Introduction
Mechanical ventilation (MV) is essential for
respiratory support and employs positive pressure to overcome
airway resistance and lung elasticity, inevitably causing pulmonary
stretching and strain (1). While
necessary for homeostasis, excessive mechanical forces during MV
[such as high tidal volumes (HTVs)] trigger epithelial dysfunction
(2,3). This maladaptive response, termed
‘biotrauma’, involves the release of cytokines, such as IL-6, TNF-α
and IL-1β, which exacerbates local damage, potentially leading to
systemic inflammation and multiple organ failure, collectively
known as ventilator-induced lung injury (VILI) (4,5). Key
features of VILI include disruption of the lung epithelial barrier,
a critical defence mechanism, and pronounced proinflammatory
signalling originating from these cells (6,7).
However, the precise molecular sensors that translate harmful
mechanical stretch into this injurious biochemical response within
epithelial cells remain incompletely understood.
Mechanosensitive ion channels translate physical
stimuli into biochemical signals within the cellular
microenvironment. As the frontline sensors of MV-associated
stretch, lung epithelial cells utilize these channels to detect
mechanical forces (8). Piezo1 is a
ubiquitously expressed, membrane tension-gated, nonselective cation
channel activated by forces such as shear stress, compression,
stretching and osmotic pressure (9). It is broadly expressed and implicated
in diverse physiological and pathophysiological processes,
including vascular development and osmoregulation (10). In the lung, Piezo1 is present in
epithelial cells and its activation has been linked to processes
such as stretch-induced epithelial-mesenchymal transition (11). While other mechanosensors (for
example, TRPV4) have been associated with VILI (12,13),
the specific role of Piezo1 in mediating inflammatory injury in
airway epithelia during MV has not been defined.
It has been hypothesized that Piezo1 may act as a
critical mechanotransducer in lung epithelial cells in VILI,
orchestrating downstream inflammatory signalling. Therefore, the
present study aimed to investigate the role of Piezo1 in lung
epithelial cells in VILI, and to elucidate its downstream
signalling mechanisms using genetic and pharmacological approaches
in vivo and in vitro.
Materials and methods
Materials and reagents
Cyclosporine A (CsA; cat. no. SML1018) and FK506
(cat. no. F4679) were purchased from MilliporeSigma (Merck KGaA).
Myeloperoxidase (MPO) detection kits (cat. no. A044-1-1) were
obtained from Nanjing Jiancheng Bioengineering Institute.
calcineurin activity assay kits (cat. no. AB139461) were purchased
from Abcam. ELISA kits for TNF-α (cat. no. EMC102a), IL-1β (cat.
no. EMC001b) and IL-6 (cat. no. EMC004) were acquired from
Neobioscience Technology Co., Ltd. The anti-NFATc3 antibody (cat.
no. ab245501) was obtained from Abcam and the anti-Piezo1 antibody
(cat. no. A23380) was obtained from ABclonal Biotech Co., Ltd.
Fluo-3 AM (cat. no. S1056), anti-β-actin (cat. no. AF5003),
anti-GAPDH (cat. no. AF1186), anti-Lamin B1 (cat. no. AF1408) and
horseradish peroxidase (HRP)-conjugated secondary antibodies (cat.
nos. A0208 and A0216) were obtained from Beyotime
Biotechnology.
Animals and treatment
A total of 30 male C57BL/6 mice (weight, 20–25 g;
age, 6–8 weeks) were procured from Shulaibao (Wuhan) Biotechnology
Co., Ltd. The animals were maintained under specific pathogen-free
conditions, with a controlled temperature (25°C), humidity (50±10%)
and a 12-h light/dark cycle, with ad libitum access to food and
water. The mice were randomly assigned to the sham and HTV groups
(n=6/group) using a computer-generated randomization sequence.
Histopathological evaluation of lung sections was performed by an
investigator who was blinded to the group allocation. A total of 2
mice failed intubation and 1 mouse died during the ventilation
protocol; these 3 mice were excluded from the final analysis. All
experimental protocols adhered to the guidelines set forth in the
National Institutes of Health Guide for the Care and Use of
Laboratory Animals (14).
A murine model of VILI was established via MV
following a previously described procedure (15). Briefly, anaesthesia was induced by
intraperitoneal injection of pentobarbital sodium (50 mg/kg).
Following anaesthesia, oral endotracheal intubation was performed
using an 18 G catheter, and the mice were ventilated in
volume-controlled mode (55-7062; Harvard Apparatus) for 6 h with
the following parameters: Tidal volume, 35 ml/kg; respiratory rate,
80 breaths/min; zero positive end-expiratory pressure (HTV group).
The fraction of inspired oxygen was maintained at 0.21 throughout.
The mice in the sham group underwent intubation and spontaneous
breathing for 6 h.
At the end of the experiments, mice were deeply
anesthetized with an intraperitoneal injection of sodium
pentobarbital (50 mg/kg). Bronchoalveolar lavage fluid (BALF) was
collected by cannulating the trachea and instilling 0.4 ml sterile
PBS into the left lung three times. Following BALF collection, all
mice were euthanized via an intraperitoneal injection of an
overdose of sodium pentobarbital (150 mg/kg). Death was confirmed
by the cessation of spontaneous breathing and heartbeat, followed
by cervical dislocation as a secondary physical method to ensure
euthanasia. Immediately after sacrifice, the right lung lobes were
harvested separately, the superior lobe was fixed in 4%
paraformaldehyde for histopathological analysis, the middle lobe
was used for wet-to-dry weight ratio measurement, and the inferior
and post-caval lobes were snap-frozen in liquid nitrogen and stored
at −80°C for subsequent MPO activity, western blotting and reverse
transcription-quantitative PCR (RT-qPCR) analyses.
Generation of conditional Piezo1
conditional knockout (Piezo1CKO) mice
To establish male mice with tamoxifen-inducible,
lung epithelial-specific deletion of Piezo1
(Piezo1CKO), Piezo1F/F mice
(Piezo1tm2.1Apat/J; Jackson Laboratory) were bred
with Sftpc-CreERT mice (Jackson Laboratory) in our
laboratory. The resulting offspring were genotyped in our
laboratory via PCR with primers targeting the Cre recombinase and
the floxed Piezo1 alleles according to a previously
described protocol (10). To
induce Piezo1 knockout, 3-week-old Piezo1CKO mice
(n=20) received daily intraperitoneal injections of tamoxifen (cat.
no. HY-13757A; MedChemExpress) dissolved in corn oil (cat. no.
HY-Y1888; MedChemExpress) at a concentration of 10 mg/ml (0.1
ml/injection) for 7 consecutive days. Age- and weight-matched
Piezo1F/F littermates that did not carry the Cre
transgene (n=20) were utilized as control animals for the
Piezo1CKO mice cohort. All mice were
housed under the same specific pathogen-free conditions as
described for the C57BL/6 mice, with controlled temperature (25°C),
humidity (50±10%) and a 12-h light/dark cycle, with ad
libitum access to food and water. Efficient and specific
ablation of Piezo1 in lung epithelial cells was subsequently
verified by PCR genotyping and immunofluorescence costaining. Both
the control and Piezo1CKO mice were subjected to
HTV at 8 weeks of age (weight, 22–26 g). Immediately after the 6-h
ventilation protocol, the mice were euthanized by intraperitoneal
injection of an overdose of sodium pentobarbital (150 mg/kg)
followed by cervical dislocation, as described in the
aforementioned section, and lung tissues were collected for
subsequent analyses.
Cell culture and mechanical
stretching
The MLE-12 mouse alveolar epithelial cell line,
acquired from the American Type Culture Collection, was cultured in
Dulbecco's modified Eagle's medium (cat. no. C11995500BT; Gibco;
Thermo Fisher Scientific, Inc.) supplemented with 10% foetal bovine
serum (cat. no. 10099141; Gibco; Thermo Fisher Scientific, Inc.)
and antibiotics (100 U/ml penicillin and 100 µg/ml streptomycin;
cat. no. 15140122; Gibco; Thermo Fisher Scientific, Inc.). The
cells were maintained at 37°C in a humidified atmosphere containing
5% CO2.
For gene silencing studies, short hairpin (sh)RNA
targeting Piezo1 (shPiezo1) and scrambled control shRNA (Scr-shRNA)
were designed and synthesized. The shRNA sequences were cloned into
the pLKO.1-puro lentiviral vector (cat. no. SHC001;
MilliporeSigma). Lentiviral particles were generated using a
second-generation packaging system. Briefly, 293T cells (cat. no.
CRL-3216; American Type Culture Collection) were co-transfected
with the lentiviral transfer plasmid (pLKO.1-shPiezo1 or
pLKO.1-Scr; 10 µg), the packaging plasmid psPAX2 (7.5 µg; cat. no.
12260; Addgene, Inc.), and the envelope plasmid pMD2.G (2.5 µg;
cat. no. 12259; Addgene, Inc.) using Lipofectamine® 3000
transfection reagent (cat. no. L3000015; Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's instructions.
Transfection was carried out at 37°C for 6 h, after which the
medium was replaced with fresh culture medium. Lentiviral particles
were collected from the supernatant 48 and 72 h post-transfection,
to maximize viral yield, filtered through 0.45-µm filters, and
concentrated using Lenti-X Concentrator (cat. no. 631232; Takara
Bio, Inc.) according to the manufacturer's protocol. The virus
pellets were then resuspended in cold culture medium and stored at
−80°C until use.
MLE-12 cells were seeded in 6-well plates at a
density of 5×105 cells/well. At 60–70% confluence,
MLE-12 cells were transduced with lentiviral particles carrying
shPiezo1 or Scr-shRNA in the presence of 8 µg/ml Polybrene
(MilliporeSigma) at 37°C in a humidified 5% CO2
incubator. The target sequences for the mouse shPiezo1 and
Scr-shRNA (negative control) used in the present study are as
follows: shPiezo1 (mouse target sequence):
5′-GAGCCAGAAGCTAAGCTGGAA-3′; Scr-shRNA (target sequence):
5′-TTCTCCGAACGTGTCACGT-3′. Lentiviral particles carrying shPiezo1
or Scr-shRNA were used at a multiplicity of infection of 20, with a
viral titre of 1×108 TU/ml, as determined by qPCR. A
total of 24–48 h post-transduction (to allow for viral genomic
integration and expression of the resistance gene), cells were
subjected to puromycin selection (2 µg/ml) for 7 days to establish
stable cell lines, and maintained in medium containing 0.5 µg/ml
puromycin thereafter. Gene silencing efficiency was confirmed using
qPCR.
For mechanical stimulation assays, MLE-12 cells were
seeded on flexible-bottom 6-well BioFlex plates at 5×105
cells/well and grown to 80% confluence. The culture medium was
replaced with fresh medium containing 0.5% foetal bovine serum for
12 h prior to stretching. A Flexcell Tension Plus system (FX-5000
T; Flexcell International Corporation) was employed to apply cyclic
stretch (18% elongation, 0.5 Hz, 30 cycles/min) at 37°C in a
humidified atmosphere with 5% CO2 for the indicated
durations (3 or 6 h, or 5–30 min). The peak strain was validated
using the calibration loading station of the system prior to the
experiments. For calcineurin inhibition experiments, cells were
pretreated with CsA (10 µM) or FK506 (10 µM) at 37°C for 1 h prior
to mechanical stimulation. Cells were then subjected to cyclic
stretch for 6 h. Non-stretched cells cultured on identical plates
served as the sham group.
Histopathological evaluation
The superior lobe of the right lung was fixed in 4%
paraformaldehyde at room temperature for 24 h, embedded in paraffin
and sectioned at a thickness of 5 µm. Haematoxylin and eosin
staining was performed on these sections using haematoxylin (5 min)
and eosin (3 min) at room temperature. Stained sections were
examined under a light microscope (BX53; Olympus Corporation). Lung
injury was assessed using a semi-quantitative scoring system
(16) that evaluated four
parameters: i) Alveolar congestion, ii) haemorrhage, iii) leukocyte
infiltration and iv) alveolar wall thickness, each graded from 0
(normal) to 3 (severe), and the four scores were summed to obtain
the total lung injury score (range 0–12).
Analysis of BALF
Following anaesthesia with intraperitoneal injection
of pentobarbital sodium (50 mg/kg), the trachea was exposed and
cannulated with an 18-gauge needle. The right lung hilum was
ligated, and the left lung was consistently lavaged by instilling
and withdrawing 0.4 ml sterile PBS three times. Immediately after
BALF collection, mice were euthanized by cervical dislocation while
maintained under deep anaesthesia. The collected BALF was
centrifuged at 400 × g for 10 min at 4°C. After centrifugation, the
cell pellet was resuspended in PBS and total cell counts were
determined using a haemocytometer under a light microscope. The
results are expressed as the total number of cells per ml of BALF.
The protein concentration in the BALF supernatant was quantified
using a bicinchoninic acid (BCA) protein assay kit (Beyotime
Biotechnology).
Assessment of pulmonary oedema
The wet weight of the right middle lung lobe was
recorded immediately after excision. The tissue was then dried at
80°C for 48 h to obtain the dry weight. The wet/dry weight ratio
was calculated to evaluate lung oedema.
Measurement of MPO activity
The inferior lung lobe was homogenized in PBS and
centrifuged at ~30,000 × g for 30 min at 4°C. MPO activity in the
supernatant was determined using a commercial MPO assay kit
according to the manufacturer's instructions.
Calcineurin activity assay
Lung tissue homogenates or cell lysates were
prepared in ice-cold lysis buffer and centrifuged at 12,000 × g for
10 min at 4°C. The supernatants were collected, and protein
concentrations were determined using a bicinchoninic acid (BCA)
protein assay kit (Beyotime Biotechnology). Calcineurin activity
was then assessed based on the release of inorganic phosphate from
the synthetic phosphopeptide substrate RII (DLDVPIPGRFDRRVSVAAE) in
the presence of calmodulin. The reaction mixture (containing the
sample, RII peptide and calmodulin) was incubated at 30°C for 30
min and the reaction was terminated by adding molybdate reagent.
The absorbance was then measured at 636 nm using a microplate
reader (Synergy Neo HTS; BioTek; Agilent Technologies, Inc.).
Results were expressed as units per milligram of protein (U/mg
protein), where one unit represents the release of 1 µmol
phosphate/min.
Intracellular calcium imaging
Cytosolic Ca2+ levels were monitored
using a Fluo-3 AM fluorescent probe according to the manufacturer's
guidelines. After mechanical stimulation, the cells were harvested,
washed with PBS and incubated with 5 µM Fluo-3 AM in serum-free
medium at 37°C for 30 min. For fluorescence imaging, cells loaded
with Fluo-3 AM were visualized under a fluorescence microscope
(IX73; Olympus Corporation) at the indicated time points, and
representative images were captured and analysed. For agonist
experiments, cells were loaded with Fluo-3 AM as aforementioned,
then treated with the Piezo1 agonist Yoda1 (10 µM; cat. no. 5586;
Tocris Bioscience) at 37°C. Fluorescence intensity was measured at
5, 15 and 30 min after Yoda1 treatment using a microplate reader
(Synergy Neo HTS). For inhibitor experiments, cells were pretreated
with the Piezo1 inhibitor GsMTx4 (5 µM; cat. no. ab141871; Abcam)
at 37°C for 30 min prior to mechanical stimulation. Cells were then
subjected to mechanical stretch for 30 min, and intracellular
Ca2+ levels were measured using a microplate reader
(Synergy Neo HTS). The fluorescence values are expressed as
ΔF/F0, where F0 is the baseline fluorescence
prior to stimulation.
Cytokine concentration
measurements
The concentrations of TNF-α, IL-6 and IL-1β in the
BALF and cell culture supernatants were measured using commercially
available ELISA kits according to the manufacturer's protocols.
Samples and standards were incubated for 2 h (TNF-α) or 4 h (IL-6,
IL-1β) at room temperature. The detection limits of the assays were
4 pg/ml for TNF-α, 2 pg/ml for IL-6 and 1 pg/ml for IL-1β.
Western blotting
Cytoplasmic and nuclear protein extracts were
prepared from cultured MLE-12 cells using a commercial extraction
kit (Beijing Solarbio Science & Technology Co., Ltd.). Protein
concentrations were determined using a BCA protein assay kit,
according to the manufacturer's instructions. Equal amounts of
protein (30 µg/lane) were separated by SDS-PAGE on 10% gels and
transferred onto polyvinylidene fluoride membranes
(MilliporeSigma). The membranes were blocked with 5% non-fat milk
in Tris-buffered saline containing 0.1% Tween-20 (TBST) at room
temperature for 1 h. Subsequently, the membranes were incubated
overnight at 4°C with primary antibodies against Piezo1 (1:500),
NFATc3 (1:500), β-actin (1:1,000), GAPDH (1:1,000) and Lamin B1
(1:1,000). GAPDH was used as the loading control for whole-cell
lysates, whereas β-actin and Lamin B1 served as loading controls
for cytoplasmic and nuclear fractions, respectively. After washing
with TBST, the membranes were incubated with HRP-conjugated
secondary antibodies (1:5,000) at room temperature for 1 h. Protein
bands were visualized using an enhanced chemiluminescence detection
reagent (Beyotime Biotechnology) and band intensity was
semi-quantified using ImageJ software (version 1.53; National
Institutes of Health).
RT-qPCR
Total RNA was isolated from cells using TRIzol
reagent (Invitrogen; Thermo Fisher Scientific, Inc.). cDNA
synthesis was performed using the PrimeScript™ RT reagent Kit with
gDNA Eraser (Takara Bio, Inc.) according to the manufacturer's
protocol (RT at 37°C for 15 min, followed by enzyme inactivation at
85°C for 5 sec). qPCR was carried out using TB Green®
Premix Ex Taq™ (Tli RNaseH Plus) (cat. no. RR420A; Takara Bio,
Inc.) on an Applied Biosystems 7500 Fast Real-Time PCR System
(Applied Biosystems; Thermo Fisher Scientific, Inc.). The
thermocycling conditions were as follows: Initial denaturation at
95°C for 30 sec, followed by 40 cycles at 95°C for 5 sec and 60°C
for 34 sec. A dissociation curve analysis was performed at the end
of each run to verify specific amplification. The primer sequences
were as follows: Mouse Piezo1, forward
5′-TCATCATCCTTAACCACATGGTG-3′, reverse
5′-TGAAGACGATAGCTGTCATCCA-3′; mouse Gapdh, forward
5′-AGGTCGGTGTGAACGGATTTG-3′, reverse 5′-TGTAGACCATGTAGTTGAGGTCA-3′.
Relative gene expression was analysed using the 2−ΔΔCq
method (17), with GAPDH serving
as the internal control.
Immunohistochemical analysis
Lung tissue samples were fixed in 4%
paraformaldehyde at room temperature for 24 h, embedded in paraffin
and sectioned at a thickness of 5 µm. After deparaffinization and
rehydration, antigen retrieval was performed by heating the
sections in citrate buffer (pH 6.0) at 95°C for 20 min. Endogenous
peroxidase activity was blocked by incubating the sections with 3%
hydrogen peroxide in methanol for 10 min at room temperature, and
the sections were then incubated with 5% normal goat serum (cat.
no. S-1000; Vector Laboratories; Maravai LifeSciences) in PBS for 1
h at room temperature to block non-specific binding sites.
The sections were then incubated overnight at 4°C
with primary antibody against Piezo1 (1:200). After washing with
PBS, the sections were incubated with biotin-conjugated goat
anti-rabbit IgG secondary antibody (1:200; cat. no. BA-1000; Vector
Laboratories; Maravai LifeSciences) for 30 min at room temperature,
followed by incubation with streptavidin-horseradish peroxidase
conjugate (1:500; cat. no. SA-5004; Vector Laboratories; Maravai
LifeSciences) for 30 min at room temperature. Immunoreactivity was
visualized using DAB substrate kit (cat. no. SK-4100; Vector
Laboratories; Maravai LifeSciences) according to the manufacturer's
instructions. Sections were counterstained with hematoxylin room
temperature for 5 min, dehydrated through graded ethanol and
xylene, and mounted with permanent mounting medium. Stained
sections were examined under a light microscope (BX53) and images
were captured using Olympus CellSens Imaging software (version
1.16; http://www.olympus-lifescience.com/en/software/cellsens/).
Immunofluorescence staining
Lung tissue sections staining
Lung tissue samples were fixed in 4%
paraformaldehyde at room temperature for 24 h, embedded in paraffin
and sectioned at a thickness of 5 µm. After deparaffinization and
rehydration, antigen retrieval was performed by heating the
sections in citrate buffer (pH 6.0) at 95°C for 20 min. The
sections were then permeabilized with 0.3% Triton X-100 in PBS for
10 min at room temperature and blocked with 5% normal goat serum in
PBS for 1 h at room temperature. The sections were incubated
overnight at 4°C with rabbit anti-Piezo1 primary antibody (1:200)
and mouse anti-cytokeratin 8 primary antibody (CK8; 1:200; cat. no.
ab9023; Abcam). After washing with PBS, the sections were incubated
with Alexa Fluor 488-conjugated goat anti-mouse IgG (1:500; cat.
no. A11001; Invitrogen; Thermo Fisher Scientific, Inc.) and Alexa
Fluor 594-conjugated goat anti-rabbit IgG (1:500;cat. no. A11012;
Invitrogen; Thermo Fisher Scientific, Inc.) for 1 h at room
temperature in the dark. Nuclei were counterstained with DAPI (1
µg/ml; cat. no. C1005; Beyotime Biotechnology) for 5 min at room
temperature. Stained sections were mounted with anti-fade mounting
medium and visualized under a confocal laser scanning microscope
(Zeiss LSM 880; Zeiss AG). Images were captured and analysed using
Zeiss Zen 2011 software (https://www.zeiss.com.cn/microscopy/products/microscope-software/zen.html).
Cultured cell staining
MLE-12 cells seeded on coverslips at a density of
5×105 cells per well in 6-well plates, then fixed with
4% paraformaldehyde for 15 min at room temperature, permeabilized
with 0.3% Triton X-100 in PBS for 10 min and blocked with 5% normal
goat serum in PBS for 1 h at room temperature. For F-actin
staining, cells were incubated with FITC Phalloidin (1:200; cat.
no. RM02836; ABclonal Biotech Co., Ltd.) for 30 min at room
temperature in the dark. For NFAT isoform detection, cells were
incubated overnight at 4°C with primary antibodies against NFATc1
(1:100; cat. no. ab2796; Abcam), NFATc2 (1:100; cat. no. ab2722;
Abcam), NFATc3 (1:100) or NFATc4 (1:100; cat. no. ab99431; Abcam).
After washing, cells were incubated with Alexa Fluor 594-conjugated
goat anti-rabbit IgG (1:500; cat. no. A11012; Invitrogen; Thermo
Fisher Scientific, Inc.) for 1 h at room temperature in the dark.
Nuclei were counterstained with DAPI (1 µg/ml) for 5 min.
Coverslips were mounted onto glass slides with anti-fade mounting
medium and visualized under a confocal laser scanning microscope
(Zeiss LSM 880). Images were captured and analysed using ZEN
software.
Statistical analysis
Data were analysed using GraphPad Prism 7.04
(Dotmatics). Normality was assessed using the Shapiro-Wilk test.
For comparisons between two groups, normally distributed continuous
data were analysed using an unpaired Student's t-test and are
presented as the mean ± SD. By contrast, nonnormally distributed
continuous data or ordinal data (for example, lung injury score)
were analysed using the Mann-Whitney U test and are presented as
the median with interquartile range (IQR). For experiments
involving more than two groups, oneway analysis of variance (ANOVA)
followed by Bonferroni's post hoc test was used for normally
distributed data, whereas the Kruskal-Wallis test followed by
Dunn's post hoc test was applied for nonnormally distributed or
ordinal data. For factorial designs with two independent factors
(for example, genotype and ventilation status), twoway ANOVA
followed by Bonferroni's post hoc test was performed. P<0.05 was
considered to indicate a statistically significant difference. All
experiments included at least six biological replicates per
group.
Results
Piezo1 expression is upregulated in
lung epithelium during VILI in vivo and in vitro
To investigate the potential association between
Piezo1 protein abundance and VILI, immunofluorescence staining for
Piezo1 was performed on lung sections from experimental mice.
Immunofluorescence analysis revealed constitutive expression of
Piezo1 in the lung epithelium of sham-operated control mice, as
evidenced by its colocalization with the epithelial marker CK8
(Fig. 1A). Notably, compared wiin
the sham control mice, mice subjected to 6 h of HTV MV exhibited a
marked increase in Piezo1 immunofluorescence intensity within the
lung epithelium. In line with these findings, immunohistochemical
analysis revealed a clear increase in Piezo1 protein expression in
the lung tissue of mice exposed to MV (Fig. 1B).
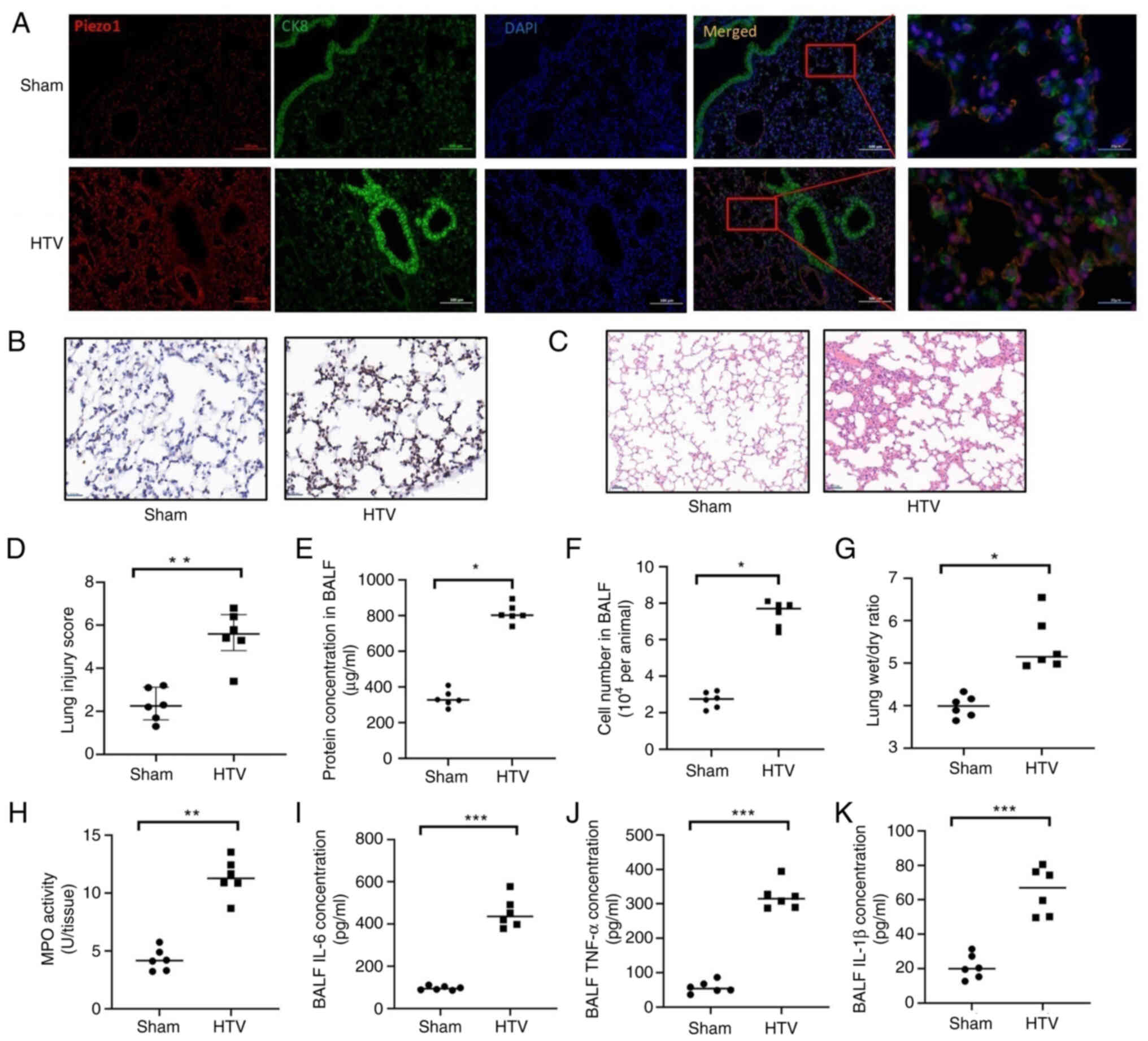 | Figure 1.Piezo1 expression is upregulated in
the lung epithelium during ventilator-induced lung injury. (A)
Representative immunofluorescence images of lung sections from
sham-operated control mice and mice subjected to 6 h of mechanical
ventilation. Piezo1 protein (red) is constitutively expressed and
colocalizes (yellow in merged panels) with the epithelial cell
marker CK8 (green). Nuclei were counterstained with DAPI (blue).
Scale bar, 100 µm. (B) Representative images of immunohistochemical
staining for Piezo1 expression in lung epithelium from
sham-operated mice and mice subjected to 6 h of mechanical
ventilation. Scale bar, 20 µm (original magnification, ×400). (C)
Representative haematoxylin and eosin-stained lung sections from
sham-operated and HTV-ventilated mice. Scale bar, 50 µm. (D)
Quantitative lung injury score was determined based on histological
evaluation of alveolar congestion, haemorrhage, leukocyte
infiltration and alveolar wall thickness, each graded from 0
(normal) to 3 (severe). Data are presented as the median
(interquartile range); n=6 mice/group and Pvalues were determined
by Mann-Whitney U test. Lung injury was further evaluated on the
basis of (E) protein concentration, (F) cell number in BALF and (G)
wet/dry weight ratio. (H) MPO activity in lung tissue.
Proinflammatory cytokines in BALF were evaluated using the
following ELISA kits: (I) IL-6, (J) TNF-α and (K) IL-β. (E-K) Data
are presented as the mean ± SD, n=6 mice/group. Pvalues were
determined by Student's t-test. *P<0.05, **P<0.01,
***P<0.001. BALF, bronchoalveolar lavage fluid; CK8, cytokeratin
8; HTV, high tidal volume; MPO, myeloperoxidase. |
Consistent with the established VILI pathology,
histopathological evaluation revealed that ventilation with HTV
induced marked lung injury, as evidenced by alveolar wall
thickening, inflammatory cell infiltration, alveolar congestion and
haemorrhage in lung tissue sections (Fig 1C) The lung injury score was
significantly elevated in the HTV group [median (IQR): 5.60
(4.83–6.50)] compared with in the sham group [2.25 (1.60–3.13)
(P<0.01; Fig. 1C and D).
Barrier dysfunction was evidenced by a ~2.4-fold increase in BALF
protein concentration (813.8±51.63 vs. 335.7±45.48 µg/ml in the
sham group; P<0.05; Fig. 1E),
and a ~2.7-fold increase in the total BALF cell count (P<0.05;
Fig. 1F). The lung wet/dry weight
ratio, indicating oedema, was greater in mice in the HTV group
(5.44±0.64) compared with that in sham mice (3.98±0.54; P<0.05;
Fig. 1G). Inflammatory marker
levels were also elevated in the HTV group: Lung tissue MPO
activity increased by ~2.6-fold (P<0.01), whereas the BALF
levels of IL-6, TNF-α, and IL-1β were increased by ~4.7-, 5.6- and
3.1-fold, respectively (all P<0.001 vs. sham; Fig. 1H-K).
The current study also assessed whether mechanical
stress could directly upregulate Piezo1 expression in lung
epithelial cells in vitro. Compared with in control cells,
MLE-12 cells subjected to mechanical stretch (18% elongation, 0.5
Hz) exhibited a time-dependent increase in Piezo1 mRNA expression,
which was upregulated ~3.2-fold at 3 h and ~6.5-fold at 6 h (both
P<0.001; Fig. 2A). The protein
levels followed a similar trend, increasing by ~1.4-fold at 3 h and
~2.0-fold at 6 h post-stretching (P<0.05 and P<0.001,
respectively; Fig. 2B and C).
Concomitantly, after 6 h of stretch-induced epithelial injury, the
F-actin cytoskeleton was notably disrupted (Fig. 2D), and the levels of IL-6, TNF-α
and IL-1β in the supernatant were increased by ~3.9-, 7.1- and
6.1-fold, respectively (all P<0.001 vs. sham; Fig. 2E-G).
Genetic ablation of epithelial Piezo1
attenuates VILI in vivo
To establish causality, a lung epithelial-specific
Piezo1 knockout mouse model (Piezo1CKO) was used
(Fig. 3A). Among ventilated mice,
the lung injury score was significantly lower in the
Piezo1CKO group than in the Piezo1F/F group
[median (IQR): 3.25 (2.95–4.68) vs. 5.80 (5.00–6.58); P<0.05;
Fig. 3B and C]. Barrier function
was preserved, with the BALF protein concentration decreased by
~53.2% (P<0.001) and the total BALF cell count reduced by ~43.4%
(P<0.05) relative to those in the ventilated
Piezo1F/F group (Fig. 3D
and E). The lung wet/dry weight ratio and MPO activity were
also reduced by ~26.1 and 51.1%, respectively (both P<0.001;
Fig. 3F and G). Notably, HTV
MV-induced elevations in the levels of cytokines (IL-6, TNF-α, and
IL-1β) in the BALF were nearly abolished in Piezo1CKO
mice, remaining at baseline levels (IL-6 and IL-1β: P<0.05;
TNF-α: P<0.01 vs. ventilated Piezo1F/F mice; Fig. 3H-J).
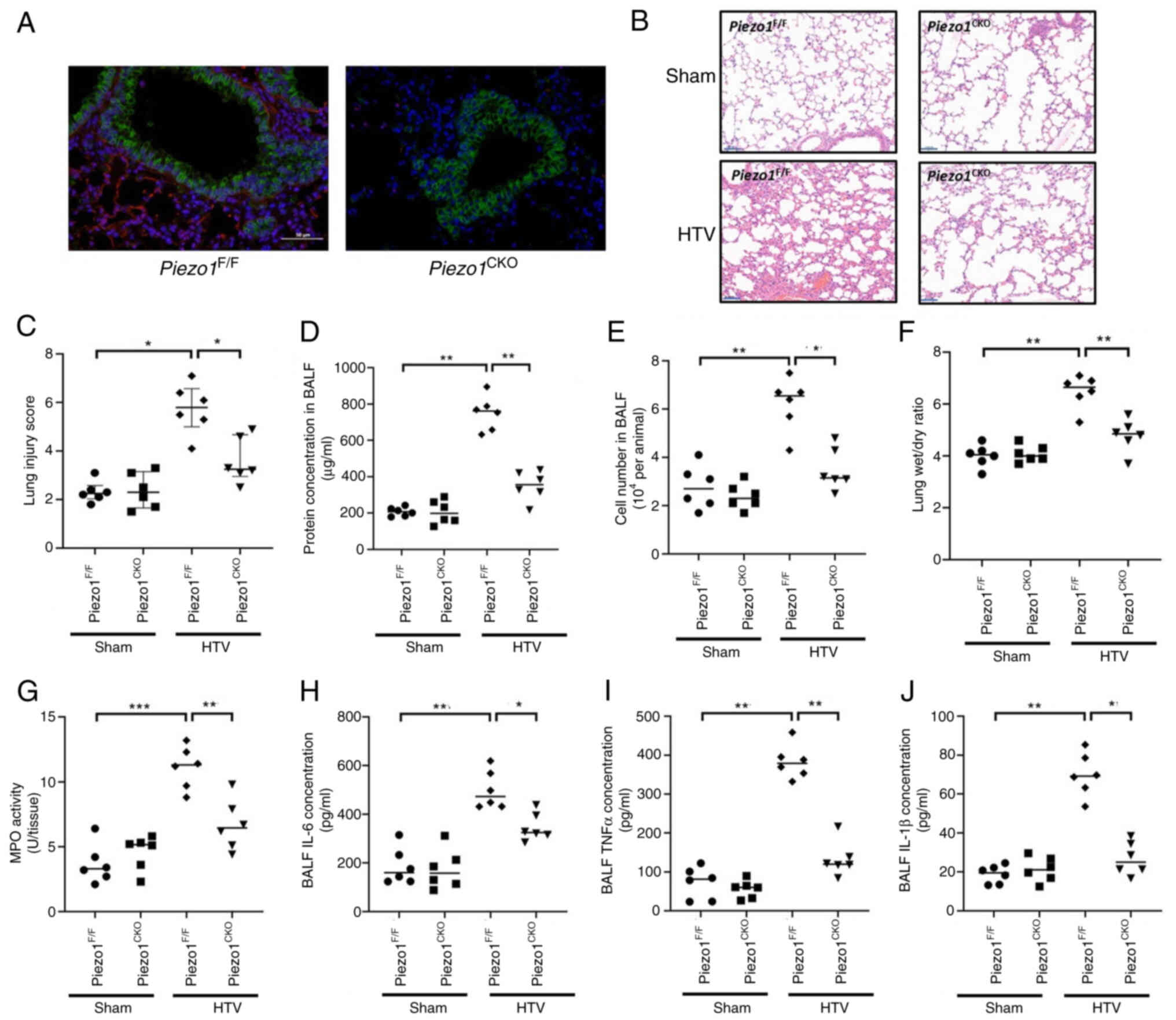 | Figure 3.Lung epithelial-specific Piezo1
deletion attenuates ventilator-induced lung injury. (A) Validation
of Piezo1 knockout efficiency in the lung epithelium.
Representative immunofluorescence images of lung sections from
control and Piezo1CKO mice showing the expression of the
Piezo1 protein (red), the epithelial cell marker CK8 (green), and
nuclei (DAPI, blue). Scale bar, 50 µm. (B) Representative
haematoxylin and eosin-stained lung sections from control and
Piezo1CKO mice after 6 h of mechanical ventilation.
Scale bar, 50 µm. (C) Quantitative histopathological lung injury
score. Data are presented as the median (IQR); n=6/group. Data were
analysed using the Kruskal-Wallis test followed by Dunn's post hoc
test. (D) Total protein concentration in BALF. (E) Total cell
counts in the BALF. (F) Lung wet/dry weight ratio. (G) Lung MPO
activity. BALF concentrations of the inflammatory cytokines (H)
IL-6, (I) TNF-α and (J) IL-1β. (D-J) Data are presented as the mean
± SD, n=6 mice/group. Pvalues were determined by oneway ANOVA.
*P<0.05, **P<0.01, ***P<0.001. BALF, bronchoalveolar
lavage fluid; CK8, cytokeratin 8; CKO, conditional; HTV, high tidal
volume; MPO, myeloperoxidase. |
Genetic inhibition of Piezo1 protects
against stretch-induced injury in vitro
Compared with in the Scr-shRNA control group, stable
knockdown of Piezo1 in MLE-12 cells (shPiezo1) resulted in a 62.1%
reduction in mRNA expression and a 78.5% reduction in protein
expression (P<0.001; Fig.
4A-C). In the Scr-shRNA cells, 6 h of stretching markedly
increased cytokine secretion (IL-6, 6.5-fold; TNF-α, 4.3-fold;
IL-1β, 4.0-fold; all P<0.001 vs. unstretched control cells;
Fig. 4D-F). This response was
almost completely abolished in shPiezo1 cells (all P<0.01 vs.
stretched Scr-shRNA cells). Furthermore, while stretching severely
disrupted the F-actin network in control cells, cytoskeletal
integrity was largely preserved in shPiezo1 cells under identical
conditions (Fig. 4G).
Piezo1 mediates mechanical
stretch-induced Ca2+ influx and activates calcineurin
signalling
Using Fluo-3 AM, initial analysis confirmed that
treatment with the Piezo1 agonist Yoda1 for 30 min significantly
increased intracellular Ca2+ levels to approximately
twice the baseline level in control cells (P<0.001; Fig. 5A). Similarly, mechanical stretching
increased intracellular Ca2+ levels in Fluo-3 AM-loaded
MLE-12 cells (Fig. 5B). This
Ca2+ response was blocked in cells transfected with
shPiezo1 (122% of baseline, P<0.001) or by the Piezo1 inhibitor
GsMTx4 (5 µM) (peak reduced to 125% of baseline, P<0.001 vs.
stretch alone) (Fig. 5C and
D).
To elucidate the mechanism by which Piezo1-mediated
Ca2+ influx regulates epithelial inflammation, the
involvement of calcineurin, a Ca2+/calmodulin-dependent
serine/threonine phosphatase, was investigated. Stretch increased
calcineurin activity by 2.2-fold in control cells (P<0.001), but
this increase was reduced to 1.3-fold in cells transfected with
shPiezo1 (P<0.001; Fig. 5E). To
define the role of calcineurin in Piezo1-dependent inflammation,
the pharmacological inhibitors CsA and FK506 were used.
Pretreatment with the calcineurin inhibitors CsA or FK506 (both at
10 µM) significantly attenuated mechanical stretch-induced cytokine
release, reducing IL-6, TNF-α, and IL-1β levels by 70–85% (all
P<0.001; Fig. 5F-H).
Furthermore, the notable disruption in the F-actin network of
stretched cells was alleviated by the pharmacological inhibitors
CsA and FK506 (Fig. 5I).
Piezo1/Ca2+/calcineurin
axis drives NFATc3 activation in stretched epithelial cells
The NFAT family consists of five members, first
known as transcriptional regulators of T-cell development,
maturation and activation. Among these genes, NFATc1 to NFATc4 are
activated by Ca2+/calcineurin signalling signalling
(18). The effects of mechanical
stretching on NFAT activation in lung epithelial cells were
assessed in the present study by immunostaining with anti-NFAT
antibodies. The results revealed that the four isoforms of NFAT
were distributed mainly in the cytoplasm of resting cells. Upon
mechanical stretch treatment, compared with NFATc1, NFATc2 and
NFATc4, NFATc3 exhibited the most pronounced translocation to the
nucleus (Fig. 6A). Western blot
analysis of nuclear fractions revealed a 2.8-fold increase in
nuclear NFATc3 protein expression post-stretching compared with
non-stretched control cells (P<0.001), which was prevented by
Piezo1 knockdown (1.2-fold; P<0.001 vs. stretched control cells)
(Fig. 6B-D). Furthermore, compared
with stretching alone, the calcineurin inhibitors CsA and FK506
significantly inhibited mechanical stretch-induced NFATc3 nuclear
translocation by 60–75% (P<0.001; Fig. 6E-G).
 | Figure 6.Piezo1 mediates CS-induced
inflammation via calcineurin/NFATc3 signalling. (A)
Immunofluorescence screening of the nuclear translocation of NFAT
isoforms (NFATc1-4) upon CS. NFATc3 showed the most prominent
nuclear translocation. Scale bar, 10 µm. (B) Representative western
blot images showing NFATc3 protein levels in the nuclear and
cytoplasmic fractions of MLE-12 cells transfected with Scr-shRNA or
shPiezo1, with or without CS (6 h). GAPDH and Lamin B1 were used as
loading controls for cytoplasmic and nuclear fractions,
respectively. (C) Semi-quantitative analysis of cytoplasmic NFATc3
protein expression normalized to GAPDH. (D) Semi-quantitative
analysis of nuclear NFATc3 protein expression normalized to Lamin
B1. (E) Representative western blot images showing NFATc3 protein
levels in the nuclear and cytoplasmic fractions of MLE-12 cells
treated with or without calcineurin inhibitors (CsA, 10 µM; FK506,
10 µM) prior to CS (6 h). GAPDH and Lamin B1 were used as loading
controls for cytoplasmic and nuclear fractions, respectively. (F)
Semi-quantitative analysis of cytoplasmic NFATc3 protein expression
normalized to GAPDH. (G) Semi-quantitative analysis of nuclear
NFATc3 protein expression normalized to Lamin B1. Data are
presented as the mean ± SD, n=6 each. Pvalues were determined by
oneway ANOVA. *P<0.05, **P<0.01, ***P<0.001. CS, cyclic
stretch; CsA, cyclosporine A; Scr, scrambled; sh, short
hairpin. |
Discussion
The present study revealed that Piezo1 is an
essential mechanotransducer in lung epithelial cells that drives
VILI through the activation of a specific Ca2+-dependent
signalling cascade. To the best of our knowledge, a previously
unrecognized pathway, Piezo1/Ca2+/calcineurin/NFATc3,
has been identified, which converts physical stretch into the
release of proinflammatory cytokines, such as IL-6, TNF-α and
IL-1β, and cytoskeletal disorganization, providing a mechanistic
link between mechanical force and the ‘biotrauma’ of VILI.
The rapid and notable upregulation of Piezo1
expression in lung epithelial cells in response to mechanical
stress in vitro and in vivo observed in the current
study suggests the presence of a feed-forward mechanism that
amplifies the mechanosensitive response. This is distinct from the
role of constitutively active mechanosensors (19) and positions Piezo1 as a master
regulator of epithelial maladaptation to excessive stretch. The
in vivo data demonstrating that epithelial-specific deletion
of Piezo1 significantly ameliorated VILI pathology provide
compelling evidence for its indispensability. The concordant
protective effects of Piezo1 knockdown in vitro preserved
cytoskeletal integrity and abrogated cytokine production,
confirming that the observed benefits are directly attributable to
the loss of Piezo1 in epithelial cells rather than indirect
systemic effects. These findings align with and markedly extend
those of previous studies indicating that Piezo1 is involved in
endothelial activation and acute lung injury (20–22)
by pinpointing the lung epithelium as a critical site of
action.
One key mechanistic insight is the coupling of
Piezo1-mediated Ca2+ entry to the activation of
calcineurin activity (23) and the
subsequent nuclear translocation of NFATc3. The moderate
Ca2+ selectivity of Piezo1 appears uniquely suited to
activate calcineurin, a Ca2+/calmodulin-dependent
phosphatase that requires sustained, intermediate-level
Ca2+ signals for its activation (24). This specificity explains why the
selective inhibition of Piezo1 was so effective in abrogating the
inflammatory response, as it disrupted this precise signalling
node. Notably, among the NFAT isoforms, NFATc3 has emerged as the
primary responder to mechanical stretch in epithelial cells. This
isoform specificity is notable, as it identifies a distinct
transcriptional regulator in epithelial mechanoinflammation,
differing from the canonical roles of NFATc1/c2 in immune cells
(25,26); accordingly, the present data
highlight NFATc3 as a key mediator of proinflammatory responses in
mechanically stressed epithelial cells. Both Piezo1 knockdown and
calcineurin inhibitors completely blocked NFATc3 nuclear
translocation, and the production of TNF-α, IL-6 and IL-1β,
supporting a linear signalling cascade. These cytokines are
established NFAT target genes, and their coordinated upregulation
provides a direct molecular explanation for the inflammatory milieu
characteristic of VILI.
It is important to contextualize the present
findings within the broader literature, which presents a nuanced
view of the role of Piezo1 in barrier regulation. For example,
Zhong et al (21) suggested
that endothelial Piezo1 serves a protective role in maintaining
vascular barrier function under alveolar stretch. This apparent
contradiction may reflect cell type-specific signalling outcomes,
differences in the magnitude or pattern of mechanical stress, or
the engagement of distinct downstream effectors. The present data
firmly establish an injurious role for epithelial Piezo1 in the
context of HTV-induced VILI, underscoring the complexity of
mechanosignalling in multicellular organ responses.
From a therapeutic perspective, the protective
effects of both genetic and pharmacological Piezo1 inhibition
highlight its potential as a novel target. GsMTx4, a selective
Piezo1 blocker (27), effectively
mitigated injury, suggesting that locally administered Piezo1
inhibitors (such as via nebulization) could serve as a viable
adjunctive therapy to lung-protective ventilation. Targeting Piezo1
may offer distinct advantages over broad-spectrum calcium channel
blockers because of its restricted expression in specific cell
types and its activation mechanism, which is independent of
membrane potential, allowing for more selective disruption of
pathological mechanosignalling. Furthermore, the current data
suggested that targeting the downstream effectors calcineurin and
NFATc3 may achieve comparable protection. While the systemic use of
calcineurin inhibitors is limited by immunosuppression and toxicity
(28), their local delivery to the
lungs via inhalation could harness their anti-inflammatory benefits
while minimizing systemic side effects (29), a strategy worthy of future
exploration.
Despite these advances, several intriguing questions
remain. First, the mechanism underlying the rapid transcriptional
upregulation of Piezo1 itself by mechanical stretch is unknown. The
Hippo pathway effectors YAP and TAZ, which are mechanosensitive
transcriptional coactivators, are prime candidates, as they have
been shown to bind and regulate the Piezo1 promoter in other
contexts (30). Second, the
spatial organization of this signalling pathway is likely crucial.
It is plausible that Piezo1 channels form signalling nanodomains
with calcineurin and possibly other partners near the plasma
membrane, facilitating efficient and specific Ca2+
transfer. Super-resolution imaging techniques could be employed to
visualize this potential ‘mechanotransducosome’. Third, the clear
preservation of the F-actin cytoskeleton in Piezo1-knockdown cells
indicates a dual role for Piezo1 in coordinating both inflammation
and structural remodelling. This crosstalk may be mediated through
Ca2+-dependent proteases such as calpains, which can
cleave cytoskeletal and junctional proteins, and their interplay
with integrins warrants further investigation (31). Finally, the relevance of this
pathway in human biology must be firmly established. While murine
models are invaluable, human lung epithelia may exhibit differences
in mechanosensitivity. Validating these findings in more complex
human systems, such as primary human alveolar epithelial cells,
lung organoids or precision-cut lung slices, will be a critical
step towards clinical translation.
The current management of VILI relies predominantly
on limiting tidal volumes and plateau pressures (32). However, VILI still develops in a
notable subset of patients, indicating an unmet need for
mechanistically targeted therapies (33). The present study indicated that
Piezo1 and its downstream signalling molecules are potential
targets for pharmacological intervention. Monitoring soluble Piezo1
levels in BALF or serum could be explored as a ‘mechanobiological
biomarker’ to identify patients at high risk for or in the early
stages of VILI. Therapeutically, repurposing existing drugs such as
calcineurin inhibitors for inhaled delivery, or developing novel
Piezo1 antagonists such as optimized versions of GsMTx4, represent
promising options. Future research should focus on optimizing the
delivery, efficacy and safety of such targeted anti-mechanosensing
strategies to complement existing ventilatory protocols and improve
outcomes for patients requiring life-supporting MV.
Acknowledgements
Not applicable.
Funding
This work was supported by the Scientific Research Project of
Hubei Provincial Health Commission (grant no. WJ2023M123).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
ML, SLZ and FY performed the experiments. ML and SLZ
analysed and interpreted the data. DF contributed to the conception
and design of the study and participated in data interpretation. ML
and SLZ wrote the original draft. ML, SLZ, FY and DF revised the
original draft. DF secured funding. All authors read and approved
the final manuscript. ML and DF confirm the authenticity of all the
raw data.
Ethics approval and consent for
participation
The animal experiments were approved by the Ethics
Committee of the Traditional Chinese and Western Medicine Hospital
of Wuhan, Tongji Medical College, Huazhong University of Science
and Technology (approval no. 2024-103). All procedures adhered to
the Guide for the Ethical Review of Animal Welfare (GB/T
35892-2018).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Liu G, Dong BB, Ding ZH, Lan C, Zhu CJ and
Liu Q: Unphysiological lung strain promotes ventilation-induced
lung injury via activation of the PECAM-1/Src/STAT3 signaling
pathway. Front Pharmacol. 15:14697832025. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bates JHT, Nieman GF, Kollisch-Singule M
and Gaver DP: Ventilator-induced lung injury as a dynamic balance
between epithelial cell damage and recovery. Ann Biomed Eng.
51:1052–1062. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Spassov SG, Kessler C, Jost R and Schumann
S: Ventilation-like mechanical strain modulates the inflammatory
response of BEAS2B epithelial cells. Oxid Med Cell Longev.
2019:27697612019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Slutsky AS and Ranieri VM:
Ventilator-induced lung injury. N Engl J Med. 369:2126–2136. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dolinay T, Himes BE, Shumyatcher M,
Lawrence GG and Margulies SS: Integrated stress response mediates
epithelial injury in mechanical ventilation. Am J Respir Cell Mol
Biol. 57:193–203. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang Y, Fang X, Yang Y, Chen L, Xiong W,
Song L, Li B, Zhou T, Yu Y, Yang X, et al: Death-associated protein
kinase 1 promotes alveolar epithelial cell apoptosis and
ventilator-induced lung injury through P53 pathway. Shock.
57:140–150. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Englert JA, Macias AA, Amador-Munoz D,
Pinilla Vera M, Isabelle C, Guan J, Magaoay B, Suarez Velandia M,
Coronata A, Lee A, et al: Isoflurane ameliorates acute lung injury
by preserving epithelial tight junction integrity. Anesthesiology.
123:377–388. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Solis AG, Bielecki P, Steach HR, Sharma L,
Harman CC, Yun S, de Zoete MR, Warnock JN, Toomer SD, Kosenko RA,
et al: Mechanosensation of cyclical force by PIEZO1 is essential
for innate immunity. Nature. 573:69–74. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Fang XZ, Zhou T, Xu JQ, Wang YX, Sun MM,
He YJ, Pan SW, Xiong W, Peng ZK, Gao XH and Shang Y: Structure,
kinetic properties and biological function of mechanosensitive
Piezo channels. Cell Biosci. 11:132021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang J, Jing F, Zhao Y, You Z, Zhang A and
Qin S: Piezo1: Structural pharmacology and mechanotransduction
mechanisms. Trends Pharmacol Sci. 46:752–770. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Fang XZ, Li M, Wang YX, Zhang P, Sun MM,
Xu JX, Yang YY, He YJ, Yu Y, Li RT, et al: Mechanosensitive ion
channel Piezo1 mediates mechanical ventilation-exacerbated
ARDS-associated pulmonary fibrosis. J Adv Res. 53:175–186. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pairet N, Mang S, Fois G, Keck M, Kühnbach
M, Gindele J, Frick M, Dietl P and Lamb DJ: TRPV4 inhibition
attenuates stretch-induced inflammatory cellular responses and lung
barrier dysfunction during mechanical ventilation. PLoS One.
13:e01960552018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li M, Wang Y, Hu Z, Huang S, Chen P, Chen
L, Wu J, Wu Z, Yao S and Yang Y: PTEN-mediated senescence of lung
epithelial cells drives ventilator-induced pulmonary fibrosis.
Theranostics. 15:8360–8376. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
National Research Council, . Guide for the
care and use of laboratory animals. 8th edition. The National
Academies Press; Washington, DC: 2011
|
|
15
|
Li M, Fang XZ, Liu XT, Zheng YF, Xie YB,
Ma XD, Xia Y and Shao DH: Inhibition of calcineurin/NFATc4
signaling attenuates ventilatorinduced lung injury. Mol Med Rep.
21:607–614. 2020.PubMed/NCBI
|
|
16
|
Hong W, Zhi FX, Kun TH, Hua FJ, Huan Ling
L, Fang F, Wen C, Jie W and Yang LC: 6-Gingerol attenuates
ventilator-induced lung injury via anti-inflammation and
antioxidative stress by modulating the PPARγ/NF-κB
signalling pathway in rats. Int Immunopharmacol. 92:1073672021.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using realtime quantitative PCR and
the 2 (−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chaudhry MZ, Borkner L, Kulkarni U,
Berberich-Siebelt F and Cicin-Sain L: NFAT signaling is
indispensable for persistent memory responses of MCMV-specific CD8+
T cells. PLoS Pathog. 20:e10120252024. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wen D, Gao Y, Ho C, Yu L and Zhang Y, Lyu
G, Hu D, Li Q and Zhang Y: Focusing on mechanoregulation axis in
fibrosis: Sensing, transduction and effecting. Front Mol Biosci.
9:8046802022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jiang L, Zhang Y, Lu D, Huang T, Yan K,
Yang W and Gao J: Mechanosensitive Piezo1 channel activation
promotes ventilator-induced lung injury via disruption of
endothelial junctions in ARDS rats. Biochem Biophys Res Commun.
556:79–86. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhong M, Wu W, Kang H, Hong Z, Xiong S,
Gao X, Rehman J, Komarova YA and Malik AB: Alveolar stretch
activation of endothelial Piezo1 protects adherens junctions and
lung vascular barrier. Am J Respir Cell Mol Biol. 62:168–177. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Liang GP, Xu J, Cao LL, Zeng YH, Chen BX,
Yang J, Zhang ZW and Kang Y: Piezo1 induced apoptosis of type II
pneumocytes during ARDS. Respir Res. 20:1182019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li Z, Jiang Q, Wei J, Dang D, Meng Z and
Wu H: Piezo1 promotes the progression of necrotizing enterocolitis
by activating the Ca2+/CaMKII-dependent pathway. Commun Biol.
8:4172025. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Crabtree GR and Olson EN: NFAT signaling:
Choreographing the social lives of cells. Cell. 109:S67–S79. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xu T, Keller A and Martinez GJ: NFAT1 and
NFAT2 differentially regulate CTL differentiation upon acute viral
infection. Front Immunol. 10:1842019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nagai J, Lin J and Boyce JA: Macrophage
P2Y6 receptor signaling selectively activates NFATC2 and suppresses
allergic lung inflammation. J Immunol. 209:2293–2303. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang W, Huang M, Huang X, Ma K, Luo M and
Yang N: GsMTx4-blocked PIEZO1 channel promotes myogenic
differentiation and alleviates myofiber damage in Duchenne muscular
dystrophy. Skelet Muscle. 15:132025. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Diker Cohen T, Dotan I, Calvarysky B and
Robenshtok E: Endocrine effects of long-term calcineurin inhibitor
use in solid organ transplant recipients. Eur J Endocrinol.
193:R1–R16. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Neurohr C, Kneidinger N, Ghiani A,
Monforte V, Knoop C, Jaksch P, Parmar J, Ussetti P, Sole A,
Müller-Quernheim J, et al: A randomized controlled trial of
liposomal cyclosporine A for inhalation in the prevention of
bronchiolitis obliterans syndrome following lung transplantation.
Am J Transplant. 22:222–229. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hasegawa K, Fujii S, Matsumoto S, Tajiri
Y, Kikuchi A and Kiyoshima T: YAP signaling induces Piezo1 to
promote oral squamous cell carcinoma cell proliferation. J Pathol.
253:80–93. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Nourse JL and Pathak MM: How cells channel
their stress: Interplay between Piezo1 and the cytoskeleton. Semin
Cell Dev Biol. 71:3122017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Acute Respiratory Distress Syndrome
Network, . Brower RG, Matthay MA, Morris A, Schoenfeld D, Thompson
BT and Wheeler A: Ventilation with lower tidal volumes as compared
with traditional tidal volumes for acute lung injury and the acute
respiratory distress syndrome. N Engl J Med. 342:1301–1308. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tonetti T, Vasques F, Rapetti F, Maiolo G,
Collino F, Romitti F, Camporota L, Cressoni M, Cadringher P,
Quintel M and Gattinoni L: Driving pressure and mechanical power:
New targets for VILI prevention. Ann Transl Med. 5:2862017.
View Article : Google Scholar : PubMed/NCBI
|




















