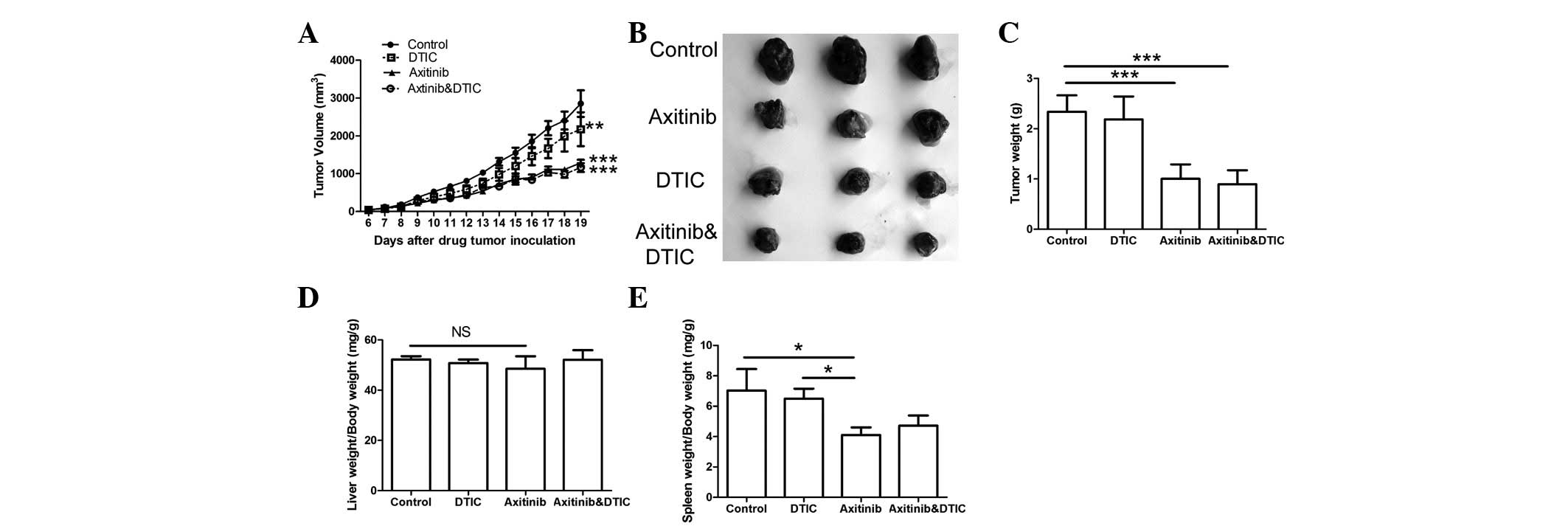
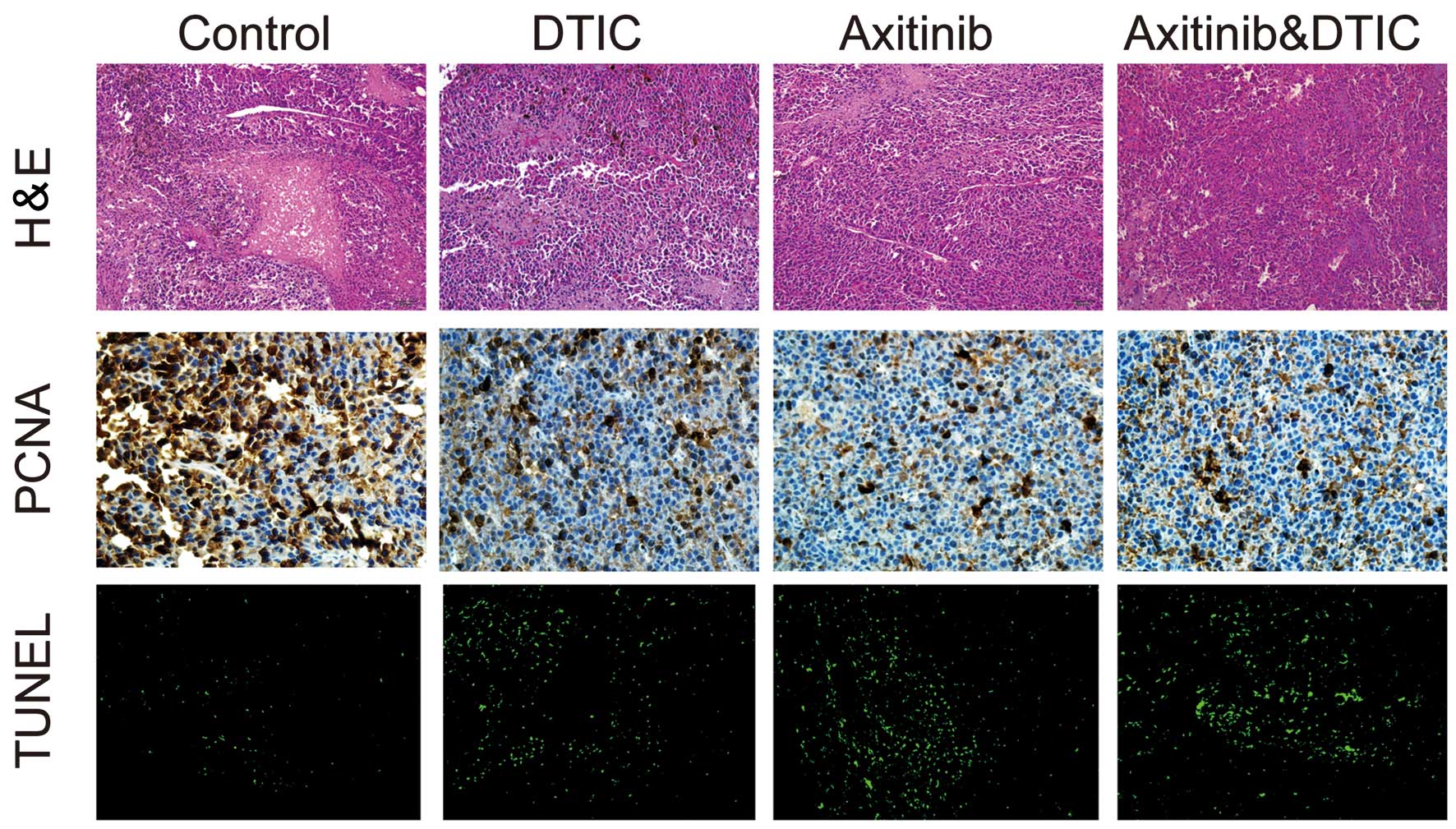
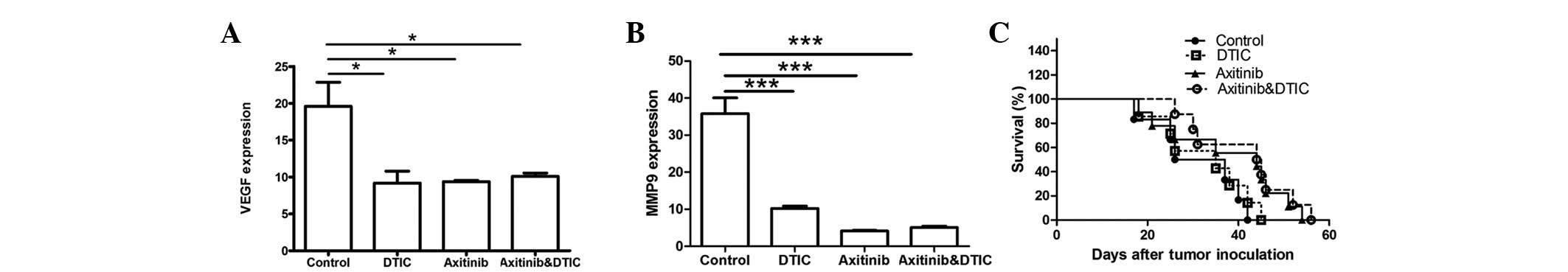
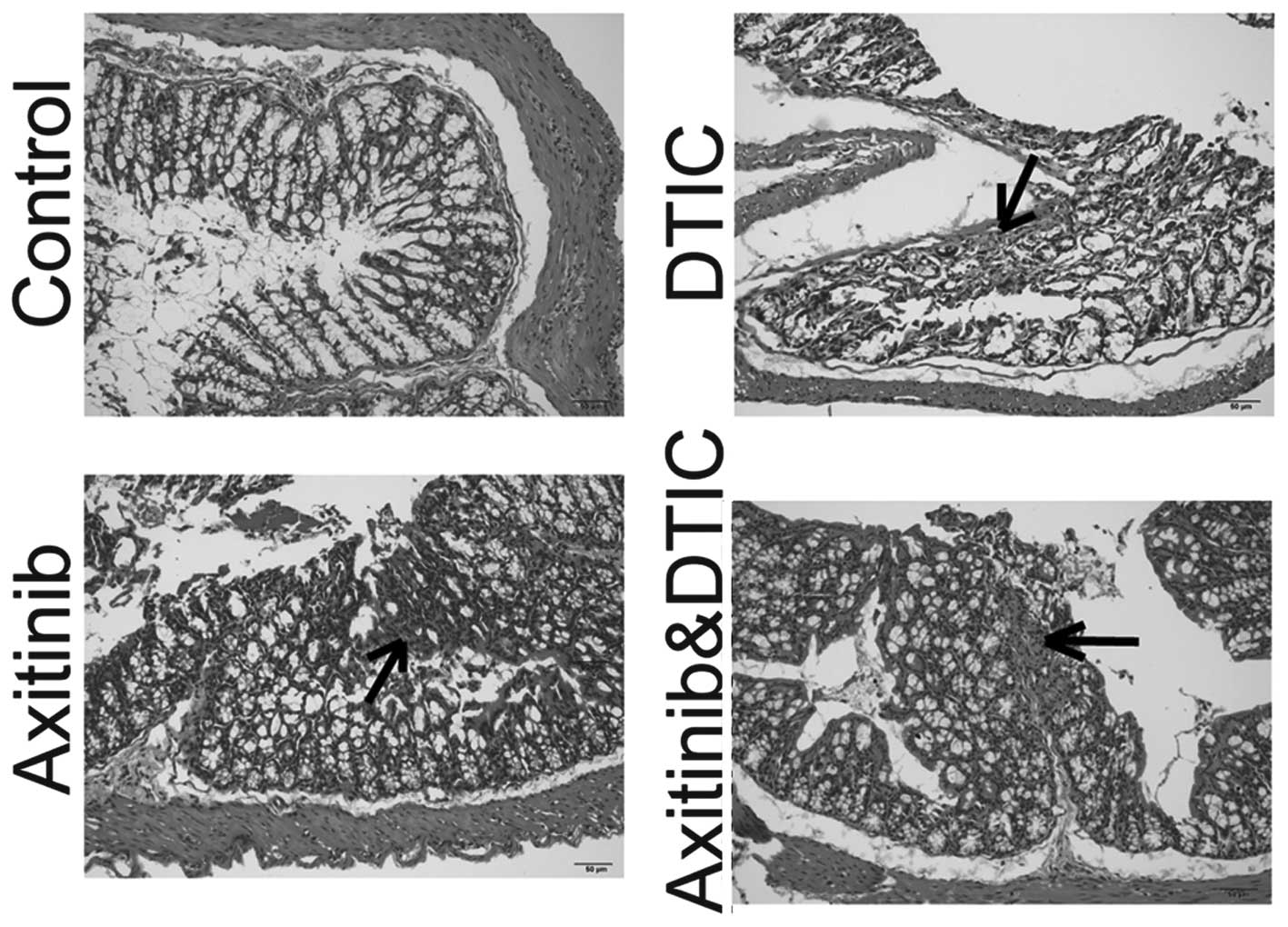
|
1.
|
Garbe C and Leiter U: Melanoma
epidemiology and trends. Clin Dermatol. 27:3–9. 2009. View Article : Google Scholar
|
|
2.
|
Balch CM, Soong SJ, Gershenwald JE, et al:
Prognostic factors analysis of 17,600 melanoma patients: validation
of the American Joint Committee on Cancer melanoma staging system.
J Clin Oncol. 19:3622–3634. 2001.PubMed/NCBI
|
|
3.
|
Agarwala SS: Current systemic therapy for
metastatic melanoma. Expert Rev Anticancer Ther. 9:587–595. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Bedikian AY, Millward M, Pehamberger H, et
al: Bcl-2 antisense (oblimersen sodium) plus dacarbazine in
patients with advanced melanoma: the Oblimersen Melanoma Study
Group. J Clin Oncol. 24:4738–4745. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Rini BI, Escudier B, Tomczak P, et al:
Comparative effectiveness of axitinib versus sorafenib in advanced
renal cell carcinoma (AXIS): a randomised phase 3 trial. Lancet.
378:1931–1939. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Rixe O, Bukowski RM, Michaelson MD, et al:
Axitinib treatment in patients with cytokine-refractory metastatic
renal-cell cancer: a phase II study. Lancet Oncol. 8:975–984. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Cohen EE, Rosen LS, Vokes EE, et al:
Axitinib is an active treatment for all histologic subtypes of
advanced thyroid cancer: results from a phase II study. J Clin
Oncol. 26:4708–4713. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Schiller JH, Larson T, Ou SH, et al:
Efficacy and safety of axitinib in patients with advanced
non-small-cell lung cancer: results from a phase II study. J Clin
Oncol. 27:3836–3841. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Hersey P, Bastholt L, Chiarion-Sileni V,
et al: Small molecules and targeted therapies in distant metastatic
disease. Ann Oncol. 20(Suppl 6): vi35–vi40. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Hu-Lowe DD, Zou HY, Grazzini ML, et al:
Nonclinical anti-angiogenesis and antitumor activities of axitinib
(AG-013736), an oral, potent, and selective inhibitor of vascular
endothelial growth factor receptor tyrosine kinases 1, 2, 3. Clin
Cancer Res. 14:7272–7283. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Fruehauf J, Lutzky J, McDermott D, et al:
Multicenter, phase II study of axitinib, a selective
second-generation inhibitor of vascular endothelial growth factor
receptors 1, 2, and 3, in patients with metastatic melanoma. Clin
Cancer Res. 17:7462–7469. 2011. View Article : Google Scholar
|
|
12.
|
Emmett MS, Dewing D and Pritchard-Jones
RO: Angiogenesis and melanoma - from basic science to clinical
trials. Am J Cancer Res. 1:852–868. 2011.PubMed/NCBI
|
|
13.
|
Ma J and Waxman DJ: Dominant effect of
antiangiogenesis in combination therapy involving cyclophosphamide
and axitinib. Clin Cancer Res. 15:578–588. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Ma J and Waxman DJ: Modulation of the
antitumor activity of metronomic cyclophosphamide by the
angiogenesis inhibitor axitinib. Mol Cancer Ther. 7:79–89. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Richardson, G and Dobish R: Chemotherapy
induced diarrhea. J Oncol Pharm Pract. 13:181–198. 2007. View Article : Google Scholar
|
|
16.
|
Jemal A, Siegel R, Xu J and Ward E: Cancer
statistics, 2010. CA Cancer J Clin. 60:277–300. 2010. View Article : Google Scholar
|
|
17.
|
Wilmes LJ, Pallavicini MG, Fleming LM, et
al: AG-013736, a novel inhibitor of VEGF receptor tyrosine kinases,
inhibits breast cancer growth and decreases vascular permeability
as detected by dynamic contrast-enhanced magnetic resonance
imaging. Magn Reson Imaging. 25:319–327. 2007. View Article : Google Scholar
|
|
18.
|
Nakahara T, Norberg SM, Shalinsky DR,
Hu-Lowe DD and McDonald DM: Effect of inhibition of vascular
endothelial growth factor signaling on distribution of extravasated
antibodies in tumors. Cancer Res. 66:1434–1445. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Spano JP, Chodkiewicz C, Maurel J, et al:
Efficacy of gemcitabine plus axitinib compared with gemcitabine
alone in patients with advanced pancreatic cancer: an open-label
randomised phase II study. Lancet. 371:2101–2108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Kindler HL, Ioka T, Richel DJ, et al:
Axitinib plus gemcitabine versus placebo plus gemcitabine in
patients with advanced pancreatic adenocarcinoma: a double-blind
randomised phase 3 study. Lancet Oncol. 12:256–262. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Rugo HS, Stopeck AT, Joy AA, et al:
Randomized, placebo-controlled, double-blind, phase II study of
axitinib plus docetaxel versus docetaxel plus placebo in patients
with meta-static breast cancer. J Clin Oncol. 29:2459–2465. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Bendell JC, Tournigand C, Bednarczyk M, et
al: Axitinib or bevacizumab (bev) plus FOLFOX or FOLFIRI as
second-line therapy in patients (pts) with metastatic colorectal
cancer (mCRC). J Clin Oncol. 29(suppl 4): abstr 478. 2011.
|
|
23.
|
Hiratsuka S, Nakamura K, Iwai S, et al:
MMP9 induction by vascular endothelial growth factor receptor-1 is
involved in lung-specific metastasis. Cancer Cell. 2:289–300. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Shafi MA and Bresalier RS: The
gastrointestinal complications of oncologic therapy. Gastroenterol
Clin North Am. 39:629–647. 2010. View Article : Google Scholar
|