Introduction
Breast cancer (BC) is the most frequently diagnosed
cancer and the leading cause of cancer-related mortality among
females worldwide (1). It is
estimated that one in eight females will develop BC in their
lifetime. Staging in BC is based on tumor size, nodal involvement
and distant metastasis, similar to other solid tumors. However,
clinical staging is not the only relevant factor in the management
of BC. Gene expression profiling studies have revealed that BC
consists of heterogeneous subtypes that show substantial variation
in their molecular and clinical characteristics (2–4).
Clinically, this heterogeneous disease is categorized into the
three following therapeutic groups (5): Estrogen receptor (ER)-positive, human
epidermal growth factor receptor 2 (HER2)-amplified and
triple-negative BC [lacking ER, progesterone receptor (PR) and
HER2]. This high heterogeneity is associated with
significant challenges in BC management, and these molecular
features are key factors in the response to therapy and disease
outcome.
Chemotherapy is an important modality in the
treatment of patients with BC. Systemic therapy improves the
disease-free survival of patients with BC, but does not lead to
remission in the majority of patients with advanced or metastatic
disease (6). The resistance to
chemotherapeutic agents commonly results in the subsequent
recurrence and metastasis of cancer cells. Currently, there is no
sensitive and specific biomarker to predict the response to
chemotherapy in BC (7).
Extensive studies over the last decade have
indicated that microRNAs (miRNA/miRs) are important regulators of
numerous cellular processes. Widespread miRNA deregulation in
cancer has resulted in the identification of their roles in
oncogenesis and tumor-suppression, and has indicated their
involvement in cancer initiation, progression and metastasis
(8). A number of oncogenic miRNAs
have been identified that are associated with BC (9). In recent years, circulating miRNAs
have drawn wide interest due to their potential as cancer
biomarkers. Several studies have indicated the potential of
circulating miRNAs as predictive biomarkers for the early detection
and prognosis of cancer (10,11).
Although it has been suggested that circulating miRNAs may be
associated with the response to treatment, only a few studies have
analyzed the predictive value of the pretreatment levels of
circulating miRNAs as a marker of chemoresistance in BC (12–14).
In concordance with a recent study (15), in early unpublished experiments we
determined a number of BC-associated miRNAs, which are at
relatively low (let-7, miR-10b, miR-34, miR-155 and miR-200c) or
abundant (miR-21, miR-195 and miR-221) levels in the blood plasma
of patients with BC. The aim of the present study was to
investigate the changes in the plasma levels of these miRNA
molecules in response to treatment and their association with the
clinicopathological parameters.
Materials and methods
Patients
This study included a cohort of 25 female patients
with invasive ductal carcinoma of the breast. The patients had been
diagnosed with primary operable (T2-3N1) or locally advanced
(T1-3N2 or T4Nx) disease. The patient characteristics are shown in
Table I. The inclusion criteria for
enrolment in the study were normal bone marrow, no contraindication
for chemotherapy, no cardiac, renal or hepatic limitations and the
provision of informed consent. Peripheral blood was obtained prior
to treatment and at the end of the fourth cycle of neoadjuvant
chemotherapy (four cycles of 90 mg/m2 epirubicin and 600
mg/m2 cyclophosphamide). The study was approved by the
Istanbul Medical Faculty Local Ethics Committee of Istanbul
University (Istanbul, Turkey) and the experiments were undertaken
with the understanding and written consent of each subject. The
study conforms with The Code of Ethics of the World Medical
Association (Declaration of Helsinki).
 | Table IClinicopathological characteristics of
the patients. |
Table I
Clinicopathological characteristics of
the patients.
| Characteristics | Value |
|---|
| Age, years |
| Median | 47.5 |
| Range | 36–68 |
| Staging, n |
| Stage II | 12 |
| Stage III–IV | 13 |
| Her-2 status, n |
| Positive | 12 |
| Negative | 13 |
| Estrogen receptor,
n |
| High positive | 12 |
| Low positive | 5 |
| Negative | 8 |
| Progesterone
receptor, n |
| High positive | 6 |
| Low positive | 8 |
| Negative | 11 |
| Ki-67 index, n |
| High | 13 |
| Low | 12 |
| Pathological
response, n |
| Minimal/good | 17 |
| Complete | 8 |
miRNA quantification in plasma
Cell-free RNA molecules were extracted from the
plasma using TRIzol reagent (Applichem, Düren, Germany). Briefly,
200 μl of the plasma sample was mixed with 800 μl TRIzol reagent,
and homogenized. The mixture was added to 200 μl chloroform and
incubated on ice for 5 min. Following centrifugation at 12,550 × g
for 5 min, the upper RNA phase was transferred into 550 μl propanol
containing tubes. Following centrifugation at 12,550 × g, the RNA
containing pellet was washed with 75% ethanol, air-dried and
dissolved in polymerase chain reaction (PCR)-grade water.
cDNA was synthesized using the miScript Reverse
Transcription kit (Qiagen, Valencia, CA, USA) according to the
manufacturer’s instructions. The kit includes a poly-A polymerase
and a reverse transcriptase. The first step adds a poly-A tail to
the 3′-end of the small RNA molecules, while in the second step,
the reverse transcriptase converts RNA to cDNA. To quantitate the
miRNAs, miScript Primer assays (Qiagen) were used, which include a
universal primer specific to the poly-A tail and a miRNA-specific
primer. SYBR Green (Qiagen) was used as the florescent molecule.
The amplified PCR product had a size of ~80 bp. The small RNA
molecule, RNU1A (Qiagen), was used as a control.
Quantitative PCR was performed using LightCycler
Instrument 480 (Roche Diagnostics, Mannheim, Germany). The PCR
program included a fast start step of 10 min at 95°C followed by 45
cycles of amplification. Each cycle consisted of denaturation at
95°C for 10 sec, annealing at 60°C for 10 sec and elongation at
70°C for 10 sec.
As the reference molecule, miR-205 was used, which
showed relatively stable expression [cross-over threshold (Ct) of
<30)] throughout all samples and was not significantly affected
by the therapy. For quantitation, the comparative ΔCt method was
used. Serial dilutions of cDNA were generated using primers for the
internal control of miR-205, which exhibited a linear decrease in
expression (correlation coefficient=0.99). These dilution series
were co-amplified in each PCR session, and the expression values of
the internal control and miRNAs from a given sample were derived
from the Ct values using the dilution standard. The quantitative
experiments were performed twice and the mean values were
calculated.
Statistical analyses
The Pearson correlation test was used to evaluate
the correlation between the individual miRNA molecules.
Mann-Whitney U and Wilcoxon tests were utilized to assess the
univariate differences between the pretreatment plasma levels and
clinicopathological parameters, or between the changes during
neoadjuvant chemotherapy and clinicopathological parameters.
Statistical analyses were conducted with SPSS version 16.0 (SPSS,
Inc., Chicago, IL, USA). P<0.05 was used to indicate a
statistically significant difference.
Results
Pretreatment plasma levels of miRNAs and
their association with clinicopathological parameters
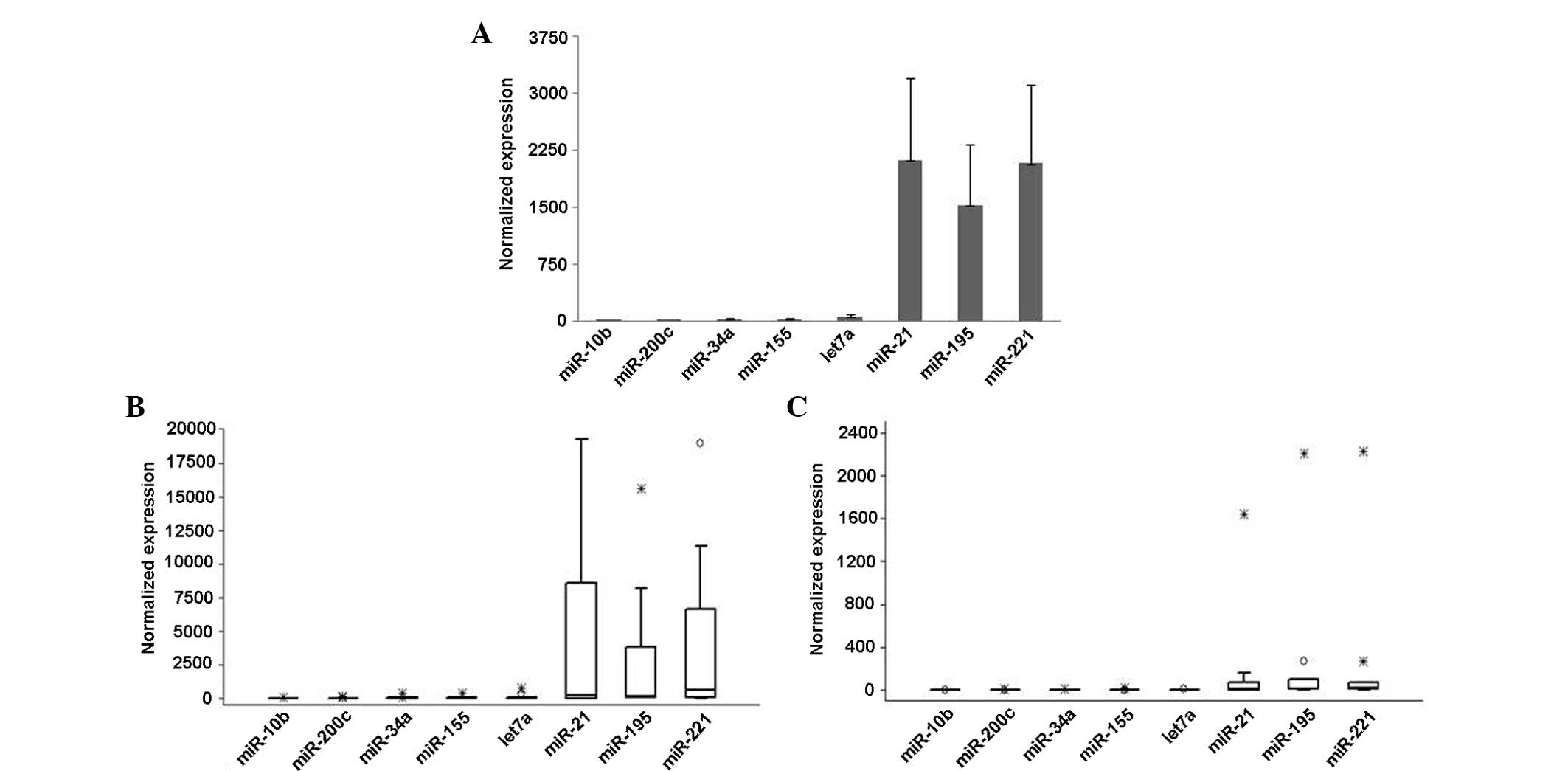
miR-21, miR-221 and miR-195 had the highest basal
expression levels in the blood plasma, while other miRNAs were
expressed at much lower levels (Fig.
1A). The basal plasma levels of eight miRNAs were highly
variable between the patients; for example, the relative expression
level of miR-221 ranged from 2.3 to 18,900 (median, 63; mean,
2,080). The rates were similar for miR-21 and miR-195. A high
correlation was identified between individual miRNAs (P<0.01 for
all), with the exception of miR-34, where the correlation with
miR-21 (P=0.04) and miR-195 (P=0.06) was somewhat weaker.
The association between the pretreatment miRNA
levels and clinicopathological parameters were investigated, and
the plasma levels of all miRNAs were observed to be higher in the
patients with stage II tumors (n=13) than in those with higher
tumor stages (stage III–IV; n=12) (Fig.
1B and C). The difference was most significant for miR-155,
miR-21 (both P=0.05) and miR-221 (P=0.07). For the discrimination
of ER-positive and -negative cases, the most useful molecule was
let-7, with pretreatment levels that were lower in the patients
with ER-negative tumors. No associations were observed between the
basal miRNA levels and PR status, c-erbB2 levels or Ki67 index.
Change of the miRNA levels during
neoadjuvant chemotherapy and their association with
clinicopathological parameters
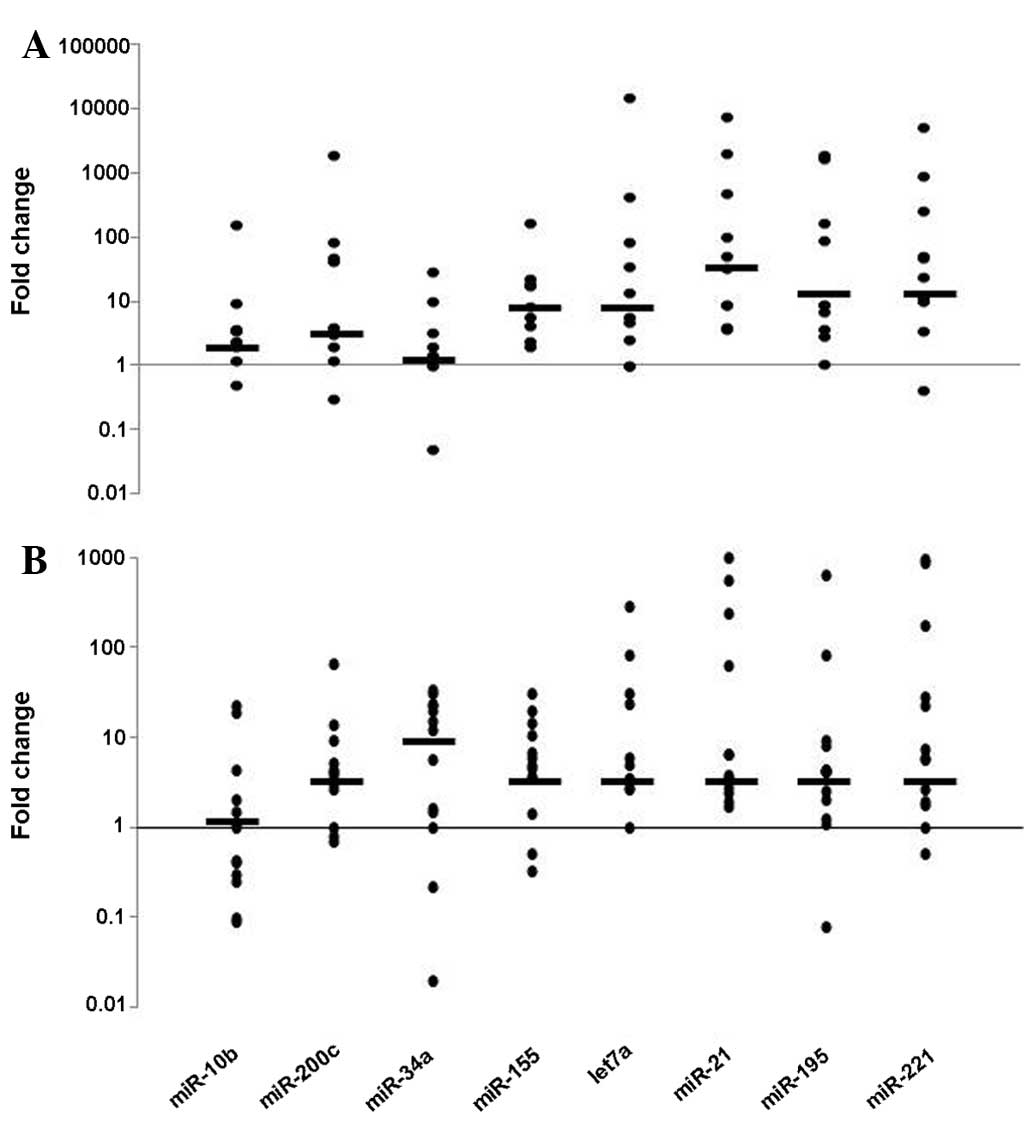
When compared with the basal levels, the plasma
levels of the majority of the miRNAs at the fourth cycle of
neoadjuvant chemotherapy had decreased in half of the patients
(n=13; Fig. 2A) whilst they had
increased in the other half (n=13; Fig.
2B). The rate of change was highly variable among the miRNA
molecules. In general, for the molecules that had high expression
levels (such as miR-21, miR-195 and miR-221), the extent of change
was also higher among all samples (~500-fold on average). For
samples exhibiting an increase following therapy, the highest
change was 980-fold. The samples in which the miRNA levels declined
exhibited a much more pronounced change (≤15,500-fold). Notably,
decreasing levels were more frequent in patients with early-stage
disease, and the change was statistically significant for miR-21
and miR-195 (P<0.05). In patients with a lower Ki-67 index,
decreasing levels were more frequently observed for the let-7
molecule (P=0.02), while the change was not significant for other
molecules. When the clinical pathological response was categorized
as ‘adequate’ versus ‘complete’, no significant differences were
observed between the pre- and post-treatment levels.
Discussion
The present study was conducted to investigate the
manner in which systemic therapy in the neoadjuvant setting affects
circulating miRNAs of low or high levels in patients with BC.
Considering the pretreatment levels of eight miRNAs, miR-21,
miR-221 and miR-195 had the highest expression levels. This is in
concordance with previous studies describing high levels of these
miRNAs in the blood circulation of patients with BC (12,16,17).
As expected, the plasma levels of the miRNAs were highly variable
among the patients; those with stage II tumors exhibited higher
plasma levels than those with higher tumor stages. miR-155 was the
most distinct molecule for discrimination, however, this finding
was not in concordance with a study by Chen et al (18), which reported higher miR-155
expression in advanced tumors. Another notable observation in the
present study was the lower plasma let-7 levels in patients with
ER-negative tumors. Consistent with this finding, previous studies
have revealed that let-7 is a negative regulator of ERα signaling
(19,20), indicating that plasma let-7 levels
may serve as a marker for the ER status.
The effect of chemotherapy on plasma miRNA levels
was found to vary between patients in the present study. The extent
of change was also highly variable for the individual miRNAs. The
change was much more pronounced (≤15,500-fold) for the samples in
which the miRNA levels decreased compared with the samples which
showed an increase. This may present as a further indication that
these miRNAs are highly expressed in breast tumors. Notably, in
patients with stage II tumors who had higher miRNA plasma levels
than those with advanced stages, the rate of decrease following
neoadjuvant chemotherapy was more frequent, which may reflect
shrinkage of the tumor. This observation is also consistent with a
study indicating that miR-221 induces cell survival and resistance
in cancer (21), since a favorable
therapy response was observed in all patients. A similar finding
during chemotherapy has been described for miR-155 (7). Decreasing levels of let-7 were more
frequent in patients with a lower Ki-67 index (P=0.02), while no
significant differences were observed among the other molecules.
Thus, the let-7 molecule may be a candidate to follow the response
of BC patients who have a low proliferation index. In this series,
almost all patients had a fair/complete pathological response to
therapy, even though certain patients exhibited declining plasma
miRNA levels while others exhibited increasing miRNA levels. A
previous study by Zhao et al (12) reported that patients with varying
miR-221 plasma levels exhibited significant differences in the
overall response rate, but not in the pathological complete
response rate. This indicated that patients with a good
pathological response may exhibit varying plasma levels for a given
miRNA.
In conclusion, miR-21, miR-195 and miR-221 are
highly expressed in the blood circulation of BC patients and are
affected most frequently by chemotherapy, particularly in patients
with early-stage tumors. This information may be valuable in future
studies when assessing the response of patients to therapy.
Individual miRNAs, such as let-7, appear to bear potential for use
in assessing patients subgroups with given clinicopathological
characteristics, such as ER status or Ki-67 index.
Acknowledgements
This study was supported by the Istanbul University
Scientific Projects Coordination Unit (project no. 9661).
References
|
1
|
Jemal A, Bray F, Center MM, et al: Global
cancer statistics. CA Cancer J Clin. 61:69–90. 2011.
|
|
2
|
Perou CM, Sørlie T, Eisen MB, et al:
Molecular portraits of human breast tumours. Nature. 406:747–752.
2000.
|
|
3
|
Sotiriou C, Neo SY, McShane LM, et al:
Breast cancer classification and prognosis based on gene expression
profiles from a population-based study. Proc Natl Acad Sci USA.
100:10393–10398. 2003.
|
|
4
|
Sotiriou C and Pusztai L: Gene-expression
signatures in breast cancer. N Engl J Med. 360:790–800. 2009.
|
|
5
|
Cancer Genome Atlas Network. Comprehensive
molecular portraits of human breast tumours. Nature. 490:61–70.
2012.
|
|
6
|
Moulder S: Intrinsic resistance to
chemotherapy in breast cancer. Womens Health (Lond Engl).
6:821–830. 2010.
|
|
7
|
Sun Y, Wang M, Lin G, et al: Serum
microRNA-155 as a potential biomarker to track disease in breast
cancer. PLoS One. 7:e470032012.
|
|
8
|
Braicu C, Calin GA and Berindan-Neagoe I:
MicroRNAs and cancer therapy - from bystanders to major players.
Curr Med Chem. 20:3561–3573. 2013.
|
|
9
|
Harquail J, Benzina S and Robichaud GA:
MicroRNAs and breast cancer malignancy: an overview of
miRNA-regulated cancer processes leading to metastasis. Cancer
Biomark. 11:269–280. 2012.
|
|
10
|
Madhavan D, Cuk K, Burwinkel B and Yang R:
Cancer diagnosis and prognosis decoded by blood-based circulating
microRNA signatures. Front Genet. 4:1162013.
|
|
11
|
Cuk K, Zucknick M, Heil J, et al:
Circulating microRNAs in plasma as early detection markers for
breast cancer. Int J Cancer. 132:1602–1612. 2013.
|
|
12
|
Zhao R, Wu J, Jia W, et al: Plasma miR-221
as a predictive biomarker for chemoresistance in breast cancer
patients who previously received neoadjuvant chemotherapy.
Onkologie. 34:675–680. 2011.
|
|
13
|
Jung EJ, Santarpia L, Kim J, et al: Plasma
microRNA 210 levels correlate with sensitivity to trastuzumab and
tumor presence in breast cancer patients. Cancer. 118:2603–2614.
2012.
|
|
14
|
Wang H, Tan G, Dong L, et al: Circulating
MiR-125b as a marker predicting chemoresistance in breast cancer.
PLoS One. 7:e342102012.
|
|
15
|
Liu H: MicroRNAs in breast cancer
initiation and progression. Cell Mol Life Sci. 69:3587–3599.
2012.
|
|
16
|
Heneghan HM, Miller N, Kelly R, Newell J
and Kerin MJ: Systemic miRNA-195 differentiates breast cancer from
other malignancies and is a potential biomarker for detecting
noninvasive and early stage disease. Oncologist. 15:673–682.
2010.
|
|
17
|
Kumar S, Keerthana R, Pazhanimuthu A and
Perumal P: Overexpression of circulating miRNA-21 and miRNA-146a in
plasma samples of breast cancer patients. Indian J Biochem Biophys.
50:210–214. 2013.
|
|
18
|
Chen J, Wang BC and Tang JH: Clinical
significance of microRNA-155 expression in human breast cancer. J
Surg Oncol. 106:260–266. 2012.
|
|
19
|
Zhao Y, Deng C, Wang J, et al: Let-7
family miRNAs regulate estrogen receptor alpha signaling in
estrogen receptor positive breast cancer. Breast Cancer Res Treat.
127:69–80. 2011.
|
|
20
|
Sun X, Qin S, Fan C, et al: Let-7: a
regulator of the ERα signaling pathway in human breast tumors and
breast cancer stem cells. Oncol Rep. 29:2079–2087. 2013.
|
|
21
|
Zhao G, Cai C, Yang T, et al: MicroRNA-221
induces cell survival and cisplatin resistance through PI3K/Akt
pathway in human osteosarcoma. PloS One. 8:e539062013.
|