1b cells secrete lower amounts of IgM, they can
alleviate metabolic dysregulation in adipose tissue by suppressing
the pro-inflammatory responses of M1 macrophages (24,25).
In adipose tissue under homeostatic conditions, conventional
follicular B-2 cells are relatively sparse; however, certain B-2
cell subsets with functional similarities to B-1 cells contribute
to the maintenance of adipose tissue homeostasis through the
production of natural IgM (26).
Similar to FOB, MZB are also present at low frequencies in adipose
tissue, but their innate capacity for rapid responses to
blood-borne antigens enables them to participate in local immune
surveillance. In addition, a small population of Bregs with
immunosuppressive functions may exist within homeostatic adipose
tissue. By producing anti-inflammatory factors such as IL-10 and
cooperating with Tregs, these Bregs help suppress local
inflammatory responses and maintain immune equilibrium, thereby
playing a notable role in preserving white adipose tissue
homeostasis (27). However, their
precise proportion and phenotypic characteristics within
homeostatic adipose tissue remain to be elucidated (Fig. 1).
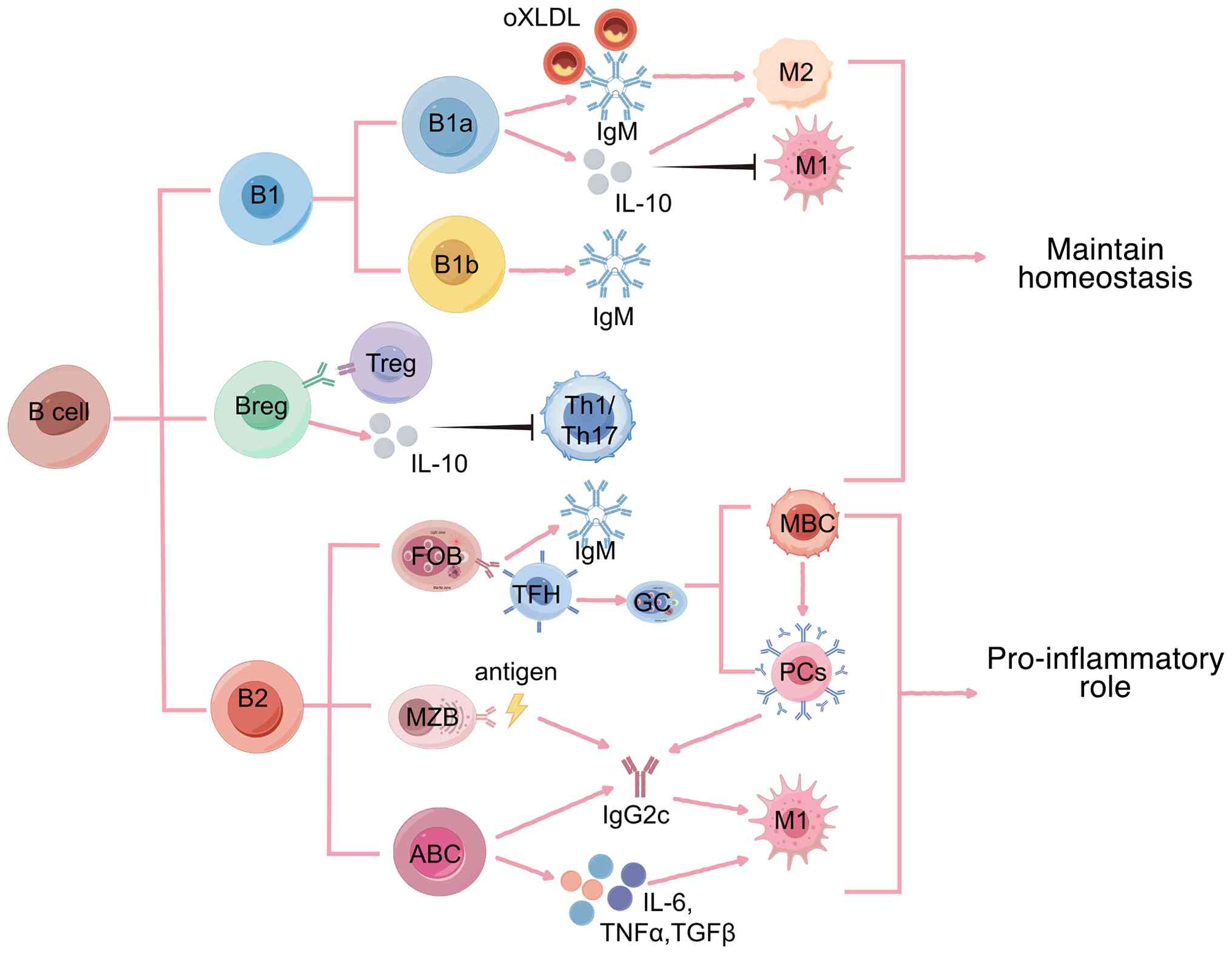 | Figure 1.Functions of B-cell subsets in
adipose tissue. Under physiological conditions, B-1 cells
constitute a major population of B cells in adipose tissue and can
be further subdivided into B-1a and B-1b subsets. B-1a cells
predominantly secrete natural IgM, which recognizes and clears
oxidized low-density lipoprotein and produce the anti-inflammatory
cytokine IL-10, promoting macrophage polarization toward the M2
phenotype while suppressing M1 activation, thereby exerting
anti-inflammatory effects. B-1b cells also mainly secrete IgM,
contributing to innate immune defense and the maintenance of tissue
homeostasis. Bregs, through IL-10 secretion and cooperation with
Tregs, suppress the proportion and effector functions of Th1/Th17
cells, thereby inhibiting inflammatory responses. Under obese
conditions, B-2 cells become the predominant B-cell population in
adipose tissue and can further differentiate into FOB, MZB and
ABCs. Upon antigen stimulation and with the help of Tfh cells, FOB
cells enter germinal centers and differentiate into MBCs and PCs,
producing IgM or undergoing class-switch recombination. MZB cells
rapidly respond to antigenic stimulation and contribute to the
production of pro-inflammatory antibodies, such as IgG2c. ABCs,
characterized by senescence-associated phenotypes and strong
pro-inflammatory features, preferentially produce autoreactive
antibodies (such as IgG2c) and secrete pro-inflammatory cytokines,
including IL-6, TNF-α and TGF-β, thereby promoting M1 macrophage
activation and amplifying adipose tissue inflammation.
Collectively, these pro-inflammatory B-cell responses drive the
development of chronic inflammation. Pink arrows indicate
stimulation or activation; black arrows indicate inhibition or
downregulation. B reg, B regulatory cell; Treg, T regulatory cell;
Th, T helper cell; FOB, follicular B cells; ABCs, age-associated B
cells; MZB, marginal zone B cells; tfh, T follicular helper; MBCs,
memory B cells; PCs, plasma cells. |
In adipose tissue, B cells are not randomly
dispersed but instead form loose aggregates through interactions
with macrophages, T cells and innate lymphoid cells (ILCs). These
structures are referred to as fat-associated lymphoid clusters
(FALCs). FALCs represent a form of tertiary lymphoid structure that
was initially identified in the mesenteric adipose tissue of both
mice and humans (28), and B cells
constitute one of the central cellular components of FALCs
(29).
Alterations and mechanisms of B-cell subsets
in obesity
Under metabolic perturbations such as obesity, the
adipose tissue microenvironment undergoes notable remodeling,
leading to the disruption of immune homeostasis. In this context,
marked alterations are observed in B-cell abundance, spatial
organization and functional phenotypes. Although accumulating
evidence suggests that obesity-associated B-cell changes are
generally characterized by enhanced pro-inflammatory activity
accompanied by impaired immunoregulatory capacity, these findings
are not entirely consistent across different experimental models,
species or adipose depots, highlighting a pronounced
context-dependent regulation. In high-fat diet (HFD)-induced obese
C57BL/6J mice, both the number and size of FALCs are markedly
increased within visceral adipose tissue (VAT) (29,30).
Concomitant with the expansion of FALCs, a substantial number of B
cells are recruited from the circulation and bone marrow into
adipose tissue, resulting in an increase in the proportion of B
cells within the stromal vascular fraction from ~10% in lean
conditions to ~20% in obesity. These recruited cells predominantly
exhibit a conventional B-2 cell phenotype (31). In addition to white adipose tissue,
an increased proportion of B cells has also been observed in murine
brown adipose tissue (BAT) under HFD conditions; however, the
precise functional roles of B cells in BAT remain largely undefined
(32). To date, studies addressing
B-cell function within BAT are notably limited, and it remains
unclear whether these cells actively participate in metabolic
regulation or merely reflect the spillover of systemic
inflammation. Consequently, this area represents a substantial gap
in current knowledge. Of note, in human adipose tissue, systematic
quantitative analyses of B-cell abundance and spatial distribution
are still lacking, which substantially constrains the direct
extrapolation of findings derived from animal models.
In VAT, B-cell accumulation occurs at an early stage
of obesity. As early as 4 weeks of HFD feeding, both B-1 and B-2
cell numbers are markedly increased in murine epididymal adipose
tissue (33). However, despite the
numerical expansion of B-1 cells, their functional protective
properties, such as the production of natural IgM, are often
attenuated in obesity settings, suggesting that the obesogenic
microenvironment may drive a state of functional exhaustion in B-1
cells. In parallel, pro-inflammatory B-2 cells and T-bet+CD21-CD23-
age-associated B cells (ABCs) progressively accumulate during
weight gain in mice and with increasing body mass index (BMI) in
humans (14,34,35).
Concomitantly, B cell-derived antibody profiles shift from
predominantly protective IgM toward pathogenic IgG isotypes. These
ABCs are hypothesized to exacerbate chronic inflammation and IR by
promoting IgG antibody production and pro-inflammatory cytokine
secretion. Nevertheless, in human studies, the definition of ABCs
remains heterogeneous, and direct evidence for their presence and
functional relevance within adipose tissue is still limited
(36).
In human studies, data regarding Breg subsets remain
relatively limited. Available evidence indicates that patients with
T2DM exhibit reduced Breg frequencies and IL-10 production in the
peripheral blood and certain tissues (37–40),
suggesting a compromised immunosuppressive capacity. However, most
of these studies are cross-sectional in nature, and can therefore
not establish whether alterations in Bregs are a cause or a
consequence of metabolic dysregulation. Furthermore, findings
derived from peripheral blood may not fully reflect the local
immune landscape within adipose tissue.
By contrast, murine studies have provided stronger
causal support for the protective role of Bregs, as the adoptive
transfer of Bregs has been shown to ameliorate adipose tissue
inflammation and IR in diet-induced obesity (DIO) models.
Mechanistically, obesity-associated chronic inflammation and the
senescence-associated secretory phenotype have been implicated in
driving B-cell polarization toward pro-inflammatory phenotypes
while suppressing Breg function through sustained activation of the
NF-κB and Janus kinase-signal transducer and activator of
transcription (JAK/STAT) signaling pathways (10,41,42).
In addition, the persistent activation of the p38 mitogen-activated
protein kinase (p38/MAPK) and protein phosphatase 2A signaling
pathways disrupts the balance between B-cell proliferation and
differentiation, thereby impairing the maintenance of Breg function
(43).
FALCs are considered notable microenvironmental
niches that support aberrant B-cell expansion and activation in
obesity. Under homeostatic conditions, M2-polarized macrophages
promote the recruitment of B-1 cells into adipose tissue through
the secretion of C-X-C motif chemokine ligand 13 (CXCL13) (44,45).
In the obese state, group 2 ILCs within mesenteric adipose
tissue-associated FALCs secrete IL-5, thereby enhancing natural IgM
production by B-1 cells and potentially exerting metabolically
protective effects. However, with the progression of obesity,
sustained increases in CXCL13, IL-7 and other stromal-derived
factors from adipocyte progenitors and fibroblasts drive excessive
B-cell recruitment and activation. Activated B cells, in turn,
reinforce chemokine production by stromal cells, establishing a
positive feedback loop between B cells and the stromal compartment
that promotes pathological FALC expansion and the maintenance of
chronic inflammation (46). In
aged mice, the reduced mRNA stability of E47 (TCF3) in visceral
adipose tissue impairs activation-induced cytidine
deaminase-mediated somatic hypermutation and class-switch
recombination, leading to defective germinal center responses and
limited antibody affinity maturation. These alterations
collectively contribute to obesity-associated inflammation and
immune dysregulation (47).
Nevertheless, most of the available evidence is derived from global
knockout or pharmacological intervention models, making it
difficult to disentangle B-cell-intrinsic effects from systemic
inflammatory changes.
Inflammatory cytokines exert a ‘double-edged sword’
effect on B-cell chemotaxis and functional remodeling. In obese
mice, adipocytes within VAT secrete a range of chemokines,
including CXCL10, C-C motif chemokine ligand 2 (CCL2) and CCL5,
which preferentially recruit B-2 cells through their corresponding
receptors, such as C-C motif chemokine receptor 2 (CCR2) and CCR3
(11,48). In HFD mice, the lipid-derived
inflammatory mediator leukotriene B4 promotes the recruitment of
B-2 cells into adipose tissue via leukotriene B4 receptor 1 and
directly induces a pro-inflammatory B-cell phenotype. This, in
turn, exacerbates IR through the activation of CD4+ and CD8+ T
cells as well as M1-polarized macrophages (31). IL-6 and TNF-α, produced by both
adipocytes and B cells, are widely recognized as key drivers of
pro-inflammatory B-cell polarization (15,49).
Of note, IL-6 exhibits context-dependent effects, as it has also
been shown to improve lipid metabolism and insulin sensitivity in
murine models (50,51). In addition, the IL-6 family member
cardiotrophin-like cytokine factor 1 is upregulated in the BAT of
obese mice and suppresses mitochondrial biogenesis and thermogenic
capacity by activating STAT3 and inhibiting PGC-1α/β transcription,
thereby promoting BAT ‘whitening’, as indicated by adipocyte
hypertrophy, lipid droplet accumulation, uncoupling protein 1
(UCP-1) suppression, and mitochondrial dysfunction, initiates and
accelerates obesity progression (52). In patients with T2DM, TNF-α acts as
a major driver of adipose tissue inflammation by inducing immune
cell polarization and suppressing regulatory immune functions.
Furthermore, TNF-α enhances lipolysis and increases free fatty acid
release, further aggravating IR (53).
Adipocytes not only serve as energy storage units,
but also function as endocrine cells, secreting a variety of
adipokines that play notable regulatory roles in B-cell biology. In
obese individuals, leptin, a key adipocyte-derived hormone, is
positively associated with BMI. B cells express leptin receptor
(LEPR/Ob-Rb) on their surface, and leptin binding activates B cells
from both young and elderly peripheral blood, inducing the
secretion of pro-inflammatory cytokines, including IL-6 and TNF-α,
primarily through the activation of the JAK2/STAT3 and p38
MAPK/ERK1/2 signaling pathways (54,55).
In addition, in obese individuals, leptin secreted by adipose
tissue mediates the metabolic reprogramming of B cells through the
activation of the mechanistic target of rapamycin complex 1
pathway, enhancing glycolytic and biosynthetic activity and driving
differentiation toward a pro-inflammatory phenotype. This is
characterized by the upregulation of CD25 and human leukocyte
antigen DR expression and increased secretion of IL-6 and TNF-α
(56). Clinical studies have
indicated that adiponectin levels are reduced in obese individuals,
and that exerts anti-inflammatory effects by inhibiting
AMP-activated protein kinase signaling, thereby enhancing signal
transduction and STAT3 activity to promote IL-10 transcription
(57–59). Furthermore, adiponectin can induce
B1 cells to secrete the peptide inhibitor of transendothelial
migration, derived from the 14-3-3ζδ protein, which modulates
endothelial adhesion molecules and sphingosine-1-phosphate
signaling, thereby limiting T-cell trafficking and inflammatory
responses (60,61). In HFD-fed mice, adiponectin
indirectly suppresses the pro-inflammatory phenotype of B cells
through multilayered immunoregulatory mechanisms. On one hand,
adiponectin upregulates Sirtuin 1 and peroxisome
proliferator-activated receptor γ whereas inhibiting the Th17
lineage-specifying transcription factor retinoic acid-related
orphan receptor γt markedly suppressing Th1 and Th17
differentiation and pro-inflammatory cytokine production, and
concurrently promoting FoxP3 expression; this leads to an expansion
of the number and function of Tregs, which shifts immune responses
toward tolerance. On the other hand, adiponectin remodels dendritic
cell phenotypes by downregulating major histocompatibility complex
(MHC) class II and costimulatory molecules CD80/CD86, suppressing
the secretion of pro-inflammatory cytokines such as IL-12, and
upregulating inhibitory molecules such as programmed cell death
ligand 1 (PD-L1). This attenuates the dendritic cell-mediated
activation of effector T cells while promoting Treg induction.
Within this immunoregulatory environment, Treg-mediated suppression
and reduced pro-inflammatory T-cell responses collectively
constrain aberrant B-cell activation and pro-inflammatory function,
thereby indirectly limiting B-cell inflammatory phenotypes
(62–64). Furthermore, obesity-associated
hyperglycemia, cell death and lipid spillover can activate
antigen-specific B cells, leading to their expansion and pathogenic
antibody production. In db/db mice, elevated glucose concentrations
inhibit IgM secretion by B-1 cells and promote apoptosis (65). Free fatty acids, such as palmitate,
enhance B-cell IL-10 secretion and survival in adipose tissue via
Toll-like receptor 4 (TLR4) signaling, indicating that
tissue-derived metabolic cues regulate the fate of B-cell (21,38).
The ‘gut-adipose axis’ also plays a notable role in
B-cell immunoregulation (66).
High-fat diets alter the gut microbiota in humans and mice,
compromise intestinal barrier integrity and lead to endotoxemia,
which activates adipose tissue B cells and induces the expansion of
IL-10+ Breg cells (57,67). In DIO mice, gut-derived
lipopolysaccharide (LPS) activates the TLR4-myeloid differentiation
primary response 88-NF-κB signaling pathway, inducing the high
expression of pro-inflammatory cytokines such as TNF-α and IL-6,
thereby amplifying B-cell–mediated inflammatory responses. Although
fatty acids themselves are not direct TLR4 ligands, they can
indirectly activate this pathway by promoting LPS translocation or
altering cellular metabolism, exacerbating B-cell inflammatory
activation (68–70). Peritoneal B-1a cells aberrantly
activated by commensal bacteria promote insulin resistance (IR) by
enhancing systemic and adipose tissue inflammation, facilitating
macrophage polarization toward the pro-inflammatory M1 phenotype,
and impairing insulin signaling through sustained activation of
inflammatory pathways, including NF-κB, JNK and p38 MAPK; following
bariatric surgery, the gut microbiota shifts toward a lean
phenotype, characterized by reduced adiposity, improved insulin
sensitivity, normalized glucose metabolism, and attenuated
inflammatory responses, thereby restoring adipose tissue B-cell
function (58,71). Obesity also reduces the number of
intestinal IgA+ B cells and decreases secretory IgA levels
(66), IgA deficiency accelerates
B-cell senescence, leading to impaired B-cell function and
increased autoantibody production, further aggravating glucose
intolerance and reducing insulin sensitivity. In addition, in
obesity settings, elevated 25-hydroxycholesterol levels in Peyer's
patches inhibit antigen-specific IgA+ B-cell differentiation,
whereas cholesterol-25-hydroxylase deficiency alleviates this
effect (72) (Fig. 2).
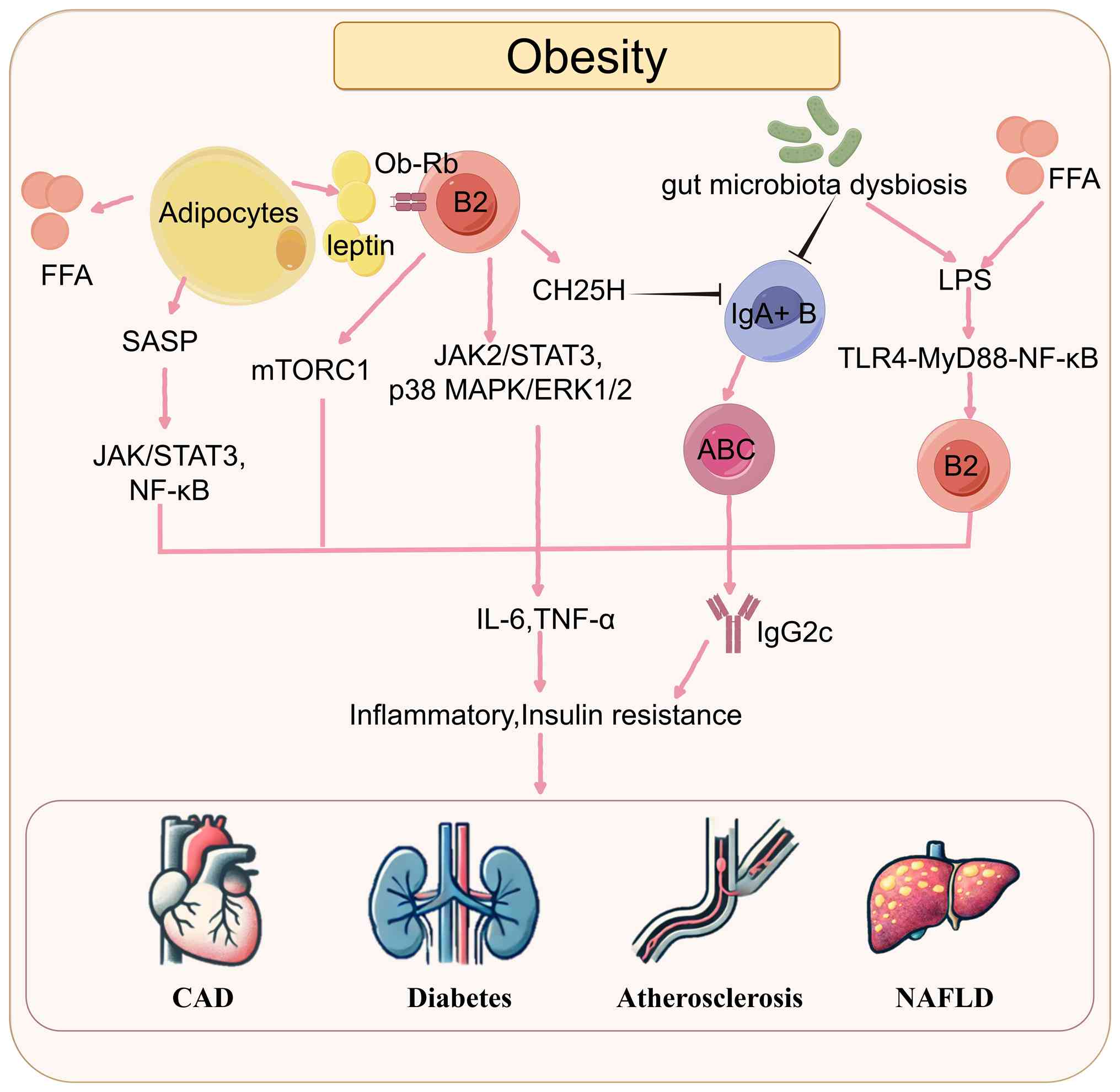 | Figure 2.Adipose tissue microenvironment
promotes B-cell functional reprogramming and regulates metabolic
inflammation. Under obese conditions, enhanced lipolysis in
adipocytes leads to elevated levels of FFAs, accompanied by
increased leptin secretion and the formation of SASP. Leptin acts
on B-2 cells via its receptor Ob-Rb, activating mTORC1 as well as
the JAK2/STAT3 and p38 MAPK/ERK1/2 signaling pathways, thereby
promoting pro-inflammatory activation of B-2 cells. SASP
simultaneously activates the JAK/STAT3 and NF-κB pathways,
amplifying inflammatory signaling. Moreover, obesity-associated gut
dysbiosis and high FFA levels increase LPS levels in adipose
tissue. LPS, through the TLR4-MyD88-NF-κB signaling axis, promotes
B-2 cell expansion and inflammatory responses. At the same time,
B-2 cells increase the synthesis and secretion of CH25H, which
inhibits the differentiation and maintenance of IgA+ B cells,
further impairing the intestinal IgA barrier. The reduction in IgA+
B cells also promotes B-cell senescence, increasing the proportion
of ABCs and the secretion of IgG2c antibodies, thereby exacerbating
inflammation. These converging signaling pathways collectively
elevate levels of pro-inflammatory cytokines such as IL-6 and
TNF-α, inducing chronic adipose tissue inflammation and aggravating
insulin resistance, ultimately contributing to the development of
CAD, diabetes, atherosclerosis and NAFLD. Pink arrows indicate
stimulation or activation; black arrows indicate inhibition or
downregulation. FFA, free fatty acid; LPS, lipopolysaccharide;
ABCs, associated B cells; NAFLD, non-alcoholic fatty liver disease;
CAD, coronary artery disease; SASP, senescence-associated secretory
phenotype. |
Collectively, current evidence supports that obesity
drives B-cell dysfunction through the synergistic actions of
inflammatory cytokines, adipokines, metabolic products and gut
microbiota, thereby promoting chronic adipose tissue inflammation
and metabolic dysregulation. However, most mechanistic conclusions
are primarily based on murine models, and the temporal
relationships, causality and tissue specificity in humans remain
incompletely elucidated. Further integrative multi-omics and
functional validation studies are warranted to address these
knowledge gaps.
Roles and mechanisms of B cells in
obesity-related metabolic diseases
In obesity-related metabolic diseases, adipose
tissue B cells are increasingly recognized as a notable regulatory
node linking immune inflammation and metabolic dysregulation.
Accumulating evidence indicates that, under obese conditions,
aberrantly activated B cells engage in complex interactions with
adipocytes, T cells and macrophages through antigen presentation,
cytokine secretion and antibody production, thereby exacerbating
adipose tissue inflammation and promoting IR (10). Clinical studies have shown that
patients with T2DM exhibit increased B-cell activation in
peripheral blood, accompanied by elevated secretion of inflammatory
mediators such as IL-8, which is closely associated with chronic
inflammatory responses (39,73).
In aged mice, the local depletion of VAT-resident B cells markedly
improves insulin sensitivity (5).
Under HFD conditions, the deletion of the B cell-specific Oct
co-activator from B cells, a key regulator of B-cell development,
notably ameliorates adipose tissue inflammation and IR in mice
(74,75). Similarly, the deletion of the
inhibitor of DNA binding 3 gene increases B-1 cell abundance in
white adipose tissue, leading to reduced adipose inflammation and
improved glucose tolerance (24).
Adoptive transfer of B-1 cells into HFD-fed B cell-deficient obese
mice also alleviates VAT inflammation and improves IR (12). Collectively, these findings provide
direct evidence of the pathogenic role of adipose tissue B cells in
metabolic homeostasis disruption; however, the applicability of
these strategies in humans remains to be determined.
Studies have further demonstrated that distinct
B-cell subsets exert divergent effects on adipose tissue and
cardiovascular homeostasis. In HFD-fed apolipoprotein E (Apoe)-/-
mice, B-1a and B-1b cells secrete natural IgM antibodies in the
spleen and adipose tissue to neutralize oxidized low-density
lipoprotein (oxLDL) and promote macrophage polarization toward an
anti-inflammatory phenotype, thereby suppressing myocardial
inflammation and atherosclerosis. By contrast, B-2 cells secrete
TNF-α, IL-6 and TGF-β, activate cardiac fibroblasts and promote
collagen deposition and ventricular remodeling (11,76).
In addition, B cells can modulate T-cell responses by promoting
pro-atherogenic Th1 immunity while suppressing anti-atherogenic
interleukin-17 production, thereby accelerating atherosclerosis
progression (77). Clinical and
experimental studies have also revealed the presence of large
B-cell clusters in epicardial adipose tissue of patients with
coronary artery disease (78). Of
note, as early as 3 days after myocardial infarction, B-cell
numbers are markedly increased in murine pericardial adipose
tissue, with no notable changes observed in the myocardium itself,
suggesting that local B-cell proliferation is the predominant
source. These B cells secrete granulocyte-macrophage
colony-stimulating factor (GM-CSF), which promotes the recruitment
and maintenance of dendritic cells in pericardial adipose tissue
and accelerates post-infarction fibrosis (79).
Adipose tissue B cells further contribute to
metabolic dysfunction by releasing pathogenic IgG antibodies and
inflammatory mediators, thereby promoting monocyte infiltration
into FALCs and sustaining the release of pro-inflammatory factors,
ultimately leading to adipose tissue dysfunction and metabolic
imbalance (11,80). In obese mice, B-2 cell infiltration
into mesenteric adipose tissue precedes that of T cells and
macrophages (81). In HFD-induced
obese mice, activated B cells produce large amounts of
pro-inflammatory cytokines, including TNF-α and GM-CSF, which
recruit macrophages and promote their differentiation toward an
inflammatory phenotype, thereby inducing IR (33,82).
During obesity, B cells within FALCs become activated and function
as APCs, presenting internalized lipids or other antigens to TFH
cells. TFH cells subsequently provide key costimulatory signals,
such as CD40L and IL-21, thereby promoting B-cell differentiation
into plasma cells and driving the production of pro-inflammatory
Immunoglobulin G2c (IgG2c) antibodies. These IgG2c antibodies
accumulate within adipocyte crown-like structures, where they form
immune complexes with oxidatively modified lipoproteins (such as
oxLDL) or adipose tissue-derived self-antigens. The resulting
immune complexes activate macrophages through Fc γ receptor
receptors, thereby amplifying adipose tissue inflammation. Of note,
the adoptive transfer of IgG isolated from DIO mice into B
cell-deficient mice is sufficient to induce macrophage polarization
toward the M1 phenotype, leading to the increased production of
pro-inflammatory mediators (33,39,83).
Furthermore, B cell-derived IgG can drive Th1/Th2 polarization
through MHC-dependent mechanisms, leading to the secretion of TNF-α
and IFN-γ, which further stimulate B cells and establish a positive
feedback loop that exacerbates IR and adipose tissue inflammation
(84).
In metabolic diseases such as obesity and diabetes,
both the number and function of Bregs are markedly reduced,
resulting in decreased IL-10 production, impaired anti-inflammatory
capacity, increased release of pro-inflammatory factors and
exacerbation of adipose tissue inflammation and IR (85). By contrast, in autoimmune diseases
and cancer, B-1 and Breg cells can exert anti-inflammatory effects
by expressing immune checkpoint molecules, such as programmed cell
death protein 1 (PD-1), programmed death-ligand 1 (PD-L1) and
CD39/CD73, and by secreting IgM antibodies and IL-10 (86). Of note, infection with
Schistosoma japonicum induces a marked upregulation of IL-10
production in CD19+CD9+ B cells, promoting the expansion of Tregs
and Th2 cells, while reducing Th1 and Th17 responses. This immune
shift alleviates inflammation and improves insulin sensitivity in
HFD-fed mice, suggesting that enhancing CD9+ B cells or
IL-10-producing Breg function may represent a novel strategy for
modulating obesity-associated inflammation and metabolic
dysregulation (87,88). Consistently, studies have
demonstrated that in HFD-fed mice and patients with T2DM, IL-10
suppresses pro-inflammatory cytokine production through the
activation of the STAT3 signaling pathway, promotes Treg
differentiation, induces macrophage polarization toward the M2
phenotype, protects adipose tissue from inflammation and
counteracts IR (23,89,90).
In summary, B cells exert dual pro- and
anti-inflammatory roles in obesity-related metabolic diseases.
While pathogenic B-cell responses exacerbate inflammation and
metabolic dysfunction, protective B-cell subsets contribute to the
maintenance of metabolic homeostasis. These findings provide a
theoretical basis for the development of B cell-targeted
therapeutic strategies in metabolic diseases (91,92).
Targeting B cells as a therapeutic strategy
in obesity and obesity-related metabolic diseases
Direct B-cell-depleting therapeutic strategies have
been widely applied in the treatment of B-cell non-Hodgkin lymphoma
and autoimmune diseases such as rheumatoid arthritis (93). Given the pivotal role of B cells in
obesity-associated chronic inflammation and metabolic
dysregulation, B-cell-targeted interventions are increasingly
recognized as a potential metabolic therapeutic strategy. Although
current evidence is largely derived from animal models and studies
conducted in other disease contexts, these findings provide an
important theoretical foundation for the application of
B-cell-targeted therapies in obesity-related metabolic
disorders.
CD20-mediated B-cell depletion
Rituximab is the first clinically approved anti-CD20
monoclonal antibody and selectively depletes mature B cells while
sparing plasma cells and B-cell precursors (94). In murine models of obesity,
CD20-mediated B-cell depletion predominantly affects
pro-inflammatory B-2 cells, whereas B-1 cells are relatively
preserved. This intervention also reduces C-C motif chemokine
ligand 7-mediated monocyte recruitment, thereby attenuating tissue
inflammation, highlighting its potential therapeutic value in
obesity-associated metabolic dysfunction (77,95).
In human studies, although CD20-targeted therapies have not been
directly applied to obesity or metabolic diseases, multiple
preclinical investigations have demonstrated that this strategy
markedly reduces B-2 cell numbers and delays the progression of
atherosclerosis. The underlying mechanisms may involve the
suppression of B-cell-dependent monocyte recruitment to the
vasculature or enhancement of immunosuppressive dendritic cell
populations and Tregs (96,97).
These findings suggested that selectively depleting
pro-inflammatory B-2 cells while preserving B-cell subsets with
immunoregulatory functions may represent a key advantage of
B-cell-targeted therapies. However, systemic B-cell depletion is
known to be associated with adverse effects, including increased
infection risk and hypogammaglobulinemia, in the treatment of
autoimmune diseases and lymphomas (98,99).
When considered for chronic metabolic diseases, long-term safety
profiles constitute a primary concern. Furthermore, non-selective
depletion of most B-2 cells may also compromise beneficial Breg
populations, potentially leading to immune homeostasis imbalance
(100,101).
Targeting the B-cell activating factor
(BAFF)/a proliferation-inducing ligand (APRIL) system
BAFF, a member of the TNF ligand family, is a
notable cytokine required for B-cell survival and maturation. Under
metabolic dysregulation conditions such as obesity and IR, BAFF can
be secreted by multiple immune cell types (102). Among BAFF-related pathways, the
BAFF/APRIL system plays a central role in the survival and
proliferation of B-2 cells. Kim et al (103) and Sanchez et al (104) demonstrated that circulating BAFF
levels are positively associated with IR and endothelial
dysfunction in obese individuals, providing clinical evidence for
its pathogenic role in metabolic diseases. In systemic lupus
erythematosus (SLE) mouse models, excessive BAFF drives the
expansion of inflammatory B-2 cells and synergizes with B-cell
receptor and TLR signaling pathways to amplify B-cell responses,
thereby promoting the production of pathogenic antibodies and
pro-inflammatory cytokines such as IL-6 and TNF-α (102,105). Furthermore, the primary
BAFF-shedding enzyme A disintegrin and metalloproteinase 17 is
markedly activated in Apoe-/- mice fed an atherogenic diet, further
enhancing BAFF-associated pro-inflammatory signaling (106). Nevertheless, BAFF also exhibits
functional duality in metabolic diseases; some studies have shown
that in mice fed a HFD for 8 weeks, decreased BAFF levels are
accompanied by increased adipose tissue inflammation, suggesting
that BAFF may exert protective effects during early disease stages
by maintaining Breg survival and immune tolerance (38,102,105). Therefore, the role of BAFF
appears to be both stage- and dose-dependent.
Based on these mechanisms, treatment with
BAFF-neutralizing monoclonal antibodies (such as single-stranded
DNA-binding protein 2) and BAFF receptor (BAFFR) has been shown to
reduce overall B-cell numbers and selectively deplete B-2 and
plasma cells. Belimumab, a monoclonal antibody targeting BAFF, is
the first drug approved in nearly 60 years for the treatment of SLE
(107). Other agents targeting
the BAFF/APRIL system, including the BAFF inhibitor blisibimod, the
BAFFR-targeting antibody ianalumab and the fusion protein
atacicept, composed of the Ig fragment of transmembrane activator
and CAML interactor (TACI) and capable of binding both BAFF and
APRIL (108), have not yet
entered routine clinical use. Of note, the BAFF/APRIL system is
essential for B-cell homeostasis, primarily through its
interactions with BAFF-R, TACI and BCMA, which collectively
regulate B-cell survival, differentiation and antibody production.
BAFF-R signaling is critical for the maintenance of transitional
and mature naïve B cells, whereas TACI and BCMA control humoral
immune responses, immunoglobulin class-switch recombination and
long-lived plasma cell survival. These effects are mainly mediated
by activation of the non-canonical NF-κB2 pathway, together with
PI3K/Akt and MAPK signaling cascades. Moreover, BAFF availability
serves as a key checkpoint during B-cell development and peripheral
selection, thereby maintaining immune homeostasis (108,109). BAFF may also exert
B-cell-independent anti-inflammatory effects through binding to
TACI expressed on myeloid cells, and its long-term inhibition may
impair humoral immune defense and increase susceptibility to
infections (106). Animal studies
have shown that BAFF deficiency can exacerbate atherosclerosis by
disrupting TACI signaling in myeloid cells, which activates
pro-inflammatory pathways (TLR9-IRF7), increases CXCL10 and IFIT2
expression, and promotes both local plaque inflammation and
systemic immune activation, leading to larger, less stable plaques
(106). Therefore, the long-term
efficacy and safety of targeting the BAFF/APRIL pathway in chronic
metabolic diseases must be carefully evaluated. The development of
tissue-specific or subset-selective intervention strategies may
represent a safer therapeutic direction.
Exploratory strategies targeting
costimulatory signals and B-cell functional regulation
The CD40-CD40L pathway represents an notable
costimulatory signal required for full B-cell activation. Its
blockade suppresses pathological interactions between B cells and T
cells, thereby reducing aberrant antibody production and
inflammatory amplification loops (110). Under HFD conditions, the genetic
deficiency of CD40L attenuates diet-induced obesity, hepatic
steatosis and systemic IR in mice (111). In studies on atherosclerosis,
preclinical evidence from CD40- or CD40L-deficient models generally
supports a pro-atherogenic role of this signaling axis. However,
despite the widely accepted view that T-cell-dependent and
CD40-dependent B-cell responses promote the progression of
atherosclerosis, elevated levels of circulating CD40+ B cells have
been associated with a reduced risk for stroke. This seemingly
paradoxical observation is thought to be associated with the marked
role of CD40 signaling in Breg differentiation (112–114). It should be noted that CD40
signaling plays a central regulatory role in multiple immune
functions, and systemic inhibition may compromise host immune
defense, weaken vaccine responses and increase susceptibility to
infections (111). Consequently,
any therapeutic intervention targeting CD40 must be approached with
caution.
Aberrant B-cell activation in obesity is also
extensively regulated by metabolic and inflammatory
microenvironments (10). Studies
have shown that the adoptive transfer of T-bet+ B cells exacerbates
metabolic dysfunction in obese mice, whereas the B-cell-specific
deletion of T-box transcription Factor 21 reduces serum IgG2c
levels, inflammatory cytokine production and inflammatory
macrophage accumulation in adipose tissue, thereby alleviating
metabolic abnormalities (35). In
addition, mice with B-cell-specific Tlr9 deficiency exhibit
increased adipose tissue inflammation, weight gain and impaired
glucose and insulin tolerance (115). These findings indicated that
selectively targeting T-bet+ B cells or modulating TLR9 signaling
pathways may offer therapeutic benefits for improving
obesity-associated metabolic homeostasis in animal models.
Overall, multiple B-cell-targeted strategies have
demonstrated potential value in ameliorating inflammation and
metabolic abnormalities in animal models and related disease
contexts. However, with the exception of a limited number of murine
studies (33,116), robust clinical evidence
supporting the efficacy of B-cell depletion or costimulatory
molecule-targeted therapies in obesity-induced type II diabetes and
IR is lacking. Future studies using disease models more closely
related to human pathology and well-designed prospective clinical
trials are required to systematically evaluate long-term efficacy,
safety and appropriate patient populations.
Summary and perspectives
Adipose tissue B cells have emerged as key drivers
of obesity-associated metabolic diseases, shifting from protective
regulators under homeostatic conditions to pathogenic ‘disruptors’
in the obese state. Through both antibody-dependent and
antibody-independent mechanisms, B cells promote chronic adipose
tissue inflammation and metabolic dysregulation. Targeting B cells,
particularly specific pathogenic subsets or functional programs,
therefore represents a promising therapeutic avenue for the
treatment of obesity and its related complications.
Future studies should prioritize the establishment
of comprehensive human adipose tissue B-cell atlases, coupled with
an in-depth characterization of the heterogeneity of adipose tissue
B-cell subsets. Elucidating the molecular mechanisms through which
distinct B-cell populations contribute to obesity-associated
metabolic diseases will be essential for the precise targeting of
pathogenic B-cell subsets, such as ABCs, while preserving
protective populations including B-1 cells and Bregs. In parallel,
efforts should be directed toward accelerating the clinical
translation of these insights.
In conclusion, as notable regulators within the
adipose tissue immune microenvironment, B cells play a central role
in shaping obesity-related inflammatory responses. A deeper
understanding of their functional diversity not only advances our
mechanistic insight into obesity-associated inflammation but also
provides an important theoretical foundation and practical guidance
for the development of more precise and safer immunometabolic
therapeutic strategies.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National Natural Science
Foundation of China (grant nos. 82000525 and 81873883), Science and
Technology Support Plan for Youth Innovation of Colleges and
Universities of Shandong Province of China (grant no. 2021KJ106)
and Shandong Provincial Natural Science Foundation, China (grant
no. ZR2023QH248).
Availability of data and materials
Not applicable.
Authors' contributions
HW and HL contributed to the writing of the original
draft and manuscript design. XZ participated in the literature
search, analysis and manuscript design. LW and ML were responsible
for reviewing and editing the manuscript, as well as funding
acquisition. All authors read and approved the final version of the
manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chakarov S, Bleriot C and Ginhoux F: Role
of adipose tissue macrophages in Obesity-related disorders. J Exp
Med. 219:e202119482022. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bluher M: Obesity: Global epidemiology and
pathogenesis. Nat Rev Endocrinol. 15:288–298. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Koenen M, Hill MA, Cohen P and Sowers JR:
Obesity, adipose tissue and vascular dysfunction. Circ Res.
128:951–68. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bapat SP, Myoung Suh J, Fang S, Liu S,
Zhang Y, Cheng A, Zhou C, Liang Y, Leblanc M, Liddle C, et al:
Depletion of fat-resident Treg cells prevents age-associated
insulin resistance. Nature. 528:137–141. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Camell CD, Gunther P, Lee A, Goldberg EL,
Spadaro O, Youm YH, Bartke A, Hubbard GB, Ikeno Y, Ruddle N H, et
al: Aging Induces an Nlrp3 Inflammasome-dependent expansion of
Adipose B cells that impairs metabolic homeostasis. Cell Metab.
30:1024–39.e6. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chavakis T, Alexaki VI and Ferrante AW Jr:
Macrophage function in adipose tissue homeostasis and metabolic
inflammation. Nat Immunol. 24:757–766. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Schaum N, Lehallier B, Hahn O, Palovics R,
Hosseinzadeh S, Lee SE, Sit R, Lee DP, Losada PM, Zardeneta ME, et
al: Ageing hallmarks exhibit organ-specific temporal signatures.
Nature. 583:596–602. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fang W, Deng Z, Benadjaoud F, Yang D, Yang
C and Shi GP: Regulatory T cells promote adipocyte beiging in
subcutaneous adipose tissue. Faseb J. 34:9755–9770. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Altintas MM, Azad A, Nayer B, Contreras G,
Zaias J, Faul C, Reiser J and Nayer A: Mast cells, macrophages, and
crown-like structures distinguish subcutaneous from visceral fat in
mice. J Lipid Res. 52:480–488. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Frasca D, Garcia D, Diaz A, Romero M,
Thaller S and Blomberg BB: Phenotypic and functional features of B
cells from two different human subcutaneous adipose depots. PLoS
One. 18:e02850252023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Meher AK and Mcnamara CA: B-1 lymphocytes
in adipose tissue as innate modulators of inflammation linked to
cardiometabolic disease. Immunol Rev. 324:95–103. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shen L, Chng MH, Alonso MN, Yuan R, Winer
DA and Engleman EG: B-1a lymphocytes attenuate insulin resistance.
Diabetes. 64:593–603. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Melchers F: Checkpoints that control B
cell development. J Clin Invest. 125:2203–2210. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Srikakulapu P and Mcnamara CA: B
lymphocytes and adipose tissue inflammation. Arterioscler Thromb
Vasc Biol. 40:1110–1122. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Menendez A, Wanczyk H, Walker J, Zhou B,
Santos M and Finck C: Obesity and adipose tissue dysfunction: From
pediatrics to adults. Genes (Basel). 13:18662022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Borocz K, Szinger D, Simon D, Berki T and
Nemeth P: Regulators and conductors of immunity: Natural immune
system in health and autoimmunity. Int J Mol Sci. 26:54132025.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Banko Z, Pozsgay J, Szili D, Toth M, Gati
T, Nagy G, Rojkovich B and Sarmay G: Induction and differentiation
of IL-10-producing regulatory B cells from healthy blood donors and
rheumatoid arthritis patients. J Immunol. 198:1512–1520. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chekol Abebe E, Asmamaw Dejenie T, Mengie
Ayele T, Dagnew Baye N, Agegnehu Teshome A and Tilahun Muche Z: The
role of regulatory B cells in health and diseases: A systemic
review. J Inflamm Res. 14:75–84. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yanaba K, Bouaziz JD, Matsushita T,
Tsubata T and Tedder TF: The development and function of regulatory
B cells expressing IL-10 (B10 cells) requires antigen receptor
diversity and TLR signals. J Immunol. 182:7459–7472. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hieronimus L and Huaux F: B-1 cells in
immunotoxicology: Mechanisms underlying their response to chemicals
and particles. Front Toxicol. 5:9608612023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yanaba K, Bouaziz JD, Haas KM, Poe JC,
Fujimoto M and Tedder TF: A regulatory B cell subset with a unique
CD1dhiCD5+ phenotype controls T cell-dependent inflammatory
responses. Immunity. 28:639–650. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Soriano FG, Barbeiro HV and Barbeiro DF:
Inflammatory response: Role of B1 cells. Shock. 39 (Suppl 1):S5–S9.
2013. View Article : Google Scholar
|
|
23
|
Wong SC, Puaux AL, Chittezhath M, Shalova
I, Kajiji TS, Wang X, Abastado JP, Lam KP and Biswas SK: Macrophage
polarization to a unique phenotype driven by B cells. Eur J
Immunol. 40:2296–2307. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Harmon DB, Srikakulapu P, Kaplan JL,
Oldham SN, Mcskimmin GC, Garmey JC, Perry HM, Kirby JL, Prohaska
TA, Gonen A, et al: Protective role for B-1b B cells and IgM in
Obesity-associated inflammation, glucose intolerance, and insulin
resistance. Arterioscler Thromb Vasc Biol. 36:682–691. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Srikakulapu P, Pattarabanjird T, Upadhye
A, Bontha SV, Osinski V, Marshall MA, Garmey J, Deroissart J,
Prohaska TA, Witztum JL, et al: B-1b cells have unique functional
traits compared to B-1a cells at homeostasis and in aged
hyperlipidemic mice with atherosclerosis. Front Immunol.
13:9094752022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Reynolds AE, Kuraoka M and Kelsoe G:
Natural IgM is produced by CD5-plasma cells that occupy a distinct
survival niche in bone marrow. J Immunol. 194:231–242. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Agrawal M, Kern PA and Nikolajczyk BS: The
immune system in obesity: Developing paradigms amidst inconvenient
truths. Curr Diab Rep. 17:872017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Moro K, Yamada T, Tanabe M, Takeuchi T,
Ikawa T, Kawamoto H, Furusawa J, Ohtani M, Fujii H and Koyasu S:
Innate production of T(H)2 cytokines by adipose tissue-associated
c-Kit(+)Sca-1(+) lymphoid cells. Nature. 463:540–544. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Stutte S, Ishikawa-Ankerhold H, Lynch L,
Eickhoff S, Nasiscionyte S, Guo C, Van Den Heuvel D, Setzensack D,
Colonna M, Maier-Begandt D, et al: High-fat diet rapidly modifies
trafficking, phenotype, and function of plasmacytoid dendritic
cells in adipose tissue. J Immunol. 208:1445–1455. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mcdonnell ME, Ganley-Leal LM, Mehta A,
Bigornia SJ, Mott M, Rehman Q, Farb MG, Hess DT, Joseph L, Gokce N,
et al: B lymphocytes in human subcutaneous adipose crown-like
structures. Obesity (Silver Spring). 20:1372–1378. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ying W, Wollam J, Ofrecio JM,
Bandyopadhyay G, El Ouarrat D, Lee YS, Oh DY, Li P, Osborn O and
Olefsky JM: Adipose tissue B2 cells promote insulin resistance
through leukotriene LTB4/LTB4R1 signaling. J Clin Invest.
127:1019–1030. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Peterson KR, Flaherty DK and Hasty AH:
Obesity Alters B cell and macrophage populations in brown adipose
tissue. Obesity (Silver Spring). 25:1881–1884. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Winer DA, Winer S, Shen L, Wadia PP,
Yantha J, Paltser G, Tsui H, Wu P, Davidson MG, Alonso MN, et al: B
cells promote insulin resistance through modulation of T cells and
production of pathogenic IgG antibodies. Nat Med. 17:610–617. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Cancro MP: Age-Associated B cells. Annu
Rev Immunol. 38:315–340. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hagglof T, Vanz C, Kumagai A, Dudley E,
Ortega V, Siller M, Parthasarathy R, Keegan J, Koenigs A, Shute T
and Leadbetter EA: T-bet+ B cells accumulate in adipose tissue and
exacerbate metabolic disorder during obesity. Cell Metab.
34:1121–1136.e6. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sachinidis A, Xanthopoulos K and
Garyfallos A: Age-Associated B cells (ABCs) in the prognosis,
diagnosis and therapy of systemic lupus erythematosus (SLE).
Mediterr J Rheumatol. 31:311–318. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Garcia-Hernandez MH, Rodriguez-Varela E,
Garcia-Jacobo RE, Hernandez-De La Torre M, Uresti-Rivera EE,
Gonzalez-Amaro R and Portales-Perez DP: Frequency of regulatory B
cells in adipose tissue and peripheral blood from individuals with
overweight, obesity and normal-weight. Obes Res Clin Pract.
12:513–519. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nishimura S, Manabe I, Takaki S, Nagasaki
M, Otsu M, Yamashita H, Sugita J, Yoshimura K, Eto K, Komuro I, et
al: Adipose Natural Regulatory B Cells Negatively Control Adipose
Tissue Inflammation. Cell Metab. 18:759–766. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Jagannathan M, Mcdonnell M, Liang Y,
Hasturk H, Hetzel J, Rubin D, Kantarci A, Van Dyke TE, Ganley-Leal
LM and Nikolajczyk BS: Toll-like receptors regulate B cell cytokine
production in patients with diabetes. Diabetologia. 53:1461–1471.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhai X, Qian G, Wang Y, Chen X, Lu J,
Zhang Y, Huang Q and Wang Q: Elevated B cell activation is
associated with type 2 diabetes development in obese subjects. Cell
Physiol Biochem. 38:1257–1266. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Rosser EC and Mauri C: The emerging field
of regulatory B cell immunometabolism. Cell Metab. 33:1088–1097.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Frasca D, Romero M, Diaz A, Garcia D,
Thaller S and Blomberg BB: B cells with a Senescent-associated
secretory phenotype accumulate in the adipose tissue of individuals
with obesity. Int J Mol Sci. 22:18392021. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Frasca D, Romero M, Landin AM, Diaz A,
Riley RL and Blomberg BB: Protein phosphatase 2A (PP2A) is
increased in old murine B cells and mediates p38
MAPK/tristetraprolin dephosphorylation and E47 mRNA instability.
Mech Ageing Dev. 131:306–314. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ansel KM, Harris RB and Cyster JG: CXCL13
is required for B1 cell homing, natural antibody production, and
body cavity immunity. Immunity. 16:67–76. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Mancuso P: The role of adipokines in
chronic inflammation. Immunotargets Ther. 5:47–56. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Jackson-Jones LH, Duncan SM, Magalhaes MS,
Campbell SM, Maizels RM, Mcsorley HJ, Allen JE and Benezech C:
Fat-associated lymphoid clusters control local IgM secretion during
pleural infection and lung inflammation. Nat Commun. 7:126512016.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Frasca D, Diaz A, Romero M, Vazquez T and
Blomberg BB: Obesity induces pro-inflammatory B cells and impairs B
cell function in old mice. Mech Ageing Dev. 162:91–99. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Frasca D and Blomberg BB: Adipose tissue
Inflammation Induces B cell inflammation and Decreases B cell
function in aging. Front Immunol. 8:10032017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Apostolopoulos V, De Courten MP,
Stojanovska L, Blatch GL, Tangalakis K and De Courten B: The
complex immunological and inflammatory network of adipose tissue in
obesity. Mol Nutr Food Res. 60:43–57. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Cheng L, Wang J, Dai H, Duan Y, An Y, Shi
L, Lv Y, Li H, Wang C, Ma Q, et al: Brown and beige adipose tissue:
A novel therapeutic strategy for obesity and type 2 diabetes
mellitus. Adipocyte. 10:48–65. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Li H, Dong M, Liu W, Gao C, Jia Y, Zhang
X, Xiao X, Liu Q and Lin H: Peripheral IL-6/STAT3 signaling
promotes beiging of white fat. Biochim Biophys Acta Mol Cell Res.
1868:1190802021. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Ding M, Xu HY, Zhou WY, Xia YF, Li BY, Shi
YJ, Dou X, Yang QQ, Qian SW, Tang Y, et al: CLCF1 signaling
restrains thermogenesis and disrupts metabolic homeostasis by
inhibiting mitochondrial biogenesis in brown adipocytes. Proc Natl
Acad Sci USA. 120:e23057171202023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Biondi G, Marrano N, Borrelli A, Rella M,
Palma G, Calderoni I, Siciliano E, Lops P, Giorgino F and
Natalicchio A: Adipose tissue secretion pattern influences β-Cell
wellness in the transition from obesity to type 2 diabetes. Int J
Mol Sci. 23:55222022. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Frasca D, Diaz A, Romero M and Blomberg
BB: Leptin induces immunosenescence in human B cells. Cell Immunol.
348:1039942020. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Gupta S, Agrawal S and Gollapudi S:
Increased activation and cytokine secretion in B cells stimulated
with leptin in aged humans. Immun Ageing. 10:32013. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Mihaylova MM and Shaw RJ: The AMPK
signalling pathway coordinates cell growth, autophagy and
metabolism. Nat Cell Biol. 13:1016–1023. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Frasca D, Ferracci F, Diaz A, Romero M,
Lechner S and Blomberg BB: Obesity decreases B cell responses in
young and elderly individuals. Obesity (Silver Spring). 24:615–625.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Rosser EC, Oleinika K, Tonon S, Doyle R,
Bosma A, Carter NA, Harris KA, Jones SA, Klein N and Mauri C:
Regulatory B cells are induced by gut microbiota-driven
interleukin-1 β and interleukin-6 production. Nat Med.
20:1334–1339. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Menon M, Blair PA, Isenberg DA and Mauri
C: A regulatory feedback between plasmacytoid dendritic cells and
regulatory B cells is aberrant in systemic lupus erythematosus.
Immunity. 44:683–697. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Chimen M, Mcgettrick HM, Apta B, Kuravi
SJ, Yates CM, Kennedy A, Odedra A, Alassiri M, Harrison M, Martin
A, et al: Homeostatic regulation of T cell trafficking by a B
cell-derived peptide is impaired in autoimmune and chronic
inflammatory disease. Nat Med. 21:467–475. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Hopkin SJ, Nathan P, Pezhman L, Begum J,
Manning JE, Quinn LM, Rainger GE, Mcgettrick HM, Iqbal AJ and
Chimen M: Rejuvenation of leukocyte trafficking in aged mice
through PEPITEM intervention. NPJ Aging. 10:332024. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Tsang JY, Li D, Ho D, Peng J, Xu A, Lamb
J, Chen Y and Tam PK: Novel immunomodulatory effects of adiponectin
on dendritic cell functions. Int Immunopharmacol. 11:604–609. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Zhang K, Guo Y, Ge Z, Zhang Z, Da Y, Li W,
Zhang Z, Xue Z, Li Y, Ren Y, et al: Adiponectin Suppresses T Helper
17 cell differentiation and limits autoimmune CNS inflammation via
the SIRT1/PPARγ/RORγt pathway. Mol Neurobiol. 54:4908–4920. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Li W, Geng L, Liu X, Gui W and Qi H:
Recombinant adiponectin alleviates abortion in mice by regulating
Th17/Treg imbalance via p38MAPK-STAT5 pathway. Biol Reprod.
100:1008–1017. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Jennbacken K, Stahlman S, Grahnemo L,
Wiklund O and Fogelstrand L: Glucose impairs B-1 cell function in
diabetes. Clin Exp Immunol. 174:129–138. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Luck H, Khan S, Kim JH, Copeland JK,
Revelo XS, Tsai S, Chakraborty M, Cheng K, Tao Chan Y, Nohr MK, et
al: Gut-associated IgA+ immune cells regulate obesity-related
insulin resistance. Nat Commun. 10:36502019. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Cani PD, Bibiloni R, Knauf C, Waget A,
Neyrinck AM, Delzenne NM and Burcelin R: Changes in gut microbiota
control metabolic endotoxemia-induced inflammation in high-fat
diet-induced obesity and diabetes in mice. Diabetes. 57:1470–1481.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Jia L, Bai X, Ni W, Yin S, Shi J, Yang Y
and Li J: B cells in metabolic dysfunction-associated steatotic
liver disease (MASLD): From mechanisms to therapeutic exploration.
Int Immunopharmacol. 166:1155532025. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Lancaster GI, Langley KG, Berglund NA,
Kammoun HL, Reibe S, Estevez E, Weir J, Mellett NA, Pernes G,
Conway JRW, et al: Evidence that TLR4 Is not a receptor for
saturated fatty acids but mediates lipid-induced inflammation by
reprogramming macrophage metabolism. Cell Metab. 27:1096–110.e5.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Caesar R, Tremaroli V, Kovatcheva-Datchary
P, Cani PD and Backhed F: Crosstalk between gut microbiota and
dietary lipids aggravates WAT inflammation through TLR signaling.
Cell Metab. 22:658–668. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Magouliotis DE, Tasiopoulou VS, Sioka E,
Chatedaki C and Zacharoulis D: Impact of Bariatric surgery on
metabolic and gut microbiota profile: A systematic review and
Meta-analysis. Obes Surg. 27:1345–1357. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Guillemot-Legris O, Mutemberezi V, Cani PD
and Muccioli GG: Obesity is associated with changes in oxysterol
metabolism and levels in mice liver, hypothalamus, adipose tissue
and plasma. Sci Rep. 6:196942016. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Jagannathan M, Hasturk H, Liang Y, Shin H,
Hetzel JT, Kantarci A, Rubin D, Mcdonnell ME, Van Dyke TE,
Ganley-Leal LM and Nikolajczyk BS: TLR cross-talk specifically
regulates cytokine production by B cells from chronic inflammatory
disease patients. J Immunol. 183:7461–7470. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Daley AD and Benezech C: Fat-associated
lymphoid clusters: Supporting visceral adipose tissue B cell
function in immunity and metabolism. Immunol Rev. 324:78–94. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Carey A, Nguyen K, Kandikonda P, Kruglov
V, Bradley C, Dahlquist K JV, Cholensky S, Swanson W, Badovinac VP,
Griffith TS and Camell CD: Age-associated accumulation of B cells
promotes macrophage inflammation and inhibits lipolysis in adipose
tissue during sepsis. Cell Rep. 43:1139672024. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Lovell JP, Duque C, Rousseau S, Bhalodia
A, Bermea K, Cohen CD and Adamo L: B cell-mediated antigen
presentation promotes adverse cardiac remodeling in chronic heart
failure. bioRxiv. Jun 5–2024.doi: 10.1101/2024.05.08.593153.
|
|
77
|
Ait-Oufella H, Herbin O, Bouaziz JD,
Binder CJ, Uyttenhove C, Laurans L, Taleb S, Van Vre E, Esposito B,
Vilar J, et al: B cell depletion reduces the development of
atherosclerosis in mice. J Exp Med. 207:1579–1587. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Hamze M, Desmetz C, Berthe ML, Roger P,
Boulle N, Brancherau P, Picard E, Guzman C, Tolza C and Guglielmi
P: Characterization of resident B cells of vascular walls in human
atherosclerotic patients. J Immunol. 191:3006–3016. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Horckmans M, Bianchini M, Santovito D,
Megens RTA, Springael JY, Negri I, Vacca M, Di Eusanio M, Moschetta
A, Weber C, et al: Pericardial adipose tissue regulates
granulopoiesis, fibrosis, and cardiac function after myocardial
infarction. Circulation. 137:948–960. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Capasso M, Rashed Alyahyawi A and Spear S:
Metabolic control of B cells: More questions than answers. Front
Immunol. 6:802015. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Barrow F, Khan S, Wang H and Revelo XS:
The emerging role of B cells in the pathogenesis of NAFLD.
Hepatology. 74:2277–2286. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Plubell DL, Fenton AM, Wilmarth PA,
Bergstrom P, Zhao Y, Minnier J, Heinecke JW, Yang X and Pamir N:
GM-CSF driven myeloid cells in adipose tissue link weight gain and
insulin resistance via formation of 2-aminoadipate. Sci Rep.
8:114852018. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Nimmerjahn F and Ravetch JV: Fcgamma
receptors as regulators of immune responses. Nat Rev Immunol.
8:34–47. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Lu Z, Li Y and Song J: Characterization
and treatment of inflammation and insulin resistance in obese
adipose tissue. Diabetes Metab Syndr Obes. 13:3449–3460. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Gao F, Litchfield B and Wu H: Adipose
tissue lymphocytes and obesity. J Cardiovasc Aging. 4:52024.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Jansen K, Cevhertas L, Ma S, Satitsuksanoa
P, Akdis M and Van De Veen W: Regulatory B cells, A to Z. Allergy.
76:2699–2715. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Li M, Wang H, Ni Y, Li C, Xu X, Chang H,
Xu Z, Hou M and JI M: Helminth-induced CD9+ B-cell subset
alleviates obesity-associated inflammation via IL-10 production.
Int J Parasitol. 52:111–123. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Ni Y, Xu Z, Li C, Zhu Y, Liu R, Zhang F,
Chang H, Li M, Sheng L, Li Z, et al: Therapeutic inhibition of
miR-802 protects against obesity through AMPK-mediated regulation
of hepatic lipid metabolism. Theranostics. 11:1079–1099. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Rajbhandari P, Thomas BJ, Feng AC, Hong C,
Wang J, Vergnes L, Sallam T, Wang B, Sandhu J, Seldin MM, et al:
IL-10 signaling remodels adipose chromatin architecture to limit
thermogenesis and energy expenditure. Cell. 172:218–33.e17. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Winer DA, Winer S, Chng MH, Shen L and
Engleman EG: B Lymphocytes in obesity-related adipose tissue
inflammation and insulin resistance. Cell Mol Life Sci.
71:1033–1043. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Ip BC, Hogan AE and Nikolajczyk BS:
Lymphocyte roles in metabolic dysfunction: Of men and mice. Trends
Endocrinol Metab. 26:91–100. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Frasca D, Romero M, Garcia D, Diaz A and
Blomberg BB: Obesity accelerates Age-associated defects in Human B
cells through a metabolic reprogramming induced by the fatty acid
palmitate. Front Aging. 2:8286972022. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Kaegi C, Wuest B, Schreiner J, Steiner UC,
Vultaggio A, Matucci A, Crowley C and Boyman O: Systematic review
of safety and efficacy of rituximab in treating Immune-mediated
disorders. Front Immunol. 10:19902019. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Renner C: 20 years of rituximab treatment:
What have we learnt? Future Oncol. 15:4119–4121. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Kyaw T, Tay C, Khan A, Dumouchel V, Cao A,
To K, Kehry M, Dunn R, Agrotis A, Tipping P, et al: Conventional B2
B cell depletion ameliorates whereas its adoptive transfer
aggravates atherosclerosis. J Immunol. 185:4410–4419. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Kerekes G, Soltesz P, Der H, Veres K,
Szabo Z, Vegvari A, Szegedi G, Shoenfeld Y and Szekanecz Z: Effects
of rituximab treatment on endothelial dysfunction, carotid
atherosclerosis, and lipid profile in rheumatoid arthritis. Clin
Rheumatol. 28:705–710. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Novikova DS, Popkova TV, Lukina GV,
Luchikhina EL, Karateev DE, Volkov AV, Novikov AA, Aleksandrova EN
and Nasonov EL: The effects of rituximab on lipids, arterial
stiffness and Carotid Intima-media thickness in rheumatoid
arthritis. J Korean Med Sci. 31:202–207. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Athni TS and Barmettler S:
Hypogammaglobulinemia, Late-onset neutropenia, and infections
following rituximab. Ann Allergy Asthma Immunol. 130:699–712. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Veeken LD, Opdam MAA, Verhoef LM, Popa C,
Van Crevel R and Den Broeder AA: Infection incidence, timing and
dose dependency in rheumatoid arthritis patients treated with
rituximab: A retrospective cohort study. Rheumatology (Oxford).
63:1246–1250. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Bodogai M, Lee Chang C, Wejksza K, Lai J,
Merino M, Wersto RP, Gress RE, Chan AC, Hesdorffer C and Biragyn A:
Anti-CD20 antibody promotes cancer escape via enrichment of
tumor-evoked regulatory B cells expressing low levels of CD20 and
CD137L. Cancer Res. 73:2127–2138. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Horikawa M, Minard-Colin V, Matsushita T
and Tedder TF: Regulatory B cell production of IL-10 inhibits
lymphoma depletion during CD20 immunotherapy in mice. J Clin
Invest. 121:4268–4280. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Mackay F, Schneider P, Rennert P and
Browning J: BAFF AND APRIL: A tutorial on B cell survival. Annu Rev
Immunol. 21:231–264. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Kim YH, Choi BH, Cheon HG and Do MS: B
cell activation factor (BAFF) is a novel adipokine that links
obesity and inflammation. Exp Mol Med. 41:208–216. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Sanchez DCV, Castellanos SG, Sandoval MEV
and Garcia AG: B-Cell activating factor increases related to
adiposity, insulin resistance, and endothelial dysfunction in
overweight and obese subjects. Life (Basel). 12:6342022.PubMed/NCBI
|
|
105
|
Jackson SW and Davidson A: BAFF inhibition
in SLE-Is tolerance restored? Immunol Rev. 292:102–119. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Tsiantoulas D, Sage AP, Goderle L,
Ozsvar-Kozma M, Murphy D, Porsch F, Pasterkamp G, Menche J,
Schneider P, Mallat Z and Binder CJ: B Cell-activating factor
neutralization aggravates atherosclerosis. Circulation.
138:2263–2273. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Shaikh MF, Jordan N and D'cruz DP:
Systemic lupus erythematosus. Clin Med (Lond). 17:78–83. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Samy E, Wax S, Huard B, Hess H and
Schneider P: Targeting BAFF and APRIL in systemic lupus
erythematosus and other antibody-associated diseases. Int Rev
Immunol. 36:3–19. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Dechkhajorn W, Benjathummarak S, Glaharn
S, Chaisri U, Viriyavejakul P and Maneerat Y: The activation of
BAFF/APRIL system in spleen and lymph nodes of Plasmodium
falciparum infected patients. Sci Rep. 10:38652020. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Lee EG and Oh JE: From neglect to
spotlight: The underappreciated role of B cells in cutaneous
inflammatory diseases. Front Immunol. 15:13287852024. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Sawada K, Chung H, Softic S,
Moreno-Fernandez ME and Divanovic S: The bidirectional immune
crosstalk in metabolic dysfunction-associated steatotic liver
disease. Cell Metab. 35:1852–1871. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Sage AP, Tsiantoulas D, Binder CJ and
Mallat Z: The role of B cells in atherosclerosis. Nat Rev Cardiol.
16:180–196. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Tay C, Liu YH, Kanellakis P, Kallies A, LI
Y, Cao A, Hosseini H, Tipping P, Toh BH, Bobik A and Kyaw T:
Follicular B Cells promote atherosclerosis via T Cell-mediated
differentiation into plasma cells and secreting pathogenic
immunoglobulin G. Arterioscler Thromb Vasc Biol. 38:e71–e84. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Mantani PT, Ljungcrantz I, Andersson L,
Alm R, Hedblad B, Bjorkbacka H, Nilsson J and Fredrikson GN:
Circulating CD40+ and CD86+ B cell subsets demonstrate opposing
associations with risk of stroke. Arterioscler Thromb Vasc Biol.
34:211–218. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Wang P, Yang X, Zhang L, Sha S, Huang J,
Peng J, Gu J, Pearson JA, Hu Y, Zhao H, et al: Tlr9 deficiency in B
cells leads to obesity by promoting inflammation and gut dysbiosis.
Nat Commun. 15:42322024. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Shu CJ, Benoist C and Mathis D: The immune
system's involvement in obesity-driven type 2 diabetes. Semin
Immunol. 24:436–442. 2012. View Article : Google Scholar : PubMed/NCBI
|
















