Introduction
Urothelial carcinoma (UC) is known to progress
through two divergent pathways: one is the pathway characterized by
superficial papillary tumors that harbor gain-of-function mutations
of H-RAS, FGFR3 and phosphatidylinositol 3-kinase (PI3K), and the
other is that of invasive tumors that exhibit defects in p53,
retinoblastoma protein (RB), and phosphatase and tensin homolog
deleted on chromosome 10 (PTEN) (1).
PTEN is a key negative regulator of the PI3K-protein
kinase B (PKB/Akt) signaling pathway (2). PI3K, activated by growth factors,
catalyzes the phosphorylation of phosphatidylinositol (4,5)-biphosphate (PIP2) to
phosphatidylinositol (3,4,5)-triphosphate (PIP3). PIP3 recruits
3-phosphoinositide-dependent kinase (PDK), which phosphorylates and
activates Akt (3). Akt is a signal
transduction protein that plays a key role in multiple signaling
pathways, including cell proliferation, apoptosis and transcription
(4). Loss of PTEN expression
results in an increased concentration of PIP3 and causes Akt to
transform into its phosphorylated, active form (pAkt) (2,5).
Heat shock protein 90 (HSP90) is a molecular
chaperone required for the stability and function of various client
proteins, which mediate a major property necessary for the
transformation of a normal cell to a cancer cell (6,7). HSP90
has two isoforms, HSP90α and HSP90β, but it has been reported that
only HSP90α is capable of activating oncogenic kinases (8). Inhibition of HSP90 induces the
degradation and inactivation of a number of HSP90 client proteins,
including protein kinases [human epidermal growth factor receptor 2
(HER2) and Akt], steroid hormone receptors and mutant oncoproteins
(mutant p53 and B-Raf), leading to an antitumor effect (9,10).
DJ-1 is a conserved protein, coded by the gene
Parkinson disease 7 (PARK7). It is associated with an
autosomal recessive early-onset Parkinson's disease (PD) (11). This protein is ubiquitously present
in cells and is involved in diverse cell processes such as cell
transformation, control of protein-RNA interaction and preventing
cell death from oxidative stress-induced apoptosis (12). DJ-1 promotes cell survival by
modulating PTEN (13), and high
DJ-1 levels have been reported during initiation and progression in
certain types of cancer (14–16).
The alterations of PTEN, pAkt and PI3K have been
found to contribute to bladder carcinogenesis (1,17,18).
However, DJ-1 and HSP90α expression in bladder tumors compared with
clinicopathological parameters has yet to be studied, although
total HSP90 expression in bladder tumors has rarely been described
(19,20).
In the present study, we analyzed the expression of
DJ-1, HSP90α, PTEN, pAkt and PI3K-p110α (p110α subunit of PI3K) in
bladder tumors and their association with pathological parameters
to determine the role of these proteins in the prognostic
significance of UC.
Materials and methods
Patients and samples
Tissue samples from 102 UC patients who underwent
transurethral resection of bladder tumor (88 cases) or radical
cystectomy (14 cases) at the Eulji Medical Center, Eulji University
School of Medicine (Seoul, Korea), between 2004 and 2008, were
enrolled in this study. The pathology slides were reviewed, and
representative sections of each tumor and tumor-free bladder
tissues, considered as ‘normal’, from the 14 radical cystectomy
specimens were selected as a control. The histological grade of the
bladder tumors was determined according to the 2004 World Health
Organization-International Society of Urological Pathology
(WHO-ISUP) classification (21).
Table I shows the clinical and
pathological data of patients. Following the review of all 102 UC
sections, the most representative areas of tumor tissues (2 mm in
diameter) were removed from the paraffin blocks and arranged in a
new recipient block for tissue microarray (TMA). TMAs were
constructed using a microarray instrument (Unitma Co., Seoul,
Korea). The institutional review board of Eulji Medical Center
approved this study.
 | Table IPatient and tumor characteristics. |
Table I
Patient and tumor characteristics.
| Characteristics | Number of
patients |
|---|
|
|
|---|
| n | (%) |
|---|
| Age |
| <70 | 46 | 45.1 |
| ≥70 | 56 | 54.9 |
| Gender |
| Male | 83 | 81.4 |
| Female | 19 | 18.6 |
| Growth pattern |
| Papillary | 66 | 64.7 |
| Non-papillary | 36 | 35.3 |
| Tumor stage |
| Ta | 34 | 33.3 |
| T1 | 40 | 39.2 |
| T2 | 16 | 15.7 |
| T3 | 12 | 11.8 |
| Tumor grade |
| Low | 51 | 50 |
| High | 51 | 50 |
Immunohistochemistry
Immunohistochemical staining was performed using
Dako Autostainer (DakoCytomation, Carpinteria, CA, USA). Tissue
sections (4-μm) were obtained from TMA blocks and transferred onto
poly-L-lysine-coated slides. Following deparaffinization and
rehydration, antigen retrieval was performed using citrate buffer
(pH 6.0) at 121°C for 10 min. Endogenous peroxidase activity was
blocked with 3% hydrogen peroxide for 5 min, and the sections were
incubated with antibodies against DJ-1 (Abcam, Cambridge, UK;
1:1,000), HSP90α (Abcam; 1:10,000), pAkt (Abcam; 1:500), PI3K-p110α
(Cell Signaling, Danvers, MA, USA; 1:500) and PTEN (Epitomics,
Burlingame, CA, USA; 1:250). Color was developed using
diaminobenzidine, and the slides were counterstained with
hematoxylin. Normal urinary bladder mucosa was used as a positive
control for DJ-1, HSP90α and PTEN. Endothelial cells were used as
positive controls for PI3K-p110α and breast carcinoma was used as a
positive control for pAkt. Cases not treated with primary
antibodies served as negative controls.
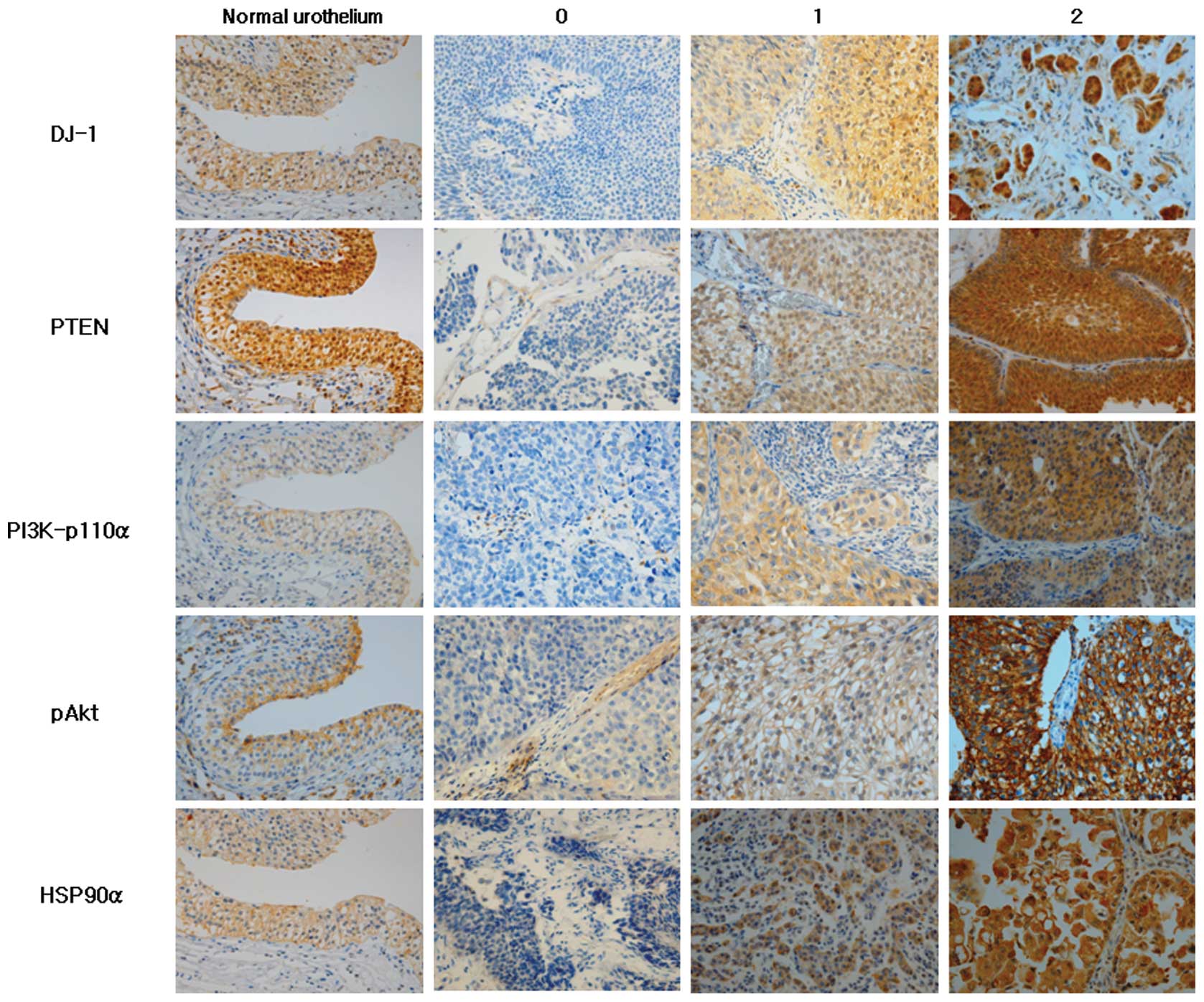
Immunohistochemical scoring of all antibodies used
for this study was based on the intensity of staining from the most
intensely stained area. The staining intensity was scored as: 0, no
expression; 1+, low expression (weak intensity); and 2+, high
expression (moderate to strong intensity) (Fig. 1).
Statistical analysis
The Chi-square test and Fisher's exact test were
used to evaluate the correlation between the immunoreactivity of
DJ-1, HSP90α, PTEN, pAkt and PI3K-p110α, and clinicopathological
characteristics. The Spearman's rank correlation test was used to
analyze the association between markers. P<0.05 was considered
to indicate a statistically significant difference. The analyses
were performed using the IBM SPSS version 19.0.
Results
Staining patterns of DJ-1, PTEN, PI3K-p110α, pAkt
and HSP90α in normal urothelium and UC with varying intensity are
shown in Fig. 1. DJ-1 in the normal
urothelium was localized in the nuclei, cytoplasm and cell membrane
in all cases. DJ-1 expression in UC was mostly cytoplasmic with
some nuclear and membranous staining. DJ-1 was expressed in 84.3%
(low 17.6% and high expression 66.7%) of UCs. HSP90α exhibited
cytoplasmic and membranous localization in normal urothelial cells
and in 91.2% of UCs (low 25.5% and high expression 65.7%). A strong
expression of DJ-1 and HSP90α was observed more in non-papillary,
high-grade, invasive UCs compared to papillary, low-grade and
non-invasive ones (Fig. 1). PTEN
expression was observed in the cytoplasm and nuclei of normal
urothelial cells in all cases and 63.7% of UCs (low 42.2% and high
expression 21.5%). In contrast to DJ-1 and HSP90α expression, PTEN
expression was much stronger in papillary, non-invasive and
low-grade UCs compared to non-papillary, high-grade, invasive UCs
(Fig. 1). PI3K-p110α exhibited only
membranous staining in the normal urothelium, but membranous and
cytoplasmic staining in UC. PI3K-p110α was expressed in 67.6% (low
41.2% and high expression 26.4%) of UC cases. pAkt expression was
heterogeneous with speckled cytoplasmic and membranous staining in
normal urothelium and UCs. pAkt was expressed in 64.7% (low 32.35%
and high expression 32.35%) of UCs.
Table II shows the
correlation between markers analyzed and clinically important
histopathological features of the tumors. The expression of DJ-1
and HSP90α was positively correlated with the invasiveness of the
tumor, with a greater expression in T1-T3 tumors compared to
non-invasive tumors (Ta) [Spearman's correlation coefficient (cc),
0.201; P=0.043 and cc, 0.303; P=0.002, respectively], but not with
growth pattern (papillary vs. non-papillary) and histological
grade. A significant negative correlation was observed between PTEN
expression and non-papillary growth pattern (cc, −0.324; P=0.001),
invasive tumor (cc, −0.246; P=0.013) and a high histological grade
(cc, −0.365; P<0.001). However, PI3K-p110α and pAkt expression
did not correlate with any histopathological parameters. A
statistically significant positive correlation was present between
DJ-1 and PI3K-p110α expression (cc, 0.367; <0.001) as well as
between PI3K-p110α and PTEN expression (cc, 0.242; P=0.014) in UC.
Table III shows the positive
correlation between the markers in a two by two comparison using
the Chi-square test. The majority of the PI3K-p110α-positive UCs
also stained positively for DJ-1 (64/69, 92.7%). Of 33 negative
samples for PI3K-p110α, 22 cases (66.7%) were DJ-1-positive
(Table IIIA). PTEN-positive
tumors were more common in PI3K-p110α-positive samples (71%, 49/69
cases) compared to PI3K-p110α-negative samples (48.5%, 16/33 cases;
Table IIIB). No significant
correlation was found among other markers.
 | Table IICorrelation between histopathological
variables and expression of each protein. |
Table II
Correlation between histopathological
variables and expression of each protein.
| DJ-1 | PTEN | PI3K-p110α | pAkt | HSP90α |
|---|
| Non-papillary | 0.790a |
0.001a | 0.274a | 0.451 | 0.184 |
| ≥T1 | 0.043 |
0.013a | 0.422 | 0.078 | 0.002 |
| High grade | 0.632 |
<0.001a | 0.832 | 0.120 | 0.081 |
| DJ-1 | - | 0.072 |
<0.001 | 0.872 | 0.170 |
| PTEN | | - | 0.014 | 0.375 | 0.141a |
| PI3K-p110α | | | - | 0.055 | 0.569 |
| pAkt | | | | - | 0.495 |
| HSP90α | | | | | - |
 | Table IIITwo by two comparisons of PI3K-p110α
and DJ-1, and PI3K-p110α and PTEN. |
Table III
Two by two comparisons of PI3K-p110α
and DJ-1, and PI3K-p110α and PTEN.
| A, Comparison of
PI3K-p110α and DJ-1a. |
|---|
|
|---|
| PI3K-p110α | − | + | Total |
|---|
| DJ-1 |
| − | 11 | 5 | 16 |
| + | 22 | 64 | 86 |
| Total | 33 | 69 | 102 |
|
| B, Comparison of
PI3K-p110α and PTENb. |
|
| PI3K-p110α | − | + | Total |
|
| PTEN |
| − | 17 | 20 | 37 |
| + | 16 | 49 | 65 |
| Total | 33 | 69 | 102 |
The histopathological characteristics associated
with DJ-1, PTEN and HSP90α staining intensity are shown in Table IV. The higher expression (score 2)
of DJ-1 and HSP90α was significantly associated with invasive
tumors (P=0.026 and P=0.004, respectively) compared to non-invasive
tumors (Ta). The loss of PTEN expression (score 0) was
significantly correlated with non-papillary, invasive and
high-grade tumors (P=0.005, P=0.034 and P<0.001,
respectively).
 | Table IVHistopathological characteristics of
DJ-1, PTEN and HSP90α staining intensity. |
Table IV
Histopathological characteristics of
DJ-1, PTEN and HSP90α staining intensity.
| Marker | Score | 0 (%) | 1 (%) | 2 (%) | Total | P-value |
|---|
| DJ-1 |
| Growth
pattern | | | | | | 0.657 |
| Papillary | | 11 (16.7) | 10 (15.1) | 45 (68.2) | 66 | |
|
Non-papillary | | 5 (13.9) | 8 (22.2) | 23 (63.9) | 36 | |
| Stage | | | | | | 0.026 |
| Ta | | 10 (29.4) | 5 (14.7) | 19 (55.9) | 34 | |
| T1-T3 | | 6 (8.8) | 13 (19.1) | 49 (72.1) | 68 | |
| Histological
grade | | | | | | 0.857 |
| Low grade | | 9 (17.6) | 9 (17.6) | 33 (64.8) | 51 | |
| High grade | | 7 (13.7) | 9 (17.6) | 35 (68.7) | 51 | |
| PTEN |
| Growth
pattern | | | | | | 0.005 |
| Papillary | | 17 (25.8) | 30 (45.5) | 19 (28.8) | 66 | |
|
Non-papillary | | 20 (55.6) | 13 (36.1) | 3 (8.3) | 36 | |
| Stage | | | | | | 0.034 |
| Ta | | 8 (23.5) | 14 (41.2) | 12 (35.3) | 34 | |
| T1-T3 | | 29 (42.6) | 29 (42.6) | 10 (14.7) | 68 | |
| Histological
grade | | | | | |
<0.001 |
| Low grade | | 9 (17.6) | 27 (52.9) | 15 (29.4) | 51 | |
| High grade | | 28 (54.9) | 16 (31.4) | 7 (13.7) | 51 | |
| HSP90α |
| Growth
pattern | | | | | | 0.294 |
| Papillary | | 6 (9.1) | 20 (30.3) | 40 (60.6) | 66 | |
|
Non-papillary | | 3 (8.3) | 6 (16.7) | 27 (75.0) | 36 | |
| Stage | | | | | | 0.004 |
| Ta | | 4 (11.8) | 15 (44.1) | 15 (44.1) | 34 | |
| T1-T3 | | 5 (7.4) | 11 (16.2) | 52 (76.5) | 68 | |
| Histological
grade | | | | | | 0.151 |
| Low grade | | 5 (9.8) | 17 (33.3) | 29 (56.9) | 51 | |
| High grade | | 4 (7.8) | 9 (17.6) | 38 (74.5) | 51 | |
Discussion
DJ-1 improves tumor cell survival by modulating the
Akt/PI3K axis (13,14), inhibiting apoptosis through
repression of the p53-Bax-caspase pathway (22) and stabilizing the antioxidant
transcriptional master regulator Nrf2 (23). Downregulation of DJ-1 by
transfection with small interfering RNA (siRNA) targets DJ-1
inhibited cell proliferation and enhances apoptosis of laryngeal
squamous cell carcinoma (SCC) Hep-2 cells (24). DJ-1 has been reported to be
overexpressed in several types of human cancer, including lung,
breast, pancreas and esophageal (13–16).
However, DJ-1 expression status in bladder tumors has not yet been
reported. In the present study, a high expression of DJ-1 was
detected in invasive tumors (T1-T3) compared to non-invasive tumors
(Ta) of the bladder. This result supports previous findings that
high levels of DJ-1 expression are associated with a higher
pathological tumor stage in SCCs of the esophagus and glottis
(14,24). These studies report that a higher
expression of DJ-1 was observed in pT3-pT4 glottic SCCs compared
with pTis-pT2 tumors (24) and in
T4 esophageal SCCs compared with T1-T3 tumors (14). Taken together, these results
indicate that a high expression of DJ-1 is associated with tumor
invasiveness in carcinomas.
Deletion of PTEN or reduced PTEN expression has been
well established in invasive UC (17,25,26). A
strong expression of PTEN observed in the nucleus and cytoplasm in
normal urothelium was lost or reduced in the nucleus and cytoplasm
in UCs in our study, which was in concordance with findings from
previous studies (17,27). Loss of PTEN expression was
significantly associated with non-papillary, invasive and
high-grade tumors. Similarly, the proportion of strong PTEN
expression positive cases was lower in non-papillary, invasive and
high-grade tumors compared to papillary, non-invasive and low-grade
tumors.
Mutation of PI3K-p110α, a catalytic subunit of PI3K,
has been found in a significant proportion of low-grade and low
stage UCs (28). In another study,
PI3K-p110α mutation was detected in 25% of UC samples and 26% of
UC-derived cell lines with mutations in a helical domain (29). PI3K-p110α was weakly expressed in
the membrane of normal urothelium and in the cytoplasm or membrane
of UC tumor cells, but showed no difference in immunoreactivity
according to the growth pattern, grade and stage of UC in our
study.
An activating mutation of Akt was identified in
renal pelvic UC (30) as well as
renal cell carcinoma (31) and
prostatic adenocarcinoma (32), but
not in urinary bladder UC (33). In
bladder UC, pAkt loss was described in invasive UC compared to
benign urothelium and non-invasive UC (26,27).
In the present study, pAkt was heterogeneously stained in the
cytoplasm and membrane of normal urothelium as well as UC tumor
cells. However, a significant loss or overexpression of pAkt was
not observed in UC according to the pathological
characteristics.
HSP90 overexpression is known to be correlated with
the evolution of a number of tumors, and HSP90 inhibitors, such as
geldanamycin derivatives, are currently undergoing clinical trials
for the treatment of advanced carcinomas of the breast, lung, colon
and esophagus, as well as melanoma and sarcomas (34–36).
In cases of UC, 17-allylamino-17-demethoxygeldanamycin (17-AAG) was
applied to human urinary bladder cancer cell lines that resulted in
cell cycle arrest and apoptosis by inducing the downregulation of
HSP90 client proteins (37). HSP90
expression in UC is not as well understood as in other tumors.
Cardillo et al (20)
reported that a higher level of HSP90 expression was shown in
high-grade and muscle-invasive UCs than in low-grade and
superficial UCs. Contrary to this result, Lebret et al
(19) demonstrated that a loss of
HSP90 expression was associated with the risk of developing an
infiltrating recurrence of UC. However, in their study, the sample
size of infiltrating recurrent tumors was limited to 5 cases and
the difference of pathological features according to the intensity
of HSP90 expression was not mentioned. Differential expression of α
and β isoforms of HSP90 was studied in gastrointestinal stromal
tumor (GIST), and it appears that HSP90α is more relevant to the
intrinsic aggressiveness of GIST (38). Initially, we used HSP90α and HSP90β
antibodies; however, HSP90β was not detected in any normal
urothelium or UC samples, thus HSP90β was excluded from this study
(data not shown). In the present study, the expression of HSP90α
was higher in invasive (T1–T3) compared to non-invasive tumors
(Ta), suggesting that a higher level of HSP90α is linked to
invasiveness in UC and supports the potential use of HSP90
inhibitors as an adjunctive therapeutic agent in invasive UC.
The interaction between DJ-1 and the PI3K/Akt
pathway genes was studied in experiments using Drosophila as
a model system. Yang et al (39) inhibited the function of a
Drosophila DJ-1 homologue (DJ-1A) by transgenic RNA
interference (RNAi) and DJ-1A RNAi flies exhibited an accumulation
of reactive oxygen species, photoreceptor neuronal loss and eye
degeneration. An enhancement of eye degeneration was observed when
PTEN was coexpressed with the DJ-1A RNAi transgene, whereas a clear
suppression of the DJ-1A RNAi phenotype was observed and the eyes
were restored to normal size when the wild-type form of the PI3K
catalytic subunit Dp110 was coexpressed. These authors also found
reduced phosphorylation of Akt in DJ-1A RNAi animals, suggesting an
impairment of PI3K/Akt signaling by DJ-1A downregulation. Our data
showed a positive correlation between DJ-1 and PI3K-p110α
expression, which concurred with the findings from the study by
Yang et al (39), although
the data did not show a negative correlation between the expression
of DJ-1 and PTEN, nor a positive correlation between the expression
of DJ-1 and pAkt. A positive correlation between the expression of
PI3K-p110α and PTEN in our data was noteworthy as PTEN exerts
enzymatic activity as a PIP3 phosphatase, opposing the activity of
PI3K (40). PTEN-inactivating and
PI3K-activating mutations is known to coexist in endometrial,
breast and colorectal cancer (41).
In UC, loss of PTEN has been associated with invasive behavior and
the mutation of PI3K has been reported in low-grade papillary
tumors. Our finding that 71% of PI3K-p110α-positive samples stained
positively for PTEN suggests that alteration of these proteins may
have non-canonical effects on the development of UC, as Platt et
al (29) previously proposed.
Platt et al demonstrated that overexpression of PI3K-p110α
does not necessarily involve amplification of the gene. Moreover,
no significant correlation was found between the alteration of PI3K
pathway members PI3K-p110α, TSC1 and PTEN, suggesting that the
mutation of these members may be independently distributed in
UC.
In conclusion, findings of our study have shown that
the overexpression of DJ-1 and HSP90α is associated with
invasiveness of UC, and that loss of PTEN is associated with
non-papillary histology, high-grade and invasive UCs. The
expression of PI3K-p110α is correlated with DJ-1 and PTEN
expression in UC. However, a correlation for DJ-1, HSP90α and PTEN
expression was not observed. Additional studies through mutational
analysis of these markers are required to better understand their
contribution to bladder tumorigenesis and molecular prognostic
markers.
Acknowledgements
We thank Deoksu Kim for technical support including
tissue microarray construction and immunohistochemistry.
References
|
1
|
Castillo-Martin M, Domingo-Domenech J,
Karni-Schmidt O, Matos T and Cordon-Cardo C: Molecular pathways of
urothelial development and bladder tumorigenesis. Urol Oncol.
28:401–408. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Morgensztern D and McLeod HL:
PI3K/Akt/mTOR pathway as a target for cancer therapy. Anticancer
Drugs. 16:797–803. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Alessi DR, James SR, Downes CP, et al:
Characterization of a 3-phosphoinositide-dependent protein kinase
which phosphorylates and activates protein kinase B alpha. Curr
Biol. 7:261–269. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Song G, Ouyang G and Bao S: The activation
of Akt/PKB signaling pathway and cell survival. J Cell Mol Med.
9:59–71. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Di Cristofano A and Pandolfi PP: The
multiple roles of PTEN in tumor suppression. Cell. 100:387–390.
2000.PubMed/NCBI
|
|
6
|
Whitesell L and Lindquist SL: HSP90 and
the chaperoning of cancer. Nat Rev Cancer. 5:761–772. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Xu W and Neckers L: Targeting the
molecular chaperone heat shock protein 90 provides a multifaceted
effect on diverse cell signaling pathways of cancer cells. Clin
Cancer Res. 13:1625–1629. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Millson SH, Truman AW, Racz A, et al:
Expressed as the sole Hsp90 of yeast, the alpha and beta isoforms
of human Hsp90 differ with regard to their capacities for
activation of certain client proteins, whereas only Hsp90beta
generates sensitivity to the Hsp90 inhibitor radicicol. FEBS J.
274:4453–4463. 2007. View Article : Google Scholar
|
|
9
|
Solit DB, Ivy SP, Kopil C, et al: Phase I
trial of 17-allylamino-17-demethoxygeldanamycin in patients with
advanced cancer. Clin Cancer Res. 13:1775–1782. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Basso AD, Solit DB, Chiosis G, Giri B,
Tsichlis P and Rosen N: Akt forms an intracellular complex with
heat shock protein 90 (Hsp90) and Cdc37 and is destabilized by
inhibitors of Hsp90 function. J Biol Chem. 277:39858–39866. 2002.
View Article : Google Scholar
|
|
11
|
Bonifati V, Rizzu P, Squitieri F, et al:
DJ-1 (PARK7), a novel gene for autosomal recessive, early onset
parkinsonism. Neurol Sci. 24:159–160. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Taira T, Saito Y, Niki T, Iguchi-Ariga SM,
Takahashi K and Ariga H: DJ-1 has a role in antioxidative stress to
prevent cell death. EMBO Rep. 5:213–218. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kim RH, Peters M, Jang Y, et al: DJ-1, a
novel regulator of the tumor suppressor PTEN. Cancer Cell.
7:263–273. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yuen HF, Chan YP, Law S, et al: DJ-1 could
predict worse prognosis in esophageal squamous cell carcinoma.
Cancer Epidemiol Biomarkers Prev. 17:3593–3602. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Le Naour F, Misek DE, Krause MC, et al:
Proteomics-based identification of RS/DJ-1 as a novel circulating
tumor antigen in breast cancer. Clin Cancer Res. 7:3328–3335.
2001.PubMed/NCBI
|
|
16
|
Tian M, Cui YZ, Song GH, et al: Proteomic
analysis identifies MMP-9, DJ-1 and A1BG as overexpressed proteins
in pancreatic juice from pancreatic ductal adenocarcinoma patients.
BMC Cancer. 8:2412008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Knowles MA, Platt FM, Ross RL and Hurst
CD: Phosphatidylinositol 3-kinase (PI3K) pathway activation in
bladder cancer. Cancer Metastasis Rev. 28:305–316. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Askham JM, Platt F, Chambers PA, Snowden
H, Taylor CF and Knowles MA: AKT1 mutations in bladder cancer:
identification of a novel oncogenic mutation that can co-operate
with E17K. Oncogene. 29:150–155. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lebret T, Watson RW, Molinie V, et al:
Heat shock proteins HSP27, HSP60, HSP70, and HSP90: expression in
bladder carcinoma. Cancer. 98:970–977. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cardillo MR, Sale P and Di Silverio F:
Heat shock protein-90, IL-6 and IL-10 in bladder cancer. Anticancer
Res. 20:4579–4583. 2000.PubMed/NCBI
|
|
21
|
Ebele JN, Sauter G, Epstein JI and
Sesterhenn IA; World Health Organization. Classification of Tumors.
Pathology and Genetics of Tumours of the Urinary System and Male
Genital Organs. IARC Press; Lyon: 2004
|
|
22
|
Fan J, Ren H, Jia N, et al: DJ-1 decreases
Bax expression through repressing p53 transcriptional activity. J
Biol Chem. 283:4022–4030. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Clements CM, McNally RS, Conti BJ, Mak TW
and Ting JP: DJ-1, a cancer- and Parkinson's disease-associated
protein, stabilizes the antioxidant transcriptional master
regulator Nrf2. Proc Natl Acad Sci USA. 103:15091–15096. 2006.
|
|
24
|
Zhu XL, Wang ZF, Lei WB, Zhuang HW, Jiang
HY and Wen WP: DJ-1: A novel independent prognostic marker for
survival in glottic squamous cell carcinoma. Cancer Sci.
101:1320–1325. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Puzio-Kuter AM, Castillo-Martin M, Kinkade
CW, et al: Inactivation of p53 and Pten promotes invasive bladder
cancer. Genes Dev. 23:675–680. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Harris LD, De La Cerda J, Tuziak T, et al:
Analysis of the expression of biomarkers in urinary bladder cancer
using a tissue microarray. Mol Carcinog. 47:678–685. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Schultz L, Albadine R, Hicks J, et al:
Expression status and prognostic significance of mammalian target
of rapamycin pathway members in urothelial carcinoma of urinary
bladder after cystectomy. Cancer. 116:5517–5526. 2010. View Article : Google Scholar
|
|
28
|
Lopez-Knowles E, Hernandez S, Malats N, et
al: PIK3CA mutations are an early genetic alteration associated
with FGFR3 mutations in superficial papillary bladder tumors.
Cancer Res. 66:7401–7404. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Platt FM, Hurst CD, Taylor CF, Gregory WM,
Harnden P and Knowles MA: Spectrum of phosphatidylinositol 3-kinase
pathway gene alterations in bladder cancer. Clin Cancer Res.
15:6008–6017. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Qian CN, Furge KA, Knol J, et al:
Activation of the PI3K/AKT pathway induces urothelial carcinoma of
the renal pelvis: identification in human tumors and confirmation
in animal models. Cancer Res. 69:8256–8264. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Pantuck AJ, Seligson DB, Klatte T, et al:
Prognostic relevance of the mTOR pathway in renal cell carcinoma:
implications for molecular patient selection for targeted therapy.
Cancer. 109:2257–2267. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
King JC, Xu J, Wongvipat J, et al:
Cooperativity of TMPRSS2-ERG with PI3-kinase pathway activation in
prostate oncogenesis. Nat Genet. 41:524–526. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yoo LI, Liu DW, Le Vu S, Bronson RT, Wu H
and Yuan J: Pten deficiency activates distinct downstream signaling
pathways in a tissue-specific manner. Cancer Res. 66:1929–1939.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ramanathan RK, Egorin MJ, Erlichman C, et
al: Phase I pharmacokinetic and pharmacodynamic study of
17-dimethylaminoethylamino-17-demethoxygeldanamycin, an inhibitor
of heat-shock protein 90, in patients with advanced solid tumors. J
Clin Oncol. 28:1520–1526. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Modi S, Stopeck A, Linden H, et al: HSP90
inhibition is effective in breast cancer: a phase II trial of
tanespimycin (17-AAG) plus trastuzumab in patients with
HER2-positive metastatic breast cancer progressing on trastuzumab.
Clin Cancer Res. 17:5132–5139. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Pacey S, Wilson RH, Walton M, et al: A
phase I study of the heat shock protein 90 inhibitor alvespimycin
(17-DMAG) given intravenously to patients with advanced solid
tumors. Clin Cancer Res. 17:1561–1570. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Karkoulis PK, Stravopodis DJ, Margaritis
LH and Voutsinas GE: 17-Allylamino-17-demethoxygeldanamycin induces
downregulation of critical Hsp90 protein clients and results in
cell cycle arrest and apoptosis of human urinary bladder cancer
cells. BMC Cancer. 10:4812010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li CF, Huang WW, Wu JM, et al: Heat shock
protein 90 overexpression independently predicts inferior
disease-free survival with differential expression of the alpha and
beta isoforms in gastrointestinal stromal tumors. Clin Cancer Res.
14:7822–7831. 2008. View Article : Google Scholar
|
|
39
|
Yang Y, Gehrke S, Haque ME, et al:
Inactivation of Drosophila DJ-1 leads to impairments of
oxidative stress response and phosphatidylinositol 3-kinase/Akt
signaling. Proc Natl Acad Sci USA. 102:13670–13675. 2005.PubMed/NCBI
|
|
40
|
Georgescu MM: PTEN tumor suppressor
network in PI3K-Akt pathway control. Genes Cancer. 1:1170–1177.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Yuan TL and Cantley LC: PI3K pathway
alterations in cancer: variations on a theme. Oncogene.
27:5497–5510. 2008. View Article : Google Scholar : PubMed/NCBI
|