Introduction
Phyllodes tumor (PT) is a rare type of breast tumor,
accounting for less than 1% of benign and malignant breast tumors
(1). PT is classified as benign,
borderline or malignant, with approximately 10% of PT being
malignant. Malignant transformation of PT usually occurs in the
stromal component, and is rare in the epithelial component. The
occurrence of PT and BC involves a twofold pattern: a separate
coexistence within an ipsilateral or contralateral breast, and BC
occurring in PT. The incidence of breast carcinoma (BC) in PT is
thought to be only 1–2% of all PTs (2,3). To
the best of our knowledge, 27 PT cases with 28 BCs have been
reported in the literature, and 15 out of the 28 BCs were reported
to be carcinoma in situ (CIS) (4–29).
A 53-year-old female with a benign PT with a ductal
carcinoma in situ (DCIS) within the tumor was evaluated. The
literature available was also reviewed.
Materials and methods
Patient
A firm, painless, well-demarcated tumor in the left
breast, measuring 4–5 cm, was found in a 53-year-old female
patient. Over the course the previous 14 years, she underwent
excision of a breast tumor four times at the same site in the left
breast. The pathological diagnosis of the first tumor was a
fibroadenoma (FA), and those of the following three tumors were
benign PTs. The tumor was the 5th one noted in the 14 years
following the previously recorded surgeries.
A firm tumor with a diameter of 3.5 cm was located
beneath the scar from the previous surgery, just above the nipple
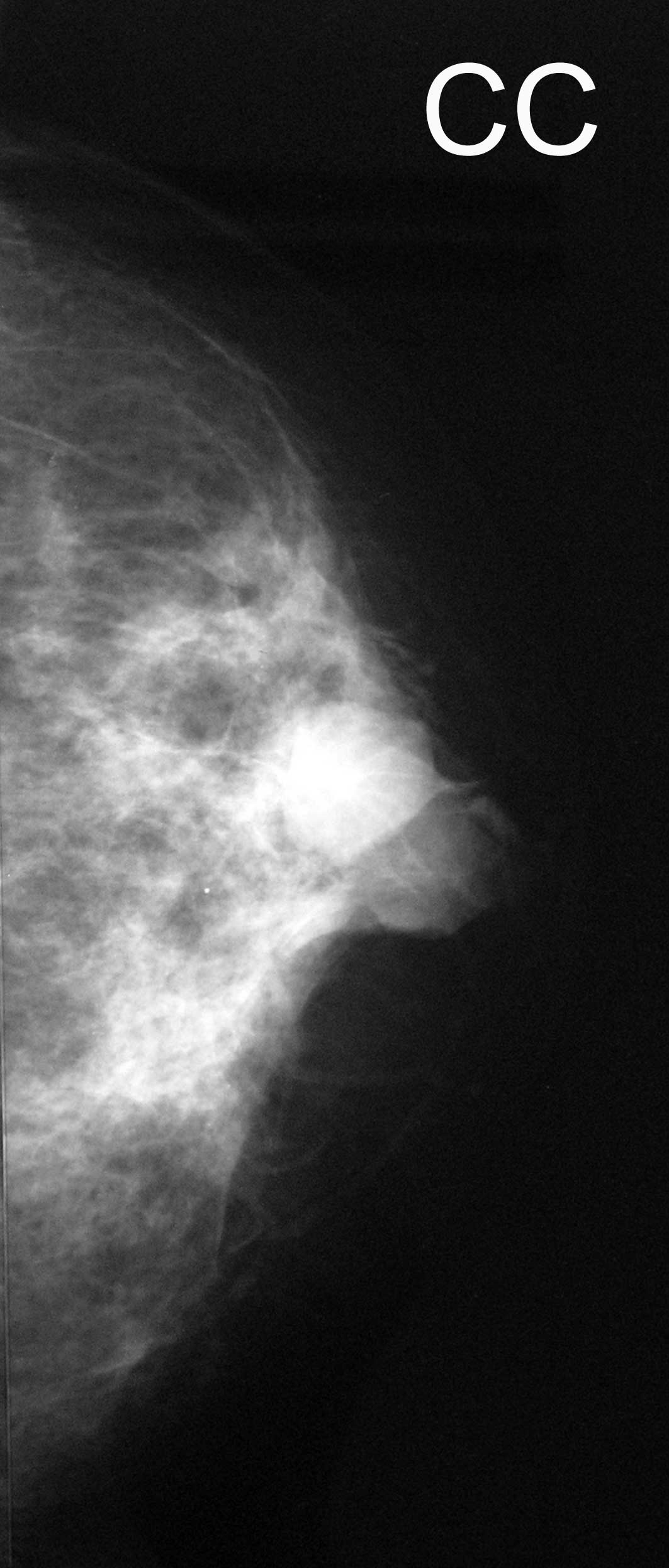
of the left breast. Mammography revealed a high-density
irregularly-shaped mass with a clear margin (Fig. 1), and an ultrasound showed low but
heterogeneous echogenicity (Fig.
1). A computed tomography (CT) scan displayed a well-defined
enhanced tumor (Fig. 1). The image
examinations were compatible with recurrent PT. Fine-needle
aspiration (FNA) cytology revealed that the tumor was likely a
benign FA.
Surgery
The patient underwent local excision (LoEx) with a
1.0 cm margin from the tumor edge. The firm, attached scar tissue
was also resected.
Results
Macroscopic findings
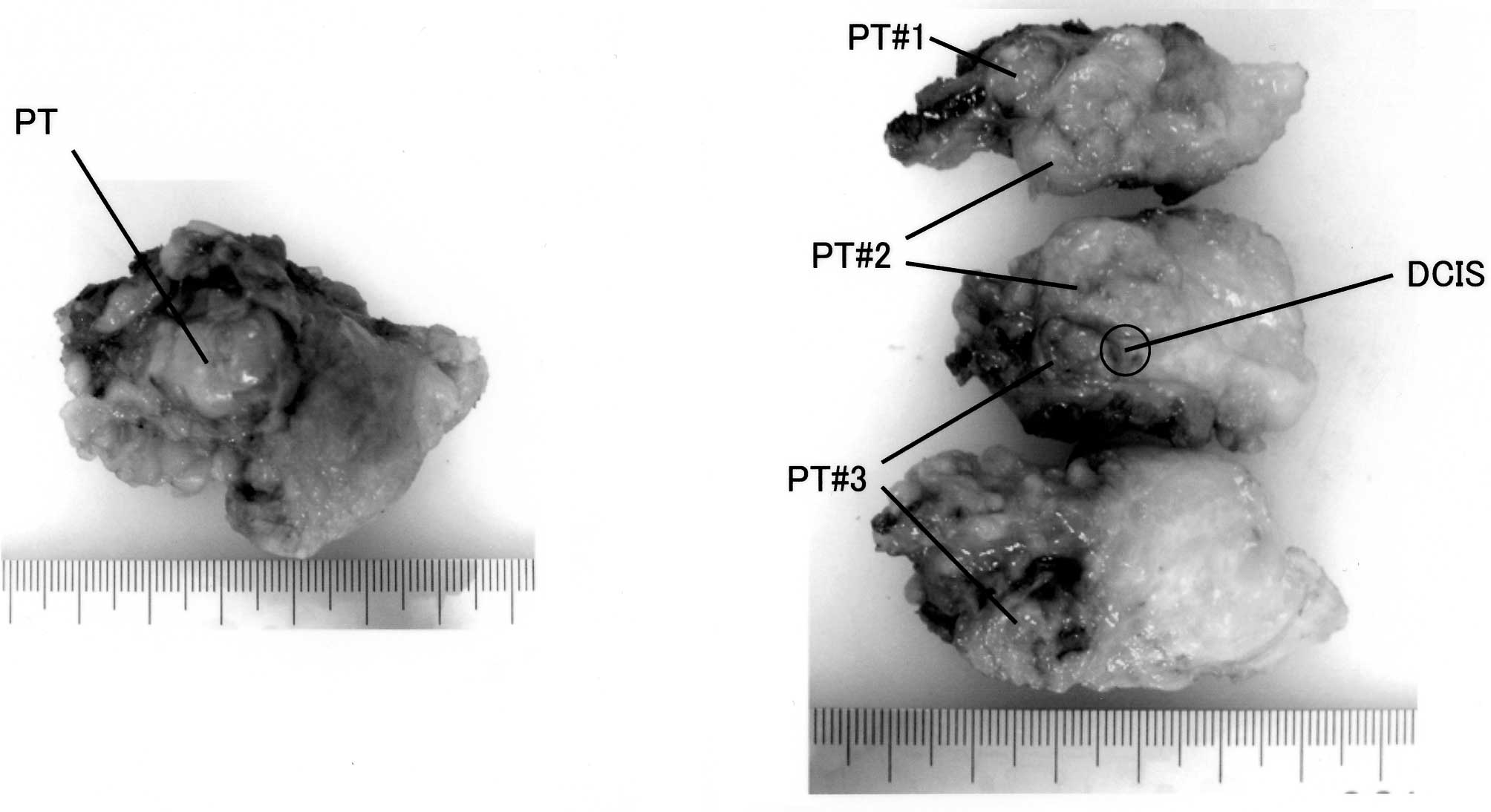
The macroscopic examination revealed a hard elastic
mass, which was encapsulated by thin fibrous tissue and which
adhered firmly to the adjacent scar tissue. The tumor comprised 3
discrete PT nodules (Fig. 2). The
overall size of the PT was 3.5×3.0×2.7 cm.
Microscopic findings
A histopathological examination showed that the
tumor had two components; epithelium and stroma. The stroma
consisted of monotonous, uniform and spindle-shaped tumor cells
without atypia or mitosis, indicating that the tumor was benign
(Fig. 3). The epithelium lined the
elongated ductal structures or leaf-like processes protruding into
dilated ducts formed by the overgrowth of the stromal component.
The epithelium lining the ducts also exhibited benign features in
the majority of areas, but, in part, showed significant nuclear
atypia and a prominent proliferation in a cribiform pattern,
definite features of low to intermediate grade DCIS. The DCIS was
~5 mm in diameter (Fig. 3).
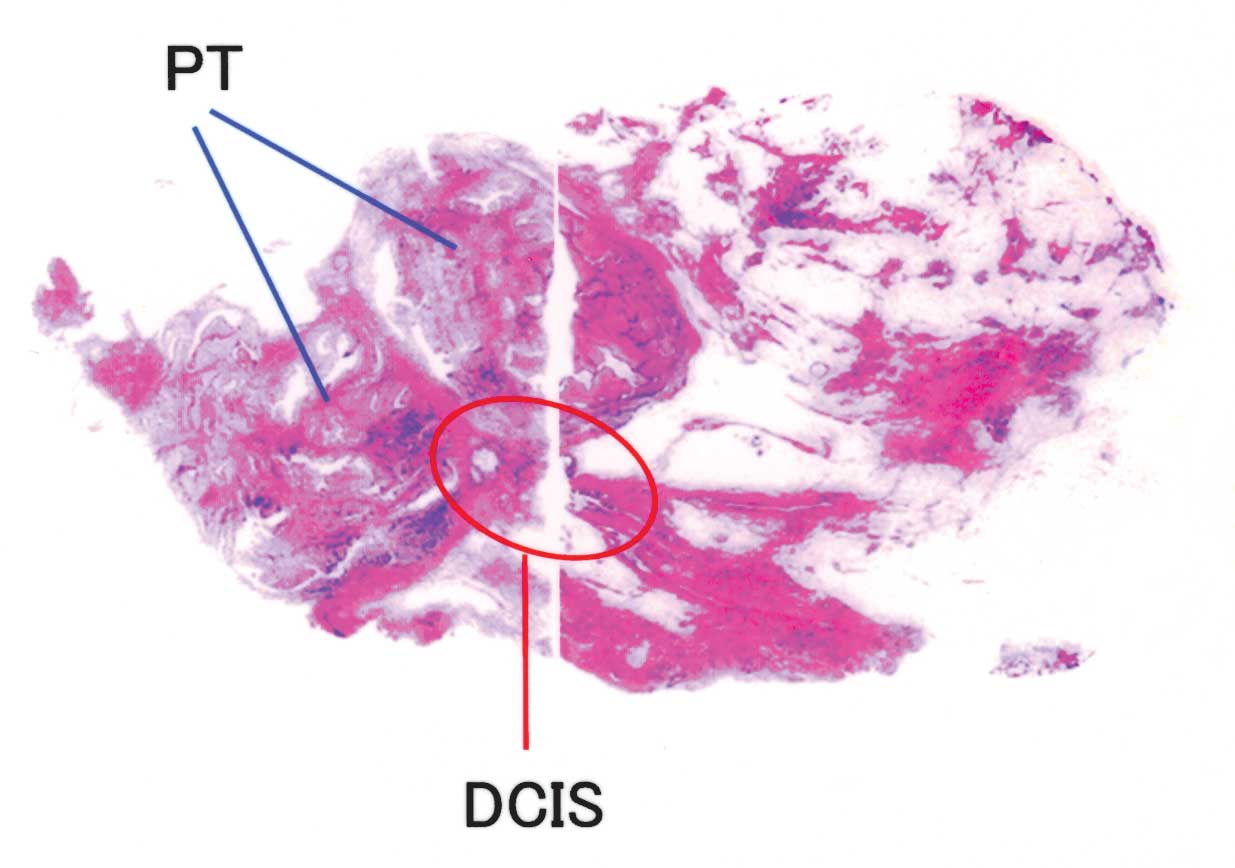 | Figure 3(A) Panoramic view of the microscopic
features. (B) PT lesion; the benign stromal component consisted of
monotonous, uniform and spindle-shaped tumor cells without atypia
or mitosis. H&E stain, magnification, × 100. (C) PT, the
epithelial component lining the ductal structure shows benign
features in the majority of areas of PT lesions. H&E stain,
magnification, × 400. (D) DCIS lesion with a cribiform pattern of 5
mm in diameter. H&E stain; magnification, × 100. (E) DCIS
lesion. H&E stain, magnification, × 400. PT, phyllodes tumor;
DCIS, ductal carcinoma in situ. |
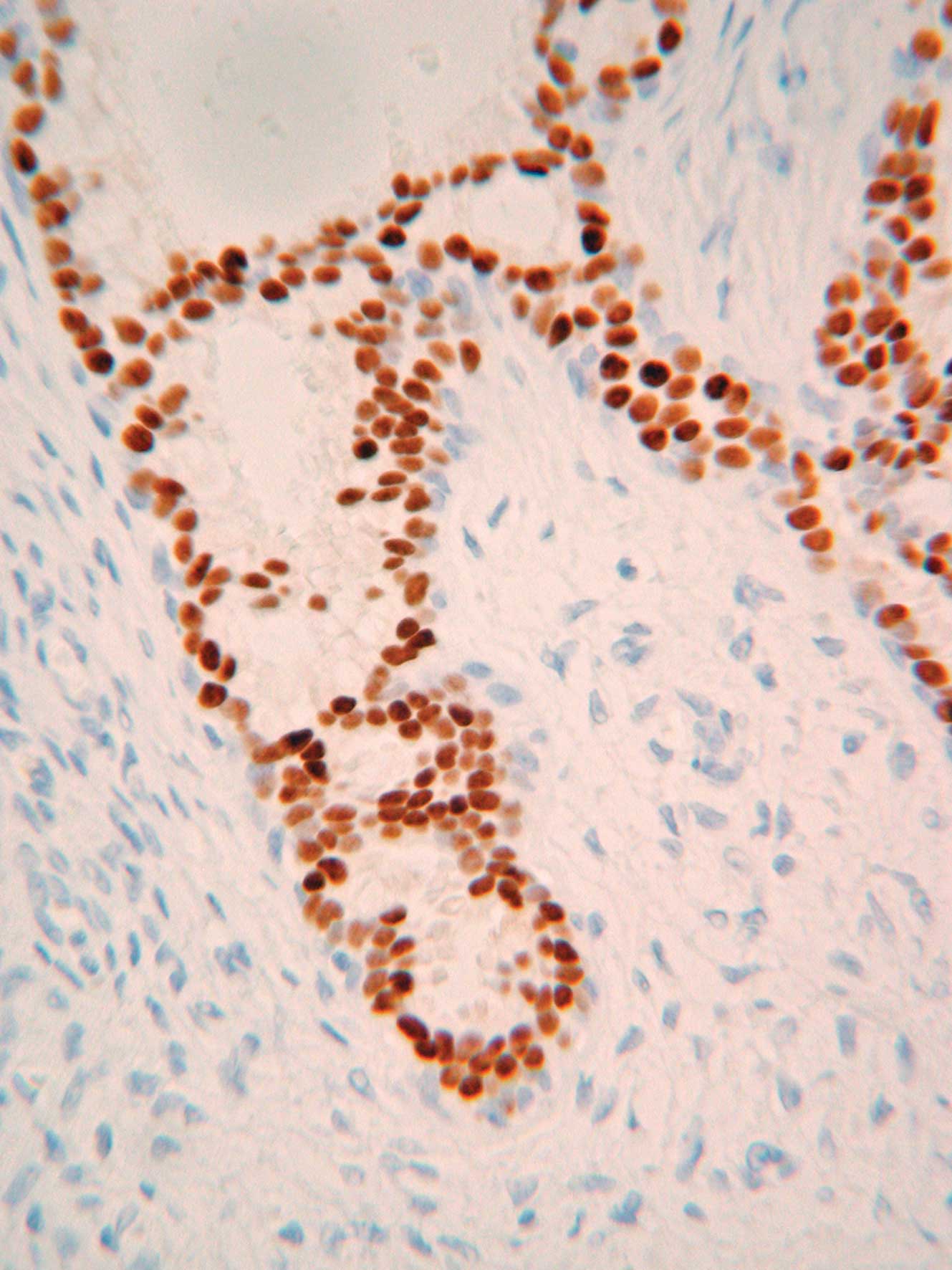
Fig. 4 shows the
results of immunohistochemical staining. The DCIS cells were
strongly positive for estrogen (ER) and progesterone receptors
(PgR), but HER2 expression was negative (score 0).
Post-surgical course
The patient received local irradiation (50 Gy)
following surgery and no evidence of recurrence or metastasis was
detected in the 2 years following surgery.
The previous cases are shown in Table I. A total of 1 patient had 2 BCs in
2 PTs. The first BC was a DCIS and the second one was a tubular
carcinoma (5). The patient ages
ranged between 26 and 80 years (average 52.7). Of the 28 PTs, 12
cases were identified as malignant and 14 as benign, with 1
borderline case. The diameters of the tumors ranged between 2.0 and
21 cm (average 8.0). The combined BCs included 15 CISs, 12 invasive
BCs, and 1 patient had recurrent PTs twice in combination with a
lobular carcinoma in situ (LCIS) at the first recurrence and
a tubular carcinoma at the second recurrence. A total of 15 CISs
included 10 DCISs and 3 LCISs, and 2 cases presented with both DCIS
and LCIS. The CIS sizes were not described in 7 cases, but the
majority of the remaining CISs were focal, and the largest CIS was
2.0 cm in diameter. On the other hand, 13 invasive BCs included 7
invasive ductal carcinomas (IDCs), 4 squamous cell carcinomas
(SCCs), 1 invasive lobular carcinoma (ILC) and 1 tubular carcinoma.
The tumor size was not described in 12 BC cases, and in 1 case the
diameter was 2.5 cm. Axillary lymph nodes (AxLNs) were involved in
2 of 7 invasive BC cases reported, but no lymph nodes (LNs) were
involved in any of the 7 CIS cases described. The LN involvement
noted was metastasized from BCs, but not from PTs.
 | Table INon-invasive carcinoma within
phyllodes tumor. |
Table I
Non-invasive carcinoma within
phyllodes tumor.
| No. | Author | Year | Age | Surgery | AxDx | PT | CIS | Refs. |
|---|
| | | | | |
|
| |
|---|
| | | | | | Type | Size (cm) | LNI | Type | Size (cm) | LNI | |
|---|
| 1 | Seemayer et
al | 1975 | 27 | MX | (−) | Malignant | 6.0 | | DCIS | Focal | | 4 |
| 2 | Leong et
al | 1980 | 49 | LoEx | (−) | Benign | 6.0 | | LCIS | - | | 5 |
| 3 | Cole-Beuglet et
al | 1983 | 55 | LoEx | (−) | Benign | 3.5 | | DCIS+LCIS | - | | 6 |
| 4 | Grove et
al | 1986 | 71 | MX | (+) | Benign | 19.0 | (−) | DCIS | 2.0 | (−) | 7 |
| 5 | Ward et
al | 1986 | 55 | MX | - | Benign | 4.0 | | LCIS | Focal | | 8 |
| 6 | Knudsen et
al | 1987 | 71 | MX | (+) | Benign | 7.0 | (−) | DCIS+LCIS | Multi-focal | (−) | 9 |
| 7 | De Rosa et
al | 1989 | 77 | MX | (+) | Benign | 5.0 | (−) | DCIS | 0.3 | (−) | 10 |
| 8 | Schwickerath et
al | 1992 | 47 | MX | (+) | Malignant | 2.0 | (−) | DCIS | - | (−) | 11 |
| 9 | Padmanabhan et
al | 1997 | 47 | MX | (+) | Malignant | 7.5 | (−) | LCIS | Focal | (−) | 12 |
| 10 | Naresh | 1997 | 51 | LoEx | (−) | Borderline | 14.0 | | DCIS | Focal | | 13 |
| 11 | Nishimura et
al | 1998 | 80 | LoEx | (−) | Malignant | 10.5 | | DCIS | - | | 14 |
| 12 | Alo et
al | 2001 | 39 | MX | - | Malignant | 9.0 | | DCIS | | | 15 |
| 13 | Lim et
al | 2005 | 45 | MX | (−) | Malignant | 12.0 | | DCIS | 0.6 | | 16 |
| 14 | Nomura et
al | 2006 | 75 | MX | (−) | Malignant | 3.5 | | DCIS | - | | 17 |
| 15 | Yamaguchi et
al | 2008 | 54 | MX | (−) | Benign | 15.0 | | DCIS | Focal | | 18 |
| 16 | Present case | - | 53 | LoEx | (−) | Benign | 3.5 | | DCIS | 0.5 | | - |
Discussion
The coexistence of PT and BC includes two patterns;
a separate coexistence within an ipsilateral or contralateral
breast, and a BC arising within PT. The present case was the latter
type, a DCIS arising in a benign PT. To the best of our knowledge,
the literature shows a total of 28 BCs arising in PT in 27
patients.
A variety of therapies were applied to the various
cases. The present case received a LoEx with a margin of 1 cm from
the tumor edge, and local irradiation at a total dose of 50 Gy
since the PT tumors locally recurred four times at the same site.
Two years have elapsed since the previous surgery and the patient
remains disease-free. The additional surgeries included 5 LoExs, 21
mastectomies (MXs), 3 no descriptions, AxLNs were dissected in 10
cases, 12 patients received no Ax dissection (Dx) and 6 were not
described. It appears that MX or LoEx was selected according to the
size of the PT. MX with AxDx was applied for large PTs, but LoEx
was applied for small PTs. Overall, 4 cases received LoEx first and
then MX again according to the pathological diagnosis of malignancy
or combination with invasive BC (10,12,20,23).
AxDx was also applied for large PTs, but no LNs involving PTs were
noted in the 10 cases described. AxDx may thus be restricted to
patients suspected of having LN involvement by image diagnosis.
Post-surgical radiotherapy (RT), chemotherapy and/or
endocrine therapy were applied for large PTs or those with combined
invasive BCs. Overall, 1 patient with a 15.5 cm malignant PT with
SCC of the breast received chemotherapy with cyclophosphamide,
methotrexate and 5-fluorouracil (CMF) following surgery (22), and 1 patient with a benign PT with a
diameter of 15 cm received chemotherapy with oral Tegafur for 2
years following surgery. This second patient has been disease-free
for 5 years (23). A patient with a
3.3 cm benign PT with AxLN involving a combined invasive BC
received chemotherapy with cyclophosphamide, epirubicin and
5-fluorouracil (CEF) and local RT following surgery, followed by
tamoxifen (TAM), and was disease-free for 3 years (25). A patient with a 21 cm malignant PT
with LN involving combined BC received chemotherapy with 4-cycle
adriamycin and cyclophosphamide (AC), local RT and TAM, and was
disease-free for 11 months (28). A
patient with a 12 cm benign PT received TAM alone since the
combined DCIS was ER- and PgR-positive, and remained disease-free
for 1 year (18). A patient with a
recurring benign PT combined with an invasive BC received local RT
at 5500 rad following surgery, similar to the present case, and
this patient was disease-free for 21 months (5). Notably, a patient was diagnosed with
SCC of the breast by FNA biopsy, and post-surgical pathology showed
an SCC arising in malignant PT. The patient received preoperative
chemotherapy with FEC followed by PTX. However, no post-surgical
chemotherapy was administered, and the patient succumbed to PT lung
metastasis 40 months following surgery (27). Since no standard therapy for PT has
been established, a variety of combinations of surgery,
chemotherapy, endocrine therapy and/or RT are applied.
The patient outcomes were described in 12 cases, and
the majority of these patients were followed for a number of months
or years. A total of 2 patients survived for 5 years following
surgery (16,23). A total of 2 patients succumbed to
the disease, 1 is mentioned above (27) and 1 developed lung metastasis and
succumbed 10 months following surgery (14).
In conclusion, a rare case of a DCIS arising in
benign PT is reported. Various types of carcinoma have been
reported to arise in PT, such as IDC, ILC, DCIS, LCIS, SCC and
tubular carcinoma. The etiological relationship between PT and
carcinoma has yet to be elucidated. This type of combination
therefore remains to be investigated.
Abbreviations:
|
BC
|
breast carcinoma
|
|
IDC
|
invasive ductal carcinoma
|
|
SCC
|
squamous cell carcinoma
|
|
PT
|
phyllodes tumor
|
|
CIS
|
carcinoma in situ
|
|
MX
|
mastectomy
|
|
LoEx
|
local excision
|
|
LNI
|
lymph node involvement
|
|
AxDx
|
axillary dissection
|
|
DCIS
|
ductal carcinoma in situ
|
|
LCIS
|
lobular carcinoma in situ
|
References
|
1
|
Tavassoli FA and Eusebi V: Tumors of the
mammary gland. Atlas of Tumor Pathology, 4th Series. Armed Forces
Institute of Pathology; Washington, DC: pp. 323–333. 2009
|
|
2
|
Rosen PP and Urban JA: Coexistent mammary
carcinoma and cystosarcoma phyllodes. Breast. 1:9–15. 1975.
|
|
3
|
Ozzello L and Gump FE: The management of
patients with carcinomas in fibroadenomatous tumors of the breast.
Surg Gynecol Obstet. 160:99–104. 1985.PubMed/NCBI
|
|
4
|
Seemayer TA, Tremblay G and Shibata H: The
unique association of mammary stromal sarcoma with intraductal
carcinoma. Cancer. 36:599–605. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Leong AS and Meredith DJ: Tubular
carcinoma developing within a recurring cystosarcoma phyllodes of
the breast. Cancer. 46:1863–1867. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cole-Beuglet C, Soriano R, Kurtz AB, Meyer
JE, Kopans DB and Goldberg BB: Ultrasound, x-ray mammography, and
histopathology of cystosarcoma phylloides. Radiology. 146:481–486.
1983. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Grove A and Kristensen LD: Intraductal
carcinoma within a phyllodes tumor of the breast: a case report.
Tumori. 72:187–190. 1986.PubMed/NCBI
|
|
8
|
Ward RM and Evans HL: Cystosarcoma
phyllodes. A clinicopathologic study of 26 cases. Cancer.
58:2282–2289. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Knudsen PJT and Ostergaard J: Cystosarcoma
phylloides with lobular and ductal carcinoma in situ. Arch Pathol
Lab Med. 111:873–875. 1987.PubMed/NCBI
|
|
10
|
De Rosa G, Ferrara G, Goglia P, Ghicas C
and Zeppa P: In situ and microinvasive carcinoma with squamoid
differentiation arising in a phyllodes tumor: report of a case.
Tumori. 75:514–517. 1989.PubMed/NCBI
|
|
11
|
Schwickerath J, Blessing MH and Wolff E:
Seltene Erscheinungsform eines Kombinationstumors aus Cystosarcoma
phylloides malignum und eines intraduktalen Karzinoms. Geburtsh u
Frauenheilk. 52:557–559. 1992. View Article : Google Scholar
|
|
12
|
Padmanabhan V, Dahlstrom JE, Chomg GC and
Bennett G: Phyllodes tumor with lobular carcinoma in situ and
liposarcomatous stroma. Pathology. 29:224–226. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Naresh KN: Cancerization of phyllodes
tumour. Histopathology. 30:98–99. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nishimura R, Hasebe T, Imoto S and Mukai
K: Malignant phyllodes tumour with a noninvasive ductal carcinoma
component. Virchows Arch. 432:89–93. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Alo PL, Andreano T, Monaco S, Sebastiani
V, Eleuteri Serpieri D and Di Tondo U: Tumore filloide maligno
della mammella con aspetti di carcinoma intraduttale. Pathologica.
93:124–127. 2001.PubMed/NCBI
|
|
16
|
Lim M and Tan PH: Ductal carcinoma in situ
within phyllodes tumour: a rare occurrence. Pathology. 37:393–396.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Nomura M, Inoue Y, Fujita S, Sakao J,
Hirota M, Souda S and Ohshima M: A case of non-invasive ductal
carcinoma arising in malignant phyllodes tumor. Breast Cancer.
13:89–94. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yamaguchi R, Tanaka M, Kishimoto Y, Ohkuma
K, Ishida M and Kojiro M: Ductal carcinoma in situ arising in a
benign phyllodes tumor: report of a case. Surg Today. 38:42–45.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bassermann R: Cystosarcoma phyllodes
mammae und doppelseitiges Mammakarcinom. Pathologe. 1:155–158.
1980.
|
|
20
|
Klausner JM, Leleuk S, Ilia B, Inbar M,
Hammer B, Skornik Y and Rozin RR: Breast carcinoma originating in
cystosarcoma phyllodes. Clin Oncol. 9:71–74. 1983.PubMed/NCBI
|
|
21
|
Ishida T, Izuo M and Kawai T: Breast
carcinoma arising in cystosarcoma phyllodes: report of a case with
a review of the literature. Jpn J Clin Oncol. 14:99–106.
1984.PubMed/NCBI
|
|
22
|
Hunger E, Turk R and Wurster K: Malignes
Cystosarcoma plylloides und Plattenepithelkarzinom der Mamma. Eine
seltene Tumorkombination Geburtsh u Frauenheilk. 44:640–642. 1984.
View Article : Google Scholar
|
|
23
|
Yasumura T, Matsui S, Hamajima T,
Nagashima K, Yamagishi H, Aikawa I and Oka T: Infiltrating ductal
carcinoma developing within cystosarcoma phyllodes – a case report.
Jpn J Surg. 18:326–329. 1988.
|
|
24
|
Kodama T, Kameyama K, Mukai M, Sugiura H,
Ikeda T and Okada Y: Invasive lobular carcinoma arising in
phyllodes tumor of the breast. Virchows Arch. 442:614–616.
2003.PubMed/NCBI
|
|
25
|
Parfitt JR, Armstrong C, O'Malley F, Ross
J and Tuck AB: In-situ and invasive carcinoma within a phyllodes
tumor associated with lymph node metastases. World J Sur Oncol.
2:462004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ramdass MJ and Dindyal S: Phyllodes breast
tumour showing invasive squamous-cell carcinoma with invasive
ductal, clear-cell, secretory, and squamous components. Lancet
Oncol. 7:8802006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sugie T, Takeuchi E, Kunishima F,
Yatsumoto F and Kono Y: A case of ductal carcinoma with squamous
differentiation in malignant phyllodes tumor. Breast Cancer.
14:327–332. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Korula A, Varghese J, Thomas M, Vyas F and
Korula A: Malignant phyllodes tumour with intraductal and invasive
carcinoma and lymph node metastasis. Singapore Med J. 49:318–321.
2008.PubMed/NCBI
|
|
29
|
Macher-Goeppinger S, Marme F, Goeppert B,
Penzel R, Schirmacher P, Sinn HP and Aulmann S: Invasive ductal
breast cancer within a malignant phyllodes tumor: case report and
assessment of clonality. Hum Pathol. 41:293–296. 2010. View Article : Google Scholar : PubMed/NCBI
|