Extracellular vesicles (EVs) are small vesicular
structures released by cells, and contain a variety of bioactive
molecules, including proteins, nucleic acids and lipids, which
facilitate the transfer of information between cells. EVs play
critical roles in a plethora of physiological processes and
pathological mechanisms. These roles include transporting bioactive
molecules, facilitating intercellular communication and modulating
the functions of target cells via receptor interaction. Moreover,
EVs have emerged as valuable tools in the fields of disease
diagnosis and therapeutic intervention, particularly in the context
of conditions like cardiovascular disorders and neurological
ailments (1,2). Mesenchymal stem cells (MSCs) are
pluripotent stem cells with self-renewal properties that serve a
role in cell therapy due to their potent immunomodulatory and
regenerative effects (2). Recent
studies have highlighted the therapeutic potential of MSCs through
their paracrine products. MSCs can regulate the immune response,
promote angiogenesis and exhibit anti-inflammatory effects by
secreting a series of bioactive factors and MSC-derived EVs
(MSC-EVs), thus potentially offering therapeutic benefits for a
variety of diseases (3–5). Isolation and purification techniques
for MSC-EVs continue to be refined, emphasizing the importance of
determining their identity. Although EVs have broad application
prospects in the medical field, there are still some challenges in
their separation and purification techniques, such as improving the
separation efficiency and avoiding contamination. A number of
methods, including electron microscopy, nanotrack analysis, western
blotting and flow cytometry, have been employed to identify MSC-EVs
(2). Studies have shown that
MSC-EVs exert similar biological effects as MSCs, including
modulation of inflammatory responses, promotion of tissue repair
and antioxidant properties (6–9).
These features enable them to serve important roles in the
regulation and treatment of numerous diseases. In the context of
infectious diseases, such as renal tuberculosis (TB), urinary TB
and TB, MSC-EVs have demonstrated marked efficacy in improving
renal function and quality of life through mechanisms such as
reduction of renal injury, stimulation of renal cell regeneration
and repair, suppression of inflammatory responses, and
antiviral/antibacterial pathways (10–13).
Furthermore, comprehensive research and clinical trials are key in
fully unlocking the therapeutic potential of MSC-EVs in disease
treatment (10,14,15).
A core theory behind the enhanced biological effects
of MSC-EVs is their ability to transfer bioactive molecules to
target cells, thereby modulating numerous cellular processes.
MSC-EVs can promote tissue repair, reduce inflammation and regulate
immune responses, making them a promising therapeutic option for a
range of diseases (6). The present
research is primarily concerned with addressing challenges
pertaining to the separation efficiency, stability and safety of
MSC-EVs. Moving forward, additional enhancements in research
methodologies and the accumulation of clinical trial data will be
necessary to facilitate the progression of MSC-EVs therapy towards
clinical implementation.
Exosomes have emerged as a promising alternative to
MSCs, offering similar benefits without the risk of injection
complications (71–73). Evidence has suggested that MSC-EVs,
including HUC-MSC-EVs, are a safer therapeutic option, free from
systemic allergic reactions, hemolysis, pyrogenic reactions,
abnormal hematological changes, and vascular and muscle irritation
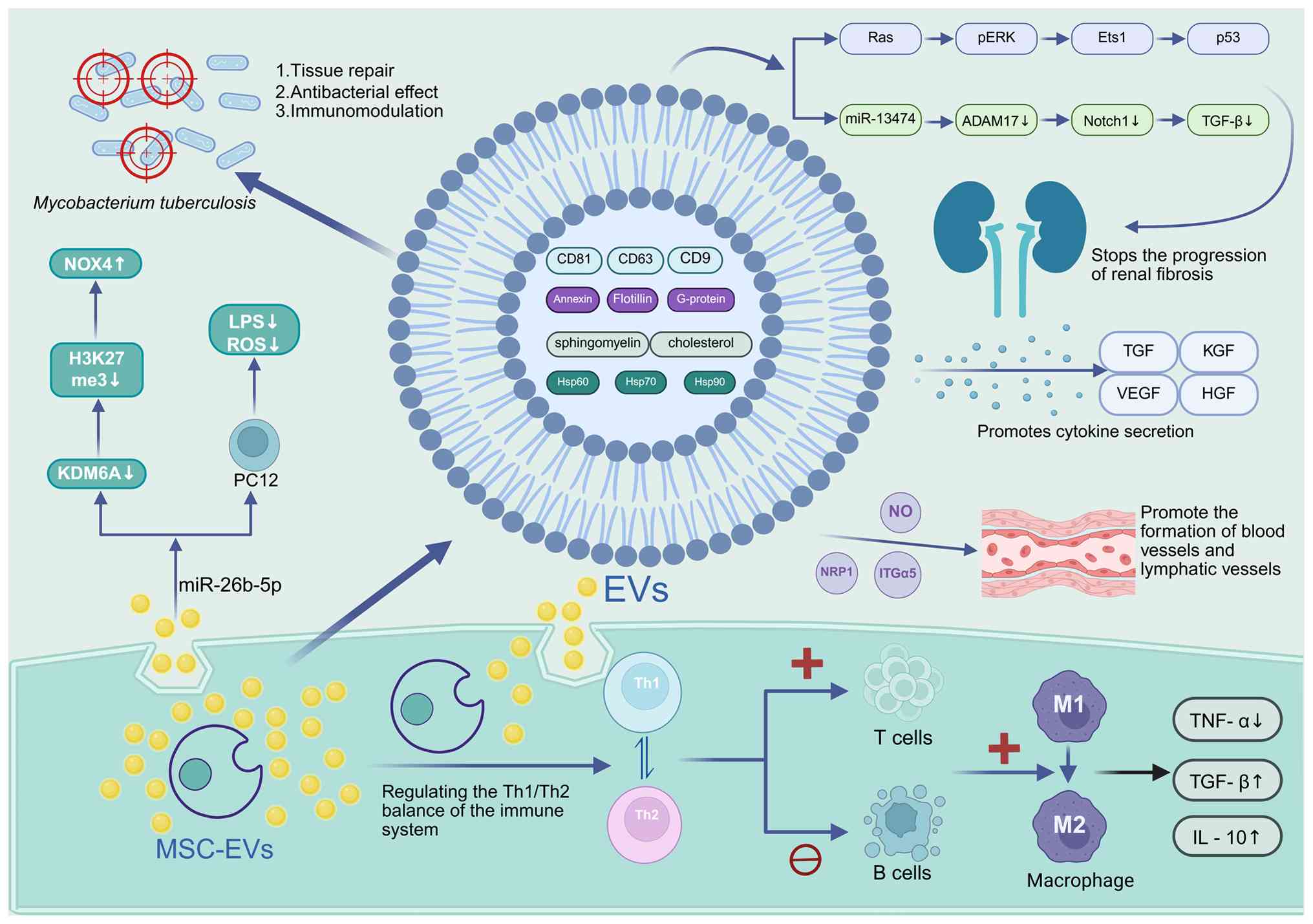
problems (23). MSC-EVs contain a
diverse range of biological molecules, including numerous types of
RNA such as mRNA and microRNA (miRNA/miR), as well as proteins.
These molecules serve a central role in promoting epithelial
repair, tissue regeneration and the secretion of important
cytokines, including TGF, keratinocyte growth factor, VEGF and
hepatocyte growth factor, amongst others (74). Research has shown that MSC-EVs
function by reducing the levels of TNF-α while increasing the
levels of molecules such as TGF-β and IL-10 (75). In addition, MSC-EVs possess the
ability to modulate the immune system by balancing the ratio of T
helper cells (Th1/Th2, promoting the generation of regulatory T
cells (Tregs) and inhibiting B cell activity (76). MSC-EVs not only boost the
expression of indoleamine 2,3-dioxygenase and activate the CD39 and
CD73 adenosine signaling pathways, but also help in converting
inflammatory M1 macrophages into anti-inflammatory M2 macrophages.
This transformation is important in regulating inflammation and
decreasing the levels of TNF-α (14,15,77).
MSC-EVs have been shown to stimulate angiogenesis and
lymphangiogenesis in endothelial cells (ECs) within ischemic areas
by transporting proteins and miRNA. This research is of great note,
as it demonstrates the crucial role played by MSC-EVs, which may be
regulated by integrin subunit α5 and neuropilin-1 proteins
(78). MSC-EVs with elevated
programmed death-ligand 1 (PD-L1) expression have demonstrated
potential in the therapeutic management of autoimmune disorders.
Through the employment of lentivirus-mediated gene transfection
strategies, scientists have engineered MSC-small EVs (sEVs) that
overexpress PD-L1 to modulate the immune response within targeted
tissues. This innovative approach has demonstrated the capacity of
MSC-sEVs-PD-L1 to orchestrate the conversion of multiple activated
immune cells towards an immunosuppressive phenotype, thereby
facilitating the restoration of the local immune microenvironment
in autoimmune pathology (79). Pei
et al (80) examined the
immunomodulatory effects of MSC-EVs on sepsis-associated liver
dysfunction in a mouse model induced by lipopolysaccharide (LPS).
Their findings indicated that MSC-EVs could mitigate liver tissue
damage and induce the transformation of M1 macrophages into M2
macrophages. In vitro experiments demonstrated that
treatment with MSC-EVs markedly reduced the expression of
glycolytic enzymes, diminished glycolytic activity and effectively
suppressed the inflammatory response of macrophages by
downregulating hypoxia-inducible factor 1α (HIF-1α) expression
(80). MSC-EVs can regulate the
immune system of the body, have antibacterial effects and promote
tissue repair by containing a variety of bioactive substances, such
as cytokines, growth factors and nucleic acids. These bioactive
substances can regulate the physiological processes within the body
through various signaling pathways, thereby playing a role in
treating diseases, such as drug-resistant infections, sepsis and
viral pneumonia (14,80–83)
(Fig. 1) (14,15,20,74,76–78,84–87).
MSC-EVs, noted for their enhanced functionality,
have attracted considerable research attention. Previous studies
(21,32,73)
have made notable advancements in uncovering their therapeutic
potential. Research utilizing bone marrow-derived MSC-EVs
(BM-MSC-EVs) has demonstrated their effects on ECs within ischemic
tissue, both in vitro and in vivo. MSC-EVs have been
demonstrated to promote angiogenesis and lymphangiogenesis in ECs
located within ischemic regions. This effect is mediated by the
transfer of proteins and microRNAs by MSC-EVs, demonstrating their
pivotal role in facilitating these physiological processes
(78). In a mouse model of severe
hind limb ischemia, MSC-EVs were found to enhance the recovery of
blood flow in ischemic muscle tissue, leading to an increase in
blood vessel density in vivo. This pro-angiogenic effect was
accompanied by an increase in nitric oxide production in ischemic
muscle (78). The findings have
indicated that fibroblasts-EVs showed the worst effect on
chondrogenesis, while juvenile chondrocyte-EVs and adult
chondrocyte-EVs showed a comparable effect on chondrogenic
differentiation as BM-MSC-EVs, and BM-MSC-EVs showed the best
effect on cell proliferation and migration. In the rat model, a
small intestinal submucosal acellular extracellular matrix hydrogel
carrying BM-MSC-EVs was successfully utilized to treat articular
cartilage defects, resulting in a marked improvement in cartilage
regeneration (88). An additional
investigation of miRNA-26b-5p-enriched MSC-EVs in spinal cord
injury (SCI) revealed that MSC-EVs could ameliorate motor
dysfunction, inflammation and oxidative stress in SCI rats. The
delivery of miR-26b-5p by MSC-EVs to PC12 cells led to reduced
inflammatory responses and reactive oxygen species (ROS) production
induced by LPS, along with enhanced cell viability. In addition,
miR-26b-5p suppresses the activity of KDM6A, which reduces H3K27me3
levels at the NOX4 promoter and thereby promotes NOX4 expression.
However, overexpression of either KDM6A or NOX4 abrogates the
protective effects conferred by MSC-EVs (89). Previous research has also
demonstrated that HUC-MSC-EVs could effectively and safely improve
chronic kidney disease. Following treatment with HUC-MSC-EVs,
notable improvements were observed in the estimated glomerular
filtration rate, serum creatinine levels, blood urea levels and the
urine albumin-creatinine ratio (90). In addition, Ma et al
(84) reported that in a mouse
model of renal ischemia-reperfusion injury, administration of human
placental MSC-EVs led to marked improvements in renal function, a
decrease in the severity of acute kidney injury and a reduction in
chronic interstitial fibrosis. MSC-EVs mainly achieved
renoprotection by regulating Bax/Bcl-2-dependent apoptosis during
acute kidney injury and mostly reduced tubular atrophy and kidney
interstitial fibrosis by regulating Ras-pERK-Ets1-p53
pathway-dependent cell senescence (84). Researchers have also explored the
potential enhancement of EVs derived from induced MSCs for treating
acute kidney injury through stimulation by stimulating them with
pan-peroxisome proliferator-activating receptor agonists (81). Shi et al (85) investigated potential therapeutic
interventions for renal fibrosis and found that HUC-MSC-EVs could
retard the progression of renal fibrosis by suppressing ADAM17.
Notably, miR-13474 was highly enriched in HUC-MSC-EVs and
efficiently targeted ADAM17 mRNA for inhibition, thereby reducing
Notch1 activation, blocking TGF-β signaling pathway engagement and
decreasing collagen deposition, all of which contribute to
mitigating fibrotic development (85). Preclinical studies have also shown
promising results in the treatment of acute and chronic renal
failure and bladder reconstruction, For instance, preclinical
studies have been applied in the treatment of acute and chronic
renal failure, as well as in bladder reconstruction (91,92).
In addition, it has been reported that Mtb can successfully evade
the effects of drugs and inflammatory cytokines within MSCs during
the treatment of infectious diseases. MSCs offer a protective
environment for Mtb, aiding in its resistance to anti-TB
medications. Despite possessing phagosome maturation capabilities,
MSCs readily engulf Mtb and facilitate its proliferation. In
contrast to the behavior of Mtb within macrophages, which is known
to be susceptible to anti-TB drugs, Mtb residing within MSCs
demonstrates a pronounced resistance or tolerance to the same
drugs, primarily attributed to the presence of ATP binding cassette
subfamily C member 1, ATP binding cassette subfamily G member 2 and
vacuolar H+-ATPase (93).
In the case of TB infection, MSC-EV treatment can
lead to the following benefits: i) Immune balance reconstruction by
inhibiting the excessive activation of macrophages and reducing
inflammatory damage in the vicinity of granulomas to combat
intracellular Mtb; ii) enhancing host defense through the delivery
of miRNA, where MSC-EVs activate macrophage autophagy, aiding in
the clearance of Mtb and preventing immune evasion by Mtb; and iii)
tissue repair, where MSC-EVs contribute to the reduction of tissue
fibrosis by inhibiting fibroblast transformation and downregulating
the TGF-β/Smad pathway (10,20,94).
MSCs are a natural reservoir for latent Mtb infection, whereas
macrophages promote an environment conducive for Mtb replication.
Mtb can enter a dormant state within MSCs, prompting Mtb to enter a
quiescent state. This process leads to the synthesis of lipid
droplets by MSCs, which helps Mtb evade the immune response of the
host. Successful treatment of TB necessitates eradication of both
actively replicating and dormant bacteria. Dormant bacteria
typically do not respond to conventional antibiotics; however, they
can be targeted by autophagy induction. Thus, the combination of
antibiotics and autophagy inducers is a promising approach for the
effective treatment of TB (95).
Studies have shown that MSC-EVs from different sources (such as the
bone marrow, umbilical cord and adipose tissue) may have different
therapeutic effects. Specifically, BM-MSC-EVs have demonstrated
promising results in stimulating angiogenesis. On the other hand,
HUC-MSC-EVs exhibit superior capabilities in modulating
anti-inflammatory responses and regulating the immune system
(23,96). In addition, adipose-derived MSC-EVs
exhibit marked effects in the treatment of fat metabolism-related
diseases such as diabetic nephropathy, but their antibacterial
activity may not be as good as that of other MSC-EVs (97). Therefore, for the treatment of TB,
the selection of MSC-EVs from different sources and the role of
MSC-EVs require further in-depth research.
A primary technique for EV separation is
differential centrifugation, which separates EVs based on their
differing settling velocities in a liquid medium. By adjusting
factors such as the centrifugal force, time and rotor type,
different particle sizes can be effectively separated. For example,
extending the centrifugation time can enhance the yields of RNA and
proteins in EVs (100). While
this method is easy to use and cost-effective, it is heavily
reliant on specific equipment and can introduce impurities, such as
cell debris, thereby impacting the purity of the final product
(100–102).
By size exclusion chromatography, exosomes can be
separated based on their particle size, resulting in high purity
and enhanced functionality. This method is characterized by its
simplicity, excellent repeatability, scalability, affordability and
the fact that it does not require specialized equipment or expert
knowledge (103). However,
relying solely on size-exclusion chromatography can result in
residual impurities. Therefore, the combination of size-exclusion
chromatography and ultracentrifugation has garnered interest, as it
effectively enhances the quality and purity of exosomes in serum
(104).
This technique involves the use of antibody-coated
magnetic beads to selectively isolate exosomes that express
specific surface markers with high specificity. Particularly
beneficial for processing low concentration samples, this method
has a higher cost and is limited by the specificity and efficacy of
the antibodies used (99,105). Brambilla et al (106) introduced an innovative capture
and release approach utilizing DNA-directed fixation of anti-CD63
antibodies. By employing deoxyribonuclease I, the authors
effectively isolated all EVs and conducted a comprehensive analysis
using advanced imaging technologies, including nanoparticle
tracking analysis, transmission electron microscopy and
single-particle interferometric reflectance imaging sensing
(101,106).
A main principle of this technique is to separate
vesicles based on their size and to concentrate them from
biological fluids using centrifugal enrichment equipment. Although
this method is fast, its ability to separate small vesicles is
limited (99). Membranes with low
protein-binding properties are ideal for this process because they
reduce the adherence of EV proteins and enhance their recovery
(101,102). Typically, ultrafiltration is
combined with other methods (such as use of differential
ultracentrifugation, filtration, concentration and high-resolution
density-gradient fractionation) to enhance the purity and
separation efficiency (102,107).
This approach utilizes centrifugal force for vesicle
separation. The benefits of ultra-centrifugation encompass superior
purity, consistency and functional efficacy of isolated EVs,
ultimately leading to a potential increase of >22% in the
activity of hypoxic cells. However, drawbacks include the risk of
vesicle rupture, difficulty in handling small sample volumes, high
cost and the specialized nature of ultracentrifuges (108). In EV research, the importance of
separation efficiency and sample purity is indisputable. A previous
study conducted a comparative analysis of two EV purification
techniques, namely ultrafiltration combined with liquid
chromatography and ultracentrifugation itself. The findings
revealed that the combination of ultrafiltration and liquid
chromatography resulted in a notable enhancement of EV production
compared with ultracentrifugation, while maintaining the protein
composition of EVs. Notably, experimental data demonstrated that
EVs purified using ultrafiltration and liquid chromatography
retained their inherent biophysical characteristics (107). In another experiment, a
combination of analytical techniques, including dynamic light
scattering, scanning electron microscopy and flow cytometry, were
utilized to isolate and characterize EVs. The findings of this
investigation demonstrate that the efficient loading of ovalbumin
(OVA) onto SMC-EVs had been successfully optimized. Specifically,
it was ascertained that the most advantageous outcome was achieved
when the initial concentration of OVA was 500 µg/ml and the
incubation duration was set at 6 h (105). However, there are variations in
the secretion and function of MSC-derived EVs from different
sources. Data have revealed that MSCs for exosome-enriched
fractions studies have been isolated from a variety of tissues
(26). Based on the diverse
advantages and disadvantages of different EV isolation technologies
(as summarized in Table II),
researchers should carefully select an isolation method that aligns
with their objectives.
Although research on MSC-EVs in the treatment of
intrapulmonary or extrapulmonary TB is still limited, preliminary
findings have shown promising results (10,20).
This necessitates further investigation and summarization of the
current research in this area.
According to statistics from the World Health
Organization, the global TB incidence reached 10.8 million in 2023,
which was a 0.9% increase from 10.7 million in 2022 (109). Among extrapulmonary TB cases,
genitourinary TB, particularly renal TB, is the most prevalent form
(110). Previous studies have
increasingly suggested that MSCs hold therapeutic promise for
inflammatory and sclerotic diseases, including renal TB (11,12).
Supporting this potential, one study has evaluated BM-MSCs as a
combination therapy for experimental renal TB model in rabbits.
Researchers induced renal TB by injecting TB H37Rv into the renal
cortex under ultrasound guidance. All animals developed renal
failure within 2.5 months. Anti-TB drugs reduced the levels of
albumin, copper protein and elastase, improved the balance of
mediators/inhibitors, and enhanced the inflammatory response, while
BM-MSC transplantation exhibited different benefits. At 1 month
post-transplant, inflammatory protein levels decreased,
kidney-specific destructive inflammation subsided and mature
connective tissue was formed, indicating activated repair
mechanisms (86). Researchers
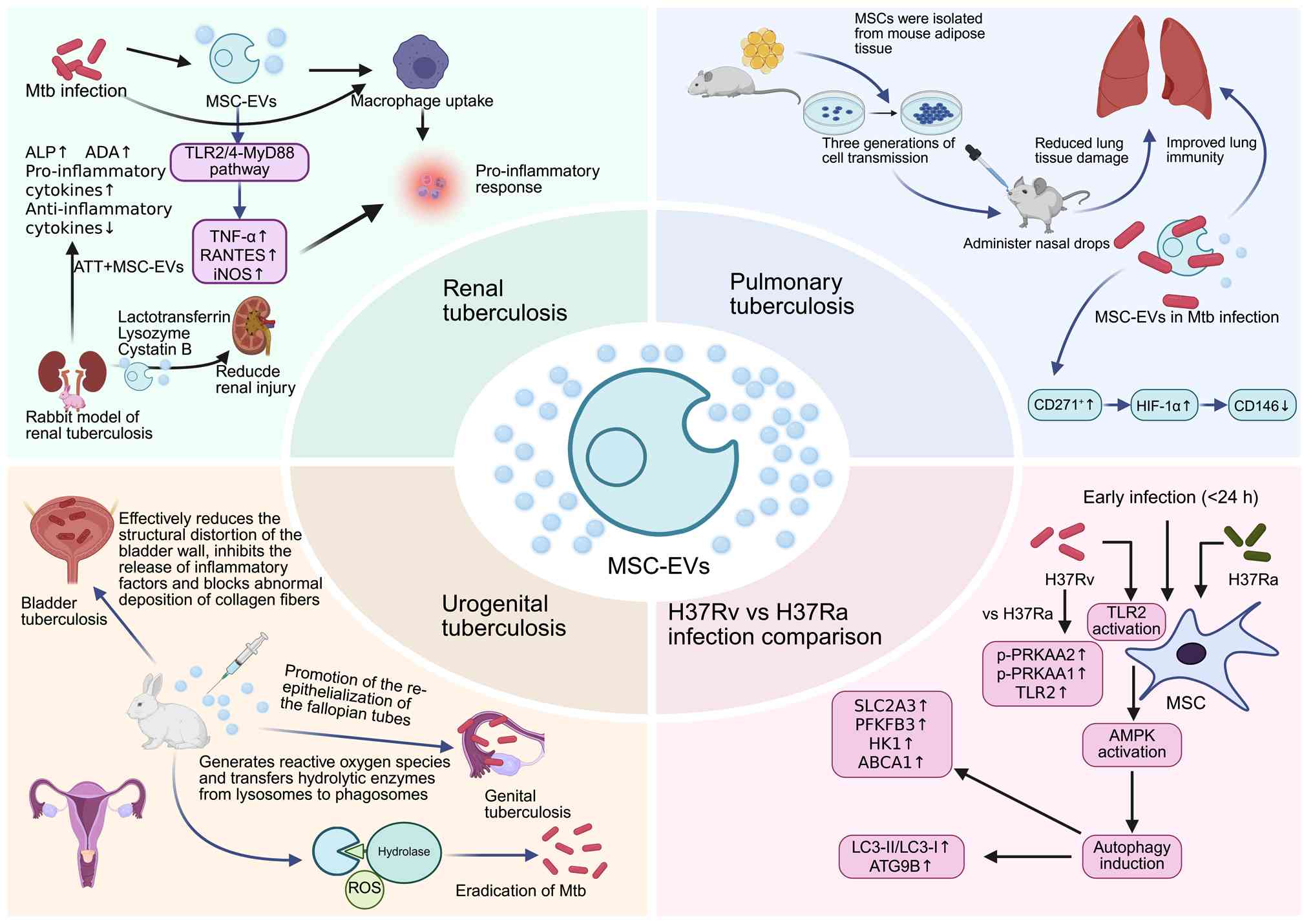
compared the cellular reactions of MSCs to the virulent H37Rv
strain and the attenuated H37Ra strain. The results showed that
early infection (≤24 h) with H37Rv led to a marked increase in the
phosphorylation of toll-like receptor (TLR)-2, protein kinase
AMP-activated catalytic subunit-α (PRKAA)-1 and PRKAA2 in MSCs
compared with attenuated H37Ra infection. Activation of the
TLR2-AMPK pathway enhanced autophagic induction in H37Rv-infected
MSCs, as demonstrated by elevated Atg9b expression and an increased
LC3-II/LC3-I ratio. In addition, metabolic analysis revealed that,
compared with H37Ra infection, H37Rv infection led to increased
expression of SLC2A3, PFKFB3, HK1 and ABCA1 in MSCs. Despite the
differences in autophagy induction, the bacterial loads between the
two strains remained similar, indicating a potential role of
apoptosis and immune inflammation in the control of mycobacterial
growth (111).
EVs exhibit therapeutic relevance in TB. Host- or
pathogen-derived EVs carry pathogenic antigens, regulating immune
responses, metabolism and cell death (112). Specifically, EVs from
Mtb-infected cells control inflammatory cytokines through antigen
presentation, transmit immunoregulatory molecules to T helper cells
and promote adaptive immune responses (113,114). Studies have shown that infection
with Mtb promotes the release of MSC-EVs without affecting MSC
proliferation; these vesicles can be internalized by macrophages
and induce time-dependent pro-inflammatory responses through
upregulation of TNF-α, RANTES and iNOS. This process is primarily
mediated via the TLR2/4-MyD88 signaling pathway, and in vivo
experiments further demonstrate that EVs derived from Mtb-infected
MSCs (Mtb-MSC-EVs) can trigger pro-inflammatory responses in mice
even during anti-TB treatment (87). A rabbit model of renal TB was
established by injecting Mtb H37Rv into the renal cortical
parenchyma, followed by intravenous administration of MSC-EVs in
combination with standard anti-TB therapy (ATT). Compared with ATT
alone, the combination treatment significantly increased serum
anti-inflammatory cytokine levels while reducing pro-inflammatory
cytokines and improving alkaline phosphatase, adenosine deaminase
and body weight. Further analyses indicated that MSC-EVs are
enriched with antimicrobial peptides such as lactotransferrin,
lysozyme and cystatin B, as well as proteins with anti-inflammatory
and immunomodulatory properties, suggesting a potential role in
alleviating renal injury (15).
Beyond therapy, Mtb-derived membrane vesicles show
potential as next-generation vaccines, eliciting stronger
adjuvant-free immunity compared with the traditional Bacillus
Calmette-Guérin (BCG) (115).
EV-associated genes may also serve as biomarkers or therapeutic
targets (114). Given their
efficacy in preclinical models of acute/chronic kidney inflammation
and functional impairment, MSC-EVs represent a promising strategy
for renal TB treatment (10,91,116).
Genitourinary TB commonly results in bladder
contracture and diminished bladder capacity, and in severe cases, a
notably reduced bladder size until the bladder becomes completely
atretic (59). Conservative
treatment of stage IV bladder TB is typically ineffective,
warranting consideration of surgical intervention. Although
numerous surgical strategies have proven to be effective, there is
a substantial risk of complications (110). Electrolyte imbalances, metabolic
abnormalities, excessive mucus production, stone formation,
recurrent urinary infections and altered drug metabolism should be
considered when addressing potential serious complications and
surgical techniques in the context of the urinary system (123). In a study utilizing a New Zealand
rabbit model of bladder TB, the efficacy of interstitial injection
of MSCs combined with standard anti-TB treatment in restoring
bladder function was determined. To track the localization of MSCs
in tissues, cells were tagged with superparamagnetic iron oxide
nanoparticles. The findings indicated that the nanoparticles were
readily absorbed by the cells without compromising their
proliferation ability. A single administration of MSCs directly
into the bladder mucosa proved to be effective in reducing bladder
wall deformation and inflammation (54). In a treatment trial involving 20
female ‘Totoro’ rabbits with experimental genital TB, the
administration of MSCs markedly enhanced the repair process of the
body and effectively promoted re-epithelialization of the fallopian
tubes 2 months after inoculation. MSCs also exhibit a bactericidal
mechanism similar to that of macrophages, involving the production
of ROS and the transfer of hydrolases from lysosomes to phagosomes.
In addition, MSCs exhibit the potential to enhance the bactericidal
capacity of macrophages by regulating their phenotypes. These
findings serve as an important theoretical foundation for the
development of novel approaches for TB treatment using EVs
(55). A number of studies
regarding the use of MSC-EVs for TB treatment are ongoing and
summarized in Fig. 2 (15,54,55,87,111,119,120,123). In addition, a summary of research
on EVs specifically targeting TB is provided in Table III. This includes studies
involving EVs derived from Mtb-infected cells and patients, EVs
administered to mice for immunization purposes, EVs isolated from
serum samples of TB patients, EVs investigated in TB animal models
and EVs discovered in pleural fluid samples (10,20,124–133).
MSC-EVs show marked promise in TB treatment,
offering potent immunomodulation, direct antibacterial effects and
tissue repair capabilities. MSC-EVs improve pathological outcomes,
reduce inflammation and combat Mtb through numerous mechanisms.
Furthermore, MSC-EVs improve kidney function, promote weight gain
and restore serum adenosine deaminase activity. Targeted
elimination of HIF-1α-high/CD146-low MSCs within hypoxic niches
could potentially eradicate latent TB infections. MSCs can
eliminate Mtb through delivery of ROS and lysosomal enzymes as
aforementioned. Technologically, methods such as ultrafiltration
combined with liquid chromatography enhance the purity and
functional capacity of MSC-EVs. Notably, their therapeutic efficacy
is dependent on the tissue source of the parent MSCs. Key
challenges requiring resolution include establishing standardized
protocols for mass production and determining long-term safety
(134–137). In summary, the primary focus of
current research encompasses enhancing the preparation techniques
of MSC-EVs, elucidating the mechanisms underlying the biological
action of MSC-EVs and utilizing MSC-EVs for the treatment of
infectious diseases such as TB.
Future research directions include engineering EVs
(such as MSC-EVs with high PD-L1 expression, conducting multi-omics
studies and accelerating clinical translation. From a technological
standpoint, there is a need to continue optimizing existing
technologies and to investigate novel isolation strategies in order
to improve the purity, yield and efficiency of MSC-EVs. This will
provide strong support for the utilization of MSC-EVs in disease
diagnosis, treatment and biomarker research, thereby advancing the
field of EV-related research towards clinical applications. A key
aspect in MSC-EVs clinical translation involves understanding how
these conditions, such as temperature, storage duration and
freeze-drying affect key properties such as morphology,
functionality and therapeutic payload. Research has shown that
MSC-EVs maintain their core biological activities, including
anti-inflammatory effects and enhanced angiogenesis promotion, for
up to 4–6 weeks when stored at temperatures of −20°C and −80°C. In
addition, freeze-drying successfully preserved these functional
properties (138). A combination
of in vitro experiments and in vivo wound healing
models has demonstrated that optimal storage conditions not only
preserve stability but also ensure the functionality of miRNA and
long non-coding RNA cargos within EVs (138). While there is currently a dearth
of research specifically investigating the effects of MSC-EVs on
Mtb infection utilizing integrated omics methodologies, findings
from related studies offer encouraging prospects for utilizing
MSC-EVs as therapeutic agents for TB. For example, a recent study
took a comprehensive approach by integrating proteomics,
metabolomics and transcriptomics analyses to elucidate the
inhibitory mechanism of MSC-EVs on the HepG2 cell line of
hepatocellular carcinoma, thereby contributing to advancements in
this field (139). Despite their
potential, current production platforms for MSC-EVs face a number
of limitations during clinical application, including variability
arising from donor sources, scalability challenges and inconsistent
therapeutic outcomes. To effectively address these shortcomings, it
is imperative to establish a scalable, standardized production
platform that integrates high-quality MSC-EVs manufacturing with
artificial intelligence-integrated, fully automated, Good
Manufacturing Practice of Medical Products-compliant manufacturing
of therapeutic EVs suitable for clinical translation (140). Collectively, MSC-EVs represent a
promising novel therapeutic strategy with the potential to address
drug-resistant TB and other challenging infections (125,141–143). However, prior to clinical
translation, it is important to perform a thorough risk assessment,
including analysis of factors such as immunogenicity, potential
pro-inflammatory effects and the possibility of promoting TB
dormancy.
Not applicable.
The present review was supported by the Sichuan Science and
Technology Program (grant nos. 2022YFS0607, 23NSFSC1456 and 2024
NSFSC0709) and the National Natural Science Foundation of China
(grant nos. 31972909, 82070502 and 32171099).
Not applicable.
YY and LY conceived the review idea and obtained the
funding. DF was responsible for organizing the manuscript,
designing the narrative to enhance coherence and completing the
writing of the manuscript. YY and LY edited the manuscript and
provided supervision. Data authentication is not applicable. All
authors have read and approved the final version of the
manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Mehaffy C, Dobos KM, Nahid P and
Kruh-Garcia NA: Second generation multiple reaction monitoring
assays for enhanced detection of ultra-low abundance
Mycobacterium tuberculosis peptides in human serum. Clin
Proteomics. 14:212017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ibrahim SA and Khan YS: Histology,
extracellular vesicles. StatPearls. Publishing LLC.; Treasure
Island, FL: 2025
|
|
3
|
Pakdaman Kolour SS, Nematollahi S,
Dehbozorgi M, Fattahi F, Movahed F, Esfandiari N, Kahrizi MS,
Ghavamikia N and Hajiagha BS: Extracecellulr vesicles (EVs)
microRNAs (miRNAs) derived from mesenchymal stem cells (MSCs) in
osteoarthritis (OA); detailed role in pathogenesis and possible
therapeutics. Heliyon. 11:e422582025. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chang M, Liu R, Chen B, Xu J, Wang W, Ji
Y, Gao Z, Liu B, Yao X, Sun H, et al: hBMSC-EVs alleviate
weightlessness-induced skeletal muscle atrophy by suppressing
oxidative stress and inflammation. Stem Cell Res Ther. 16:462025.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zheng B, Wang X, Guo M and Tzeng CM:
Current development of mesenchymal stem cell-derived extracellular
vesicles. Cell Transplant. 34:96368972412976232025. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jafarinia M, Alsahebfosoul F, Salehi H,
Eskandari N and Ganjalikhani-Hakemi M: Mesenchymal stem
cell-derived extracellular vesicles: A novel cell-free therapy.
Immunol Invest. 49:758–780. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Poh BM, Liew LC, Soh YNA, Lai RC, Lim SK,
Ho YS and Soh BS: MSC-derived small extracellular vesicles exert
cardioprotective effect through reducing VLCFAs and apoptosis in
human cardiac organoid IRI model. Stem Cells. 42:416–429. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Amaro-Prellezo E, Gómez-Ferrer M, Hakobyan
L, Ontoria-Oviedo I, Peiró-Molina E, Tarazona S, Salguero P,
Ruiz-Saurí A, Selva-Roldán M, Vives-Sanchez R and Sepúlveda P:
Extracellular vesicles from dental pulp mesenchymal stem cells
modulate macrophage phenotype during acute and chronic cardiac
inflammation in athymic nude rats with myocardial infarction.
Inflamm Regen. 44:252024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Beljanski V, Moreno Hollweg MJ, Potens R,
Blaylock T, Irausquin AB, Paleati N and Nathanson L: Functional and
molecular characterization of extracellular vesicles enriched in
exosomes released by bone marrow mesenchymal stromal cells exposed
to IFNγ in combination with autophagy modulators tamoxifen or
chloroquine. Noncoding RNA. 12:12025.PubMed/NCBI
|
|
10
|
Yudintceva N, Bobkov D, Sulatsky M,
Mikhailova N, Oganesyan E, Vinogradova T, Muraviov A, Remezova A,
Bogdanova E, Garapach I, et al: Mesenchymal stem cells-derived
extracellular vesicles for therapeutics of renal tuberculosis. Sci
Rep. 14:44952024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang X, Xie Q, Ye Z, Li Y, Che Z, Huang M
and Zeng J: Mesenchymal stem cells and tuberculosis: Clinical
challenges and opportunities. Front Immunol. 12:6952782021.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Devi A, Pahuja I, Singh SP, Verma A,
Bhattacharya D, Bhaskar A, Dwivedi VP and Das G: Revisiting the
role of mesenchymal stem cells in tuberculosis and other infectious
diseases. Cell Mol Immunol. 20:600–612. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kaur S, Angrish N, Vasudevan M and Khare
G: Global proteomics reveals pathways of mesenchymal stem cells
altered by Mycobacterium tuberculosis. Sci Rep.
14:306772024. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yudintceva N, Mikhailova N, Fedorov V,
Samochernych K, Vinogradova T, Muraviov A and Shevtsov M:
Mesenchymal stem cells and MSCs-derived extracellular vesicles in
infectious diseases: From basic research to clinical practice.
Bioengineering (Basel). 9:6622022. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yuan D, Bao Y and El-Hashash A:
Mesenchymal stromal cell-based therapy in lung diseases; from
research to clinic. Am J Stem Cells. 13:37–58. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Abbasi R, Alamdari-Mahd G, Maleki-Kakelar
H, Momen-Mesgin R, Ahmadi M, Sharafkhani M and Rezaie J: Recent
advances in the application of engineered exosomes from mesenchymal
stem cells for regenerative medicine. Eur J Pharmacol.
989:1772362025. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Noh MY, Lim SM, Oh KW, Cho KA, Park J, Kim
KS, Lee SJ, Kwon MS and Kim SH: Mesenchymal stem cells modulate the
functional properties of microglia via TGF-β secretion. Stem Cells
Transl Med. 5:1538–1549. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Abiko M, Mitsuhara T, Okazaki T, Imura T,
Nakagawa K, Otsuka T, Oshita J, Takeda M, Kawahara Y, Yuge L and
Kurisu K: Rat cranial bone-derived mesenchymal stem cell
transplantation promotes functional recovery in ischemic stroke
model rats. Stem Cells Dev. 27:1053–1061. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yang Q, Zhou Y, Farooq W, Liu Q, Duan J,
Xing L, Wu C and Dong L: The immunomodulatory effects of
mesenchymal stem cells on THP-1-derived macrophages against
Mycobacterium tuberculosis H37Ra infection. Tuberculosis
(Edinb). 150:1025932025. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yan K, Xu G and Li Z: MicroRNA-20b carried
by mesenchymal stem cell-derived extracellular vesicles protects
alveolar epithelial type II cells from Mycobacterium
tuberculosis infection in vitro. Infect Genet Evol.
101:1052922022. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Din MAU, Wan A, Chu Y, Zhou J, Yan Y and
Xu Z: Therapeutic role of extracellular vesicles from human
umbilical cord mesenchymal stem cells and their wide therapeutic
implications in inflammatory bowel disease and other inflammatory
disorder. Front Med (Lausanne). 11:14065472024. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Grillo MA: Extracellular synaptic vesicles
in the mouse heart. J Cell Biol. 47:547–553. 1970. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yaghoubi Y, Movassaghpour A, Zamani M,
Talebi M, Mehdizadeh A and Yousefi M: Human umbilical cord
mesenchymal stem cells derived-exosomes in diseases treatment. Life
Sci. 233:1167332019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wei YN, Yan CY, Zhao ML and Zhao XH: The
role and application of vesicles in triple-negative breast cancer:
Opportunities and challenges. Mol Ther Oncolytics. 31:1007522023.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Latifkar A, Cerione RA and Antonyak MA:
Probing the mechanisms of extracellular vesicle biogenesis and
function in cancer. Biochem Soc Trans. 46:1137–1146. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Elahi FM, Farwell DG, Nolta JA and
Anderson JD: Preclinical translation of exosomes derived from
mesenchymal stem/stromal cells. Stem Cells. 38:15–21. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
van Niel G, D'Angelo G and Raposo G:
Shedding light on the cell biology of extracellular vesicles. Nat
Rev Mol Cell Biol. 19:213–228. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Abels ER and Breakefield XO: Introduction
to extracellular vesicles: Biogenesis, RNA cargo selection,
content, release, and uptake. Cell Mol Neurobiol. 36:301–312. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang C, Xue Y, Duan Y, Mao C and Wan M:
Extracellular vesicles and their engineering strategies, delivery
systems, and biomedical applications. J Control Release.
365:1089–1123. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mehaffy C, Ryan JM, Kruh-Garcia NA and
Dobos KM: Extracellular vesicles in mycobacteria and tuberculosis.
Front Cell Infect Microbiol. 12:9128312022. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Carrière J, Barnich N and Nguyen HT:
Exosomes: From functions in host-pathogen interactions and immunity
to diagnostic and therapeutic opportunities. Rev Physiol Biochem
Pharmacol. 172:39–75. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Börger V, Bremer M, Ferrer-Tur R, Gockeln
L, Stambouli O, Becic A and Giebel B: Mesenchymal stem/stromal
cell-derived extracellular vesicles and their potential as novel
immunomodulatory therapeutic agents. Int J Mol Sci. 18:14502017.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Qiao Z, Wang X, Zhao H, Deng Y, Zeng W,
Yang K, Chen H, Yan Q, Li C, Wu J and Chen Y: The effectiveness of
cell-derived exosome therapy for diabetic wound: A systematic
review and meta-analysis. Ageing Res Rev. 85:1018582023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lin YN, Mesquita T, Sanchez L, Chen YH,
Liu W, Li C, Rogers R, Wang Y, Li X, Wu D, et al: Extracellular
vesicles from immortalized cardiosphere-derived cells attenuate
arrhythmogenic cardiomyopathy in desmoglein-2 mutant mice. Eur
Heart J. 42:3558–3571. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang X, Wu Y, Cheng Q, Bai L, Huang S and
Gao J: Extracellular vesicles in cardiovascular diseases: Diagnosis
and therapy. Front Cell Dev Biol. 10:8753762022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Dang Y, Hua W, Zhang X, Sun H, Zhang Y, Yu
B, Wang S, Zhang M, Kong Z, Pan D, et al: Anti-angiogenic effect of
exo-LncRNA TUG1 in myocardial infarction and modulation by remote
ischemic conditioning. Basic Res Cardiol. 118:12023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Giacomini E, Scotti GM, Vanni VS,
Lazarevic D, Makieva S, Privitera L, Signorelli S, Cantone L,
Bollati V, Murdica V, et al: Global transcriptomic changes occur in
uterine fluid-derived extracellular vesicles during the endometrial
window for embryo implantation. Hum Reprod. 36:2249–2274. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kim KY, Shin KY and Chang KA:
Brain-derived exosomal proteins as effective biomarkers for
Alzheimer's disease: A systematic review and meta-analysis.
Biomolecules. 11:9802021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Jeyaraman M, Rajendran RL, Muthu S,
Jeyaraman N, Sharma S, Jha SK, Muthukanagaraj P, Hong CM, Furtado
da Fonseca L, Santos Duarte Lana JF, et al: An update on stem cell
and stem cell-derived extracellular vesicle-based therapy in the
management of Alzheimer's disease. Heliyon. 9:e178082023.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Liu C, Wang J, Hu J, Fu B, Mao Z, Zhang H,
Cai G, Chen X and Sun X: Extracellular vesicles for acute kidney
injury in preclinical rodent models: a meta-analysis. Stem Cell Res
Ther. 11:112020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Tang TT, Lv LL, Lan HY and Liu BC:
Extracellular vesicles: Opportunities and challenges for the
treatment of renal diseases. Front Physiol. 10:2262019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Burdeyron P, Giraud S, Lepoittevin M,
Jordan N, Brishoual S, Jacquard M, Ameteau V, Boildieu N, Lemarie
E, Daniel J, et al: Dynamic conditioning of porcine kidney grafts
with extracellular vesicles derived from urine progenitor cells: A
proof-of-concept study. Clin Transl Med. 14:e700952024. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Psaraki A, Ntari L, Karakostas C,
Korrou-Karava D and Roubelakis MG: Extracellular vesicles derived
from mesenchymal stem/stromal cells: The regenerative impact in
liver diseases. Hepatology. 75:1590–1603. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tieu A, Hu K, Gnyra C, Montroy J,
Fergusson DA, Allan DS, Stewart DJ, Thébaud B and Lalu MM:
Mesenchymal stromal cell extracellular vesicles as therapy for
acute and chronic respiratory diseases: A meta-analysis. J
Extracell Vesicles. 10:e121412021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Sánchez SV, Otavalo GN, Gazeau F, Silva
AKA and Morales JO: Intranasal delivery of extracellular vesicles:
A promising new approach for treating neurological and respiratory
disorders. J Control Release. 379:489–523. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Jiang Q, Ning F, Jia Q, Wang H, Xue W,
Wang J, Wang Y, Zhu Z and Tian L: MiR-148a-3p loaded human
umbilical cord mesenchymal stem cell-derived extracellular vesicles
alleviates silica-induced pulmonary fibrosis by inhibiting
β-catenin signaling. Int J Nanomedicine. 20:4319–4336. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Shen Q, Huang Z, Yao J and Jin Y:
Extracellular vesicles-mediated interaction within intestinal
microenvironment in inflammatory bowel disease. J Adv Res.
37:221–233. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
You B, Yang Y, Wei J, Zhou C and Dong S:
Pathogenic and therapeutic roles of extracellular vesicles in
sepsis. Front Immunol. 16:15354272025. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ding JY, Chen MJ, Wu LF, Shu GF, Fang SJ,
Li ZY, Chu XR, Li XK, Wang ZG and Ji JS: Mesenchymal stem
cell-derived extracellular vesicles in skin wound healing: Roles,
opportunities and challenges. Mil Med Res. 10:362023.PubMed/NCBI
|
|
50
|
Wu J, Wu J, Liu Z, Gong Y, Feng D, Xiang
W, Fang S, Chen R, Wu Y, Huang S, et al: Mesenchymal stem
cell-derived extracellular vesicles in joint diseases: Therapeutic
effects and underlying mechanisms. J Orthop Translat. 48:53–69.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Zhuang Y, Jiang S, Yuan C and Lin K: The
potential therapeutic role of extracellular vesicles in
osteoarthritis. Front Bioeng Biotechnol. 10:10223682022. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Chen Y, Li B, Christelle M, Eugene N, Han
W, Zhou H, Qiu N, Zhang H and Xu J: Modifying MSCs-derived EVs with
esterase-responsive and charge-reversal cationic polymers enhances
bone regeneration. iScience. 27:1108012024. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Kuang H, Dou G, Cheng L, Wang X, Xu H, Liu
X, Ding F, Yang X, Liu S, Bao L, et al: Humoral regulation of iron
metabolism by extracellular vesicles drives antibacterial response.
Nat Metab. 5:111–128. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Yudintceva NM, Bogolyubova IO, Muraviov
AN, Sheykhov MG, Vinogradova TI, Sokolovich EG, Samusenko IA and
Shevtsov MA: Application of the allogenic mesenchymal stem cells in
the therapy of the bladder tuberculosis. J Tissue Eng Regen Med.
12:e1580–e1593. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Ariel BM, Guseinova FM, Vinogradova TI,
Zabolotnykh NV, Niaury DA, Yudintceva NM, Vitovskaya ML, Dogonadze
MZ, Rubtsova OL and Yablonsky PK: Mesenchymal stem cells of the
bone marrow in treatment of genital tuberculosis in rabbits
(experimental research with morphological control). Rev Clin Pharm
Drug Ther. 15:47–55. 2017. View Article : Google Scholar
|
|
56
|
Ikeda G, Santoso MR, Tada Y, Li AM,
Vaskova E, Jung JH, O'Brien C, Egan E, Ye J and Yang PC:
Mitochondria-rich extracellular vesicles from autologous stem
cell-derived cardiomyocytes restore energetics of ischemic
myocardium. J Am Coll Cardiol. 77:1073–1088. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Tominaga Y, Kawamura T, Ito E, Takeda M,
Harada A, Torigata K, Sakaniwa R, Sawa Y and Miyagawa S:
Pleiotropic effects of extracellular vesicles from induced
pluripotent stem cell-derived cardiomyocytes on ischemic
cardiomyopathy: A preclinical study. J Heart Lung Transplant.
43:85–99. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Zheng X, Zhang L, Kuang Y, Venkataramani
V, Jin F, Hein K, Zafeiriou MP, Lenz C, Moebius W, Kilic E, et al:
Extracellular vesicles derived from neural progenitor cells-a
preclinical evaluation for stroke treatment in mice. Transl Stroke
Res. 12:185–203. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Sun C, Sha S, Shan Y, Gao X, Li L, Xing C,
Guo Z and Du H: Intranasal delivery of BACE1 siRNA and berberine
via engineered stem cell exosomes for the treatment of Alzheimer's
disease. Int J Nanomedicine. 20:5873–5891. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Nan C, Zhang Y, Zhang A, Shi Y, Yan D, Sun
Z, Jin Q, Huo H, Zhuo Y and Zhao Z: Exosomes derived from human
umbilical cord mesenchymal stem cells decrease neuroinflammation
and facilitate the restoration of nerve function in rats suffering
from intracerebral hemorrhage. Mol Cell Biochem. 480:309–323. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Chen F, Chen Z, Wu HT, Chen XX, Zhan P,
Wei ZY, Ouyang Z, Jiang X, Shen A, Luo MH, et al: Mesenchymal stem
cell-derived exosomes attenuate murine cytomegalovirus-infected
pneumonia via NF-κB/NLRP3 signaling pathway. Viruses. 16:6192024.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Costa-Ferro ZSM, Rocha GV, da Silva KN,
Paredes BD, Loiola EC, Silva JD, Santos JLDS, Dias RB, Figueira CP,
de Oliveira CI, et al: GMP-compliant extracellular vesicles derived
from umbilical cord mesenchymal stromal cells: Manufacturing and
pre-clinical evaluation in ARDS treatment. Cytotherapy.
26:1013–1025. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Jeong J, Park JK, Shin J, Jung I, Kim HW,
Park A, Cho H, Kang SM, Shin S, Park E, et al: Inflammatory
cytokine-primed MSC-derived extracellular vesicles ameliorate acute
lung injury via enhanced immunomodulation and alveolar repair. Stem
Cell Res Ther. 16:4502025. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Krishnan A, Harikrishnan VS, Sabareeswaran
A and Kasoju N: Human Wharton's Jelly mesenchymal stem cells and
their extracellular vesicles in the management of bleomycin-induced
lung injury in model animals: A comparative preclinical study
focused on histomorphometric analysis. Curr Stem Cell Res Ther.
20:1184–1197. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Figueroa-Valdés AI, Luz-Crawford P,
Herrera-Luna Y, Georges-Calderón N, García C, Tobar HE, Araya MJ,
Matas J, Donoso-Meneses D, de la Fuente C, et al: Clinical-grade
extracellular vesicles derived from umbilical cord mesenchymal
stromal cells: Preclinical development and first-in-human
intra-articular validation as therapeutics for knee osteoarthritis.
J Nanobiotechnology. 23:132025. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Sarkar S, Barnaby R, Faber Z, Taub L,
Roche C, Vietje B, Taatjes DJ, Wargo MJ, Weiss DJ, Bonfield TL, et
al: Extracellular vesicles derived from mesenchymal stromal cells
reduce pseudomonas aeruginosa lung infection and inflammation in
mice. bioRxiv [Preprint]. 2025.03.30.646208. 2025.
|
|
67
|
Mirsanei Z, Jamshidi-Adegani F, Vakilian
S, Elumalai P, Hazrati A, Soufihasanabad S, Ahangari F, Karima S,
Salmani F, Ebadi Asl P, et al: Extracellular vesicles derived from
MSCs carrying dexamethasone alleviate inflammatory damage in septic
mice. Eur J Pharmacol. 1005:1780422025. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Aghayan AH, Mirazimi Y, Fateh K, Keshtkar
A, Rafiee M and Atashi A: Therapeutic effects of mesenchymal stem
cell-derived extracellular vesicles in sepsis: A systematic review
and meta-analysis of preclinical studies. Stem Cell Rev Rep.
20:1480–1500. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Choi HY, Kim TY, Lee M, Kim SH, Jhee JH,
Lee YK, Kim HJ and Park HC: Kidney mesenchymal stem cell-derived
extracellular vesicles engineered to express erythropoietin improve
renal anemia in mice with chronic kidney disease. Stem Cell Rev
Rep. 18:980–992. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Yuan F, Liu J, Zhong L, Liu P, Li T, Yang
K, Gao W, Zhang G, Sun J and Zou X: Enhanced therapeutic effects of
hypoxia-preconditioned mesenchymal stromal cell-derived
extracellular vesicles in renal ischemic injury. Stem Cell Res
Ther. 16:392025. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Rani S, Ryan AE, Griffin MD and Ritter T:
Mesenchymal stem cell-derived extracellular vesicles: Toward
cell-free therapeutic applications. Mol Ther. 23:812–823. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Hassan N, Hussein DM, Malak FA, Abdelaziz
MA, Boushra MI, Shaalan W and Elzayat EM: Mesenchymal stem cells: A
therapeutic approach in fertility restoration in premature ovarian
insufficiency. Stem Cell Rev Rep. 21:2089–2102. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Aldoghachi AF, Loh JK, Wang ML, Yang YP,
Chien CS, Teh HX, Omar AH, Cheong SK, Yeap SK, Ho WY and Ong AHK:
Current developments and therapeutic potentials of exosomes from
induced pluripotent stem cells-derived mesenchymal stem cells. J
Chin Med Assoc. 86:356–365. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Keshtkar S, Azarpira N and Ghahremani MH:
Mesenchymal stem cell-derived extracellular vesicles: Novel
frontiers in regenerative medicine. Stem Cell Res Ther. 9:632018.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Shahryari F, Jafarinia M, Jafarinia M and
Azimzadeh M: Immunomodulatory and neuroprotective effects of
miR-146a-enriched MSC-derived extracellular vesicles in
experimental autoimmune encephalomyelitis. Int Immunopharmacol.
166:1155842025. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Colombo M, Raposo G and Théry C:
Biogenesis, secretion, and intercellular interactions of exosomes
and other extracellular vesicles. Annu Rev Cell Dev Biol.
30:255–289. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Favaro E, Carpanetto A, Lamorte S, Fusco
A, Caorsi C, Deregibus MC, Bruno S, Amoroso A, Giovarelli M, Porta
M, et al: Human mesenchymal stem cell-derived microvesicles
modulate T cell response to islet antigen glutamic acid
decarboxylase in patients with type 1 diabetes. Diabetologia.
57:1664–1673. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Łabędź-Masłowska A, Vergori L,
Kędracka-Krok S, Karnas E, Bobis-Wozowicz S, Sekuła-Stryjewska M,
Sarna M, Andriantsitohaina R and Zuba-Surma EK: Mesenchymal stem
cell-derived extracellular vesicles exert pro-angiogenic and
pro-lymphangiogenic effects in ischemic tissues by transferring
various microRNAs and proteins including ITGa5 and NRP1. J
Nanobiotechnology. 22:602024. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Xu F, Fei Z, Dai H, Xu J, Fan Q, Shen S,
Zhang Y, Ma Q, Chu J, Peng F, et al: Mesenchymal stem cell-derived
extracellular vesicles with high PD-L1 expression for autoimmune
diseases treatment. Adv Mater. 34:e21062652022. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Pei L, Li R, Wang X, Xu D, Gong F, Chen W,
Zheng X, Liu W, Zhao S, Wang Q, et al: MSCs-derived extracellular
vesicles alleviate sepsis-associated liver dysfunction by
inhibiting macrophage glycolysis-mediated inflammatory response.
Int Immunopharmacol. 128:1115752024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Kim H, Lee SK, Hong S, Park TS, Kim J, Kim
S and Kim TM: Pan PPAR agonist stimulation of induced MSCs produces
extracellular vesicles with enhanced renoprotective effect for
acute kidney injury. Stem Cell Res Ther. 15:92024. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Manzoor T, Saleem A, Farooq N, Dar LA,
Nazir J, Saleem S, Ismail S, Gugjoo MB, Shiekh PA and Ahmad SM:
Extracellular vesicles derived from mesenchymal stem cells-a novel
therapeutic tool in infectious diseases. Inflamm Regen. 43:172023.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Hong S, Kim H, Kim J, Kim S, Park TS and
Kim TM: Extracellular vesicles from induced pluripotent stem
cell-derived mesenchymal stem cells enhance the recovery of acute
kidney injury. Cytotherapy. 26:51–62. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Ma M, Zeng J, Zhu M, Li H, Lin T, Yang H,
Wei X and Song T: Human umbilical cord mesenchymal stem
cells-derived extracellular vesicles ameliorate kidney
ischemia-reperfusion injury by suppression of senescent tubular
epithelial cells: Experimental study. Int J Surg. 111:394–410.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Shi L, Hu Y, Zeng H, Shi H, Xu W, Sun Y,
Chu H, Ji C and Qian H: Mesenchymal stem cell-derived extracellular
vesicles ameliorate renal interstitial fibrosis via the
miR-13474/ADAM17 axis. Sci Rep. 14:177032024. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Muraviov AN, Vinogradova TI, Remezova AN,
Ariel BM, Gorelova AA, Orlova NV, Yudintceva NM, Esmedliaeva DS,
Dyakova ME, Dogonadze MZ, et al: The use of mesenchymal stem cells
in the complex treatment of kidney tuberculosis (experimental
study). Biomedicines. 10:30622022. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Liu M, Wang Z, Ren S and Zhao H: Exosomes
derived from Mycobacterium tuberculosis-infected MSCs induce
a pro-inflammatory response of macrophages. Aging (Albany NY).
13:11595–11609. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Zhu W, Shi J, Weng B, Zhou Z, Mao X, Pan
S, Peng J, Zhang C, Mao H, Li M and Zhao J: EVs from cells at the
early stages of chondrogenesis delivered by injectable SIS dECM
promote cartilage regeneration. J Tissue Eng.
15:204173142412681892024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Xu J, Ren Z, Niu T and Li S: Epigenetic
mechanism of miR-26b-5p-enriched MSCs-EVs attenuates spinal cord
injury. Regen Ther. 25:35–48. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Nassar W, El-Ansary M, Sabry D, Mostafa
MA, Fayad T, Kotb E, Temraz M, Saad AN, Essa W and Adel H:
Umbilical cord mesenchymal stem cells derived extracellular
vesicles can safely ameliorate the progression of chronic kidney
diseases. Biomater Res. 20:212016. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Grange C, Skovronova R, Marabese F and
Bussolati B: Stem cell-derived extracellular vesicles and kidney
regeneration. Cells. 8:12402019. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Perico L, Morigi M, Rota C, Breno M, Mele
C, Noris M, Introna M, Capelli C, Longaretti L, Rottoli D, et al:
Human mesenchymal stromal cells transplanted into mice stimulate
renal tubular cells and enhance mitochondrial function. Nat Commun.
8:9832017. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Jain N, Kalam H, Singh L, Sharma V, Kedia
S, Das P, Ahuja V and Kumar D: Mesenchymal stem cells offer a
drug-tolerant and immune-privileged niche to Mycobacterium
tuberculosis. Nat Commun. 11:30622020. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Alipoor SD and Elieh-Ali-Komi D:
Significance of extracellular vesicles in orchestration of immune
responses in Mycobacterium tuberculosis infection. Front
Cell Infect Microbiol. 14:13980772024. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Fatima S, Kamble SS, Dwivedi VP,
Bhattacharya D, Kumar S, Ranganathan A, Van Kaer L, Mohanty S and
Das G: Mycobacterium tuberculosis programs mesenchymal stem
cells to establish dormancy and persistence. J Clin Invest.
130:655–661. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Bárcia RN, Santos JM, Filipe M, Teixeira
M, Martins JP, Almeida J, Água-Doce A, Almeida SC, Varela A, Pohl
S, et al: What makes umbilical cord tissue-derived mesenchymal
stromal cells superior immunomodulators when compared to bone
marrow derived mesenchymal stromal cells? Stem Cells Int.
2015:5839842015. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Harman RM, Yang S, He MK and Van de Walle
GR: Antimicrobial peptides secreted by equine mesenchymal stromal
cells inhibit the growth of bacteria commonly found in skin wounds.
Stem Cell Res Ther. 8:1572017. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Konoshenko MY, Lekchnov EA, Vlassov AV and
Laktionov PP: Isolation of extracellular vesicles: General
methodologies and latest trends. Biomed Res Int. 2018:85453472018.
View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Lane RE, Korbie D, Trau M and Hill MM:
Purification protocols for extracellular vesicles. Methods Mol
Biol. 1660:111–130. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Cvjetkovic A, Lötvall J and Lässer C: The
influence of rotor type and centrifugation time on the yield and
purity of extracellular vesicles. J Extracell Vesicles.
3:10.3402/jev.v3.23111. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Abramowicz A, Widlak P and Pietrowska M:
Proteomic analysis of exosomal cargo: The challenge of high purity
vesicle isolation. Mol Biosyst. 12:1407–1419. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Zhang Q, Jeppesen DK, Higginbotham JN,
Franklin JL and Coffey RJ: Comprehensive isolation of extracellular
vesicles and nanoparticles. Nat Protoc. 18:1462–1487. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Sidhom K, Obi PO and Saleem A: A review of
exosomal isolation methods: Is size exclusion chromatography the
best option? Int J Mol Sci. 21:64662020. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Yang J, Gao X, Xing X, Huang H, Tang Q, Ma
S, Xu X, Liang C, Li M, Liao L and Tian W: An isolation system to
collect high quality and purity extracellular vesicles from serum.
Int J Nanomedicine. 16:6681–6692. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Dehnavi S, Khodadadi A, Asadirad A and
Ghadiri A: Loading ovalbumin into mesenchymal stem cell-derived
exosomes as a nanoscale carrier with immunomodulatory potential for
allergen-specific immunotherapy. Rep Biochem Mol Biol. 11:626–634.
2023.PubMed/NCBI
|
|
106
|
Brambilla D, Sola L, Ferretti AM, Chiodi
E, Zarovni N, Fortunato D, Criscuoli M, Dolo V, Giusti I, Murdica
V, et al: EV separation: Release of intact extracellular vesicles
immunocaptured on magnetic particles. Anal Chem. 93:5476–5483.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Nordin JZ, Lee Y, Vader P, Mäger I,
Johansson HJ, Heusermann W, Wiklander OPB, Hällbrink M, Seow Y,
Bultema JJ, et al: Ultrafiltration with size-exclusion liquid
chromatography for high yield isolation of extracellular vesicles
preserving intact biophysical and functional properties.
Nanomedicine. 11:879–883. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Ansari FJ, Tafti HA, Amanzadeh A, Rabbani
S, Shokrgozar MA, Heidari R, Behroozi J, Eyni H, Uversky VN and
Ghanbari H: Comparison of the efficiency of ultrafiltration,
precipitation, and ultracentrifugation methods for exosome
isolation. Biochem Biophys Rep. 38:1016682024.PubMed/NCBI
|
|
109
|
Lee H, Kim J, Kim J and Park YJ: Review of
the global burden of tuberculosis in 2023: Insights from the WHO
global tuberculosis report 2024. Jugan Geongang Gwa Jilbyeong. 18
(11 Suppl):S55–S69. 2025.(In Korean). PubMed/NCBI
|
|
110
|
Muneer A, Macrae B, Krishnamoorthy S and
Zumla A: Urogenital tuberculosis-epidemiology, pathogenesis and
clinical features. Nat Rev Urol. 16:573–598. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Li H, Yin Y, Cao W, Chen S, Chen J, Xing Y
and Yang H: Enhanced autophagy and cholesterol efflux in mouse
mesenchymal stem cells infected with H37Rv compared to H37Ra.
Microb Pathog. 199:1071992025. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Sun X, Li W, Zhao L, Fan K, Qin F, Shi L,
Gao F and Zheng C: Current landscape of exosomes in tuberculosis
development, diagnosis, and treatment applications. Front Immunol.
15:14018672024. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Yan Z, Wang H, Mu L, Hu ZD and Zheng WQ:
Regulatory roles of extracellular vesicles in immune responses
against Mycobacterium tuberculosis infection. World J Clin
Cases. 9:7311–7318. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Arya R, Shakya H, Chaurasia R, Haque MA
and Kim JJ: Exploring the role of extracellular vesicles in the
pathogenesis of tuberculosis. Genes (Basel). 15:4342024. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Prados-Rosales R, Carreño LJ,
Batista-Gonzalez A, Baena A, Venkataswamy MM, Xu J, Yu X, Wallstrom
G, Magee DM, LaBaer J, et al: Mycobacterial membrane vesicles
administered systemically in mice induce a protective immune
response to surface compartments of Mycobacterium
tuberculosis. mBio. 5:e01921–14. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Fang Y, Bouari S, Hoogduijn MJ, Ijzermans
JNM, de Bruin RWF and Minnee RC: Therapeutic efficacy of
extracellular vesicles to suppress allograft rejection in
preclinical kidney transplantation models: A systematic review and
meta-analysis. Transplant Rev (Orlando). 36:1007142022. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Sershen CL, Salim T and May EE:
Investigating the comorbidity of COPD and tuberculosis, a
computational study. Front Syst Biol. 3:9400972023. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Erokhin VV, Vasil'eva IA, Konopliannikov
AG, Chukanov VI, Tsyb AF, Bagdasarian TR, Danilenko AA, Lepekhina
LA, Kal'sina S, Semenkova IV and Agaeva EV: Systemic
transplantation of autologous mesenchymal stem cells of the bone
marrow in the treatment of patients with multidrug-resistant
pulmonary tuberculosis. Probl Tuberk Bolezn Legk. 3–6. 2008.(In
Russian). PubMed/NCBI
|
|
119
|
Garhyan J, Bhuyan S, Pulu I, Kalita D, Das
B and Bhatnagar R: Preclinical and clinical evidence of
Mycobacterium tuberculosis persistence in the hypoxic niche
of bone marrow mesenchymal stem cells after therapy. Am J Pathol.
185:1924–1934. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Chenari A, Hazrati A, Hosseini AZ, Motiee
M and Soudi S: The effect of mesenchymal stem cell-derived
supernatant nasal administration on lung inflammation and immune
response in BCG-vaccinated BALB/c mice. Life Sci. 317:1214652023.
View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Khan A, Mann L, Papanna R, Lyu MA, Singh
CR, Olson S, Eissa NT, Cirillo J, Das G, Hunter RL and Jagannath C:
Mesenchymal stem cells internalize Mycobacterium
tuberculosis through scavenger receptors and restrict bacterial
growth through autophagy. Sci Rep. 7:150102017. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Li Q, Wang C, Gou J, Kitanovski S, Tang X,
Cai Y, Zhang C, Zhang X, Zhang Z, Qiu Y, et al: Deciphering lung
granulomas in HIV & TB co-infection: Unveiling macrophages
aggregation with IL6R/STAT3 activation. Emerg Microbes Infect.
13:23663592024. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Zuban O and Chotchaev R:
Intestinocystoplasty in tuberculosis of the urinary bladder. Exp
Clin Urol. 15:115–121. 2022.
|
|
124
|
Ji L, Fu Y and Xiong S: Chimeric antigen
carried by extracellular vesicles induces stronger protective
immunity against Mycobacterium tuberculosis infection.
Immunobiology. 229:1528342024. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Lyu L, Jia H, Liu Q, Ma W, Li Z, Pan L and
Zhang X: Individualized lipid profile in urine-derived
extracellular vesicles from clinical patients with Mycobacterium
tuberculosis infections. Front Microbiol. 15:14095522024.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Li L, Cheng Y, Emrich S and Schorey J:
Activation of endothelial cells by extracellular vesicles derived
from Mycobacterium tuberculosis infected macrophages or
mice. PLoS One. 13:e01983372018. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Arya R, Dabral D, Faruquee HM, Mazumdar H,
Patgiri SJ, Deka T, Basumatary R, Kupa RU, Semy C, Kapfo W, et al:
Serum small extracellular vesicles proteome of tuberculosis
patients demonstrated deregulated immune response. Proteomics Clin
Appl. 14:e19000622020. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Carranza C, Herrera MT, Guzmán-Beltrán S,
Salgado-Cantú MG, Salido-Guadarrama I, Santiago E, Chávez-Galán L,
Gutiérrez-González LH and González Y: A dual marker for monitoring
MDR-TB treatment: Host-derived miRNAs and M.
tuberculosis-derived RNA sequences in serum. Front Immunol.
12:7604682021. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Chen GZ, Fan FF, Deng SQ, Xia XY, Bian XC,
Ren YH and Wei L: Extraction of extracelluar vesicles derived from
Mycobacterium tuberculosis and their effect on the
production of reactive oxygen species and expression of
inflammatory factors in mouse bone marrow-derived dendritic cells.
Sichuan Da Xue Xue Bao Yi Xue Ban. 54:122–127. 2023.(In Chinese).
PubMed/NCBI
|
|
130
|
García-Martínez M, Vázquez-Flores L,
Álvarez-Jiménez VD, Castañeda-Casimiro J, Ibáñez-Hernández M,
Sánchez-Torres LE, Barrios-Payán J, Mata-Espinosa D, Estrada-Parra
S, Chacón-Salinas R, et al: Extracellular vesicles released by
J774A.1 macrophages reduce the bacterial load in macrophages and in
an experimental mouse model of tuberculosis. Int J Nanomedicine.
14:6707–6719. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Alvarez-Jiménez VD, Leyva-Paredes K,
García-Martínez M, Vázquez-Flores L, García-Paredes VG,
Campillo-Navarro M, Romo-Cruz I, Rosales-García VH,
Castañeda-Casimiro J, González-Pozos S, et al: Extracellular
vesicles released from Mycobacterium tuberculosis-infected
neutrophils promote macrophage autophagy and decrease intracellular
mycobacterial survival. Front Immunol. 9:2722018. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Cheng Y and Schorey JS: Extracellular
vesicles deliver Mycobacterium RNA to promote host immunity
and bacterial killing. EMBO Rep. 20:e466132019. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Jindal N, Sharma P, Punia S, Dass M,
Anthwal D, Gupta RK, Bhalla M, Singhal R, Behera A, Yadav R, et al:
Utility of pleural fluid-derived extracellular vesicles as a source
of Mycobacterium tuberculosis antigens MPT51 and MPT64 for
pleural TB diagnosis: A proof-of-concept study. Tuberculosis
(Edinb). 150:1025782025. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Nguyen VVT, Welsh JA, Tertel T, Choo A,
van de Wakker SI, Defourny KAY, Giebel B, Vader P, Padmanabhan J,
Lim SK, et al: Inter-laboratory multiplex bead-based surface
protein profiling of MSC-derived EV preparations identifies MSC-EV
surface marker signatures. J Extracell Vesicles. 13:e124632024.
View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Zhang J, Qiu X, Lei Y, Chen H, Wu D, Wang
T, Sui X, Xiao J, Jiang C, Zhang H, et al: Engineered EVs from
LncEEF1G-overexpressing MSCs promote fibrotic liver regeneration by
upregulating HGF release from hepatic stellate cells. Exp Mol Med.
57:584–600. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Zhu L, Wang Q, Guo M, Fang H, Li T, Zhu Y,
Jiang H, Xiao P and Hu M: Mesenchymal stem cell-derived exosomes in
various chronic liver diseases: Hype or hope? J Inflamm Res.
17:171–189. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Boysen AT, Whitehead B, Revenfeld ALS,
Gupta D, Petersen T and Nejsum P: Urine-derived stem cells serve as
a robust platform for generating native or engineered extracellular
vesicles. Stem Cell Res Ther. 15:2882024. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Levy D, Jeyaram A, Born LJ, Chang KH,
Abadchi SN, Hsu ATW, Solomon T, Aranda A, Stewart S, He X, et al:
Impact of storage conditions and duration on function of native and
cargo-loaded mesenchymal stromal cell extracellular vesicles.
Cytotherapy. 25:502–509. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Gao WY, Boonyarat C, Samar N, Sethabouppha
B and Waiwut P: Multiomics analysis of molecules associated with
cancer in mesenchymal-stem-cell-(MSC)-derived exosome-treated
hepatocellular carcinoma cells. Curr Issues Mol Biol.
46:13296–13310. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Gong S, Li N, Peng Q, Wang F, Du R, Zhang
B, Wang J, Han L, Zhang Y, Ning Z, et al: A scalable platform for
EPSC-Induced MSC extracellular vesicles with therapeutic potential.
Stem Cell Res Ther. 16:4262025. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Elnegris HM, Abdelrahman AA and El-Roghy
ES: The potential therapeutic effects of exosomes derived from bone
marrow mesenchymal stem cells on ileum injury of a rat sepsis model
(histological and immunohistochemical study). Ultrastruct Pathol.
48:274–296. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Mardpour S, Hamidieh AA, Taleahmad S,
Sharifzad F, Taghikhani A and Baharvand H: Interaction between
mesenchymal stromal cell-derived extracellular vesicles and immune
cells by distinct protein content. J Cell Physiol. 234:8249–8258.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
You J, Fu Z and Zou L: Mechanism and
potential of extracellular vesicles derived from mesenchymal stem
cells for the treatment of infectious diseases. Front Microbiol.
12:7613382021. View Article : Google Scholar : PubMed/NCBI
|