Introduction
Pituitary adenomas (PAs) are among the most common
intracranial tumors, accounting for 10–15% of all primary brain
neoplasms (1). Previous studies
have shown that 30–40% of PAs exhibit varying degrees of invasive
behavior (2), most commonly
involving invasion of the cavernous sinus, sellar dura mater,
osseous structures and sphenoid sinus (3). Invasive growth markedly increases the
difficulty of achieving gross-total resection and is closely
associated with an increased risk of postoperative tumor residue
and recurrence (4). The mechanisms
underlying invasive growth in PAs have been investigated in
experimental and clinical studies (5–8).
However, despite advances in surgical techniques and adjuvant
therapies, effective clinical control of invasive behavior remains
suboptimal. Intraoperative and pathological observations suggest a
recurrent inferior extension pattern: After breaching the sellar
dura and eroding the sellar floor, the tumor may extend toward/into
the sphenoid sinus, whereas frank sinonasal epithelial presentation
beyond the sphenoid sinus mucosa is uncommon (2). The sphenoid sinus mucosa is now
recognized as an immunologically active tissue, in which resident
and recruited immune cells cooperatively maintain barrier
integrity, immune surveillance, tissue homeostasis and post-injury
repair (9–11). Limited clinical evidence also
suggests the presence of mucosal immune activation during pituitary
apoplexy (12). Despite these
observations, little progress has been made in the study of the
sphenoid sinus mucosa since its initial description in association
with PAs in 1987 (13).
Consequently, a critical gap remains in the
understanding of how invasive PA cells interact with the sphenoid
sinus microenvironment. The cellular composition, activation states
and regulatory programs of mucosal immune populations at the
tumor-mucosa interface remain poorly defined. Addressing this gap
is therefore essential for refining current models of
tumor-microenvironment interactions and for identifying tractable
immunological targets to limit invasive progression. The present
study characterized the immune microenvironment of sphenoid sinus
mucosa adjacent to sphenoid sinus-invasive PAs, compared it with
appropriate control mucosa, and evaluated the associations between
mucosal immune features and patterns of local invasion. These
insights may provide a mechanistic basis for strategies aimed at
preventing or mitigating invasive behavior in PAs.
Materials and methods
Definition of invasiveness
PAs are classified into subtypes based on
immunohistochemical profiles and clinical manifestations (14). Non-invasive tumors (NITs) were
defined using a multimodal approach: i) Radiologically,
preoperative sellar MRI with contrast-enhanced coronal T1-weighted
imaging confirmed the distinct boundaries between the PA and
internal carotid artery (ICA) (15); ii) intraoperatively, direct
visualization by an experienced pituitary surgeon revealed intact
tumor pseudocapsules without cavernous sinus infiltration or
tumor-ICA spatial invasion (3);
and iii) routine clinical histopathological examination of the
anterior sphenoid sinus dural specimens, performed by the
Department of Pathology as part of routine clinical care, showed no
evidence of tumor cell infiltration based on H&E staining,
indicating the absence of dural invasion at the sampled site
(16).
Dural-invasive tumors (DITs) were identified
histopathologically, requiring both tumor cell infiltration into
the dural collagenous layer and structural disruption of dural
continuity. Sphenoid sinus-invasive tumors (SSITs) were defined as
follows: i) Preoperative MRI criteria included destruction of the
sphenoid sinus structure with tumor filling the sinus cavity; ii)
intraoperative criteria involved direct visualization of mucosa
displacement and sinus cavity loss after anterior wall resection;
and iii) tumor invasion into the sphenoid sinus was confirmed by
H&E staining according to the routine clinical pathology report
(17).
Study population
Consecutive patients who underwent endoscopic
transsphenoidal surgery for pituitary tumors at Tongji Hospital,
Tongji Medical College, Huazhong University of Science and
Technology (Wuhan, China) between January 2023 and January 2024
were screened for inclusion. Patients were excluded if they were
younger than 18 years of age or had undergone previous pituitary
surgery. Following application of these predefined criteria, a
total of 63 patients were included in the final statistical
analysis. The study cohort consisted of 29 men (mean age, 52.4±12.5
years; age range, 19–77 years) and 34 women (mean age, 40.7±14.4
years; age range, 19–69 years). Following application of the
predefined criteria, a total of 63 patients were included in the
final statistical analysis. Patients were categorized into NIT
(n=32), DIT (n=21) and SSIT (n=10) groups. Clinical imaging and
routine pathology information used for classification was retrieved
from the medical records, including preoperative MRI (sellar region
images and tumor size measurements were extracted from radiology
reports) and routine clinical histopathology reports.
H&E staining, immunohistochemistry
(IHC) and immunofluorescence staining
Clinical tissues were fixed in 4% formaldehyde at
4°C for 24 h, embedded in paraffin and sectioned into 4-µm-thick
slices. Sections were baked at 60°C for 2 h, deparaffinized in
xylene and rehydrated through a descending graded ethanol series
(100, 95, 80 and 70% ethanol; 5 min each) to water. For IHC and
immunofluorescence staining, antigen retrieval was performed by
heating slides in EDTA buffer (G1203; Wuhan Servicebio Technology
Co., Ltd.) at 95°C for 20 min.
H&E staining
After deparaffinization and rehydration, sections
were exposed to hematoxylin (G1004; Wuhan Servicebio Technology
Co., Ltd.) for 5 min at room temperature, briefly differentiated in
1% acid-alcohol for 10 sec, blued under running water for 5 min and
counterstained with eosin for 1 min at room temperature. Slides
were then dehydrated using ethanol, cleared in xylene and mounted.
Images were acquired using a bright-field light microscope (Olympus
Corporation) and analyzed using ImageJ (v1.53; National Institutes
of Health).
IHC
Following antigen retrieval, endogenous peroxidase
was quenched with 3% H2O2 for 15 min at room
temperature. To minimize non-specific binding, sections were
blocked in 5% BSA (GC305006-100 g; Wuhan Servicebio Technology Co.,
Ltd.) for 60 min at room temperature and then incubated with
primary antibodies overnight at 4°C. Biotinylated secondary
antibodies were applied for 2 h at room temperature (Table SI), after which the
streptavidin-biotin complex was added for 30 min. Signals were
developed using a 3,3′-diaminobenzidine kit [GK600510; Gene
Technology (Shanghai) Co., Ltd.] and nuclei were counterstained
with hematoxylin. Images were acquired using bright-field light
microscopy.
Immunofluorescence staining
Following antigen retrieval, sections were
permeabilized with 0.1% Triton X-100 in PBS for 10 min at room
temperature and blocked with 5% BSA (GC305006-100 g; Wuhan
Servicebio Technology Co., Ltd.) for 60 min at room temperature.
Sections were then incubated with primary antibodies overnight at
4°C (Table SI), followed by
incubation with the appropriate secondary antibodies for 2 h at
room temperature (Table SI).
Nuclei were labeled with DAPI for 15 min at room temperature and
sections were mounted in an anti-fade medium. Images were acquired
using a fluorescence microscope and analyzed using ImageJ (v1.53;
National Institutes of Health) using threshold-based segmentation
or grayscale intensity measurements.
Masson staining
Paraffin-embedded tissue sections (4 µm) were baked
(60°C), deparaffinized in xylene (room temperature) and rehydrated
through graded ethanols to water (room temperature). Sections were
fixed overnight in Bouin's or Zenker's solution (room temperature),
washed with distilled water (room temperature) and stained with
Harris hematoxylin (8 min; room temperature). Differentiation in
0.8% acid alcohol (10 sec; room temperature) was followed by bluing
in lithium carbonate (30 sec; room temperature). Sections were
stained with picrosirius red (10 min; room temperature),
differentiated in phosphotungstic acid (5 min; room temperature),
counterstained with aniline blue (5 min; room temperature),
dehydrated (room temperature), cleared (room temperature) and
mounted (room temperature). Observations were conducted using
bright-field light microscopy (Olympus Corporation), and
quantification was performed using ImageJ (v1.53; National
Institutes of Health).
Histological quantification
Mucosal thickness was quantified on histological
sections using ImageJ (v1.53; National Institutes of Health).
Thickness was defined as the perpendicular distance from the
epithelial surface (basement membrane) to the outer boundary of the
submucosa. For each specimen, measurements were taken at ≥5
randomly selected positions per section (avoiding folds/tears),
averaged to obtain a specimen-level value and then summarized by
group for statistical analysis.
Primary cell isolation and
culture
Primary cells were isolated from pituitary tumor
tissues obtained from 3 of the aforementioned 63 patients, and
samples from each patient were processed and cultured separately
without pooling. Fresh mucosal tissue samples from surgical
resection were stored on ice, with the digestion process completed
within 1.5 h (no longer than 2 h). The tissue blocks were washed
three times with pre-chilled PBS (1% penicillin-streptomycin) at
4°C to remove residual blood and necrotic components. Subsequently,
the tissue was finely chopped into 1–3-mm3 fragments.
For the generation of digested mucosal culture (DMC), the minced
mucosal tissue was transferred to a tube containing 10 ml digestion
solution consisting of 2 mg/ml collagenase type I (cat. no.
40507ES60; Shanghai Yeasen Biotechnology Co., Ltd.) in complete
DMEM [DMEM (G4523; Wuhan Servicebio Technology Co., Ltd.)
supplemented with 10% FBS (BMC1021; Abbkine Scientific Co., Ltd.)
and 1% penicillin-streptomycin (G4003; Wuhan Servicebio Technology
Co., Ltd.; 100 U/ml penicillin and 100 µg/ml streptomycin)] and
incubated at 37°C in a shaking incubator (100 rpm) for 1 h. Every
20 min, gentle pipetting was performed to facilitate tissue
dissociation. The digestion was terminated when the tissue became
translucent and the solution became turbid. After digestion, the
mixture was filtered through a 40-µm cell strainer, and cells were
collected by centrifugation at 300 × g for 5 min at 4°C. The cell
pellet was resuspended in complete DMEM and seeded into
collagen-coated culture dishes. Cells were incubated at 37°C with
5% CO2, with the medium changed after 6 h to remove
non-adherent cells and debris. For mucosal tissue culture (MTC),
intact sphenoid sinus mucosal tissue fragments were placed onto
0.4-µm polycarbonate membrane inserts, with the membrane positioned
at the same level as the culture medium to establish an air-liquid
interface. For primary PA cells, PA tissue was treated as
aforementioned, and digestion was carried out with 0.25% trypsin
(G4001; Wuhan Servicebio Technology Co., Ltd.) at 37°C for 30 min.
Complete DMEM was added to halt digestion. Cells were centrifuged
(300 × g; 5 min; 4°C), resuspended in complete DMEM and
subsequently seeded. Before co-culture, a pilot experiment was
conducted to assess the growth of digested primary PA cells and
mucosal cells after plating. Cell morphology and growth were
observed under an inverted light microscope on days 1, 3 and 5
after seeding. For the tumor-mucosal co-culture model, the cells
(2×105/well) were seeded in the bottom of a 24-well
plate, while a non-contact co-culture system was established by
seeding mucosal cells (2×105/well) in 0.4-µm
polycarbonate membrane inserts (3470; Corning, Inc.). After
incubation for 48 h at 37°C, cells and supernatants were collected
for subsequent functional assays.
Cell cycle analysis
TtT/GF cells (~1×106) were treated with
the indicated concentrations of IFN-γ (0–100 ng/ml; cat. no.
RP01038; ABclonal Biotech Co., Ltd.) or IL-6 (0–100 ng/ml; cat. no.
RP00201; ABclonal Biotech Co., Ltd.) alone for 48 h at 37°C. Cells
treated with ruxolitinib alone were incubated with ruxolitinib (5
µM; cat. no. HY-50856; MedChemExpress) for 48 h at 37°C. For
combination treatments, cells were pretreated with ruxolitinib for
30 min at 37°C before the simultaneous addition of IFN-γ and/or
IL-6, followed by incubation for 48 h at 37°C. Cells
(~1×106) were washed with ice-cold PBS, fixed in 70%
ethanol at 4°C for 24 h and stained with PI/RNase working solution
(BD Pharmingen; BD Biosciences) for 30 min at 37°C in the dark.
Data were acquired on a CytoFLEX cytometer (Beckman Coulter, Inc.)
and cell cycle fractions (G0/G1, S and
G2/M) were modeled in ModFit LT 6.0 (version 6.0; Verity
Software House, Inc.) using the Dean-Jett-Fox algorithm.
Apoptosis analysis
Pituitary tumor cells (1×105) were
resuspended in 1X binding buffer, labeled using an Annexin
V-FITC/PI kit (Shanghai Yeasen Biotechnology Co., Ltd.) for 15 min
at room temperature in the dark, diluted with 400 µl binding buffer
and analyzed immediately using the CytoFLEX cytometer. FlowJo v10.8
(version 10.8; BD Biosciences) was used for gating. Annexin
V+/PI− events were considered as early
apoptotic and Annexin V+/PI+ as late
apoptotic/necrotic.
RNA extraction, reverse transcription
and quantitative PCR (qPCR)
RAW264.7 macrophages in the M0, M1 or M2 state were
treated with IgG (10 µg/ml; cat. no. 14-4714-85; Invitrogen; Thermo
Fisher Scientific, Inc.) or anti-CD47 monoclonal antibody (mAb) (10
µg/ml; cat. no. 16-0479-85; Invitrogen; Thermo Fisher Scientific,
Inc.) at 37°C for 12 h. For polarization, M1 macrophages were
induced with lipopolysaccharide (100 ng/ml; cat. no. HY-D1056;
MedChemExpress) plus IFN-γ (20 ng/ml; cat. no. RP01070; ABclonal
Biotech Co., Ltd.) for 24 h at 37°C, whereas M2 macrophages were
induced with IL-4 (20 ng/ml; cat. no. RP01161; ABclonal Biotech
Co., Ltd.) for 24 h at 37°C. RAW264.7 mouse macrophages were
scraped and immediately lysed in TRIzol reagent (Invitrogen; Thermo
Fisher Scientific, Inc.). Total RNA was purified according to the
manufacturer's instructions, including chloroform phase separation,
isopropanol precipitation and a 75% ethanol wash, and quantified on
a NanoDrop spectrophotometer. Using 500 ng of RNA, first-strand
cDNA was generated with the Hifair III 1st Strand cDNA Synthesis
Kit (Shanghai Yeasen Biotechnology Co., Ltd.) at 42°C for 15 min.
qPCR was carried out with Hifair qPCR SYBR Green Master Mix (Low
Rox Plus) (Shanghai Yeasen Biotechnology Co., Ltd.) on an ABI 7500
Real-Time PCR system (Applied Biosystems; Thermo Fisher Scientific,
Inc.). The PCR thermocycling conditions were as follows: Initial
denaturation at 95°C for 5 min, followed by 40 cycles of 95°C for
10 sec and 60°C for 30 sec. GAPDH was used as the internal
reference gene (18). Relative
gene expression levels were calculated using the 2−ΔΔCq
method (18). Primer sequences are
listed in Table SII and were
designed using the National Center for Biotechnology Information
Primer-Basic Local Alignment Search tool (19).
Western blotting
TtT/GF cells were pretreated with ruxolitinib (5 µM;
cat. no. HY-50856; MedChemExpress) at 37°C for 30 min, followed by
treatment with IFN-γ (0–100 ng/ml; cat. no. RP01038; ABclonal
Biotech Co., Ltd.), IL-6 (100 ng/ml; cat. no. RP00201; ABclonal
Biotech Co., Ltd.) or a combination of IFN-γ (50 ng/ml each) and
IL-6 (50 ng/ml each) at 37°C for 48 h, as indicated. Cells were
lysed in RIPA lysis buffer (Wuhan Servicebio Technology Co., Ltd.)
supplemented with protease inhibitor cocktail (G2008; Wuhan
Servicebio Technology Co., Ltd.) and phosphatase inhibitor cocktail
(G2007; Wuhan Servicebio Technology Co., Ltd.) on ice for 30 min.
Protein concentrations were determined using a BCA protein assay
kit (Wuhan Servicebio Technology Co., Ltd.). Equal amounts of
protein (30 µg per lane) were separated using 10% SDS-PAGE gels
(G2037; Wuhan Servicebio Technology Co., Ltd.) and transferred onto
PVDF membranes (IPVH00010; MilliporeSigma). Membranes were blocked
with NcmBlot blocking buffer (P30500; Suzhou Xinsaimei
Biotechnology Co., Ltd.) for 1 h at room temperature, and then
incubated with primary antibodies (Table SI) at 4°C overnight. After washing
three times with Tris-buffered saline containing 0.1% Tween-20,
membranes were incubated with HRP-conjugated secondary antibodies
(SA00001-1/SA00001-2; 1:5,000; Proteintech Group, Inc.) for 2 h at
room temperature. Protein bands were visualized using an ECL
chemiluminescence kit (P10100; Guangzhou Saiguo Biotech Co., Ltd.),
and images were captured using a GeneGnome XRQ imaging system
(Syngene Europe). Band intensities were semi-quantified using
ImageJ software (version 1.53; National Institutes of Health).
ELISA
Supernatants were collected and clarified by
centrifugation (12,000 × g; 10 min; 4°C). Human IFN-γ, IL-1β, IL-6,
IL-10, TGF-β and TNF-α levels were quantified using commercial
ELISA kits (Human IFN-γ ELISA kit, cat. no. E-EL-H0108; Human IL-1β
ELISA kit, cat. no. E-EL-H0149; Human IL-6 ELISA kit, cat. no.
E-EL-H0102; Human IL-10 ELISA kit, cat. no. E-EL-H0103; Human TGF-β
ELISA kit, cat. no. E-EL-H0110; Human TNF-α ELISA kit, cat. no.
E-EL-H0109; Wuhan Elabscience Biotechnology Co., Ltd.). Briefly,
100 µl of sample or serially diluted standards, together with a
medium-only negative control and a recombinant-protein positive
control, were dispensed into each well and samples were incubated
for 90 min at room temperature. Plates were then treated
sequentially with biotinylated detection antibody and
streptavidin-HRP (SA00001 series; 100 µl/well; Proteintech Group,
Inc.), with incubation for 30 min at 37°C in the dark for each
step. 3,3′,5,5′-Tetramethylbenzidine substrate (E-IR-R307; Wuhan
Elabscience Biotechnology Co., Ltd.) was added, color was allowed
to develop for 15 min at room temperature in the dark, and the
reaction was stopped with 50 µl of 2 M H2SO4.
Absorbance at 450 nm was recorded on a Tecan Infinite F50 plate
reader (Tecan Group, Ltd.). Cytokine concentrations were calculated
from the standard curve and normalized to total protein content
determined using a BCA assay.
Green fluorescent protein (GFP)-TtT/GF
generation
GFP-expressing TtT/GF cells were generated by
lentiviral transduction. Parental TtT/GF cells (CL-0561; Procell
Life Science & Technology Co., Ltd.) were used in the present
study. Lentiviral particles were produced in 293T cells
(ATCC® CRL-3216™; American Type Culture Collection)
using a third-generation packaging system by co-transfecting the
enhanced green fluorescent protein (EGFP)-expressing lentiviral
transfer plasmid pLVX-CMV-EGFP-PGK-Puro together with the packaging
plasmids pMDLg/pRRE and pRSV-Rev and the envelope plasmid pMD2.G at
a mass ratio of 4:2:1:1, corresponding to 10, 5, 2.5 and 2.5 µg,
respectively, per 10-cm dish. All plasmids (transfer, packaging and
envelope plasmids) were constructed in-house by DesignGene
Biotechnology. Transfection was performed at 37°C for 6–8 h,
followed by replacement with fresh complete medium. Viral
supernatants were collected at 48 and 72 h post-transfection,
filtered through a 0.45-µm membrane and used immediately or stored
at −80°C. Parental TtT/GF cells were transduced at an MOI of 10 in
the presence of polybrene (8 µg/ml) for 12–16 h, followed by medium
replacement. Transduced cells were selected with puromycin (2
µg/ml) for 5–7 days and maintained in puromycin (1 µg/ml).
Subsequent experimentation was initiated 14 days after the start of
lentiviral transduction. This 14-day interval ensured complete
antibiotic selection, recovery of cellular homeostasis and the
establishment of stable GFP expression prior to functional assays.
GFP expression was confirmed by fluorescence microscopy. Cells were
cultured at 37°C with 5% CO2 in complete DMEM as
recommended by the supplier.
pHrodo™ phagocytosis assay
RAW264.7 mouse macrophages (ATCC®
TIB-71™; American Type Culture Collection) were maintained in
complete DMEM at 37°C with 5% CO2. Macrophages were
polarized for 24 h in complete DMEM as follows: M1 polarization was
induced by lipopolysaccharide (LPS; 100 ng/ml; L2880;
Sigma-Aldrich; Merck KGaA) and IFN-γ (20 ng/ml; 575302; BioLegend,
Inc.), and M2 polarization was induced by 20 ng/ml IL-4 (PHC0045;
Thermo Fisher Scientific, Inc.), both for 24 h at 37°C in complete
DMEM supplemented with 10% FBS. Polarized macrophages were treated
with an anti-CD47 mAb (10 µg/ml; 16-0479-85; Invitrogen; Thermo
Fisher Scientific, Inc.) for 12 h at 37°C, followed by incubation
for an additional 12 h at 37°C in serum-free DMEM. GFP-labeled
TtT/GF cells were pre-stained with pHrodo red dye (1:10,000 in PBS;
P35372; Thermo Fisher Scientific, Inc.) for 30 min at 37°C, and
then washed three times with PBS containing 1% BSA to remove
unbound dye. The co-culture system was established by seeding
polarized macrophages and pHrodo-labeled tumor cells at a 1:1 ratio
(5×105 cells each) into 6-well plates, followed by
incubation at 37°C with 5% CO2 for 24 h. After
incubation, cells were washed with PBS. Phagocytosis of GFP-labeled
TtT/GF cells by RAW264.7 macrophages was observed under a
fluorescence microscope (Olympus Corporation). Phagocytic events
were defined as RAW264.7 cells positive for pHrodo fluorescence
(excitation/emission, 560/585 nm), indicating uptake of
pHrodo-labeled tumor cells. Phagocytosis was quantified by flow
cytometry using a CytoFLEX V5-B5-R3 flow cytometer (Beckman
Coulter, Inc.) as the percentage of RAW264.7 cells positive for
pHrodo fluorescence, and data were analyzed using FlowJo (v10.8; BD
Biosciences) (20).
Wound healing assay
TtT/GF cells were plated in 6-well plates and
cultured in complete DMEM at 37°C with 5% CO2 until 100%
confluency. Vertical scratches were created with a 200-µl pipette
tip. Cells were maintained in serum-free DMEM and treated with
IFN-γ (0–100 ng/ml; cat. no. RP01038; ABclonal Biotech Co., Ltd.),
IL-6 (100 ng/ml; cat. no. RP00201; ABclonal Biotech Co., Ltd.) or a
combination of IFN-γ and IL-6 (50 ng/ml each) at 37°C for 72 h, as
indicated. Where applicable, cells were pretreated with ruxolitinib
(5 µM; cat. no. HY-50856; MedChemExpress) for 30 min at 37°C prior
to cytokine stimulation. During the 3-day culture period in
serum-free DMEM, wound closure was monitored and images were
captured every 24 h using an inverted bright-field light
microscope. Cell migration distances were quantified using ImageJ
software (version 1.53; National Institutes of Health).
5-ethynyl-2′-deoxyuridine (EdU) cell
proliferation assay
TtT/GF cells were seeded in 6- or 24-well plates and
cultured at 37°C with 5% CO2 until reaching 50–70%
confluency. For direct cytokine treatment experiments, TtT/GF cells
were treated with IFN-γ (0–100 ng/ml; cat. no. RP01038; ABclonal
Biotech Co., Ltd.) at 37°C for 48 h, as indicated. For co-culture
experiments, RAW264.7 macrophages were polarized for 24 h at 37°C
with lipopolysaccharide plus IFN-γ or with IL-4, using the same
inducing conditions as aforementioned, followed by treatment with
anti-CD47 mAb (10 µg/ml; cat. no. 16-0479-85; Invitrogen; Thermo
Fisher Scientific, Inc.) at 37°C for 12 h. Anti-CD47 mAb-treated
polarized macrophages were then co-cultured with TtT/GF cells for
EdU analysis. Cells were then incubated with pre-warmed EdU (50 µM)
in complete culture medium for 2 h at 37°C. After incubation, cells
were washed with PBS, fixed with freshly prepared 4%
paraformaldehyde for 20 min at room temperature, and permeabilized
with 0.5% Triton X-100. Cells were then blocked with 3% BSA for 30
min at room temperature. Subsequently, the cells were incubated
with the Click-iT reaction cocktail from the Click-iT™ EdU Cell
Proliferation Kit for Imaging (Alexa Fluor™ 594; cat. no. C10339;
Invitrogen; Thermo Fisher Scientific, Inc.), prepared according to
the manufacturer's instructions, for 30 min at room temperature in
the dark. Nuclei were counterstained with DAPI for 5 min at room
temperature in the dark, after which cells were washed with PBS and
mounted using an antifade mounting medium. EdU-positive cells were
visualized using a fluorescence microscope, and proliferation was
quantified using ImageJ software (version 1.53; National Institutes
of Health).
Statistical analysis
All statistical analyses were performed using
GraphPad Prism 6 (Dotmatics). Data are presented as the mean ± SD
from ≥3 independent experiments/biological replicates. Where
appropriate, measurements were normalized to the relevant control
and expressed as the percentage or fold-change. Statistical tests
were selected based on the experimental design and underlying
assumptions. For comparisons between two independent groups, an
unpaired two-sided Student's t-test was used. For paired
measurements, a paired two-sided t-test was applied. For one-factor
multi-group comparisons, one-way ANOVA followed by Tukey's post hoc
multiple comparisons test was performed. For experiments involving
two independent factors, two-way ANOVA with Bonferroni's multiple
comparisons test was used. For longitudinal/repeated measurements
on the same samples, repeated-measures ANOVA followed by
Bonferroni's multiple comparisons test was applied. When data did
not meet the assumptions for parametric testing, nonparametric
tests were used. Specifically, the Mann-Whitney U test was applied
for comparisons between two independent groups, the Wilcoxon
signed-rank test was used for paired nonparametric data, and the
Kruskal-Wallis test followed by Dunn's multiple comparisons test
was employed for multi-group comparisons. Bonferroni correction was
applied for multiple comparisons where appropriate. Correlations
were assessed using Pearson's correlation coefficient. P≤0.05 was
considered to indicate a statistically significant difference.
Results
Mucosa prevents tumor breach of the
sphenoid sinus
Intraoperative and pathological assessments
demonstrated that, in all SSIT cases included in the present study,
the lesion occupied the sphenoid sinus lumen but did not breach the
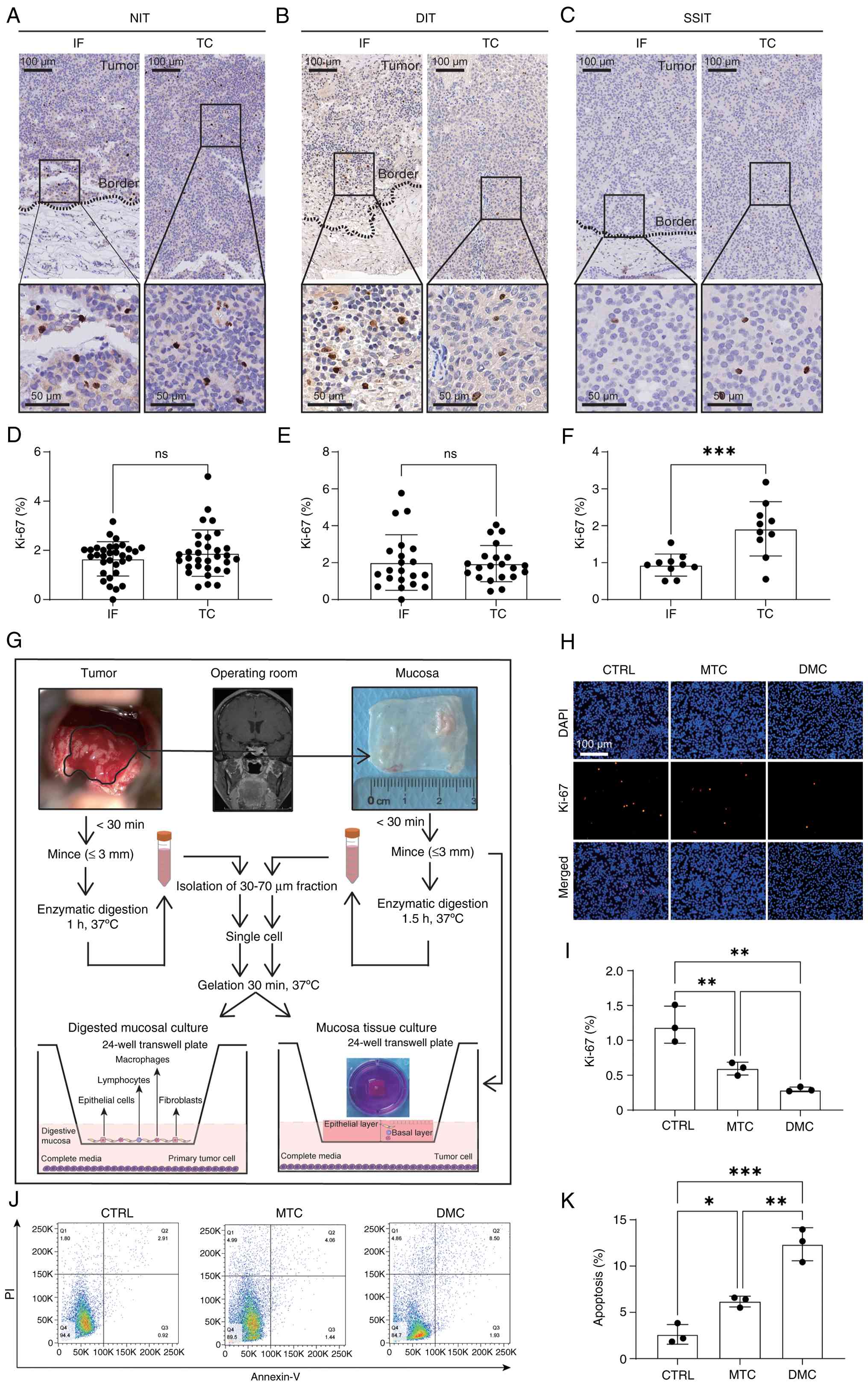
sinus mucosal wall to allow further extension (Fig. 1A). For a PA, inferior extension
into the sphenoid sinus requires a stepwise traversal of barriers,
including penetration of the tumor pseudocapsule, violation of the
dura mater and erosion of the sellar floor, before reaching the
sinus cavity (Fig. 1B). Consistent
with this trajectory, H&E staining demonstrated tumor cell
infiltration into the pseudocapsule with focal dural disruption,
while there was no infiltration or destruction of the basal layer
of the sinus mucosa (Fig. 1C).
Masson's trichrome staining revealed densely arranged collagen
fibers within the sphenoid sinus mucosa in the SSIT group, and the
mucosal thickness was comparable to that observed in the NIT and
DIT groups (Fig. 1D). These
observations suggested that pre-mucosal anatomical barriers
(pseudocapsule, dura and sellar floor) alone may be insufficient to
restrain tumor progression once erosion occurs, although they
constitute relevant physical obstacles (21–23).
Despite the larger tumor size in the SSIT group compared with that
in the NIT and DIT groups (Fig.
1E), the proliferation index at the SSIT invasive front (IF)
was significantly lower than that observed in the DIT group, but
not significantly different from the NIT group (Fig. S1A and B).
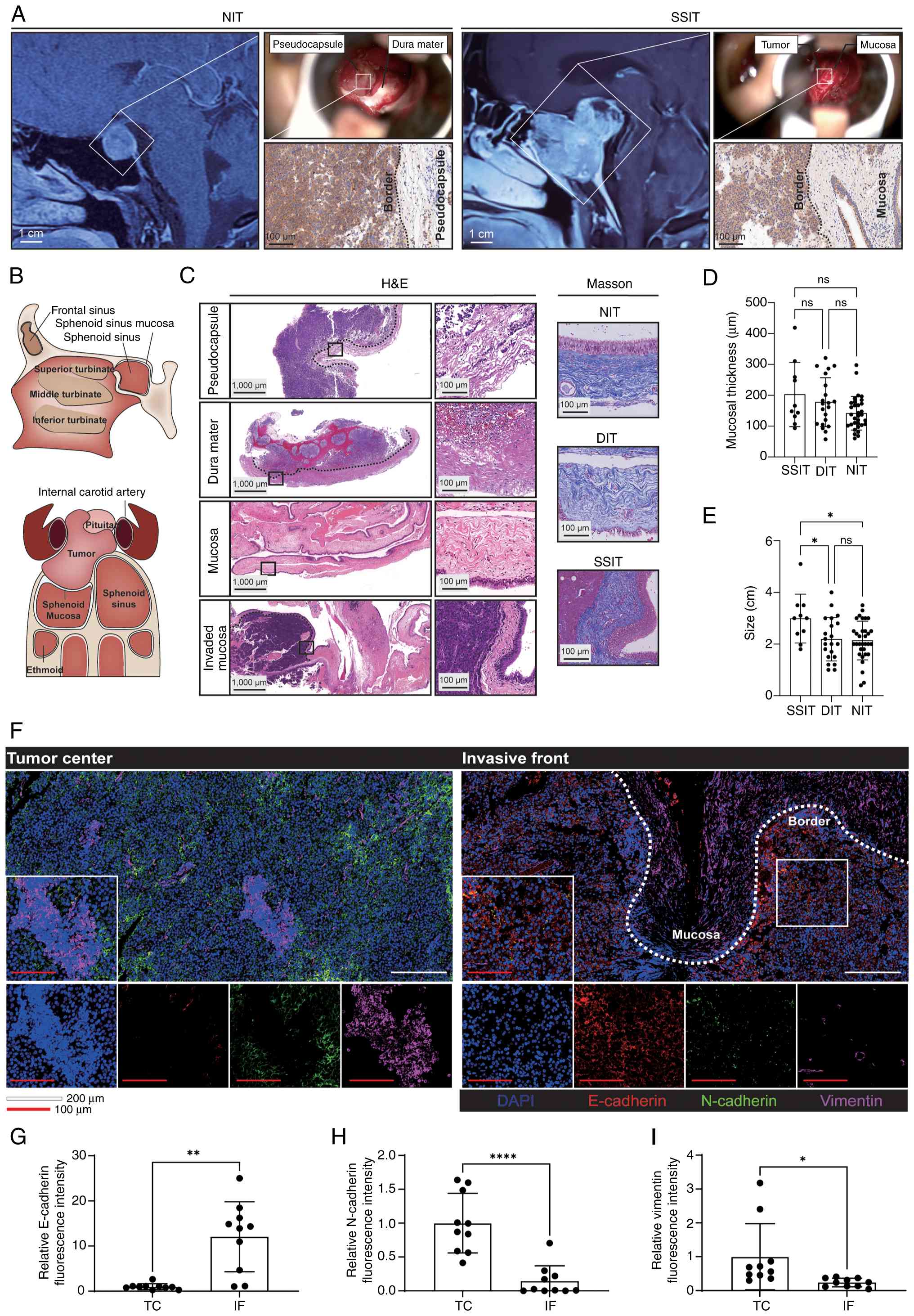 | Figure 1.Mucosal integrity at the tumor-sinus
interface. (A) Contrast-enhanced sagittal MRI, intraoperative
microscope view and prolactin immunohistochemistry staining from 2
representative cases. (B) Schematic of the sellar floor and
sphenoid sinus architecture, highlighting sequential barriers
encountered during inferior tumor extension. (C) Histopathological
characterization of invasion interfaces; H&E and Masson's
trichrome staining at the IF of pseudocapsular (n=32), dural (n=21)
and sphenoid sinus (n=10) sites, and in non-invaded sphenoid mucosa
(n=53), with (D) quantitative analyses of mucosal collagen
thickness. (E) Tumor dimension analysis for SSITs (n=10), DITs
(n=21) and NITs (n=32). (F) Multiplex immunofluorescence staining
of epithelial-mesenchymal transition markers at the IF vs. TC,
including E-cadherin (red), N-cadherin (green) and vimentin
(magenta). (G-I) Quantitative analysis of (G) E-cadherin, (H)
N-cadherin and (I) vimentin levels in paired IF vs. TC samples
(n=10). (D and E) One-way ANOVA with Tukey's post hoc multiple
comparisons test. (G-I) Two-tailed paired t-test. *P<0.05,
**P<0.01, ****P<0.0001. DIT, dural-invasive tumor; IF,
invasive front; NIT, non-invasive tumor; ns, not significant; SSIT,
sphenoid sinus-invasive tumor; TC, tumor core. |
As invasive behavior is often associated with
epithelial—mesenchymal transition (EMT) and stromal remodeling
(7,24), the present study subsequently
investigated EMT-related phenotypes. Multiplex immunofluorescence
staining demonstrated relative suppression of EMT at the SSIT IF
versus the tumor core (TC), characterized by higher E-cadherin
expression, and lower N-cadherin and vimentin expression (Fig. 1F-I). In parallel, IHC revealed
reduced MMP-2/9 expression at the IF compared with the TC in SSIT
cases (Fig. S1C and D). Taken
together, these findings were consistent with the notion that the
sphenoid sinus mucosa may exert an active, context-dependent
anti-invasive effect rather than serving solely as a passive
physical barrier.
Sphenoid sinus mucosa suppresses
growth of primary tumors
To further delineate the contribution of the
sphenoid sinus mucosa to PA progression, tumor cell proliferation
at the IF versus the TC was compared across SSIT (n=10), NIT (n=32)
and DIT (n=21) cases. Ki-67 IHC demonstrated lower proliferative
activity at the IF than at the paired TC in SSIT cases (Fig. 2A-F). By contrast, no significant
differences between the IF and TC were observed in NIT or DIT
cases. Mucosa-derived inhibitory effects on primary PA cells were
next evaluated using two complementary ex vivo models: i)
Air-liquid interface culture of sphenoid sinus mucosal tissue
(MTC); and ii) co-culture of enzymatically dissociated mucosal cell
preparations (DMC) with primary PA cells (Fig. 2G).
Air-liquid interface culture demonstrated that
mucosal tissue viability declined markedly by day 7 (Fig. S2A and B). Following enzymatic
digestion of the mucosa, fibroblast outgrowth from DMC preparations
became evident from day 5 onward, indicating progressive changes in
cellular composition during prolonged culture (Fig. S2C and D). Therefore, co-culture
assays were conducted within the validated viability window, and
48-h co-culture with either MTC or DMC significantly reduced tumor
cell proliferation (Fig. 2H and I)
and increased tumor cell death (Fig.
2J and K) compared with those of tumor cells cultured alone
(CTRL).
Mucosal tissues exhibit enrichment of
macrophages and B cells
As part of the respiratory mucosa, the sphenoid
sinus mucosa harbors abundant resident and recruited immune
populations (25). Immune
cell-subset distribution was profiled by IHC in pseudocapsule
(n=32), dural (n=31) and mucosal (n=63) tissues. Relative to the
pseudocapsule and dura mater, mucosal tissues displayed higher
infiltration densities of macrophages [ionised calcium binding
adaptor molecule 1 (IBA-1)+], CD4+ T cells,
CD8+ T cells and B cells (CD19+) (Figs. 3A-D and S3A).
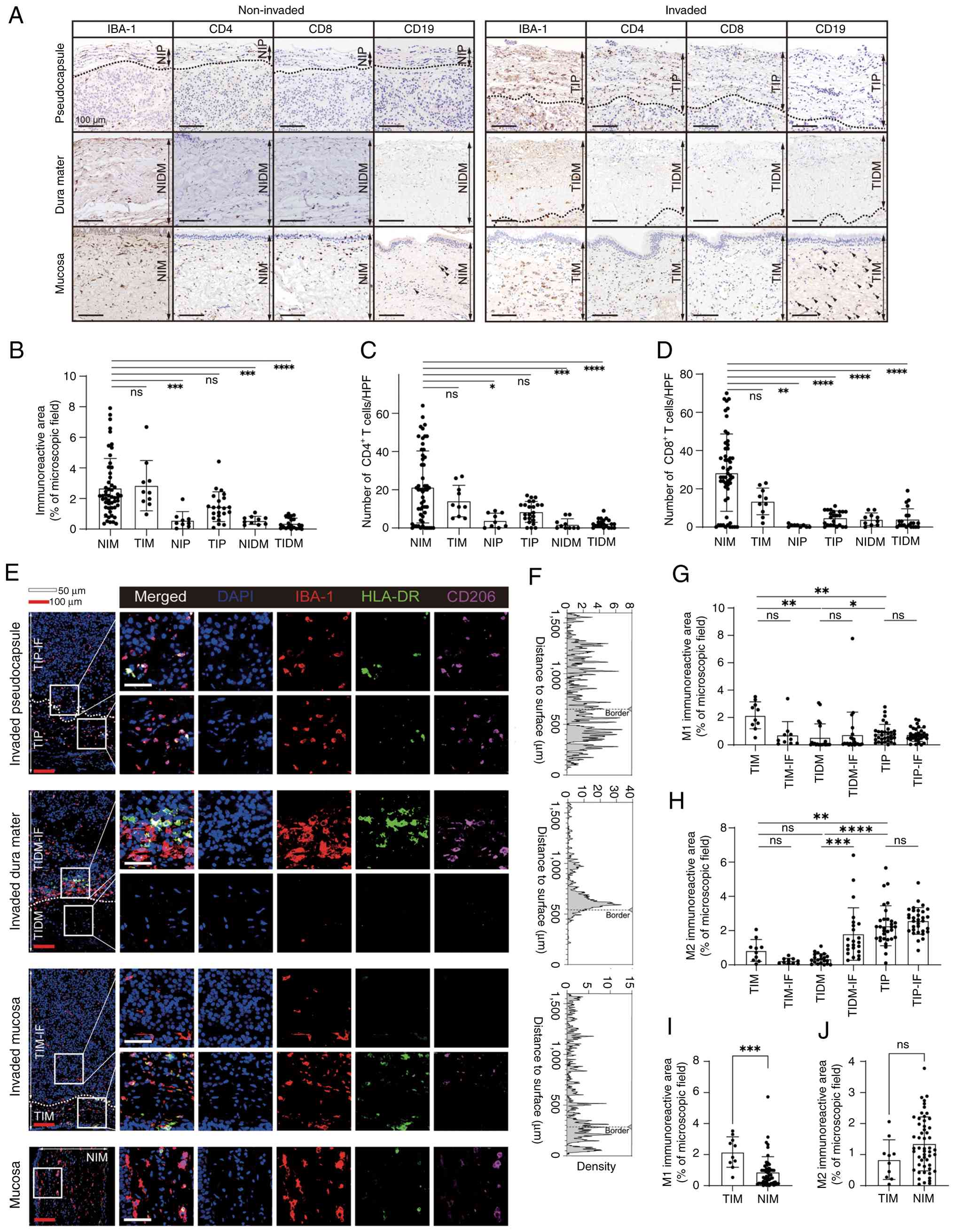 | Figure 3.Spatial heterogeneity of immune cells
in the tumor microenvironment. Immunophenotypic profiling across
invasion states: Non-invaded pseudocapsule (n=9; pathologically
tumor-free), tumor-invaded pseudocapsule (n=23), control dura mater
from non-invasive tumor cases (n=10), tumor-invaded dura mater
(n=21), tumor-invaded mucosa (n=10) and non-invaded mucosa (n=53).
(A) Representative immunohistochemistry images for the detection of
macrophages (IBA-1+), CD4+ T cells,
CD8+ T cells and CD19+ B cells. Arrows
indicate CD19-positive cells. (B) Quantification of macrophage
burden (IBA-1+ immunoreactive area; %). (C)
Quantification of CD4+ T-cell density (cells per HPF).
(D) Quantification of CD8+ T-cell density (cells per
HPF). (E-J) Spatial heterogeneity of macrophage phenotypes. (E)
Multiplex immunofluorescence images showing IBA-1+
(red), HLA-DR+ (green; M1-like) and CD206+
(magenta; M2-like) macrophage distributions at the IF of the
pseudocapsule, dura mater and mucosa, and in non-invaded mucosa.
(F) Grayscale-intensity distributions for IBA-1 quantified using
ImageJ. (G) M1 immunoreactive area (% of microscopic field) in each
group (TIM, TIM-IF, TIDM, TIDM-IF, TIP and TIP-IF). (H) M2
immunoreactive area (% of microscopic field) in each group (TIM,
TIM-IF, TIDM, TIDM-IF, TIP and TIP-IF). (I) M1 immunoreactive area
(% of microscopic field) in the TIM and NIM groups. (J) M2
immunoreactive area (% of microscopic field) in the TIM and NIM
groups. (B-D) Kruskal-Wallis test with prespecified Dunn's post hoc
planned comparisons (NIM vs. TIM/NIP/TIP/NIDM/TIDM) and Bonferroni
correction. (G and H) Unpaired comparisons among TIM, TIDM and TIP,
and separately among TIM-IF, TIDM-IF and TIP-IF, were performed
using the Kruskal-Wallis test followed by Dunn's
multiple-comparisons test, whereas paired comparisons between each
tumor region and its matched IF region were performed using the
two-tailed Wilcoxon signed-rank test. Bonferroni correction was
applied across all nine comparisons performed in this analysis. (I
and J) Unpaired comparisons were analyzed using a two-tailed
Mann-Whitney U test. *P<0.05, **P<0.01, ***P<0.001,
****P<0.0001. HPF, high-power field; IBA-1, ionised calcium
binding adaptor molecule 1; IF, invasive front; NIDM, non-invaded
dura mater; NIM, non-invaded mucosa; NIP, non-invaded
pseudocapsule; ns, not significant; TIDM, tumor-invaded dura mater;
TIM, tumor-invaded mucosa; TIP, tumor-invaded pseudocapsule. |
To resolve macrophage heterogeneity, multiplex
immunofluorescence staining was used to phenotype IBA-1+
macrophages into HLA-DR+ (M1-like; pro-inflammatory) and
CD206+ (M2-like; immunoregulatory) states (26). Macrophages were enriched at the IF
of pseudocapsular and dural tissues (Fig. 3E and F). In mucosal tissues, the
proportion of HLA-DR+ (M1-like) macrophages was higher
in tumor-invaded mucosa than in pseudocapsule/dura tissues, whereas
non-invaded mucosa remained M2-skewed (Figs. 3G-J and S3B-D). Collectively, these data
indicated context-dependent remodeling of mucosal macrophage states
adjacent to invasion and were consistent with a microenvironment
associated with constraints on local tumor expansion.
IFN-γ suppresses tumor invasion and
growth
Cytokine dynamics were profiled using ELISAs in two
co-culture systems. ELISA revealed that IFN-γ, IL-6 and IL-10 were
increased in both the MTC and DMC group compared with the CTRL
group, with higher IFN-γ levels in the DMC group (Fig. 4A). By contrast, TGF-β
concentrations were reduced in both the MTC and DMC groups relative
to the CTRL group (Fig. 4A).
Spatial analyses demonstrated enrichment of TNF-α and IL-1β at the
tumor-mucosa interface, consistent with a pro-inflammatory gradient
in regions of invasion (Fig.
S4A-D). Immunofluorescence staining indicated that the IFN-γ
signal was enriched in mucosal macrophages and exceeded that in
intratumoral macrophages, which was in line with the correlation
between macrophage density and IFN-γ intensity (Figs. 4B and S4E). IFN-γ expression in SSIT mucosal
macrophages also exceeded that in non-invaded mucosa (Fig. 4C and D).
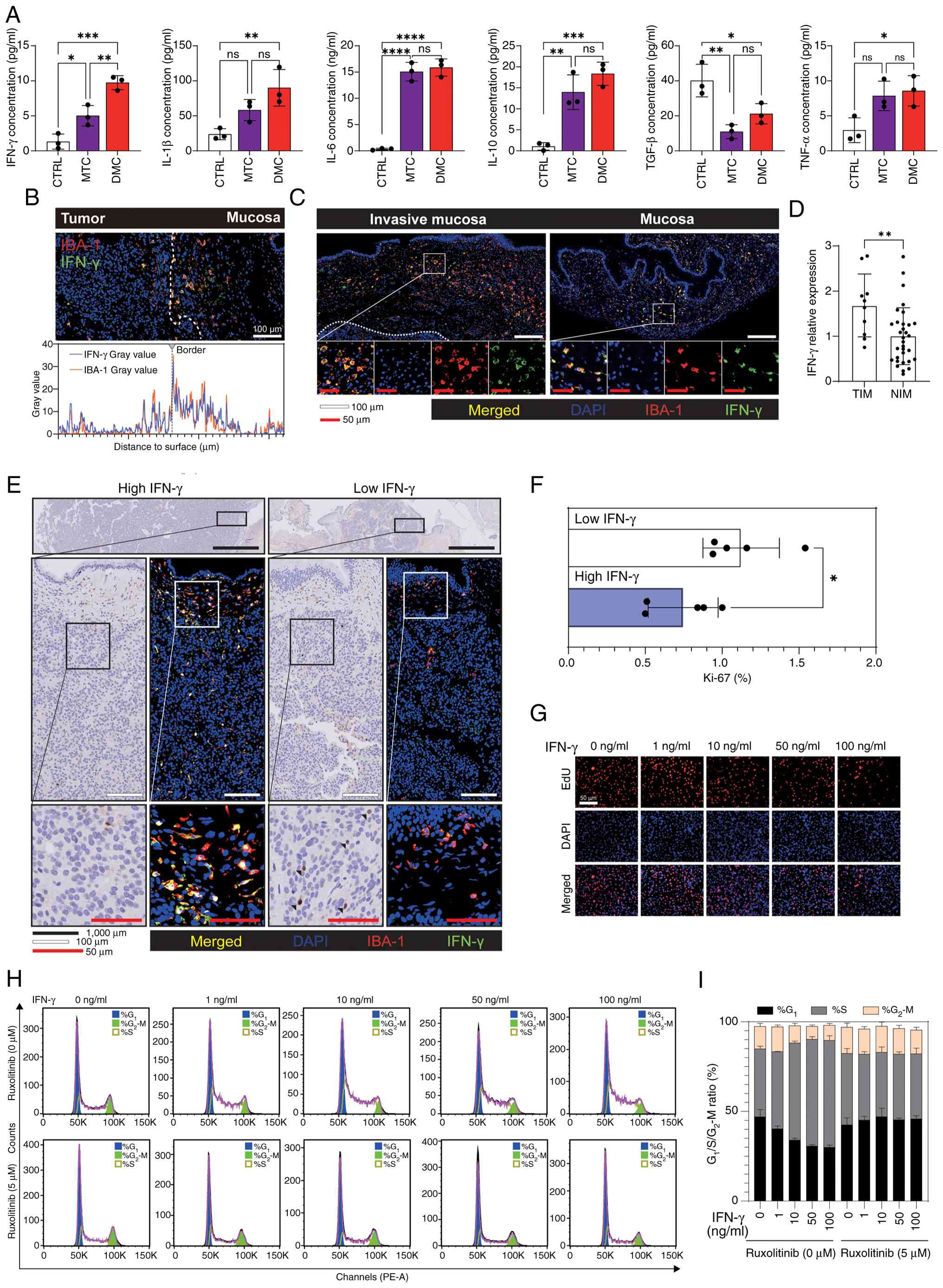 | Figure 4.IFN-γ suppresses tumor growth and
invasion. (A) Cytokine profiling of co-culture supernatants via
ELISAs: IFN-γ, IL-1β, IL-6, IL-10, TGF-β and TNF-α. (B-D) Spatial
expression patterns of IFN-γ. (B) Immunofluorescence imaging of the
invasive front in SSIT, showing DAPI (blue), IBA-1+
macrophages (red) and IFN-γ+ signals (green), and
grayscale intensity distribution. (C) Immunofluorescence imaging of
TIM and NIM, showing DAPI (blue), IBA-1+ macrophages
(red) and IFN-γ+ signals (green). (D) Quantification of
relative IFN-γ expression in TIM and NIM. (E) Representative Ki-67
immunohistochemistry images of SSIT cases stratified into
IFN-γ-high and IFN-γ-low groups (n=5 each; median split). (F)
Quantification of Ki-67 index comparing the two groups. (G) EdU
staining demonstrating dose-dependent suppression of TtT/GF
pituitary adenoma cell proliferation by IFN-γ (0–100 ng/ml; 48 h).
(H) Representative flow cytometry histograms for cell cycle
analysis of cells treated with IFN-γ (0–100 ng/ml) in the absence
(0 µM) or presence (5 µM) of ruxolitinib. (I) Stacked bar plot
showing the percentages of cells in the G1, S and
G2/M phases under the same treatment conditions. (A)
One-way ANOVA with Tukey's post hoc multiple comparisons test. (D
and F) Unpaired two-tailed Student's t-test. *P<0.05,
**P<0.01, ***P<0.001, ****P<0.0001. CTRL, control; DMC,
digested mucosal culture; EdU, 5-ethynyl-2′-deoxyuridine; IBA-1,
ionised calcium binding adaptor molecule 1; MTC, mucosal tissue
culture; NIM, non-invaded mucosa; ns, not significant; PE-A,
phycoerythrin-area; SSIT, sphenoid sinus-invasive tumor; TIM,
tumor-invaded mucosa. |
Guided by prior evidence linking IFN-γ to antitumor
activity via direct anti-proliferative effects and
microenvironmental remodeling (27,28),
SSIT cases were stratified into high- and low-IFN-γ groups based on
the median IFN-γ expression. Ki-67 IHC showed significantly lower
proliferation indices in the high-IFN-γ cohort (Fig. 4E and F). Mechanistic investigation
using the TtT/GF PA cell line revealed dose-dependent suppression
of proliferation by exogenous IFN-γ (0–100 ng/ml), as indicated by
reduced EdU incorporation (Figs.
4G and S4F). Wound healing
assays further demonstrated IFN-γ-mediated inhibition of migration
(Fig. S4G and H). Flow cytometry
cell cycle analysis indicated S-phase arrest following IFN-γ
treatment, which was reversed by the Janus kinase (JAK) inhibitor
ruxolitinib (5 µM) (Figs. 4H and
I, and S4I-K).
Semi-quantitative analysis of the western blot data confirmed that
ruxolitinib (5 µM) significantly reduced IFN-γ-induced STAT1
phosphorylation at concentrations of 0–50 ng/ml, whereas no
significant inhibition was observed at 100 ng/ml (Fig. S4L and M). Taken together, these
findings support a model in which mucosal macrophages constitute a
predominant source of IFN-γ, and where IFN-γ constrains PA growth
and invasion through JAK-STAT1 activation.
Elevated IgG levels drive M2-to-M1
macrophage polarization to suppress tumor growth
Tertiary lymphoid structures were observed in
selected mucosal regions (Fig.
S5A) and have been associated with localized antitumor immunity
(29). To investigate mucosal B
cell involvement (30,31), SSIT cases were stratified into
high- and low-CD19+ groups based on the median CD19
expression. Sequential immunofluorescence staining of serial
sections for CD19 and macrophage markers revealed reduced M2
proportions and increased M1 polarization in high-CD19+
regions (Fig. 5A and B). IHC
revealed strong IgG signal in B cells with undetectable IgA
(Fig. S5B). Notably, IgG-high
mucosal tissues exhibited significantly greater M1 macrophage
proportions than IgG-low counterparts (Fig. 5C and D).
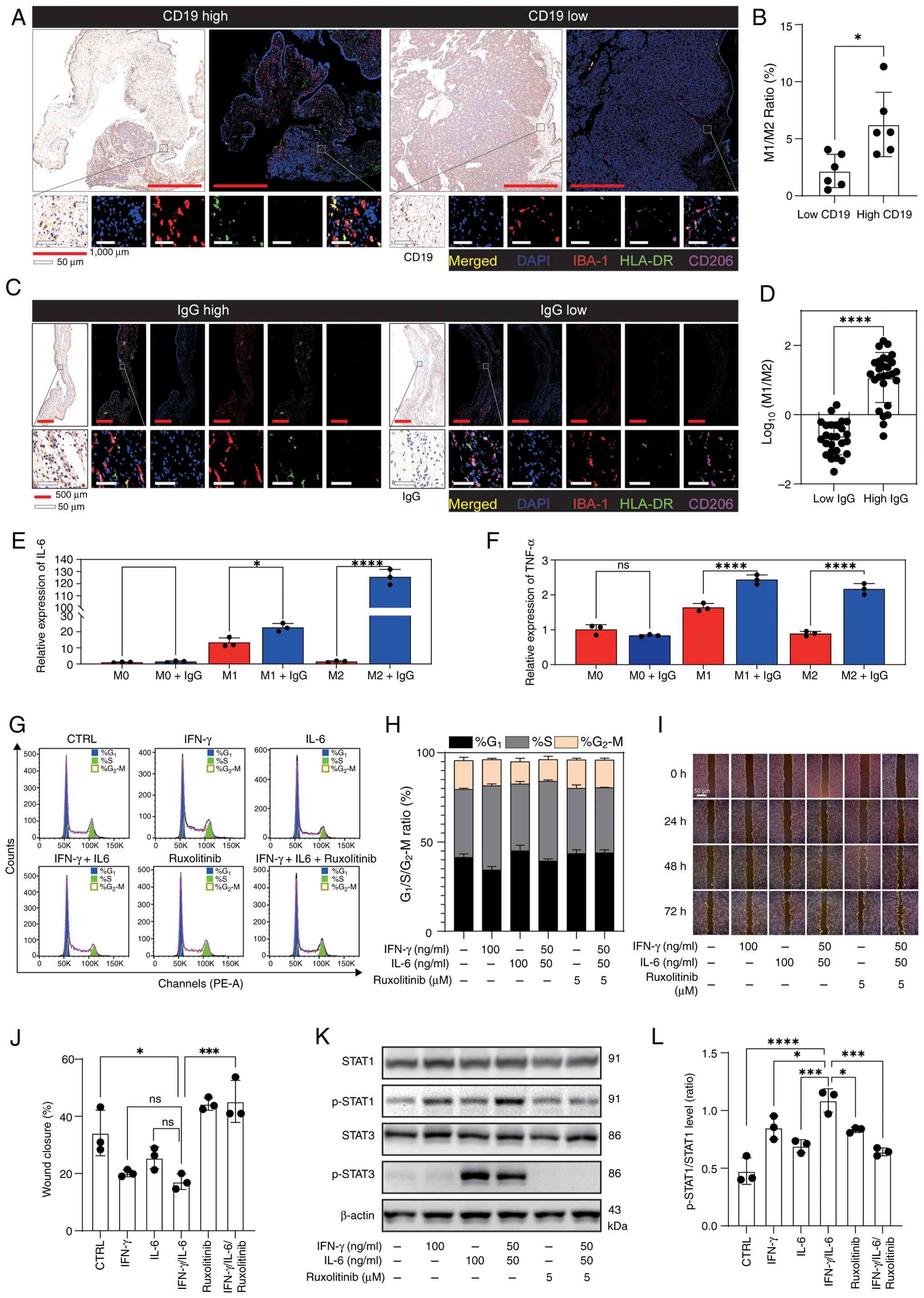 | Figure 5.Elevated IgG levels drive macrophage
M2-to-M1 reprogramming. (A) Sphenoid sinus-invasive tumor cases
stratified into CD19-high (n=5) and CD19-low (n=5) groups based on
the cohort median of CD19+ B cell density, with (B)
quantitative analyses of macrophage polarization (M1-like versus
M2-like). (C) Dural-invasive tumor and non-invasive tumor cases
stratified into IgG-high (n=27) and IgG-low (n=26) groups based on
the cohort median of relative IgG immunohistochemistry staining
intensity, with (D) quantitative analyses of M1-like/M2-like
macrophage proportions. (E and F) RAW264.7 macrophages were
pre-polarized with IL-4 (20 ng/ml) or with lipopolysaccharide (100
ng/ml) plus IFN-γ (20 ng/ml) for 24 h, followed by IgG (10 µg/ml)
exposure. Relative (E) IL-6 and (F) TNF-α mRNA expression in
RAW264.7 macrophages pre-polarized to M0, M1 or M2 states. (G)
Representative flow cytometric cell-cycle profiles of TtT/GF cells
following the indicated treatments. (H) Stacked bar plot
summarizing the percentages of cells from (G) in G1, S
and G2/M phases. (I) Representative images from the
scratch wound assay at 0, 24, 48 and 72 h under the indicated
treatments. (J) Quantification of scratch wound closure. (K)
Representative western blot images showing total STAT1, p-STAT1,
total STAT3, p-STAT3 and β-actin levels in cells treated with IFN-γ
(100 ng/ml), IL-6 (100 ng/ml), IFN-γ + IL-6 (50 ng/ml each),
ruxolitinib (5 µM) or IFN-γ + IL-6 (50 ng/ml each) plus ruxolitinib
(5 µM), as indicated. (L) Densitometric semi-quantification of
p-STAT1/STAT1 (ratio). (B and D) Unpaired two-tailed Student's
t-test. (E, F, J and L) One-way ANOVA with Tukey's post hoc
multiple comparisons test. *P<0.05, ***P<0.001,
****P<0.0001. CTRL, control; IBA-1, ionised calcium binding
adaptor molecule 1; ns, not significant; p-, phosphorylated; PE-A,
phycoerythrin-area. |
To probe the mechanistic contribution of IgG,
RAW264.7 macrophages polarized with IL-4 (20 ng/ml) or LPS (100
ng/ml) plus IFN-γ (20 ng/ml) were treated with IgG (10 µg/ml) for
12 h. qPCR analysis showed that IgG treatment increased IL-4
expression and reduced IL-18 expression in M0 macrophages, induced
upregulation of IL-6, IL-27, TNF-α and IL-10 with concomitant
downregulation of IL-18 in M1 macrophages, and increased IL-6,
IL-27 and TNF-α while decreasing IL-4 in M2 macrophages, with IL-18
and IL-10 remaining unchanged in the latter (Figs. 5E and F, and S5C).
Consistently, multiplex immunofluorescence staining
detected higher IL-6 expression in the high-CD19+ group
compared with the low-CD19+ group (Fig. S5D and E). Given previous evidence
indicating that high concentrations of IL-6 can suppress PA
progression (32), dose-response
assays (0–100 ng/ml) were performed in TtT/GF cells. Flow cytometry
cell cycle analysis indicated IL-6-induced G1-phase
arrest (Fig. S5F-J), in contrast
to IFN-γ-mediated S-phase arrest (Fig.
4H-I). Combined treatment with IFN-γ (50 ng/ml) and IL-6 (50
ng/ml) altered the cell-cycle phase distribution (Figs. 5G and H, and S5K-M), and reduced migration (scratch
wound closure) to a level comparable to the level observed after
IFN-γ treatment alone (100 ng/ml) or IL-6 treatment alone (100
ng/ml) (Fig. 5I and J). Western
blotting indicated enhanced phosphorylation of STAT1
(phosphorylated-STAT1/STAT1 ratio), whereas STAT3 phosphorylation
was attenuated relative to IL-6 monotherapy. This effect was
abrogated by the JAK inhibitor ruxolitinib (5 µM) (Figs. 5K and L, and S5N). Taken together, these data support
a model in which elevated mucosal IgG levels reprogram macrophages
toward an M1-dominant phenotype and, together with IFN-γ,
cooperatively amplify JAK-STAT1 signaling to constrain PA cell
proliferation and migration.
Anti-CD47 mAb enhances
antibody-dependent cellular phagocytosis (ADCP) to suppress tumor
cell proliferation
Immune checkpoint blockade targeting programmed cell
death protein-1, programmed death-ligand-1, CD47 or signal
regulatory protein-α (SIRPα) has improved outcomes in preclinical
murine tumor models (33,34). During progression, tumor cells can
evade macrophage phagocytosis via upregulation of CD47, a key
component of the ‘don't-eat-me’ axis with SIRPα. Immunofluorescence
staining detected pronounced CD47+ signal at the tumor
IF (Fig. 6A and B), supporting the
rationale for anti-CD47 intervention.
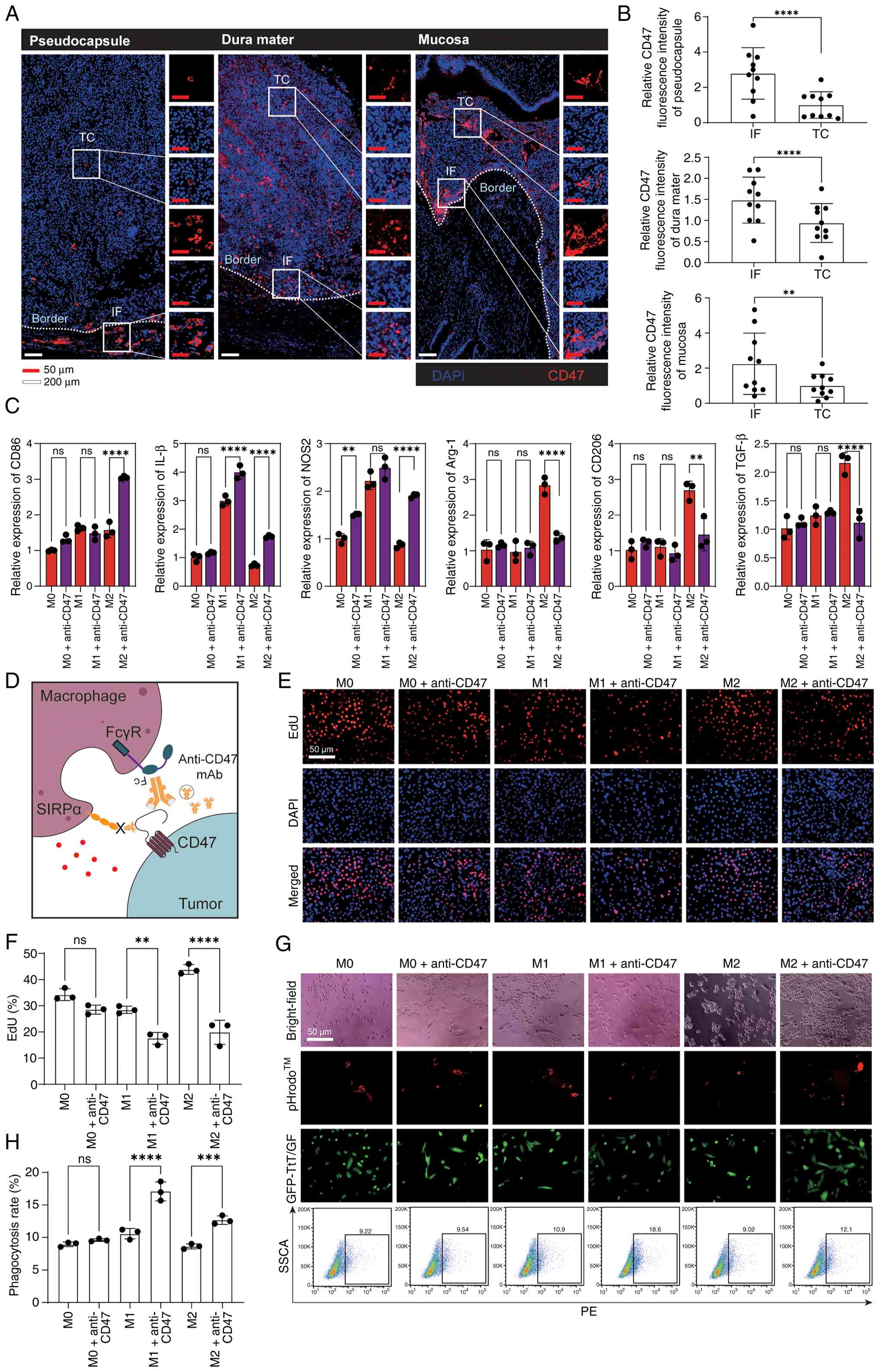 | Figure 6.Anti-CD47 mAb enhances ADCP to
suppress tumor cell proliferation. (A) Immunofluorescence staining
of CD47 (red) and DAPI (blue) in a representative subset of
non-invasive tumor, dural-invasive tumor and sphenoid
sinus-invasive tumor cases (n=10 per group). (B) Paired comparison
of CD47 fluorescence intensity at the IF versus the TC. (C)
RAW264.7 macrophages were pre-polarized with IL-4 (20 ng/ml) or
with lipopolysaccharide (100 ng/ml) plus IFN-γ (20 ng/ml) for 24 h,
followed by anti-CD47 mAb (10 µg/ml) treatment for 12 h.
Quantitative PCR was used to analyze polarization/activation
markers. (D) Schematic illustrating anti-CD47 mAb-mediated blockade
of the CD47-SIRPα axis and enhancement of ADCP. (E) EdU assay of
TtT/GF cell proliferation in a Transwell co-culture with anti-CD47
mAb-treated polarized macrophages. (F) Quantification of
EdU-positive cells. (G) Representative microscopy images and flow
cytometry plots showing macrophage phagocytosis of pHrodo™
Red-labeled GFP-TtT/GF cells. (H) Quantification of phagocytosis
(%). (B) Paired two-tailed Student's t-test. (C, F and H) One-way
ANOVA with Tukey's post hoc multiple comparisons test. **P<0.01,
***P<0.001, ****P<0.0001. ADCP, antibody-dependent cellular
phagocytosis; Arg-1, arginase 1; EdU, 5-ethynyl-2′-deoxyuridine;
FcγR, Fcγ receptor; GFP, green fluorescent protein; IF, invasive
front; mAb, monoclonal antibody; NOS2, nitric oxide synthase 2; ns,
not significant; PE, phycoerythrin; SIRPα, signal regulatory
protein-α; SSCA, side scatter area; TC, tumor core. |
To evaluate the therapeutic potential, M1- and
M2-polarized macrophages were exposed to anti-CD47 mAb (10 µg/ml)
for 12 h. Anti-CD47 mAb treatment elicited transcriptional changes
in M2 macrophages, marked by increased CD86, IL-1β and NOS2
expression together with reduced Arg-1, CD206 and TGF-β levels
(Fig. 6C). By comparison,
anti-CD47 mAb treatment induced only modest effects in M0 (NOS2
upregulation) and M1 macrophages (IL-1β upregulation) (Fig. 6C). Previous studies have
demonstrated that anti-CD47 mAb augments macrophage phagocytosis
and immune activation (35,36)
(Fig. 6D). In macrophage-TtT/GF
co-culture assays, anti-CD47 mAb treatment did not significantly
affect tumor cell proliferation or phagocytic activity in the
presence of M0 macrophages (Fig. 6F
and H). By contrast, anti-CD47 mAb treatment in M1- and
M2-polarized macrophages significantly reduced tumor cell
proliferation, as determined by EdU incorporation, and
significantly increased ADCP compared with isotype controls
(Fig. 6F and H).
Collectively, these findings are summarized in the
working model shown in Fig. 7.
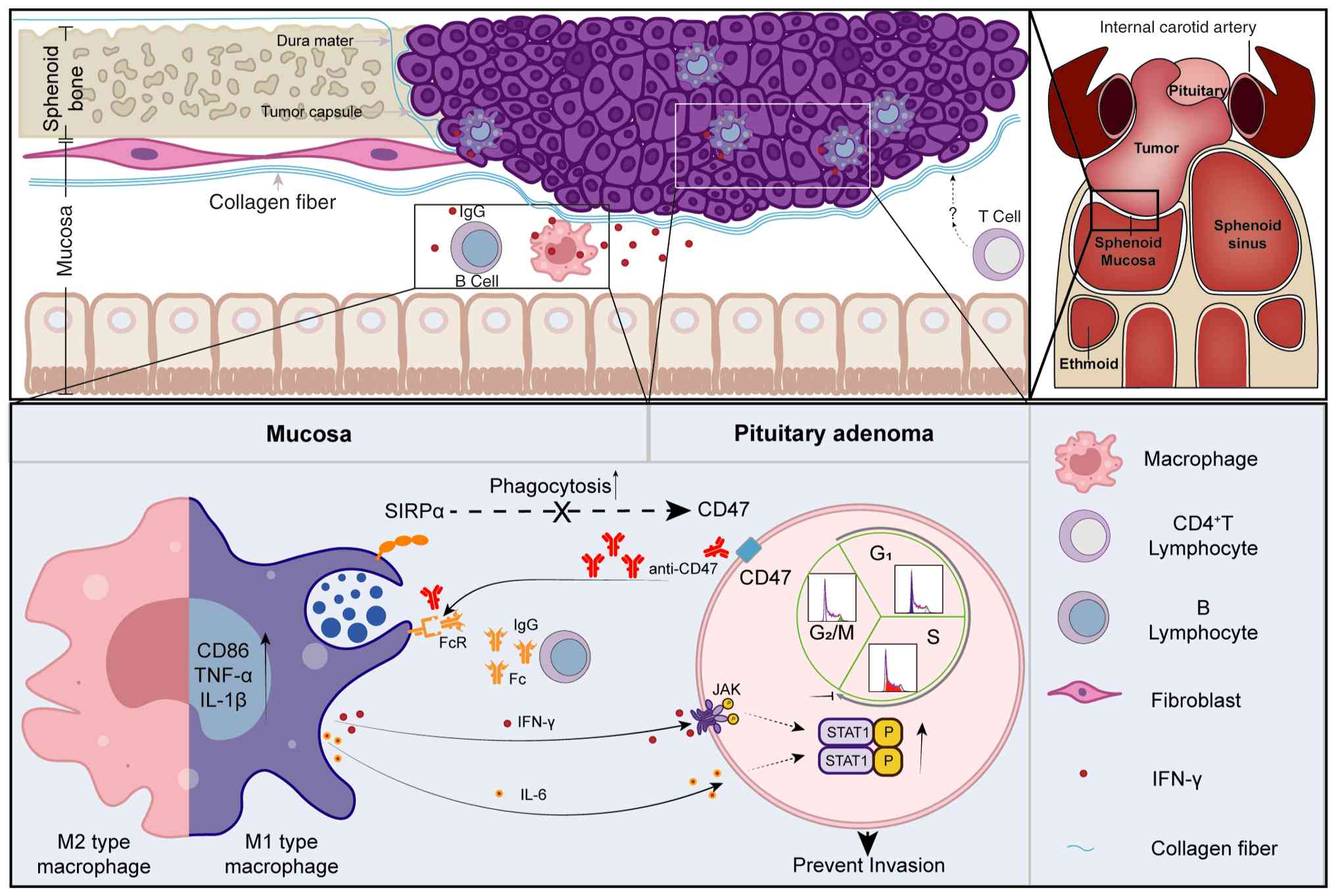 | Figure 7.Summary graphic illustration. This
illustration summarizes the proposed model during pituitary adenoma
invasion. The tumor invasive front abuts an intact sphenoid sinus
mucosa, forming a distinct boundary. The mucosal compartment is
enriched for ionised calcium binding adaptor molecule 1-positive
macrophages with an M1-like predominance and IgG-high B cells. B
cell-derived IgG promotes M2-to-M1 macrophage reprogramming, while
coordinated IFN-γ and IL-6 production establishes a
tumor-suppressive cytokine gradient that decreases from mucosa
toward the tumor core, constraining proliferation and migration via
JAK-STAT1 activation. Therapeutically, anti-CD47 monoclonal
antibody blocks the CD47-SIRPα ‘don't-eat-me’ axis and augments
antibody-dependent cellular phagocytosis, highlighting a strategy
for immune checkpoint-targeted therapy that may complement surgical
management. FcR, Fc receptor; JAK, Janus kinase; mAb, monoclonal
antibody; p-, phosphorylated; SIRPα, signal regulatory
protein-α. |
Discussion
Persistent hormonal hypersecretion and invasion into
adjacent critical structures remain major challenges in the
management of Pas (37,38). Infiltrative growth into the
cavernous sinus, dura mater and bone confers a substantial risk of
recurrence (4). Unlike
well-characterized IFs in hepatocellular or other solid tumors
(39), the tumor-host interface in
PAs remains underexplored. Intraoperative and histopathological
observations in the present cohort indicated that the sphenoid
sinus mucosa could maintain structural integrity even when tumors
breached the pseudocapsule and dura, suggesting a potential barrier
function analogous to that noted in other tissues (22,40).
Immunohistochemical profiling revealed heterogeneous
immune infiltration at the tumor-mucosa interface, with macrophages
(IBA-1+) constituting the predominant population. Prior
studies in colorectal and ovarian cancer have shown that a higher
ratio of pro-inflammatory (M1) to immunoregulatory (M2) macrophages
at the tumor-stromal interface is associated with a favorable
prognosis (41–43). In the present study, multiplex
immunofluorescence staining demonstrated an increased M1 proportion
in tumor-invaded mucosa relative to both TCs and non-invaded
mucosa. In parallel, cytokine analyses delineated a
pro-inflammatory gradient at the IF, including increased IFN-γ
expression in mucosal macrophages. Given the established role of
IFN-γ in constraining tumorigenesis through STAT1 activation
(27), functional assays in PA
models corroborated JAK-STAT1-dependent anti-proliferative and
anti-migratory effects.
In addition to macrophage-mediated immune
regulation, the mucosal niche also contained abundant B cells.
Accumulating evidence indicates that B cells can mediate antitumor
functions within the tumor microenvironment, and that B
cell-enriched tertiary lymphoid structures are associated with
durable antitumor immunity and favorable clinical outcomes
(44,45). In the present study, the mucosal
niche also contained abundant B cells, and tertiary lymphoid
structures were observed in discrete areas. Within this context,
IgG emerged as a candidate immunomodulator. Tissue-level analyses
showed an association between IgG-high mucosa and increased M1
macrophage proportions, and cell-based experiments showed that IgG
exposure increased macrophage IL-6 expression and was accompanied
by enhanced IFN-γ-dependent STAT1 signaling. As PA invasion entails
degradation of membranous and stromal barriers (46), strategies that preserve mucosal
architecture, potentially by leveraging B cell-IgG axes, merit
systematic evaluation.
Therapeutically, the CD47-SIRPα checkpoint
represents a rational target in settings where tumor cells
upregulate CD47 levels at the IF. In the present study, anti-CD47
mAb treatment increased activation markers in M2-polarized
macrophages, including CD86 and NOS2, reduced PA cell proliferation
under co-culture conditions, and enhanced ADCP, in line with prior
reports (33,47). Nonetheless, clinical translation is
complicated by on-target effects on erythrocytes and attendant
anemia (48).
Tissue engineering concepts may offer complementary
avenues: Prior work showing growth restraint of MCF7 ×enografts by
adipose tissue grafts raises the broader hypothesis that
perioperative preservation or augmentation of protective mucosal
elements, potentially combined with macrophage-directed
immunotherapy, could be explored alongside transsphenoidal surgery
(49).
The focus of the present study on polarized
macrophages overlooks subsets lacking classical M1/M2 markers or
exhibiting mixed phenotypes, as reported in other systems (42,43).
The roles of T lymphocytes, neutrophils and intercellular crosstalk
remain unaddressed. Future studies should isolate fresh mucosal
immune cells for single-cell transcriptomic profiling and spatial
transcriptomics to elucidate their plasticity within the IF of the
tumor microenvironment (50).
Beyond its macrophage-modulating effects, it should be emphasized
that IgG exhibits divergent modulatory effects on tumor cell
proliferation (30,51), an aspect not systematically
addressed in the present study. Constrained by limitations in the
in vitro passaging of tumor cells and the proliferative
dynamics of mucosal tissue, the present study focused on analyzing
the inhibitory effect of the mucosa on tumor growth. Future studies
will employ functional assays, such as Matrigel invasion assays of
primary tumor cells under organoid culture conditions and mucosal
explant models, to determine whether the mucosa actively restricts
tumor invasion.
In conclusion, the intricate dynamics between
sphenoid sinus mucosa and invasive PAs, as revealed in the present
study, provide novel insights into tumor-host interactions. Far
from a passive barrier, the mucosa orchestrates an active defense
system, recruiting macrophages, B cells and cytokines to counteract
tumor-driven structural disruption. Understanding and leveraging
this natural defense strategy may inspire novel approaches to
inhibit tumor invasion.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was funded by the National Natural Science
Foundation of China (grant nos. 82173136 and 82203683).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
YH and TL conceived the study. XL, ZL and LX
contributed to experimental design and execution of experimental
procedures. XL, ZL, ZW, QW, QJ, LX and TL performed experiments and
acquired data. XL performed formal analysis. XL, ZL and SL
collected, organized and verified the clinical and experimental
data, and ensured the accuracy and completeness of the datasets
used for analysis, and SL contributed to data interpretation and
figure preparation. XL and ZL were involved in validation. YH, ZL
and SL were involved in visualization. XL and LX wrote the original
manuscript. TL, SL and HZ and YH reviewed and edited the
manuscript. TL and HZ provided resources, and HZ contributed to
interpretation of the clinical/pathological data and critically
revised the manuscript for important intellectual content. YH and
TL supervised the study. TL, XL and YH were involved in project
administration. TL acquired funding. XL and YH confirm the
authenticity of all the raw data. All authors have read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Medical Ethics
Committee of Tongji Hospital (Wuhan, China; approval no.
TJ-IRB20220325) and conducted in accordance with The Declaration of
Helsinki. All participants provided written informed consent before
participation.
Patient consent for publication
All patients provided written informed consent for
publication of their anonymized data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Melmed S: Pituitary-tumor
endocrinopathies. N Engl J Med. 382:937–950. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Thompson LD, Seethala RR and Müller S:
Ectopic sphenoid sinus pituitary adenoma (ESSPA) with normal
anterior pituitary gland: A clinicopathologic and immunophenotypic
study of 32 cases with a comprehensive review of the english
literature. Head Neck Pathol. 6:75–100. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lu L, Wan X, Xu Y, Chen J, Shu K and Lei
T: Classifying pituitary adenoma invasiveness based on
radiological, surgical and histological features: A retrospective
assessment of 903 cases. J Clin Med. 11:24642022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Buchfelder M and Schlaffer SM: Surgical
treatment of aggressive pituitary adenomas and pituitary
carcinomas. Rev Endocr Metab Disord. 21:253–261. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang J, Voellger B, Benzel J, Schlomann U,
Nimsky C, Bartsch JW and Carl B: Metalloproteinases ADAM12 and
MMP-14 are associated with cavernous sinus invasion in pituitary
adenomas. Int J Cancer. 139:1327–1339. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wan X, Yan Z, Tan Z, Cai Z, Qi Y, Lu L, Xu
Y, Chen J and Lei T: MicroRNAs in dopamine agonist-resistant
prolactinoma. Neuroendocrinology. 112:417–426. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang Q, Yao B, Long X, Chen Z, He M, Wu
Y, Qiao N, Ma Z, Ye Z, Zhang Y, et al: Single-cell sequencing
identifies differentiation—related markers for molecular
classification and recurrence prediction of PitNET. Cell Rep Med.
4:1009342023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang F, Zhang Q, Zhu J, Yao B, Ma C, Qiao
N, He S, Ye Z, Wang Y, Han R, et al: Integrated proteogenomic
characterization across major histological types of pituitary
neuroendocrine tumors. Cell Res. 32:1047–1067. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Viola MF and Boeckxstaens G:
Niche-specific functional heterogeneity of intestinal resident
macrophages. Gut. 70:1383–1395. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ruder B and Becker C: At the forefront of
the mucosal barrier: The role of macrophages in the intestine.
Cells. 9:21622020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rugtveit J, Nilsen EM, Bakka A, Carlsen H,
Brandtzaeg P and Scott H: Cytokine profiles differ in newly
recruited and resident subsets of mucosal macrophages from
inflammatory bowel disease. Gastroenterology. 112:1493–1505. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vaphiades MS: Pituitary ring sign plus
sphenoid sinus mucosal thickening: Neuroimaging signs of pituitary
apoplexy. Neuroophthalmology. 41:306–309. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Schteingart DE, Chandler WF, Lloyd RV and
Ibarra-Perez G: Cushing's syndrome caused by an ectopic pituitary
adenoma. Neurosurgery. 21:223–227. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lopes MBS: The 2017 world health
organization classification of tumors of the pituitary gland: a
summary. Acta Neuropathol. 134:521–535. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Araujo-Castro M, Cancela AA, Vior C,
Pascual-Corrales E and Rodriguez Berrocal V: Radiological knosp,
revised-knosp, and hardy-wilson classifications for the prediction
of surgical outcomes in the endoscopic endonasal surgery of
pituitary adenomas: Study of 228 cases. Front Oncol. 11:8070402021.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cooper O, Bonert V, Mamelak AN, Bannykh S
and Melmed S: Dural invasion as a marker of aggressive pituitary
adenomas. Neurosurgery. 90:775–783. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Asa SL, Mete O, Perry A and Osamura RY:
Overview of the 2022 WHO classification of pituitary tumors. Endocr
Pathol. 33:6–26. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ye J, Coulouris G, Zaretskaya I,
Cutcutache I, Rozen S and Madden TL: Primer-BLAST: A tool to design
target-specific primers for polymerase chain reaction. BMC
Bioinformatics. 13:1342012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xu C, Wu H, Liu Y, Li F, Manne RK and Lin
HK: Protocol for detecting macrophage-mediated cancer cell
phagocytosis in vitro and in vivo. STAR Protoc. 4:1019402023.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nakamura K, Tsukasaki M, Tsunematsu T, Yan
M, Ando Y, Huynh NC, Hashimoto K, Gou Q, Muro R, Itabashi A, et al:
The periosteum provides a stromal defence against cancer invasion
into the bone. Nature. 634:474–481. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Conway JRW, Dinç DD, Follain G,
Paavolainen O, Kaivola J, Boström P, Hartiala P, Peuhu E and Ivaska
J: IGFBP2 secretion by mammary adipocytes limits breast cancer
invasion. Sci Adv. 9:eadg18402023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li H, Zheng Y, Han YL, Cai S and Guo M:
Nonlinear elasticity of biological basement membrane revealed by
rapid inflation and deflation. Proc Natl Acad Sci USA.
118:e20224221182021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Dongre A and Weinberg RA: New insights
into the mechanisms of epithelial-mesenchymal transition and
implications for cancer. Nat Rev Mol Cell Biol. 20:69–84. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chen CL, Wang YT, Yao Y, Pan L, Guo B, Zhu
KZ, Ma J, Wang N, Li XL, Deng YK and Liu Z: Inflammatory endotypes
and tissue remodeling features in antrochoanal polyps. Allergy
Asthma Immunol Res. 13:863–881. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Schoenfeld DA, Moutafi M, Martinez S,
Djureinovic D, Merkin RD, Adeniran A, Braun DA, Signoretti S,
Choueiri TK, Parisi F, et al: Immune dysfunction revealed by
digital spatial profiling of immuno-oncology markers in progressive
stages of renal cell carcinoma and in brain metastases. J
Immunother Cancer. 11:e0072402023. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ding H, Wang G, Yu Z, Sun H and Wang L:
Role of interferon-gamma (IFN-gamma) and IFN-gamma receptor 1/2
(IFNgammaR1/2) in regulation of immunity, infection, and cancer
development: IFN-gamma-dependent or independent pathway. Biomed
Pharmacother. 155:1136832022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lyu L, Jiang Y, Ma W, Li H, Liu X, Li L,
Shen A, Yu Y, Jiang S, Li H, et al: Single-cell sequencing of
PIT1-positive pituitary adenoma highlights the pro-tumour
microenvironment mediated by IFN-γ-induced tumour-associated
fibroblasts remodelling. Br J Cancer. 128:1117–1133. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
MacFawn IP, Magnon G, Gorecki G, Kunning
S, Rashid R, Kaiza ME, Atiya H, Ruffin AT, Taylor S, Soong TR, et
al: The activity of tertiary lymphoid structures in high grade
serous ovarian cancer is governed by site, stroma, and cellular
interactions. Cancer Cell. 42:1864–1881.e1865. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang Y, Liu G, Zeng Q, Wu W, Lei K, Zhang
C, Tang M, Zhang Y, Xiang X, Tan L, et al: CCL19-producing
fibroblasts promote tertiary lymphoid structure formation enhancing
anti-tumor IgG response in colorectal cancer liver metastasis.
Cancer Cell. 42:1370–1385.e1379. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mester S, Chan C, Lustig M, Foss S, Jansen
JHM, Herigstad M, Evers M, Nilsen J, Reiding KR, Damen JM, et al:
Engineering of anticancer human immunoglobulin A equipped with
albumin for enhanced plasma half-life. PNAS Nexus. 4:pgaf0422025.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Borg SA, Kerry KE, Baxter L, Royds JA and
Jones TH: Expression of interleukin-6 and its effects on growth of
HP75 human pituitary tumor cells. J Clin Endocrinol Metab.
88:4938–4944. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nie W, Wu G, Zhang J, Huang LL, Ding J,
Jiang A, Zhang Y, Liu Y, Li J, Pu K and Xie HY: Responsive exosome
nano-bioconjugates for synergistic cancer therapy. Angew Chem Int
Ed Engl. 59:2018–2022. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Xia Y, Rao L, Yao H, Wang Z, Ning P and
Chen X: Engineering macrophages for cancer immunotherapy and drug
delivery. Adv Mater. 32:e20020542020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Osorio JC, Smith P, Knorr DA and Ravetch
JV: The antitumor activities of anti-CD47 antibodies require
Fc-FcgammaR interactions. Cancer Cell. 41:2051–2065. e20562023.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jain S, Van Scoyk A, Morgan EA, Matthews
A, Stevenson K, Newton G, Powers F, Autio A, Louissaint A, Pontini
G, et al: Targeted inhibition of CD47-SIRPα requires Fc-FcγR
interactions to maximize activity in T-cell lymphomas. Blood.
134:1430–1440. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fleseriu M, Langlois F, Lim DST, Varlamov
EV and Melmed S: Acromegaly: pathogenesis, diagnosis, and
management. Lancet Diabetes Endocrinol. 10:804–826. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Raverot G, Jouanneau E and Trouillas J:
Management of endocrine disease: Clinicopathological classification
and molecular markers of pituitary tumours for personalized
therapeutic strategies. Eur J Endocrinol. 170:R121–R132. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wu L, Yan J, Bai Y, Chen F, Zou X, Xu J,
Huang A, Hou L, Zhong Y, Jing Z, et al: An invasive zone in human
liver cancer identified by Stereo-seq promotes hepatocyte-tumor
cell crosstalk, local immunosuppression and tumor progression. Cell
Res. 33:585–603. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhou Q, Peng RQ, Wu XJ, Xia Q, Hou JH,
Ding Y, Zhou QM, Zhang X, Pang ZZ, Wan DS, et al: The density of
macrophages in the invasive front is inversely correlated to liver
metastasis in colon cancer. J Transl Med. 8:132010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang M, He Y, Sun X, Li Q, Wang W, Zhao A
and Di W: A high M1/M2 ratio of tumor-associated macrophages is
associated with extended survival in ovarian cancer patients. J
Ovarian Res. 7:192014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Pinto ML, Rios E, Durães C, Ribeiro R,
Machado JC, Mantovani A, Barbosa MA, Carneiro F and Oliveira MJ:
The two faces of tumor-associated macrophages and their clinical
significance in colorectal cancer. Front Immunol. 10:18752019.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Edin S, Wikberg ML, Dahlin AM, Rutegård J,
Öberg Å, Oldenborg PA and Palmqvist R: The distribution of
macrophages with a M1 or M2 phenotype in relation to prognosis and
the molecular characteristics of colorectal cancer. PLoS One.
7:e470452012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Cabrita R, Lauss M, Sanna A, Donia M,
Larsen MS, Mitra S, Johansson I, Phung B, Harbst K,
Vallon-Christersson J, et al: Tertiary lymphoid structures improve
immunotherapy and survival in melanoma. Nature. 577:561–565. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Petitprez F, de Reyniès A, Keung EZ, Chen
TW, Sun CM, Calderaro J, Jeng YM, Hsiao LP, Lacroix L, Bougoüin A,
et al: B cells are associated with survival and immunotherapy
response in sarcoma. Nature. 577:556–560. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Harris FS and Rhoton AL: Anatomy of the
cavernous sinus. A microsurgical study. J Neurosurg. 45:169–180.
1976. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Willingham SB, Volkmer JP, Gentles AJ,
Sahoo D, Dalerba P, Mitra SS, Wang J, Contreras-Trujillo H, Martin
R, Cohen JD, et al: The CD47-signal regulatory protein alpha
(SIRPa) interaction is a therapeutic target for human solid tumors.
Proc Natl Acad Sci USA. 109:6662–6667. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Advani R, Flinn I, Popplewell L, Forero A,
Bartlett NL, Ghosh N, Kline J, Roschewski M, LaCasce A, Collins GP,
et al: CD47 blockade by Hu5F9-G4 and rituximab in Non-Hodgkin's
lymphoma. N Engl J Med. 379:1711–1721. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Silva MMA, Kokai LE, Donnenberg VS, Fine
JL, Marra KG, Donnenberg AD, Neto MS and Rubin JP: Oncologic safety
of fat grafting for autologous breast reconstruction in an animal
model of residual breast cancer. Plast Reconstr Surg. 143:103–112.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Wu X, Han X, Zhu H, Li M, Gong L, Jing S,
Xie W, Liu Z, Li C and Zhang Y: Single-cell transcriptomics
identify a novel macrophage population associated with bone
invasion in pituitary neuroendocrine tumors. J Exp Clin Cancer Res.
44:272025. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Gao J, Gu D, Yang K, Zhang J, Lin Q, Yuan
W, Zhu X, Dixit D, Gimple RC, You H, et al: Infiltrating plasma
cells maintain glioblastoma stem cells through IgG-tumor binding.
Cancer Cell. 43:122–143.e128. 2025. View Article : Google Scholar : PubMed/NCBI
|