Introduction
The high mobility group box 1 (HMGB1) protein is an
abundant non-histone component of chromatin. It is well known for
its two DNA binding domains, HMG box A and HMG box B. The HMG box
motive was also detected in a number of regulatory factors
including UBF, Lef-1, SRY and SSRP (1). The main characteristics of HMGB1
protein are i) its capability to recognize and bind with high
affinity to distorted DNA, such as cruciform DNA, UV and
cisplatin-damaged DNA (2–4) and ii) its ability to induce kinks in
linear DNA fragments (5,6). These characteristics identify the
protein as an ‘architectural’ factor, facilitating the assembly of
certain nucleoprotein complexes, and render it a key participant in
fundamental nuclear events including repair (7), replication (8) and remodeling (9–10). In
previous years, the extracellular functions of HMGB1 protein have
gained scientific interest. It was revealed that
monocytes/macrophages actively secreted HMGB1 in response to
various stimuli (11) and that,
once secreted, HMGB1 was capable of activating other cells involved
in immune response or inflammatory reactions, thus acting as a
cytokine itself (12,13). The protein can also be passively
released by damaged or necrotic cells, leading to inflammation
(14). A number of
post-translational modifications including acetylation (15), phosphorylation (16), methylation (17) and poly(ADP)-ribosylation (18) have been suggested to direct HMGB1 to
the secretory pathway.
The HMGB1 protein has been associated with cancer
progression. An elevated expression of HMGB1 was observed in
certain primary tumors including melanoma and colon, prostate,
pancreatic, and breast cancers (19). In the majority of cases, HMGB1 was
associated with invasion and metastasis. The protein was proposed
to be directly involved in tumor cell metastasis through its
ability to promote cell migration, modulate the adhesive properties
of cells and modify components of the extracellular matrix
(20,21). The experiments carried out using C6
glioma cells, an example of highly invasive cells, demonstrated
that inhibition of HMGB1 expression by anti-sense oligonucleotides
or inhibition of the protein function by anti-HMGB1 antibodies
markedly inhibited cell migration (21). Mounting evidence show that the main
signaling pathway is activated through the interaction of HMGB1 and
its Receptor for Advanced Glycation End products (RAGE). The
significance of this pathway in vivo was indicated by the
observation that blockade of the HMGB1/interaction suppressed tumor
growth and metastasis in lung cancer (22). One probable mechanism is that the
HMGB1/RAGE complex induces depletion of macrophages in colon cancer
(23). The lack of host defense,
therefore, becomes conducive to tumor spread.
RAGE is constitutively expressed during embryonic
development and its expression is downregulated in adult life.
However, known exceptions are the skin and lung, which
constitutively express RAGE throughout life. The majority of other
cells, including monocytes/macrophages, endothelial and smooth
muscle cells, fibroblasts and neuronal cells, do not produce
significant amounts of RAGE under physiological conditions but may
be induced to express RAGE in situations where ligands accumulate
(24). Several findings have
indicated that the elevated expression of RAGE was not always a
prerequisite of poor prognosis of tumor development. The cellular
localization of the receptor should also be taken into
consideration. In colorectal adenomas, the cytosolic pattern was
associated with mild atypia and small tumor size, whereas the
membranous pattern was correlated to severe atypia, villous
histological type and elevated levels of HMGB1 protein. These
results indicated that RAGE expression, particularly with a
membranous pattern, was associated with the malignant potential of
colorectal adenomas (25).
Immunohistochemistry revealed that RAGE exhibited dot-like
cytoplasmic localization in primary hepatocellular and colorectal
carcinomas, which changed to dense brown staining across the
metastatic cells due to membranous expression (26).
The data concerning the expression of HMGB1 protein
and its receptor RAGE in various tissues and tumor cells mainly
reflect the overall production of the proteins. However, these data
do not refer to the cellular localization of HMGB1 and RAGE and
there is no direct evidence for the formation of a stable complex
between the two proteins. We examined the expression of HMGB1
protein and its receptor RAGE in different rat organs and in Guerin
ascites tumor cells in respect to their localization and complex
formation.
Materials and methods
Preparation of total protein extract
Guerin ascites tumor cells were inoculated in albino
rats. The ascite liquid was collected 7 days after transplantation
together with 2 g of tissue samples. The total protein extracts
were prepared as described by Dignam (27). The material was manually homogenized
on ice in lysis buffer [5 mM Tris-HCl, pH 7.4, 2 mM EDTA, 1% Triton
100, 1 mM PMSF and protease inhibitor mix (Boehringer, Mannheim,
Germany)], sonicated, centrifuged at 500 × g for 30 min and
aliquoted at −80°C.
Preparation of soluble and membrane
protein extracts
The tissue samples were washed in cold
phosphate-buffered saline (PBS), fast-frozen in liquid nitrogen,
homogenized in 5 mM Tris-HCl, pH 7.4, 2 mM EDTA, 1% Triton 100, 1
mM PMSF and protease inhibitor mix (Boehringer) and centrifuged at
500 × g for 30 min at 4°C. The samples were then washed twice with
the same buffer and the collected supernatants were centrifuged at
45,000 × g for 30 min at 4°C. The supernatant was considered as the
‘soluble fraction’. The pellet was suspended in 75 mM Tris, pH 7.4,
12.5 mM MgCl2, 5 mM EDTA and considered as the ‘membrane
fraction’. The samples were aliquoted and stored at −80°C.
Immunoblotting
Samples (80–100 μg of protein extract) were resolved
on SDS-18% polyacrylamide gel and transferred to nitrocellulose
membranes (Schleicher & Schuell, Dassell, Germany) at 4°C for 1
h at 8 V/cm in a buffer containing 25 mM Tris, pH 8.2, 192 mM
glycine and 15% methanol. The membranes were blocked for 1 h in 10%
(w/v) non-fat dry milk in TBST buffer (10 mM Tris, pH 7.5, 150 mM
NaCl, 0.1% Tween-20) and incubated for 2 h at room temperature with
various primary antibodies diluted at 1:1000. The antibodies
included: polyclonal rabbit anti-HMGB1 antibody (Upstate),
monoclonal mouse anti-RAGE antibody (Santa Cruz Biotechnology,
Santa Cruz, CA, USA) and mouse monoclonal anti-actin antibody AC-15
as a reference (Sigma Immunochemicals, St. Louis, MO, USA). The
secondary antibodies 680 conjugated goat (polyclonal) anti-mouse
IgG (LI-COR, Lincoln, NE, USA) and 800 CW conjugated goat
(polyclonal) anti-rabbit IgG (LI-COR) were used at a dilution of
1:3000 to visualize immunoreactivity. Immunoreactivity was detected
using the chemiluminescent method (Odyssey Infrared Imaging System,
Amersham, Buckinghamshire, UK).
Immunoprecipitation
Immunoprecipitation with the monoclonal anti-RAGE
antibody (Santa Cruz Biotechnology) was carried out according to
the manufacturer’s instructions. The antibody (2 μg) was incubated
with 100 μl (5 μg/μl) protein extract at 4°C with gentle agitation.
After 1 h, Protein A-Sepharose (Zymed Laboratories, San Francisco,
CA, USA) was added for an additional incubation period of 1 h. The
sepharose beads were washed twice with RIPA buffer (50 mM Tris-HCl,
pH 7.4, 1% NP-40, 0.25% Na-deoxycholate, 150 mM NaCl, 1 mM EDTA and
protease inhibitors) and PBS, and boiled in 0.5% SDS for 10 min.
The proteins were analyzed using sodium dodecyl sulfate
polyacrylamide gel electrophoresis and Western blotting.
Results
Expression level of HMGB1 protein and its
receptor RAGE in rat normal tissues and tumor cells and their
distribution in soluble and membrane fractions
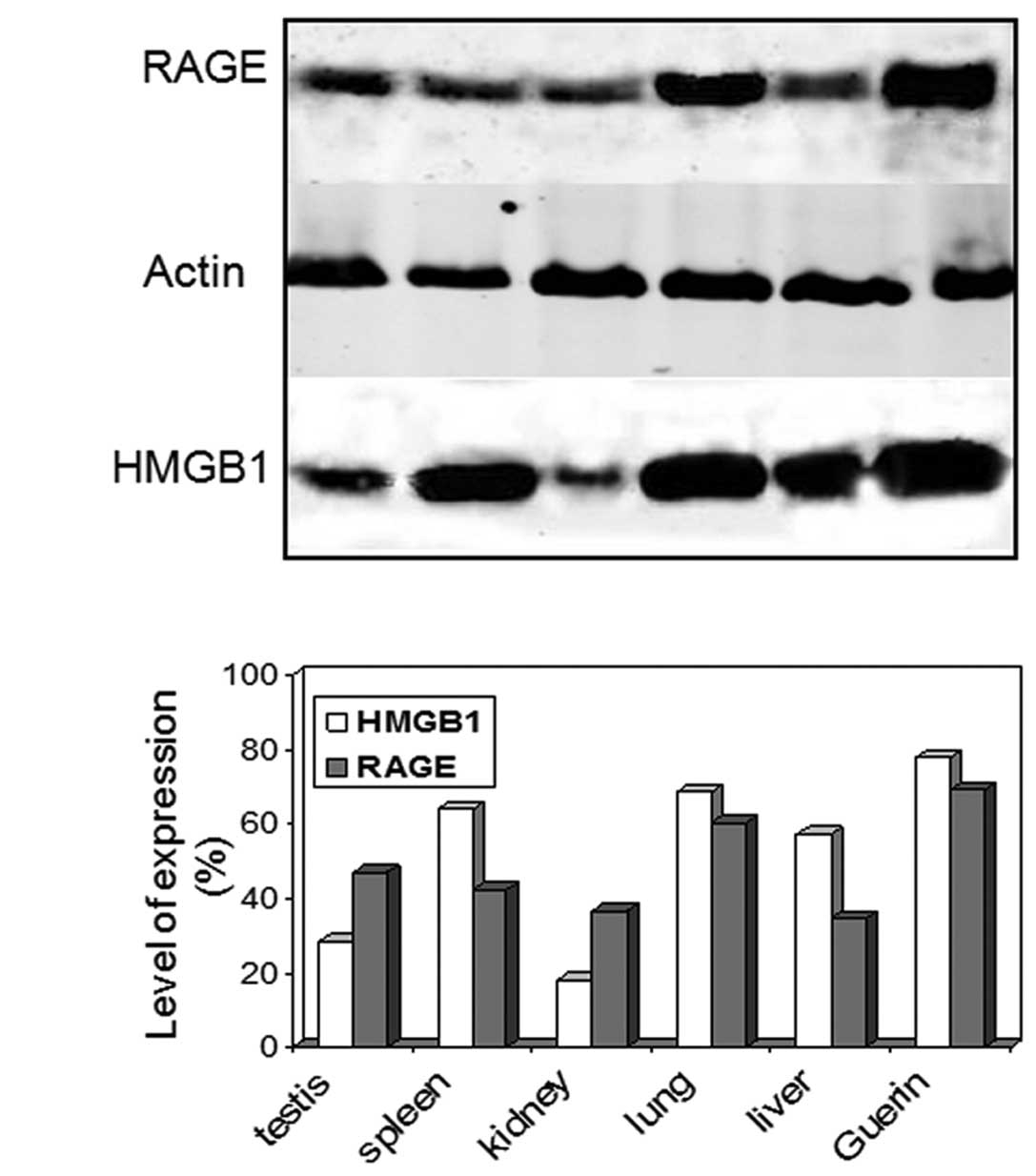
Using immunoblotting, we examined the total
expression level of HMGB1 protein and its receptor RAGE in various
rat organs including the liver, lung, testis and spleen, as well as
Guerin ascites tumor cells. The two proteins were detected in the
tested specimens as their amount varied in the different organs and
tumor cells (Fig. 1). The HMGB1
production was higher in the lung, liver and spleen and lower in
the kidney as previously reported for older animals (28). In the normal tissues RAGE levels
were elevated in the lung, which constitutively expressed the
receptor throughout life. It should be noted that the highest
protein amounts were registered in the Guerin tumor cells. In the
majority of cases the increased production of HMGB1 and RAGE was
correlated to tumor development and poor prognosis (19); however, a number of exceptions were
reported. Although HMGB1 was overexpressed in the majority of
tumors, tumors devoid of HMGB proteins have also been found. For
example, adrenal gland carcinoma exhibited no HMGB1 expression
(29). A marked intertumoral
variation of HMGB1 expression was observed in various breast
cancers (30). This was also the
case for RAGE: in non-small cell lung carcinomas RAGE expression
was strongly reduced (31).
Given that the overall expression of HMGB1 and RAGE
is not always considered to be an indicator for cancer progression,
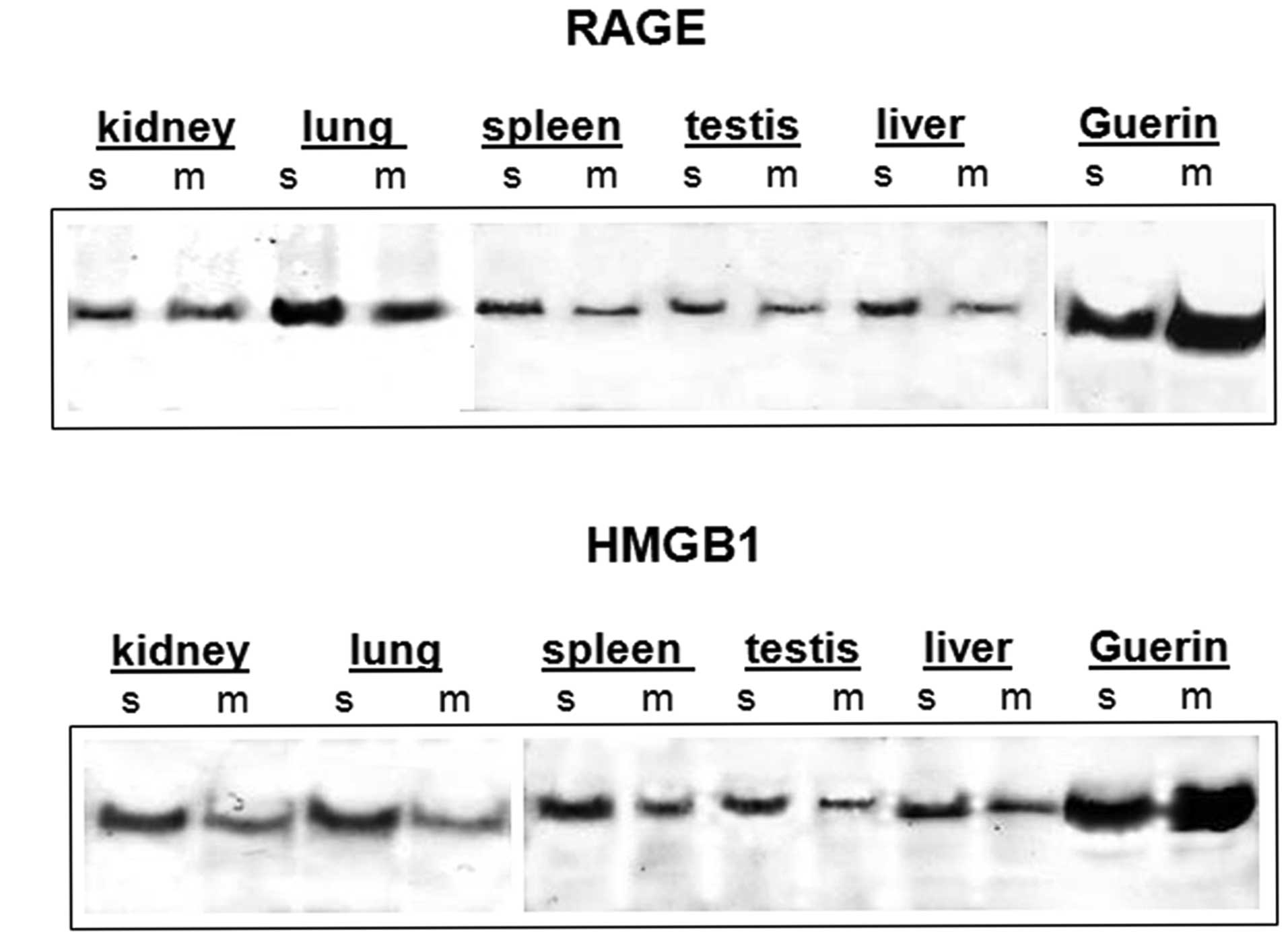
we examined the distribution of the two proteins in soluble and
membrane fractions of normal tissues and tumor cells. The results
are shown in Fig. 2. In the rat
organ specimens, HMGB1 protein was mainly observed in the soluble
fraction and lesser amounts were recovered from the insoluble
membrane fractions (Fig. 2B). These
findings are in agreement with previously reported data that the
bulk of the protein remained in the supernatant following high
speed centrifugation; however, part of the protein sedimented with
the microsomal membrane fraction (32). Similar results were obtained for
RAGE distribution in normal tissues (Fig. 2A). Although the receptor was
considered a membrane protein it was expressed as the full-length,
membrane-bound form and various soluble forms lacking the
transmembrane domain. Soluble RAGE was produced by the proteolytic
cleavage of full-length RAGE and alternative mRNA splicing. The
soluble isoforms included the extracellular domains but lacked the
transmembrane and cytoplasmic domains (33). Again, the tumor cells exhibited a
markedly different localization pattern for HMGB 1 protein and its
receptor RAGE in comparison with the normal tissues. The two
proteins were predominantly observed in the insoluble membrane
fraction (Fig. 2A and B, compare
soluble/membrane Guerin with soluble/membrane normal tissue).
Notably, the overall expression of RAGE was almost comparable in
lung and tumor cells (Fig. 1B).
However, whereas in lung tissue the receptor was mainly observed in
its soluble form, RAGE was predominantly membrane-bound in the
Guerin tumor cells. The distribution pattern also differed for
HMGB1 protein: HMGB1 was chiefly soluble in the protein extract
from normal tissues, whereas it mainly occurred in its insoluble
membrane form in cancer cells (Fig.
2B). Our results confirm the hypothesis that the cellular
localization of HMGB1 protein and its receptor RAGE should be
considered as a more reliable indicator of tumor progression
instead of the total protein expression level.
HMGB1 protein interacts with its receptor
RAGE only in tumor cells
The question remains as to whether the
over-expression of HMGB1 and its receptor RAGE always leads to the
formation of a ligand/receptor complex. To examine this
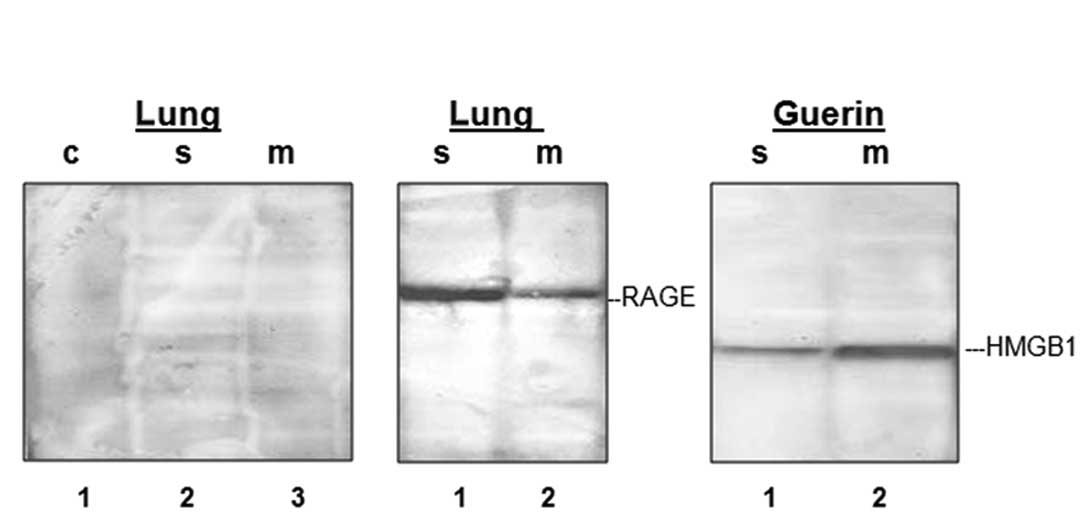
possibility, we performed immunoprecipitation with anti-RAGE
antibody of the soluble and membrane fractions and revealed
immunoreactivity by incubation with anti-HMB1 antibody. We selected
the protein extracts from the lung as a control, since the RAGE
level was higher in this normal tissue. We applied the same
approach to Guerin tumor cells. Immuno-precipitation with anti-RAGE
antibody and the subsequent findings of the immunoblot analysis
with anti-HMB1 antibody provided no signal in the soluble or
membrane fraction derived from rat lung samples (Fig. 3A, lanes 2 and 3). To verify that the
immunoprecipitation was effective, we performed the same experiment
with the exception that the transfer membrane was incubated with
anti-RAGE antibody. The positive signals (Fig. 3, lanes 1 and 2) in the soluble and
membrane fractions demonstrated that receptor molecules were bound
to the antibody, but the receptor clearly did not carry the HMGB1
protein. By contrast, in the case of the tumor cells visualization
with the anti-HMGB1 antibody of the anti-RAGE precipitated complex
provided a clearly positive signal in the membrane fraction,
indicating that the HMGB1 protein formed a stable complex with its
receptor RAGE (Fig. 3, lane 2). In
the soluble extract, HMGB1 was also associated with the receptor
but the interaction was probably weaker as a less intensive signal
was detected (Fig. 3, lane 1).
Discussion
The coexistence in the cell of the HMGB1 protein and
its receptor RAGE in relatively higher levels is not a necessary
prerequisite for complex formation and cancer development. The
quantities of the proteins distributed in the membrane and soluble
fractions should be investigated. As we demonstrated, in the normal
tissue the receptor was predominantly observed in its soluble form,
whereas in the Guerin ascites tumor cells it changed to the
membrane-bound form. The soluble RAGE (sRAGE) acts as a decoy that
prevents ligands from interacting with the cell surface receptor.
The application of sRAGE in vitro and in vivo
resulted in an effective blockade of RAGE, in accordance with this
decoy mechanism, in a range of animal models (24). sRAGE prevented the development of
micro-and macrovascular diseases in rodents and also protected the
animals from tumor metastasis and growth of primary tumors
(22). By contrast, membrane-bound
RAGE was generally associated with metastatic potential and poor
prognosis (25,26). The bulk of HMGB1 protein was
generally found in the soluble fraction following high speed
centrifugation (32); however, in
certain cases the protein may alter its subcellular localization.
For example, upon platelet activation, part of HMGB1 was associated
with the plasma membrane (34). In
murine erythroleukemia (MEL) cells, following induction with
hexamethylene bisacetamide, HMGB1 protein accumulated in a
membrane-bound form (35). In our
case, we observed increased quantities of HMGB1 protein in the
tumor cells with prevailing membrane localization, which we
consider to be a clear characteristic of cancer development.
Furthermore, we determined a stable HMGB1/RAGE complex in the tumor
cells alone, mainly occurring in the membrane fraction. The data
presented in this study indicate that during tumorigenesis, HMGB1
protein and its receptor RAGE undergo cellular redistribution
required for the stable protein/protein interaction. Other factors
are also indispensible for the formation of a stable
receptor/ligand complex, since in the normal tissues the two
proteins coexist in the membrane fraction in smaller quantities.
However, no association was identified.
Acknowledgements
This study was supported by grant DTK 02/80 from the
National Science Fund.
References
|
1
|
Thomas JO and Travers A: HMG1 and 2, and
related ‘architectural’ DNA-binding proteins. Trends Biochem
Science. 26:167–174. 2001.
|
|
2
|
Bianchi ME, Bertrame M and Paonessa G:
Specific recognition of cruciform DNA by nuclear protein HMG1.
Science. 243:1056–1059. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Pil PM and Lippard SJ: Specific binding of
chromosomal protein HMG1 to DNA damaged by the anticancer drug
cisplatin. Science. 256:234–237. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pasheva EA, Pashev IG and Favre A:
Preferential binding of high mobility group 1 protein to UV-damaged
DNA. Role of the COOH-terminal domain. J Biol Chem.
273:24730–24736. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Paull T, Hakynson MJ and Johnson RC: The
nonspecific DNA-binding and bending proteins HMG1 and HMG2 promote
the assembly of complex nucleoprotein structures. Genes Dev.
7:1521–1534. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Onate SA, Prendergast P, Wagner JP, Nissen
M, Reeves R, Pettijhon DE and Edwards DP: The DNA-bending protein
HMG-1 enhances progesterone receptor binding to its target DNA
sequences. Mol Cell Biol. 14:3375–3391. 1994.PubMed/NCBI
|
|
7
|
Ugrinova I, Zlateva S, Pashev IG and
Pasheva EA: Native HMGB1 protein inhibits repair of
cisplatin-damaged nucleosomes in vitro. Int J Biochem Cell Biol.
41:1556–1562. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Topalova D, Ugrinova I, Pashev IG and
Pasheva EA: HMGB1 protein inhibits DNA replication in vitro: a role
of the acetylation and the acidic tail. Int J Biochem Cell Biol.
40:1536–1542. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bonaldi T, Langst G, Strohner R, Becker PB
and Bianchi ME: The DNA chaperone HMGB1 facilitates ACF/CHRAC
dependent nucleosome sliding. EMBO J. 21:6865–6873. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ugrinova I, Pashev IG and Pasheva EA:
Nucleosome binding properties and co-remodeling activities of
native and in vivo acetylated HMGB-1 and HMGB-2 proteins.
Biochemistry. 48:6502–6507. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang H, Bloom O, Zhang M, Vishnubhakat JM,
Ombrellino M, Che J, Frazier A, Yang H, Ivanova S, Borovikova L,
Manogue KR, Faist E, Abraham E, Andersson J, Andersson U, Molina
PE, Abumrad NN, Sama A and Tracey KJ: HMG-1 as a late mediator of
endotoxin lethality in mice. Science. 285:248–251. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Andersson U, Wang H, Palmblad K, Aveberger
AC, Bloom O, Erlandsson-Harris H, Janson A, Kokkola R, Zhang M,
Yang H and Tracey KJ: High mobility group 1 protein (HMG-1)
stimulates proinflammatory cytokine synthesis in human monocytes. J
Exp Med. 192:565–570. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Degryse B and De Virgilio M: The nuclear
protein HMGB1, a new kind of chemokine. FEBS Lett. 553:11–17. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Scaffidi P, Misteli T and Bianchi ME:
Release of chromatin protein HMGB1 by necrotic cells triggers
inflammation. Nature. 418:191–195. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bonaldi T, Talamo F, Scaffidi P, Ferrera
D, Porto A, Bachi A, Rubartelli A, Agresti A and Bianchi ME:
Monocytic cells hyperacetylate chromatin protein HMGB1 to redirect
it towards secretion. EMBO J. 22:5551–5560. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Youn JH and Shin JS: Nucleocytoplasmic
shuttling of HMGB1 is regulated by phosphorylation that redirects
it toward secretion. J Immunol. 177:7889–7897. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ito I, Fukuzawa J and Yoshida M:
Post-translational methylation of high mobility group box 1 (HMGB1)
causes its cytoplasmic localization in neutrophils. J Biol Chem.
282:16336–16344. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ditsworth D, Zong WX and Thompson CB:
Activation of poly(ADP)-ribosepolymerase (PARP-1) induces release
of the pro-inflammatory mediator HMGB1 from the nucleus. J Biol
Chem. 282:17845–17854. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ellerman JE, Brown CK, De Vera M, Zeh HJ,
Billiar T, Rubartelli A and Lotze M: Masquerader: high mobility
group box-1 and cancer. Clin Cancer Res. 13:2836–2848. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sasahira T, Kirita T, Oue N, Bhawal UK,
Yamamoto K, Fujii K, Ohmori H, Luo Y, Yasui W, Bosserhoff AK and
Kuniyasu H: High mobility group box-1-inducible melanoma inhibitory
activity is associated with nodal metastasis and lymphangiogenesis
in oral squamous cell carcinoma. Cancer Sci. 99:1806–1812.
2008.PubMed/NCBI
|
|
21
|
Fages C, Nolo R, Huttunen H, Eskelinen E
and Rauvala H: Regulation of cell migration by amphoterin. J Cell
Science. 113:611–620. 2000.
|
|
22
|
Taguchi A, Blood DC, Del Toro G, Canet A,
Lee DC, Qu W, Tanji N, Lu Y, Lalla E, Fu C, Hofmann MA, Kislinger
T, Ingram M, Lu A, Tanaka H, Hori O, Ogawa S, Stern DM and Schmidt
AM: Blockade of RAGE-amphoterin signalling suppresses tumour growth
and metastases. Nature. 405:354–360. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kuniyasu H, Chihara Y and Takahashi T:
Co-expression of receptor for advanced glycation end products and
the ligand amphoterin associates closely with metastasis of
colorectal cancer. Oncol Rep. 10:445–448. 2003.PubMed/NCBI
|
|
24
|
Bierhaus A, Humpert P, Morcos M, Wendt T,
Chavakis T, Arnold B, Stern D and Nawroth P: Understanding RAGE,
the receptor for advanced glycation end products. J Mol Med.
83:876–886. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sasahira T, Akama Y, Fujii K and Kuniyasu
H: Expression of receptor for advanced glycation end products and
HMGB1/amphoterin in colorectal adenomas. Virchows Arch.
446:411–415. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kostova N, Zlateva S, Ugrinova I and
Pasheva E: The expression of HMGB1 protein and its receptor RAGE in
human malignant tumors. Mol Cell Biochem. 337:251–258. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dignam JD: Preparation of extracts from
higher eukaryotes. Methods Enzymol. 182:194–203. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Prasad S and Thakur MK: Distribution of
high mobility group proteins in different tissues of rats during
aging. Biochem Int. 20:687–695. 1990.PubMed/NCBI
|
|
29
|
Muller S, Ronfani L and Bianchi M:
Regulated expression and subcellular localization of HMGB1, a
chromatin protein with a cytokine function. J Int Med. 255:332–343.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Flohr A, Rogalla P, Meiboom M, Borrmann L,
Krohn M, Thode-Halle B and Bullrdiek J: Variation of HMGB1
expression in breast cancer. Anticancer Res. 21:3881–3885.
2001.PubMed/NCBI
|
|
31
|
Bartling B, Hofmann H, Weiglel B, Silber S
and Simm A: Down-regulation of the receptor for advanced glycation
end-products (RAGE) supports non-small cell lung carcinoma.
Carcinogenesis. 26:293–301. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Merenmies J, Pihlaskari R, Laitinen J,
Wartiovaarall J and Rauvala H: 30-kDa heparin-binding protein of
brain (amphoterin) involved in neurite outgrowth. J Biol Chem.
266:16722–16729. 1991.PubMed/NCBI
|
|
33
|
Sparvero L, Asafu-Adjei D, Kang R, Tang D,
Amin N, Im J, Rutledge R, Lin B, Amoscato A, Zeh H and Lotze M:
RAGE (Receptor for Advanced Glycation End products), RAGE ligands
and their role in cancer and inflammation. J Transl Med. 7:17–39.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rouhiainen A, Imai S, Rauvala H and
Parkkinen J: Occurrence of amphoterin (HMG1) as an endogenous
protein of human platelets that is exported to the cell surface
upon platelet activation. Thromb Haemost. 84:1087–1094.
2000.PubMed/NCBI
|
|
35
|
Passalacqua M, Zicca A, Sparatore B,
Patrone M, Melloni E and Pontremoli S: Secretion and binding of
HMG1 protein to the external surface of the membrane are required
for murine erythroleukemia cell differentiation. FEBS Lett.
400:275–279. 1997. View Article : Google Scholar : PubMed/NCBI
|