Introduction
Hepatic function is one of the most significant
indices for cancer patients who undergo chemoradiation treatment
(1,2). The patients were required to take a
biochemical test prior to radiotherapy or chemotherapy. Patients
with hepatic dysfunction were not permitted to continue the
treatment.
Hepatitis B is a common infectious disease in Asia,
particularly in China (3–5). Approximately 10% of the Chinese
population are hepatitis B virus (HBV) carriers and numerous
patients are likely to develop hepatic cirrhosis and liver cancer
(6,7). A significant proportion of hepatic
cancer patients also present with hepatitis B; however, there are
numerous patients with non-hepatic cancer who also carry hepatitis
B in China. The latter patients were also found to be more likely
to develop hepatic dysfunction during the process of radiotherapy
or chemotherapy (8–11).
Previously, numerous studies focused on patients
with lymphoma and hepatitis B treated with rituximab (12–14).
Few studies investigated other patients with other types of cancer
with hepatitis B. Cheng et al (1) studied 62 postgastrectomy patients
treated with concomitant chemoradiotherapy (CCRT) and observed that
HBV infection was the only independent factor associated with
chemoradiation-induced liver disease. However, only 11 patients
were HBV carriers and all the patients were treated with
postgastrectomy CCRT. Since hepatic function is extremely important
for cancer patients, this study aims to investigate the factors
associated with hepatic function in a larger population of
postgastrectomy cancer patients carrying HBV.
Patients and methods
Patient population and
characteristics
Patients with stage IB-IV M0 gastric
adenocarcinoma carrying HBV who underwent total or subtotal
gastrectomy and regional lymph node dissection and were treated in
the Abdominal Cancer Department of the West China Hospital
(Chengdu, China) between October 2006 and October 2010 were
reviewed. The clinical staging system of gastric cancer (AJCC 2005
version) was adopted for this study. This study was approved by the
Ethics Committee of Sichuan University. All patients gave informed
consent to participate in the study. Patients who had hepatic
dysfunction, HBV activation or hepatic cirrhosis prior to treatment
were excluded from this study. Patients who failed to complete the
radiotherapy for reasons other than hepatic dysfunction or HBV
activation were also excluded. In total 44 patients remained who
were confirmed to have gastric adenocarcinoma by the Pathological
Department of our hospital. The patients were diagnosed as HBV
carriers by the Laboratory Department of our hospital with serum
HBV DNA <5 pg/ml. Patients with grade I hepatic dysfunction who
recovered soon after completing treatment were also included. The
characteristics of these patients are listed in Table I.
 | Table IPatient characteristics (n=44). |
Table I
Patient characteristics (n=44).
| Patient
characteristics | No. of patients
(%) |
|---|
| Discrete
variables |
| Gender |
| Male | 28 (63.64) |
| Female | 16 (36.36) |
| AJCC stage |
| IB | 4 (9.09) |
| II | 7 (15.91) |
| III | 18 (40.91) |
| IV | 15 (34.09) |
| Radiotherapy |
| Yes | 17 (38.64) |
| No | 27 (61.36) |
| Chemotherapy
cycles |
| <6 | 26 (59.09) |
| ≥6 | 18 (40.91) |
| Continuous
variables |
| Age (years), median
(range) | 56 (35–73) |
Chemotherapy
A total of 44 gastric cancer patients received
postgastrectomy chemotherapy: CF, FOLFIRI, XELOX and EOF regimens
were each used in four patients; the PTX+DDP+5-F protocol was used
in two; the ECF regimen was used in three patients; mFOLFOX 6,
mFOLFOX 7 or FOLFOX 4 regimens were used in the remaining patients.
For the patients who received CCRT, the dosage of chemotherapy was
reduced by 0–20% following the start of radiotherapy according to
the reaction of the patient.
Radiotherapy
A total of 16 patients received CCRT. In general,
the radiotherapy began with the second cycle of chemotherapy.
Intensity modulated radiotherapy (IMRT) or image-guided
radiotherapy (IGRT) were used in all the patients. The radiation
techniques were introduced in previous studies (15). Briefly, the patients were
immobilized in the Stereotactic Body Frame (Elekta, Stockholm,
Sweden), which uses a vacuum pillow in a rigid frame, with an
abdominal compression device. CT-guided simulation was performed
and the gross tumor volume (GTV) was defined. The target and normal
adjacent structures were contoured on the planning CT scan.
Multileaf collimator (MLC) blocking was used to block normal
tissues outside of the intended targeted tissues. Treatment was
delivered once daily with 1.8–2.0 Gy, five fractions per week by a
6-MV linear accelerator. For the patients who received CCRT, the
median dose was 50.4 Gy (range, 45–50.4). The clinical target
volume (CTV) included the preoperative stomach volume, surgical
bed, gastric remnant and perigastric lymph nodes. Other lymph node
areas, including mediastinal, porta hepatis, splenic hilum,
pancreaticoduodenal and peripancreatic, were included if they were
at risk based on the primary tumor location or pathological
involvement of the lymph nodes. With regard to the bowel, the
intestinal loops outside the planning treatment volume (PTV) were
contoured, but not the whole abdominal space. To account for daily
setup error and organ motion, the CTV to PTV expansion was
typically 5–10 mm. Normal structures were also contoured, including
kidneys, liver, spinal cord and bowel. The mean hepatic dose and
dose to 30% volume of liver (V30) was maintained at <30 Gy
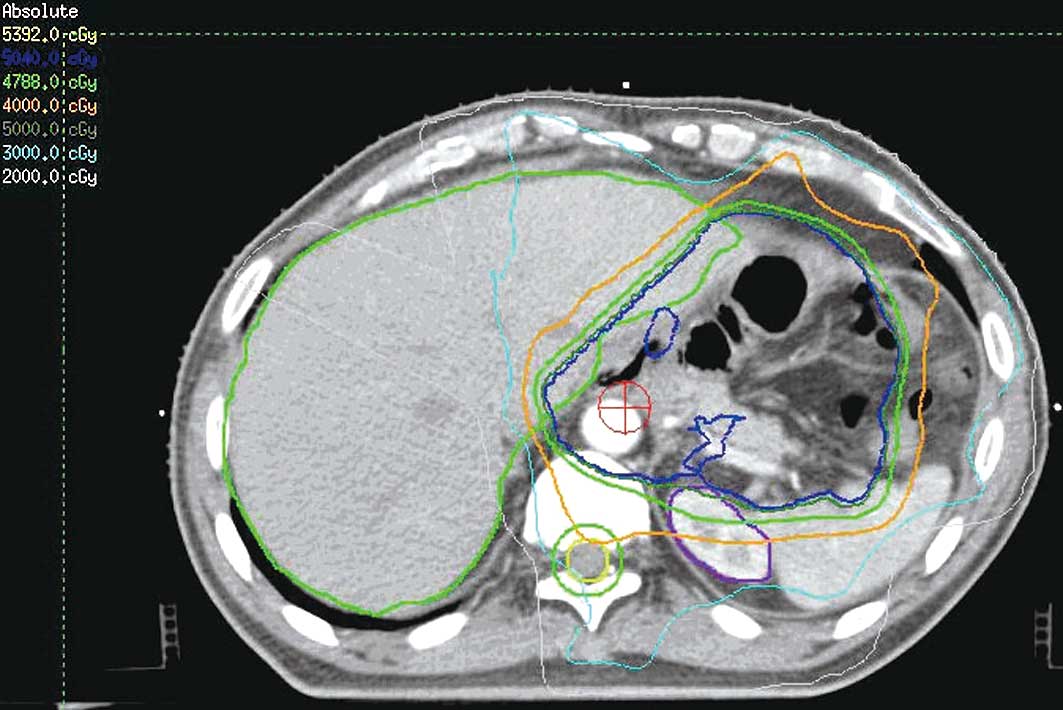
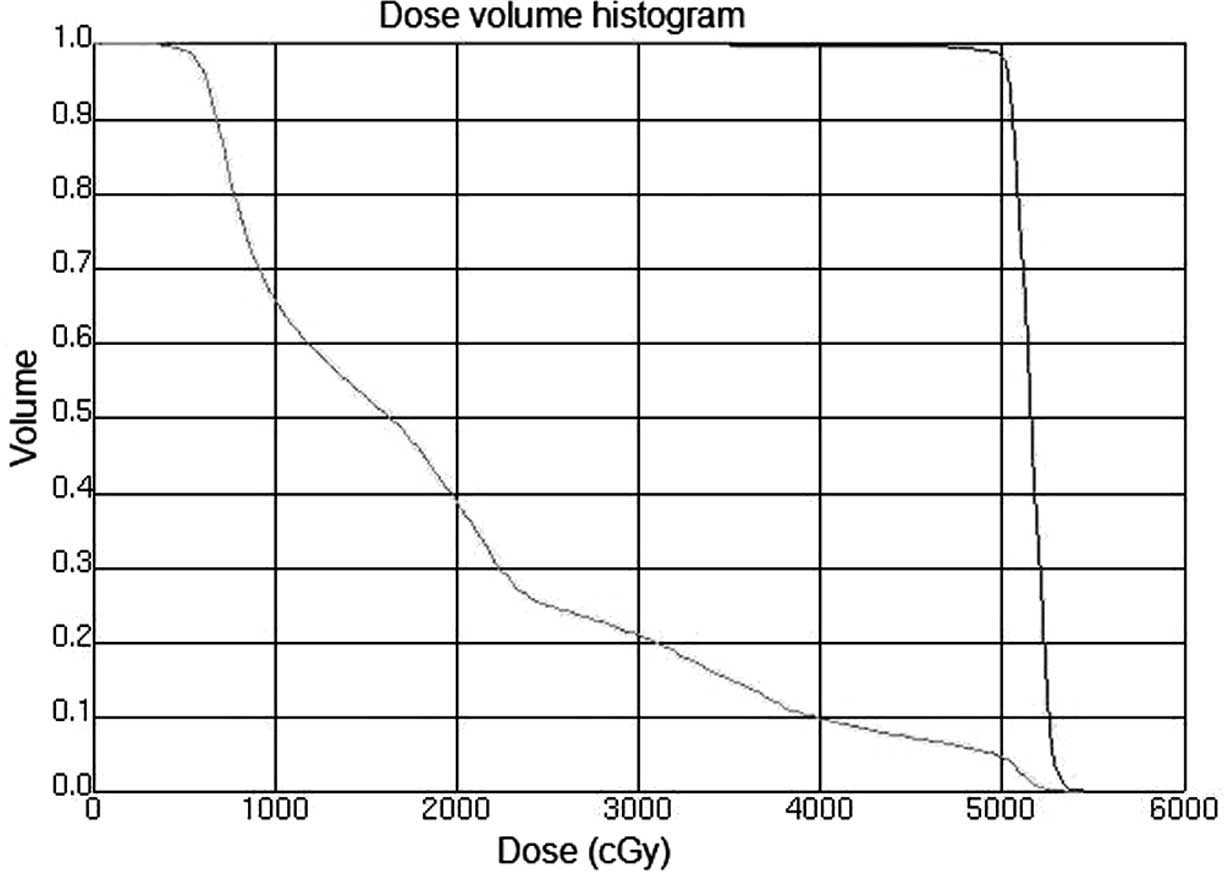
(V30<30%). Figs. 1 and 2 show the CTV delineation and dose volume
histograms (DVHs) for the PTV of one representative patient treated
with the IMRT technique, respectively.
Follow-up
The patients were examined weekly during the
treatment period, involving taking patient history, physical
examination, blood test and biochemical test, and were followed up
every month after finishing the treatment for at least 3 months.
HBV DNA copies were examined at the beginning of the treatment and
repeated for the patients who had been examined for hepatic
dysfunction during the treatment. The cases of hepatic dysfunction
were recorded according to the WHO guideline and levels of serum
HBV DNA >5 pg/ml were defined as HBV reactivation (1). The patients who had serious hepatic
dysfunction had received corresponding treatment, including
tiopronin, glutathione, multi-phosphatidylcholine and vitamin B.
Patients who had HBV DNA reactivation had received lamivudine
treatment for at least half a year, besides which, the patients did
not receive any other treatments. All 44 patients completed
treatment by July 2011.
Statistical analysis
The SPSS software program (version 16.0; SPSS Inc.,
Chicago, IL, USA) was used for all statistical analyses. For the
univariate analysis, a Student’s t-test was used to compare
continuous variables between the patients with or without hepatic
dysfunction and a Chi-square test was used to compare discrete
variables. Logistic regression analysis was used to investigate the
multivariable impact on the occurrence of hepatic dysfunction.
P<0.05 was considered to indicate a statistically significant
result.
Results
Patient characteristics
Following treatment, 17 patients developed hepatic
dysfunction. The characteristics of these patients are listed in
Table II. HBV had reactivated in
eight out of 17 patients, five of whom had received CCRT. Findings
of the univariate analysis showed that only radiotherapy was a
significant associating factor of patients who had developed
hepatic dysfunction (P=0.001). Other factors that were not
statistically significant include gender (P=0.165), age (P=0.393),
AJCC stage (P=0.377) and chemotherapy cycles (P=0.203; Table III). Results of the multivariate
analysis showed that only radiotherapy was a significant
associating factor for patients who had also developed hepatic
dysfunction [odds ratio, 10.560; 95% confidence interval (CI),
2.539–43.918; P=0.001; Table
IV].
 | Table IICharacteristics of patients with
hepatic dysfunction (n=17). |
Table II
Characteristics of patients with
hepatic dysfunction (n=17).
| Patient
characteristics | No. of patients
(%) |
|---|
| Discrete
variables |
| Gender | |
| Male | 13 (76.47) |
| Female | 4 (23.53) |
| AJCC stage |
| IB | 0 (0) |
| II | 4 (23.53) |
| III | 10 (58.82) |
| IV | 3 (17.65) |
| Radiotherapy |
| Yes | 12 (70.59) |
| No | 5 (29.41) |
| Chemotherapy
cycles |
| <6 | 8 (47.06) |
| ≥6 | 9 (52.94) |
| Virus
reactivation |
| Yes | 8 (47.06) |
| No | 9 (52.94) |
| Continuous
variables |
| Age (years), median
(range) | 51 (35–72) |
 | Table IIIUnivariate analysis of patients who
developed hepatic dysfunction. |
Table III
Univariate analysis of patients who
developed hepatic dysfunction.
| Patient
characteristics | No. of patients
(%) | P-value |
|---|
| Discrete
variables |
| Gender |
| Male | 28 (63.64) | 0.165 |
| Female | 16 (36.36) | |
| AJCC stage |
| IB | 4 (9.09) | 0.377 |
| II | 7 (15.91) | |
| III | 18 (40.91) | |
| IV | 15 (34.09) | |
| Radiotherapy |
| Yes | 17 (38.64) | 0.001 |
| No | 27 (61.36) | |
| Chemotherapy
cycles |
| <6 | 26 (59.09) | 0.203 |
| ≥6 | 18 (40.91) | |
| Continuous
variables |
| Age (years),
median (range) | 56 (35–73) | 0.393 |
 | Table IVMultivariate analysis of patients who
developed hepatic dysfunction. |
Table IV
Multivariate analysis of patients who
developed hepatic dysfunction.
| | | | | | | 95% CI for
Exp(B) |
|---|
| | | | | | |
|
|---|
| Factor | B | SE | Wald | df | Sig. | Exp(B) | Lower | Upper |
|---|
| Radiotherapy | 2.357 | 0.727 | 10.507 | 1 | 0.001 | 10.560 | 2.539 | 43.918 |
Dosimetric characteristics
To determine the internal dosimetric correlation
with hepatic dysfunction, we listed the dosimetric characteristics
of the 16 patients who received radiotherapy, including normal
liver volume, isocenter dose, V30 and mean liver dose (mean ± SD;
Table V). A Student’s t-test was
used to analyze the correlation between the dosimetric
characteristics and hepatic dysfunction and we observed that mean
liver dose was a significant associating factor (P=0.002; Table V).
 | Table VDosimetric characteristics and
univariate analysis (Student’s t-test) of patients who received
radiotherapy (n=16). |
Table V
Dosimetric characteristics and
univariate analysis (Student’s t-test) of patients who received
radiotherapy (n=16).
| Variable | Mean ± SD | P-value |
|---|
| Normal liver volume
(ml) | 1266±284 | 0.834 |
| Isocenter dose
(Gy) | 50.7±2.8 | 0.198 |
| V30 (%) | 26.4±2.4 | 0.730 |
| Mean liver dose
(cGy) | 2133±139 | 0.002 |
Severe hepatic dysfunction
A total of five patients developed severe (grade III
or IV) hepatic dysfunction in the present study (Table VI). The five patients were male,
aged 35–71 years old and the AJCC stages were either stage II or
IIIb. The patients had received radiotherapy and the number of
chemotherapy cycles ranged from 4 to 7, including EOF, mFOLFOX6 and
mFOLFOX7 regimens. In these five patients, the hepatic dysfunctions
were mainly enhanced aminotransferase levels, including alanine
aminotransferase (ALT) and aspartate aminotransferase (AST). The
bilirubins appeared almost normal. The HBV was reactivated in these
five patients, all of whom recovered following lamivudine
treatment.
 | Table VICharacteristics of five patients who
developed grade III or IV hepatic dysfunction. |
Table VI
Characteristics of five patients who
developed grade III or IV hepatic dysfunction.
| Patient no. |
|---|
|
|
|---|
|
Characteristics | 1 | 2 | 3 | 4 | 5 |
|---|
| Gender | M | M | M | M | M |
| Age (years) | 43 | 41 | 43 | 35 | 71 |
| AJCC stage | IIIb | II | IIIb | II | IIIb |
| Radiotherapy | Yes | Yes | Yes | Yes | Yes |
| Chemo.
regimens | mFOLFOX6 | EOF | mFOLFOX7 | mFOLFOX7 | mFOLFOX7 |
| Chemo. cycles | 7 | 4 | 5 | 7 | 5 |
| Isocenter dose
(Gy) | 50.4 | 50.4 | 50.4 | 50.4 | 50.4 |
| Liver volume
(ml) | 1297 | 875 | 1410 | 1177 | 1420 |
| Mean liver dose
(cGy) | 2137 | 2313 | 2181 | 2164 | 2191 |
| V30 (%) | 26 | 22 | 24 | 25 | 27 |
| ALT (IU/l) | 538 | 245 | 326 | 341 | 240 |
| AST (IU/l) | 315 | 265 | 294 | 209 | 210 |
| Tbil (μmol/l) | N | N | N | N | N |
| Dbil (μmol/l) | 9.1 | N | 9.6 | N | N |
| HBV DNA
reactivation | Yes | Yes | Yes | Yes | Yes |
| Lamivudine
treatment | Yes | Yes | Yes | Yes | Yes |
| Outcome of
dysfunction | R | R | R | R | R |
Discussion
Chronic HBV infection is a major public health
problem in Asian countries and the reactivation of HBV prior to or
following treatment with immunosuppressive or anticancer agents has
been documented since 1975 (6,
16). The most commonly reported
types of chemotherapy associated with HBV reactivation are those
used for the treatment of hematological malignancies, including
acute leukemia, myeloproliferative disorders, lymphoproliferative
disorders and plasma cell dyscrasias (17–19).
One of the most frequently studied examples is HBV reactivation in
lymphoma treated with rituximab since it is able to specifically
induce the apoptosis of B lymphocytes and leads to a marked
immunosuppression in patients. However, few studies concerning the
effect of chemotherapy on HBV reactivation in other cancer
patients, including gastric cancer, have been performed.
Radiotherapy, as a locoregional treatment, may also
cause human immunosuppression (1,11,20,21).
Therefore radiotherapy may affect HBV reactivation in the treatment
of cancer patients, especially in patients with liver located in
the radiation field. Cheng et al (11) compared 65 HBV-related hepatocellular
cancer (HCC) patients and 24 other HCC patients treated with
three-dimensional conformal radiotherapy (3D-CRT) and found that
the HCC patients who were HBV carriers presented with a
statistically significantly greater susceptibility to
radiation-induced liver disease following 3D-CRT. Kim et al
(22) compared 32 HBV-related HCC
patients treated with 3D-CRT and 43 HBV-related HCC patients
without any treatment and observed that the cumulative rate of HBV
reactivation was significantly greater in the treatment group. In
the radiotherapy treatment of gastric cancer, part of the liver is
located in the radiation field. Investigation into the influence of
radiotherapy on HBV reactivation in gastric cancer patients is
needed.
Chemoradiation treatment is unable to cause HBV
reactivation in all the cancer patients who have hepatitis B
(16,23) and the patients who did not develop
HBV reactivation may also have hepatic dysfunction. Therefore, we
studied the associating factors of hepatic function in 44
postgastrectomy patients who carried HBV and were treated in our
department.
In the present study, 17 patients developed hepatic
dysfunction, five of whom were of grade III or IV. The five
patients had received CCRT, thus of the total 16 patients that
received CCRT, the rate of patients that developed grade III or IV
hepatic dysfunction was 31.25% (5/16). This rate is lower than that
observed in the study by Cheng et al (1), which was 54.55% (6/11). In the present
study, HBV was reactivated in eight patients, five of whom had
received CCRT, thus of the 16 patients that received CCRT, the rate
of patients who had HBV reactivated was 31.25% (5/16) in comparison
to the incidence rate of 36.36% (4/11) in the study by Cheng et
al (1). Therefore the study by
those authors (1) had a higher
severe hepatic dysfunction rate and HBV reactivation rate than our
study. When comparing the treatment differences between the present
study and that by Cheng et al, we observed that although we
had used the IMRT technique, the rates of the isocenter dose, dose
to the liver and V30 were higher in our study than those reported
Cheng et al (1). Therefore,
we suggest that this difference is due to the majority of the
patients from the study by Cheng et al undergoing
neoadjuvant chemotherapy (93.55%), which none of our patients
received. However, of the 51 HBV-negative postgastrectomy patients
who received CCRT in the study by Cheng et al (1), the rate of grade III or IV hepatic
dysfunction was 3.92% (2/51), which was markedly lower than the
incidence rate in the present study. Therefore, we suggest that HBV
is a significant factor that causes severe hepatic dysfunction.
In the univariate and multivariate analyses,
radiotherapy was the only significant associating factor of hepatic
function in the 44 patients. In the study by Cheng et al
(1), radiotherapy was not suggested
to be an associating factor since their patients had received CCRT.
In the present study, chemotherapy cycle was not found to be a
significant associating factor which was similar to the findings of
Cheng et al (1). However,
whether chemotherapy or different chemotherapy regimens were
significant associating factors requires further investigation
since patients in the current study had received postgastrectomy
chemotherapy and most of the chemotherapy regimens were similar.
Although radiotherapy was an independent risk factor of hepatic
function, it remains unclear whether radiation to the liver was the
main cause. However, we suggest excluding the liver from radiation
field.
Since radiotherapy was the only significant
associating factor of hepatic function in this study, we studied
the dosimetric difference of 16 patients who had received CCRT. The
characteristics of these 16 patients are listed in Table V and we observed that the mean liver
dose was a significant associating factor of hepatic function using
the Student’s t-test (P=0.002). However, there were only 16
patients who had received radiotherapy, thus it is difficult to
reach a firm conclusion.
The five patients who developed grade III or IV
hepatic dysfunction received CCRT and also had HBV reactivated. In
the study by Cheng et al (1), the HBV status was the only significant
associating factor correlated with the grade III or IV hepatic
dysfunction. Therefore we suggest that HBV reactivation played a
significant role in the development of serious hepatic dysfunction.
The major abnormalities of these five patients were
aminotransferase activity although the bilirubin levels were rarely
affected.
In the present study, the patients who had HBV
reactivated had received lamivudine treatment regularly and
recovered following treatment. HBV reactivation is a serious
complication for hepatitis B patients and can occasionally be fatal
(10,24,25),
therefore it is vital to take regular antiviral treatment.
Lamivudine is the most commonly used drug for HBV reactivation
(26,27). However, results of numerous studies
have indicated that entecavir may be more effective (28–30).
In conclusion, radiotherapy was an independent risk
factor of hepatic function in postgastrectomy adenocarcinoma
patients positive for HBV. We suggest excluding the liver from the
radiation field. HBV is important in the development of severe
hepatic dysfunction and patients in whom HBV has reactivated should
take immediate and regular antiviral treatment.
Acknowledgements
This study was supported by grants from the National
Basic Research Program of China (No. 2011CB935800).
References
|
1
|
Cheng JC, Liu MC, Tsai SY, Fang WT,
Jer-Min Jian J and Sung JL: Unexpectedly frequent hepatitis B
reactivation by chemoradiation in postgastrectomy patients. Cancer.
101:2126–2133. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Stange MA, Tutarel O, Pischke S, Schneider
A, Strassburg CP, Becker T, Barg-Hock H, Basturk M, Wursthorn K,
Cornberg M, et al: Fulminant hepatic failure due to
chemotherapy-induced hepatitis B reactivation: role of rituximab. Z
Gastroenterol. 48:258–263. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lubel JS and Angus PW: Hepatitis B
reactivation in patients receiving cytotoxic chemotherapy:
diagnosis and management. J Gastroenterol Hepatol. 25:864–871.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Koo YX, Tan DS, Tan IB, Tao M, Chow WC and
Lim ST: Hepatitis B virus reactivation and role of antiviral
prophylaxis in lymphoma patients with past hepatitis B virus
infection who are receiving chemoimmunotherapy. Cancer.
116:115–121. 2010.PubMed/NCBI
|
|
5
|
Lim SM, Jang JW, Kim BW, Choi H, Choi KY,
Park SJ and Han CW: Hepatitis B virus reactivation during
chlorambucil and prednisolone treatment in an HBsAg-negative and
anti-HBs-positive patient with B-cell chronic lymphocytic leukemia.
Korean J Hepatol. 14:213–218. 2008.(In Korean).
|
|
6
|
Wu JM, Huang YH, Lee PC, Lin HC and Lee
SD: Fatal reactivation of hepatitis B virus in a patient who was
hepatitis B surface antigen negative and core antibody positive
before receiving chemotherapy for non-Hodgkin lymphoma. J Clin
Gastroenterol. 43:496–498. 2009. View Article : Google Scholar
|
|
7
|
Liu CJ, Chen PJ, Chen DS and Kao JH:
Hepatitis B virus reactivation in patients receiving cancer
chemotherapy: natural history, pathogenesis, and management.
Hepatol Int. 14:142011.PubMed/NCBI
|
|
8
|
Yagci M, Ozkurt ZN, Yegin ZA, Aki Z, Sucak
GT and Haznedar R: Hepatitus B virus reactivation in HBV-DNA
negative and positive patients with hematological malignancies.
Hematology. 15:240–244. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang B, Wang J, Xu W, Wang L and Ni W:
Fatal reactivation of occult hepatitis B virus infection after
rituximab and chemotherapy in lymphoma: necessity of antiviral
prophylaxis. Onkologie. 33:537–539. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Day FL, Karnon J and Rischin D:
Cost-effectiveness of universal hepatitis B virus screening in
patients beginning chemotherapy for solid tumors. J Clin Oncol.
29:3270–3277. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cheng JC, Wu JK, Lee PC, Liu HS, Jian JJ,
Lin YM, Sung JL and Jan GJ: Biologic susceptibility of
hepatocellular carcinoma patients treated with radiotherapy to
radiation-induced liver disease. Int J Radiat Oncol Biol Phys.
60:1502–1509. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Evens AM, Jovanovic BD, Su YC, Raisch DW,
Ganger D, Belknap SM, Dai MS, Chiu BC, Fintel B, Cheng Y, et al:
Rituximab-associated hepatitis B virus (HBV) reactivation in
lymphoproliferative diseases: meta-analysis and examination of FDA
safety reports. Ann Oncol. 22:1170–1180. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Matsue K, Kimura S, Takanashi Y, Iwama K,
Fujiwara H, Yamakura M and Takeuchi M: Reactivation of hepatitis B
virus after rituximab-containing treatment in patients with
CD20-positive B-cell lymphoma. Cancer. 116:4769–4776. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Niitsu N, Hagiwara Y, Tanae K, Kohri M and
Takahashi N: Prospective analysis of hepatitis B virus reactivation
in patients with diffuse large B-cell lymphoma after rituximab
combination chemotherapy. J Clin Oncol. 28:5097–5100. 2010.
View Article : Google Scholar
|
|
15
|
Minn AY, Hsu A, La T, Kunz P, Fisher GA,
Ford JM, Norton JA, Visser B, Goodman KA, Koong AC and Chang DT:
Comparison of intensity-modulated radiotherapy and 3-dimensional
conformal radiotherapy as adjuvant therapy for gastric cancer.
Cancer. 116:3943–3952. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ji D, Cao J, Hong X, Li J, Wang J, Chen F,
Wang C and Zou S: Low incidence of hepatitis B virus reactivation
during chemotherapy among diffuse large B-cell lymphoma patients
who are HBsAg-negative/HBcAb-positive: a multicenter retrospective
study. Eur J Haematol. 85:243–250. 2010. View Article : Google Scholar
|
|
17
|
Roche B and Samuel D: The difficulties of
managing severe hepatitis B virus reactivation. Liver Int. 31(Suppl
1): 104–110. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Artz AS, Somerfield MR, Feld JJ, Giusti
AF, Kramer BS, Sabichi AL, Zon RT and Wong SL: American Society of
Clinical Oncology provisional clinical opinion: chronic hepatitis B
virus infection screening in patients receiving cytotoxic
chemotherapy for treatment of malignant diseases. J Clin Oncol.
28:3199–3202. 2010. View Article : Google Scholar
|
|
19
|
Marinone C and Mestriner M: HBV disease:
HBsAg carrier and occult B infection reactivation in haematological
setting. Dig Liver Dis. 43(Suppl 1): S49–56. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chou CH, Chen PJ, Jeng YM, Cheng AL, Huang
LR and Cheng JC: Synergistic effect of radiation and interleukin-6
on hepatitis B virus reactivation in liver through STAT3 signaling
pathway. Int J Radiat Oncol Biol Phys. 75:1545–1552. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cheng JC, Liu HS, Wu JK, Chung HW and Jan
GJ: Inclusion of biological factors in parallel-architecture
normal-tissue complication probability model for radiation-induced
liver disease. Int J Radiat Oncol Biol Phys. 62:1150–1156. 2005.
View Article : Google Scholar
|
|
22
|
Kim JH, Park JW, Kim TH, Koh DW, Lee WJ
and Kim CM: Hepatitis B virus reactivation after three-dimensional
conformal radiotherapy in patients with hepatitis B virus-related
hepatocellular carcinoma. Int J Radiat Oncol Biol Phys. 69:813–819.
2007. View Article : Google Scholar
|
|
23
|
Ide Y, Ito Y, Takahashi S, Tokudome N,
Kobayashi K, Sugihara T, Hattori M, Yokoyama M, Uchiyama A, Inoue
K, et al: Hepatitis B virus reactivation in adjuvant chemotherapy
for breast cancer. Breast Cancer. Jul 24–2010.(Epub ahead of
print).
|
|
24
|
Kusumoto S, Tanaka Y, Mizokami M and Ueda
R: Clinical significance of hepatitis B virus (HBV)-DNA monitoring
to detect HBV reactivation after systemic chemotherapy. J Clin
Oncol. 29:e100author reply e101. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Day FL, Link E, Thursky K and Rischin D:
Current hepatitis B screening practices and clinical experience of
reactivation in patients undergoing chemotherapy for solid tumors:
a nationwide survey of medical oncologists. J Oncol Pract.
7:141–147. 2011. View Article : Google Scholar
|
|
26
|
Huang H, Cai Q, Lin T, Lin X, Liu Y, Gao Y
and Peng R: Lamivudine for the prevention of hepatitis B virus
reactivation after high-dose chemotherapy and autologous
hematopoietic stem cell transplantation for patients with advanced
or relapsed non-Hodgkin’s lymphoma single institution experience.
Expert Opin Pharmacother. 10:2399–2406. 2009.
|
|
27
|
You CR, Jang JW, Choi JK, Bae SH, Yoon SK,
Kay CS and Choi JY: Hepatic failure caused by reactivation of YMDD
mutants occurring during preemptive lamivudine therapy. Gut Liver.
4:262–265. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Colson P, Borentain P, Coso D, Chabannon
C, Tamalet C and Gérolami R: Entecavir as a first-line treatment
for HBV reactivation following polychemotherapy for lymphoma. Br J
Haematol. 143:148–150. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Brost S, Schnitzler P, Stremmel W and
Eisenbach C: Entecavir as treatment for reactivation of hepatitis B
in immunosuppressed patients. World J Gastroenterol. 16:5447–5451.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Watanabe M, Shibuya A, Takada J, Tanaka Y,
Okuwaki Y, Minamino T, Hidaka H, Nakazawa T and Koizumi W:
Entecavir is an optional agent to prevent hepatitis B virus (HBV)
reactivation: a review of 16 patients. Eur J Intern Med.
21:333–337. 2010. View Article : Google Scholar : PubMed/NCBI
|