Introduction
Hypopharyngeal carcinoma is a type of malignancy
that arises from the mucosal epithelia in the hypopharynx (1). At present, in addition to surgery,
adjuvant chemotherapy is important for the multidisciplinary
treatment of hypopharyngeal cancer. However, the resistance of
cancer cells to chemotherapeutic agents remains an unquestionable
entity, which hampers treatment planning and successful tumor
control in routine clinical practice. Among various causes
responsible for therapeutic resistance, hypoxia of the cancer
microenvironment is proposed to be important (2). Hypoxia evokes a series of
intracellular signal cascades, resulting in cell cycle arrest,
limited diffusion of drugs and changes in gene transcription
activity, which contributes to tumor growth, angiogenesis and
metastasis and, thereby, enhances the resistance of cancer cells to
chemotherapeutic drugs. Previous studies have revealed that tumor
cells adapt to hypoxia by hypoxia-inducible factor (HIF)-dependent
and -independent pathways (3,4). It
has been previously demonstrated that HIF-1α is induced by mild to
moderate hypoxia with oxygen concentrations ranging between 5 and
0.1%, in vitro and in vivo. By contrast, the
induction of HIF-1α in response to severe hypoxia (oxygen
concentration, <0.02%) is considered a functionally different
state (3,5) and requires further investigation.
Due to an insufficient blood supply and poor
oxygenation in solid tumors, including head and neck squamous cell
carcinoma (HNSCC), severe hypoxia in the tumor region, particularly
in the central section of the tumor bulk, is a persisting status.
Although cancer cells are considered to be unable to survive in
tumor tissues with extremely low oxygen concentrations, they are
well adapted to complete anoxia by HIF-independent pathways, for
example the unfolded protein response (UPR), and exhibit prolonged
survival times (6). It has been
previously identified that hypoxia activates UPR and causes an
increase in glucose-regulated protein 78 (GRP78) expression, the
latter of which is also referred to as the immunoglobulin heavy
chain binding protein. GRP78 is an important molecular chaperone
that resides in the endoplasmic reticulum (ER) and belongs to the
HSP70 protein family. In non-stressed cells, GRP78 binds to three
transmembrane ER sensors, PERK, IRE1a and ATF6, and maintains them
in an inactive form. In tumor cells suffering from stresses,
including glucose starvation, hypoxia and oxidative stimulation,
unfolded proteins accumulate in the ER lumen, which, in turn,
causes dissociation of GRP78 from the three ER transmembrane
sensors, leading to their activation and triggering of UPR
(7).
GRP78, the most important marker of UPR, has been
reported to be elevated in a variety of tumors, including breast
(8), ovarian (9) and prostate (10). In human cancers, GRP78 protects
tumor cells from apoptosis, contributing to tumor cell
proliferation, survival and therapeutic resistance. However, the
roles of GRP78 in regulating the growth and survival of
hypopharyngeal carcinoma cells with regard to the severity of
hypoxia remain to be elucidated. The aim of the present study was
to investigate the changes of GRP78 in response to various
conditions of hypoxia, and its association with chemoresistance in
hypopharyngeal carcinoma. It was demonstrated that the induction of
GRP78 by severe hypoxia is a cause of chemoresistance and knockdown
of GRP78 with short hairpin RNA (shRNA) enhances the
chemosensitivity of hypopharyngeal carcinoma cells to cisplatin
(DDP) in response to severe hypoxia.
Materials and methods
Cell line and lentivirus infection
FaDu hypopharyngeal squamous carcinoma cell line was
purchased from the Type Culture Collection of the Chinese Academy
of Sciences (Shanghai, China). Cells were grown at 37°C in a
CO2 incubator with 5% CO2 and 95% humidity
and Dulbecco’s modified Eagle’s medium (with 4.5 mg/ml glucose)
supplemented with 10% fetal bovine serum (Hangzhou Sijiqing Biology
Engineering Materials Co., Ltd., Hangzhou, China), 100 U/ml
penicillin, 100 μg/ml streptomycin and 4 mM L-glutamine (Gibco,
Paisley, United Kingdom). The shRNA eukaryotic plasmid expression
vector kit was purchased from GeneCopoeia, Inc. (Guangzhou, China).
For infection, 3×105/2 ml FaDu cells were plated in
six-well plates, infected with the lentivirus containing a shRNA
targeting GRP78 (shGRP78; 5′-TTCCGGTCTACTATGAAGC-3′) or a scrambled
oligonucleotide (scshRNA; 5′-GCTTCGCGCCGTAGTCTTA-3′) and sorted by
flow cytometry to ensure 100% positivity. Established stable cell
lines were named GRP78(+/+) and GRP78(−/−) and cultured in the
medium described above (containing 2.5 μg/ml puromycin) for
subsequent usage.
Hypoxia incubation and DDP treatment
Hypoxic conditions were achieved and maintained by
regulating the gas flow rates under 95% N2 and 5%
CO2. The oxygen level within the incubation chamber was
monitored with an anaerobic indicator (Zirconia Oxygen Analyzer;
Nanjing Nensite Instrument Co., Ltd., Nanjing, China). To acquire
moderate hypoxia, gas purging (3 l/min) with 95% N2 and
5% CO2 was performed for 8–9 min until the oxygen level
reached 1%, when the purge gas was replaced by a gas mixture of 1%
O2, 94% N2 and 5% CO2. The gas
flow rate was adjusted steadily at 0.8 l/min and oxygen levels were
maintained at 1%. To achieve severe hypoxia, gas purging (3 l/min)
was performed for 12–13 min until the oxygen level reached 0.02%
and, simultaneously, the gas flow rate was adjusted steadily at 0.8
l/min. The oxygen level had to be maintained for the duration of
the procedure for each separate experiment.
To investigate the effect of various hypoxic
conditions on the protein expression of GRP78 and C/EBP homology
protein (CHOP), FaDu cells were cultured in six-well plates under
moderate and severe hypoxia for the indicated time periods. For
immunocytochemical analysis, cover glasses were placed in the wells
of the six-well plates, to which 3.0×105 GRP78(+/+) or
GRP78(−/−) cells were seeded per well and grown overnight in an
incubator (37°C and 20% O2), followed by culture under
severely hypoxic conditions for 24 h. To investigate the effects of
GRP78 on the chemosensitivity of hypopharyngeal carcinoma cells,
GRP78(+/+) and GRP78(−/−) cells were seeded in 96-well plates at a
density of 5×103 per well, incubated under severe
hypoxia and treated with various concentrations of DDP for 24 h. In
the additional experiments, including cell apoptosis and protein
regulation assays, GRP78(+/+) and GRP78(−/−) cells were plated at
3×105 cells/well in six-well plates and treated with DDP
(40 μmol/l) under severe hypoxia for 24 h.
Fluorescent quantitative real-time
polymerase chain reaction (qPCR)
Total RNA was isolated using TRIzol reagent
(Invitrogen Life Technologies, Carlsbad, CA, USA) and the cDNA was
synthesized by reverse transcription according to the
manufacturer’s instructions for the HiFi-MMLV cDNA kit (cat. no.
CW0744; CoWin Biotech Co., Ltd., Beijing, China). qPCR was
performed using the ABI Prism 7500HT Sequence Detection system
(Applied Biosystems, Inc., Foster City, CA, USA). Reactions were
performed in 20 μl volumes. qPCR parameters were as follows:
Initial denaturation at 95°C for 10 min, followed by 40 cycles of
95°C for 15 sec and 60°C for 60 sec. The mRNA expression level was
determined using the 2−ΔΔCt method and actin was used as
internal control. The primers used for qPCR were as follows:
Forward: 5′-CTGTAGCGTATGGTGCTGCTGTCC-3′ and reverse:
5′-TGACACCTCCCACAGTTTCAATACCA-3′ for GRP78; and forward:
5′-ACTTAGTTGCGTTACACCCTT-3′ and reverse: 5′-GTCACCTTCACCGTTCCA-3′
for actin.
Western blot analysis
Cells from corresponding experiments were harvested
and washed with ice-cold phosphate-buffered saline (PBS) and total
protein was extracted with ice-cold lysis buffer. Protein
concentrations were quantified according to the manufacturer’s
instructions for the BCA protein assay kit (Pierce Biotechnology
Inc., Rockford, IL, USA). For protein denaturation, the mixture of
total protein and 3X loading buffer [0.1 M Tris-Cl, (pH 6.8), 4%
SDS, 0.2% bromophenol blue and 20% glycerol] was heated at 97°C for
5 min in a Thermomixer comfort (Eppendorf, Hamburg, Germany). Equal
amounts of protein (30 μg) were separated by 12% SDS-PAGE and
transferred to a polyvinylidene fluoride membrane, the latter of
which was blocked in 5% skimmed milk for 2 h at 37°C.
Immunoblotting was performed using primary antibodies, including
GRP78 (1:1000), CHOP/GADD153 (1:200) and HIF-1α (1:1000) (all
Abcam, Cambridge, UK), and Bcl-2, Bax and β-actin (all 1:200; Santa
Cruz Biotechnology, Inc., Santa Cruz, CA, USA). Horseradish
peroxidase-conjugated goat anti-mouse or anti-rabbit antibody was
used as a secondary antibody (1:5000; Beijing Zhongshan
Goldenbridge Bio-technology Co., Ltd., Beijing, China). Protein
expression levels were detected using the RPN2132 enhanced
chemiluminescence plus western blotting detection system (GE
Healthcare Life Sciences, Amersham, UK). The bands were exposed to
film in a dark chamber and the relative expression levels of each
separate protein were determined by densitometry using a gel image
analyzing system (UVP, LLC, Upland, CA, USA).
Immunocytochemistry
Cover glasses grown with GRP78(+/+) and GRP78(−/−)
cells were washed twice with PBS (pH 7.4) and fixed with 4%
paraformaldehyde for 20 min. Cells on cover glasses were
permeabilized with 0.3% Triton X-100 for 30 min and incubated with
goat serum for 20 min at 37°C to block the non-specific binding of
the antibody. Subsequently, cells were incubated with primary
antibody (rabbit antihuman GRP78 polyclonal antibody, 1:500; and
mouse antihuman CHOP monoclonal antibody, 1:100; Abcam) at 4°C
overnight. Following washing, cells were incubated with
goat-anti-rabbit antibody for 10 min at room temperature, labeled
with horseradish-peroxidase-labeled pronase avidin and
3,3′-Diaminobenzidine reagent. Cells were then counterstained with
hematoxylin for 5 min and dehydrated with gradient alcohol
solutions. Cover glasses were mounted onto the glass slide with
neutral resin and visualized using an optical microscope
(magnification, ×200; VANOX-S, Olympus, Tokyo, Japan). Optical
density (OD) values were measured using Image-Pro Plus software
(Media Cybernetics Inc., Rockville, MD, USA).
MTT cell viability assay
MTT reagent (20 μl; 5 mg/ml; Sigma-Aldrich, St.
Louis, MO, USA) was added to each well of a 96-well plate, 4 h
prior to the indicated time points. Following 4 h of incubation at
37°C, the culture medium was removed from each well and the
precipitate from each representative well was dissolved in 150 μl
dimethyl sulfoxide. The OD values were measured at 409 nm using an
enzyme-linked immunosorbent detector (Model 550; Bio-Rad, Hercules,
CA, USA).
Flow cytometry of cell apoptosis
Treated cells in each well were trypsinized and
collected by centrifugation at 200 × g for 4 min. Next, the
harvested cells were washed twice with cold PBS and fixed with 70%
ethanol at 40°C overnight. Cells were resuspended in PBS containing
40 μg/ml propidium iodide (PI), 0.1 mg/ml RNase A and 0.1% triton
X-100 in a dark room for 30 min at 37°C. Counting of apoptotic
cells was carried out by measuring sub-G1 DNA content using the PI
method with a FACS Aria flow cytometer (Becton-Dickinson, Franklin
Lakes, NJ, USA).
Statistical analysis
Data are presented as the mean ± standard deviation.
One-way analysis of variance and Student’s t-test were performed
for statistical evaluation of data. Statistical comparisons were
performed using SPSS software for Windows (version 13.0; SPSS,
Inc., Chicago, IL, USA), and P<0.05 was considered to indicate a
statistically significant difference.
Results
Establishment of FaDu hypopharyngeal
carcinoma cell line with knockdown of GRP78
To investigate the role of GRP78 in regulating the
proliferation and growth of FaDu cells under hypoxic conditions,
lentivirus containing shGRP78 or scshRNA was used to infect FaDu
cells, generating stable cell lines defined as GRP78(−/−) and
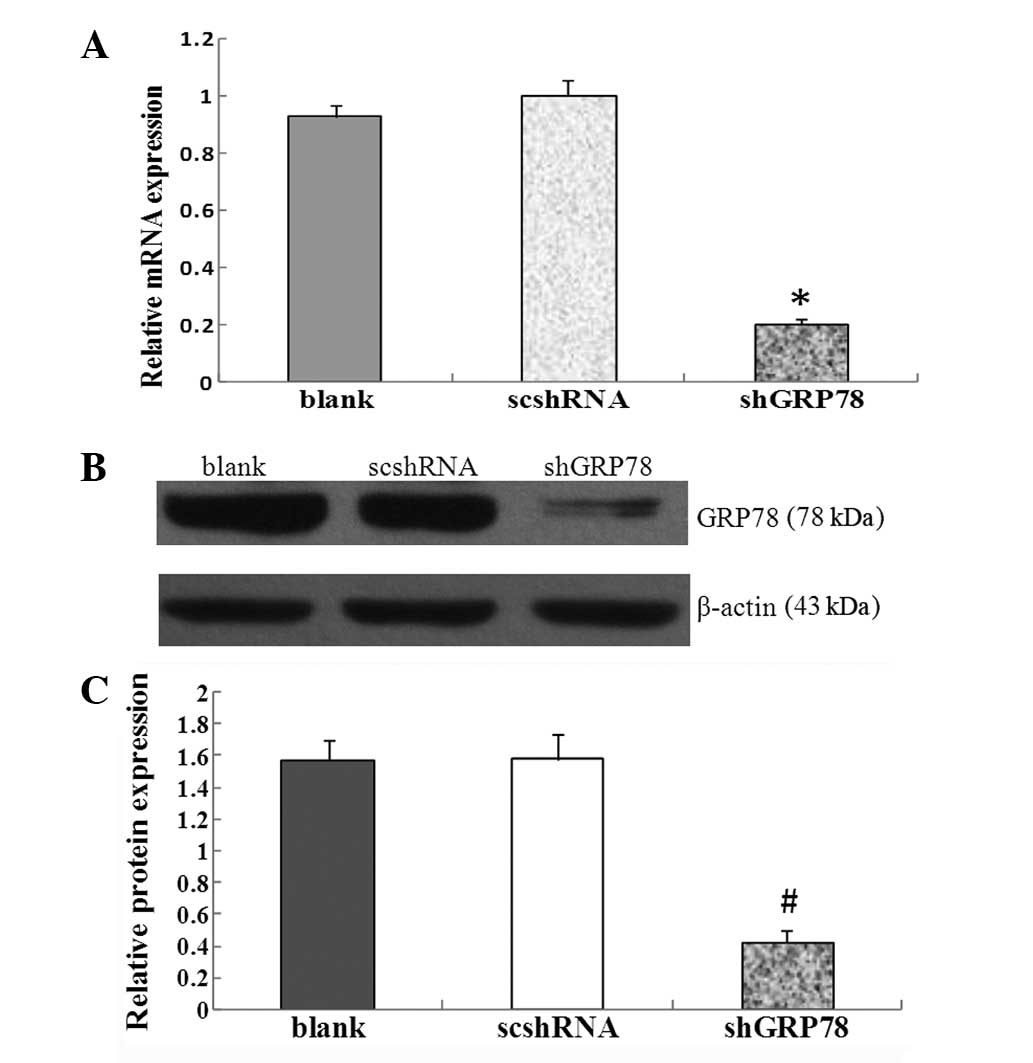
GRP78(+/+). As demonstrated by qPCR and western blot analysis,
downregulation of GRP78 was at mRNA and protein levels in
GRP78(−/−) cells as opposed to GRP78(+/+) cells. In quantitative
analysis, the GRP78 shRNA plasmid caused a significant reduction in
GRP78 expression at mRNA (P<0.01) and protein (P<0.01) levels
under normoxic conditions (Figs. 1A and
B). It was demonstrated that the hypopharyngeal carcinoma cell
line with knockdown of GRP78 [GRP78(−/−)] was successfully
established.
Effects of various hypoxic conditions on
the expression of major proteins in UPR pathways in hypopharyngeal
carcinoma cells
GRP78 and CHOP are two main regulator proteins
downstream of the UPR pathways. The expression of GRP78 and CHOP
under various hypoxic conditions was investigated in FaDu cells by
western blot analysis. Under normoxic conditions, the expression of
GRP78 and CHOP remained almost at the same level for up to 24 h
(Fig. 2A). However, expression of
GRP78 and CHOP was upregulated in FaDu cells under severe hypoxia
(Fig. 2C). The expression of GRP78
protein, a marker in pro-survival signaling of UPR, increased
gradually under severe hypoxia between 3 and 9 h and remained
almost unchanged for up to 24 h. Severe hypoxia also leads to the
induction of CHOP, a major proapoptotic arm of the UPR. CHOP
protein levels were observed to increase gradually between 12 and
24 h, and peaked at 24 h (Fig. 2C).
Under moderate hypoxia, HIF-1α, an important marker of hypoxia,
increased gradually between 6 and 24 h, and peaked at 24 h.
However, under severe hypoxia, the expression of HIF-1α was
markedly increased within 3 h, maintained at an even level between
6 and 12 h, and significantly reduced at 24 h. This indicated that
in the later stages of severe hypoxia, HIF-1α-independent pathways,
including UPR, may dominate the response of the cell to hypoxic
conditions.
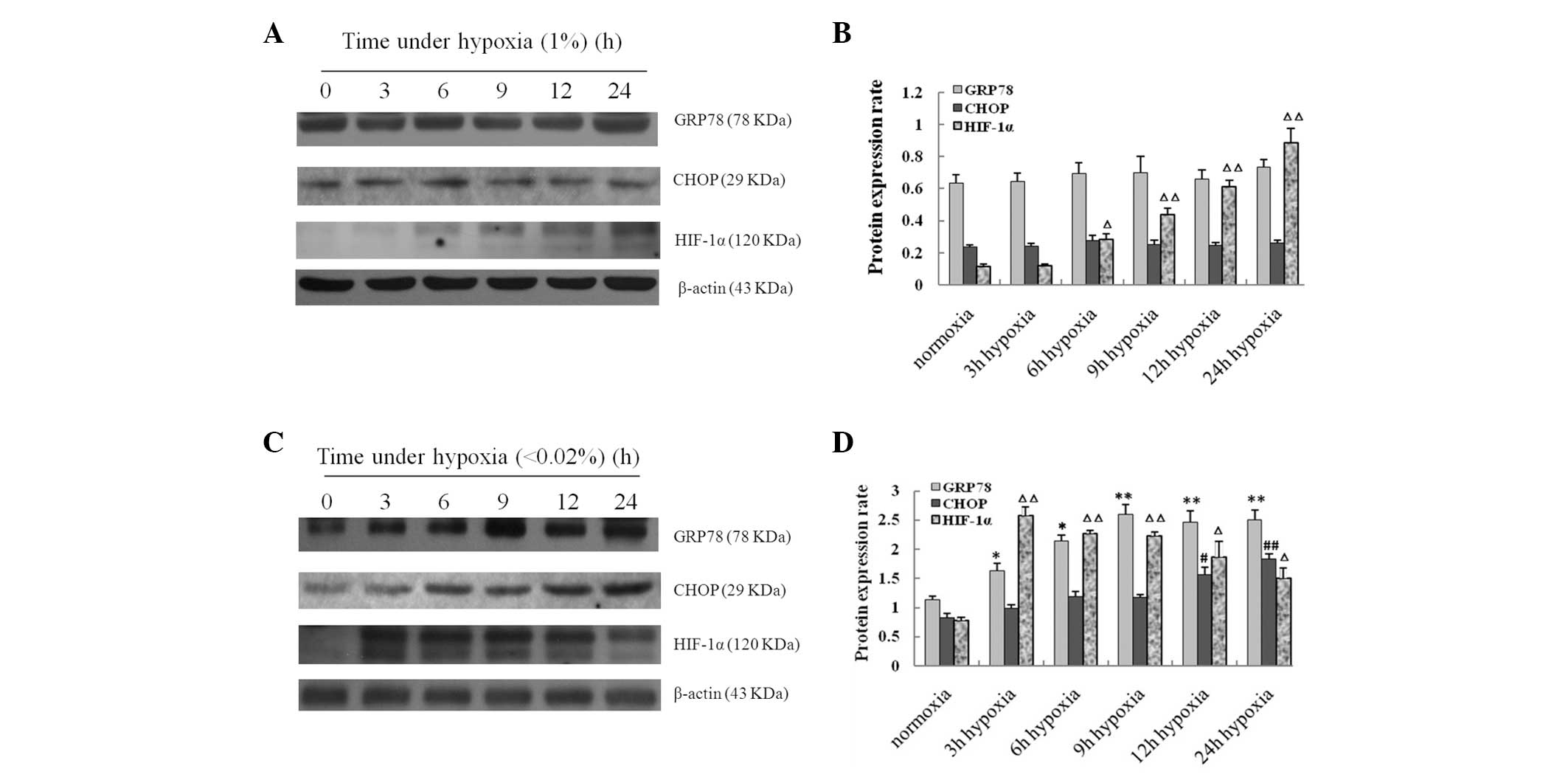 | Figure 2Changes of GRP78, CHOP and HIF-1α
expression levels in FaDu cells following exposure to moderate and
severe hypoxia for 3, 6, 9, 12 and 24 h, respectively. Expression
of GRP78, CHOP and HIF-1α were detected by western blot analysis.
Results from western blot analysis in FaDu cells exposed to (A)
moderate and (C) severe hypoxia. (B and D) Histograms of the
expression levels of GRP78, CHOP and HIF-1α proteins in FaDu cells
determined by western blot analysis. Data shown are from three
individual experiments. *P<0.05 and
**P<0.01, vs. normoxic conditions (GRP78);
#P<0.05, ##P<0.01, vs. normoxic
conditions (CHOP); ΔP<0.05, ΔΔP<0.01,
vs. normoxic conditions (HIF-1α). GRP78, glucose-regulated protein
78; CHOP, C/EBP homology protein; HIF-1α, hypoxia-inducible
factor-1α. |
Induction of GRP78 by severe hypoxia is
associated with chemoresistance of hypopharyngeal carcinoma cells
to DDP
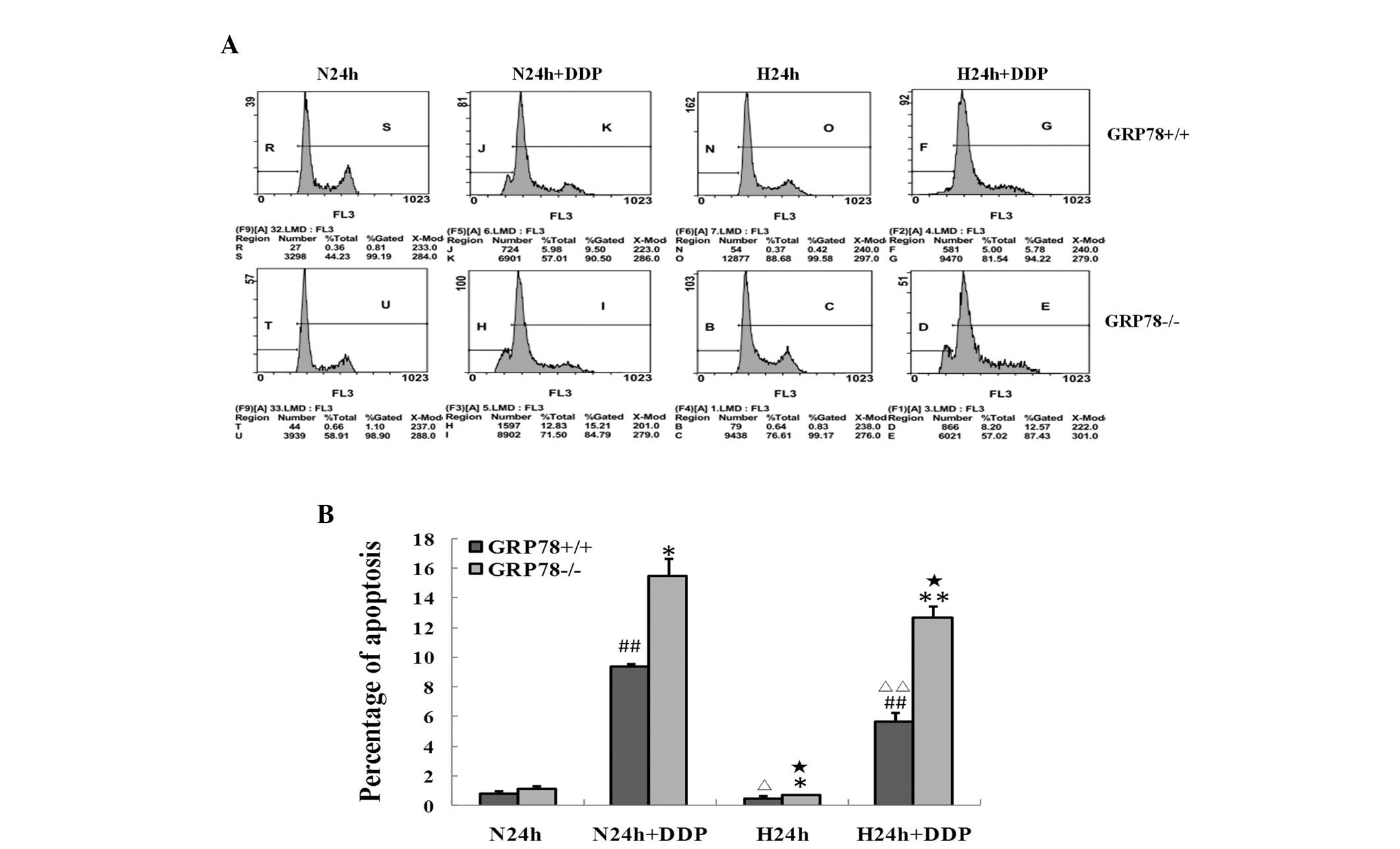
To investigate the role of GRP78 in regulating cell
proliferation under severely hypoxic conditions, GRP78(+/+) and
GRP78(−/−) cells were cultured under severe hypoxia and treated
with various concentrations of DDP for 24 h. As demonstrated by MTT
assay (Fig. 3B), DDP inhibited the
proliferation of GRP78(+/+) and GRP78(−/−) cells in a
dose-dependent manner. However, the proliferation inhibition rates
of GRP78(+/+) cells under severe hypoxia were markedly lower than
those under normoxic conditions (P<0.01; Fig. 3A), indicating that severe hypoxia is
a major cause of chemoresistance of hypopharyngeal carcinoma cells
to DDP. Moreover, a stronger inhibitory effect was observed in
GRP78(−/−) cells compared with that in GRP78(+/+) cells
(P<0.01), indicating that induction of GRP78 under hypoxia
impacts the chemosensitivity of hypopharyngeal carcinoma cells to
DDP. Consistently, DDP induced apoptosis in GRP78(+/+) and
GRP78(−/−) cells under normoxic and severely hypoxic conditions
(Figs. 4A and B). However, the
apoptosis rates induced by DDP were markedly less in GRP78(+/+)
cells (P<0.01) compared with GRP78(−/−) cells under severe
hypoxia. These observations indicate that GRP78 confers
hypopharyngeal carcinoma cells to chemoresistance induced by severe
hypoxia and knockdown of GRP78 sensitized DDP-induced apoptosis of
hypopharyngeal carcinoma cells under severely hypoxic
conditions.
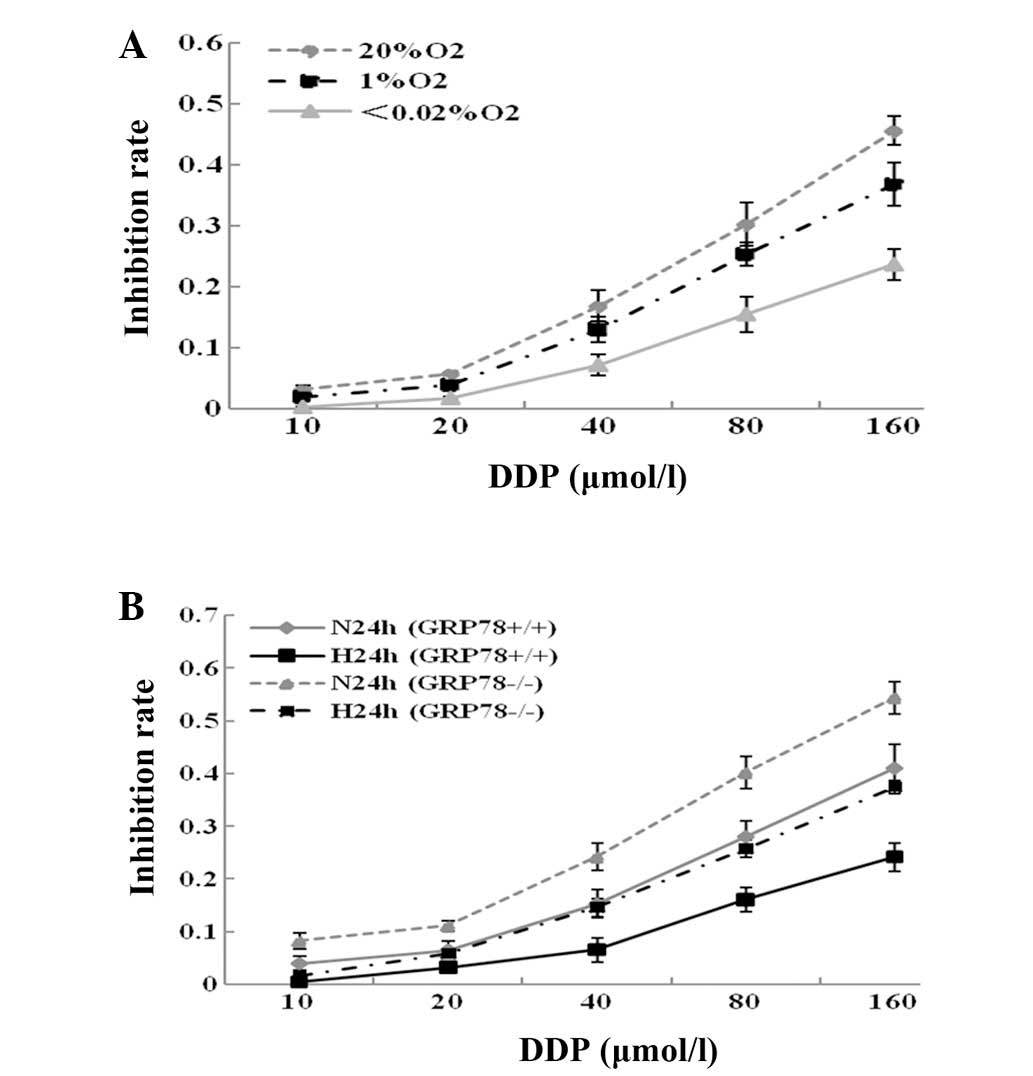 | Figure 3Results of cell proliferation
inhibition assays by MTT. (A) FaDu cells were treated with 10, 20,
40, 80 and 160 μmol/l of DDP for 24 h under normoxia, and moderate
and severe hypoxia. Compared with FaDu cells treated under normoxic
conditions, the proliferation inhibition rates of FaDu cells were
significantly decreased under moderate (P<0.05) and severe
(P<0.01) hypoxia. (B) GRP78(+/+) and GRP78(−/−) hypopharyngeal
carcinoma cells were treated with various concentrations (10, 20,
40, 80 and 160 μmol/l) of DDP for 24 h under normoxia and severe
hypoxia. Results showed that knockdown of GRP78 with shRNA greatly
increased cell proliferation inhibition induced by DDP under
normoxia and severe hypoxia in hypopharyngeal carcinoma cells (all
P<0.01). All experiments were performed in triplicate. GRP78,
glucose-regulated protein 78; N24h, normoxia for 24 h; H24h, severe
hypoxia for 24 h; shRNA, short hairpin RNA; DDP, cisplatin. |
GRP78 regulates the expression of CHOP,
Bcl-2 and Bax under severe hypoxia
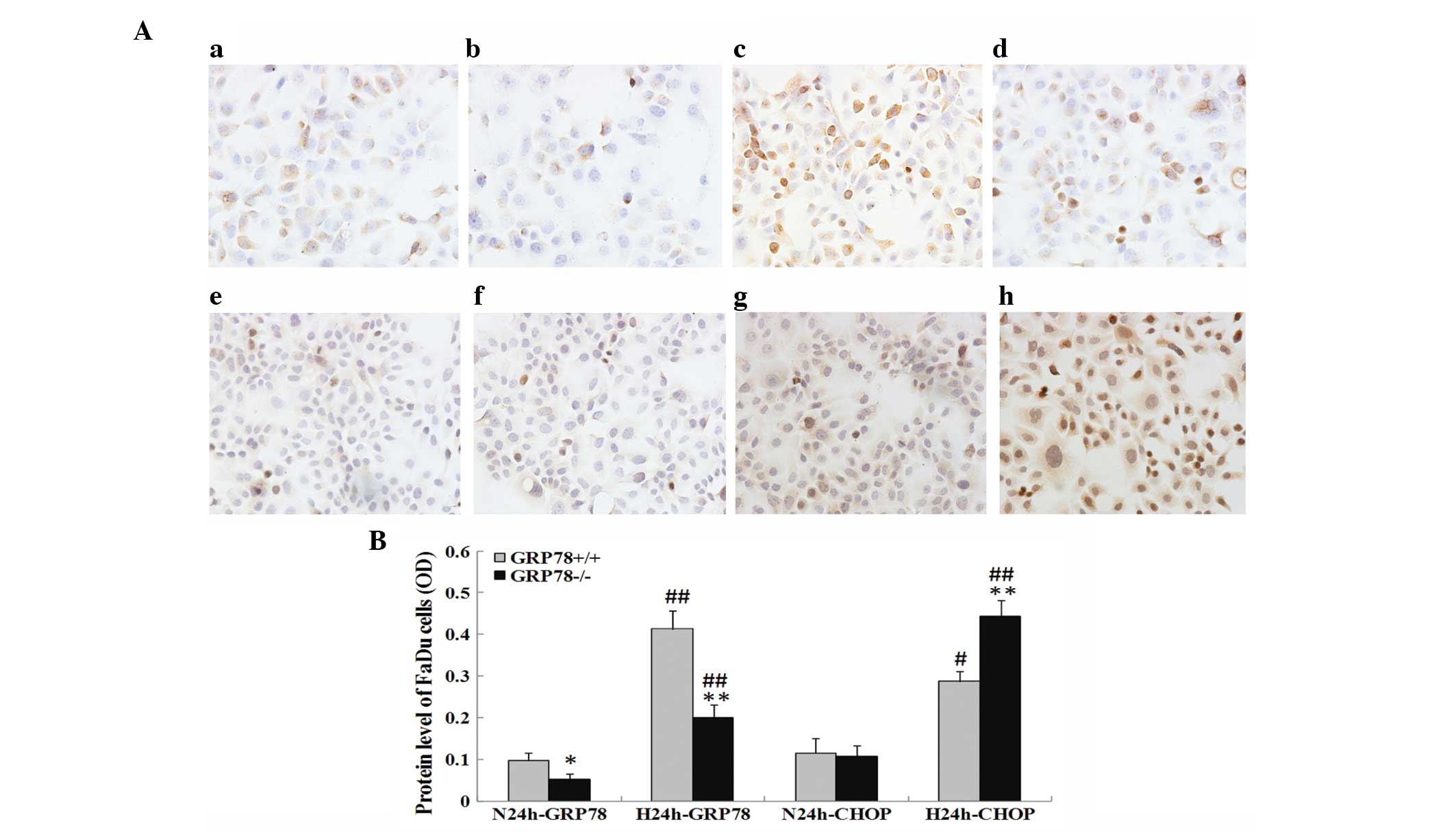
Expression of GRP78 and CHOP in GRP78(+/+) and
GRP78(−/−) cells under severe hypoxia was initially investigated by
immunohistochemistry. It was revealed that the expression of GRP78
was mainly located in the cytoplasm or cytomembrane, whereas the
expression of CHOP was mainly found in the cytoplasm or nucleus
(Fig. 5A). Compared with normoxic
conditions, severe hypoxia induced a significant increase in the
protein expression levels of GRP78 (P<0.01) and CHOP (P<0.05)
(Fig. 5B). Notably, knockdown of
GRP78 led to significantly increased CHOP expression under severe
hypoxia (P<0.01; Figs. 5A and
B).
The expression of GRP78 and CHOP and their
downstream effector proteins, Bcl-2 and Bax, in GRP78(+/+) and
GRP78(−/−) cells was also investigated by western blot analysis
following incubation under normoxic and severely hypoxic conditions
(Figs. 6A and B). It was found that
GRP78 expression was significantly decreased in GRP78(−/−) cells
(P<0.01). The increase in the protein level of CHOP was observed
under severe hypoxia in GRP78(+/+) cells (P<0.05), however, it
was more prominent in GRP78(−/−) cells. This indicated that
knockdown of GRP78 may promote the expression of CHOP in the later
stages of UPR induced by hypoxia. Blocking GRP78 by shRNA also
resulted in the downregulation of Bcl-2 and upregulation of Bax
under severe hypoxia (all P<0.01).
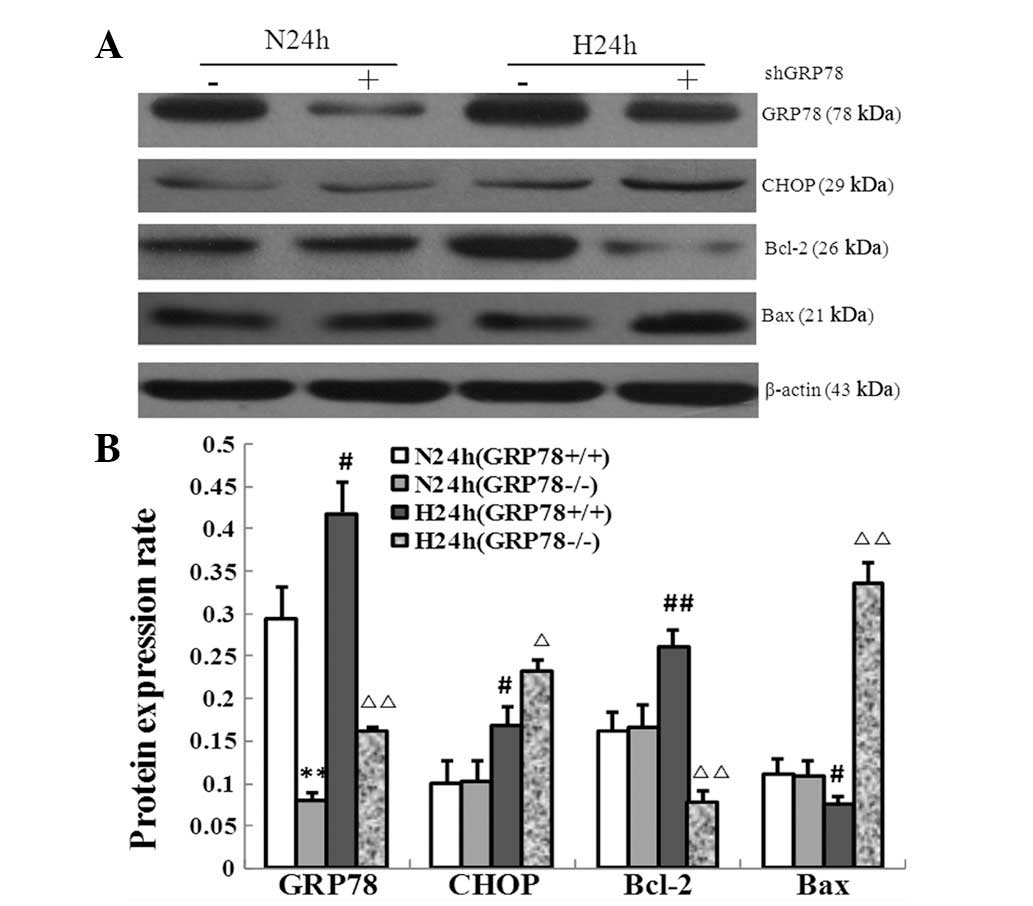 | Figure 6Changes in the expression of
apoptosis-regulating proteins following the knockdown of GRP78 with
shRNA. (A) Protein levels of GRP78, CHOP, Bcl-2 and Bax following
knockdown of GRP78 in hypopharyngeal carcinoma cells were measured
by immunoblot analysis under normoxic and severely hypoxic cultures
for 24 h. (B) Histogram of the protein expression levels of GRP78,
CHOP, Bcl-2 and Bax in GRP78(+/+) and GRP78(−/−) cells under
normoxia and severe hypoxia for 24 h. **P<0.01, vs.
N24h GRP78(+/+); #P<0.05 and ##P<0.01,
vs. N24h GRP78(+/+); ΔP<0.05 and
ΔΔP<0.01, vs. H24h GRP78(+/+). Data shown are from
three individual experiments. GRP78, glucose-regulated protein 78;
CHOP, C/EBP homology protein; shRNA, short hairpin RNA; N24h,
normoxia; H24h, severe hypoxia. |
Discussion
HNSCC ranks as the sixth most common malignancy of
the human body worldwide and exhibits formidable features of
resistance to chemotherapeutic agents (11). Hypoxia is a major cause of
therapeutic resistance and prevails in the tumor bulks
simultaneously with the development and progression of solid
tumors, including hypopharyngeal carcinoma. Typically, tumor cells
suffer from stresses, including glucose starvation, hypoxia and
oxidative stress, due to an insufficient supply of nutrients, which
activates intracellular signaling pathways, including UPR, to
overwhelm intra-plasma disorders and protect against cell death
(12).
UPR is a biological behavior of cancer cells, which
functions to degrade unfolded protein for the preservation of
energy and activation of anti-apoptotic factors. For example,
GRP78, the most important marker of UPR, has been identified as a
survival factor for rescuing cells from cell death and
overexpression of GRP78 has been reported to be associated with
poor prognosis in a number of malignant tumors (13,14).
Nevertheless, the role of GRP78 in regulating the proliferation and
growth of hypopharyngeal carcinoma cells under severe hypoxia
remains to be unveiled. The present study demonstrated that
hypopharyngeal carcinoma cells are more resistant to DDP under
severe hypoxia, which was made evident by results from the
proliferation inhibition and apoptosis assays. In addition, the
rates of proliferation inhibition and apoptosis induced by DDP were
markedly increased in GRP78(−/−) cells, indicating that the
induction and activation of GRP78 is responsible for
chemoresistance in response to severe hypoxia.
Activation of GRP78 may vary with the severity of
hypoxia (15). It has been observed
in the present study that GRP78 was substantially upregulated under
severe hypoxia when compared with normoxia, which is consistent
with observations from previous studies (16). By contrast, normoxia or moderate
hypoxia brought almost no change to GRP78 expression. Notably, it
was found that hypoxic conditions (oxygen concentration, <0.02%)
are necessary for UPR activation. Therefore, severe hypoxia rather
than moderate hypoxia leads to UPR activation in hypopharyngeal
carcinoma cells.
In addition, the present study revealed that severe
hypoxia leads to the induction of CHOP, an additional significant
regulating protein of the proapoptotic arm of UPR. Moreover, the
induction of CHOP was preceded by the upregulation of GRP78,
indicating that the gene expression of UPR in response to severe
hypoxia is during dynamic changes, depending on the stage and
status of hypoxia. This is partly reflected by the corresponding
changes observed in HIF-1α, an important marker of hypoxia.
To further explore the mechanism of the knockdown of
GRP78 for the sensitization of hypopharyngeal carcinoma cells to
DDP under severe hypoxia, the interaction between GRP78 and CHOP
and its effects on the downstream apoptosis-regulating proteins
Bcl-2 and Bax was investigated. A previous study by Pyrko et
al demonstrated that the inhibition of GRP78 may upregulate
CHOP expression in the absence of any treatment under normoxic
conditions in malignant glioma cells (17). Similarly, the current study found
that the expression levels of GRP78 and CHOP were significantly
increased under severe hypoxia and downregulation of GRP78 by shRNA
was accompanied with the simultaneous upregulation of CHOP. The
results indicate that similar regulating mechanisms may exist
between GRP78 and CHOP in hypopharyngeal carcinoma cells and a
checkpoint of the pro-apoptosis pathway is initiated when the
pro-survival pathway, dominated by GRP78, is blocked under severe
hypoxia.
Elevated GRP78 expression prevents tumor cells from
stress-induced apoptosis, suppressing apoptosis and protecting
cells against cisplatin and hypoxia. It has been previously
reported that a fraction of GRP78 exists as an ER transmembrane
protein that forms a complex with caspase-7 or caspase-12. The
formation of this complex is dependent on its ATP binding domain
and directly inhibits the activity of proapoptotic effectors
localized in the ER (18). GRP78
overexpression inhibits Bax activation, which is also occurring in
hypopharyngeal carcinoma cells under severe hypoxia as observed in
the present study. In addition, knockdown of GRP78 leads to Bax
activation (19) and provokes
cytochrome c release from the mitochondria. Functionally,
GRP78 and antiapoptotic protein Bcl-2, are capable of forming
separate complexes with BIK. Elevated GRP78 expression leads to a
reduction of Bcl-2 binding to BIK and Bcl-2 sequestration by BIK at
the endoplasmic reticulum (20),
which prevents endoplasmic reticulum Ca2+ release,
thereby, suppressing apoptosis. Downregulation of GRP78 may
upregulate the pro-apoptosis factor of CHOP, which has been
reported to downregulate the expression of antiapoptotic Bcl-2
(21) and enhance apoptosis. In a
previous study analyzing heterozygous knockout mice, it was
identified that a lower level of GRP78 leads to the specific
induction of CHOP and activation of executioner caspase-3 and
caspase-7 (22). CHOP is also known
as growth arrest and DNA damage-inducible gene 153, which implies
that it induced apoptosis through direct inhibition, causing DNA
damage. The crosstalk between the mitochondria and ER through
interactions between GRP78 and CHOP determines the fate of the cell
growth and proliferation of hypopharyngeal carcinoma cells
following treatment with DDP in response to severe hypoxia.
In conclusion, induction of GRP78 through UPR is
determined by the severity of hypoxia. Severe hypoxia causes UPR
with induction of GRP78 expression in hypopharyngeal carcinoma,
which is a major cause of chemoresistance to DDP. Downregulation of
GRP78 upregulates CHOP and the proapoptotic factor Bax, inhibiting
the expression of the antiapoptotic factor Bcl-2, thus, resulting
in the inhibition of cell growth and proliferation. Knockdown of
GRP78 sensitizes hypopharyngeal carcinoma cells to DDP treatment
and alleviates hypoxia-induced chemoresistance to DDP. Therefore,
downregulation of GRP78 may become a promising adjuvant treatment
strategy for overcoming the therapeutic resistance of HNSCC.
Acknowledgement
This study was granted in part by Natural Science
Foundation of Hebei Province (no. H2012505011) and by Wu Jieping
Medical Research Foundation (no. 320.6750.10121).
References
|
1
|
Chien CY, Su CY, Hwang CF, Chuang HC,
Hsiao YC, Wu SL and Huang CC: Clinicopathologic significance of
CD105 expression in squamous cell carcinoma of the hypopharynx.
Head Neck. 28:441–446. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Brown JM and Wilson WR: Exploiting tumour
hypoxia in cancer treatment. Nat Rev Cancer. 4:437–447. 2004.
View Article : Google Scholar
|
|
3
|
Wouters BG and Koritzinsky M: Hypoxia
signalling through mTOR and the unfolded protein response in
cancer. Nat Rev Cancer. 8:851–864. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ghosh R, Lipson KL, Sargent KE, Mercurio
AM, Hunt JS, Ron D and Urano F: Transcriptional regulation of
VEGF-A by the unfolded protein response pathway. PLoS ONE.
5:e95752010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rzymski T, Milani M, Pike L, et al:
Regulation of autophagy by ATF4 in response to severe hypoxia.
Oncogene. 29:4424–4435. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Koumenis C and Wouters BG: ‘Translating’
tumor hypoxia: unfolded protein response (UPR)-dependent and
UPR-independent pathways. Mol Cancer Res. 4:423–436. 2006.
|
|
7
|
Rutkowski DT and Kaufman RJ: A trip to the
ER: coping with stress. Trends Cell Bio. 14:20–28. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Grkovic S, O’Reilly VC, Han S, Hong M,
Baxter RC and Firth SM: IGFBP-3 binds GRP78, stimulates autophagy
and promotes the survival of breast cancer cells exposed to adverse
microenvironments. Oncogene. 32:2412–2420. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kandala PK and Srivastava SK: Regulation
of macroautophagy in ovarian cancer cells in vitro and in vivo by
controlling glucose regulatory protein 78 and AMPK. Oncotarget.
3:435–449. 2012.PubMed/NCBI
|
|
10
|
de Ridder G, Ray R, Misra UK and Pizzo SV:
Modulation of the unfolded protein response by GRP78 in prostate
cancer. Methods Enzymol. 489:245–257. 2011.PubMed/NCBI
|
|
11
|
Jemal A, Siegel R, Ward E, Hao Y, Xu J,
Murray T and Thun MJ: Cancer statistics, 2008. CA Cancer J Clin.
58:71–96. 2008. View Article : Google Scholar
|
|
12
|
Sun Q, Li X, Lu X and Di B: Cancer stem
cells may be mostly maintained by fluctuating hypoxia. Med
Hypotheses. 76:471–473. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Huang LW, Lin CY, Lee CC, Liu TZ and Jeng
CJ: Overexpression of GRP78 is associated with malignant
transformation in epithelial ovarian tumors. Appl Immunohistochem
Mol Morphol. 20:381–385. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kuroda K, Horiguchi A, Asano T, et al:
Glucose-regulated protein 78 positivity as a predictor of poor
survival in patients with renal cell carcinoma. Urol Int.
87:450–456. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Koumenis C, Bi M, Ye J, Feldman D and
Koong AC: Hypoxia and the unfolded protein response. Methods
Enzymol. 435:275–293. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Krivoruchko A and Storey KB: Activation of
the unfolded protein response during anoxia exposure in the turtle
Trachemys scripta elegans. Mol Cell Biochem. 374:91–103.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pyrko P, Schönthal AH, Hofman FM, Chen TC
and Lee AS: The unfolded protein response regulator GRP78/BiP as a
novel target for increasing chemosensitivity in malignant gliomas.
Cancer Res. 67:9809–9816. 2007. View Article : Google Scholar
|
|
18
|
Healy SJ, Gorman AM, Mousavi-Shafaei P,
Gupta S and Samali A: Targeting the endoplasmic reticulum-stress
response as an anticancer strategy. Eur J Pharmacol. 625:234–246.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fu Y, Li J and Lee AS: GRP78/BiP inhibits
endoplasmic reticulum BIK and protects human breast cancer cells
against estrogen starvation-induced apoptosis. Cancer Res.
67:3734–3740. 2007. View Article : Google Scholar
|
|
20
|
Zhou H, Zhang Y, Fu Y, Chan L and Lee AS:
Novel mechanism of anti-apoptotic function of 78-kDa
glucose-regulated protein (GRP78): endocrine resistance factor in
breast cancer, through release of B-cell lymphoma 2 (BCL-2) from
BCL-2-interacting killer (BIK). J Biol Chem. 286:25687–25696. 2011.
View Article : Google Scholar
|
|
21
|
McCullough KD, Martindale JL, Klotz LO, Aw
TY and Holbrook NJ: Gadd153 sensitizes cells to endoplasmic
reticulum stress by down-regulating Bcl2 and perturbing the
cellular redox state. Mol Cell Biol. 21:1249–1259. 2001. View Article : Google Scholar
|
|
22
|
Dong D, Ni M, Li J, et al: Critical role
of the stress chaperone GRP78/BiP in tumor proliferation, survival,
and tumor angiogenesis in transgene-induced mammary tumor
development. Cancer Res. 68:498–505. 2008. View Article : Google Scholar : PubMed/NCBI
|