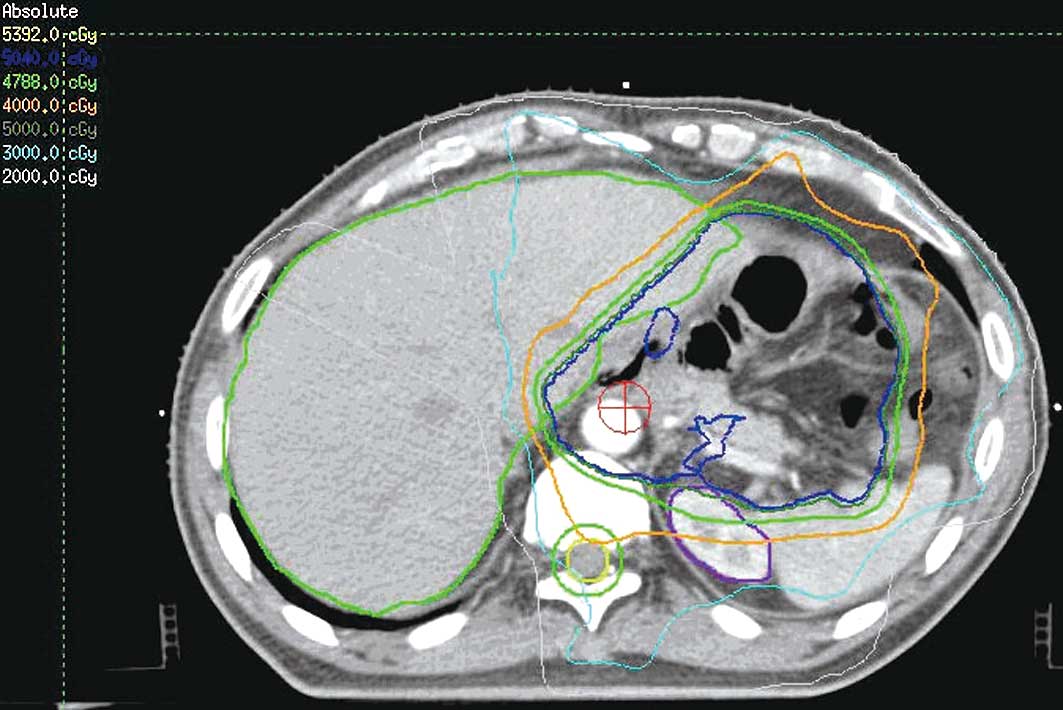
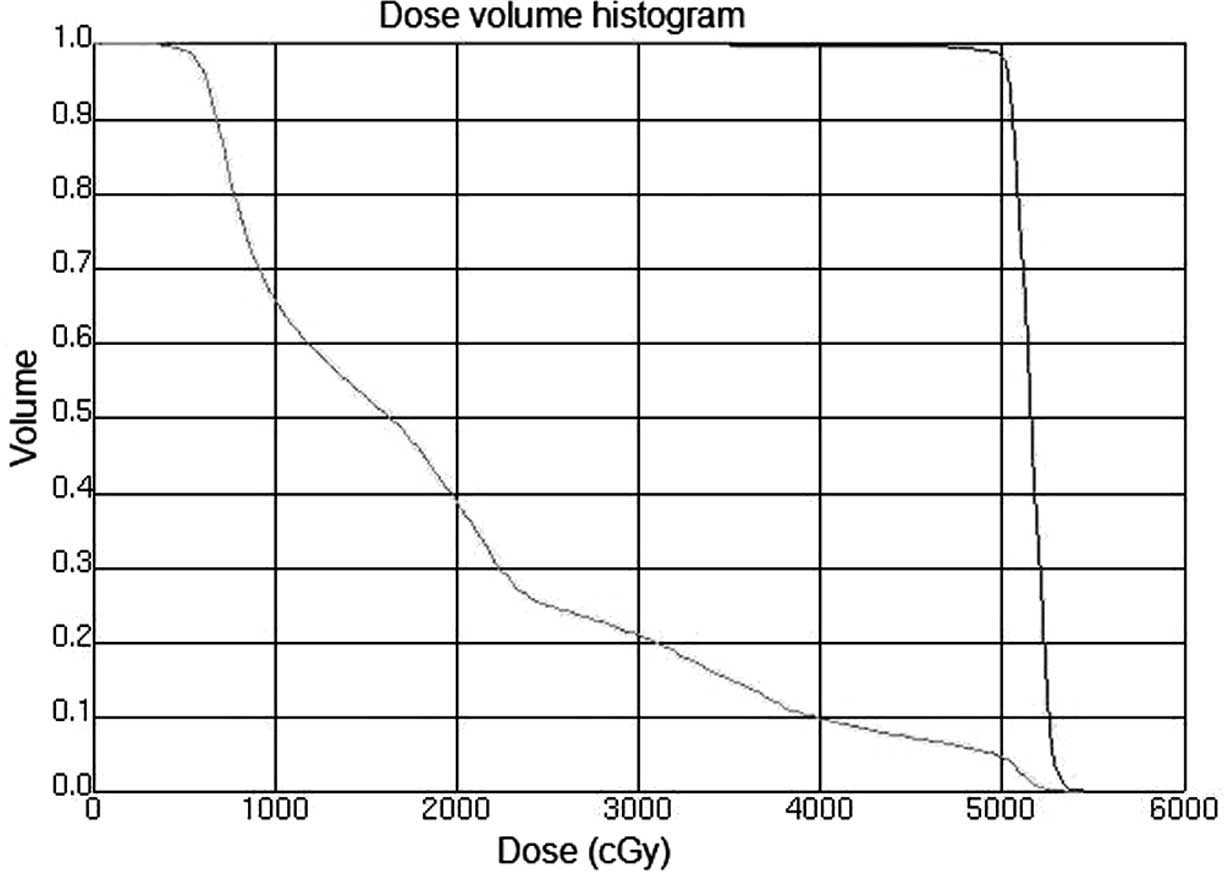
|
1
|
Cheng JC, Liu MC, Tsai SY, Fang WT,
Jer-Min Jian J and Sung JL: Unexpectedly frequent hepatitis B
reactivation by chemoradiation in postgastrectomy patients. Cancer.
101:2126–2133. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Stange MA, Tutarel O, Pischke S, Schneider
A, Strassburg CP, Becker T, Barg-Hock H, Basturk M, Wursthorn K,
Cornberg M, et al: Fulminant hepatic failure due to
chemotherapy-induced hepatitis B reactivation: role of rituximab. Z
Gastroenterol. 48:258–263. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lubel JS and Angus PW: Hepatitis B
reactivation in patients receiving cytotoxic chemotherapy:
diagnosis and management. J Gastroenterol Hepatol. 25:864–871.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Koo YX, Tan DS, Tan IB, Tao M, Chow WC and
Lim ST: Hepatitis B virus reactivation and role of antiviral
prophylaxis in lymphoma patients with past hepatitis B virus
infection who are receiving chemoimmunotherapy. Cancer.
116:115–121. 2010.PubMed/NCBI
|
|
5
|
Lim SM, Jang JW, Kim BW, Choi H, Choi KY,
Park SJ and Han CW: Hepatitis B virus reactivation during
chlorambucil and prednisolone treatment in an HBsAg-negative and
anti-HBs-positive patient with B-cell chronic lymphocytic leukemia.
Korean J Hepatol. 14:213–218. 2008.(In Korean).
|
|
6
|
Wu JM, Huang YH, Lee PC, Lin HC and Lee
SD: Fatal reactivation of hepatitis B virus in a patient who was
hepatitis B surface antigen negative and core antibody positive
before receiving chemotherapy for non-Hodgkin lymphoma. J Clin
Gastroenterol. 43:496–498. 2009. View Article : Google Scholar
|
|
7
|
Liu CJ, Chen PJ, Chen DS and Kao JH:
Hepatitis B virus reactivation in patients receiving cancer
chemotherapy: natural history, pathogenesis, and management.
Hepatol Int. 14:142011.PubMed/NCBI
|
|
8
|
Yagci M, Ozkurt ZN, Yegin ZA, Aki Z, Sucak
GT and Haznedar R: Hepatitus B virus reactivation in HBV-DNA
negative and positive patients with hematological malignancies.
Hematology. 15:240–244. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang B, Wang J, Xu W, Wang L and Ni W:
Fatal reactivation of occult hepatitis B virus infection after
rituximab and chemotherapy in lymphoma: necessity of antiviral
prophylaxis. Onkologie. 33:537–539. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Day FL, Karnon J and Rischin D:
Cost-effectiveness of universal hepatitis B virus screening in
patients beginning chemotherapy for solid tumors. J Clin Oncol.
29:3270–3277. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cheng JC, Wu JK, Lee PC, Liu HS, Jian JJ,
Lin YM, Sung JL and Jan GJ: Biologic susceptibility of
hepatocellular carcinoma patients treated with radiotherapy to
radiation-induced liver disease. Int J Radiat Oncol Biol Phys.
60:1502–1509. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Evens AM, Jovanovic BD, Su YC, Raisch DW,
Ganger D, Belknap SM, Dai MS, Chiu BC, Fintel B, Cheng Y, et al:
Rituximab-associated hepatitis B virus (HBV) reactivation in
lymphoproliferative diseases: meta-analysis and examination of FDA
safety reports. Ann Oncol. 22:1170–1180. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Matsue K, Kimura S, Takanashi Y, Iwama K,
Fujiwara H, Yamakura M and Takeuchi M: Reactivation of hepatitis B
virus after rituximab-containing treatment in patients with
CD20-positive B-cell lymphoma. Cancer. 116:4769–4776. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Niitsu N, Hagiwara Y, Tanae K, Kohri M and
Takahashi N: Prospective analysis of hepatitis B virus reactivation
in patients with diffuse large B-cell lymphoma after rituximab
combination chemotherapy. J Clin Oncol. 28:5097–5100. 2010.
View Article : Google Scholar
|
|
15
|
Minn AY, Hsu A, La T, Kunz P, Fisher GA,
Ford JM, Norton JA, Visser B, Goodman KA, Koong AC and Chang DT:
Comparison of intensity-modulated radiotherapy and 3-dimensional
conformal radiotherapy as adjuvant therapy for gastric cancer.
Cancer. 116:3943–3952. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ji D, Cao J, Hong X, Li J, Wang J, Chen F,
Wang C and Zou S: Low incidence of hepatitis B virus reactivation
during chemotherapy among diffuse large B-cell lymphoma patients
who are HBsAg-negative/HBcAb-positive: a multicenter retrospective
study. Eur J Haematol. 85:243–250. 2010. View Article : Google Scholar
|
|
17
|
Roche B and Samuel D: The difficulties of
managing severe hepatitis B virus reactivation. Liver Int. 31(Suppl
1): 104–110. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Artz AS, Somerfield MR, Feld JJ, Giusti
AF, Kramer BS, Sabichi AL, Zon RT and Wong SL: American Society of
Clinical Oncology provisional clinical opinion: chronic hepatitis B
virus infection screening in patients receiving cytotoxic
chemotherapy for treatment of malignant diseases. J Clin Oncol.
28:3199–3202. 2010. View Article : Google Scholar
|
|
19
|
Marinone C and Mestriner M: HBV disease:
HBsAg carrier and occult B infection reactivation in haematological
setting. Dig Liver Dis. 43(Suppl 1): S49–56. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chou CH, Chen PJ, Jeng YM, Cheng AL, Huang
LR and Cheng JC: Synergistic effect of radiation and interleukin-6
on hepatitis B virus reactivation in liver through STAT3 signaling
pathway. Int J Radiat Oncol Biol Phys. 75:1545–1552. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cheng JC, Liu HS, Wu JK, Chung HW and Jan
GJ: Inclusion of biological factors in parallel-architecture
normal-tissue complication probability model for radiation-induced
liver disease. Int J Radiat Oncol Biol Phys. 62:1150–1156. 2005.
View Article : Google Scholar
|
|
22
|
Kim JH, Park JW, Kim TH, Koh DW, Lee WJ
and Kim CM: Hepatitis B virus reactivation after three-dimensional
conformal radiotherapy in patients with hepatitis B virus-related
hepatocellular carcinoma. Int J Radiat Oncol Biol Phys. 69:813–819.
2007. View Article : Google Scholar
|
|
23
|
Ide Y, Ito Y, Takahashi S, Tokudome N,
Kobayashi K, Sugihara T, Hattori M, Yokoyama M, Uchiyama A, Inoue
K, et al: Hepatitis B virus reactivation in adjuvant chemotherapy
for breast cancer. Breast Cancer. Jul 24–2010.(Epub ahead of
print).
|
|
24
|
Kusumoto S, Tanaka Y, Mizokami M and Ueda
R: Clinical significance of hepatitis B virus (HBV)-DNA monitoring
to detect HBV reactivation after systemic chemotherapy. J Clin
Oncol. 29:e100author reply e101. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Day FL, Link E, Thursky K and Rischin D:
Current hepatitis B screening practices and clinical experience of
reactivation in patients undergoing chemotherapy for solid tumors:
a nationwide survey of medical oncologists. J Oncol Pract.
7:141–147. 2011. View Article : Google Scholar
|
|
26
|
Huang H, Cai Q, Lin T, Lin X, Liu Y, Gao Y
and Peng R: Lamivudine for the prevention of hepatitis B virus
reactivation after high-dose chemotherapy and autologous
hematopoietic stem cell transplantation for patients with advanced
or relapsed non-Hodgkin’s lymphoma single institution experience.
Expert Opin Pharmacother. 10:2399–2406. 2009.
|
|
27
|
You CR, Jang JW, Choi JK, Bae SH, Yoon SK,
Kay CS and Choi JY: Hepatic failure caused by reactivation of YMDD
mutants occurring during preemptive lamivudine therapy. Gut Liver.
4:262–265. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Colson P, Borentain P, Coso D, Chabannon
C, Tamalet C and Gérolami R: Entecavir as a first-line treatment
for HBV reactivation following polychemotherapy for lymphoma. Br J
Haematol. 143:148–150. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Brost S, Schnitzler P, Stremmel W and
Eisenbach C: Entecavir as treatment for reactivation of hepatitis B
in immunosuppressed patients. World J Gastroenterol. 16:5447–5451.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Watanabe M, Shibuya A, Takada J, Tanaka Y,
Okuwaki Y, Minamino T, Hidaka H, Nakazawa T and Koizumi W:
Entecavir is an optional agent to prevent hepatitis B virus (HBV)
reactivation: a review of 16 patients. Eur J Intern Med.
21:333–337. 2010. View Article : Google Scholar : PubMed/NCBI
|