Introduction
Rectal carcinoids are low-grade malignancies, which
are slow-growing and usually become symptomatic late in the course
of the disease (1,2). The incidence of rectal carcinoids is
rising: In the United States, the age-adjusted incidence has
increased by 800–1000% in the last 35 years. The incidence of
rectal carcinoids is the highest among gastrointestinal carcinoid
tumors (3). The incidences of
carcinoids of the rectum, stomach and small bowels have also
multiplied (4). The reasons for
these epidemiological changes are not yet understood. Screening
colonoscopy, in addition to decreasing colorectal adenocarcinoma
mortality, is useful in diagnosing carcinoid tumors at an earlier
stage and in decreaing mortality form malignant colorectal
carcinoid tumors (5,6).
Small carcinoid tumors measuring <1 cm in
diameter may therefore be managed endoscopically and preoperative
assessment with endoscopic miniprobe ultrasonography (EUS) with out
recurrence or spread (7,8). Various methods for complete endoscopic
resection of rectal carcinoid tumors have been reported, such as
endoscopic submucosal dissection (ESD) or conventional endoscopic
mucosal resection (EMR) (9,10). Increased surgical time and
complication risks with ESD remain problematic (11). The selection of endoscopic treatment
should be made after taking factors, such as cost-effectiveness,
expertise and experience into careful consideration (12). In this study, we review and
summarize the endoscopic and pathological features of small rectal
carcinoid tumors (≤1 cm in diameter) of twenty-one cases. We also
evaluate the safety and efficacy of complete resection of small
rectal carcinoid tumors using EMR and endoscopic submucosal
resection with cap aspiration technique (ESMR-C).
Patients and methods
Patient cases
A series of 22 rectal carcinoid tumors in 21
patients (age range, 37 to 78 years; 2 females and 19 males) were
treated at The PLA General Hospital, Beijing, China, between June
1995 and March 2010. All clinical data and pathological records
were reviewed. All cases were detected by colonoscopy without
clinical symptoms. The study was approved by the Ethics Committee
of The PLA General Hospital, Beijing, China. Written informed
consent was obtained from the patients.
Devices
CF-H260AL colonoscopy (Olympus, Tokyo, Japan) and
micro ultrasound probe UM2R and UM3R (Olympus) were used with
frequencies of 12MHZ and 20MHZ. The aerodynamic endoscopic ligation
device MD-48709 was employed (Olympus).
Methods
Under colonoscopy, the rectal carcinoid tumors were
identified to have smooth surfaces and normal mucosa. In 16 cases,
EUS was performed using the degassing and water filling method to
observe the size of the lesion and the shape of the border, the
internal echo intensity, sources and the relationship between the
lesion and the surrounding wall. All patients were followed-up
after 2 months, 6 months and 1 year by colonoscopy, EUS and local
biopsy.
Results
Lesion location, size and shape
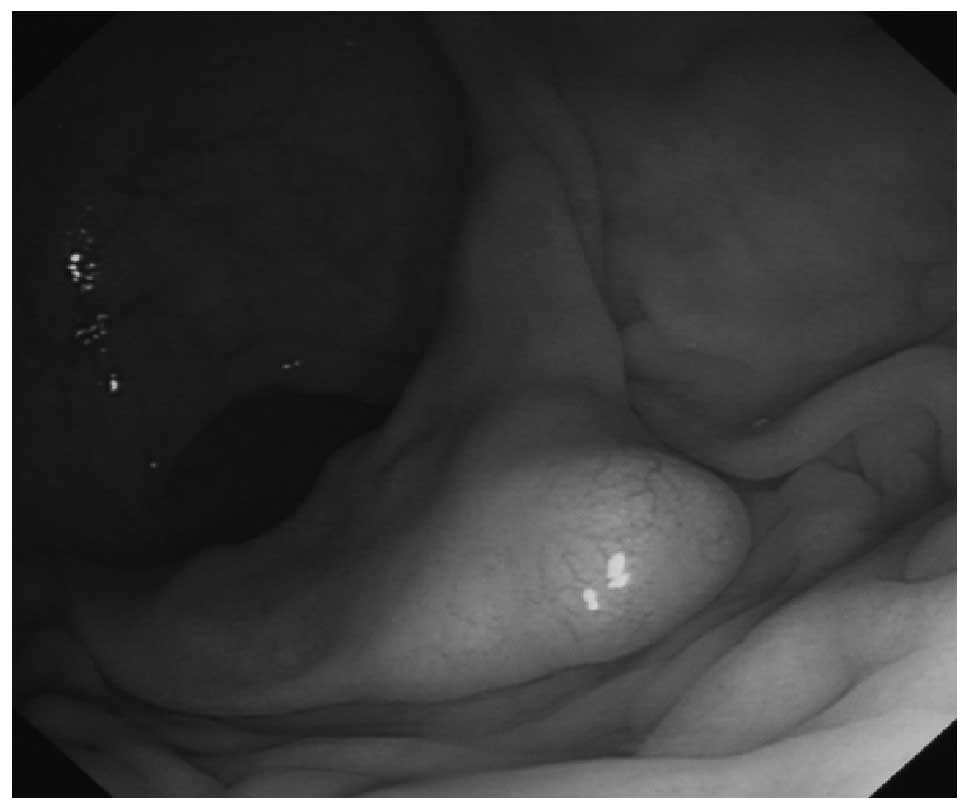
Only 1 patient had a lesion that was 15 cm from the
anus, while the rest were within the range of 3–10 cm from the
anus. Two lesions were found 6 cm away, and 1 case was 8 cm away
from the anus. The size of the lesions ranged from 0.3 to 1.0 cm.
Surface erosion was found in 1 case, while the remaining cases were
smooth. Lesions were yellow in 7 cases and slightly red in 3 cases.
The remaining lesions were similar in colour to the surrounding
mucosa. The tumor shapes were hemispherical bulges in 13 cases and
flat-shaped bulges in 9 cases. Upon being touched with bopsy
forceps, a sliding movement was observed in 8 cases (Fig. 1).
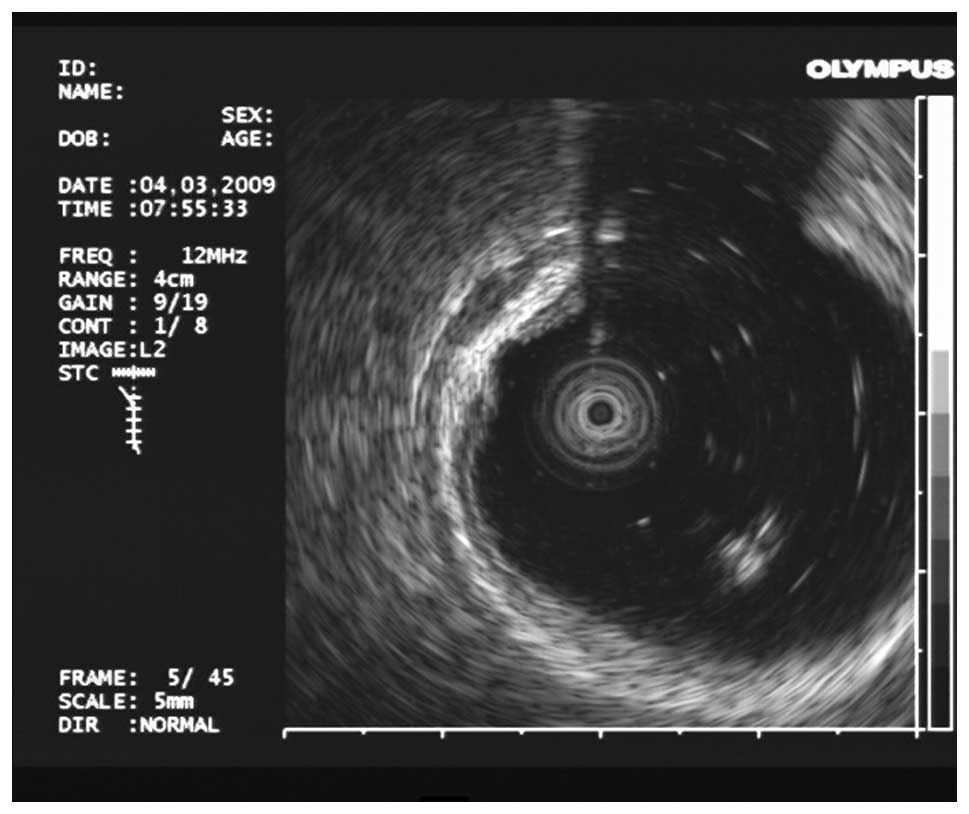
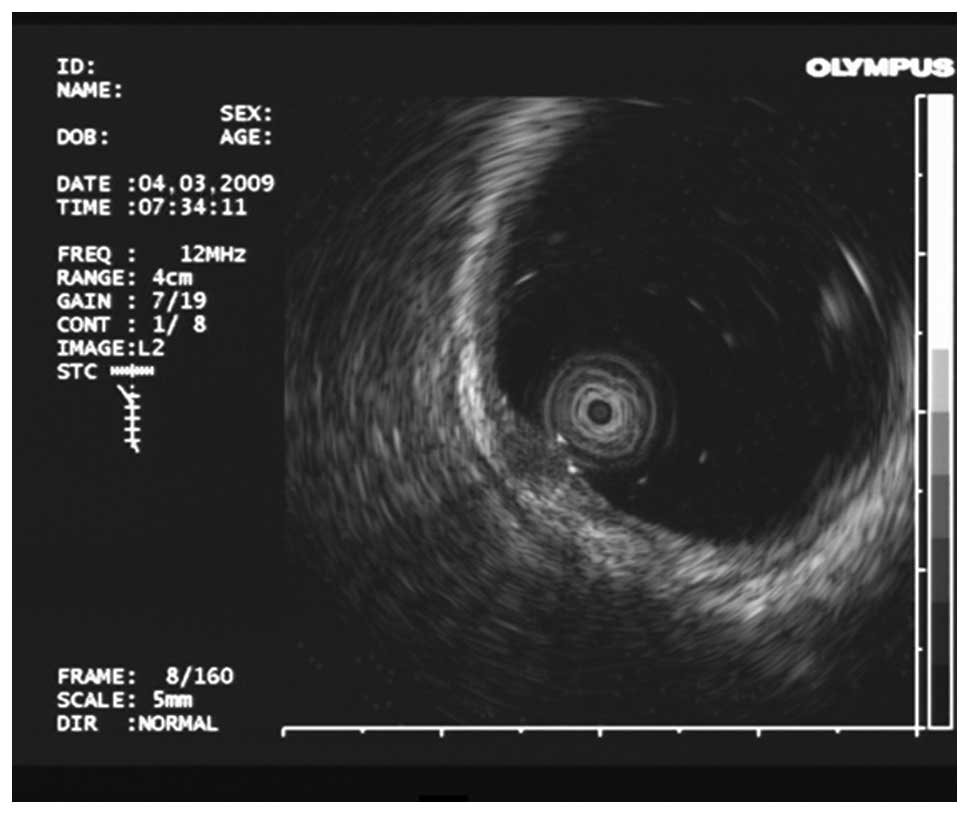
EUS results
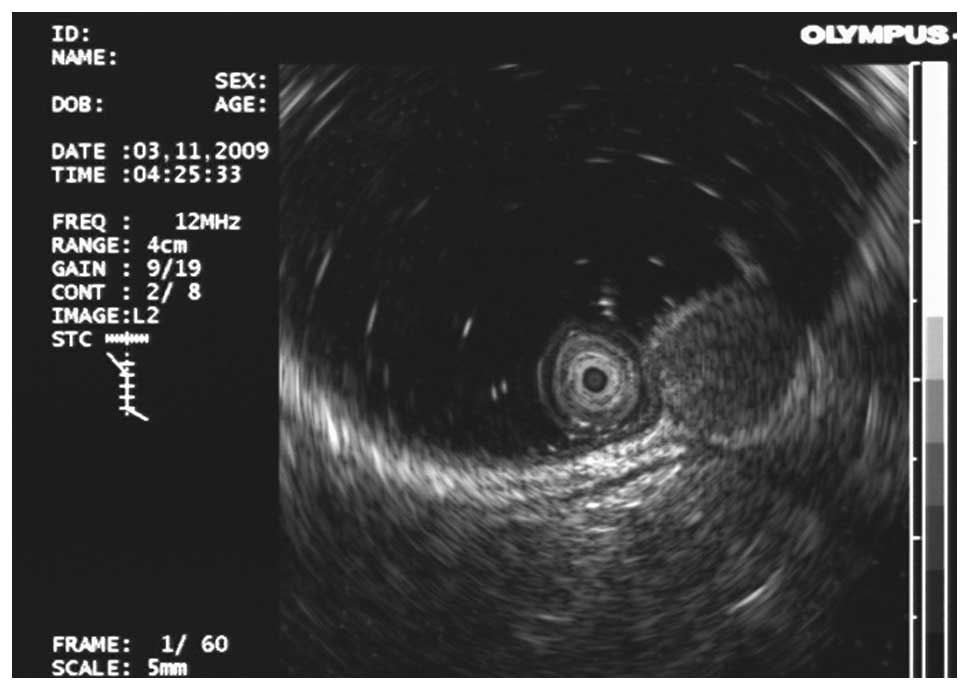
Among the 16 cases of rectal carcinoid lesions, 12
were hypoechoic nodules and 4 were isoechoic nodules. Six lesions
were located in the mucosa and 10 lesions were located in the
submucosa. Sixteen lesions showed clear boundaries (Fig. 2).
Treatment and follow-up results
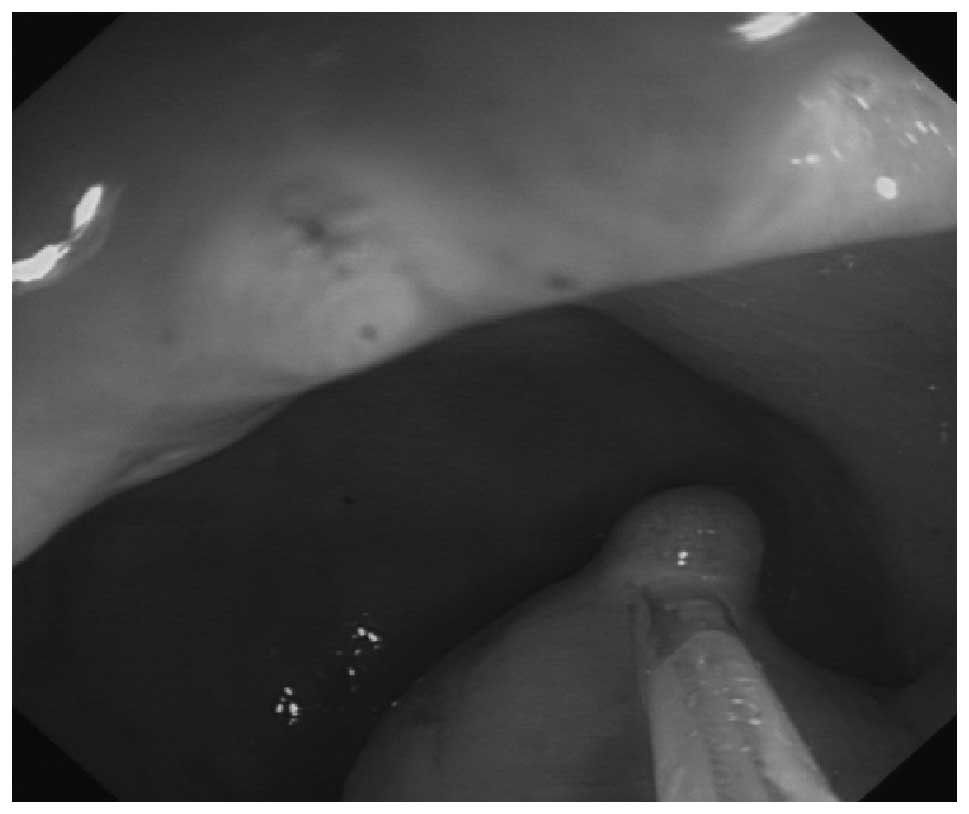
Different treatments were selected according to the
size and shape of the lesion. For lesions with a diameter of ∼0.5
cm and a semisphere shape, a snare resection was performed by use
of an electrosurgical current following a submucosal injection of
physiological saline solution around the lesion to lift it off the
muscle layer (Fig. 3). For flat
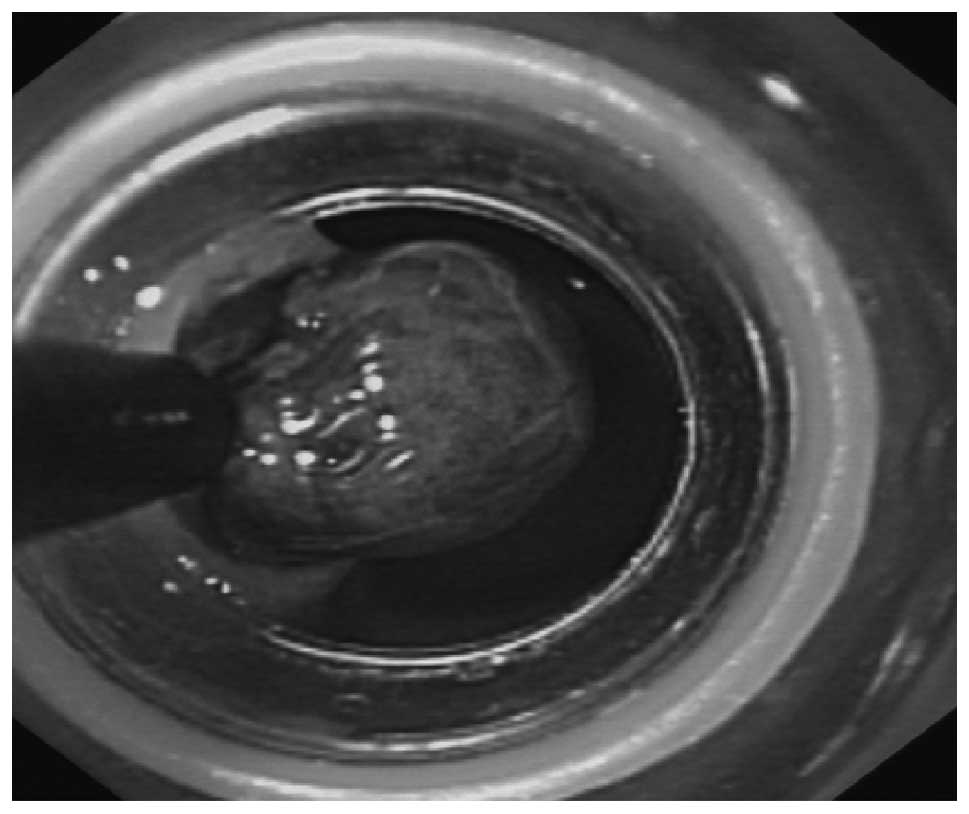
lesions or lesions with a diameter of >0.5 cm, prior to the
snare resection, the lesion was immobilized with suction using an
endoscopic ligation device (Olympus) (Fig. 4). For 1 case with a minor lesion of
0.2 cm, the lesion was clamped using biopsy forceps and then
resected by argon plasma coagulation (APC) using a VIO 300D
electrosurgical system (ERBE, Tuebingen, Germany). The specimens
were pathologically examined and immunohistochemistry was conducted
for some specimens.
Following excision of 18 lesions, pathological
examination revealed a clear cutting edge and base. After 2 months,
the follow-up found no carcinoid tumor tissue. Three cases reported
carcinoid tissue in the margin. After 2 months, the
ultrasonographic and pathological examinations showed no local
carcinoma (Fig. 5). One case
reported carcinoid tissue in the margin. After 2 months, the
ultrasonographic and pathological examinations still found
carcinoid tissue in the margin and surgical operations were
undertaken (Fig. 6).
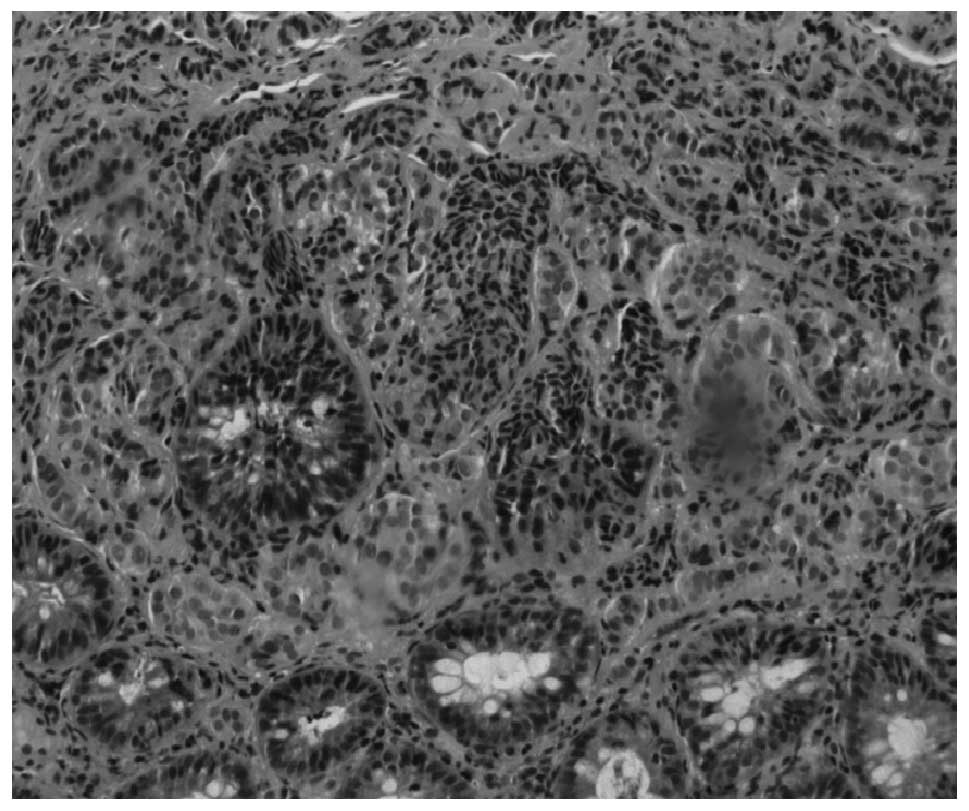
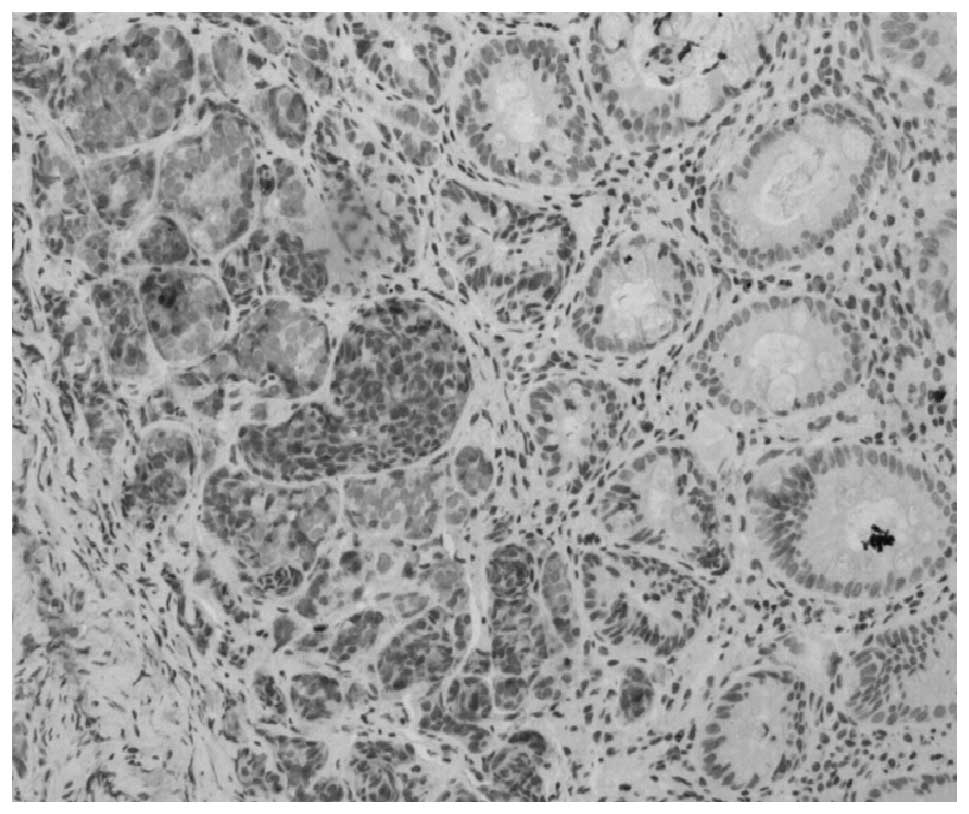
Pathological results
Pathological examination revealed chronic
inflammation in the surface of the rectal mucosa. Microscopically,
the tumor cells showed solid- or cord-like appearances with round
or polygonal shapes and small nuclei. The majority of the tumor
cells were confined to the mucosa or the submucosa (Fig. 7). Eleven samples of 21 cases
received immunohistochemistry evaluation. Among these, vesicle
synaptophysin (SYN) was positive in 11 cases (Fig. 8), neuron-specific enolase (NSE) was
positive in 9 cases (Fig. 9),
chromogranin (CgA) was positive in 5 cases and CK was positive in 2
cases. Ki-67 and CEA were negative.
Discussion
Rectal carcinoid tumors originate from the Kulchisty
cells (or chromaffin cells) in the deep intestinal crypt. These
amine precursor uptake decarboxylase (APUD) cells, known as
neuroendocrine tumors, are low invasive with limited infiltrating
growth. A single tumor is common, while multiple tumors account for
only 2 to 4.5% (13)t. Compared
with other tumors, lymphatic and hematogenous metastasis is less.
Among our cases, only 1 case was multiple. It has been reported
that multiple tumors are only 0.5 cm away from each other, probably
due to submucosal lymphatic metastasis, rather than multiple
germinal generation (13).
In general, rectal carcinoid tumors are more common
in the front or lateral wall of the rectum, usually 3 to 8 cm away
from the anus. The early small nodules in the lamina propria
gradually develop into the submucosal and muscle layer. Colonoscopy
has revealed a flat round nodule with a clear border and a smooth
surface, coated with the normal mucosa, presenting a white or light
yellow color and movability with biopsy clamp touch. As the
diameter of early lesions is <1 cm, they are initially diagnosed
as submucosal tumors.
As the shape of the rectal carcinoid tumor resembles
sessile polyps, certain endoscopists may regard carcinoid tumors
<0.5 cm as hyperplastic polyps and ignore them, thus delaying
the treatment. Although occasionally biopsy for pathological
examination is conducted, the tumor tissue is difficult to biopsy
since it is located in the submucosa (14). It is also difficult to correctly
determine the size, the intestinal origin and the histological
features of the rectal carcinoid tumor with common colonoscopy.
Rectal endoscopic ultra-sound is the most accurate method to
determine the depth of tumor invasion. With high ultrasound
frequency (12–20 MHz), EUS is able to clearly show the five-layer
structure of the digestive tract wall, determine the source of the
tumor and its relationship with the rectum wall, and the nature of
the lesion, which aids in making accurate judgements in order to
determine the treatment. EUS is the best for diagnosis of tumors of
gastrointestinal mucosa and submucosa. EUS boasts high accuracy for
the identification of benign and malignant rectal carcinoid and
other submucosal tumors, as well as extraintestinal oppressive
lesions (15,16).
In our study, we used this examination technique in
16 cases of rectal carcinoid lesions, which all showed hypoechoic
or isoechoic nodules with clear boundaries. Six lesions were
located in the mucosa and 10 lesions were located in the submucosa.
According to the literature, leiomyoma from the muscularis mucosa
also present with sonographic features of hypoechoic nodules with
clear boundary and uniform internal echo. The sonographic features
of the rectal leiomyoma and the rectal carcinoid tumor require
further research. No leiomyoma of the rectum has been confirmed
pathologically in our department, due to the much lower incidence
than rectal carcinoid tumors under endoscopy. Therefore, priority
EUS should be given to rectal carcinoid displaying EUS features,
such as hypoechoic or isoechoic nodule with clear boundaries
located in the mucosa or submucosa (17).
Tumor size is closely correlated with the degree of
malignancy. A diameter of 2.0 cm is the key indicator to determine
the level of malignancy of the rectal carcinoid tumor (18). The rate of metastasis of tumors with
diameters <1.0 cm is <2%. For 2/3 of rectal carcinoid tumor,
the diameter is <1.0 cm. The diameters of the lesions were
between 0.3 and 1.0 cm with clear borders. We believe that
endoscopic treatment should be first considered. The resection
should include the normal mucosa 0.5 cm away from the border of the
tumor and the depth should reach the muscle layer. Pathological
results confirmed the carcinoid tumor, and that the edge and base
were intact without residue. For hemispherical elevated lesions
<0.5 cm in diameter, direct snare electrocoagulation resection
was performed after submucosal injection of saline. For lesions
with diameter >0.5 cm or flat lesions, with the application of
endoscopic air-driven ligator (transparent cap attached to the tip
of the intestinal endoscope) and the formation of polypoid bulge,
the electrical incision ring was used to remove the lesions (EMR).
To avoid residual tumor, EMR is more effective (19). Electrocautery may be used to stop
bleeding after EMR. For residual tumor confirmed by pathology, due
to the thermal conductive effects, the residual tumor tissue may be
in coagulation and necrosis. Close follow-up will be required
instead of expanding the surgery. EUS and endoscopic biopsy are
needed after 1 to 2 months. This approach has been successfully
utilized to treat 21 cases of rectal carcinoid tumors with a
diameter of <1 cm in our department. No complications, such as
bleeding or perforation, occurred. Six months to 1 year follow-up
showed no recurrence.
Immunohistochemical examination is essential for
determining the diagnosis of carcinoid tumor. All tissues in solid
nest-like, island-like nodules, beam-like, banded or rose-like
structure or with abundant capillaries or sinusoids should
undertake the routine neuroendocrine markers immunohistochemical
examination (20–22). Among known neuroendocrine markers,
SYN, NSE and CgA are relatively specific, and SYN is relatively
sensitive in rectal carcinoids. Expression of Ki-67 is important
for assessment of tumor cell proliferation activity and prognosis
prediction (23). In our study, 11
cases undertook routine immunohistochemistry examination, of which
11 were positive for Syn, 9 were CgA positive and 5 were positive
for NSE, while CEA was negative in all cases. The expression rate
of Ki-67 in rectal carcinoid tumors is low. In this study, the
expression rate of Ki-67 in tumors lesions <1 cm was <5% or
negative. These were attributed to the low degree of malignancy of
rectal carcinoid tumors.
In this study, we showed that colonoscopy combined
with ultrasonic micro-probe was able to precisely determine the
location, size and range of the rectal carcinoid tumor, which may
guide endoscopic treatment. For lesions <1 cm with clear border,
we believe that endoscopic resection, particularly EMR, should be
considered first. Pathological examination is needed for final
diagnosis and immunohistochemical examination is necessary if the
lesion is considered malignant to improve the preoperative
diagnosis of carcinoid tumor rate. Due to the fact that other
tumors in the colon or other parts are concomitant with a high
incidence, we should fully evaluate the colon instead of only
treating the local lesions.
References
|
1
|
Wang HB and Wang LP: Analysis on clinical
characteristics of rectal neuroendocrine tumors. Zhonghua Yi Xue Za
Zhi. 90:1686–1689. 2010.In Chinese.
|
|
2
|
Avenel P, McKendrick A, Silapaswan S, et
al: Gastrointestinal carcinoids: an increasing incidence of rectal
distribution. Am Surg. 76:759–763. 2010.PubMed/NCBI
|
|
3
|
Nakanome T, Yokoyama K and Takeuchi H:
Rectal granular-cell tumor difficult to distinguish from carcinoid
tumor. Dig Endosc. 22:325–328. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Varas-Lorenzo MJ, Muñoz-Agel F,
Espinós-Pérez JC and Bardají-Bofill M: Gastrointestinal carcinoid
tumors. Rev Esp Enferm Dig. 102:533–537. 2010.
|
|
5
|
Scherübl H: Rectal carcinoids are on the
rise: early detection by screening endoscopy. Endoscopy.
41:162–165. 2009.PubMed/NCBI
|
|
6
|
Onozato Y, Kakizaki S, Iizuka H, Sohara N,
Mori M and Itoh H: Endoscopic treatment of rectal carcinoid tumors.
Dis Colon Rectum. 53:169–176. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang XD, Wang ZQ, Huang QW, et al: The
ultrasonographic characteristicsof the rectal carcinoid tumor.
Zhonghua Xiao Hua Nei Jing Za Zhi. 22:98–100. 2005.In Chinese.
|
|
8
|
Chen TH, Lin CJ, Wu RC, et al: The
application of miniprobe ultrasonography in the diagnosis of
colorectal subepithelial lesions. Chang Gung Med J. 33:380–388.
2010.PubMed/NCBI
|
|
9
|
Lee DS, Jeon SW, Park SY, et al: The
feasibility of endoscopic submucosal dissection for rectal
carcinoid tumors: comparison with endoscopic mucosal resection.
Endoscopy. 42:647–651. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Baek IH: Endoscopic submucosal dissection
or conventional endoscopic mucosal resection is an effective and
safe treatment for rectal carcinoid tumors: a retrospective study.
J Laparoendosc Adv Surg Tech A. 20:329–331. 2010. View Article : Google Scholar
|
|
11
|
Onozato Y, Kakizaki S, Ishihara H, et al:
Endoscopic submucosal dissection for rectal tumors. Endoscopy.
39:423–427. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Steinhagen E, Chang G and Guillem JG:
Initial experience with transanal endoscopic microsurgery: the need
for understanding the limitations. J Gastrointest Surg. 15:958–962.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ghassemi KA, Ou H and Roth BE: Multiple
rectal carcinoids in a patient with neurofibromatosis. Gastrointest
Endosc. 71:216–218. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shim KN, Yang SK, Myung SJ, et al:
Atypical endoscopic features of rectal carcinoids. Endoscopy.
36:313–316. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhou PH, Yao LQ, Xu MD, Zhong YS, Zhang YQ
and Chen WF: Endoscopic ultrasonography and submucosal resection in
the diagnosis and treatment of rectal carcinoid tumors. Chin Med J
(Engl). 120:1938–1939. 2007.PubMed/NCBI
|
|
16
|
Ishii N, Horiki N, Itoh T, et al:
Endoscopic submucosal dissection and preoperative assessment with
endoscopic ultrasonography for the treatment of rectal carcinoid
tumors. Surg Endosc. 24:1413–1419. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fu KI, Mashimo Y and Matsuda T: Is
endoscopic ultrasonography necessary for depth evaluation of rectal
carcinoid tumors <or=10 mm? Dis Colon Rectum. 49:1238–1239.
2006.PubMed/NCBI
|
|
18
|
Nakase H, Matsuura M, Uza N, Ueno S,
Nishio A and Chiba T: Rectal carcinoid tumor mimicking colonic
adenomatous lesion. Gastrointest Endosc. 62:976–977. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sohn DK, Han KS, Hong CW, Chang HJ, Jeong
SY and Park JG: Selection of cap size in endoscopic submucosal
resection with cap aspiration for rectal carcinoid tumors. J
Laparoendosc Adv Surg Tech A. 18:815–818. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kawahara M, Kammori M, Kanauchi H, et al:
Immunohistochemical prognostic indicators of gastrointestinal
carcinoid tumours. Eur J Surg Oncol. 28:140–146. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ishida M, Kushima R and Okabe H: Claudin
expression in rectal well-differentiated endocrine neoplasms
(carcinoid tumors). Oncol Rep. 21:113–117. 2009.PubMed/NCBI
|
|
22
|
Conlon JM: Granin-derived peptides as
diagnostic and prognostic markers for endocrine tumors. Regul Pept.
165:5–11. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Modlin IM, Kidd M, Latich I, Zikusoka MN
and Shapiro MD: Current status of gastrointestinal carcinoids.
Gastroenterology. 128:1717–1751. 2005. View Article : Google Scholar : PubMed/NCBI
|