Ulcerative colitis (UC) is a chronic, non-specific
inflammatory bowel disease (IBD) that is characterized by
persistent inflammation and ulceration of the colonic mucosa.
Patients with UC typically present with abdominal pain, diarrhea
and stools containing blood and mucus (1). Although the precise pathogenesis of
UC remains to be fully elucidated, it is generally understood to
result from the multifactorial interplay of environmental factors,
immune dysregulation, gut microbiota imbalance and genetic
predispositions (2).
Histopathological features include inflammatory cell infiltration
within the lamina propria and excessive secretion of
proinflammatory cytokines such as IL-6 and TNF-α (3). Globally, the prevalence of UC is
~10.6 per 100,000 individuals (4),
driven by an aging population and improved diagnostic recognition.
With disease progression, the risk of colonic malignancy also
increases (5). In Asian
populations, westernization of diets has been identified as a key
contributor to the increasing prevalence. Evidence from a large
cohort of 500,000 Chinese participants highlights two high-risk
dietary patterns associated with increased risk of UC: i) The
traditional Northern diet, consisting of high wheat and low rice
intake; and ii) a modern diet rich in animal-based foods and
fruits. Notably, frequent egg consumption was associated with
increased susceptibility to late-onset UC, whereas spicy food
intake showed a protective association (6). Similarly, a Japanese study has
attributed the pathogenesis of UC to reduced intake of dietary
fiber, fermented foods containing probiotics and plant-based
nutrients, accompanied by higher consumption of refined
carbohydrates and animal fats (7).
Pan-Asian analyses further support these findings, indicating that
‘Westernized’ diets, characterized by elevated refined sugar, red
meat and linoleic acid, promote UC development, while fiber-rich
fruits and vegetables exert protective effects (8).
Mechanistically, these dietary factors are
considered to impair gut barrier integrity, alter gut microbial
composition and amplify proinflammatory signaling cascades. Acute
severe colitis is experienced by ~15% of patients with UC, with
>30% ultimately requiring colectomy (9). Therapeutically, monoclonal antibodies
targeting key cytokines have been increasingly used to manage
corticosteroid-refractory UC (10–12).
However, their long-term efficacy is limited by adverse effects and
the gradual loss of therapeutic response in some patients (13), underscoring the need for continued
investigation into UC pathogenesis and the development of novel
treatment strategies.
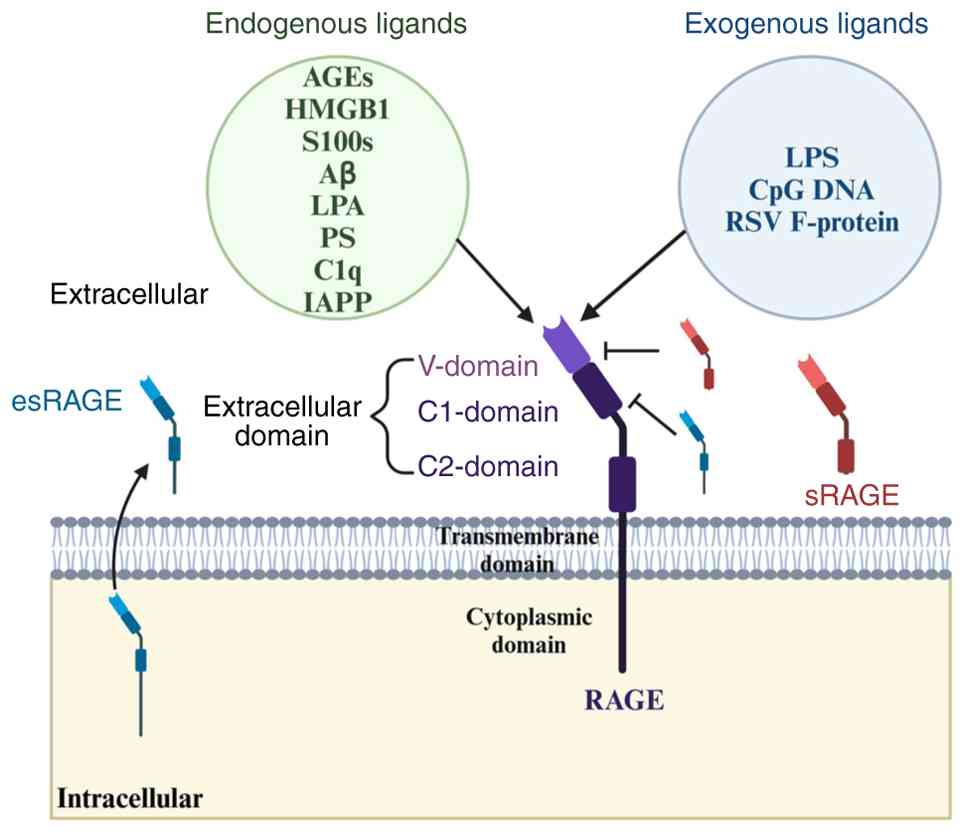
The receptor for advanced glycation end products
(RAGE), first identified in 1992, is a transmembrane protein
belonging to the immunoglobulin (Ig) receptor superfamily (10). Structurally, RAGE comprises three
domains, extracellular, transmembrane and intracellular, and exists
in both membrane-bound and soluble forms. Owing to its unique
molecular configuration, RAGE is one of the few pattern recognition
receptors capable of binding both pathogen-associated molecular
patterns and damage-associated molecular patterns (DAMPs).
Initially discovered for its ability to interact with AGEs
implicated in diabetes (14), RAGE
has since been shown to bind a wide array of non-glycation ligands,
including protein S100 (S100) calgranulins, high-mobility group box
1 (HMGB1) and amyloid β (Aβ) protein (15). Although this is most abundantly
expressed in lung tissue, RAGE signaling contributes to the
pathogenesis of numerous chronic inflammatory diseases affecting
multiple organs, including diabetic vascular complications
(16), cardiovascular disease
(17), cancer (18), Alzheimer's disease (AD) (19) and various infection-related and
autoimmune disorders (20).
Previous studies have revealed elevated RAGE expression in UC
(21) and Crohn's disease
(22). Furthermore, several DAMP
ligands, including calprotectin (23), lactoferrin (24), S100A calgranulins (25) and HMGB1 (26), are recognized biomarkers of disease
activity and prognosis in IBD. Other receptor systems implicated in
UC, including toll-like receptors (TLRs) (27), C-type lectin receptors (28) and nucleotide-binding
oligomerization domain-like receptors (29), have been well characterized.
However, the contribution of RAGE to UC pathogenesis and its
potential as a therapeutic target remain inadequately understood.
The present review, therefore, highlights the pathobiological
importance of RAGE in UC, summarizing previous advances in clinical
and translational research and exploring its implications for
targeted therapeutic intervention.
Currently, the primary pharmacologic options for
treating UC include corticosteroids, aminosalicylates,
immunomodulators and antibiotics. However, the widespread clinical
application of these drugs is constrained by high costs, notable
toxicities and frequent disease recurrence (30). Although a previous study has
explored dose de-escalation strategies using immunomodulators such
as thiopurines and methotrexate, these regimens have shown limited
efficacy in patients with moderate-to-severe UC compared with
biologic therapies and small-molecule targeted drugs (31). To address these limitations, a new
generation of small-molecule targeted therapies has been developed,
offering distinct advantages such as high oral bioavailability,
reduced risk of immunogenicity and lower manufacturing costs
(32). These agents represent a
promising alternative to conventional biologics in UC management.
Approaches primarily targeting cellular subsets or broad immune
modulation are beyond the scope of the present discussion and are
therefore not included in the classification stated in the present
review.
The IL-12 family has unique heterodimeric cytokines,
including the IL-12, IL-23, IL-27 and IL-35 cytokines; this
heterodimeric property confers a unique set of connectivity and
functional interactions in these cytokines (33). Despite their similar structural
features, the members of the IL-12 family have distinct properties.
Among them, IL-12 and IL-23 play key roles in intestinal
homeostasis and inflammation and are involved in the pathogenesis
of IBD (34). The main
contributions of IL-12 and IL-23 to UC pathogenesis are the
induction of T helper (Th)1 and Th17 cell differentiation,
respectively (35); thus, the
inflammatory effects of IL-12 and IL-23 provide a theoretical
rationale for the development of blocking agents targeting UC.
Inhibitors targeting IL-12/23 attenuate the Th1/Th17-mediated
adaptive immune response, which is a notable contributor to UC
pathogenesis (36).
JAK is a non-receptor tyrosine protein kinase
located downstream of different inflammatory cytokines. As an
intracellular-signaling mediator, JAK interacts with STAT so as to
induce phosphorylation of STAT and activate the target
transcription molecules (11). The
JAK/STAT pathway is an important signaling pathway that allows
extracellular proinflammatory cytokines to relay inflammatory
signals to the nucleus via membrane receptors. Several cytokines
that are notably associated with immunity and intestinal stromal
cell homeostasis, such as IL-6, IL-10, IL-2 and IL-22, as well as
cytokines that act as mediators of pathological responses in UC,
such as IFN-γ, IL-12, IL-23 and IL-9, are dependent on
JAK/STAT-mediated signaling (37,38).
When JAK/STAT signaling is blocked, the nucleus cannot receive
extracellular chemical signals, which reduces inflammation. Thus,
JAK inhibitors can simultaneously block multiple inflammatory
pathways. Demonstrably, JAK inhibitors can broadly affect the
immunopathogenesis of UC, influencing factors such as the
inflammatory response, intestinal epithelial barrier and fibrosis
(11).
The diversity of therapeutic targets identified thus
far underscores the multifactorial nature of UC pathogenesis.
Agents targeting cytokines, such as TNF-α (39–52)
and IL-12/23 (40,53–56),
intracellular signaling pathways, such as the JAK/STAT pathway
(57–67), epithelial barrier integrity, such
as mucin-2, phosphodiesterase 4 (PDE4) inhibitors (68,69)
and lymphocyte trafficking, for example sphingosine 1-phosphate
receptor (70–74), have demonstrated clinical efficacy
in mediating UC (11,12,72,75),
with the detailed clinical trial data of these targeted agents
systematically summarized in Table
SI (39–56,58–74);
however, these therapies typically modulate discrete components of
the inflammatory cascade or adaptive immune response. By contrast,
RAGE represents a distinct signaling axis, primarily activated by
DAMPs generated during tissue stress and injury (76). Functioning as a sensor of
persistent inflammation and cellular injury, RAGE activation
amplifies oxidative stress, perpetuates chronic inflammation and
contributes to fibrotic remodeling, key pathological processes not
fully addressed by current biologics or small molecule inhibitors
(77). The following sections
examine the mechanistic role of RAGE and its ligands in UC
pathogenesis and discuss the potential of targeting this axis as a
novel therapeutic strategy.
The RAGE gene is located on chromosome 6 within the
major histocompatibility complex class III region, which harbors
numerous genes that are important to both adaptive and innate
immune function (78). RAGE is a
50–55 kDa type I transmembrane glycoprotein composed of an
extracellular region containing three Ig-like domains, a variable,
a constant 1 and a constant 2 domain (Fig. 1). Ligand binding predominantly
occurs within the variable domain (79). The extracellular region adjoins a
single transmembrane segment, followed by a short, charged
cytoplasmic tail, the latter being important for intracellular
signal transduction (80,81). Notably, truncation of this
cytoplasmic domain abolishes downstream RAGE signaling and markedly
attenuates RAGE-mediated pathological effects (82,83).
In addition to the membrane-bound full-length RAGE (FL-RAGE), two
soluble isoforms, soluble RAGE (sRAGE) and endogenous secretory
RAGE (esRAGE), have been identified. The former arises from
proteolytic cleavage of membrane-bound RAGE, whereas esRAGE is
produced through alternative mRNA splicing. Both soluble forms can
bind circulating RAGE ligands, acting as ‘decoy’ receptors that
prevent ligand engagement with FL-RAGE and thereby dampen
inflammatory signaling (84,85).
Interactions between cell-surface RAGE and its
ligands initiate a cascade of intracellular events that promote
proinflammatory phenotypes both in vitro and in vivo,
implicating this pathway in the pathophysiology of numerous
diseases (84). In addition to
binding endogenous DAMPs, RAGE can also be activated by
pathogen-associated molecular patterns such as bacterial
lipopolysaccharide (86), viral
proteins (87), parasite-derived
proteins (88) and bacterial DNA
(76). Ligand engagement activates
multiple downstream signaling networks, including the
diaphanous-related formin 1 (89),
MAPK (90), PI3K/Akt (91) and toll-interleukin 1 receptor
domain-containing adaptor protein (92) pathways, culminating in NF-κB
activation. Notably, RAGE signaling forms a self-sustaining
positive feedback loop with NF-κB: Inflammatory stimuli activate
NF-κB, which subsequently upregulates RAGE expression, further
amplifying and prolonging inflammatory responses (93).
RAGE expression has been detected in diverse cell
types, including endothelial cells (ECs), vascular smooth muscle
cells, monocytes and macrophages, granulocytes, adipocytes and
various tumor cells (94).
Aberrant RAGE expression has also been implicated in the
pathogenesis of numerous diseases, such as diabetes (95), atherosclerosis (96), rheumatoid arthritis (97), AD (19), cardiovascular diseases (98) and chronic immune-mediated and
inflammatory disorders (84).
Furthermore, RAGE has been associated with tumor initiation and
progression across multiple types of cancer (99).
The binding of RAGE ligands to membrane-bound RAGE
initiates receptor activation and triggers a cascade of
intracellular signaling events. Increasing evidence identifies RAGE
as an important mediator in the pathogenesis of numerous chronic
inflammatory disorders (100–102). Multiple molecular mechanisms
appear to contribute to disease initiation and persistence in
patients with UC, particularly those amplifying proinflammatory
signaling (103,104). Notably, both RAGE and its ligands
exhibit elevated expression in intestinal epithelial cells from
patients with UC and experimental colitis models, and are localized
predominantly in inflamed mucosal regions (105–108). This interaction of RAGE and its
ligands plays an important role in sustaining mucosal injury and
perpetuating intestinal inflammation.
In UC, chronic inflammation and mucosal damage
promote the accumulation of AGEs, which interact with RAGE to
exacerbate inflammatory signaling. AGEs are naturally formed during
aging however, their formation is accelerated under conditions of
hyperglycemia and oxidative stress, such as in diabetes mellitus
(81). The binding of AGEs to RAGE
activates the NF-κB and MAPK signaling pathways (90), stimulating the release of
proinflammatory cytokines, including IL-6 and TNF-α, thereby
aggravating mucosal inflammation and driving UC progression
(21). AGEs are generated through
the Maillard reaction, a non-enzymatic process in which reducing
sugars react with proteins, lipids or DNA (109). This reaction proceeds from the
formation of reversible Schiff bases and Amadori intermediates to
stable, irreversible AGEs via oxidative rearrangements (110). Given that diabetes is a frequent
comorbidity among patients with UC (111), strategies aimed at glycemic
control, through dietary interventions, hypoglycemic agents or
inhibition of glycation reactions, may effectively suppress AGE
production and attenuate intestinal inflammation. Inhibitors of AGE
synthesis, including aminoguanidine, which has been evaluated in
acetic acid- (112) and
TNF-α-induced rat and murine colitis models (113), metformin, tested in oxazolone-
(114), acetic acid- (115) and dextran sulfate sodium
(DSS)-induced models (116), and
pioglitazone, previously assessed in acute and chronic DSS-driven
murine colitis (117,118), consistently alleviate
inflammation and reduce colonic mucosal injury across experimental
settings. Although some clinical evidence supports these findings,
translational validation remains limited (119).
Another therapeutic strategy involves preventing
AGE-RAGE binding, which can be achieved via soluble receptor
analogs, receptor antagonists or post-receptor signaling
inhibitors. In UC, blockade of this binding suppresses inflammatory
cascades, preserves mucosal integrity and modulates immune
responses (120). Statins, for
instance, interrupt the positive feedback loop between the AGE-RAGE
axis and C-reactive protein expression, thereby reducing
inflammation and oxidative stress (121). Statins, as a potential preventive
and therapeutic strategy for UC, have been shown to attenuate
colitis severity in animal models (122–124). However, clinical investigations
evaluating the disease-modifying and preventive potential of
statins in UC have yielded limited and inconsistent findings
(125–127). Current epidemiologic evidence is
insufficient to support the use of statins for the prevention or
treatment of UC (128).
Low-molecular-weight heparin acts as a competitive
RAGE antagonist by displacing AGE ligands, leading to notable
anti-inflammatory and antioxidant effects in UC, as supported by
clinical evidence (129–131). Collectively, AGEs contribute to
UC pathogenesis through multiple mechanisms that sustain
inflammation and tissue injury. Elucidating these pathways may
yield novel therapeutic targets and provide mechanistic insight
into AGE-RAGE-mediated intestinal pathology.
HMGB1 was the second ligand identified to bind RAGE
following the discovery of AGEs (132). HMGB1 is a highly-conserved
nuclear protein comprising two N-terminal DNA-binding domains and
an acidic C-terminal domain. It is broadly expressed in multiple
tissues, including the brain, heart, lungs, liver, spleen, kidneys
and lymphatic organs, and can be localized in the nucleus,
cytoplasm and extracellular milieu (133). Under resting conditions, HMGB1
predominantly resides within the nucleus; however, upon stimulation
by lipopolysaccharide, 5′-C-phosphate-G-3′ DNA, TNF-α or IL-1,
various immune cells, particularly monocytes and neutrophils,
actively secrete HMGB1 (134).
Passive release also occurs from necrotic cells (134). Both actively and passively
released HMGB1 can bind high-affinity receptors such as TLR2, TLR4
and RAGE on target cells (135–137). These interactions drive immune
cell activation, cytokine release and downstream inflammatory
cascades (138,139). RAGE-HMGB1 binding activates
multiple tumor-associated signaling pathways, including the ERK1/2,
p38 MAPK and NF-κB pathways, thereby promoting cancer progression
and metastasis (140).
In the context of UC, blockade of HMGB1/TLR4 or
HMGB1/RAGE signaling markedly attenuates inflammation in
experimental models (141,142).
Clinically, fecal HMGB1 levels correlate strongly with disease
severity and mucosal activity in UC (143–145), highlighting its promise as a
non-invasive biomarker for both overt and subclinical intestinal
inflammation (26,146). Therapeutic strategies targeting
HMGB1 remain in early development, but preclinical evidence
suggests potential efficacy. Pharmacological agents such as
dapagliflozin (141) and several
natural compounds, including isoliquiritin (147), 20(S)-protopanaxadiol saponins
(148) and matrine (149), have demonstrated anti-HMGB1
effects in experimental models. Nonetheless, the majority of these
interventions remain confined to animal experimental stages
(147,148,150), and rigorous clinical validation
is required to confirm their safety, pharmacokinetics and
therapeutic benefit. To date, HMGB1-targeted therapies have not
entered routine clinical practice, although ongoing advances
warrant close attention to emerging evidence.
The S100 protein family constitutes one of the
largest subgroups of calcium-binding proteins, exhibiting distinct
biological functions and tissue-specific expression patterns
(151). Extracellular S100
proteins interact with several receptors, including RAGE, TLR4,
fibroblast growth factor receptor 1 and G-protein coupled receptors
(152). Through these
interactions, they promote the transcription of proinflammatory
mediators such as TNF-α, IL-1β, IL-6 and IL-8, induce reactive
oxygen species generation and regulate apoptosis (102). Although RAGE binding is
considered a common feature of numerous S100 proteins (153), only specific members, including
S100A1, S100A2, S100A4-9, S100A11-13, S100B and S100P, have been
experimentally validated as RAGE ligands in vivo (154). Among these, S100A8/A9, also known
as calprotectin, is predominantly expressed by neutrophils and is
markedly elevated in IBD. The notable stability of calprotectin in
fecal samples has established it as a robust biomarker of
intestinal inflammation (155,156). Functionally, calprotectin
contributes to epithelial barrier dysfunction by disrupting
cytoskeletal organization and tight junction integrity via TLR4-
and RAGE-dependent pathways in endothelial and epithelial cells
(157). Calprotectin further
compromises EC integrity by downregulating junctional proteins and
increasing vascular permeability (158). The quinoline-3-carboxamide
derivative ABR-215757 binds S100A9 and S100A8/A9 complexes,
blocking their interactions with TLR4 and RAGE, thereby exerting
potent anti-inflammatory effects across several experimental models
(159–161). Localized targeting of
calprotectin in UC mucosa using monoclonal antibodies represents a
promising therapeutic strategy, supported by successful outcomes in
preclinical models of atherosclerosis (162).
Beyond HMGB1 and S100 proteins, several other
endogenous RAGE ligands have been identified, including Aβ
(15), lysophosphatidic acid (LPA)
(163), phosphatidylserine
(164), complement protein C1q
(165) and islet amyloid
polypeptide (166). Although the
involvement of DAMPs in UC remains insufficiently characterized,
emerging evidence suggests that they contribute to disease
pathogenesis or to comorbid conditions associated with UC. For
instance, modulation of adrenergic receptor signaling can preserve
intestinal barrier integrity in UC partly through the presenilin
1/β-secretase-1/Aβ axis, conferring antioxidant, anti-inflammatory
and antifibrotic effects (164).
Similarly, bamboo leaf flavonoids downregulate Aβ expression in the
brain, ameliorating both AD and UC-like inflammation (167). Furthermore, inhibition of the
autotaxin/LPA axis reduces chronic intestinal inflammation by
suppressing Th17 cell differentiation (168,169). Despite these insights, to the
best of our knowledge, no current studies have directly
demonstrated that these ligands exert anti-UC effects specifically
through RAGE signaling. Further mechanistic and translational
investigations are therefore warranted to delineate their roles
within RAGE-dependent inflammatory networks.
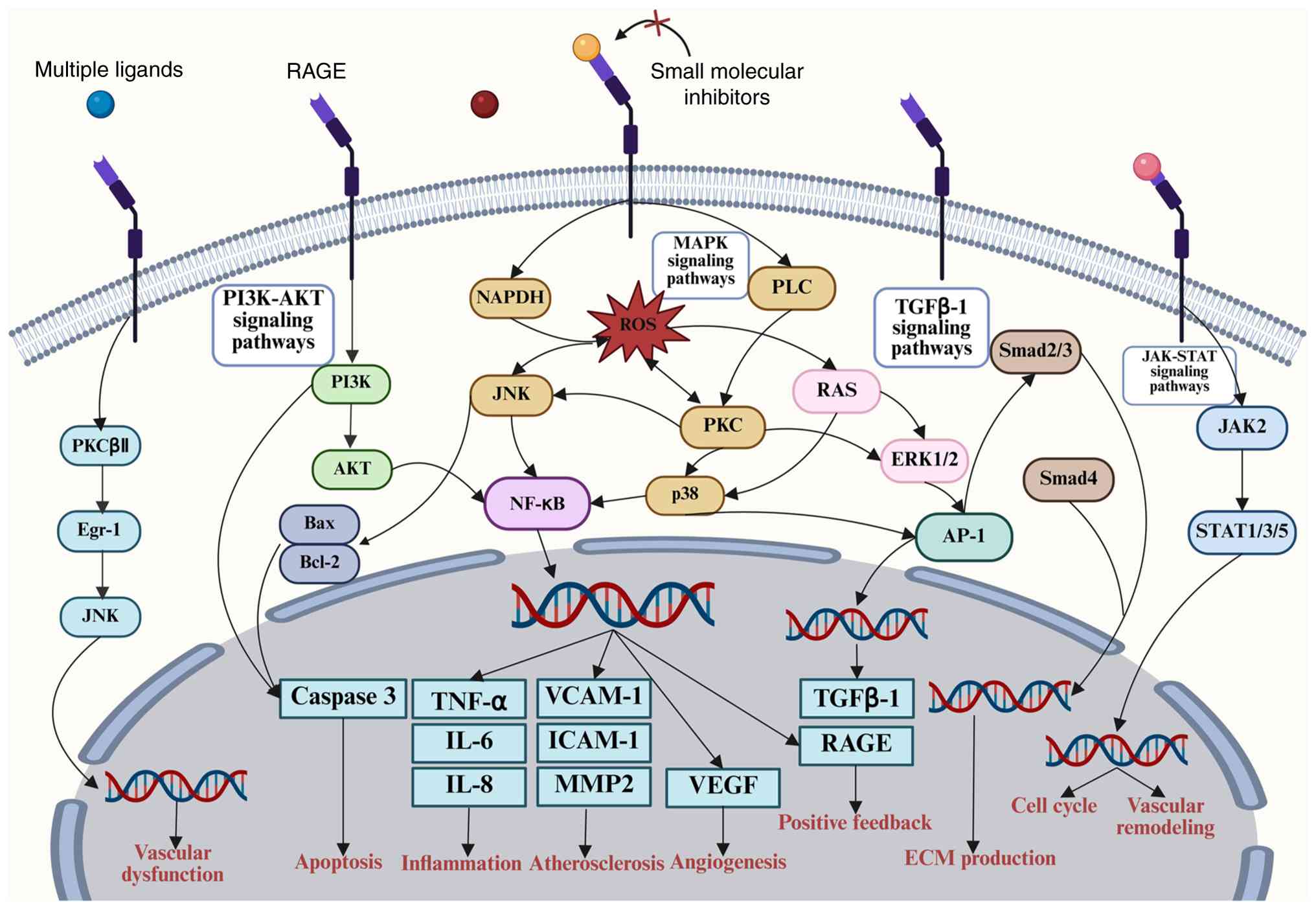
Ligand binding to RAGE activates multiple downstream
signaling cascades implicated in the pathogenesis of UC (Fig. 2). These include the Ras/MEK/ERK1/2S
(170), stress-activated protein
kinase/JNK (171), MAPK/p38
(172), PI3K/AKT (173), JAK/STAT (174), Rho GTPase (175) and vascular endothelial growth
factor (VEGF) (176) pathways.
Collectively, these cascades activate the transcription factors
NF-κB, STAT3, activator protein 1 and early growth response-1,
which subsequently induce the synthesis and secretion of vascular
cell adhesion protein 1, intercellular adhesion molecule 1, matrix
metalloproteinase-2, IL-1, IL-6 and TNF-α (15,177–180). DAMP-mediated RAGE activation
drives UC pathogenesis via three interconnected mechanisms: i) Rho
GTPase modulation alters gut microbiota composition and
metabolites, promoting disease progression (175); ii) JAK/STAT signaling regulates
inflammatory mediators and immune cell activation (181); and iii) MAPK pathway activation
induces mitochondrial dysfunction (182), autophagy (183), oxidative stress (184) and apoptosis (185). Collectively, these pathways
underscore the central role of RAGE in UC pathogenesis by
orchestrating inflammation, disrupting intestinal barrier integrity
and dysregulating immune responses. Thus, identifying and
characterizing RAGE-associated downstream targets may offer novel
insights into UC pathophysiology and reveal new therapeutic
strategies.
Angiogenesis, the formation of new capillaries from
pre-existing blood vessels in adult tissues, is a multistep process
involving EC proliferation, migration, differentiation, lumen
formation and maturation, ultimately expanding the microvascular
network (186,187). This process represents a
double-edged sword: While important for wound healing and tissue
repair, it also contributes to pathological tissue remodeling in
cancer and chronic inflammatory diseases. Beyond oncology,
angiogenesis plays a key role in several chronic inflammatory
disorders such as atherosclerosis, rheumatoid arthritis and
psoriasis (188–193). Although angiogenesis has been
implicated in UC (194),
quantitative characterization of mucosal vascular remodeling during
active inflammation remains limited. However, increasing evidence
from clinical and experimental studies demonstrates notable
angiogenesis in UC and Crohn's disease, with elevated vascular
density associating with IBD severity (195–200).
In UC, the colonic mucosa undergoes recurrent cycles
of ulceration and regeneration. This dynamic process increases
local neovascularization and enhances the recruitment of
leukocytes, nutrients and oxygen to inflamed regions (194,201). The expansion of the vascular
network during inflammation is accompanied by notable structural
and functional changes in blood vessels. Functionally, these
changes promote inflammation through several mechanisms: i)
Enhanced leukocyte infiltration; ii) augmented nutrient delivery
that sustains metabolically active immune responses; and iii) EC
activation, which drives local cytokine, chemokine and
metalloproteinase production (202). Consequently, angiogenesis and
inflammation form a self-perpetuating, chronic cycle.
Hypoxia serves as a central trigger in
inflammation-induced angiogenesis. Inflammatory and immune cells
migrate to hypoxic sites, where they release angiogenic mediators,
including growth factors, cytokines, proteases and nitric oxide,
which stimulate EC activation and vascular remodeling. The
resulting neovascularization amplifies inflammation by increasing
the delivery of oxygen, nutrients and inflammatory mediators to
affected tissues (203,204). In UC, this excessive angiogenesis
enlarges the endothelial surface area and enhances vascular
permeability, promoting plasma extravasation and worsening IBD
severity (199). Notably, the
neovessels formed during active UC differ from those generated
during normal physiological angiogenesis. UC-induced vessels are
structurally immature, highly permeable, poorly perfused, prone to
stenosis and thrombosis and display hypersensitivity to growth
factors, features that exacerbate mucosal injury and sustain
chronic inflammation (205,206). Collectively, inflammation and
angiogenesis in UC exist in a reciprocal, self-amplifying
relationship that drives disease progression (207–210).
Angiogenesis is coordinated through a balance of
pro- and anti-angiogenic molecules. Although the VEGF family is a
central player, the angiogenic cascade in UC involves the complex
interplay of multiple factors, receptors and isoforms (211). VEGF is the most extensively
studied angiogenic factor in IBD, with elevated levels in
circulation and the intestinal mucosa associating with disease
activity (212,213). The VEGF family comprises several
isoforms, such as VEGF-A, -B, -C, -D and -E, and placental growth
factor (PlGF), which exert their effects primarily by binding to
three tyrosine kinase receptors: VEGFR1, VEGFR2 and VEGFR3
(214). Although VEGFR2 is the
primary mediator of pathological angiogenesis, driving EC
proliferation, permeability and survival, the roles of other
receptors and isoforms are increasingly recognized. VEGFR1, which
has a higher affinity for VEGF-A than other isoforms but weaker
tyrosine kinase activity than other VEGFRs, acts as a decoy
receptor, thereby fine-tuning the availability of VEGF for VEGFR2
(215). The VEGF2 ligands, VEGF-B
and PlGF, are upregulated in inflammation and modulate
VEGFR1-specific signaling, influencing monocyte recruitment and
inflammatory angiogenesis (216).
Furthermore, the neuropilin (NRP) co-receptors NRP1 and NRP2, which
bind specific VEGF-A isoforms, enhance VEGFR2-signaling complex
formation and signaling potency (217).
The specific splice variants of VEGF-A are notably
important. The pro-angiogenic VEGF-Axxx isoforms, such as
VEGF-A164, dominate in the inflammatory milieu of UC (218). By contrast, the anti-angiogenic
VEGF-Axxxb isoforms, such as VEGF-A164b, are often downregulated,
creating a permissive environment for neovascularization (57). Previous evidence suggests that
restoring the balance of isoforms toward VEGF-A164b can ameliorate
experimental colitis (57).
VEGF-C, primarily known for lymphangiogenesis via VEGFR3, can also
contribute to blood vessel angiogenesis in chronic inflammation
(219).
Under the inflammatory environment of UC, cytokines
such as TNF-α and IL-1β can activate the RAGE-signaling pathway,
which, in turn, promotes the expression and release of VEGF
(21). Alterations in the
extracellular matrix and oxidative stress, both hallmarks of UC,
further amplify RAGE activation and its downstream pro-angiogenic
signals (77,177,220). The downstream signaling of VEGFR2
exhibits notable crosstalk with RAGE-activated pathways. VEGFR2
activates the ERK1/2/MAPK pathway via Ras, which is important for
EC proliferation and migration (221). VEGFR2 also activates PI3K,
leading to the activation of AKT, a central regulator of cell
survival, and small GTPases such as Rac, which guides cytoskeletal
dynamics and EC motility (222,223). Notably, both RAGE and VEGFR2
signaling converge on NF-κB activation, creating a feed-forward
loop that amplifies the production of proinflammatory cytokines and
sustains the angiogenic response (224). Supporting this interconnection,
neutralizing RAGE has been shown to markedly inhibit AGE-induced
activation of both the VEGF and NF-κB pathways (224), while RAGE silencing also inhibits
VEGF expression and angiogenesis in colorectal cancer models
(225).
Beyond VEGF, other angiogenic factor families are
active in UC. The angiopoietin (Ang)/Ang-1 receptor (Tie2) system
is important for vascular maturation and stability. Ang-1, produced
by pericytes, activates Tie2 to promote vessel quiescence and
integrity (226). In UC, the
balance is shifted toward Ang-2, which is stored in and released
from endothelial Weibel-Palade bodies upon inflammatory stimuli.
Ang-2 acts as a context-dependent antagonist of Tie2, destabilizing
vessels, priming them to be more responsive to VEGF and promoting
vascular leakage and inflammation (227). Platelet-derived growth factors
(PDGFs), particularly PDGF-BB produced by ECs and platelets, are
important for recruiting pericytes and vascular smooth muscle cells
in order to stabilize newly formed vessels. PDGF-BB is also
associated with M1 macrophages and has demonstrated notable
potential diagnostic value for active IBD (228).
Despite the central role of VEGF and its synergistic
partners in UC angiogenesis, drug development targeting VEGF has
predominantly focused on oncology. The majority of mechanistic
insights into the RAGE/VEGF axis are derived from diabetes-related
lesions and tumors (229–231). Consequently, there is a notable
gap in the comprehension of the precise mechanisms and therapeutic
potential involved in targeting the RAGE/VEGF network and its
associated angiogenic factors, specifically in UC. Future research
should therefore explore multi-target strategies that co-regulate
RAGE, specific VEGF isoforms and parallel pathways such as the
Ang-2/Tie2 pathway to achieve effective vascular normalization in
UC.
RAGE knockdown is safe in animal models, supporting
the feasibility of developing RAGE-targeted drugs (232). However, to the best of our
knowledge, no anti-RAGE drug has been approved for the treatment of
UC to date. Several investigations are exploring strategies that
interfere with RAGE activation, including antagonistic ligands,
RAGE gene deletion or small-molecule inhibitors (120,233,234). Among these, FPS-ZM1 (77,235) binds to the V-type domain of RAGE,
thereby preventing its interaction with multiple ligands such as
AGEs (84,236–238), HMGB1 (84,239–241), S100B (84,242) and Aβ (243). Other RAGE antagonists or
inhibitors (Table SII) (84,236–244), including azeliragon, alagebrium
and Tanshinone IIA (241,243,244), have been primarily developed for
other RAGE-related conditions, such as neurodegenerative disorders,
diabetes, tumors and inflammatory diseases (237,245,246). As aforementioned, angiogenesis is
a multifactorial process involving numerous cells, molecules and
signaling pathways, all of which may serve as potential therapeutic
targets in UC (21,208,210). Beyond conventional
anti-inflammatory agents, such as 5-aminosalicylic acid
derivatives, glucocorticoids and biologics, which indirectly
modulate VEGF-mediated angiogenesis (247,248), direct anti-VEGF therapies have
gained attention as potential adjunctive treatments. Representative
agents include receptor tyrosine kinase inhibitors, such as
axitinib and sunitinib, and monoclonal antibodies such as
bevacizumab, as well as investigational compounds such as
rapatinib, hesperidin and sorafenib (249–251). These agents inhibit VEGF
activity, suppress new vessel formation and consequently reduce
vascular density and permeability in the colonic mucosa, leading to
attenuation of inflammatory responses and tissue injury (247–251). Despite these promising
mechanisms, clinical outcomes of antiangiogenic drugs in UC remain
suboptimal, and further preclinical and clinical studies are
warranted to validate their efficacy and safety.
Although numerous innovative drugs and biologics
have been approved for UC treatment in previous years, the complex
multifactorial pathogenesis of UC continues to limit therapeutic
efficacy. Furthermore, individualized therapy in UC remains
underdeveloped, and the lack of reliable prognostic biomarkers
complicates the selection of optimal therapeutic regimens (40,47,50,54).
Although sRAGE and esRAGE function as both biomarkers and natural
inhibitors of RAGE-mediated pathology, their large recombinant
protein structures hinder their practical use as therapeutic
agents. Consequently, current research is increasingly focused on
developing small-molecule inhibitors that can selectively target
the extracellular ligand-binding domains of RAGE or its
intracellular signaling cascades (252–254).
Nevertheless, several key questions remain
regarding the long-term safety, pharmacodynamics and physiological
impact of RAGE blockade in humans. Further studies are required to
elucidate the molecular properties of potent RAGE inhibitors and to
clarify the systemic consequences of chronic RAGE inhibition. In
parallel, although VEGF-based antiangiogenic therapies have been
successfully used in clinical trials against various tumors
(255–257), their application in chronic
inflammatory diseases such as UC is still in its infancy.
Antiangiogenic therapy may represent a promising adjunctive
approach to preventing inflammation recurrence and persistence in
UC. Given that angiogenesis involves multiple interdependent steps,
regulated by growth and survival factors, adhesion molecules and
proteases, intestinal angiogenesis in UC provides a
multidisciplinary array of potential pharmacological targets
(258–260). Despite being generally considered
low in toxicity, antiangiogenic agents warrant comprehensive
evaluation to determine their effects on normal physiological
angiogenesis and mucosal healing dynamics. Consequently, combined
targeting of RAGE ligands and VEGF may offer a synergistic and
effective therapeutic strategy for UC. Translation to clinical
applications will require careful evaluation of human tolerability
and maintaining an appropriate physiological-pathological
angiogenesis balance.
Not applicable.
The present article is supported by the Anhui Provincial Health
Research Program (grant nos. AHWJ2023A20431 and AHWJ2023A20431) and
Clinical Research Project of Anhui University of Chinese Medicine
(grant no. 2024YFYLCZX35).
Not applicable.
CX designed the manuscript concept. CX and YL wrote
the manuscript. KH, LW, YH, DZ and FH participated in writing and
reviewing the manuscript. All authors read and approved the final
version of the manuscript. Data authentication is not
applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Nakase H, Uchino M, Shinzaki S, Matsuura
M, Matsuoka K, Kobayashi T, Saruta M, Hirai F, Hata K, Hiraoka S,
et al: Evidence-based clinical practice guidelines for inflammatory
bowel disease 2020. J Gastroenterol. 56:489–526. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kobayashi T, Siegmund B, Le Berre C, Wei
SC, Ferrante M, Shen B, Bernstein CN, Danese S, Peyrin-Biroulet L
and Hibi T: Ulcerative colitis. Nat Rev Dis Primers. 6:742020.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li J, Wang H, Zheng Z, Luo L, Wang P, Liu
K, Namani A, Jiang Z, Wang XJ and Tang X: Mkp-1 cross-talks with
Nrf2/Ho-1 pathway protecting against intestinal inflammation. Free
Radic Biol Med. 124:541–549. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hracs L, Windsor JW, Gorospe J, Cummings
M, Coward S, Buie MJ, Quan J, Goddard Q, Caplan L, Markovinović A,
et al: Global evolution of inflammatory bowel disease across
epidemiologic stages. Nature. 642:458–466. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Du L and Ha C: Epidemiology and
pathogenesis of ulcerative colitis. Gastroenterol Clin North Am.
49:643–654. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Song S, Wu Z, Lv J, Yu C, Sun D, Pei P,
Pan L, Yang L, Chen Y, Du H, et al: Dietary factors and patterns in
relation to risk of later-onset ulcerative colitis in Chinese: A
prospective study of 0.5 million people. Aliment Pharmacol Ther.
59:1425–1434. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kanai T, Matsuoka K, Naganuma M, Hayashi A
and Hisamatsu T: Diet, microbiota, and inflammatory bowel disease:
Lessons from Japanese foods. Korean J Intern Med. 29:409–415. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Prideaux L, Kamm MA, De Cruz PP, Chan FK
and Ng SC: Inflammatory bowel disease in Asia: A systematic review.
J Gastroenterol Hepatol. 27:1266–1280. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Turner D, Walsh CM, Steinhart AH and
Griffiths AM: Response to corticosteroids in severe ulcerative
colitis: A systematic review of the literature and a
meta-regression. Clin Gastroenterol Hepatol. 5:103–110. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim TW, Shin JS, Chung KS, Lee YG, Baek NI
and Lee KT: Anti-inflammatory mechanisms of koreanaside A, a lignan
isolated from the flower of forsythia koreana, against LPS-induced
macrophage activation and DSS-induced colitis mice: The crucial
role of AP-1, NF-κB, and JAK/STAT Signaling. Cells. 8:11632019.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Salas A, Hernandez-Rocha C, Duijvestein M,
Faubion W, McGovern D, Vermeire S, Vetrano S and Vande Casteele N:
JAK-STAT pathway targeting for the treatment of inflammatory bowel
disease. Nat Rev Gastroenterol Hepatol. 17:323–337. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Straatmijer T, Biemans VBC, Visschedijk M,
Hoentjen F, de Vries A, van Bodegraven AA, Bodelier A, de Boer NKH,
Dijkstra G, Festen N, et al: Superior effectiveness of tofacitinib
compared to vedolizumab in Anti-TNF-experienced ulcerative colitis
patients: A nationwide dutch registry study. Clin Gastroenterol
Hepatol. 21:182–191.e2. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Savelkoul EHJ, Thomas PWA, Derikx L, den
Broeder N, Römkens TEH and Hoentjen F: Systematic review and
Meta-analysis: Loss of response and need for dose escalation of
infliximab and adalimumab in ulcerative colitis. Inflamm Bowel Dis.
29:1633–1647. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Schmidt AM, Vianna M, Gerlach M, Brett J,
Ryan J, Kao J, Esposito C, Hegarty H, Hurley W, Clauss M, et al:
Isolation and characterization of two binding proteins for advanced
glycosylation end products from bovine lung which are present on
the endothelial cell surface. J Biol Chem. 267:14987–14997. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jangde N, Ray R and Rai V: RAGE and its
ligands: From pathogenesis to therapeutics. Crit Rev Biochem Mol
Biol. 55:555–575. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yamagishi S, Fukami K and Matsui T:
Crosstalk between advanced glycation end products (AGEs)-receptor
RAGE axis and dipeptidyl peptidase-4-incretin system in diabetic
vascular complications. Cardiovasc Diabetol. 14:22015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang L, He J, Wang J, Liu J, Chen Z, Deng
B, Wei L, Wu H, Liang B, Li H, et al: Knockout RAGE alleviates
cardiac fibrosis through repressing endothelial-to-mesenchymal
transition (EndMT) mediated by autophagy. Cell Death Dis.
12:4702021. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rigiracciolo DC, Nohata N, Lappano R,
Cirillo F, Talia M, Adame-Garcia SR, Arang N, Lubrano S, De
Francesco EM, Belfiore A, et al: Focal Adhesion Kinase
(FAK)-Hippo/YAP transduction signaling mediates the stimulatory
effects exerted by S100A8/A9-RAGE system in triple-negative breast
cancer (TNBC). J Exp Clin Cancer Res. 41:1932022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Guan L, Mao Z, Yang S, Wu G, Chen Y, Yin
L, Qi Y, Han L and Xu L: Dioscin alleviates Alzheimer's disease
through regulating RAGE/NOX4 mediated oxidative stress and
inflammation. Biomed Pharmacother. 152:1132482022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li Y, Qin M, Zhong W, Liu C, Deng G, Yang
M, Li J, Ye H, Shi H, Wu C, et al: RAGE promotes dysregulation of
iron and lipid metabolism in alcoholic liver disease. Redox Biol.
59:1025592023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Duan ZL, Wang YJ, Lu ZH, Tian L, Xia ZQ,
Wang KL, Chen T, Wang R, Feng ZY, Shi GP, et al: Wumei Wan
attenuates angiogenesis and inflammation by modulating RAGE
signaling pathway in IBD: Network pharmacology analysis and
experimental evidence. Phytomedicine. 111:1546582023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang YF, Li QP, Dou YX, Wang TT, Qu C,
Liang JL, Lin ZX, Huang XQ, Su ZR, Chen JN and Xie YL: Therapeutic
effect of Brucea javanica oil emulsion on experimental Crohn's
disease in rats: Involvement of TLR4/NF-κB signaling pathway.
Biomed Pharmacother. 114:1087662019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jukic A, Bakiri L, Wagner EF, Tilg H and
Adolph TE: Calprotectin: From biomarker to biological function.
Gut. 70:1978–1988. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gisbert JP, McNicholl AG and Gomollon F:
Questions and answers on the role of fecal lactoferrin as a
biological marker in inflammatory bowel disease. Inflamm Bowel Dis.
15:1746–1754. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Däbritz J, Langhorst J, Lügering A,
Heidemann J, Mohr M, Wittkowski H, Krummenerl T and Foell D:
Improving relapse prediction in inflammatory bowel disease by
neutrophil-derived S100A12. Inflamm Bowel Dis. 19:1130–1138. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Palone F, Vitali R, Cucchiara S,
Pierdomenico M, Negroni A, Aloi M, Nuti F, Felice C, Armuzzi A and
Stronati L: Role of HMGB1 as a suitable biomarker of subclinical
intestinal inflammation and mucosal healing in patients with
inflammatory bowel disease. Inflamm Bowel Dis. 20:1448–1457. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kordjazy N, Haj-Mirzaian A, Haj-Mirzaian
A, Rohani MM, Gelfand EW, Rezaei N and Abdolghaffari AH: Role of
toll-like receptors in inflammatory bowel disease. Pharmacol Res.
129:204–215. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Iliev ID, Funari VA, Taylor KD, Nguyen Q,
Reyes CN, Strom SP, Brown J, Becker CA, Fleshner PR, Dubinsky M, et
al: Interactions between commensal fungi and the C-type lectin
receptor Dectin-1 influence colitis. Science. 336:1314–1317. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhen Y and Zhang H: NLRP3 inflammasome and
inflammatory bowel disease. Front Immunol. 10:2762019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Le Berre C, Honap S and Peyrin-Biroulet L:
Ulcerative colitis. Lancet. 402:571–584. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chhibba T and Ma C: Is there room for
immunomodulators in ulcerative colitis? Expert Opin Biol Ther.
20:379–390. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Coskun M, Vermeire S and Nielsen OH: Novel
targeted therapies for inflammatory bowel disease. Trends Pharmacol
Sci. 38:127–142. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Vignali DA and Kuchroo VK: IL-12 family
cytokines: Immunological playmakers. Nat Immunol. 13:722–728. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Verstockt B, Salas A, Sands BE, Abraham C,
Leibovitzh H, Neurath MF and Vande Casteele N; Alimentiv
Translational Research Consortium (ATRC), : IL-12 and IL-23 pathway
inhibition in inflammatory bowel disease. Nat Rev Gastroenterol
Hepatol. 20:433–446. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Teng MW, Bowman EP, McElwee JJ, Smyth MJ,
Casanova JL, Cooper AM and Cua DJ: IL-12 and IL-23 cytokines: From
discovery to targeted therapies for immune-mediated inflammatory
diseases. Nat Med. 21:719–729. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ogino H, Fukaura K, Iboshi Y, Nagamatsu Y,
Okuno H, Nishioka K, Nishihara Y, Tanaka Y, Chinen T, Ihara E and
Ogawa Y: Role of the IL-23-T-bet/GATA3 axis for the pathogenesis of
ulcerative colitis. Inflammation. 44:592–603. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
O'Shea JJ and Plenge R: JAK and STAT
signaling molecules in immunoregulation and immune-mediated
disease. Immunity. 36:542–550. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Villarino AV, Kanno Y and O'Shea JJ:
Mechanisms and consequences of Jak-STAT signaling in the immune
system. Nat Immunol. 18:374–384. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Croft NM, Faubion WA Jr, Kugathasan S,
Kierkus J, Ruemmele FM, Shimizu T, Mostafa NM, Venetucci M,
Finney-Hayward T, Sanchez Gonzalez Y, et al: Efficacy and safety of
adalimumab in paediatric patients with moderate-to-severe
ulcerative colitis (ENVISION I): A randomised, controlled, phase 3
study. Lancet Gastroenterol Hepatol. 6:616–627. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Feagan BG, Sands BE, Sandborn WJ,
Germinaro M, Vetter M, Shao J, Sheng S, Johanns J and Panés J; VEGA
Study Group, : Guselkumab plus golimumab combination therapy versus
guselkumab or golimumab monotherapy in patients with ulcerative
colitis (VEGA): A randomised, double-blind, controlled, phase 2,
proof-of-concept trial. Lancet Gastroenterol Hepatol. 8:307–320.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Harris MS, Hartman D, Lemos BR, Erlich EC,
Spence S, Kennedy S, Ptak T, Pruitt R, Vermeire S and Fox BS:
AVX-470, an orally delivered anti-tumour necrosis factor antibody
for treatment of active ulcerative colitis: Results of a
First-in-Human trial. J Crohns Colitis. 10:631–640. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Hartman DS, Tracey DE, Lemos BR, Erlich
EC, Burton RE, Keane DM, Patel R, Kim S, Bhol KC, Harris MS and Fox
BS: Effects of AVX-470, an oral, locally acting anti-tumour
necrosis factor antibody, on tissue biomarkers in patients with
active ulcerative colitis. J Crohns Colitis. 10:641–649. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hibi T, Imai Y, Senoo A, Ohta K and Ukyo
Y: Efficacy and safety of golimumab 52-week maintenance therapy in
Japanese patients with moderate to severely active ulcerative
colitis: A phase 3, double-blind, randomized, placebo-controlled
study-(PURSUIT-J study). J Gastroenterol. 52:1101–1111. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Panés J, Colombel JF, D'Haens GR,
Schreiber S, Panaccione R, Peyrin-Biroulet L, Loftus EV Jr, Danese
S, Tanida S, Okuyama Y, et al: Higher vs standard adalimumab
induction and maintenance dosing regimens for treatment of
ulcerative colitis: SERENE UC trial results. Gastroenterology.
162:1891–1910. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Rutgeerts P, Feagan BG, Marano CW, Padgett
L, Strauss R, Johanns J, Adedokun OJ, Guzzo C, Zhang H, Colombel
JF, et al: Randomised clinical trial: A placebo-controlled study of
intravenous golimumab induction therapy for ulcerative colitis.
Aliment Pharmacol Ther. 42:504–514. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Sandborn WJ, Targan SR, Byers VS, Rutty
DA, Mu H, Zhang X and Tang T: Andrographis paniculata extract
(HMPL-004) for active ulcerative colitis. Am J Gastroenterol.
108:90–98. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sands BE, Peyrin-Biroulet L, Loftus EV Jr,
Danese S, Colombel JF, Törüner M, Jonaitis L, Abhyankar B, Chen J,
Rogers R, et al: Vedolizumab versus Adalimumab for
Moderate-to-severe ulcerative colitis. N Engl J Med. 381:1215–1226.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Schreiber S, Ben-Horin S, Leszczyszyn J,
Dudkowiak R, Lahat A, Gawdis-Wojnarska B, Pukitis A, Horynski M,
Farkas K, Kierkus J, et al: Randomized controlled trial:
Subcutaneous vs intravenous infliximab CT-P13 maintenance in
inflammatory bowel disease. Gastroenterology. 160:2340–2353. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Suzuki Y, Motoya S, Hanai H, Matsumoto T,
Hibi T, Robinson AM, Mostafa NM, Chao J, Arora V, Camez A, et al:
Efficacy and safety of adalimumab in Japanese patients with
moderately to severely active ulcerative colitis. J Gastroenterol.
49:283–294. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Syversen SW, Jørgensen KK, Goll GL, Brun
MK, Sandanger Ø, Bjørlykke KH, Sexton J, Olsen IC, Gehin JE, Warren
DJ, et al: Effect of therapeutic drug monitoring vs standard
therapy during maintenance infliximab therapy on disease control in
patients with immune-mediated inflammatory diseases: A randomized
clinical trial. JAMA. 326:2375–2384. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Tajiri H, Arai K, Kagimoto S, Kunisaki R,
Hida N, Sato N, Yamada H, Nagano M, Susuta Y, Ozaki K, et al:
Infliximab for pediatric patients with ulcerative colitis: A phase
3, open-label, uncontrolled, multicenter trial in Japan. BMC
Pediatr. 19:3512019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Tang T, Targan SR, Li ZS, Xu C, Byers VS
and Sandborn WJ: Randomised clinical trial: Herbal extract HMPL-004
in active ulcerative colitis-a double-blind comparison with
sustained release mesalazine. Aliment Pharmacol Ther. 33:194–202.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
D'Haens G, Higgins PDR, Peyrin-Biroulet L,
Sands BE, Lee S, Moses RE, Redondo I, Escobar R, Hunter Gibble T,
Keohane A, et al: Extended induction and prognostic indicators of
response in patients treated with mirikizumab with moderately to
severely active ulcerative colitis in the LUCENT trials. Inflamm
Bowel Dis. 30:2335–2346. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Danese S, Sands BE, Abreu MT, O'Brien CD,
Bravatà I, Nazar M, Miao Y, Wang Y, Rowbotham D, Leong RWL, et al:
Early symptomatic improvement after ustekinumab therapy in patients
with ulcerative colitis: 16-week data from the UNIFI trial. Clin
Gastroenterol Hepatol. 20:2858–2867.e5. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Peyrin-Biroulet L, Allegretti JR, Rubin
DT, Bressler B, Germinaro M, Huang KG, Shipitofsky N, Zhang H,
Wilson R, Han C, et al: Guselkumab in patients with moderately to
severely active ulcerative colitis: QUASAR phase 2b induction
study. Gastroenterology. 165:1443–1457. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Sands BE, Sandborn WJ, Panaccione R,
O'Brien CD, Zhang H, Johanns J, Adedokun OJ, Li K, Peyrin-Biroulet
L, Van Assche G, et al: Ustekinumab as induction and maintenance
therapy for ulcerative colitis. N Engl J Med. 381:1201–1214. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Cromer WE, Ganta CV, Patel M, Traylor J,
Kevil CG, Alexander JS and Mathis JM: VEGF-A isoform modulation in
an preclinical TNBS model of ulcerative colitis: Protective effects
of a VEGF164b therapy. J Transl Med. 11:2072013. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Danese S, Vermeire S, Zhou W, Pangan AL,
Siffledeen J, Greenbloom S, Hébuterne X, D'Haens G, Nakase H, Panés
J, et al: Upadacitinib as induction and maintenance therapy for
moderately to severely active ulcerative colitis: Results from
three phase 3, multicentre, double-blind, randomised trials.
Lancet. 399:2113–2128. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Feagan BG, Danese S, Loftus EV Jr,
Vermeire S, Schreiber S, Ritter T, Fogel R, Mehta R, Nijhawan S,
Kempiński R, et al: Filgotinib as induction and maintenance therapy
for ulcerative colitis (SELECTION): A phase 2b/3 double-blind,
randomised, placebo-controlled trial. Lancet. 397:2372–2384. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Gros B, Goodall M, Plevris N,
Constantine-Cooke N, Elford AT, O'Hare C, Noble C, Jones GR, Arnott
ID and Lees CW: Real-world cohort study on the effectiveness and
safety of filgotinib use in ulcerative colitis. J Crohns Colitis.
19:jjad1872023. View Article : Google Scholar
|
|
61
|
Sandborn WJ, Ghosh S, Panes J, Schreiber
S, D'Haens G, Tanida S, Siffledeen J, Enejosa J, Zhou W, Othman AA,
et al: Efficacy of upadacitinib in a randomized trial of patients
with active ulcerative colitis. Gastroenterology.
158:2139–2149.e14. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Sandborn WJ, Nguyen DD, Beattie DT,
Brassil P, Krey W, Woo J, Situ E, Sana R, Sandvik E, Pulido-Rios
MT, et al: Development of Gut-selective pan-janus kinase inhibitor
TD-1473 for ulcerative colitis: A translational medicine programme.
J Crohns Colitis. 14:1202–1213. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Sandborn WJ, Peyrin-Biroulet L, Quirk D,
Wang W, Nduaka CI, Mukherjee A, Su C and Sands BE: Efficacy and
safety of extended induction with tofacitinib for the treatment of
ulcerative colitis. Clin Gastroenterol Hepatol. 20:1821–1830.e3.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Sandborn WJ, Su C, Sands BE, D'Haens GR,
Vermeire S, Schreiber S, Danese S, Feagan BG, Reinisch W,
Niezychowski W, et al: Tofacitinib as induction and maintenance
therapy for ulcerative colitis. N Engl J Med. 376:1723–1736. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Sands BE, Sandborn WJ, Feagan BG,
Lichtenstein GR, Zhang H, Strauss R, Szapary P, Johanns J, Panes J,
Vermeire S, et al: Peficitinib, an oral Janus Kinase inhibitor, in
moderate-to-severe ulcerative colitis: Results from a randomised,
phase 2 study. J Crohns Colitis. 12:1158–1169. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Singh A, Midha V, Kaur K, Mahajan R, Singh
D, Kaur R, Kohli A, Chawla A, Sood K, Bansal N and Sood A:
Tofacitinib versus oral prednisolone for induction of remission in
moderately active ulcerative colitis [ORCHID]: A prospective,
open-label, randomized, pilot study. J Crohns Colitis. 18:300–307.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Sandborn WJ, Ghosh S, Panes J, Vranic I,
Su C, Rousell S and Niezychowski W; Study A3921063 Investigators, :
Tofacitinib, an oral Janus kinase inhibitor, in active ulcerative
colitis. N Engl J Med. 367:616–624. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Danese S, Neurath MF, Kopoń A, Zakko SF,
Simmons TC, Fogel R, Siegel CA, Panaccione R, Zhan X, Usiskin K and
Chitkara D: Effects of apremilast, an oral inhibitor of
phosphodiesterase 4, in a randomized trial of patients with active
ulcerative colitis. Clin Gastroenterol Hepatol. 18:2526–2534.e9.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Schreiber S, Keshavarzian A, Isaacs KL,
Schollenberger J, Guzman JP, Orlandi C and Hanauer SB: A
randomized, placebo-controlled, phase II study of tetomilast in
active ulcerative colitis. Gastroenterology. 132:76–86. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Danese S, Panaccione R, Abreu MT, Rubin
DT, Ghosh S, Dignass A, Afzali A, Wolf DC, Chiorean MV, Vermeire S,
et al: Efficacy and safety of approximately 3 years of continuous
ozanimod in moderately to severely active ulcerative colitis:
Interim analysis of the true north Open-label extension. J Crohns
Colitis. 18:264–274. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Sandborn WJ, Feagan BG, D'Haens G, Wolf
DC, Jovanovic I, Hanauer SB, Ghosh S, Petersen A, Hua SY, Lee JH,
et al: Ozanimod as induction and maintenance therapy for ulcerative
colitis. N Engl J Med. 385:1280–1291. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Sandborn WJ, Feagan BG, Wolf DC, D'Haens
G, Vermeire S, Hanauer SB, Ghosh S, Smith H, Cravets M, Frohna PA,
et al: Ozanimod induction and maintenance treatment for ulcerative
colitis. N Engl J Med. 374:1754–1762. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Sandborn WJ, Peyrin-Biroulet L, Zhang J,
Chiorean M, Vermeire S, Lee SD, Kühbacher T, Yacyshyn B, Cabell CH,
Naik SU, et al: Efficacy and safety of etrasimod in a phase 2
randomized trial of patients with ulcerative colitis.
Gastroenterology. 158:550–561. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Sandborn WJ, Vermeire S, Peyrin-Biroulet
L, Dubinsky MC, Panes J, Yarur A, Ritter T, Baert F, Schreiber S,
Sloan S, et al: Etrasimod as induction and maintenance therapy for
ulcerative colitis (ELEVATE): Two randomised, double-blind,
placebo-controlled, phase 3 studies. Lancet. 401:1159–1171. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Sánchez de Medina F, Romero-Calvo I,
Mascaraque C and Martínez-Augustin O: Intestinal inflammation and
mucosal barrier function. Inflamm Bowel Dis. 20:2394–2404. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Sims GP, Rowe DC, Rietdijk ST, Herbst R
and Coyle AJ: HMGB1 and RAGE in inflammation and cancer. Annu Rev
Immunol. 28:367–388. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Body-Malapel M, Djouina M, Waxin C,
Langlois A, Gower-Rousseau C, Zerbib P, Schmidt AM, Desreumaux P,
Boulanger E and Vignal C: The RAGE signaling pathway is involved in
intestinal inflammation and represents a promising therapeutic
target for inflammatory bowel diseases. Mucosal Immunol.
12:468–478. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Sugaya K, Fukagawa T, Matsumoto K, Mita K,
Takahashi E, Ando A, Inoko H and Ikemura T: Three genes in the
human MHC class III region near the junction with the class II:
Gene for receptor of advanced glycosylation end products, PBX2
homeobox gene and a notch homolog, human counterpart of mouse
mammary tumor gene int-3. Genomics. 23:408–419. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Fritz G: RAGE: A single receptor fits
multiple ligands. Trends Biochem Sci. 36:625–632. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Hudson BI, Kalea AZ, Del Mar Arriero M,
Harja E, Boulanger E, D'Agati V and Schmidt AM: Interaction of the
RAGE cytoplasmic domain with diaphanous-1 is required for
ligand-stimulated cellular migration through activation of Rac1 and
Cdc42. J Biol Chem. 283:34457–34468. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Kalea AZ, Schmidt AM and Hudson BI: RAGE:
A novel biological and genetic marker for vascular disease. Clin
Sci (Lond). 116:621–637. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Harja E, Bu DX, Hudson BI, Chang JS, Shen
X, Hallam K, Kalea AZ, Lu Y, Rosario RH, Oruganti S, et al:
Vascular and inflammatory stresses mediate atherosclerosis via RAGE
and its ligands in apoE-/- mice. J Clin Invest. 118:183–194. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Huttunen HJ, Fages C and Rauvala H:
Receptor for advanced glycation end products (RAGE)-mediated
neurite outgrowth and activation of NF-kappaB require the
cytoplasmic domain of the receptor but different downstream
signaling pathways. J Biol Chem. 274:19919–19924. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Hudson BI and Lippman ME: Targeting RAGE
signaling in inflammatory disease. Annu Rev Med. 69:349–364. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Piarulli F, Lapolla A, Ragazzi E, Susana
A, Sechi A, Nollino L, Cosma C, Fedele D and Sartore G: Role of
endogenous secretory RAGE (esRAGE) in defending against plaque
formation induced by oxidative stress in type 2 diabetic patients.
Atherosclerosis. 226:252–257. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Deng M, Tang Y, Li W, Wang X, Zhang R,
Zhang X, Zhao X, Liu J, Tang C, Liu Z, et al: The endotoxin
delivery protein HMGB1 mediates caspase-11-dependent lethality in
sepsis. Immunity. 49:740–753.e7. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Angioni R, Bonfanti M, Caporale N,
Sánchez-Rodríguez R, Munari F, Savino A, Pasqualato S, Buratto D,
Pagani I, Bertoldi N, et al: RAGE engagement by SARS-CoV-2 enables
monocyte infection and underlies COVID-19 severity. Cell Rep Med.
4:1012662023. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Tsubokawa D, Lee JM, Hatta T, Mikami F,
Maruyama H, Arakawa T, Kusakabe T and Tsuji N: Characterization of
the RAGE-binding protein, Strongyloides venestatin, produced by the
silkworm-baculovirus expression system. Infect Genet Evol.
75:1039642019. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Senatus L, Egaña-Gorroño L, López-Díez R,
Bergaya S, Aranda JF, Amengual J, Arivazhagan L, Manigrasso MB,
Yepuri G, Nimma R, et al: DIAPH1 mediates progression of
atherosclerosis and regulates hepatic lipid metabolism in mice.
Commun Biol. 6:2802023. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Chiappalupi S, Sorci G, Vukasinovic A,
Salvadori L, Sagheddu R, Coletti D, Renga G, Romani L, Donato R and
Riuzzi F: Targeting RAGE prevents muscle wasting and prolongs
survival in cancer cachexia. J Cachexia Sarcopenia Muscle.
11:929–946. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Tian M, Tang Y, Huang T, Liu Y and Pan Y:
Amelioration of human peritoneal mesothelial cell co-culture-evoked
malignant potential of ovarian cancer cells by acacetin involves
LPA release-activated RAGE-PI3K/AKT signaling. Cell Mol Biol Lett.
26:512021. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Sakaguchi M, Murata H, Aoyama Y, Hibino T,
Putranto EW, Ruma IM, Inoue Y, Sakaguchi Y, Yamamoto K, Kinoshita
R, et al: DNAX-activating protein 10 (DAP10) membrane adaptor
associates with receptor for advanced glycation end products (RAGE)
and modulates the RAGE-triggered signaling pathway in human
keratinocytes. J Biol Chem. 289:23389–23402. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Bierhaus A, Schiekofer S, Schwaninger M,
Andrassy M, Humpert PM, Chen J, Hong M, Luther T, Henle T, Klöting
I, et al: Diabetes-associated sustained activation of the
transcription factor nuclear factor-kappaB. Diabetes. 50:2792–2808.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Goldin A, Beckman JA, Schmidt AM and
Creager MA: Advanced glycation end products: Sparking the
development of diabetic vascular injury. Circulation. 114:597–605.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Le Bagge S, Fotheringham AK, Leung SS and
Forbes JM: Targeting the receptor for advanced glycation end
products (RAGE) in type 1 diabetes. Med Res Rev. 40:1200–1219.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Mocanu CA, Fuior EV, Voicu G, Rebleanu D,
Safciuc F, Deleanu M, Fenyo IM, Escriou V, Manduteanu I, Simionescu
M and Calin M: P-selectin targeted RAGE-shRNA lipoplexes alleviate
atherosclerosis-associated inflammation. J Control Release.
338:754–772. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Lai PH, Wang TH, Zhang NY, Wu KC, Yao CJ
and Lin CJ: Changes of blood-brain-barrier function and transfer of
amyloid beta in rats with collagen-induced arthritis. J
Neuroinflammation. 18:352021. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Selejan SR, Linz D, Mauz M, Hohl M, Huynh
AKD, Speer T, Wintrich J, Kazakov A, Werner C, Mahfoud F and Böhm
M: Renal denervation reduces atrial remodeling in hypertensive rats
with metabolic syndrome. Basic Res Cardiol. 117:362022. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Turovskaya O, Foell D, Sinha P, Vogl T,
Newlin R, Nayak J, Nguyen M, Olsson A, Nawroth PP, Bierhaus A, et
al: RAGE, carboxylated glycans and S100A8/A9 play essential roles
in colitis-associated carcinogenesis. Carcinogenesis. 29:2035–2043.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Bierhaus A, Stern DM and Nawroth PP: RAGE
in inflammation: A new therapeutic target? Curr Opin Investig
Drugs. 7:985–991. 2006.PubMed/NCBI
|
|
101
|
Chuah YK, Basir R, Talib H, Tie TH and
Nordin N: Receptor for advanced glycation end products and its
involvement in inflammatory diseases. Int J Inflam.
2013:4034602013.PubMed/NCBI
|
|
102
|
Sparvero LJ, Asafu-Adjei D, Kang R, Tang
D, Amin N, Im J, Rutledge R, Lin B, Amoscato AA, Zeh HJ, et al:
RAGE (Receptor for Advanced Glycation Endproducts), RAGE ligands,
and their role in cancer and inflammation. J Transl Med. 7:172009.
View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Moura FA, Goulart MOF, Campos SBG and da
Paz Martins AS: The close interplay of Nitro-oxidative stress,
advanced glycation end products and inflammation in inflammatory
bowel diseases. Curr Med Chem. 27:2059–2076. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Yadav V, Varum F, Bravo R, Furrer E, Bojic
D and Basit AW: Inflammatory bowel disease: Exploring gut
pathophysiology for novel therapeutic targets. Transl Res.
176:38–68. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Foell D, Kucharzik T, Kraft M, Vogl T,
Sorg C, Domschke W and Roth J: Neutrophil derived human S100A12
(EN-RAGE) is strongly expressed during chronic active inflammatory
bowel disease. Gut. 52:847–853. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Hu Z, Wang X, Gong L, Wu G, Peng X and
Tang X: Role of high-mobility group box 1 protein in inflammatory
bowel disease. Inflamm Res. 64:557–563. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Manolakis AC, Kapsoritakis AN, Tiaka EK
and Potamianos SP: Calprotectin, calgranulin C, and other members
of the s100 protein family in inflammatory bowel disease. Dig Dis
Sci. 56:1601–1611. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Yamasaki H, Mitsuyama K, Masuda J, Kuwaki
K, Takedatsu H, Sugiyama G, Yamada S and Sata M: Roles of
high-mobility group box 1 in murine experimental colitis. Mol Med
Rep. 2:23–27. 2009.PubMed/NCBI
|
|
109
|
Brownlee M: Advanced protein glycosylation
in diabetes and aging. Annu Rev Med. 46:223–234. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Yamagishi S and Matsui T: Pathologic role
of dietary advanced glycation end products in cardiometabolic
disorders, and therapeutic intervention. Nutrition. 32:157–165.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Maconi G, Furfaro F, Sciurti R, Bezzio C,
Ardizzone S and de Franchis R: Glucose intolerance and diabetes
mellitus in ulcerative colitis: Pathogenetic and therapeutic
implications. World J Gastroenterol. 20:3507–3515. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Farghaly HS and Thabit RH: L-arginine and
aminoguanidine reduce colonic damage of acetic acid-induced colitis
in rats: Potential modulation of nuclear factor-κB/p65. Clin Exp
Pharmacol Physiol. 41:769–779. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Mouzaoui S, Rahim I and Djerdjouri B:
Aminoguanidine and curcumin attenuated tumor necrosis factor
(TNF)-α-induced oxidative stress, colitis and hepatotoxicity in
mice. Int Immunopharmacol. 12:302–311. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
El-Mahdy NA, El-Sayad ME, El-Kadem AH and
Abu-Risha SE: Metformin alleviates inflammation in oxazolone
induced ulcerative colitis in rats: Plausible role of sphingosine
kinase 1/sphingosine 1 phosphate signaling pathway. Immunopharmacol
Immunotoxicol. 43:192–202. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Pandey A, Verma S and Kumar VL: Metformin
maintains mucosal integrity in experimental model of colitis by
inhibiting oxidative stress and pro-inflammatory signaling. Biomed
Pharmacother. 94:1121–1128. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Shaaban AA, Abdelhamid AM, Shaker ME,
Cavalu S, Maghiar AM, Alsayegh AA, Babalghith AO, El-Ahwany E, Amin
NA, Mohammed OA, et al: Combining the HSP90 inhibitor TAS-116 with
metformin effectively degrades the NLRP3 and attenuates
inflammasome activation in rats: A new management paradigm for
ulcerative colitis. Biomed Pharmacother. 153:1132472022. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
da Rocha GHO, de Paula-Silva M, Broering
MF, Scharf PRDS, Matsuyama LSAS, Maria-Engler SS and Farsky SHP:
Pioglitazone-mediated attenuation of experimental colitis relies on
cleaving of annexin A1 released by macrophages. Front Pharmacol.
11:5915612020. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Matsunaga H, Hokari R, Kurihara C, Okada
Y, Takebayashi K, Okudaira K, Watanabe C, Komoto S, Nakamura M,
Tsuzuki Y, et al: Omega-3 fatty acids exacerbate DSS-induced
colitis through decreased adiponectin in colonic subepithelial
myofibroblasts. Inflamm Bowel Dis. 14:1348–1357. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
El-Haggar SM, Hegazy SK, Maher MM, Bahgat
MM and Bahaa MM: Repurposing metformin as adjuvant therapy in
patients with ulcerative colitis treated with mesalamine: A
randomized controlled double-blinded study. Int Immunopharmacol.
138:1125412024. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Lin J, Wang C, Yang G, Liu Y, Da Y, Zhou
W, Zhou H, Wang F, Fan Z, Hou L, et al: Targeting the cell adhesion
related ligands MAC1 with Indirubin to inhibit AGE-RAGE signaling
and mitigate colitis in an mouse model. Phytomedicine.
149:1575042025. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Paradela-Dobarro B, Raposeiras-Roubín S,
Rodiño-Janeiro BK, Grigorian-Shamagian L, García-Acuña JM,
Aguiar-Souto P, Jacquet-Hervet M, Reino-Maceiras MV,
González-Juanatey JR and Alvarez E: Statins modulate feedback
regulation mechanisms between advanced glycation end-products and
C-reactive protein: Evidence in patients with acute myocardial
infarction. Eur J Pharm Sci. 49:512–518. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Côté-Daigneault J, Mehandru S, Ungaro R,
Atreja A and Colombel JF: Potential immunomodulatory effects of
statins in inflammatory bowel disease. Inflamm Bowel Dis.
22:724–732. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Ikeda M, Takeshima F, Isomoto H, Shikuwa
S, Mizuta Y, Ozono Y and Kohno S: Simvastatin attenuates
trinitrobenzene sulfonic acid-induced colitis, but not
oxazalone-induced colitis. Dig Dis Sci. 53:1869–1875. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Naito Y, Katada K, Takagi T, Tsuboi H,
Isozaki Y, Handa O, Kokura S, Yoshida N, Ichikawa H and Yoshikawa
T: Rosuvastatin, a new HMG-CoA reductase inhibitor, reduces the
colonic inflammatory response in dextran sulfate sodium-induced
colitis in mice. Int J Mol Med. 17:997–1004. 2006.PubMed/NCBI
|
|
125
|
Ungaro R, Chang HL, Côté-Daigneault J,
Mehandru S, Atreja A and Colombel JF: Statins associated with
decreased risk of new onset inflammatory bowel disease. Am J
Gastroenterol. 111:1416–1423. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Khalil D, Boktor M, Mortensen EM, Frei CR
and Mansi I: Comparison of frequency of inflammatory bowel disease
and noninfectious gastroenteritis among statin users versus
nonusers. Am J Cardiol. 115:1396–1401. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Dhamija P, Hota D, Kochhar R, Sachdev A
and Chakrabarti A: Randomized clinical trial: Atorvastatin versus
placebo in patients with acute exacerbation of mild to moderate
ulcerative colitis. Indian J Gastroenterol. 33:151–156. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Peppas S, Piovani D, Peyrin-Biroulet L,
Danese S and Bonovas S: Statins and inflammatory bowel disease:
Where do we stand? Eur J Intern Med. 75:10–14. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Chande N, McDonald JW, Macdonald JK and
Wang JJ: Unfractionated or low-molecular weight heparin for
induction of remission in ulcerative colitis. Cochrane Database
Syst Rev: CD006774. 2010.doi: 10.1002/14651858.CD006774.pub3.
View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Yan Y, Sun Y, Wang P, Zhang R, Huo C, Gao
T, Song C, Xing J and Dong Y: Mucoadhesive nanoparticles-based oral
drug delivery systems enhance ameliorative effects of low molecular
weight heparin on experimental colitis. Carbohydr Polym.
246:1166602020. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Yazeji T, Moulari B, Beduneau A, Stein V,
Dietrich D, Pellequer Y and Lamprecht A: Nanoparticle-based
delivery enhances anti-inflammatory effect of low molecular weight
heparin in experimental ulcerative colitis. Drug Deliv. 24:811–817.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Hori O, Brett J, Slattery T, Cao R, Zhang
J, Chen JX, Nagashima M, Lundh ER, Vijay S, Nitecki D, et al: The
receptor for advanced glycation end products (RAGE) is a cellular
binding site for amphoterin. Mediation of neurite outgrowth and
co-expression of rage and amphoterin in the developing nervous
system. J Biol Chem. 270:25752–25761. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Erlandsson Harris H and Andersson U:
Mini-review: The nuclear protein HMGB1 as a proinflammatory
mediator. Eur J Immunol. 34:1503–1512. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Kang R, Zhang Q, Zeh HJ III, Lotze MT and
Tang D: HMGB1 in cancer: Good, bad, or both? Clin Cancer Res.
19:4046–4057. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Dong Xda E, Ito N, Lotze MT, Demarco RA,
Popovic P, Shand SH, Watkins S, Winikoff S, Brown CK, Bartlett DL,
et al: High mobility group box I (HMGB1) release from tumor cells
after treatment: Implications for development of targeted
chemoimmunotherapy. J Immunother. 30:596–606. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Tang D, Kang R, Zeh HJ III and Lotze MT:
High-mobility group box 1 and cancer. Biochim Biophys Acta.
1799:131–140. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Tian J, Avalos AM, Mao SY, Chen B, Senthil
K, Wu H, Parroche P, Drabic S, Golenbock D, Sirois C, et al:
Toll-like receptor 9-dependent activation by DNA-containing immune
complexes is mediated by HMGB1 and RAGE. Nat Immunol. 8:487–496.
2007. View
Article : Google Scholar : PubMed/NCBI
|
|
138
|
Scaffidi P, Misteli T and Bianchi ME:
Release of chromatin protein HMGB1 by necrotic cells triggers
inflammation. Nature. 418:191–195. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Yanai H, Ban T, Wang Z, Choi MK, Kawamura
T, Negishi H, Nakasato M, Lu Y, Hangai S, Koshiba R, et al: HMGB
proteins function as universal sentinels for nucleic-acid-mediated
innate immune responses. Nature. 462:99–103. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Huang CY, Chiang SF, Chen WT, Ke TW, Chen
TW, You YS, Lin CY, Chao KSC and Huang CY: HMGB1 promotes
ERK-mediated mitochondrial Drp1 phosphorylation for chemoresistance
through RAGE in colorectal cancer. Cell Death Dis. 9:10042018.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Arab HH, Al-Shorbagy MY and Saad MA:
Activation of autophagy and suppression of apoptosis by
dapagliflozin attenuates experimental inflammatory bowel disease in
rats: Targeting AMPK/mTOR, HMGB1/RAGE and Nrf2/HO-1 pathways. Chem
Biol Interact. 335:1093682021. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Chen L, Li J, Ye Z, Sun B, Wang L, Chen Y,
Han J, Yu M, Wang Y, Zhou Q, et al: Anti-high mobility group box 1
neutralizing-antibody ameliorates dextran sodium sulfate colitis in
mice. Front Immunol. 11:5850942020. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Davé SH, Tilstra JS, Matsuoka K, Li F,
DeMarco RA, Beer-Stolz D, Sepulveda AR, Fink MP, Lotze MT and Plevy
SE: Ethyl pyruvate decreases HMGB1 release and ameliorates murine
colitis. J Leukoc Biol. 86:633–643. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Maeda S, Hikiba Y, Shibata W, Ohmae T,
Yanai A, Ogura K, Yamada S and Omata M: Essential roles of
high-mobility group box 1 in the development of murine colitis and
colitis-associated cancer. Biochem Biophys Res Commun. 360:394–400.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Palone F, Vitali R, Cucchiara S, Mennini
M, Armuzzi A, Pugliese D, D'Incà R, Barberio B and Stronati L:
Fecal HMGB1 reveals microscopic inflammation in adult and pediatric
patients with inflammatory bowel disease in clinical and endoscopic
remission. Inflamm Bowel Dis. 22:2886–2893. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Vitali R, Stronati L, Negroni A, Di Nardo
G, Pierdomenico M, del Giudice E, Rossi P and Cucchiara S: Fecal
HMGB1 is a novel marker of intestinal mucosal inflammation in
pediatric inflammatory bowel disease. Am J Gastroenterol.
106:2029–2040. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Miao Z, Gu M, Raza F, Zafar H, Huang J,
Yang Y, Sulaiman M, Yan J and Xu Y: Isoliquiritin ameliorates
ulcerative colitis in rats through caspase 3/HMGB1/TLR4 dependent
signaling pathway. Curr Gene Ther. 24:73–92. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Chen J, Lu P, Liu J, Yang L, Li Y, Chen Y,
Wang Y, Wan J and Zhao Y: 20(S)-Protopanaxadiol saponins isolated
from Panax notoginseng target the binding of HMGB1 to TLR4 against
inflammation in experimental ulcerative colitis. Phytother Res.
37:4690–4705. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Sun K, Lin W, Hong Q, Chen S, Li J and Qiu
S: Matrine: A promising treatment for ulcerative colitis by
targeting the HMGB1/NLRP3/caspase-1 pathway. Comb Chem High
Throughput Screen. 28:654–663. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Zhu N, Ge X, Zhang L, Chen X, Xiang W and
Mei Q: HMGB1 exacerbates intestinal barrier damage by inducing
ferroptosis through the TLR4/NF-κB/GPX4 pathway in ulcerative
colitis. Mediators Inflamm. 2025:23955572025. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Gonzalez LL, Garrie K and Turner MD: Role
of S100 proteins in health and disease. Biochim Biophys Acta Mol
Cell Res. 1867:1186772020. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Donato R, Cannon BR, Sorci G, Riuzzi F,
Hsu K, Weber DJ and Geczy CL: Functions of S100 proteins. Curr Mol
Med. 13:24–57. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Xiao X, Yang C, Qu SL, Shao YD, Zhou CY,
Chao R, Huang L and Zhang C: S100 proteins in atherosclerosis. Clin
Chim Acta. 502:293–304. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Gross SR, Sin CG, Barraclough R and
Rudland PS: Joining S100 proteins and migration: For better or for
worse, in sickness and in health. Cell Mol Life Sci. 71:1551–1579.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Bjarnason I: The use of fecal calprotectin
in inflammatory bowel disease. Gastroenterol Hepatol (N Y).
13:53–56. 2017.PubMed/NCBI
|
|
156
|
Rammes A, Roth J, Goebeler M, Klempt M,
Hartmann M and Sorg C: Myeloid-related protein (MRP) 8 and MRP14,
calcium-binding proteins of the S100 family, are secreted by
activated monocytes via a novel, tubulin-dependent pathway. J Biol
Chem. 272:9496–9502. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Wang L, Luo H, Chen X, Jiang Y and Huang
Q: Functional characterization of S100A8 and S100A9 in altering
monolayer permeability of human umbilical endothelial cells. PLoS
One. 9:e904722014. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Viemann D, Barczyk K, Vogl T, Fischer U,
Sunderkötter C, Schulze-Osthoff K and Roth J: MRP8/MRP14 impairs
endothelial integrity and induces a caspase-dependent and
-independent cell death program. Blood. 109:2453–2460. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Bengtsson AA, Sturfelt G, Lood C, Rönnblom
L, van Vollenhoven RF, Axelsson B, Sparre B, Tuvesson H, Ohman MW
and Leanderson T: Pharmacokinetics, tolerability, and preliminary
efficacy of paquinimod (ABR-215757), a new quinoline-3-carboxamide
derivative: Studies in lupus-prone mice and a multicenter,
randomized, double-blind, placebo-controlled, repeat-dose,
dose-ranging study in patients with systemic lupus erythematosus.
Arthritis Rheum. 64:1579–1588. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Brunmark C, Runström A, Ohlsson L, Sparre
B, Brodin T, Aström M and Hedlund G: The new orally active
immunoregulator laquinimod (ABR-215062) effectively inhibits
development and relapses of experimental autoimmune
encephalomyelitis. J Neuroimmunol. 130:163–172. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Karussis DM, Lehmann D, Slavin S,
Vourka-Karussis U, Mizrachi-Koll R, Ovadia H, Kalland T and
Abramsky O: Treatment of chronic-relapsing experimental autoimmune
encephalomyelitis with the synthetic immunomodulator linomide
(quinoline-3-carboxamide). Proc Natl Acad Sci USA. 90:6400–6404.
1993. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Maiseyeu A, Badgeley MA, Kampfrath T,
Mihai G, Deiuliis JA, Liu C, Sun Q, Parthasarathy S, Simon DI,
Croce K and Rajagopalan S: In vivo targeting of
inflammation-associated myeloid-related protein 8/14 via gadolinium
immunonanoparticles. Arterioscler Thromb Vasc Biol. 32:962–970.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Rai V, Touré F, Chitayat S, Pei R, Song F,
Li Q, Zhang J, Rosario R, Ramasamy R, Chazin WJ and Schmidt AM:
Lysophosphatidic acid targets vascular and oncogenic pathways via
RAGE signaling. J Exp Med. 209:2339–2350. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Nasser S, Abdallah DM, Ahmed KA,
Abdel-Mottaleb Y and El-Abhar HS: The novel anti-colitic effect of
β-adrenergic receptors via modulation of PS1/BACE-1/Aβ axis and
NOTCH signaling in an ulcerative colitis model. Front Pharmacol.
13:10080852022. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Son M, Porat A, He M, Suurmond J,
Santiago-Schwarz F, Andersson U, Coleman TR, Volpe BT, Tracey KJ,
Al-Abed Y and Diamond B: C1q and HMGB1 reciprocally regulate human
macrophage polarization. Blood. 128:2218–2228. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Abedini A, Cao P, Plesner A, Zhang J, He
M, Derk J, Patil SA, Rosario R, Lonier J, Song F, et al: RAGE binds
preamyloid IAPP intermediates and mediates pancreatic β cell
proteotoxicity. J Clin Invest. 128:682–698. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Zhang T, Jia C, Ran L, Shi J, Amarmend T
and Li H: The alleviative effects comparison of four flavonoids
from bamboo leaves on ulcerative colitis in an Alzheimer mouse
model. CNS Neurosci Ther. 30:e146202024. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Dong YL, Duan XY, Liu YJ, Fan H, Xu M,
Chen QY, Nan Z, Wu H and Deng SJ: Autotaxin-lysophosphatidic acid
axis blockade improves inflammation by regulating Th17 cell
differentiation in DSS-induced chronic colitis mice. Inflammation.
42:1530–1541. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Wang Z, Shi W, Tian D, Qin H, Vallance BA,
Yang H, Yu HB and Yu Q: Autotaxin stimulates LPA2 receptor in
macrophages and exacerbates dextran sulfate sodium-induced acute
colitis. J Mol Med (Berl). 98:1781–1794. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Takahashi S, Yoshimura T, Ohkura T,
Fujisawa M, Fushimi S, Ito T, Itakura J, Hiraoka S, Okada H,
Yamamoto K and Matsukawa A: A novel role of Spred2 in the colonic
epithelial cell homeostasis and inflammation. Sci Rep. 6:375312016.
View Article : Google Scholar : PubMed/NCBI
|
|
171
|
You BH, Chae HS, Song J, Ko HW, Chin YW
and Choi YH: α-Mangostin ameliorates dextran sulfate sodium-induced
colitis through inhibition of NF-κB and MAPK pathways. Int
Immunopharmacol. 49:212–221. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Wei YY, Fan YM, Ga Y, Zhang YN, Han JC and
Hao ZH: Shaoyao decoction attenuates DSS-induced ulcerative
colitis, macrophage and NLRP3 inflammasome activation through the
MKP1/NF-κB pathway. Phytomedicine. 92:1537432021. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Li C, Gong L, Jiang Y, Huo X, Huang L, Lei
H, Gu Y, Wang D, Guo D and Deng Y: Sanguisorba officinalis ethyl
acetate extract attenuates ulcerative colitis through inhibiting
PI3K-AKT/NF-κB/STAT3 pathway uncovered by single-cell RNA
sequencing. Phytomedicine. 120:1550522023. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Salari-Sharif P and Abdollahi M:
Phosphodiesterase 4 inhibitors in inflammatory bowel disease: A
comprehensive review. Curr Pharm Des. 16:3661–3667. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Yang J, Pei G, Sun X, Xiao Y, Miao C, Zhou
L, Wang B, Yang L, Yu M, Zhang ZS, et al: RhoB affects colitis
through modulating cell signaling and intestinal microbiome.
Microbiome. 10:1492022. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Adini A, Ko VH, Puder M, Louie SM, Kim CF,
Baron J and Matthews BD: PR1P, a VEGF-stabilizing peptide, reduces
injury and inflammation in acute lung injury and ulcerative colitis
animal models. Front Immunol. 14:11686762023. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Adamopoulos C, Piperi C, Gargalionis AN,
Dalagiorgou G, Spilioti E, Korkolopoulou P, Diamanti-Kandarakis E
and Papavassiliou AG: Advanced glycation end products upregulate
lysyl oxidase and endothelin-1 in human aortic endothelial cells
via parallel activation of ERK1/2-NF-κB and JNK-AP-1 signaling
pathways. Cell Mol Life Sci. 73:1685–1698. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Byun K, Yoo Y, Son M, Lee J, Jeong GB,
Park YM, Salekdeh GH and Lee B: Advanced glycation end-products
produced systemically and by macrophages: A common contributor to
inflammation and degenerative diseases. Pharmacol Ther. 177:44–55.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Reynaert NL, Gopal P, Rutten EPA, Wouters
EFM and Schalkwijk CG: Advanced glycation end products and their
receptor in age-related, non-communicable chronic inflammatory
diseases; Overview of clinical evidence and potential contributions
to disease. Int J Biochem Cell Biol. 81:403–418. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Xu Y, Toure F, Qu W, Lin L, Song F, Shen
X, Rosario R, Garcia J, Schmidt AM and Yan SF: Advanced glycation
end product (AGE)-receptor for AGE (RAGE) signaling and
up-regulation of Egr-1 in hypoxic macrophages. J Biol Chem.
285:23233–23240. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Spalinger MR, Sayoc-Becerra A,
Ordookhanian C, Canale V, Santos AN, King SJ, Krishnan M, Nair MG,
Scharl M and McCole DF: The JAK inhibitor tofacitinib rescues
intestinal barrier defects caused by disrupted
Epithelial-macrophage interactions. J Crohns Colitis. 15:471–484.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Han J, Zhao Q, Basmadjian C, Désaubry L
and Theiss AL: Flavaglines ameliorate experimental colitis and
protect against intestinal epithelial cell apoptosis and
mitochondrial dysfunction. Inflamm Bowel Dis. 22:55–67. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Zhou M, Xu W, Wang J, Yan J, Shi Y, Zhang
C, Ge W, Wu J, Du P and Chen Y: Boosting mTOR-dependent autophagy
via upstream TLR4-MyD88-MAPK signalling and downstream NF-κB
pathway quenches intestinal inflammation and oxidative stress
injury. EBioMedicine. 35:345–360. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Hwang J, Jin J, Jeon S, Moon SH, Park MY,
Yum DY, Kim JH, Kang JE, Park MH, Kim EJ, et al: SOD1 suppresses
pro-inflammatory immune responses by protecting against oxidative
stress in colitis. Redox Biol. 37:1017602020. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Hayakawa Y, Hirata Y, Nakagawa H, Sakamoto
K, Hikiba Y, Otsuka M, Ijichi H, Ikenoue T, Tateishi K, Akanuma M,
et al: Apoptosis signal-regulating kinase 1 regulates colitis and
colitis-associated tumorigenesis by the innate immune responses.
Gastroenterology. 138:1055–1067.e1-e4. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Eelen G, Treps L, Li X and Carmeliet P:
Basic and therapeutic aspects of angiogenesis updated. Circ Res.
127:310–329. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Sajib S, Zahra FT, Lionakis MS, German NA
and Mikelis CM: Mechanisms of angiogenesis in microbe-regulated
inflammatory and neoplastic conditions. Angiogenesis. 21:1–14.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Roldán-Montero R, Pérez-Sáez JM,
Cerro-Pardo I, Oller J, Martinez-Lopez D, Nuñez E, Maller SM,
Gutierrez-Muñoz C, Mendez-Barbero N, Escola-Gil JC, et al:
Galectin-1 prevents pathological vascular remodeling in
atherosclerosis and abdominal aortic aneurysm. Sci Adv.
8:eabm73222022. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Elshabrawy HA, Chen Z, Volin MV, Ravella
S, Virupannavar S and Shahrara S: The pathogenic role of
angiogenesis in rheumatoid arthritis. Angiogenesis. 18:433–448.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Dudley AC and Griffioen AW: Pathological
angiogenesis: Mechanisms and therapeutic strategies. Angiogenesis.
26:313–347. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Ackermann M, Verleden SE, Kuehnel M,
Haverich A, Welte T, Laenger F, Vanstapel A, Werlein C, Stark H,
Tzankov A, et al: Pulmonary vascular endothelialitis, thrombosis,
and angiogenesis in Covid-19. N Engl J Med. 383:120–128. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Tarnawski AS and Ahluwalia A: The critical
role of growth factors in gastric ulcer healing: The cellular and
molecular mechanisms and potential clinical implications. Cells.
10:19642021. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Vagnucci AH Jr and Li WW: Alzheimer's
disease and angiogenesis. Lancet. 361:605–608. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Carmeliet P: Angiogenesis in health and
disease. Nat Med. 9:653–660. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Danese S, Sans M, de la Motte C, Graziani
C, West G, Phillips MH, Pola R, Rutella S, Willis J, Gasbarrini A
and Fiocchi C: Angiogenesis as a novel component of inflammatory
bowel disease pathogenesis. Gastroenterology. 130:2060–2073. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Danese S, Sans M, Spencer DM, Beck I,
Doñate F, Plunkett ML, de la Motte C, Redline R, Shaw DE, Levine
AD, et al: Angiogenesis blockade as a new therapeutic approach to
experimental colitis. Gut. 56:855–862. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Chidlow JH Jr, Langston W, Greer JJ,
Ostanin D, Abdelbaqi M, Houghton J, Senthilkumar A, Shukla D, Mazar
AP, Grisham MB and Kevil CG: Differential angiogenic regulation of
experimental colitis. Am J Pathol. 169:2014–2030. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Maconi G, Sampietro GM, Russo A, Bollani
S, Cristaldi M, Parente F, Dottorini F and Bianchi Porro G: The
vascularity of internal fistulae in Crohn's disease: An in vivo
power Doppler ultrasonography assessment. Gut. 50:496–500. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Spalinger J, Patriquin H, Miron MC, Marx
G, Herzog D, Dubois J, Dubinsky M and Seidman EG: Doppler US in
patients with crohn disease: Vessel density in the diseased bowel
reflects disease activity. Radiology. 217:787–791. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Hatoum OA and Binion DG: The vasculature
and inflammatory bowel disease: Contribution to pathogenesis and
clinical pathology. Inflamm Bowel Dis. 11:304–313. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Folkman J and Shing Y: Angiogenesis. J
Biol Chem. 267:10931–10934. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Szekanecz Z and Koch AE: Vascular
endothelium and immune responses: Implications for inflammation and
angiogenesis. Rheum Dis Clin North Am. 30:97–114. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Colgan SP, Furuta GT and Taylor CT:
Hypoxia and innate immunity: Keeping Up with the HIFsters. Annu Rev
Immunol. 38:341–363. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Ucuzian AA, Gassman AA, East AT and
Greisler HP: Molecular mediators of angiogenesis. J Burn Care Res.
31:158–175. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Danese S, Papa A, Saibeni S, Repici A,
Malesci A and Vecchi M: Inflammation and coagulation in
inflammatory bowel disease: The clot thickens. Am J Gastroenterol.
102:174–186. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Ginnan R, Guikema BJ, Halligan KE, Singer
HA and Jourd'heuil D: Regulation of smooth muscle by inducible
nitric oxide synthase and NADPH oxidase in vascular proliferative
diseases. Free Radic Biol Med. 44:1232–1245. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Benelli R, Lorusso G, Albini A and Noonan
DM: Cytokines and chemokines as regulators of angiogenesis in
health and disease. Curr Pharm Des. 12:3101–3115. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Chidlow JH Jr, Shukla D, Grisham MB and
Kevil CG: Pathogenic angiogenesis in IBD and experimental colitis:
New ideas and therapeutic avenues. Am J Physiol Gastrointest Liver
Physiol. 293:G5–G18. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Deban L, Correale C, Vetrano S, Malesci A
and Danese S: Multiple pathogenic roles of microvasculature in
inflammatory bowel disease: A Jack of all trades. Am J Pathol.
172:1457–1466. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Pousa ID, Maté J and Gisbert JP:
Angiogenesis in inflammatory bowel disease. Eur J Clin Invest.
38:73–81. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Hicklin DJ and Ellis LM: Role of the
vascular endothelial growth factor pathway in tumor growth and
angiogenesis. J Clin Oncol. 23:1011–1027. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Algaba A, Linares PM, Fernández-Contreras
ME, Ordoñez A, Trápaga J, Guerra I, Chaparro M, de la Poza G,
Gisbert JP and Bermejo F: Relationship between levels of angiogenic
and lymphangiogenic factors and the endoscopic, histological and
clinical activity, and acute-phase reactants in patients with
inflammatory bowel disease. J Crohns Colitis. 7:e569–579. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Frysz-Naglak D, Fryc B, Klimacka-Nawrot E,
Mazurek U, Suchecka W, Kajor M, Kurek J and Stadnicki A:
Expression, localization and systemic concentration of vascular
endothelial growth factor (VEGF) and its receptors in patients with
ulcerative colitis. Int Immunopharmacol. 11:220–225. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Yamazaki Y and Morita T: Molecular and
functional diversity of vascular endothelial growth factors. Mol
Divers. 10:515–527. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Melincovici CS, Boşca AB, Şuşman S,
Mărginean M, Mihu C, Istrate M, Moldovan IM, Roman AL and Mihu CM:
Vascular endothelial growth factor (VEGF)-key factor in normal and
pathological angiogenesis. Rom J Morphol Embryol. 59:455–467.
2018.PubMed/NCBI
|
|
216
|
Shibuya M: VEGF-VEGFR system as a target
for suppressing inflammation and other diseases. Endocr Metab
Immune Disord Drug Targets. 15:135–144. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Sarabipour S and Mac Gabhann F: VEGF-A121a
binding to Neuropilins-A concept revisited. Cell Adh Migr.
12:204–214. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Sandor Z, Deng XM, Khomenko T, Tarnawski
AS and Szabo S: Altered angiogenic balance in ulcerative colitis: A
key to impaired healing? Biochem Biophys Res Commun. 350:147–150.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Tacconi C, Schwager S, Cousin N, Bajic D,
Sesartic M, Sundberg JP, Neri D and Detmar M: Antibody-mediated
delivery of VEGFC ameliorates experimental chronic colitis. ACS
Pharmacol Transl Sci. 2:342–352. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Koutroubakis IE, Tsiolakidou G, Karmiris K
and Kouroumalis EA: Role of angiogenesis in inflammatory bowel
disease. Inflamm Bowel Dis. 12:515–523. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Simons M, Gordon E and Claesson-Welsh L:
Mechanisms and regulation of endothelial VEGF receptor signalling.
Nat Rev Mol Cell Biol. 17:611–625. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Dimmeler S and Zeiher AM: Akt takes center
stage in angiogenesis signaling. Circ Res. 86:4–5. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Fryer BH and Field J: Rho, Rac, Pak and
angiogenesis: Old roles and newly identified responsibilities in
endothelial cells. Cancer Lett. 229:13–23. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Chen YJ, Chan DC, Chiang CK, Wang CC, Yang
TH, Lan KC, Chao SC, Tsai KS, Yang RS and Liu SH: Advanced
glycation end-products induced VEGF production and inflammatory
responses in human synoviocytes via RAGE-NF-κB pathway activation.
J Orthop Res. 34:791–800. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Liang H, Zhong Y, Zhou S and Peng L:
Knockdown of RAGE expression inhibits colorectal cancer cell
invasion and suppresses angiogenesis in vitro and in vivo. Cancer
Lett. 313:91–98. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Sha L, Zhao Y, Li S, Wei D, Tao Y and Wang
Y: Insights to Ang/Tie signaling pathway: Another rosy dawn for
treating retinal and choroidal vascular diseases. J Transl Med.
22:8982024. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Fagiani E and Christofori G: Angiopoietins
in angiogenesis. Cancer Lett. 328:18–26. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Fang Z, Qu S, Ji X, Zheng C, Mo J, Xu J,
Zhang J and Shen H: Correlation between PDGF-BB and M1-type
macrophage in inflammatory bowel disease: A case-control study. BMC
Gastroenterol. 24:4172024. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Chen X, Zhang L, Zhang IY, Liang J, Wang
H, Ouyang M, Wu S, da Fonseca ACC, Weng L, Yamamoto Y, et al: RAGE
expression in tumor-associated macrophages promotes angiogenesis in
glioma. Cancer Res. 74:7285–7297. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Lan J, Luo H, Wu R, Wang J, Zhou B, Zhang
Y, Jiang Y and Xu J: Internalization of HMGB1 (High Mobility Group
Box 1) promotes angiogenesis in endothelial cells. Arterioscler
Thromb Vasc Biol. 40:2922–2940. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
231
|
Zheng J, Zhu W, He F, Li Z, Cai N and Wang
HH: An aptamer-based antagonist against the receptor for advanced
glycation end-products (RAGE) blocks development of colorectal
cancer. Mediators Inflamm. 2021:99580512021. View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Soro-Paavonen A, Watson AM, Li J, Paavonen
K, Koitka A, Calkin AC, Barit D, Coughlan MT, Drew BG, Lancaster
GI, et al: Receptor for advanced glycation end products (RAGE)
deficiency attenuates the development of atherosclerosis in
diabetes. Diabetes. 57:2461–2469. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Ming L, Ji J, Luo Z, Zhao Z, Li M, Sun R,
Lu W, Chen Y and Huang Q: Sishen pill alleviates DSS induced
colitis through AGE/RAGE/NLRP3 pathway based on transcriptomics
analysis. J Ethnopharmacol. 356:1208182026. View Article : Google Scholar : PubMed/NCBI
|
|
234
|
He X, Zhou C, Shang R and Wang X:
Acanthoside B attenuates NLRP3-mediated pyroptosis and ulcerative
colitis through inhibition of tAGE/RAGE pathway. Allergol
Immunopathol (Madr). 53:112–122. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
235
|
Zhang J, Jiao Y, Hou S, Tian T, Yuan Q,
Hao H, Wu Z and Bao X: S100A4 contributes to colitis development by
increasing the adherence of Citrobacter rodentium in intestinal
epithelial cells. Sci Rep. 7:120992017. View Article : Google Scholar : PubMed/NCBI
|
|
236
|
Aljohi A, Matou-Nasri S, Liu D, Al-Khafaji
N, Slevin M and Ahmed N: Momordica charantia extracts protect
against inhibition of endothelial angiogenesis by advanced
glycation endproducts in vitro. Food Funct. 9:5728–5739. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Watson AM, Gray SP, Jiaze L, Soro-Paavonen
A, Wong B, Cooper ME, Bierhaus A, Pickering R, Tikellis C, Tsorotes
D, et al: Alagebrium reduces glomerular fibrogenesis and
inflammation beyond preventing RAGE activation in diabetic
apolipoprotein E knockout mice. Diabetes. 61:2105–2113. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
238
|
Zhang M, Li Y, Rao P, Huang K, Luo D, Cai
X and Xiao J: Blockade of receptors of advanced glycation end
products ameliorates diabetic osteogenesis of adipose-derived stem
cells through DNA methylation and Wnt signalling pathway. Cell
Prolif. 51:e124712018. View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Arab HH, Abd El-Aal SA, Eid AH, Arafa EA,
Mahmoud AM and Ashour AM: Targeting inflammation, autophagy, and
apoptosis by troxerutin attenuates methotrexate-induced renal
injury in rats. Int Immunopharmacol. 103:1082842022. View Article : Google Scholar : PubMed/NCBI
|
|
240
|
He F, Gu L, Cai N, Ni J, Liu Y, Zhang Q
and Wu C: The HMGB1-RAGE axis induces apoptosis in acute
respiratory distress syndrome through PERK/eIF2α/ATF4-mediated
endoplasmic reticulum stress. Inflamm Res. 71:1245–1260. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
241
|
Tang Z, Yang G, Liao Z, Chen F, Chen S,
Wang W, Huo G, Sun X and Wang X: Tanshinone IIA reduces AQP4
expression and astrocyte swelling after OGD/R by inhibiting the
HMGB1/RAGE/NF-κB/IL-6 pro-inflammatory axis. Sci Rep. 12:141102022.
View Article : Google Scholar : PubMed/NCBI
|
|
242
|
Shihui M, Shirong Y, Jing L, Jingjing H,
Tongqian W, Tian T, Chenyu W and Fang Y: S100A4 reprofiles lipid
metabolism in mast cells via RAGE and PPAR-gamma signaling pathway.
Int Immunopharmacol. 128:1115552024. View Article : Google Scholar : PubMed/NCBI
|
|
243
|
Ding B, Lin C, Liu Q, He Y, Ruganzu JB,
Jin H, Peng X, Ji S, Ma Y and Yang W: Tanshinone IIA attenuates
neuroinflammation via inhibiting RAGE/NF-κB signaling pathway in
vivo and in vitro. J Neuroinflammation. 17:3022020. View Article : Google Scholar : PubMed/NCBI
|
|
244
|
Liu W, Wu C, Wang Q, Kuang L and Le A:
Tanshinone IIA relieves arthritis by inhibiting autophagy of
fibroblast-like synoviocytes via matrix metalloproteinase9/receptor
for advanced glycation end product/toll-like receptor 9 signal axis
in mice with collagen-induced arthritis. Phytother Res.
37:1391–1404. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
245
|
Parthasarathy G, Mauer AS, Golla N, Daniel
PV, Kim LH, Sidhu GS, Marek GW III, Loeuillard E, Krishnan A, Lee
HSK, et al: Macrophage RAGE activation is proinflammatory in NASH.
JCI Insight. 9:e1691382024.PubMed/NCBI
|
|
246
|
Zhang C, Wang L, Xu Y, Huang Y, Huang J,
Zhu J, Wang W, Li W, Sun A, Li X, et al: Discovery of novel dual
RAGE/SERT inhibitors for the potential treatment of the comorbidity
of Alzheimer's disease and depression. Eur J Med Chem.
236:1143472022. View Article : Google Scholar : PubMed/NCBI
|
|
247
|
Algaba A, Linares PM, Encarnación
Fernández-Contreras M, Figuerola A, Calvet X, Guerra I, de Pousa I,
Chaparro M, Gisbert JP and Bermejo F: The effects of infliximab or
adalimumab on vascular endothelial growth factor and angiopoietin 1
angiogenic factor levels in inflammatory bowel disease: Serial
observations in 37 patients. Inflamm Bowel Dis. 20:695–702. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
248
|
Deng X, Tolstanova G, Khomenko T, Chen L,
Tarnawski A, Szabo S and Sandor Z: Mesalamine restores angiogenic
balance in experimental ulcerative colitis by reducing expression
of endostatin and angiostatin: Novel molecular mechanism for
therapeutic action of mesalamine. J Pharmacol Exp Ther.
331:1071–1078. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
249
|
Knod JL, Crawford K, Dusing M and Frischer
JS: Murine colitis treated with multitargeted tyrosine kinase
inhibitors. J Surg Res. 200:501–507. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
250
|
Liu XM, Zhu WT, Jia ML, Li YT, Hong Y, Liu
ZQ and Yan PK: Rapamycin liposomes combined with 5-fluorouracil
inhibits angiogenesis and tumor growth of APC((Min/+))
mice and AOM/DSS-induced colorectal cancer mice. Int J
Nanomedicine. 17:5049–5061. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
251
|
Tang T, Abu-Sbeih H, Ma W, Lu Y, Luo W,
Foo WC, Richards DM, Halperin DM, Ge PS and Wang Y:
Gastrointestinal injury related to antiangiogenesis cancer therapy.
Clin Colorectal Cancer. 19:e117–e123. 2020.PubMed/NCBI
|
|
252
|
Meijer B, Hoskin T, Ashcroft A, Burgess L,
Keenan JI, Falvey J, Gearry RB and Day AS: Total soluble and
endogenous secretory receptor for advanced glycation endproducts
(RAGE) in IBD. J Crohns Colitis. 8:513–520. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
253
|
Făgărășan A and Săsăran MO: The predictive
role of plasma biomarkers in the evolution of aortopathies
associated with congenital heart malformations. Int J Mol Sci.
23:49932022. View Article : Google Scholar
|
|
254
|
Yilmaz Y, Yonal O, Eren F, Atug O and
Hamzaoglu HO: Serum levels of soluble receptor for advanced
glycation endproducts (sRAGE) are higher in ulcerative colitis and
correlate with disease activity. J Crohns Colitis. 5:402–406. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
255
|
van der Heijden MS, Powles T, Petrylak D,
de Wit R, Necchi A, Sternberg CN, Matsubara N, Nishiyama H,
Castellano D, Hussain SA, et al: Predictive biomarkers for survival
benefit with ramucirumab in urothelial cancer in the RANGE trial.
Nat Commun. 13:18782022. View Article : Google Scholar : PubMed/NCBI
|
|
256
|
Heil F, Babitzki G, Julien-Laferriere A,
Ooi CH, Hidalgo M, Massard C, Martinez-Garcia M, Le Tourneau C,
Kockx M, Gerber P, et al: Vanucizumab mode of action: Serial
biomarkers in plasma, tumor, and skin-wound-healing biopsies.
Transl Oncol. 14:1009842021. View Article : Google Scholar : PubMed/NCBI
|
|
257
|
Soria JC, Mauguen A, Reck M, Sandler AB,
Saijo N, Johnson DH, Burcoveanu D, Fukuoka M, Besse B and Pignon
JP; meta-analysis of bevacizumab in advanced NSCLC collaborative
group, : Systematic review and meta-analysis of randomised, phase
II/III trials adding bevacizumab to platinum-based chemotherapy as
first-line treatment in patients with advanced non-small-cell lung
cancer. Ann Oncol. 24:20–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
258
|
Eder P, Korybalska K, Linke K and Witowski
J: Angiogenesis-related proteins-their role in the pathogenesis and
treatment of inflammatory bowel disease. Curr Protein Pept Sci.
16:249–258. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
259
|
Mateescu RB, Bastian AE, Nichita L,
Marinescu M, Rouhani F, Voiosu AM, Benguş A, Tudoraşcu DR and Popp
CG: Vascular endothelial growth factor-key mediator of angiogenesis
and promising therapeutical target in ulcerative colitis. Rom J
Morphol Embryol. 58:1339–1345. 2017.PubMed/NCBI
|
|
260
|
Xu X, Ying H, Wang X, Hong W and Zhang M:
Identification of Angiogenesis-related gene signatures and
prediction of potential therapeutic targets in ulcerative colitis
using integrated bioinformatics. J Inflamm Res. 17:11699–11717.
2024. View Article : Google Scholar : PubMed/NCBI
|