Introduction
Angioimmunoblastic T-cell lymphoma (AITL) is a
distinct peripheral T-cell lymphoma (PTCL) subtype with uncommon
clinical and pathological features, accounting for 15–20% of PTCL
and 2% of all non-Hodgkin lymphoma (NHL). However, there is no
consensus with regard to the best approach for treating patients
with AITL and the disease often follows an aggressive clinical
course. AITL portends a poor prognosis, the median survival time is
less than three years and the five-year overall survival (OS) rate
is only approximately 30% in most studies, even when treated
intensively. However, AITL is not always lethal, with 30% of
patients being long-term survivors (1,2).
In the present study, the first case in which a
relapsed and refractory AITL patient is successfully treated with a
combination of bortezomib, mitoxantrone and dexamethasone (PAD
regimen) is reported. The diagnosis, clinical features and
bortezomib-based treatment of AITL according to this case are
described and previously published data are also discussed. The
study was approved by Ethical Principles for Conducting Research
With Humans and Other Animals committee of The Second Military
Medical University, Shanghai.
Case report
In September 2008, a 76-year-old female individual
first visited an otorhinolaryngology clinic with the chief
complaints of dyspnea and painful swelling of the throat. Physical
examination revealed that her bilateral tonsils were projected
beyond the midline with an irregular surface. Pharyngeal stenosis
and lymphoid follicular hyperplasia were observed in the posterior
wall of the pharynx. The bilateral cervical, axillary and inguinal
lymph nodes were enlarged with moderate hardness and limited
motion, as well as splenomegaly. A computed tomography (CT) scan of
the chest showed multiple swollen lymph nodes in the mediastinal
and bilateral axillary areas. Bone marrow cytomorphological
examination and biopsy at that time were normal. A diagnosis of
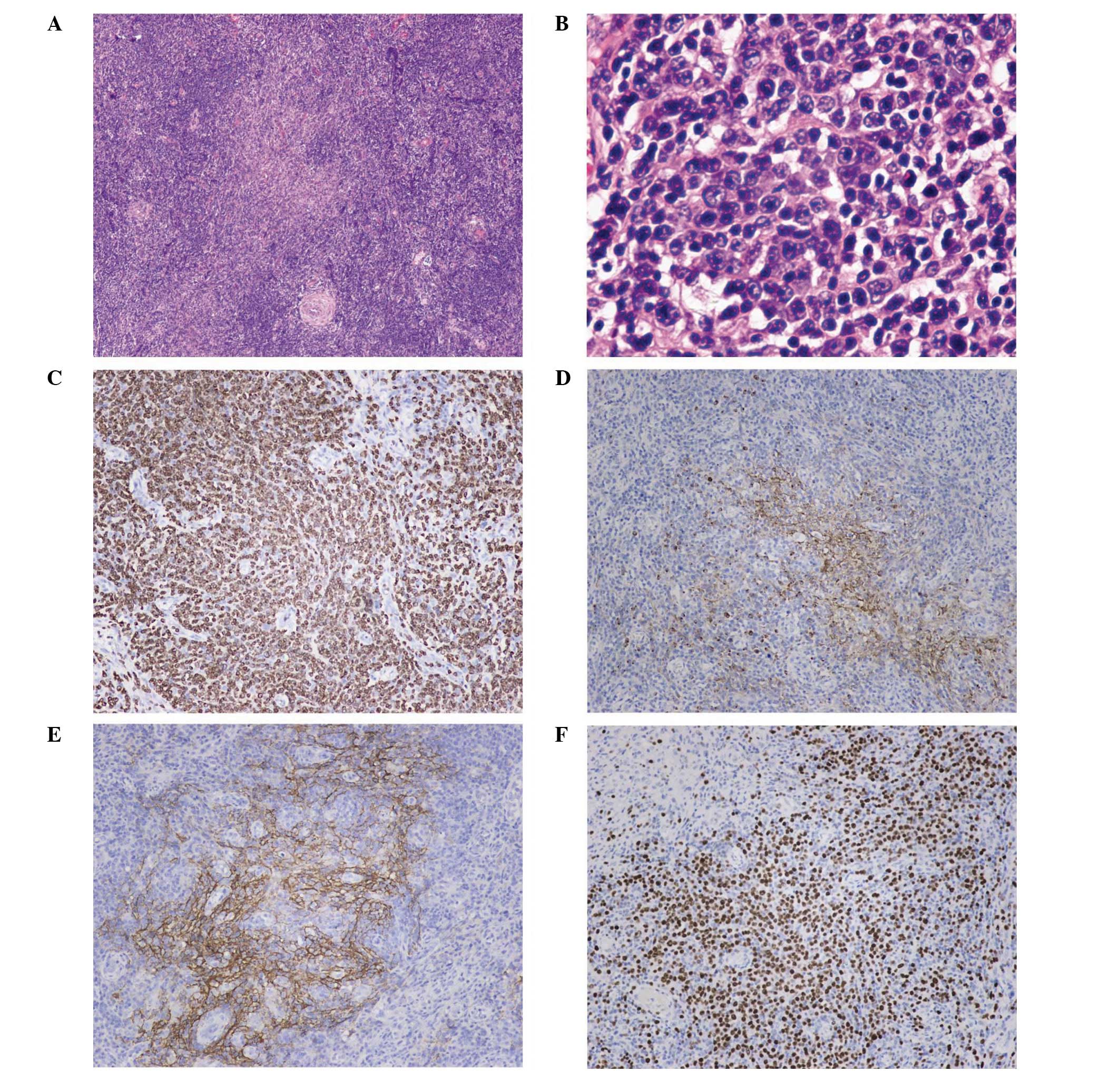
AITL was confirmed by biopsy of the enlarged cervical lymph node
and bilateral tonsils. Immunohistochemical staining of the tumor
cells showed CD4(+), CD8(+), CD10(±), Ki-67(+), CD15(+), CD30(−),
CD3(+), CD31+ (vascular), CD34+ (vascular),
CXCL13(+) and follicular dendritic cell meshworks expressing CD21
(Fig. 1). T-cell receptor (TCR) γ
gene rearrangement (+) and immunoglobulin (Ig) genes rearrangement
(−) of the lymph nodes were detected by the polymerase chain
reaction (PCR).
The patient was diagnosed with AITL IIIB. The
patient achieved a partial response to the initial treatment with
two cycles of CHOP (cyclophosphamide, doxorubicin, vincristine and
prednisolone) and three cycles of CHOEP (cyclophosphamide,
doxorubicin, vincristine, etoposide and prednisolone). However,
after four months the disease had progressed. The patient then
received two cycles of salvage therapy with EDCP (cisplatin,
etoposide, cyclophosphamide and dexamethasone). The patient also
received cyclosporine and thalidomide for two months. No response
was observed. Between March 2009 and July 2009, the patient
received five 28-day cycles of PAD regimen comprising 1.3
mg/m2 bortezomib, 2 mg/m2 mitoxantrone and 40
mg dexamethasone on days 1, 4, 8 and 11, after signed informed
consent was obtained. At the 18-month follow-up, the patient showed
persistent clinical improvement, weight gain, disappearance of
lymphadenopathy, improvement of performance status and no B
symptoms, and complete response was achieved. The patient did not
receive therapy until October 2010 when the patient was diagnosed
as relapsed by positron emission tomography (PET). Subsequently,
one cycle of COP and PAD were performed. However, severe
myelosuppression and minor therapeutic effects were observed. The
patient succumbed to a severe pulmonary infection in March
2011.
Discussion
AITL is a rare and aggressive malignancy which
affects elderly adults at an age ranging from 59 to 65 years,
clinically characterized by a sudden onset of constitutional
symptoms, lymphadenopathy, hepatosplenomegaly, skin rash, bone
marrow involvement, anemia (often hemolytic and Coombs-positive),
polyclonal hypergammaglobulinemia and hypereosinophilia. Using PCR
techniques, the detection of monoclonal or oligoclonal
rearrangement of the TCR was found to be present in the majority of
cases. In addition, the rearrangement of the Ig gene(s) was
observed in up to one-third of patients (3). As yet, the standard therapeutic option
for patients with AITL has not been clearly established. Various
treatment strategies, which range from the CHOP or CHOP-like
therapy to chemotherapy with autologous stem cell transplantation,
even allogeneic transplantation, have proved to be largely
unsuccessful in curing the disease. The lack of a demonstrated
improvement in OS supports the development of novel approaches,
including alemtuzumab (4),
zanolimumab (5), denileukin
diftitox (6), pralatrexate
(7), histone deacetylase (HDAC)
inhibitors (8), cyclosporine
(9), thalidomide (10), gemcitabine (11) and bortezomib (12).
Bortezomib is a dipeptide boronic acid that
selectively and potently inhibits the proteasome 26S complex,
exerting antitumor activity mainly via the inhibition of the NF-κB
pathway components correlated with cell proliferation, apoptosis
and angiogenesis (13). It has been
reported that abnormal PRDM1β expression reflected poor prognosis
in T-cell lymphoma, and that bortezomib is able to downregulate
PRDM1β through NF-κB inactivation (14). In vitro and in vivo
studies demonstrated that bortezomib has the ability to eliminate
tumor cell interaction with endothelial cells in T-cell
leukemia/lymphoma by affecting the expression of genes associated
with the adhesion cascades (15).
Therefore, bortezomib may play a special therapeutic role and be
safe from the emergence of acute toxicity, which is important
considering that AITL patients tend to be elderly adults with a
number of concomitant medical problems.
A PAD regimen is commonly used to treat patients
with multiple myeloma. To the best of our knowledge, this is the
first reported case of the successful management of relapsed and
refractory AITL with a combination of bortezomib, mitoxantrone and
dexamethasone (PAD regimen). This combination was well tolerated
and induced a complete and relatively sustained remission in this
patient. We believe bortezomib was involved in the therapy of this
patient since no response to anthracyclines and high-dose
glucocorticoid was observed. Martínez-Delgado et al
differentiated two subgroups of PTCL characterized by a high or
reduced expression of the NF-κB pathway genes (16). Furthermore, Martínez-Delgado et
al observed that a low expression of the NF-κB pathway genes
was significantly associated with short OS and may be an
independent prognostic factor. Bortezomib not only acted on tumor
cells themselves through NF-κB inactivation, but also removed tumor
cell interaction with endothelial cells (14,15).
Therefore, we suggest that NF-κB-positive AITL patients treated
with bortezomib have better results to treatment than
NF-κB-negative AITL patients.
Considering that heavy chemotherapy may cause
serious adverse events in elderly adults, the use of the PAD
regimen may be a reliable, safe and effective alternative for
treating relapsed/refractory AITL. However, the efficacy of
bortezomib should be elucidated in further larger-sized clinical
trials with an extended follow-up. Furthermore, continued
preclinical and clinical research is required to identify more
effective treatment options for patients with relapsed/refractory
AITL.
References
|
1
|
Vose J, Armitage J and Weisenburger D;
International T-Cell Lymphoma Project. International peripheral
T-cell and natural killer/T-cell lymphoma study: pathology findings
and clinical outcomes. J Clin Oncol. 26:4124–4130. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mourad N, Mounier N, Brière J, Raffoux E,
Delmer A, Feller A, Meijer CJ, Emile JF, Bouabdallah R, Bosly A,
Diebold J, Haioun C, Coiffier B, Gisselbrecht C and Gaulard P;
Groupe d’Etude des Lymphomes de l’Adulte. Clinical, biologic, and
pathologic features in 157 patients with angioimmunoblastic T-cell
lymphoma treated within the Groupe d’Etude des Lymphomes de
l’Adulte (GELA) trials. Blood. 111:4463–4470. 2008.PubMed/NCBI
|
|
3
|
de Leval L, Gisselbrecht C and Gaulard P:
Advances in the understanding and management of angioimmunoblastic
T-cell lymphoma. Br J Haematol. 148:673–689. 2010.PubMed/NCBI
|
|
4
|
Gallamini A, Zaja F, Patti C, Billio A,
Specchia MR, Tucci A, Levis A, Manna A, Secondo V, Rigacci L, et
al: Alemtuzumab (Campath-1H) and CHOP chemotherapy as first-line
treatment of peripheral T-cell lymphoma: results of a GITIL (Gruppo
Italiano Terapie Innovative nei Linfomi) prospective multicenter
trial. Blood. 110:2316–2323. 2007. View Article : Google Scholar
|
|
5
|
d’Amore F, Radford J, Relander T, Jerkeman
M, Tilly H, Osterborg A, Morschhauser F, Gramatzki M, Dreyling M,
Bang B and Hagberg H: Phase II trial of zanolimumab (HuMax-CD4) in
relapsed or refractory non-cutaneous peripheral T cell lymphoma. Br
J Haematol. 150:565–573. 2010.PubMed/NCBI
|
|
6
|
Dang NH, Pro B, Hagemeister FB, Samaniego
F, Jones D, Samuels BI, Rodriguez MA, Goy A, Romaguera JE,
McLaughlin P, et al: Phase II trial of denileukin diftitox for
relapsed/refractory T-cell non-Hodgkin lymphoma. Br J Haematol.
136:439–447. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
O’Connor OA, Pro B, Pinter-Brown L,
Bartlett N, Popplewell L, Coiffier B, Lechowicz MJ, Savage KJ,
Shustov AR, Gisselbrecht C, et al: Pralatrexate in patients with
relapsed or refractory peripheral T-cell lymphoma: results from the
pivotal PROPEL study. J Clin Oncol. 29:1182–1189. 2011.
|
|
8
|
Piekarz RL, Frye R, Prince HM, Kirschbaum
MH, Zain J, Allen SL, Jaffe ES, Ling A, Turner M, Peer CJ, et al:
Phase 2 trial of romidepsin in patients with peripheral T-cell
lymphoma. Blood. 117:5827–5834. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Advani R, Horwitz S, Zelenetz A and
Horning SJ: Angioimmunoblastic T cell lymphoma: treatment
experience with cyclosporine. Leuk Lymphoma. 48:521–525. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gottardi M, Danesin C, Canal F, Dei Tos
AP, Stefani PM, Calistri E, Salvadori U and Gherlinzoni F: Complete
remission induced by thalidomide in a case of angioimmunoblastic
T-cell lymphoma refractory to autologous stem cell transplantation.
Leuk Lymphoma. 49:1836–1838. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Arkenau HT, Chong G, Cunningham D, Watkins
D, Sirohi B, Chau I, Wotherspoon A, Norman A, Horwich A and Matutes
E: Gemcitabine, cisplatin and methylprednisolone for the treatment
of patients with peripheral T-cell lymphoma: the Royal Marsden
Hospital experience. Haematologica. 92:271–272. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lee J, Suh C, Kang HJ, Ryoo BY, Huh J, Ko
YH, Eom HS, Kim K, Park K and Kim WS: Phase I study of proteasome
inhibitor bortezomib plus CHOP in patients with advanced,
aggressive T-cell or NK/T-cell lymphoma. Ann Oncol. 19:2079–2083.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sunwoo JB, Chen Z, Dong G, Yeh N, Crowl
Bancroft C, Sausville E, Adams J, Elliott P and Van Waes C: Novel
proteasome inhibitor PS-341 inhibits activation of nuclear
factor-kappa B, cell survival, tumor growth, and angiogenesis in
squamous cell carcinoma. Clin Cancer Res. 7:1419–1428.
2001.PubMed/NCBI
|
|
14
|
Zhao WL, Liu YY, Zhang QL, Wang L, Leboeuf
C, Zhang YW, Ma J, Garcia JF, Song YP, Li JM, et al: PRDM1 is
involved in chemoresistance of T-cell lymphoma and down-regulated
by the proteasome inhibitor. Blood. 111:3867–3871. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shi WY, Wang L, Xiao D, Yao Y, Yang F,
Jiang XX, Leboeuf C, Janin A, Chen SJ and Zhao WL: Proteasome
inhibitor bortezomib targeted tumor-endothelial cell interaction in
T-cell leukemia/lymphoma. Ann Hematol. 90:53–58. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Martínez-Delgado B, Cuadros M, Honrado E,
Ruiz de la Parte A, Roncador G, Alves J, Castrillo JM, Rivas C and
Benítez J: Differential expression of NF-kappaB pathway genes among
peripheral T-cell lymphomas. Leukemia. 19:2254–2263.
2005.PubMed/NCBI
|