Introduction
Cervical cancer is the second leading cause of
cancer-associated mortality in women worldwide, responsible for
>275,100 deaths each year, with the mortality rate on the rise
in a number of developing countries (1). Cervical squamous cell carcinoma
(cervical SCC) is one of the most frequent types of cervical
cancer, accounting for 80–90% of all cases. A large number of
studies have confirmed that persistent infection of high-risk human
papillomavirus (HR-HPV) is a critical and indispensible risk factor
for cervical SCC and the development of precancerous lesions
(2). The high-risk HPVs contain two
important oncogenes, E6 and E7, which contribute to oncogenesis of
cervical SCC by silencing the tumor-suppressive p53 and Rb
proteins, eventually resulting in cell cycle disorder and malignant
transformation (3–5). Although the underlying pathogenesis of
cervical SCC has been identified, the molecular mechanisms in its
progression have not yet been fully elucidated.
Long non-coding RNAs (lncRNAs) are non-coding
transcripts that are >200 nucleotides in length that have
recently emerged as important molecules in both normal development
and tumorigenesis (6,7). Studies have demonstrated that these
lncRNAs have an important role in numerous biological processes,
including X-chromosome inactivation, genomic imprinting, chromatin
modification, gene transcription and splicing (8–11). A
large group of lncRNAs have exhibited deregulated expression in
human cancer types and appeared to have specific functional roles
in tumor progression. HOTAIR is a 2.2-kb lncRNA, and its expression
level was identified to be associated with breast cancer metastases
and mortality (12). Panzitt et
al identified that HULC contributed to early hepatocellular
tumorigenesis and may act as a promising biomarker in cancer
diagnostics (13). Furthermore,
lincRNA-p21 and PANDA were reported to regulate cell apoptosis in
carcinomas through a p53 gene regulatory pathway (14,15).
Metastasis-associated lung adenocarcinoma transcript
1 (MALAT1), also known as nuclear-enriched abundant transcript 2,
is a highly evolutionary conserved lncRNA with a full length of
8708 nt (16). MALAT1 is a highly
abundant nucleus-retained RNA that localizes to nuclear speckles, a
sub-nuclear domain enriched in pre-mRNA splicing factors and
affects alternative splicing of pre-mRNAs through modulating the
cellular distribution and activity of serine arginine
dipeptide-containing SR splicing factors (17–19).
Several recent studies have demonstrated that lncRNA MALAT1 is
upregulated in several solid tumor types and contributes to tumor
cell proliferation, apoptosis, migration and invasion. The first
study that linked MALAT1 to cancer was in non-small cell lung
cancer (NSCLC) patients in 2003. The authors identified that MALAT1
was a prognostic parameter for the survival of stage I lung
carcinoma patients, and that its expression was higher in NSCLC
with metastasis than that without (16). Later studies in hepatocellular
carcinoma (HCC) revealed that MALAT1 was upregulated compared with
normal liver tissue, and that the depletion of MALAT1 in HepG2
cells reduced cell viability, motility and invasiveness. Clinical
analysis further proved that MALAT1 was an independent prognostic
factor for HCC recurrence following liver transplantation (20,21). A
large number of studies have certified the specific functions of
MALAT1 in other solid tumors, but the molecular mechanism
underlying its effects remains unclear (22–26).
Materials and methods
Cervical cancer cell lines and
specimens
Human cervical cancer cell lines, HeLa
(HPV18-positive), CaSki (HPV16-positive), SiHa (HPV16-positive) and
HCC94 (HPV16-positive), as well as immortal human keratinocyte
HaCaT cells, were grown in RPMI-1640 medium (Gibco-BRL, Carlsbad,
CA, USA) supplemented with 10% fetal bovine serum (HyClone, Logan,
UT, USA). All cells were cultured in a humidified atmosphere
containing 5% CO2 at 37°C. A total of 40 cases of
HPV-positive normal and abnormal cervical squamous epithelium
specimens and 24 cases of HPV-negative normal cervical squamous
epithelium were collected from patients who had undergone cervical
Thinprep cytological test and high risk human papillomavirus
(HR-HPV) detection at Xiangya Hospital of Central South University
(Changsha, China). The samples were processed and stored in
RNAlater RNA stabilization reagent (Qiagen, Hilden, Germany) at
−20°C until RNA extraction. This study was approved by the ethics
committee of the Cancer Research Institute, Central South
University, (Changsha, China). Written informed consent and
approval were obtained from each patient.
RNA isolation from cells and
specimens
Isolation of cell total RNA was performed using
TRIzol reagent (Invitrogen Life Technologies, Carlsbad, CA, USA)
according to manufacturer’s instructions. The total RNA samples
were isolated using Total RNA kit I (Omega Bio-Tek, Inc., Norcross,
GA, USA) according to manufacturer’s instructions. All of the RNA
samples were examined for integrity and purity by an ultraviolet
spectrophotometer (OD260/OD280) (SmartSpec 3000 UV Vis
Spectrophotometer, Bio-Rad, Hercules, CA, USA).
Reverse transcription-polymerase chain
reaction (RT-PCR) analysis
A total of 1 μg RNA was absorbed for semi-quantified
RT using an RevertAid First Strand cDNA Synthesis kit (Thermo
Fisher Scientific, Inc., Pittsburgh, PA, USA) according to the
manufacturer’s instructions. All primers for PCR were designed by
Primer 5.0 (Premier, Palo Alto, CA, USA) and detected in NCBI
Blast. Primers for MALAT1 detection: forward, AGCGGAAGAACGAATGTAAC
and reverse, GAACAGAAGGAAGAGCCAAG; primers for CDK4: forward,
GGAGTGTTGGCTGTATCTTTGC and reverse, CGGATTACCTTCATCCTTATGT; primers
for CDK6: forward, TGGTCGTCACGCTGTGGTACAG and reverse,
GCAGGTGGGAATCCAGGTTTTC; primers for cyclinD1: forward,
GCATCTACACCGACAACTCC and reverse, CTCCTCCTCCTCTTCCTCCT; primers for
cyclinE: forward, GCTTATTGGGATTTCATCTTTA and reverse,
TCTGTGGGTCTGTATGTTGTGT; primers for HPV16 E6: forward,
CCACCCAGAAAGTTACCACA and reverse, TGCAACAAGACATACATCGA; and primers
for HPV16 E7: forward, TGGAGATACACCTACATTGCAT and reverse,
CCATTAACAGGTCTTCCAACGT. The PCR conditions were one cycle of 94°C
for 5 min, followed by 27 cycles of 94°C for 30 sec, 58°C for 30
sec and 72°C for 60 sec with an extension of 68°C for 10 min. The
PCR products were visualized on 1% agarose gels stained with
ethidium bromide and quantified by ImageJ software (NIH, Bethseda,
MD, USA).
Construction of expression vectors of
MALAT1 and transfection
Total RNA was extracted from HeLa cells and the
amplification of two fragments in MALAT1 (NR_002819.2) was
performed using primer M1 (4481–5481): forward,
GTTGTTTGGATATGGTAGTGTGTGG and reverse, ATA AGCACTTATCCCTAACATGCAA
[introduced with XhoI site (forward) and an BamHI site (reverse),
respectively]; and primer M2 (6419–7260): forward,
GAGTGCTTGGCTCTTCCTTCTG and reverse, ACCTGTTTTCCTCATTTTGTCC
[introduced with XhoI site (forward) and an Bsp120I site (reverse),
respectively]. The PCR products of the MALAT1 gene were purified by
a Gel Extraction kit (Omega Bio-Tek, Inc.) following 1% agarose gel
electrophoresis, then double digested and ligated into the
eukaryotic expression vector pEGFP-C1. The recombinant plasmids,
pEGFP-C1/M1 and pEGFP-C1/M2, were transformed into competent
Escherichia coli DH5α and then positive clones were
identified by PCR, double digestion and DNA sequencing. For
transfection, the recombinant plasmids were transfected into cells
using Lipofectamine 2000 transfection reagent (Invitrogen Life
Technologies) according to manufacturer’s instructions. Then, for
transient transfection, the cells were harvested following 48 h
and, for stable transfection, G418 was added as a screening reagent
and a limiting dilution method was used to select the monoclones.
The positive clone was detected by RT-PCR.
Cell proliferation assay
Cell proliferation assay was determined using an
Cell Counting kit-8 (CCK-8; Beyotime Institute of Biotechnology,
Haimen, China) based on WST-8
[2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium].
Cells were seeded in 96-well plates at a density of 103
cells/well with serum-free medium in a total volume of 100 μl, and
then washed with phosphate-buffered saline (PBS). The medium was
replaced with DMEM (Gibco-BRL) supplemented with 1% fetal bovine
serum (FBS; Hyclone) the next day. Then, WST-8 (10 μl) was added to
each well and incubated for 2 h at 37°C, every 24 h for 7 days. The
optical density (OD) was measured at 450 nm in an SM-3 automatic
enzyme-linked immune analyzer (TianShi, Beijing, China).
Cell cycle analysis
Cells were trypsinized, washed twice in PBS, counted
and then collected following fixation in 70% ethanol overnight at
4°C. A total of 1×106 cells were suspended and stained
with 500 μl propidium iodide solution (Beyotime Institute of
Biotechnology) for 30 min in the dark at 37°C. FACScan flow
cytometry instrument (Becton-Dickinson, Franklin Lakes, NJ, USA)
was used to analyze cell cycles. The CellQuest program
(Becton-Dickinson) was used for data analysis.
Wound healing assay
Cells were cultured in 6-well plates. A wound was
scratched with a 200-μl pipette tip and captured when the monolayer
cells reached subconfluency. The cells were washed three times with
PBS and cultured in DMEM medium supplemented with 1% FBS and
captured at different time points (24 or 48 h). The relative
migration rate (%) of the cells was measured and quantified by the
distance of cell migration divided by the distance measured at 0
h.
Statistical analysis
Experimental data are presented as the mean ±
standard deviation for three or more individual experiments. All
statistical analyses were performed using a two-tailed Student’s
t-test or one-way analysis of variance (SPSS 17.0; SPSS, Inc.,
Chicago, IL, USA). P<0.05 was considered to indicate a
statistically significant difference. The diagrams were drawn by
GraphPad Prism 5 (GraphPad Software, San Diego, CA, USA).
Results
MALAT1 expression in human cervical
cancer cell lines
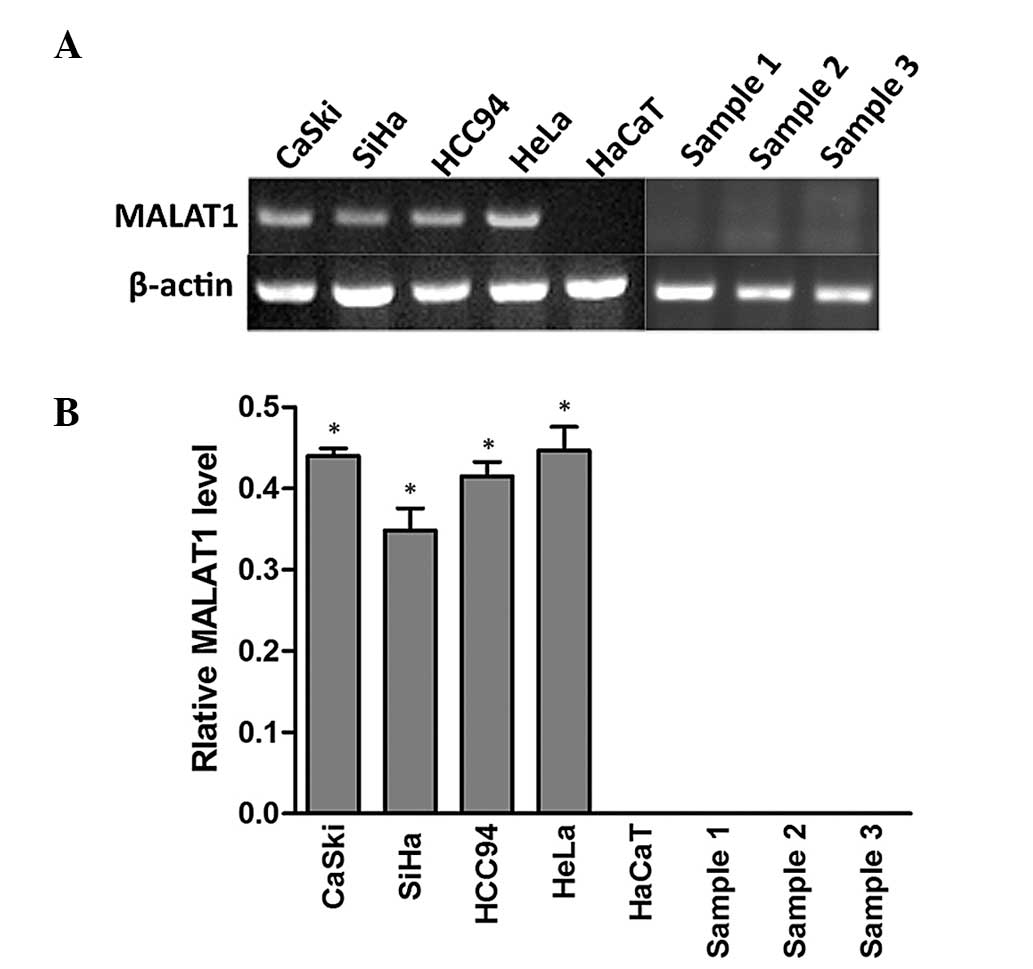
To examine MALAT1 expression levels in cervical
cancer, RT-PCR was used to detect the expression of MALAT1 in
cervical cancer cell lines (HeLa, CaSki, SiHa and HCC94), immortal
human keratinocyte HaCaT cells and in three cases of normal
cervical squamous cells. As demonstrated in Fig. 1, MALAT1 was expressed in all
cervical cancer cell lines but not in HaCaT cells and normal
samples. This suggested that MALAT1 was activated in cervical
cancer and may have an important role in tumorigenesis.
Construction of stable
MALAT1-overexpressing and -underexpressing cells
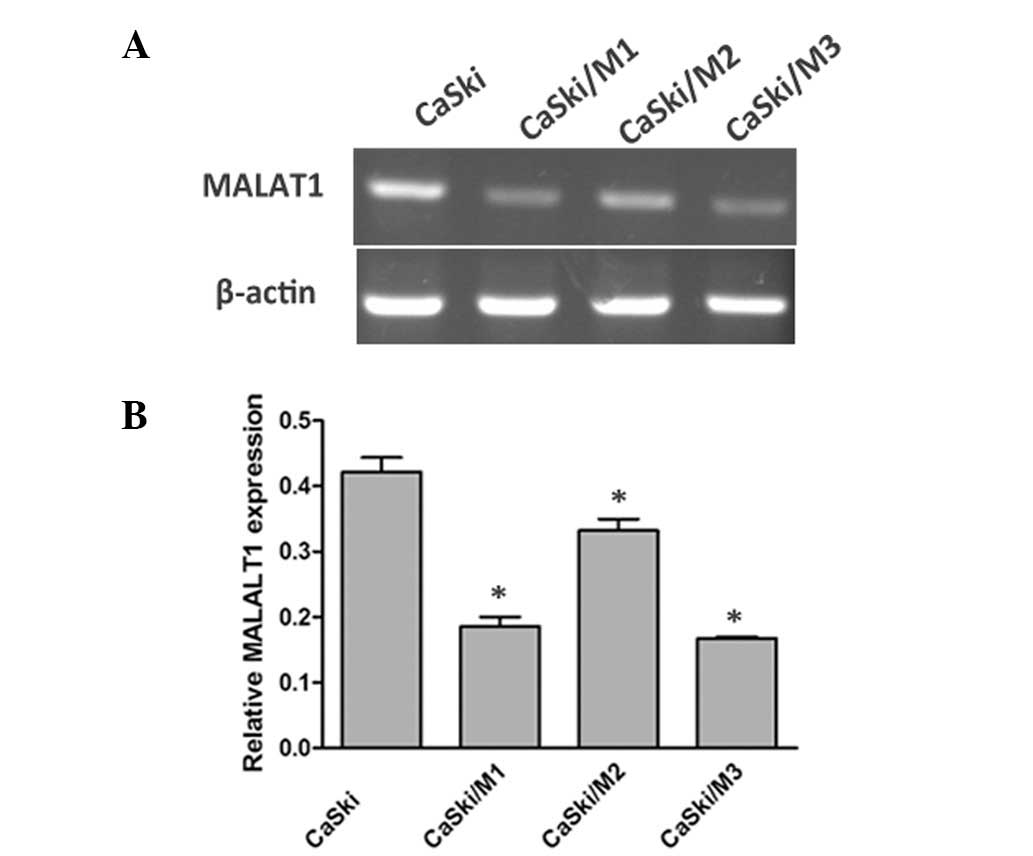
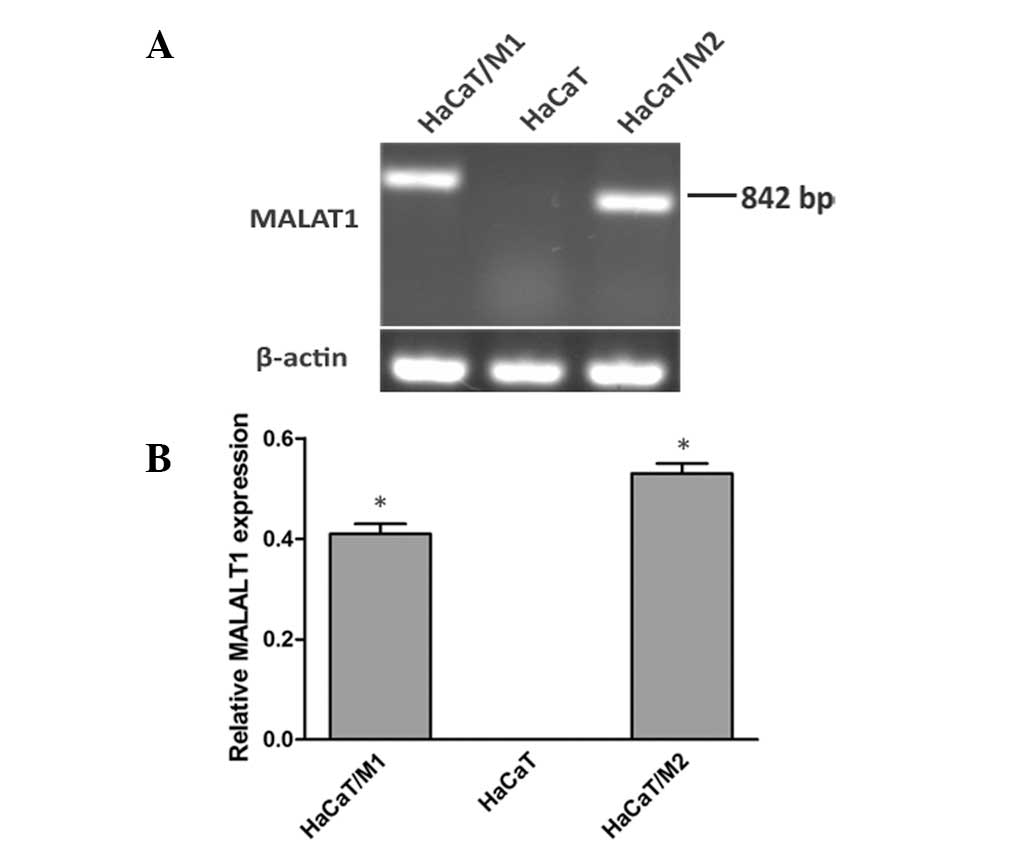
To investigate the functional role of MALAT1,
gain-of-function studies were conducted using HaCaT cells
transfected with two fragments of MALAT, respectively (HaCaT/M1,
HaCaT/M2) and loss-of-function in CaSki cells transfected with
MALAT1 shRNA expression vector pRNAT-U6.1/Neo encoding a small
hairpin RNA directed target sequences of MALAT1
5′-GACCTTGAAATCCATGACG-3′. The underexpression and overexpression
efficiency are demonstrated in Figs.
2 and 3, respectively, and
CaSki/M3 was selected as the MALAT1 downregulation group
(CaSki/si-M) for the following experiments.
MALAT1 affects cell migration and
proliferation capability
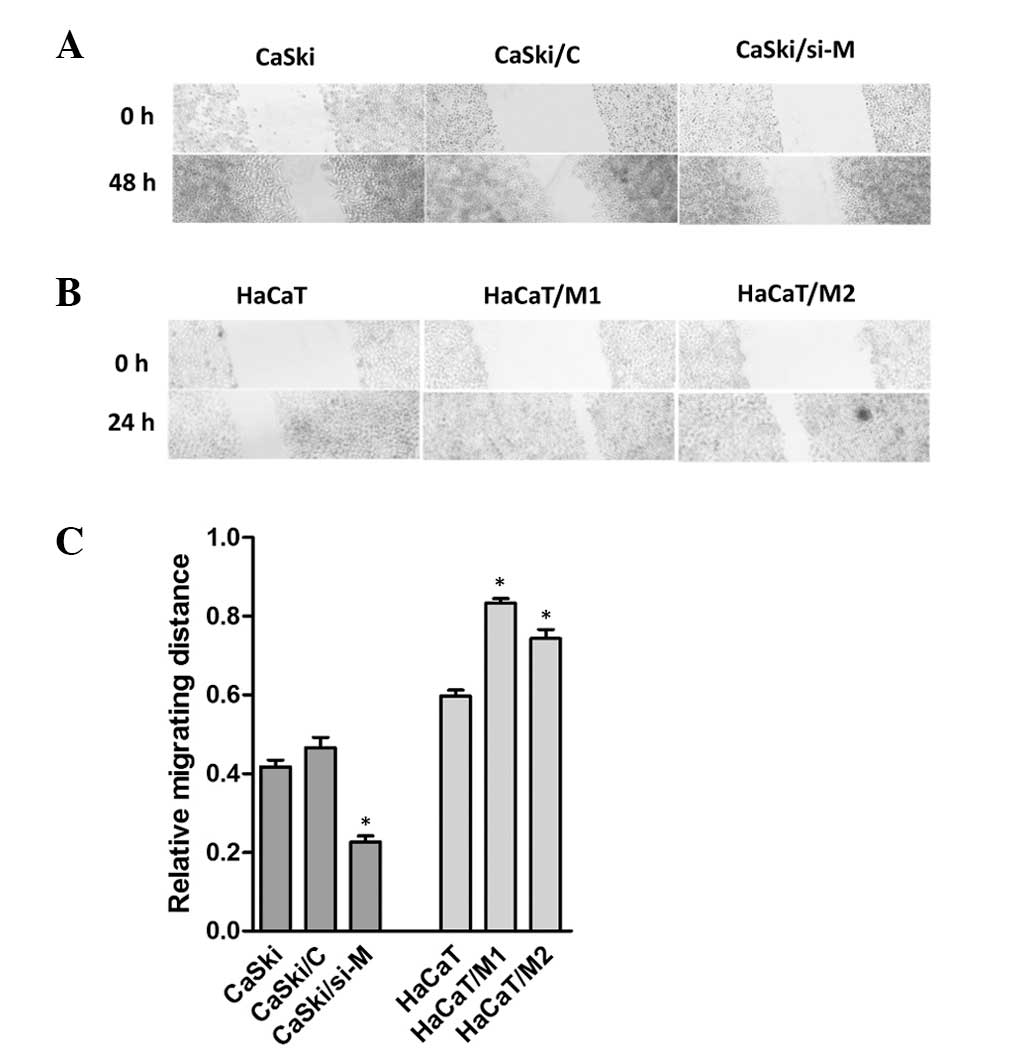
To investigate whether MALAT1 impacted cell
migration, a wound healing assay was performed. As demonstrated in
Fig. 4, the migratory speed of
CaSki/si-M was markedly slower than that of control cells,
following 48 h, and the migration distance was also shorter in
HaCaT cells than in the HaCaT/M1 or HaCaT/M2 cells following 24 h.
These data suggested that MALAT1 may promote cell migration.
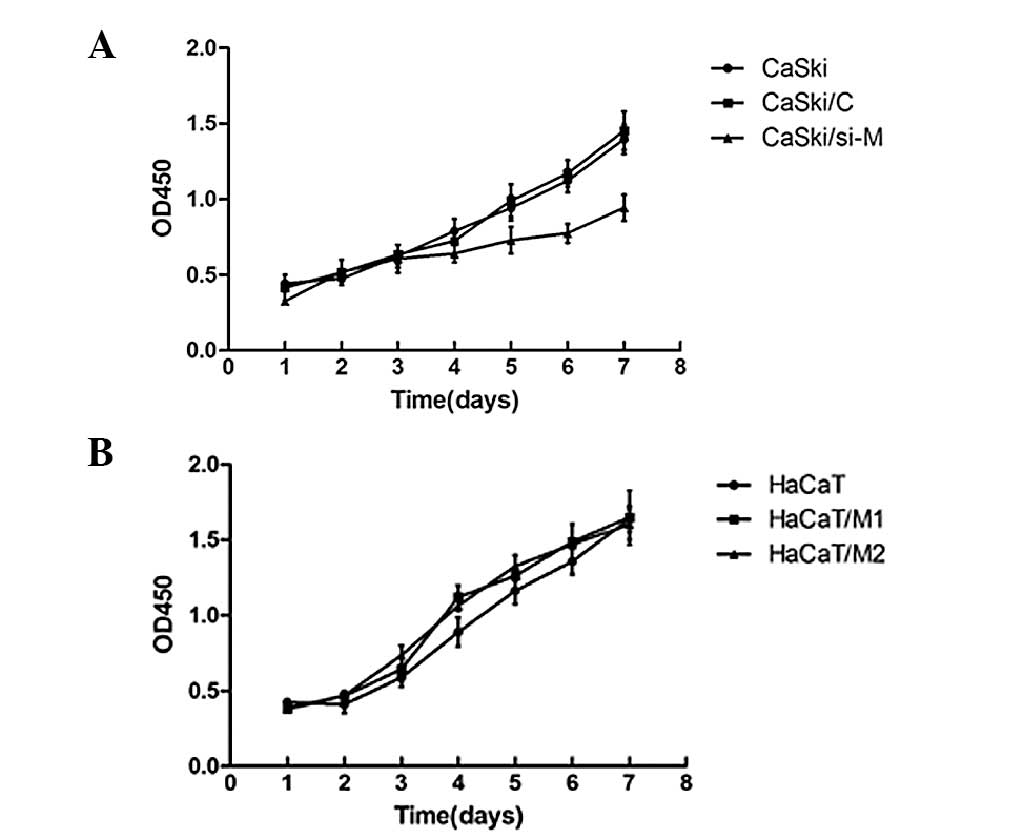
The CCK-8 assay revealed that cell proliferation was
significantly inhibited in CaSki/si-M in comparison with
non-transfectants (CaSki) and vector-control transfectants
(CaSki/C) in CaSki cells (Fig. 5A).
However, no significant difference was detected among the HaCaT/M1,
HaCaT/M2 and HaCaT cells (Fig. 5B).
These data indicate that downregulation of MALAT1 decreased cell
proliferation ability in CaSki cells.
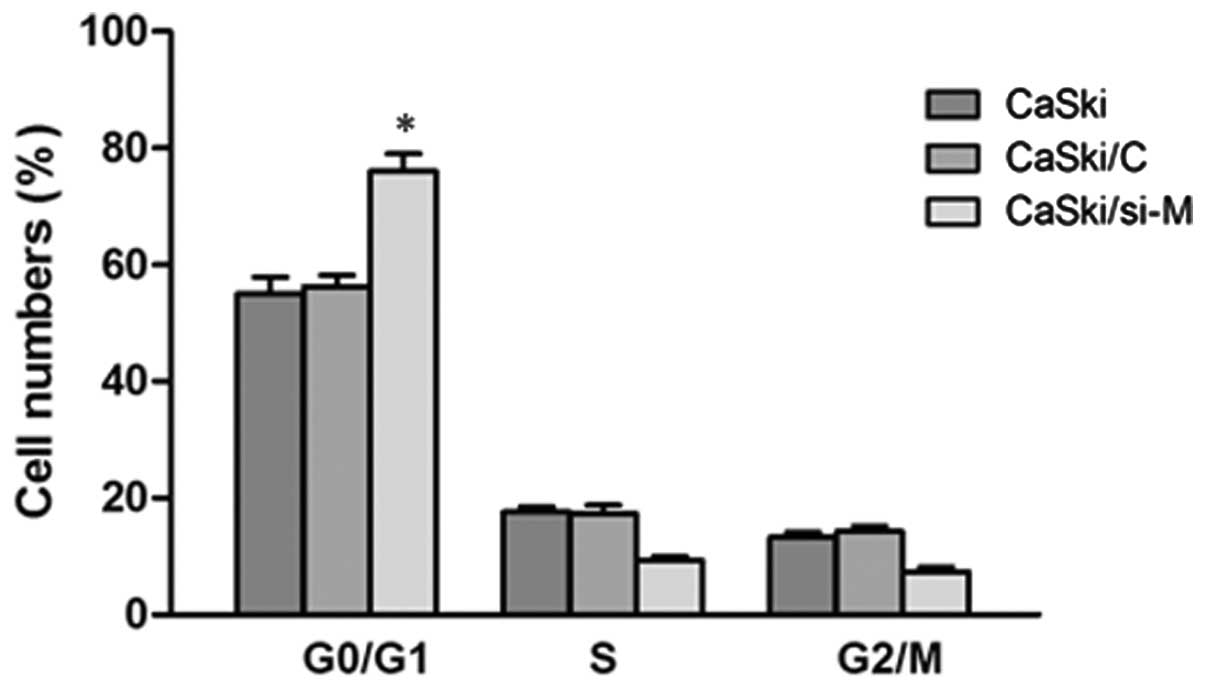
To further examine the cause for the decreasing of
cell viability, analysis of the cell cycle was conducted in CaSki
cells. As revealed in Fig. 6, there
was a significant decrease in S phase cells in CaSki/si-M cells
compared with CaSki/C or CaSki/si-M, indicating that downregulation
of MALAT1 inhibited the cell cycle at the G1/S transition.
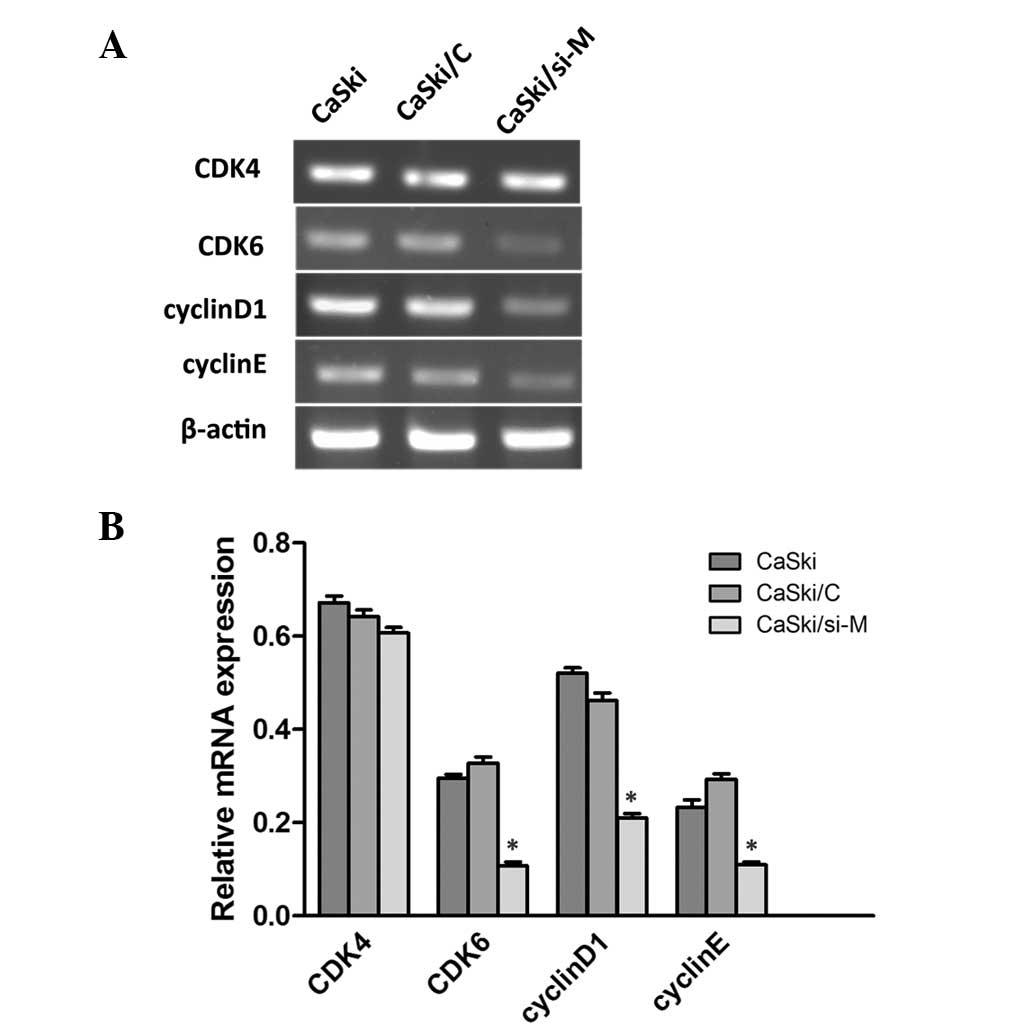
Following this, semi-quantitative RT-PCR was performed to
investigate whether cell cycle associated molecules were affected
by MALAT1. As revealed in Fig. 7,
cyclinD1, cyclinE and cyclin-dependent kinase 6 (CDK6) were
decreased in CaSki/si-M cells compared with the mock and control
groups, while CDK4 was indistinguishable. These findings indicate
that MLAT1 may increase cell proliferation by upregulating
cyclinD1, cyclinE and CDK6.
Knockdown of HPV16 E6/E7 reduces MALAT1
expression
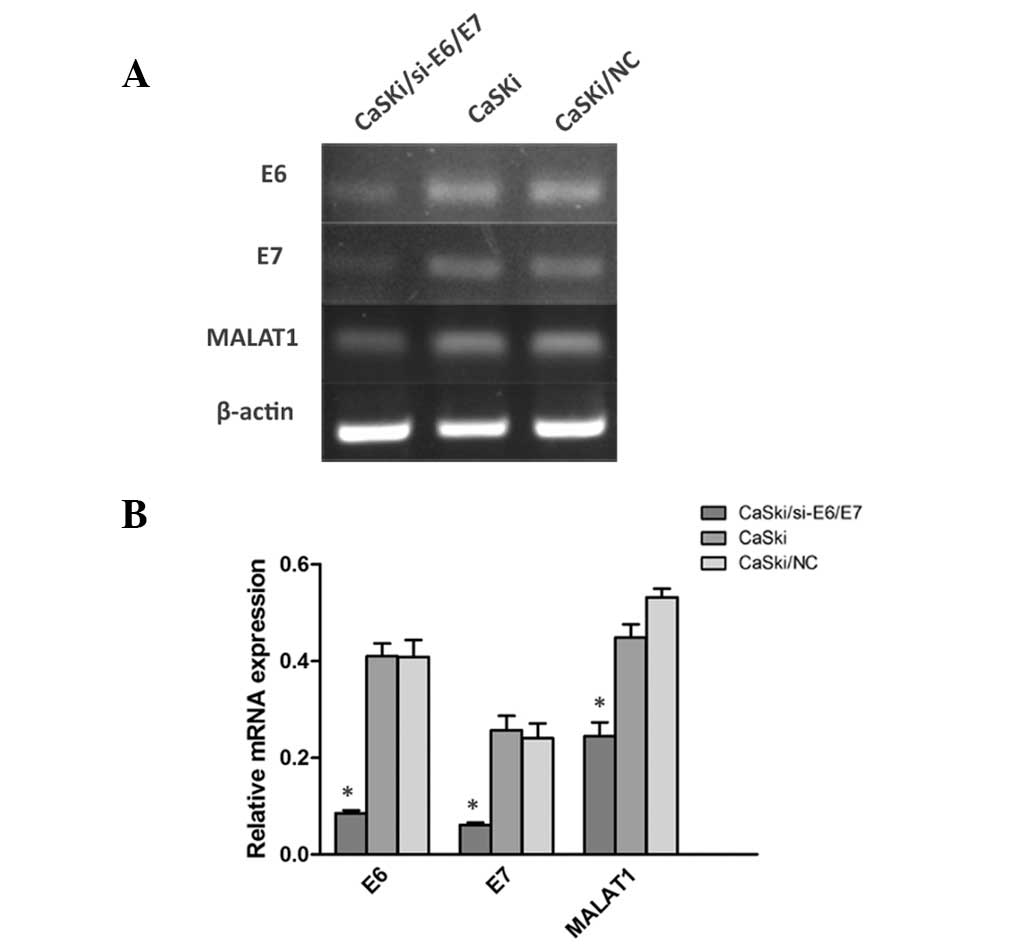
To identify the possible factors inducing MALAT1
deregulation in cervical cancer, HPV E6/E7 was incorporated into
the investigations due to its key role in cervical lesions. HPV16
E6/E7 shRNA vector GV102 targeting sequence GCAACAGTTACTGCGACGT
(GeneChem, Shanghai, China) were transfected into HPV16-positive
cell lines (CaSki cells). It was identified that the expression of
MALAT1 was reduced with the E6/E7 downregulation in CaSki cells
(Fig. 8). These results indicate
that the HPV16 E6/E7 gene is involved in the upregulation of MALAT1
in cervical cancer.
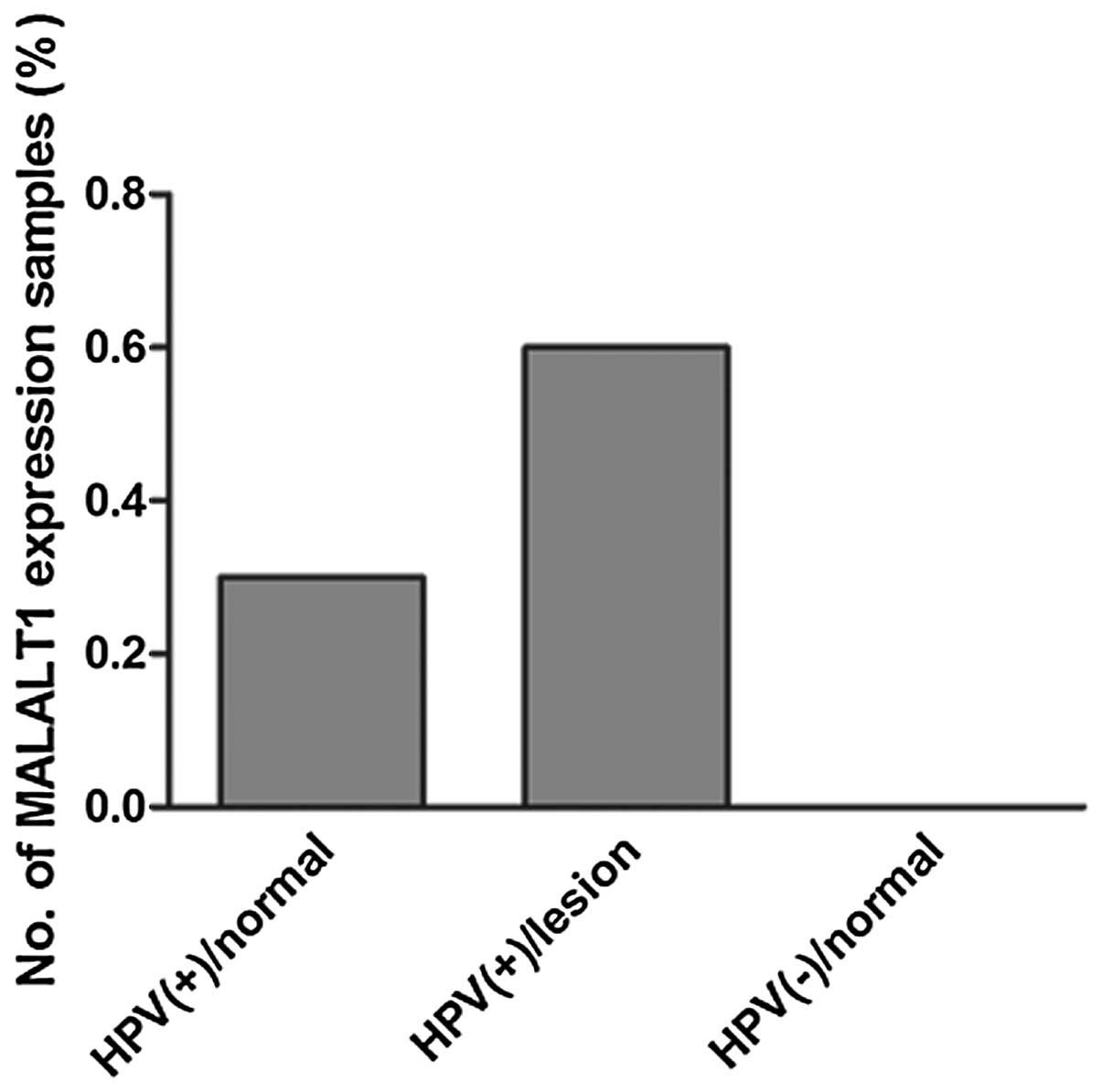
MALAT1 expression in clinical
HPV-positive normal cervical squamous cells and lesions
To further identify the correlation between MALAT1
and HPV, 64 cases of clinical cervical squamous cell samples were
collected and MALAT1 expression was identified in 6/18 cases in
HPV-positive cervical normal cells and 14/22 cases in HPV-positive
cervical lesion specimens; while in all HPV-negative normal
cervical squamous cells (n=24), MALAT1 expression was not detected
(Fig. 9). This suggested that HPV
is one of the important factors leading to MALAT1 activation in
cervical SCC.
Discussion
With the development of genomic microarrays and
whole genome and transcriptome sequencing technologies, it has been
revealed that ≥90% of the genome is actively transcribed, but
<2%of the total genome encodes for functional proteins (27,28).
These non-coding transcripts were previously argued to be spurious
transcriptional noise, but have now been identified to have an
increasingly important role in both normal development and disease,
particularly in cancer initiation through interaction with
protein-coding genes (29). MALAT1
is a lncRNA transcribed by RNA polymerase II (RNA pol II) and is
not polyadenylated. It may be spliced to a 61-nt tRNA-like
transcript and a longer transcript with a length of 6.7 kb. The
61-nt transcript may be exported to the cytoplasm the function of
which remains unclear. The longer transcript was retained in
nuclear speckles and were capable of regulating gene expresseion
through interaction with SR (30).
Several studies have reported that MALAT1 has a critical role in
cancer development, including in lung, liver, breast and cervical
cancer (16,21,23,31).
The function of MALAT1 in cervical cancer was initially studied in
our laboratory; however, the molecular mechanism in cell growth and
the factors inducing MALAT1 upregulation were unclear. Furthermore,
due to the established link between cervical cancer and HPV
infection, elucidating the correlation among MALAT1, HPV and
cervical cancer warranted further investigation.
In the present study, the expression of MALAT1 was
detected in cervical cancer cell lines (HeLa, CaSki, SiHa and
HCC94) and immortal human keratinocytes (HaCaT cells) using RT-PCR.
HaCaT cells are a type of normal epithelial cell, so were utilized
as a negative control. The results indicated that MALAT1 is
expressed in cervical cancer but not in normal epithelial cells,
and further study in normal cervical cell samples also demonstrated
this. To study the functional role of MALAT1, stable
MALAT1-underexpressing and -overexpressing cell lines were
constructed in CaSki and HaCaT cells, respectively. A series of
in vitro investigations indicated that the migration
capability was evidently altered in CaSki/si-M, HaCaT/M1 and
HaCaT/M2 compared with the control. The growth capability was
decreased in CaSki/si-M while no alteration was observed in
HaCaT/M1 and HaCaT/M2 compared with HaCaT. The explanation for this
phenomenon may be due to the fact that the function fragments of
MALAT1 in cell growth are different. A study in colorectal
carcinoma demonstrated that a fragment spanning nucleotides
6918–8441 provides growth and proliferative advantage (25). To find the molecules associated with
cell growth, the cell cycle of CaSki/si-M cells were analyzed and
it was identified that the cells were arrested in G1 phase
following MALAT1 downregulation. The cell cycle is controlled by
numerous mechanisms ensuring correct cell division. The present
study focused on the regulation of CDK by cyclins, and it was
revealed that G1/S transition regulation molecules cyclinD1,
cyclinE and CDK6 were decreased significantly in CaSki/si-M cells.
These results suggest that MALAT1 may regulate cell proliferation
through the P16INK4A/CDKs/RB pathway (32), while further studies are required to
further elucidate this.
Since HR-HPV infection was an indispensable factor
for cervical cancer and precancerous lesions, and numerous studies
have demonstrated that HPV may target a group of molecules,
including mTOR, miR-29 and MAGI-1 (33–35),
it was hypothesized that HPV may be a key factor inducing MALAT1
expression in cervical SCC. MALAT1 expression was detected in 64
cases of HPV-positive or -negative cervical squamous cells, and it
was identified that MALAT1 was expressed in ~30% of the
HPV-positive normal cells and ~60% of the HPV-positive cervical
lesion cells, but not in HPV-negative normal cells. Currently, the
two main methods for cervical cancer screening are HPV testing and
liquid-based cytology, both of which are associated with certain
limitations (36–38). Joint inspection is a tendency based
on the complete consideration of age, cost, positive rate and
future efficiency. Our further study in HPV16-positive cells using
shRNA targeting HPV16 E6/E7 proved that MALAT1 is a target of HPV.
Therefore, MALAT1 may be a potential screening and therapeutic
target for the treatment of cervical cancer. Further studies are
required to analyze the molecular mechanisms of MALAT1 in cervical
tumorigenesis, using more clinical samples to further analyze the
correlation among MALAT1, HPV and cervical cancer progression.
Acknowledgements
This study was supported by the Science and
Technology Department Research Foundation of Hunan province
(12JJ2052; China), the Open-End Fund for the Valuable and Precision
Instruments of Central South University (CSU2C2013048; Hunan China)
and the Fundamental Research Funds for the Central Universities of
Central South University (2013zzts283; Hunan, China).
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
2
|
Jablonowska D, Marszalek A and Bodnar M:
Tabacco smoking, HPV infection and changes in cervix. Przegl Lek.
69:740–743. 2012.(In Polish).
|
|
3
|
Scheffner M, Huibregtse JM, Vierstra RD
and Howley PM: The HPV-16 E6 and E6-AP complex functions as a
ubiquitin-protein ligase in the ubiquitination of p53. Cell.
75:495–505. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shaikh F, Sanehi P and Rawal R: Molecular
screening of compounds to the predicted Protein-Protein Interaction
site of Rb1-E7 with p53- E6 in HPV. Bioinformation. 8:607–612.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Goodwin EC and DiMaio D: Repression of
human papillomavirus oncogenes in HeLa cervical carcinoma cells
causes the orderly reactivation of dormant tumor suppressor
pathways. Proc Natl Acad Sci USA. 97:12513–12518. 2000. View Article : Google Scholar
|
|
6
|
Kornienko AE, Guenzl PM, Barlow DP and
Pauler FM: Gene regulation by the act of long non-coding RNA
transcription. BMC Biol. 11:592013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Spizzo R, Almeida MI, Colombatti A and
Calin GA: Long non-coding RNAs and cancer: a new frontier of
translational research? Oncogene. 31:4577–4587. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pandey RR, Mondal T, Mohammad F, et al:
Kcnq1ot1 antisense noncoding RNA mediates lineage-specific
transcriptional silencing through chromatin-level regulation. Mol
Cell. 32:232–246. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang X, Arai S, Song X, et al: Induced
ncRNAs allosterically modify RNA-binding proteins in cis to inhibit
transcription. Nature. 454:126–130. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang KC and Chang HY: Molecular mechanisms
of long noncoding RNAs. Mol Cell. 43:904–914. 2011. View Article : Google Scholar
|
|
11
|
Tsai MC, Manor O, Wan Y, et al: Long
noncoding RNA as modular scaffold of histone modification
complexes. Science. 329:689–693. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gupta RA, Shah N, Wang KC, et al: Long
non-coding RNA HOTAIR reprograms chromatin state to promote cancer
metastasis. Nature. 464:1071–1076. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Panzitt K, Tschernatsch MM, Guelly C, et
al: Characterization of HULC, a novel gene with striking
up-regulation in hepatocellular carcinoma, as noncoding RNA.
Gastroenterology. 132:330–342. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Huarte M, Guttman M, Feldser D, et al: A
large intergenic noncoding RNA induced by p53 mediates global gene
repression in the p53 response. Cell. 142:409–419. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hung T, Wang Y, Lin MF, et al: Extensive
and coordinated transcription of noncoding RNAs within cell-cycle
promoters. Nat Genet. 43:621–629. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ji P, Diederichs S, Wang W, et al:
MALAT-1, a novel noncoding RNA, and thymosin beta4 predict
metastasis and survival in early-stage non-small cell lung cancer.
Oncogene. 22:8031–8041. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yang L, Lin C, Liu W, et al: ncRNA- and
Pc2 methylation-dependent gene relocation between nuclear
structures mediates gene activation programs. Cell. 147:773–788.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Miyagawa R, Tano K, Mizuno R, et al:
Identification of cis- and trans-acting factors involved in the
localization of MALAT-1 noncoding RNA to nuclear speckles. RNA.
18:738–751. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tripathi V, Ellis JD, Shen Z, et al: The
nuclear-retained noncoding RNA MALAT1 regulates alternative
splicing by modulating SR splicing factor phosphorylation. Mol
Cell. 39:925–938. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Luo JH, Ren B, Keryanov S, et al:
Transcriptomic and genomic analysis of human hepatocellular
carcinomas and hepatoblastomas. Hepatology. 44:1012–1024. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lai MC, Yang Z, Zhou L, et al: Long
non-coding RNA MALAT-1 over expression predicts tumor recurrence of
hepatocellular carcinoma after liver transplantation. Med Oncol.
29:1810–1816. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Koshimizu TA, Fujiwara Y, Sakai N, Shibata
K and Tsuchiya H: Oxytocin stimulates expression of a noncoding RNA
tumor marker in a human neuroblastoma cell line. Life Sci.
86:455–460. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Guffanti A, Iacono M, Pelucchi P, et al: A
transcriptional sketch of a primary human breast cancer by 454 deep
sequencing. BMC Genomics. 10:1632009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yamada K, Kano J, Tsunoda H, et al:
Phenotypic characterization of endometrial stromal sarcoma of the
uterus. Cancer Sci. 97:106–112. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xu C, Yang M, Tian J, Wang X and Li Z:
MALAT-1: a long non-coding RNA and its important 3′ end functional
motif in colorectal cancer metastasis. Int J Oncol. 39:169–175.
2011.
|
|
26
|
Ying L, Chen Q, Wang Y, Zhou Z, Huang Y
and Qiu F: Upregulated MALAT-1 contributes to bladder cancer cell
migration by inducing epithelial-to-mesenchymal transition. Mol
Biosyst. 8:2289–2294. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ponting CP and Belgard TG: Transcribed
dark matter: meaning or myth? Hum Mol Genet. 19:R162–R168. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dermitzakis ET, Reymond A and Antonarakis
SE: Conserved non-genic sequences - an unexpected feature of
mammalian genomes. Nat Rev Genet. 6:151–157. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Nana-Sinkam SP and Croce CM: Non-coding
RNAs in cancer initiation and progression and as novel biomarkers.
Mol Oncol. 5:483–491. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wilusz JE, Freier SM and Spector DL: 3′
end processing of a long nuclear-retained noncoding RNA yields a
tRNA-like cytoplasmic RNA. Cell. 135:919–932. 2008.
|
|
31
|
Guo F, Li Y, Liu Y, Wang J, Li Y and Li G:
Inhibition of metastasis-associated lung adenocarcinoma transcript
1 in CaSki human cervical cancer cells suppresses cell
proliferation and invasion. Acta Biochim Biophys Sin (Shanghai).
42:224–229. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ohtani N, Yamakoshi K, Takahashi A and
Hara E: The p16INK4a-RB pathway: molecular link between cellular
senescence and tumor suppression. J Med Invest. 51:146–153. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Molinolo AA, Marsh C, El Dinali M, et al:
mTOR as a molecular target in HPV-associated oral and cervical
squamous carcinomas. Clin Cancer Res. 18:2558–2568. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li Y, Wang F, Xu J, et al: Progressive
miRNA expression profiles in cervical carcinogenesis and
identification of HPV-related target genes for miR-29. J Pathol.
224:484–495. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kranjec C and Banks L: A systematic
analysis of human papillomavirus (HPV) E6 PDZ substrates identifies
MAGI-1 as a major target of HPV type 16 (HPV-16) and HPV-18 whose
loss accompanies disruption of tight junctions. J Virol.
85:1757–1764. 2011. View Article : Google Scholar
|
|
36
|
Gök M, Heideman DA, van Kemenade FJ, et
al: HPV testing on self collected cervicovaginal lavage specimens
as screening method for women who do not attend cervical screening:
cohort study. BMJ. 340:c10402010.PubMed/NCBI
|
|
37
|
Karnon J, Peters J, Platt J, Chilcott J,
McGoogan E and Brewer N: Liquid-based cytology in cervical
screening: an updated rapid and systematic review and economic
analysis. Health Technol Assess. 8:1–78. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Cuzick J, Clavel C, Petry KU, et al:
Overview of the European and North American studies on HPV testing
in primary cervical cancer screening. Int J Cancer. 119:1095–1101.
2006. View Article : Google Scholar : PubMed/NCBI
|