Introduction
Laparoscopy has become an attractive approach in
surgical treatment in gynecologic oncology. Various studies have
been published demonstrating the feasibility and safety of this
technique. Its main advantages over laparotomy are less blood loss
and a shorter length of postoperative hospitalization and earlier
recovery, with a substantial equivalence in terms of the number of
lymph nodes removed. From these advantages, laparoscopy is
considered to be minimally invasive surgery. However, few authors
have objectively evaluated how invasive these two methods of
surgery are.
This prospective clinical study was performed to
evaluate whether the trauma is less in laparoscopic surgery
compared to laparotomy when dealing with cervical cancer. The
effects of proinflammatory cytokine interleukin-6 (IL-6),
C-reactive protein (CRP) and cortisol were analyzed for the two
groups. This study, therefore, evaluates the effects of
laparoscopic and conventional cervical surgery on specific and
non-specific responses following major abdominal surgery.
In this study, we aimed to evaluate the extent of
surgical trauma after laparoscopic and open radical hysterectomy
and lymphadenectomy by comparing the clinical data and examining
serum values of IL-6, CRP and cortisol to clarify whether patients
benefit from the laparoscopic technique and whether it is a
minimally invasive approach.
Materials and methods
Patient groups
Data were prospectively collected and recorded from
63 patients who underwent laparoscopic radical hysterectomy with
lymphadenectomy and conversional open surgery for stage IA-IIB
cervical cancer between May 2009 and July 2010. Age, body mass
index, site and type of malignancy, and the International
Federation of Gynecology and Obstetrics (FIGO) staging were noted.
Surgery was performed at the Department of Gynecologic Oncology at
the PLA General Hospital in Beijing, China. Inclusion criteria
were: good general condition, stage IA-IIB cancer, and no evidence
of lymph node metastases in imaging study (MRI and/or CT and/or
PET). A total of 9 patients accepted preoperative intra-arterial
chemotherapy. Patients were staged based on clinical evaluation
according to the most recent FIGO clinical staging system.
The patients were informed of risks and possible
complications of the surgical procedure and provided written
informed consent. The patients received a bowel preparation and a
3-dose antibiotic prophylaxis prior to the operation.
Laparoscopic surgery was performed as a
laparoscopically assisted procedure as described by other authors
(1–3). To minimize the risk of port site
metastases, the lymph nodes were placed into a bag and removed
vaginally at the end of the intervention.
Initially, ureteral bilateral stents were placed
preoperatively, in order to facilitate laparoscopic ureteral
manipulation and minimize the risk of ureteral damage. They were
removed at the first clinical control 30 days after surgery.
The urethral catheter was removed 2 days after the
intervention. Intermittent catheterization was performed three
times a day until residual urine volumes of <50 ml were obtained
at least twice.
Survival and outcomes
Follow-up consisted of a pelvic examination every 3
months during the first two years, three times a year from the
third to the fifth year, and annually thereafter. TCT and HPV tests
were performed every year. X-ray and CT were performed when
necessary.
Samples and assays
Peripheral venous blood samples were collected in
EDTA collection tubes to be analyzed for cytokines IL-6, CRP and
cortisol before surgery, 1 h and 2 h into surgery, and 1, 4 and 7
days after surgery. The samples were centrifuged at 4500 g for 10
min. Serum samples were separated into PE tubes and stored at −80°C
until analysis. Serum IL-6 and CRP were measured using commercially
available sandwich enzyme-linked immunosorbent assays (ELISA kits)
(R&D Systems, Minneapolis, MN, USA). The minimum sensitivity of
the assays was 0.70 pg/ml for IL-6 and 0.01 ng/ml for CRP. The
concentration of cortisol in serum was measured using a
radioimmunoassay (RIA) kit (The North Biological Technology Co.,
Ltd., Beijing, China) with a minimum detectable level of 2
ng/ml.
Statistical analysis
Results are expressed as the means ± SEM.
Statistical differences within each group were determined by
repeated measures of analysis of variance (ANOVA) on ranks,
followed by the Student Newman-Keuls test. The groups were compared
statistically using the Mann-Whitney rank sum test. P<0.05 was
considered to be statistically significant.
Results
Patient and surgical data
Patient data
Between May 2009 and July 2010, 63 patients
underwent laparoscopic radical hysterectomy and pelvic
lymphadenectomy for stage IA-IIB early cervical cancer. Surgery was
performed at the Department of Gynecologic Oncology of the PLA
General Hospital, Beijing, China. All 63 patients were diagnosed by
vaginoscopic biopsy. There was no patient conversion to laparotomy
in the laparoscopy group. The clinical and pathological
characteristics are summarized in Table
I.
 | Table IPatient characteristics. |
Table I
Patient characteristics.
| Laparoscopy
(n1=33) | Laparotomy
(n2=30) |
|---|
| Mean age, years
(range) | 47.55 (27–63) | 44.94 (27–79) |
| Median BMI,
kg/m2 (range) | 23.64±2.72
(18.29–30.85) | 22.72±2.92
(16.8–30.48) |
| Stage |
| IA | 4 | 2 |
| IB | 10 | 10 |
| IIA | 15 | 14 |
| IIB | 4 | 4 |
| Histology |
| Squamous | 25 | 22 |
| Adenocarcinoma | 6 | 6 |
| Adenosquamous | 1 | 1 |
| Other | Neuroendocrine
carcinoma | Clear-cell
carcinoma |
No significant difference was noted in age between
the laparoscopy group (47.55 years old) and the laparotomy group
(44.94 years old, p=0.254). Median BMI was 23.64 kg/m2
and 22.72 kg/m2, respectively (p=0.159). The predominant
histotype was squamous (25 patients in the laparoscopy group and 22
patients in the laparotomy group), followed by adenocarcinoma (6
patients in each group), adenosquamous (1 patient in each group)
and other histotypes (neuroendocrine carcinoma in 1 patient in the
laparoscopy group and clear-cell carcinoma in 1 patient in the
laparotomy group). None of the patients showed MRI or CT evidence
of lymph node involvement.
Surgical data
The surgical data are summarized in Table II. Operative time for the
laparoscopy group was 202±39 min, which differed from the
laparotomy group (128±28 min, p=0.05). The estimated blood loss for
patients undergoing laparoscopy was 317.23±217.20 ml. This blood
loss was significantly less than in the laparotomy group
(872.58±693.16 ml, p<0.0001). Laparoscopy patients had an
average of 19.74 nodes retrieved as compared to 20.35 nodes
retrieved in laparotomy patients. The differences were not
significant (p=0.712). The mean length of postoperative hospital
stay was 11.72 days in the laparoscopy group and 16.42 days in the
laparotomy group (p=0.004).
 | Table IISurgical data. |
Table II
Surgical data.
| Laparoscopy
(n1=33) | Laparotomy
(n2=30) |
|---|
| Lymph nodes
resected | 19.74±7.43
(5–38) | 20.35±6.62
(9–33) |
| Duration, min
(range) | 202±39 (135–258) | 128±28 (117–149) |
| Blood loss, ml
(range) | 317.23±217.20
(100–1000) | 872.58±693.16
(200–3400) |
| Blood transfusion
rate, % | 27.27 | 83.33 |
| Postoperative
exhaust, days (range) | 1.83±0.43 (1–3) | 2.77±0.67 (1–4) |
| Hospital stay, days
(range) | 19.4±5.15 (9–36) | 22.9±10.05
(13–68) |
| Postoperative
hospital stay (range) | 11.72±3.62
(5–23) | 16.42±10.04
(10–63) |
Serum cytokine concentrations
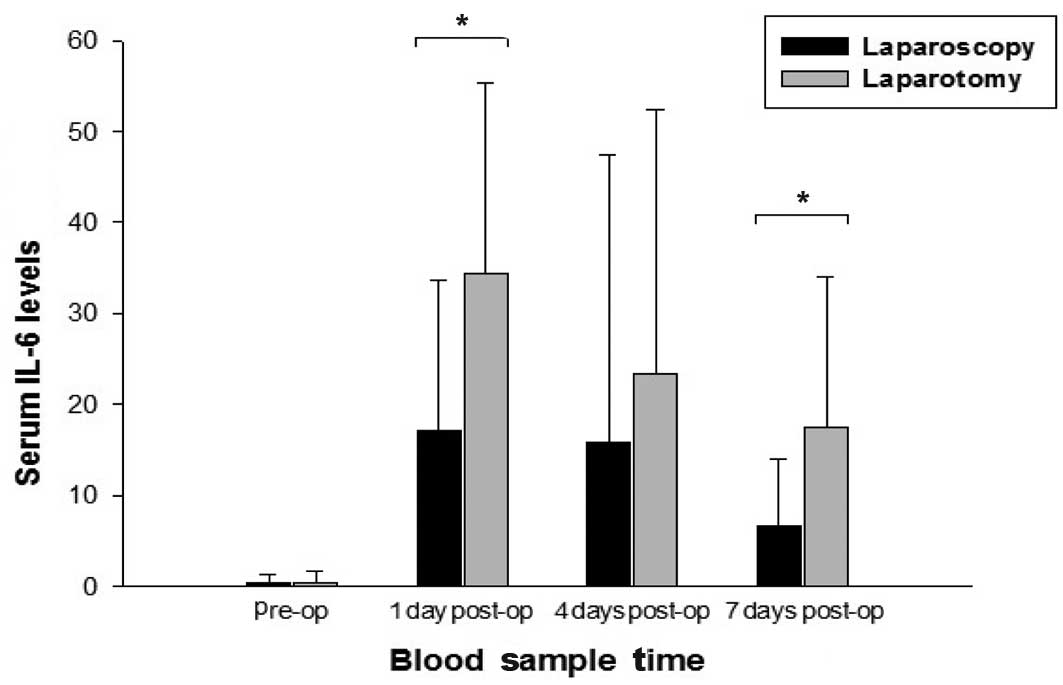
Time course of IL-6 serum levels in
laparoscopic and conventional open surgery
Both laparoscopic and conventional open surgery
caused a significant increase in serum IL-6 levels 1 and 7 days
after the operation (Fig. 1) and
the maximum increase was found 1 day after the operation. A
comparison between the laparoscopy and open surgery groups showed a
significantly less pronounced increase of IL-6 in the laparoscopy
group, which was most evident 1 and 7 days after the operation
(p<0.001). Results noted were: pre-operation: laparoscopy
0.41±0.85 pg/ml and laparotomy 0.40±1.24 pg/ml, p=0.983; day 1:
laparoscopy 17.14±16.53 pg/ml and laparotomy 34.32±20.97 pg/ml,
p<0.001; day 4: laparoscopy 15.89±31.47 pg/ml and laparotomy
23.34±29.03 pg/ml, p=0.334; day 7: laparoscopy 6.7±7.21 pg/ml and
laparotomy 17.54±16.47 pg/ml, p<0.001.
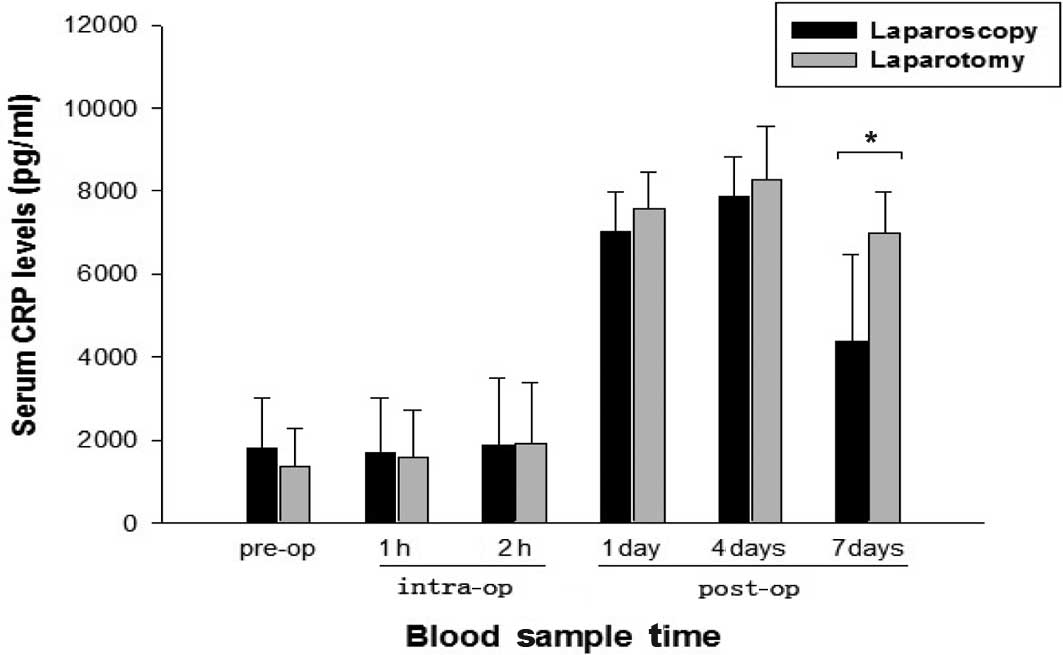
Time course of C-reactive protein
serum levels in lapascopic and conventional open surgery
Daily analysis of serum CRP values over the 7 days
following surgery showed a significant increase after the
laparoscopic and open procedures (Fig.
2). This increase was significantly higher at days 1 and 7 in
conventional surgery patients compared with laparoscopy patients
(p=0.018 and p<0.001, respectively). Results noted were:
pre-operation: laparoscopy 1796.30±1221.30 ng/ml and laparotomy
1365.30±929.57 ng/ml, p=0.123; 1 h: laparoscopy 1684.64±1330.09
ng/ml and laparotomy 1580.64±1126.57 ng/ml, p=0.74; 2 h:
laparoscopy 1889.53±1584.08 ng/ml and laparotomy 1905.52±1478.04
ng/ml, p=0.967; day 1: laparoscopy 7024.72±949.12 ng/ml and
laparotomy 7586.61±869.42 ng/ml, p=0.018; day 4: laparoscopy
7855.67±975.20 ng/ml and laparotomy 8265.33±1310.99 ng/ml, p=0.162;
day 7: laparoscopy 4357.71±2108.85 ng/ml and laparotomy
6967.96±995.02 ng/ml, p<0.001.
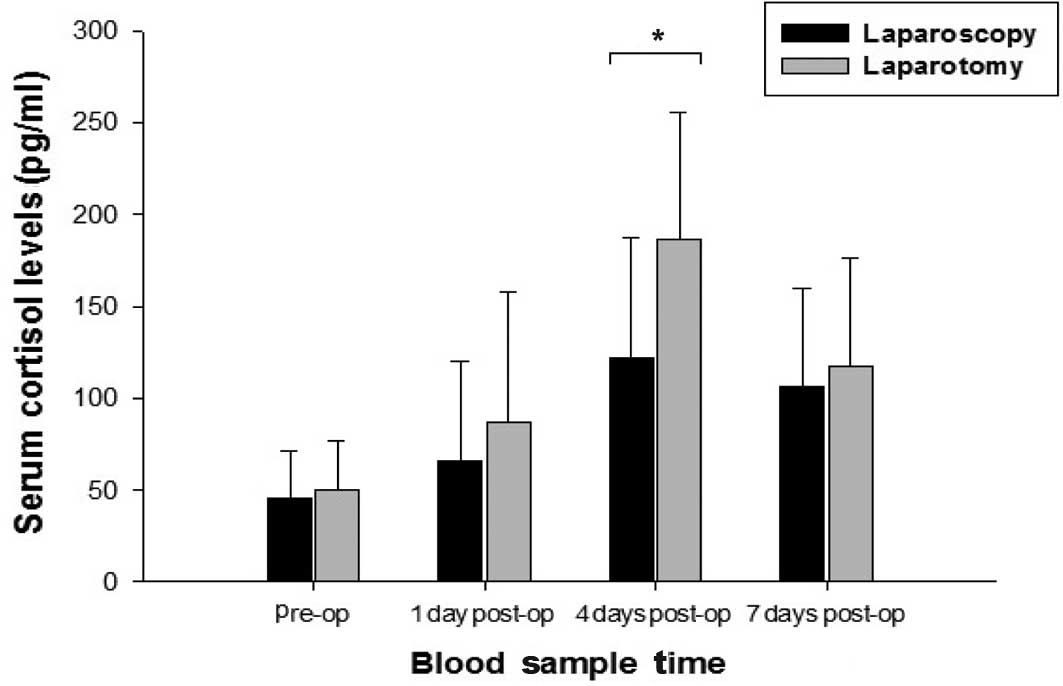
Time course of the cortisol serum
levels in laparoscopic and conventional open surgery
A significant increase of circulating cortisol
levels was observed after both laparoscopic and open surgery
(Fig. 3). This increase was
significantly higher at 4 days after the operation in conventional
surgery patients compared with laparoscopy patients (p<0.001).
Pre-operation: laparoscopy 45.15±26.53 ng/ml and laparotomy
49.63±27.42 ng/ml, p=0.513; day 1: laparoscopy 66.13±54.15 ng/ml
and laparotomy 86.96±70.85 ng/ml, p=0.192; day 4: laparoscopy
122.29±65.17 ng/ml and laparotomy 186.76±68.61 ng/ml, p<0.001;
day 7: laparoscopy 106.50±53.31 ng/ml and laparotomy 116.87±59.49
ng/ml, p=0.468.
Discussion
Research has shown that laparoscopic radical
hysterectomy is feasible and safe and induces less trauma due to
less intraoperative bleeding, less postoperative pain, a shorter
period of recovery and a shorter hospital stay, while the number of
lymph nodes removed was almost equal to that shown in conventional
open surgery (4–14). Laparoscopically assisted vaginal
hysterectomy has become the preferred approach for cervical cancer
treatment. Experimental studies have shown that laparoscopic
surgery has a low impact on cancer growth (15–16).
Mounting evidence shows that cervical cancer recurrence and
survival rates after laparoscopic surgery are comparable with those
observed after laparotomy, despite the results of prospective
randomized trials. Concomitantly, laparoscopy patients have better
cosmetic results and quality of life.
In our study, the mean blood loss was found to be
317 ml (range 100–1000) in the laparoscopy group and 872 ml
(200–3400) in the laparotomy group; a significant difference
(p<0.0001). The mean operative time was 202 min (range 135–258)
in the laparoscopy group and 128 min (range 117–149) in the
laparotomy group (p=0.05). In their study, Renaud et al
(17) reported that the operative
time was 270 min (range 180–360) in laparoscopically assisted
radical vaginal hysterectomy, whereas Spirtos et al
(18) found the operative time to
be 205 min. Recently, Puntambekar et al (19) reported that the operation time for
laparoscopy has been reduced to 92 min (range 65–120). A
standardized, reproducible procedure and a well-trained and skilled
surgeon, together with a competent team, using open surgical
techniques, should contribute towards this relatively challenging
procedure.
Postoperative immune dysfunction is a factor for
patients undergoing surgery for benign as well as malignant tumors
as it affects the rate of infectious complications as well as the
growth of disseminated tumor cells (20). Particularly in patients with cancer,
better preserved postoperative immunity may result in more
favorable long-term oncological results. It was previously reported
that the systemic immune response was better preserved after
laparoscopic surgery as compared to after conventional surgery
(21–22). The acute-phase response is a
favorable indicator of tissue injury in patients (23). Cytokines such as IL-6 and CRP are
short-lived, and previous studies have shown elevated serum levels
after infection, inflammation and trauma. Serum levels were found
to peak 4 to 48 h after injury.
IL-6 is significant in trauma research as studies
have shown that the increase in circulating IL-6 serum levels is
almost proportional to the severity of surgery trauma. Moreover,
IL-6 release may be used to evaluate the impact of injury in the
early stages regardless of the injury pattern (24–26).
In recent years, more research has been conducted on other surgical
procedures and their impact on the immune system (27). Based on these results, this
prospective clinical study was conducted to address the issue of
potential differences in postoperative immunological alterations
after laparoscopic and open surgery. The maximum increase of IL-6
in serum was found 1 day after the operation in the two groups,
followed by a decline after 4 days. In contrast to certain studies
in which no significant change was found in the CRP concentration
between females undergoing laparoscopically assisted vaginal
hysterectomy and those undergoing abdominal hysterectomy (28), we verified that there was a
significantly less severe increase in serum IL-6 levels after
laparoscopic cervical surgery. Our data are in accordance with
those of previous studies (29–30),
which reported that the serum concentration of IL-6 and CRP is
significantly different in the laparoscopy and conventional surgery
groups.
IL-6 is known to stimulate numerous immune cells and
the synthesis of acute-phase proteins, including CRP. CRP is the
prototypical acute-phase protein in humans and is a significant
mediator of immune host defense (24–26).
Normal baseline levels of circulating CRP are low, but a many-fold
increase may occur within hours of inflammation induced by
infection or injury (29). Serum
CRP levels are elevated as a result of major surgery, but have not
always reflected the magnitude of acute trauma after open and
laparoscopic surgery since CRP is a non-specific marker of an
acute-phase reaction (31).
Compared to the serum IL-6 levels, the maximum increase was delayed
by 2 days in the two groups. However, the increase in serum CRP
levels was found to be significantly less pronounced 1, 4 and 7
days after surgery in the laparoscopy group as compared to the
laparotomy group, indicating a certain benefit for the minimally
invasive procedure.
Cortisol is a glucocorticoid produced by the adrenal
cortex in response to adrenocorticotropic hormone (ACTH). Cortisol
is secreted with a circadian periodicity and peaks just prior to
waking in the morning. The production of glucocorticoids is
increased by stress; therefore, cortisol can be used as a biomarker
of stress. Serum levels of cortisol were measured 1, 4 and 7 days
after the operation. The daily comparison showed a significantly
higher increase after open surgery, particularly at 4 days after
surgery, suggesting that serum cortisol elastase levels reflect the
body’s stress after surgery, thereby indicating less pronounced
tissue injury after laparoscopic cervical cancer treatment.
The difference in increase of IL-6, CRP and cortisol
serum levels between the two groups may correlate to the difference
in tissue trauma and surgical stress caused by the two different
methods. However, the immune response may be caused by the
underlying pathology. All of the patients in our study had cervical
cancer, and the increase in IL-6, CRP and cortisol was equal, thus
appearing to be independent of the underlying pathology. Although
these substances are short-lived, their effect is believed to be
much more sustained. However, their role in inflammation has yet to
be thoroughly clarified.
From our study, it can be deduced that there is a
significant decrease in surgical trauma in the laparoscopy group
compared to the conventional open surgery group, especially on days
1 and 4. The significant differences between laparoscopic and
conventional colorectal surgery patients regarding the release of
CRP and IL-6 suggest a more pronounced proinflammatory response in
patients undergoing conventional surgery. This observation confirms
findings reported by Leung et al (32), who detected significantly smaller
peaks of circulating IL-1β, IL-6, and CRP levels in a group of 34
patients undergoing laparoscopic or conventional resection of
rectosigmoid carcinoma. It is not clear whether these differences
are immunologically relevant since a certain degree of
proinflammation is required for the initiation of host defense
mechanisms as well as for the activation of repair processes after
tissue trauma (33–34). It is nonetheless accepted that an
overwhelming inflammatory response to surgical trauma may
ultimately lead to organ dysfunction (35).
Findings of our study as well as previous ones, have
demonstrated that serum IL-6, CRP and cortisol can be used to
monitor surgical trauma. By using these parameters, the surgical
trauma after laparoscopic surgery was found to be significantly
reduced compared to that observed following the open procedure.
After more than 20 years of research, a laparoscopically assisted
vaginal radical hysterectomy can safely be performed and is
comparable in terms of side effects, complications and patient
trauma. This procedure not only involves less blood loss and a
shorter postoperative hospital stay, but also less surgical stress.
The rapid development of endoscopy and skillful operation of the
surgeon and the fact that patients usually prefer surgery with
better cosmetic results, laparoscopy is likely to become widely
used. Consistent with the results of other studies, the results of
this study demonstrated improved clinical recovery with
laparoscopy, which was of benefit to the patients.
However, any form of radical hysterectomy; both
laparoscopically assisted vaginal hystectomy and laparotomy, which
are traumatic involves significant trauma and leads to an
acute-phase response. Therefore, operations should be carried out
carefully at every step in order for minimally invasive surgery to
be less traumatic for patients.
Acknowledgements
This study was supported in part by grant
D09050703570903 (to Yuan-Qing Yao).
References
|
1
|
Querleu D, Leblanc E and Castelain B:
Laparoscopic pelvic lymphadenectomy in the staging of early
carcinoma in the cervix. Am J Obstet Gynecol. 164:579–581. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Dargent D and Mathevet P: Radical
laparoscopic vaginal hysterectomy. J Gynecol Obstet Biol Reprod.
21:709–710. 1992.PubMed/NCBI
|
|
3
|
Querleu D: Laparoscopically assisted
radical vaginal hysterectomy. Gynecol Oncol. 51:248–254. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Roy M: Laparoscopically assisted vaginal
radical hysterectomy. Best Pract Res Clin Obstet Gynaecol.
19:377–86. 2005. View Article : Google Scholar
|
|
5
|
Morgan DJ, Hunter DC, McCracken G, et al:
Is laparoscopically assisted radical vaginal hysterectomy for
cervical carcinoma safe? A case control study with follow up. BJOG.
114:537–542. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jackson KS, Das N, Naik R, et al:
Laparoscopically assisted radical vaginal hysterectomy vs. radical
abdominal hysterectomy for cervical cancer: a match controlled
study. Gynecol Oncol. 95:655–61. 2004. View Article : Google Scholar
|
|
7
|
Steed H, Rosen B, Murphy J, et al: A
comparison of laparascopic-assisted radical vaginal hysterectomy
and radical abdominal hysterectomy in the treatment of cervical
cancer. Gynecol Oncol. 93:588–593. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Malur S, Possover M and Schneider A:
Laparoscopically assisted radical vaginal versus radical abdominal
hysterectomy type II in patients with cervical cancer. Surg Endosc.
15:289–292. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sharma R, Bailey J, Anderson R, et al:
Laparoscopically assisted radical vaginal hysterectomy
(Coelio-Schauta): A comparison with open Wertheim/Meigs
hysterectomy. Int J Gynecol Cancer. 16:1927–1932. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nam JH, Kim JH, Kim DY, et al: Comparative
study of laparoscopico-vaginal radical hysterectomy and
abdominalradical hysterectomy in patients with early cervical
cancer. Gynecol Oncol. 92:277–283. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kohler C, Klemm P, Schau A, et al:
Introduction of transperitoneal lymphadenectomy in a gynecologic
oncology center: analysis of 650 laparoscopic pelvic and/or
paraaortic transperitoneal lymphadenectomies. Gynecol Oncol.
95:52–61. 2004. View Article : Google Scholar
|
|
12
|
Hertel H, Kohler C, Michels W, et al:
Laparoscopic-assisted radical vaginal hysterectomy (LARVH):
prospective evaluation of 200 patients with cervical cancer.
Gynecol Oncol. 90:505–511. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lee CL, Huang KG, Jain S, et al:
Comparison of laparoscopic and conventional surgery in the
treatment of early cervical cancer. J Am Assoc Gynecol Laparosc.
9:481–487. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dargent D and Mathevet P: Schauta’s
vaginal hysterectomy combined with laparoscopic lymphadenectomy.
Baillieres Clin Obstet Gynaecol. 9:691–705. 1995.
|
|
15
|
Allendorf JDF, Bessler M, Kayton ML,
Oesterling SD, Tret MR, Nowygrod R and Whelan RL: Increased tumor
establishment and growth after laparotomy vs laparoscopy in a
murine model. Arch Surg. 130:649–653. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mutter D, Hajri A, Tassetti V, Solis-Caxaj
C, Aprahamian M and Marescaux J: Increased tumor growth and spread
after laparoscopy vs laparotomy. Surg Endosc. 13:365–370. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Renaud MC, Plante M and Roy M: Combined
laparoscopic and vaginal radical surgery in cervical cancer.
Gynecol Oncol. 79:59–63. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Spirtos NM, Eisenkop SM, Schlaerth JB, et
al: Laparoscopic radical hysterectomy (type III) with aortic and
pelvic lymphadenectomy in patients with stage I cervical cancer:
surgical morbidity and intermediate follow-up. Am J Obstet Gynecol.
187:340–348. 2002. View Article : Google Scholar
|
|
19
|
Puntambekar SP, Palep RJ, Puntambekar SS,
et al: Laparoscopic total radical hysterectomy by the Pune
technique: our experience of 248 cases. J Minim Invasive Gynecol.
14:682–689. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Salo M: Effects of anaesthesia and surgery
on the immune response. Acta Anaesthesiol Scand. 36:201–220. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bolla G and Tuzzato G: Immunologic
postoperative competence after laparoscopy vs laparotomy. Surg
Endosc. 17:1247–1250. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gupta A and Watson DI: Effect of
laparoscopy on immune function. Br J Surg. 88:1296–1306. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lennard TW, Shenton BK, Borzotta A, et al:
The influence of surgical operations on components of the human
immune system. Br J Surg. 72:771–776. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cruickshank AM, Fraser WD, Burns HJ, van
Damme J and Shenkin A: Response of serum interleukin-6 in patients
undergoing elective surgery of varying severity. Clin Sci.
79:161–165. 1990.PubMed/NCBI
|
|
25
|
Gebhard F, Pfetsch H, Steinbach G, et al:
Is interleukin 6 an early marker of injury severity following major
trauma in humans? Arch Surg. 135:291–295. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Stensballe J, Christiansen M, Tonnesen E,
et al: The early IL-6 and IL-10 response in trauma is correlated
with injury severity and mortality. Acta Anaesthesiol Scand.
53:515–521. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Holub Z: Impact of laparoscopic surgery on
immune function. Clin Exp Obstret Gynecol. 29:77–81.
2002.PubMed/NCBI
|
|
28
|
Harkki-Siren P, Sjoberg J, Toivonen J and
Tiitinen A: Clinical outcome and tissue trauma after laparoscopic
and abdominal hysterectomy: a randomized controlled study. Acta
Obstet Gynecol Scand. 79:866–871. 2000.PubMed/NCBI
|
|
29
|
Malik E, Buchweitz O, Muller-Steinhardt M,
et al: Prospective evaluation of the systemic immune response
following abdominal, vaginal, and laparoscopically assisted vaginal
hysterectomy. Surg Endosc. 15:463–466. 2001. View Article : Google Scholar
|
|
30
|
Yuen PM, Mak TW, Yim SF, Kee WD, et al:
Metabolic and inflammatory responses after laparoscopic and
abdominal hysterectomy. Am J Obstet Gynecol. 179:1–5. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Baigrie RJ, Lamont PM, Kwiatkowski D, et
al: Systemic cytokine response after major surgery. Br J Surg.
79:757–760. 2000. View Article : Google Scholar
|
|
32
|
Leung KL, Lai PB, Ho RL, et al: Systemic
cytokine response after laparoscopic-assisted resection of
rectosigmoid carcinoma: a prospective randomized trial. Ann Surg.
231:506–511. 2000. View Article : Google Scholar
|
|
33
|
Jeschke MG, Wolf SE, DebRoy MA and Herndon
DN: The combination of growth hormone with hepatocyte growth
factors alters the acute phase response. Shock. 12:181–187. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Gabay C and Kushner I: Acute-phase
proteins and other systemic responses to inflammation. N Engl J
Med. 340:448–454. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Faist E and Wichmann MW: Immunology in the
severely injured. Chirurg. 68:1066–1070. 1997.PubMed/NCBI
|