Introduction
Pleomorphic hyalinizing angiectatic tumors (PHATs)
are rare neoplasms of the soft tissue that were first described by
Smith et al in 1996 in a series of 14 cases (1). The tumor usually occurs within the
subcutaneous tissue, particularly in the lower limbs of adults, and
is histologically characterized by clusters of thin-walled ectatic
vessels surrounded by hyalinized, fibrin and collagen material
(2). Folpe and Weiss (3) described ‘early PHAT’ as an early stage
or precursor lesion of classic PHAT that appears essentially
identical to a hemosiderotic fibrolipomatous lesion (HFLL). The
current study presents a case of PHAT arising in the thigh in which
the adipose tissue surrounding the tumor mass resembled an HFLL.
The patient was informed that data from the case would be submitted
for publication, and written consent was subsequently obtained.
Case report
A 68-year-old female presented with a solitary
asymptomatic tumor of the left medial thigh that had been rapidly
growing for ~2 months. There was no history of trauma and a
physical examination revealed an elastic soft tumor measuring ~12×7
cm in size.
Radiography showed an expansion of the soft tissue
on the medial aspect of the thigh, but no bony changes or
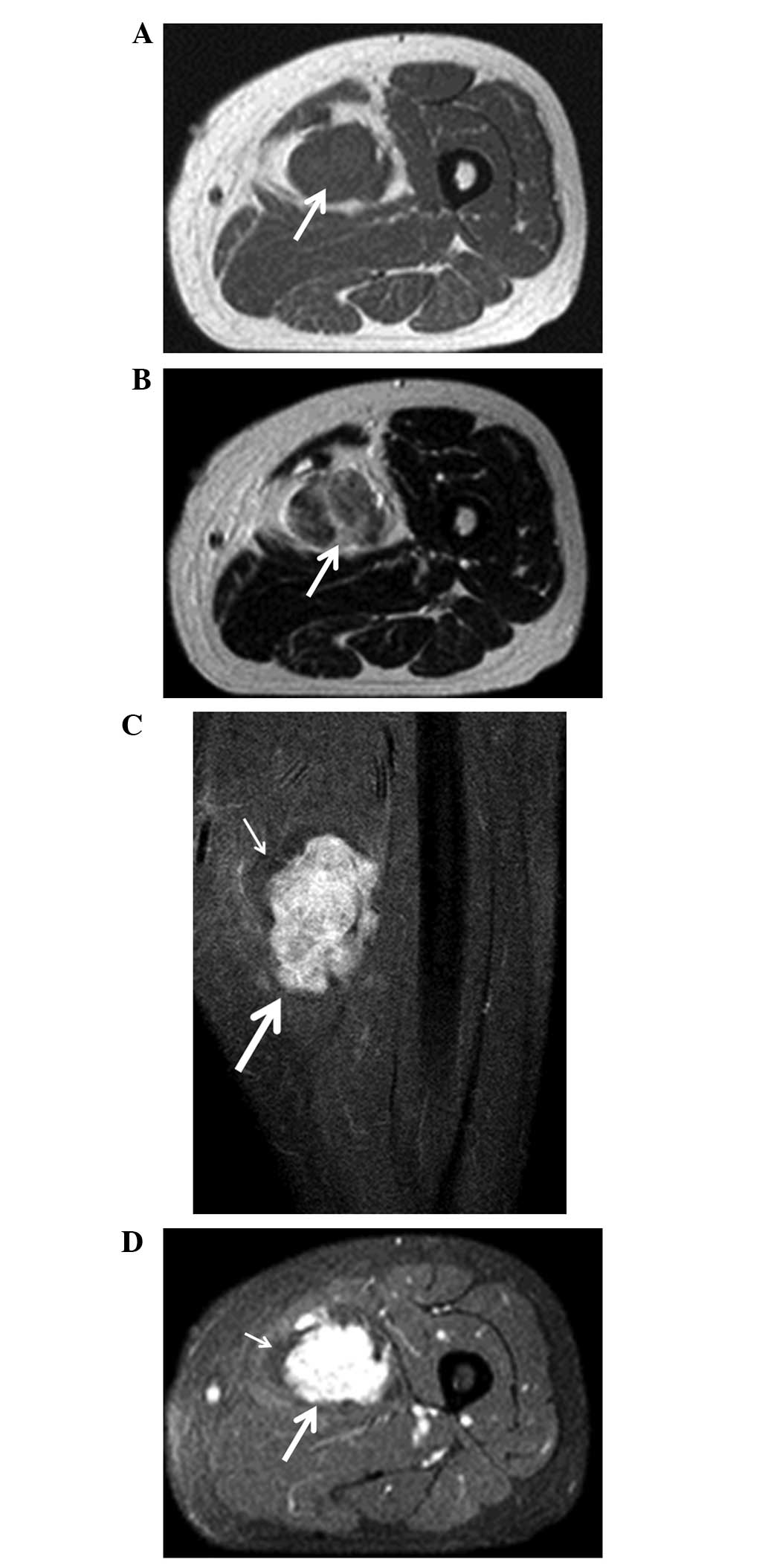
calcifications. Magnetic resonance imaging (MRI) revealed that the
tumor mass was isointense compared with the muscle on T1-weighted
images (Fig. 1A). On T2-weighted
images, the tumor was of heterogeneous high signal intensity
(Fig. 1B). The tumor enhanced
homogeneously following intravenous administration of gadolinium
with gadolinium diethylenetriaminepentacetate (Gd-DTPA). The
surrounding tissue was of high intensity on T1 and T2-weighted
images, but was not enhanced following administration of Gd-DTPA
(Fig. 1C and D). An angiogram
showed that the tumor was fed by a femoral artery and was markedly
stained. 67Gallium scintigraphy showed significantly
abnormal uptake in the tumor. Furthermore, chest radiography and
computed tomography found no evidence of lung metastasis, and the
laboratory findings were normal.
Based on a presumed diagnosis of a benign fibrous
tumor or schwannoma, tumor excision was performed. The surgical
findings concluded that the tumor was located in the femoral
abductor muscle, with the femoral artery penetrating into the
tumor. As a benign tumor was suspected according to the
intraoperative frozen section examination, the majority of the mass
was excised and the remaining portion was left attached to the
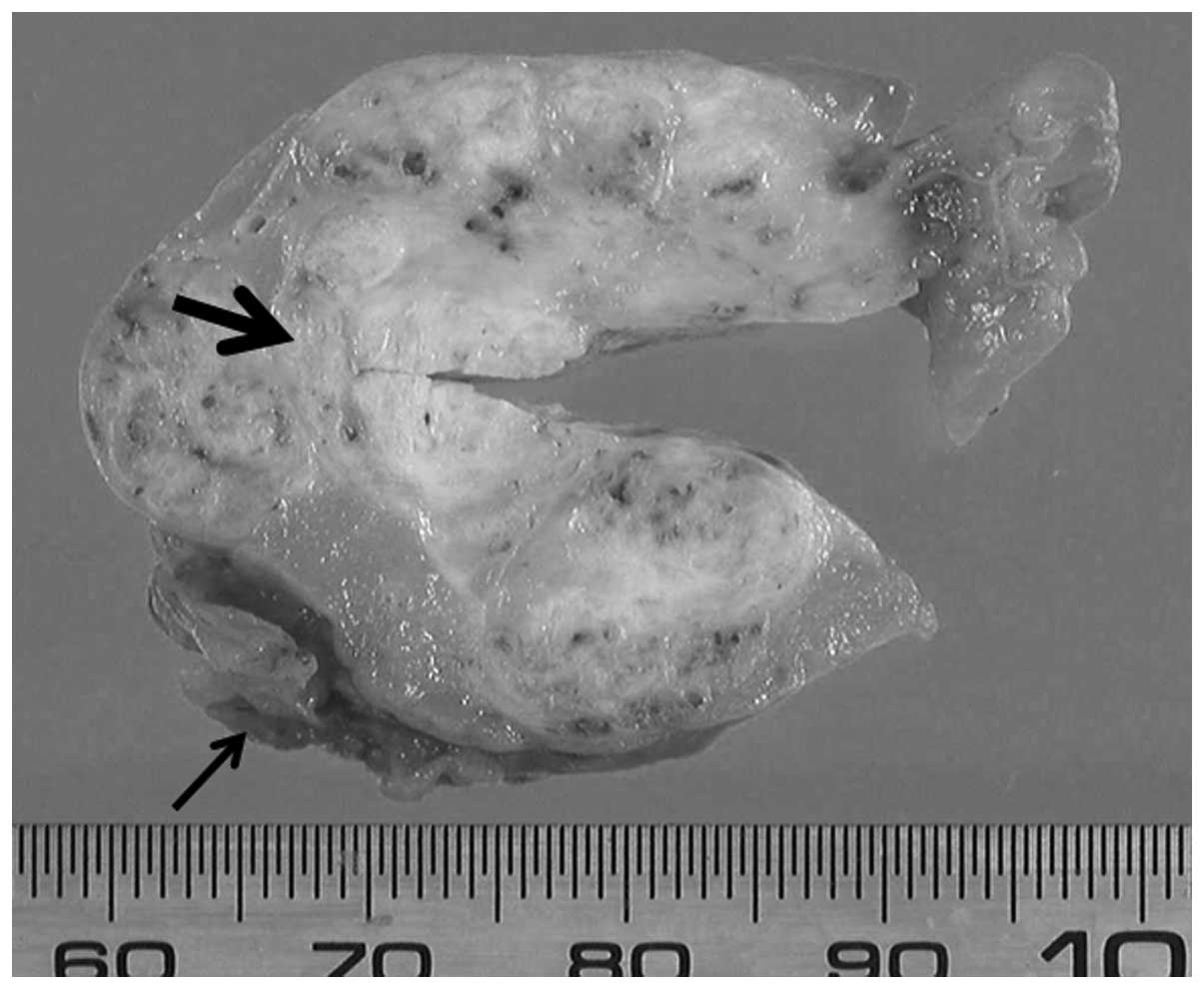
artery. The gross appearance revealed two layers of structures: An
internal lesion with a hard, white-tan colored tumor and an outer
lesion composed of lipomatous tumor tissue (Fig. 2). A wide resection was performed for
the internal lesion, whereas an intralesional resection was
performed for the outer lesion. The tumor measured 10.5×4.5×3.5 cm
and was lobular in appearance.
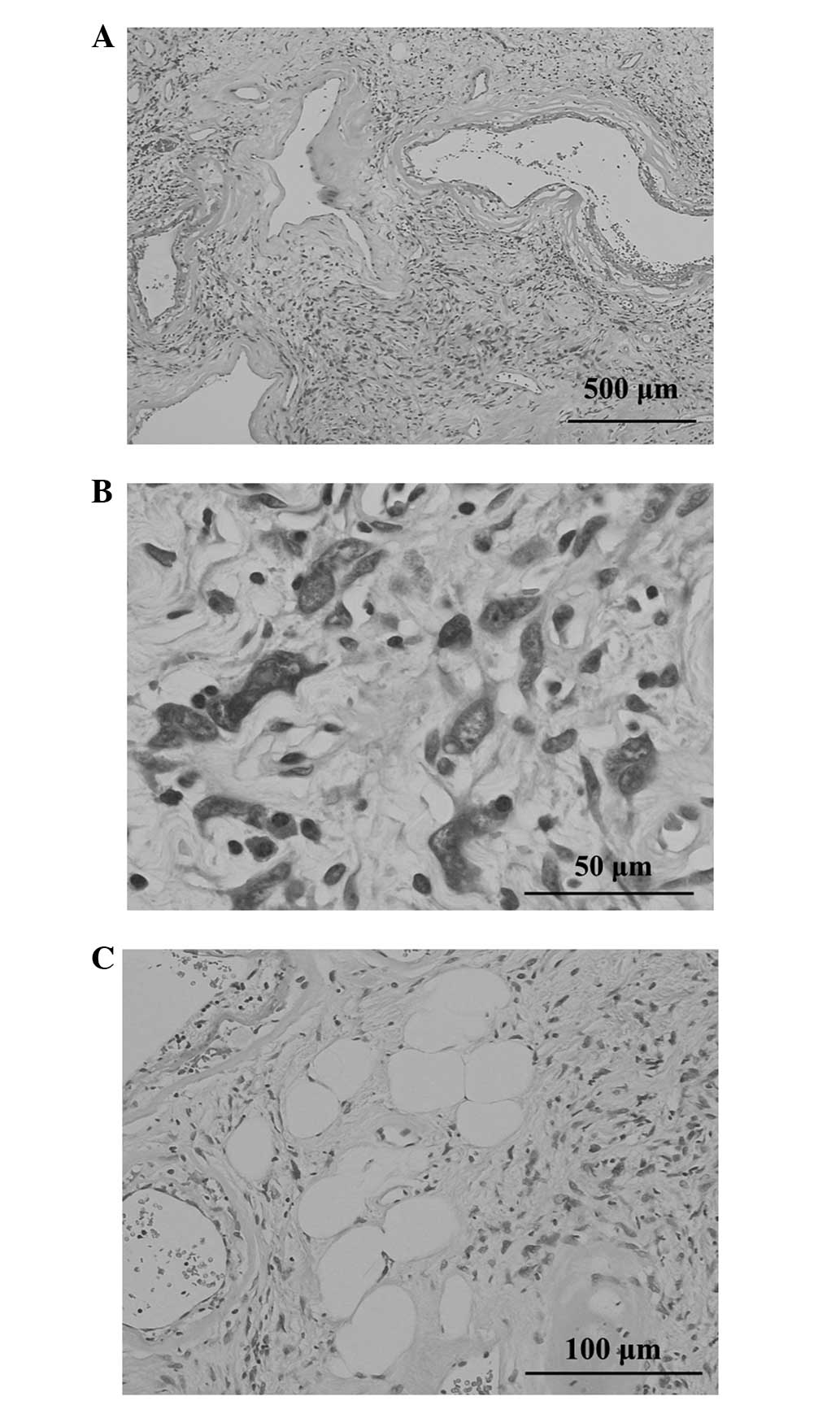
Histopathologically, the stroma revealed loose
fascicles that were arranged in whorls or a haphazard, patternless
fashion. This lesion contained a number of enlarged, thin-walled
blood vessels with rims of a fibrinous or hyalinized material and
organized thrombi and foci of hemosiderin deposition (Fig. 3A). The partially well-demarcated and
lobular lesion was composed of a proliferation of spindle cells
with hyperchromatic nuclei and nucleoplasmic bodies, as well as an
eosinophilic or palely-stained cytoplasm occasionally admixed with
pleomorphic cells (Fig. 3B). Mature
adipocytes were present within the proliferation of spindle and
pleomorphic cells (Fig. 3C).
Immunohistochemistry revealed a number of tumor
cells were positive for vimentin, cluster of differentiation
(CD)34, CD99 and B-cell lymphoma 2, whereas staining for S-100
protein, α-smooth muscle actin, desmin, AE1/AE3 and epithelial
membrane antigen was negative. Mitotic figures were rare and the
labeling index of Ki-67 was <3%. Based on these clinical and
histological findings, a PHAT was diagnosed.
No recurrence or metastases were identified during
the 6-year follow-up period.
Discussion
PHATs are non-metastasizing soft-tissue tumors of
uncertain lineage occurring within the superficial subcutaneous
tissues and muscles (3,4). Presents in adults between the ages of
10 and 83 years (median age of 51 years), PHATs are more commonly
observed in females than in males (4). The majority of affected patients
present with slowly-growing, painless masses, most commonly
involving the lower extremities. Rarer tumor sites include the arm,
chest wall, axilla, popliteal fossa, buttocks, inguinal region,
perineum, buccal mucosa and breast (2,4).
Due to its rarity, PHATs may be misdiagnosed as
other soft-tissue lesions. Malignant fibrous histiocytoma and
schwannoma should be considered within the differential diagnosis,
as cellular pleomorphism and ectatic, hyalinized blood vessels with
infiltration by variable chronic inflammatory cells is present in
those types of tumors (5–7). The characteristics of a schwannoma,
such as palisading of nuclei and the formation of Verocay bodies,
are absent in a PHAT. Immunohistochemically, a PHAT is strongly
positive for CD34, vascular endothelial growth factor and CD99, and
negative for S-100 protein (1,7). In
the present case, all findings were compatible with a diagnosis of
a PHAT.
Currently, PHATs are a benign condition according to
the World Heath Organization classification (2) and there have been no published studies
of metastasis associated with PHAT to date. However, the local
recurrence rate is 33–50% (1,3).
Generally recurrences are not destructive in their growth (2); however, one previous study has
described an aggressive recurrence that ultimately necessitated
amputation (3). In rare cases, the
tumors have recurred with the appearance of a sarcoma (3,8,9). These
findings indicated that PHATs may be low-grade sarcomas.
A previous study reported an association between
‘early PHAT’ and HFLL (3). An HFLL
is a reactive lesion that typically occurs in the foot or ankle
region of middle-aged patients, and consists of an admixture of fat
and moderately cellular fascicles of spindle cells. The lesion also
shows vascular hyalinization and scattered pleomorphic cells
(10). Folpe and Weiss (3) found a remarkable resemblance between
‘early PHAT’ and HFLL and suggested that an HFLL is a tumor related
to a PHAT rather than a reactive lesion. In the present case, the
outer lesion of the tumor may be an HFLL. As the femoral artery
penetrated into the HFLL, a wide excision of the tumor, including
the potential HFLL, was not possible. Although this portion of the
lesion was left in place, the patient did not experience recurrence
of the PHAT over the six-year follow-up period.
The published case studies of PHATs have primarily
discussed the pathological findings of the disease. To the best of
our knowledge, only one study has described the imaging
characteristics of this tumor on MRI (11). In the present report, the
association between the gross appearance and the imaging
characteristics of the PHAT were analyzed. With regard to the gross
appearance, the inside of the tumor was white-tan in color, hard
and included lipomatous tissue, while the outside was made up of a
lipomatous tumor. On MRI, the tumor was essentially isointense to
neighboring muscles on the T1-weighted images. On the T2-weighted
images, the signal intensity of the tumor was heterogeneously
hyperintense. On the Gd-DTPA-enhanced images, the tumor mass was
enhanced homogeneously. The existence of a large quantity of
lipomatous tissues in the muscle indicated the possibility of an
HFLL, and HFLLs are not enhanced following the intravenous
administration of Gd-DTPA. Due to the histological overlap and the
presence of areas that resemble HFLL and PHAT within the same
tumor, certain studies have argued that HFLL is actually a
precursor lesion to classic PHAT. Accordingly, the term ‘early
PHAT’ has been used to distinguish it from classic PHAT, which
exhibits more fully developed histological characteristics
(3), although whether it should be
considered an entity distinct from PHAT remains controversial
(12,13).
Due to the considerable potential for local
recurrence, surgical excision with a tumor-free margin is the
preferred treatment for PHAT (4).
Low-dose radiotherapy may be beneficial in cases of incomplete
resection to avoid recurrence. Currently, there are no published
reports of metastasis associated with a PHAT (1). The current study presented the case of
a PHAT that was surrounded by an HFLL. PHATs may be low-grade
malignant tumors; however, HFLLs are clearly benign lesions
according to their histopathological characteristics. In the
present study, removal was by a wide resection for the PHAT and by
intralesional resection for the HFLL. The patient did not
experience recurrence over the six-year follow-up period. This
clinical course may result in a determination of the marginal range
for this tumor. A wide resection is indicated for PHATs, but an
intralesional resection may be used for HFLLs when they are in
contact with important structures, such as an artery or nerve.
Further studies are required to delineate the clinical course and
long-term outcomes associated with this condition.
Acknowledgements
This study was supported, in part, by the
Grants-in-Aid for Scientific Research (C) 24592227 (KAKENHI).
References
|
1
|
Smith ME, Fisher C and Weiss SW:
Pleomorphic hyalinizing angiectatic tumor of soft parts. A
low-grade neoplasm resembling neurilemoma. Am J Surg Pathol.
20:21–29. 1996. View Article : Google Scholar
|
|
2
|
Weiss SW and Dei Tos AP: Pleomorphic
hyalinizing angiectatic tumour of soft parts. WHO Classification of
Tumours of Soft Tissue and Bone. Fletcher CDM, Bridge JA,
Hogendoorn PCW and Mertens F: IARC Press; Lyon: 2003
|
|
3
|
Folpe AL and Weiss SW: Pleomorphic
hyalinizing angiectatic tumor: analysis of 41 cases supporting
evolution from a distinctive precursor lesion. Am J Surg Pathol.
28:1417–1425. 2004. View Article : Google Scholar
|
|
4
|
Lee JC, Jiang XY, Karpinski RH and Moore
ED: Pleomorphic hyalinizing angiectatic tumor of soft parts.
Surgery. 137:119–121. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
El-Tal AE and Mehregan D: Pleomorphic
hyalinizing angiectatic tumor of soft parts: case report and
literature review. J Cutan Pathol. 33:361–364. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ke Q, Erbolat, Zhang HY, et al:
Clinicopathologic features of pleomorphic hyalinizing angiectatic
tumor of soft parts. Chin Med J (Engl). 120:876–881.
2007.PubMed/NCBI
|
|
7
|
Tardío JC: CD34-reactive tumors of the
skin. An updated review of an ever-growing list of lesions. J Cutan
Pathol. 36:1079–1092. 2008.
|
|
8
|
Kazakov DV, Pavlovsky M, Mukensnabl P and
Michal M: Pleomorphic hyalinizing angiectatic tumor with a
sarcomatous component recurring as high-grade myxofibrosarcoma.
Pathol Int. 57:281–284. 2007. View Article : Google Scholar
|
|
9
|
Mitsuhashi T, Barr RJ, Machtinger LA,
Machtinger LA and Cassarino DS: Primary cutaneous myxofibrosarcoma
mimicking pleomorphic hyalinizing angiectatic tumor (PHAT): a
potential diagnostic pitfall. Am J Dermatopathol. 27:322–326. 2005.
View Article : Google Scholar
|
|
10
|
Marshall-Taylor C and Fanburg-Smith JC:
Hemosiderotic fibrohistiocytic lipomatous lesion: ten cases of a
previously undescribed fatty lesion of the foot/ankle. Mod Pathol.
13:1192–1199. 2000. View Article : Google Scholar
|
|
11
|
Subhawong TK, Subhawong AP, Montgomery EA
and Fayad LM: Pleomorphic hyalinizing angiectatic tumor: imaging
findings. Skeletal Radiol. 41:1621–1626. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Browne TJ and Fletcher CDM: Haemosiderotic
fibrolipomatous tumour (so-called haemosiderotic fibrohistiocytic
lipomatous tumour): analysis of 13 new cases in support of a
distinct entity. Histopathology. 48:453–461. 2006. View Article : Google Scholar
|
|
13
|
Moretti VM, de la Cruz M, Brooks JS and
Lackman RD: Early pleomorphic hyalinizing angiectatic tumor:
precursor or distinct lesion? Orthopedics. 33:5162010.
|