1. Introduction
Tumor development is initiated by an accumulation of
genetic and epigenetic alterations, which promote tumor initiation,
invasion and metastasis (1,2). During the development of human
neoplasms, the hallmarks of cancer are acquired, including the
sustainment of proliferative signaling, the evasion of growth
suppressors, the resistance to cell death, the enabling of
replicative immortality, the induction of angiogenesis, the
activation of invasion and metastasis, genome instability and
mutations, the tumor promotion of inflammation, the deregulation of
cellular energetics and the avoidance of immune destruction. These
hallmarks aid in our understanding of the diversity of neoplastic
disease (3). With the continuous
development of cancer research, our understanding of the molecular
pathogenesis of cancer has been enhanced. Increasing efforts in
cancer research have been focused on the study of oncogenes, tumor
suppressors and signaling pathways. Since the development of a more
in-depth understanding of the molecular etiology of carcinogenesis,
specific oncogenes have been identified and have led to the
generation of ‘molecular-targeted therapy’ (4). The development of a ‘pan-cancer’
therapy may be possible by targeting an oncogene which is
ubiquitously overexpressed in almost all types of cancer and has a
regulatory role in the multistep processes of carcinogenesis
(5).
A novel gene that has been identified is AEG-1 [also
known as Metadherin (MTDH) and Lysine-rich CEACAM1 co-isolated
(LYRIC)], which has emerged as a potentially crucial mediator of
malignant tumors, and a key converging point of a complex network
of oncogenic signaling pathways (6,7).
AEG-1/MTDH presents as an ideal target for the development of the
next generation of effective cancer therapeutics.
2. Molecular cloning and structure of
AEG-1/MTDH
AEG-1/MTDH was first reported by Su et al
(8) in 2002 as a
neuropathology-associated gene induced in human fetal astrocytes
following human immunodeficiency virus-1 (HIV-1) infection or
treatment with recombinant HIV-1 envelope glycoprotein (gp120).
Subsequently, Kang et al (9)
described the full-length cloning and functional characterization
of AEG-1/MTDH. Next, Brown and Ruoslahti (10) used a phage expression library of
complementary deoxyribonucleic acid (cDNA) from a mouse model of
the lung metastasis of breast carcinoma to identify a lung homing
peptide in AEG-1/MTDH that was overexpressed in metastatic breast
cancer and promoted the homing of breast cancer cells to the lungs.
In 2004, Britt et al (11)
and Sutherland et al (12)
separately reported a novel protein, LYRIC, that colocalized with
the tight junction protein, ZO-1, in polarized prostate epithelial
cells (11) and was present in the
cytoplasm, endoplasmic reticulum (ER), perinuclear regions and
nucleolus (12).
Full-length AEG-1/MTDH cDNA includes 3,611 bp,
excluding the poly-A tail (9). The
open reading frame from nucleotide 220 to 1,968 of AEG-1/MTDH
encodes a single pass transmembrane protein (putative 582-amino
acid) of ~64 kDa and with an isoelectric point of 9.33 (9). AEG-1/MTDH orthologues are reported in
the majority of vertebrate species, but are not detected in
invertebrates. With the exception of three putative lysine-rich
nuclear localization signals (NLS), AEG-1/MTDH has no recognizable
protein domains (13), and the
presence of putative (monopartite or bipartite) NLS between amino
acids 79–91, 432–451 and 561–580 suggests that it may enter into
the nucleus (6).
The AEG-1/MTDH gene consists of 12 exons/11 introns,
as identified through the use of a genomic BLAST search (http://blast.ncbi.nlm.nih.gov/Blast.cgi), and is
located at 8q22 where cytogenetic analysis of human gliomas
suggests recurrent amplification (9). In a number of malignancies, such as
malignant glioma (14),
hepatocellular carcinoma (HCC) (15) and breast cancer (16), the location is significant. In HCC
and breast cancer, genomic amplification of AEG-1/MTDH has been
found in patients (15,16). Several protein motif analysis
methods have predicted that AEG-1/MTDH has a single transmembrane
domain (9–12). With regard to whether AEG-1/MTDH is
a type I b membrane protein (with a cytoplasmic C-terminal without
a signal peptide) (9,11,12) or
a type II protein (with an extracytoplasmic C-terminal) (10,11),
considerable debate remains. In recent years, functional and
clinical evidence significantly support an important function of
AEG-1/MTDH in cancer development, including transformation, the
evasion of apoptosis, invasion and metastasis (13). However, a generous amount of
research is required to fully characterize the molecular and
biochemical properties of AEG-1/MTDH.
3. Oncogenic functions of AEG-1/MTDH
AEG-1/MTDH mRNA is ubiquitously expressed at varying
levels in all organs, as determined by multi-tissue northern
blotting (9). The potential role of
AEG-1/MTDH is to promote tumor progression and metastasis in human
HCC cell lines and colorectal cancer (CRC) (17,18).
AEG-1/MTDH localizes in the cell membrane, cytoplasm, ER and
nucleus, and contributes to a group of signaling pathways, such as
the PI3K-AKT, nuclear factor-κB (NF-κB), mitogen-activated protein
kinase (MAPK) and Wnt pathways (19). AEG-1/MTDH is seminal in regulating
proliferation, invasion, angiogenesis, metastasis and
chemoresistance, as determined by ‘gain-of-function’ and
‘loss-of-function’ studies in human cancer cells and through the
analysis of a transgenic mouse model (20). AEG-1/MTDH promotes tumorigenesis by
modulating multiple signal transduction pathways and altering gene
expression changes (Fig. 1).
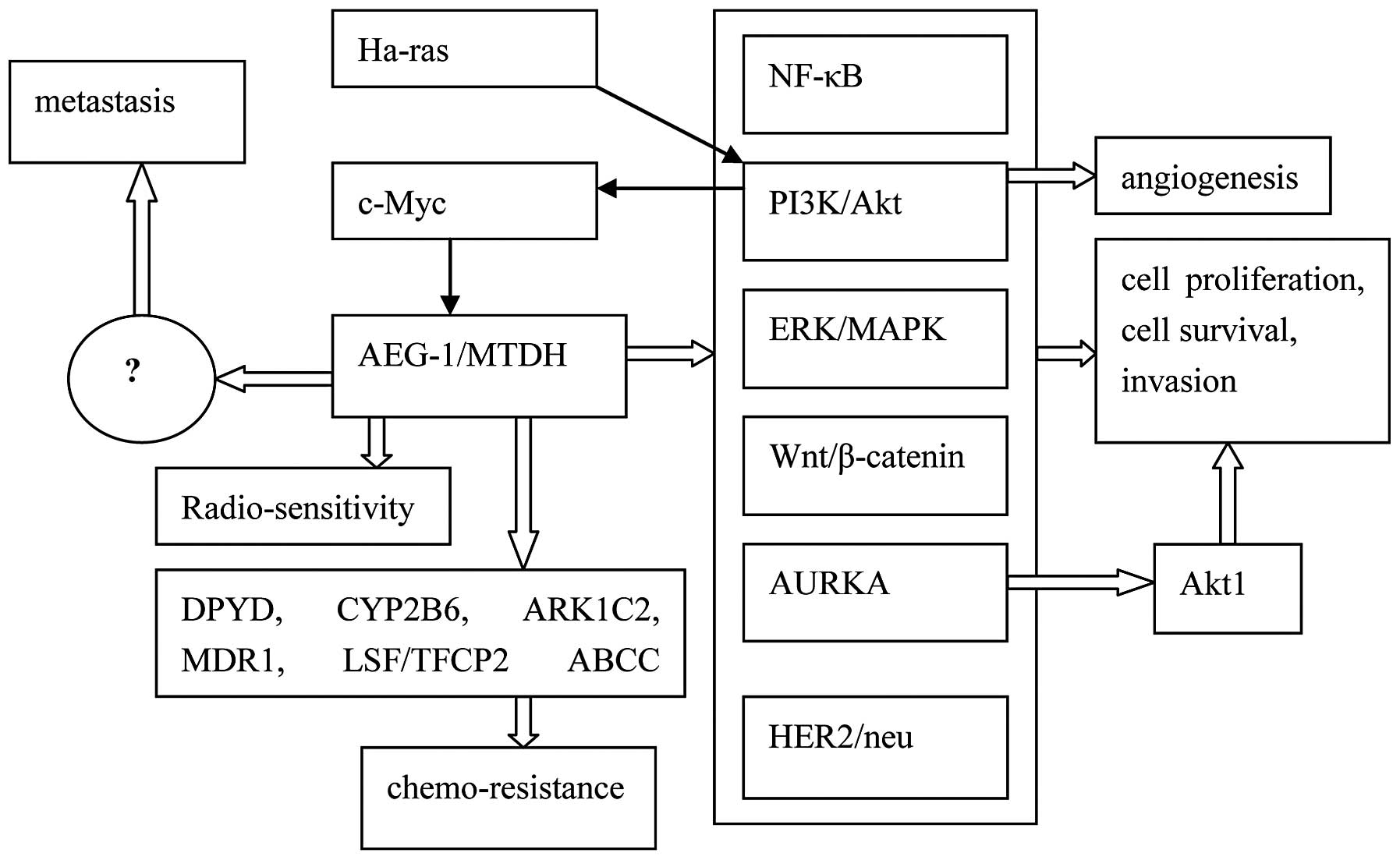 | Figure 1Hypothetical molecular mechanism of
the action of AEG-1/MTDH. The thick white arrows indicate the
regulation by AEG-1/MTDH, while the thin black arrows indicate the
mechanisms that regulate AEG-1/MTDH. AEG-1, astrocyte elevated
gene-1; NF-κB, nuclear factor-κB; MTDH, metadherin; MAPK,
mitogen-activated protein kinase; AURKA, Aurora A kinase; DPYD,
dihydropryimidine dehydrogenase; CYP2B6, cytochrome P450B6; ARK1C2,
dyhydrodiol dehydrogenase; MDR1, multidrug-resistance gene 1. |
Integration of oncogenic pathways
Overexpression of AEG-1/MTDH synergizes with
oncogenic Ha-Ras to enhance the soft agar colony formation of
non-tumorigenic immortalized melanocytes and provides evidence of
the tumor promoting activity of AEG-1/MTDH (9). AEG-1/MTDH expression is markedly
induced by Ha-Ras, which activates the PI3K/Akt pathway leading to
the binding of the transcription factor, c-Myc, to the E-box
element in the promoter region of AEG-1/MTDH and the regulation of
AEG-1/MTDH transcription (21).
AEG-1 overexpression inhibits serum starvation-induced apoptosis by
activating the Ras and PI3K-Akt signaling pathways (22). AEG-1/MTDH is also crucial in the
carcinogenesis of non-small cell lung cancer (NSCLC) and inhibits
apoptosis by enhancing the level of the antiapoptotic protein,
Bcl-2, and activating the PI3K/Akt pathway (23). AEG-1/MTDH also downregulates the
transcriptional activity of forkhead box (FOXO) 1 through the
PI3K/Akt signaling pathway in MCF-7 and MDA-MB-435 breast cancer
cells (24). In addition,
AEG-1/MTDH is important in the aggressiveness of NSCLC through the
activation of the PI3K-Akt and NF-κB signaling pathways (25).
Emdad et al (26) first reported that AEG-1/MTDH
promotes the anchorage-independent growth and invasion of Hela
cells by activating the NF-κB pathway. At the mRNA and protein
levels, AEG-1/MTDH is upregulated during CRC development and
aggressiveness (from normal mucosa to primary CRC, followed by
lymph node metastasis and finally liver metastasis) through the
NF-κB signaling pathway (27).
AEG-1/MTDH contributes to the chemoresistance of cervical cancer
cells by increasing autophagy and the activation of the ERK/NF-κB
pathway (28). AEG-1/MTDH also
modulates the BCCIPα protein levels in prostate tumor cells through
an indirect mechanism involving the NF-κB signaling pathway
(29). In malignant glioma cells,
AEG-1/MTDH regulates invasion and migration through activation of
the NF-κB signaling pathway (30),
in which AEG-1/MTDH is involved in the lipopolysaccharide
(LPS)-induced inflammatory response (31) and mediates the LPS-induced migration
and invasion of breast cancer cells (32).
In human HCC cells, AEG-1/MTDH activates the MAPK
pathways, including ERK and p38 MAPK, and is also associated with
the Wnt/β-catenin pathway through the activation of the
Raf/MEK/MAPK branch of the Ras signaling pathway (15). In proximal tubular epithelial cells,
AEG-1/MTDH is important in TGF-β1-induced epithelial-mesenchymal
transition (EMT) through activation of p38 MAPK (33). A recent study has suggested that
AEG-1/MTDH contributes to the pathogenesis of diffuse large B-cell
lymphoma mediated through regulation of the Wnt/β-catenin pathway
(34).
Furthermore, in the carcinogenesis of acute myeloid
leukemia (AML), a novel functional link has been revealed between
AEG-1/MTDH and Aurora A kinase (AURKA) with regard to Akt1
activation (35). In human AML
cells, AEG-1/MTDH overexpression is vital for the maintenance of
the malignant state via upregulation of Akt1, which is mediated by
AURKA activation (35). In breast
cancer cells, AEG-1/MTDH facilitates cancer proliferation and
invasion by upregulating HER2/neu expression (36).
Angiogenesis and metastasis
AEG-1/MTDH overexpression converts non-tumorigenic
human HCC cells into highly aggressive vascular tumors. In
addition, AEG-1/MTDH modulates the expression of genes associated
with invasion, angiogenesis, metastasis, chemoresistance and
senescence, as determined by microarray analysis (15). AEG-1/MTDH has a dominant function in
regulating oncogenic transformation and angiogenesis (37). AEG-1/MTDH expression is also
increased in multiple cancers and is crucial in oncogenic
transformation and angiogenesis (38–41).
In a phage display study, Brown and Ruoslahti (10) identified that a lung homing domain
(amino acids 378–440 in mice and 381–443 in humans) in AEG-1/MTDH
was a mediator of 4T1 mouse mammary tumor cell adhesion to the lung
vasculature, and suggested that AEG-1/MTDH is important in breast
cancer metastasis. In CRC, Jiang et al (18) showed that AEG-1/MTDH is
overexpressed in liver metastasis patients compared with patients
without liver metastasis. In addition, AEG-1/MTDH may present as a
potential novel biomarker for early liver metastasis. In a large
proportion of epithelial ovarian cancer patients with peritoneal
dissemination and/or lymph node metastasis, AEG-1/MTDH is
overexpressed and is a novel predictor of metastasis (42). In summary, AEG-1/MTDH is crucial in
lymph node metastasis (39,43–45)
and contributes to tumor progression, including transformation, the
evasion of apoptosis, invasion and metastasis (13).
Chemoresistance
One of the important hallmarks of aggressive cancers
is chemoresistance. Previous studies have suggested that AEG-1/MTDH
contributes to a broad spectrum of resistance to various
chemotherapeutics, including 5-fluorouracil, doxorubicin,
paclitaxel, cisplatin and 4-hydroxycyclophosphamide (16,46–48).
In human HCC cells, the gene expression profiles of overexpressed
AEG-1/MTDH have been identified in several drug-metabolizing
enzymes involved in chemoresistance, including dihydropyrimidine
dehydrogenase, cytochrome P450B6, dihydrodiol dehydrogenase,
ATP-binding cassette transporter 11/MRP8 and transcription factor
LSF/TFCP2 (15). AEG-1/MTDH
increases multidrug-resistance gene 1 (MDR1) protein expression,
which facilitates the association between MDR1 mRNA and polysomes,
leading to increased translation, the inhibition of ubiquitination
and the resultant proteasome-mediated degradation of the MDR1
protein (47). The inhibition of
AEG-1/MTDH may be an effective method in HCC chemotherapy (47). Bhutia et al (49) also showed that protective autophagy
is the cause of AEG-1-mediated chemoresistance, and that the
inhibition of AEG-1/MTDH results in a decrease in the protective
autophagy and chemosensitization of cancer cells. Due to the
multiple functions of AEG-1/MTDH in drug resistance, AEG-1/MTDH is
a viable target as an anticancer agent for a wide range of cancer
types (50).
Recent results have also indicated that AEG-1/MTDH
affects the radiosensitivity of cervical cancer cells (51). In summary, it has become apparent
that AEG-1/MTDH is an important oncogene, which is overexpressed in
numerous human cancer types. Through a number of signaling
cascades, AEG-1/MTDH is involved in several crucial aspects of
tumor progression, including transformation, proliferation, the
evasion of apoptosis, cell survival, migration and invasion,
angiogenesis, metastasis and chemoresistance (52). Future studies are required to
evaluate the correlation between AEG-1/MTDH function and signaling
changes and interacting partners in order to highlight novel
perspectives for AEG-1/MTDH as a significant target for the
clinical treatment of various cancers.
4. Clinical-translational advances
In keeping with the role of AEG-1/MTDH in a number
of different aspects of malignancy, AEG-1/MTDH has been found to
correlate with tumor progression and poor prognosis in a number of
cancer types, including HCC (17,53–58)
and breast (59–64), prostate (65–67),
glioma (68–70) and esophageal cancer (71) (Table
I). These studies indicate that AEG-1/MTDH may be a powerful
independent marker for poor prognosis and a viable target for
anticancer therapeutics.
 | Table IStudies on AEG-1/MTDH in a variety of
cancer types. |
Table I
Studies on AEG-1/MTDH in a variety of
cancer types.
| Cancer types | First author/s,
year (ref.) |
|---|
| HCC | Zhou et al,
2012 (17); Srivastava et
al, 2012 (53); Gong et
al, 2012 (54); Zhu et
al, 2011 (55); Yoo et
al, 2011 (56); Ahn et
al, 2013 (57) |
| Gastric cancer | Jian-bo et
al, 2011 (74); Zhang et
al, 2013 (75); Baygi et
al, 2012 (76) |
| CRC | Gnosa et al,
2012 (27); Wang et al, 2012
(78); Zhang et al,
2013(79) |
| ESCC | Yu et al,
2009 (71) |
| GBC | Sun et al,
2011 (81); Liu and Yang, 2013
(82) |
| Breast cancer | Kang et al,
2005 (9); Brown and Ruoslahti, 2004
(10); Hu et al, 2009
(16); Tokunaga et al, 2012
(59); Li et al, 2011
(60); Liu et al, 2011
(61); Wan et al (62) Kong et al, 2012 (63); Zhang et al, 2013 (64) |
| NSCLC | Ke et al,
2013 (23); Song et al, 2009
(25); Sun et al, 2012
(84) |
| RCC | Chen et al,
2010 (85); Erdem et al,
2013 (86) |
| PC | Thirkettle et
al, 2009 (65); Kikuno et
al, 2007 (66); Lee et
al, 2012 (67) |
| Glioma | Lee et al,
2011 (68); Liu et al, 2010
(69); Emdad et al, 2010
(70) |
| Neuroblastoma | Liu et al,
2009 (48) |
| Osteosarcoma | Liu et al,
2013 (87) |
| Ovarian cancer | Li et al,
2011 (42); Li et al, 2012
(88); Yuan et al, 2012
(89) |
HCC
Using transgenic mice with hepatocyte-specific
AEG-1/MTDH expression, a previous study identified novel aspects of
AEG-1/MTDH functions, including the induction of steatosis, the
inhibition of senescence and the activation of the coagulation
pathway to augment aggressive hepatocarcinogenesis (53). The results suggested that the
expression of the AEG-1/MTDH protein was significantly higher in
cancer cell lines with high metastatic potential, such as Sk-HEP-1
and MHCC-97H, than in those with low metastatic potential, such as
HepG2 and Huh7. Additionally, AEG-1/MTDH has been shown to be
closely associated with the abilities of the orientation chemotaxis
and adhesion of HCC cells (17). In
hepatitis B virus (HBV)-related HCC patients, AEG-1/MTDH expression
has been found to significantly correlate with the American Joint
Committee on Cancer (7th edition) (72) stage, T and N classification,
vascular invasion and histological differentiation (54). In addition, patients with high
AEG-1/MTDH levels have been found to exhibit poor survival rates
compared with those with low AEG-1/MTDH levels (54). In HBV-related HCC patients,
AEG-1/MTDH is a potential prognostic marker for overall survival
(OS) and tumor progression, and is a chemotherapeutic target
(54). In HCC tumors, the high
expression of AEG-1/MTDH has also been found to correlate with
microvascular invasion, pathological satellites, poor
differentiation and tumor-node-metastasis (TNM) stages II to III.
Furthermore, AEG-1/MTDH promotes HCC metastasis through induction
of the EMT process (55). In a nude
mouse model, the shRNA-mediated downregulation of AEG-1/MTDH
resulted in reduced migratory capacity in HCC cell lines, and also
reduced the number of abdominal and pulmonary metastases (55). AEG-1/MTDH overexpression and
staphylococcal nuclease domain-containing 1 (SND1) lead to
increased levels of RNA-induced silencing complex activity and
contribute to hepatocarcinogenesis (56). Additionally, AEG-1/MTDH is a
prognostic predictor of HCC following curative hepatectomy
(57). AEG-1/MTDH overexpression
has been identified in a high percentage of hepatitis B and C
virus-positive HCC cases, and is key in the regulation of
hepatocarcinogenesis (58). In
summary, the AEG-1/MTDH gene is amplified in human HCC patients and
promotes chemoresistance, angiogenesis and metastasis.
Gastric cancer
The high expression of AEG-1/MTDH is observed in
gastric cancer tissues. AEG-1/MTDH overexpression is associated
with TNM Stage (TNM Classification of Malignant Tumors) (73), Ki-67 proliferation index and poor
survival, and is an independent prognostic factor for gastric
cancer in multivariate analysis (74). Inhibition of AEG-1/MTDH expression
by specific small interfering RNA (siRNA) clearly inhibits SGC-7901
cell growth and enhances cell apoptosis by reducing the
phosphorylation of AKT and glycogen synthase kinase-3β, and
decreasing the levels of β-catenin, lymphoid enhancer binding
factor 1 and cyclin D1 (74).
Furthermore, the inhibition of cell proliferation and cell cycle
arrest in gastric carcinoma SGC-7901 cells, mediated by the
downregulation of AEG-1/MTDH expression, may be closely associated
with changes in the expression of cell cycle-related proteins,
including cdk2, cyclin D1 and p21 (75). AEG-1/MTDH overexpression is a useful
prognostic factor in patients with gastric cancer, and the
inhibition of AEG-1/MTDH may provide a novel therapeutic strategy
for gastric cancer. However, in Iranian patients, AEG-1/MTDH mRNA
expression was significantly elevated in 46.6% of examined tumor
tissues, while its expression was low in others (36.6%) (76). There is only a marginal statistical
difference between the AEG-1/MTDH gene expression in all tumor
specimens compared with their paired non-tumor specimens, and no
statistically significant association has been identified between
the grades and types of tumors (76). At the transcriptional level,
AEG-1/MTDH levels may be increased in gastric cancer tissue
samples, but with considerable heterogeneity, and it may have the
potential to be used as a target for diagnostic/therapeutic
purposes only in a subset of patients (76). Therefore, the status of AEG-1/MTDH
expression and its significance in gastric cancer remains unclear
and requires further investigation.
CRC
SND1 is a novel AEG-1/MTDH-interacting protein, and
a functionally and clinically significant mediator of metastasis in
breast cancer (77). In colon
cancer tissues, a positive correlation has been identified between
AEG-1/MTDH and SND1 expression by immunohistochemical staining;
AEG-1/MTDH- and SND1-positive expression has been found to
significantly correlate with nodal status, pathological stage and
differentiation (78). Furthermore,
OS time in colon cancer patients with positive AEG-1/MTDH and SND1
expression is significantly shorter than in those without
AEG-1/MTDH and SND1 expression (78). The positive expression of AEG-1/MTDH
and SND1 is an independent poor prognostic predictor in colon
cancer, as observed by multivariate Cox regression analysis, and
the increased expression of AEG-1/MTDH and/or SND1 is closely
associated with the carcinogenesis, progression and prognosis of
colon cancer (78). In predicting
the prognosis of colon cancer, the coexpression of AEG-1/MTDH/SND1
may be a novel distinctive marker. During CRC development and
aggressiveness, the AEG-1/MTDH mRNA and protein levels are
upregulated and have been associated with tumor location and stage
(27). Zhang et al (79) was the first to show that AEG-1/MTDH
interacts with β-catenin in SW480 CRC cell lines and that
AEG-1/MTDH expression closely correlates with the progression of
CRC. The aforementioned studies also suggest that AEG-1/MTDH may be
a potential therapeutic target in CRC.
Esophageal squamous cell carcinoma
(ESCC)
Immunohistochemical analysis of 168 ESCC specimens
revealed that 47.6% of tumors exhibit high levels of AEG-1/MTDH
expression (71). In ESCC patients,
AEG-1/MTDH overexpression has been found to significantly correlate
with the TNM stage, histological differentiation and a shorter
survival time, and is an independent poor prognostic indicator, as
determined by multivariate analysis (71).
Gallbladder carcinoma (GBC)
High AEG-1/MTDH expression is present in highly
invasive GBC-SD cell lines at the protein and mRNA levels, and in
GBC samples (63.4%) compared with normal gallbladder mucosa. In
addition, AEG-1/MTDH has been found to markedly correlate with
differentiation degree, Nevin stage (80), Ki-67 expression and liver
infiltration (81). In GBC
patients, AEG-1/MTDH overexpression leads to a shorter survival
time and is an independent prognostic marker, as determined by
multivariate analysis (81).
AEG-1/MTDH is a useful marker of GBC progression and may be a
potential therapeutic target. The immunohistochemical analysis of
96 benign and 108 malignant lesions of the gallbladder revealed
that the positive expression of erythropoietin-producing
hepatoma-amplified sequence receptor A7 (EphA7) and AEG-1/MTDH is
significantly higher in gallbladder adenocarcinoma than in benign
lesions (82). In gallbladder
adenocarcinoma, the positive expression of EphA7 and AEG-1/MTDH has
been found to significantly correlate with differentiation, tumor
masses, lymph node metastasis, invasion and OS, and is an
independent poor prognostic predictor, as determined by
multivariate analysis (82). The
elevated expression of EphA7 and/or AEG-1/MTDH has also been found
to closely correlate with the carcinogenesis, progression, clinical
biological behavior and prognosis of gallbladder adenocarcinoma
(82).
Breast cancer
AEG-1/MTDH is expressed at low levels or is absent
in the majority of normal human breast tissues, but is frequently
overexpressed in ductal carcinoma in situ (83), breast cancer cell lines or breast
tumors (9,10,16,59).
The analyses of breast tumor samples collected in the USA and Japan
revealed strikingly similar patterns of AEG-1/MTDH expression and
clinical association (16,59). AEG-1/MTDH overexpression is
significantly associated with estrogen receptor- and progesterone
receptor-negative expression, a high nuclear grade, poor
disease-free survival, a high Ki67 index, poor distant
metastasis-free survival and poor OS (59). AEG-1/MTDH overexpression has a
particularly negative impact on the prognosis of node-negative
patients and is independently associated with poor disease-free and
distant metastasis-free survival rates, as determined by
multivariate analysis (59). Li
et al (60) showed that
AEG-1/MTDH may promote EMT in breast cancer cells in driving the
progression of their aggressive behavior. Furthermore, Li et
al (61) assessed the variants
of the AEG-1/MTDH gene and their potential association with breast
cancer susceptibility. The study discovered nine novel variants and
found two variants to be associated with the susceptibility of
breast cancer. AEG-1/MTDH has a dual function in promoting
chemoresistance and metastasis, and is a key functional target of
the 8q22 genomic gain that is frequently observed in breast cancer
patients with a poor prognosis (62). In summary, AEG-1/MTDH overexpression
contributes to an aggressive phenotype, leading to a poor prognosis
in primary invasive breast cancer. Blocking AEG-1/MTDH and its
regulated pathways is likely to be beneficial in breast cancer
cells or tissues (63,64).
NSCLC
In NSCLC cell lines and tissues, AEG-1/MTDH has a
crucial function in the aggressiveness leading to a poor clinical
prognosis (25) and promotes NSCLC
metastasis by modulating matrix metalloproteinase-9 (MMP-9)
expression (84). In NSCLC,
AEG-1/MTDH overexpression has been found to significantly correlate
with clinical staging, differentiation, lymph node metastasis and a
shorter OS time (23). In L-78
cells, a previous study observed that AEG-1/MTDH siRNA treatment
significantly upregulated caspase-3, markedly decreased Bcl-2,
largely attenuated PI3K p110 protein expression and phosphorylated
Akt (23). These results suggested
that AEG-1/MTDH is crucial in the carcinogenesis of NSCLC and that
it inhibits apoptosis by enhancing the level of antiapoptotic
protein, Bcl-2, and activating the PI3K/Akt pathway (23).
Renal cell carcinoma (RCC)
A markedly higher expression of AEG-1/MTDH was
identified in eight cases of RCC tissue compared with the paired
normal tissue from the same patient by quantitative reverse
transcription polymerase chain reaction and western blot analysis
(85). At the mRNA and protein
levels, the expression of AEG-1/MTDH was also increased in four RCC
cell lines, in contrast to normal tubular epithelial human kidney
HK 2 cells (85). Furthermore,
AEG-1/MTDH overexpression has been found to significantly correlate
with tumor grade, clinical staging, T classification, metastasis
classification and a shorter survival time, as determined by
immunohistochemical analysis (85).
A microarray study showed that high AEG-1/MTDH and p53 expression
correlate with the prognostic parameters in RCC patients and may be
associated with tumor progression (86). In conclusion, the AEG-1/MTDH protein
is overexpressed in RCC and is important in tumor differentiation
and progression.
Prostate cancer (PC)
A significantly higher expression of AEG-1/MTDH has
been identified in PC samples and cell lines compared with benign
prostatic hyperplasia tissue samples and normal prostate epithelial
cells (65,66). The knockdown of AEG-1/MTDH induces
PC cell apoptosis through upregulation of FOXO3a activity, and also
attenuates the constitutive activity of NF-κB and activator protein
1 (AP-1), with a corresponding depletion in the expression of NF-κB
and AP-1-regulated genes. Knockdown also significantly decreases
the cell invasion properties of PC-3 and DU145 PC cells (66). Recent findings have suggested that
AEG-1/MTDH is overexpressed in PC cells and that cryptotanshinone
exerts antitumor activity via inhibition of hypoxia inducible
factor 1α, AEG-1/MTDH and vascular endothelial growth factor in
hypoxic PC-3 cells (67).
Glioma
Oncogenic AEG-1/MTDH is overexpressed in >90% of
brain tumors and promotes gliomagenesis, particularly tumor growth
and invasion, two primary characteristics of glioma (68). Lee et al (68) found that AEG-1/MTDH contributes to
glioma-induced neurodegeneration, a hallmark of this fatal tumor,
by regulating EAAT2 expression. Liu et al (69) suggested that AEG-1/MTDH expression
significantly correlates with the clinicopathological stage of the
glioma and contributes to glioma progression by enhancing MMP-9
transcription and promoting tumor cell invasiveness. AEG-1/MTDH
expression is significantly elevated in >90% of diverse human
brain tumor samples, including glioblastoma multiformes and
astrocytic tumors, and in human glioma cell lines compared with
normal brain tissues and normal astrocytes, as determined by
western blot analysis and immunohistochemistry (70). AEG-1/MTDH may have a crucial
function in the pathogenesis of glioma and may represent a viable
potential target for malignant glioma therapy.
AEG-1/MTDH overexpression has also been documented
in melanoma (9), neuroblastoma
(48), osteosarcoma (87) and ovarian cancer (42,88,89),
as well as in other tumors. However, certain controversy exists
regarding the localization of the AEG-1/MTDH protein in the nucleus
or cytoplasm of cancer cells, and the utility of nuclear or
cytoplasmic AEG-1/MTDH to predict the course and prognosis of
disease (20). Future studies are
required to evaluate AEG-1/MDTH interaction partners in various
cancer types and their significance in cancer progression. In
summary, AEG-1/MTDH overexpression markedly correlates with
advanced tumor characteristics and a poor clinical prognosis, and
is a promising target for novel therapeutics.
5. Clinical translation and therapeutic
targeting strategy
AEG-1/MTDH inhibition may be an effective anticancer
strategy, considering that AEG-1/MTDH is overexpressed in a variety
of cancer types and regulates the fundamental processes of
carcinogenesis. AEG-1/MTDH overexpression can be utilized to
identify subgroups of patients who require more intense treatments
and who are likely to benefit from AEG-1/MTDH-targeted therapies.
Patients with AEG-1/MTDH overexpression or amplification in
different tumors may potentially suffer from metastatic recurrence
and require close monitoring for clinical signs of relapse for
early therapeutic intervention. High-risk patients require a higher
dose of chemotherapy, the efficacy of which may be increased
through the combination of chemotherapy with AEG-1/MTDH inhibition.
Based on the molecular targeting of AEG-1/MTDH, novel cancer
treatments consist of several possible avenues.
AEG-1/MTDH-knockdown in MG-63 osteosarcoma cells significantly
decreases ET-1 expression (at the mRNA and protein levels), cell
invasion, MMP-2 expression and cell survival against cisplatin
(87). Transfection of a
recombinant plasmid containing pcDNA-AEG-1/MTDH-microRNA (miR)-4
significantly suppresses AEG-1/MTDH at the mRNA and protein levels
by >69% in MDA-MB-231 breast cancer cells, and significantly
inhibits proliferation, motility and migration compared with
controls (90). Silencing
AEG-1/MTDH expression by RNA interference was found to effectively
reduce metastasis by 3- to 10-fold in a MDA-MB-231 xenograft model
of breast cancer lung metastasis, and it was also shown to
sensitize chemoresistant MDA-MB-231 breast tumors to paclitaxel or
doxorubicin (16). Ward et
al (91) observed that
AEG-1/MTDH is a direct target of miR-375, and that
AEG-1/MTDH-knockdown partially phenocopies the effects of miR-375
on the sensitivity to tamoxifen and the reversal of EMT in breast
cancer. The treatment of tamoxifen-resistant breast cancer may be
aided by the development of potential therapeutic approaches, such
as the re-expression of miR-375 or the targeting of AEG-1/MTDH
(91). In breast cancer cells,
AEG-1/MTDH-miRNA may be important in the downregulation of
proliferation, motility and migration, and must be used as a future
potential small molecule inhibitor therapeutic targeting strategy.
AEG-1/MTDH-knockdown decreases nuclear β-catenin accumulation and
suppresses the migration and invasion of SW620 colorectal carcinoma
cell lines (79). Adenoviral
delivery of AEG-1/MTDH-targeting shRNA inhibits xenograft primary
tumor growth in HCC mice (15). The
inhibition of AEG-1/MTDH in neoadjuvant or adjuvant settings not
only increases the response rate of chemotherapy, but also reduces
tumor growth and the systemic spread of metastatic cancer (13). In MDA-MB231 human breast cancer
cells, exposure to cadmium chloride affects AEG-1/MTDH expression
(92). Current and future studies
must focus on the translational aspects of AEG-1/MTDH, and on the
understanding of AEG-1/MTDH function in physiological and
pathological processes using transgenic and knockout mouse models
in cancer. The knockdown of AEG-1/MTDH (32,33,41,45,51,93,94) or
AEG-1/MTDH inhibition (92,95,96)
can inhibit transformation, proliferation, the evasion of
apoptosis, migration and invasion, metastasis, angiogenesis and
chemoresistance. In summary, the functional characterization of
AEG-1/MTDH as a novel protein with poorly-characterized functions
is urgently required to realize its full therapeutic potential.
6. Conclusion and future perspectives
Accumulating evidence has demonstrated that
AEG-1/MTDH has clear functions in the regulation of various
physiological and pathological processes (97). AEG-1/MTDH is an extremely important
molecule for the regulation of a variety of pathological and
physiological processes by modulating the transcription and
translation of factors involved in the signaling pathways (97). It is clear that AEG-1/MTDH has a
decisive role in the process of tumorigenesis in multiple models,
as determined by in vitro and in vivo studies and
expression analysis (98). The
multiple functions of AEG-1/MTDH highlight several important
clinical implications. AEG-1/MTDH, as a ubiquitous biomarker for
aggressive tumors, may be used for the routine screening of
patients (13). AEG-1/MTDH is an
important regulator in multiple aspects of cancer development and
progression. Clinical and functional analyses suggest that the
multifunctional gene, AEG-1/MTDH, is a potentially valuable target
in cancer treatments. Chen et al (99) showed that the anti-AEG-1
autoantibody response may be a diagnostic biomarker for cancer
patients with AEG-1-positive expression, and a possible inducer,
with substantial immunity against AEG-1 by immunization boosting
with AEG-1 vaccines. In a therapeutic model, the AEG-1/MTDH vaccine
increased the chemosensitivity to doxorubicin and inhibited breast
cancer lung metastasis (100). The
AEG-1/MTDH vaccine in combination with chemotherapy may offer novel
strategies for the treatment of cancer metastasis.
In conclusion, AEG-1/MTDH is a valuable diagnostic
or prognostic biomarker based on its overexpression and
correlations with disease staging and outcome throughout a wide
range of cancer types. However, larger patient studies and a
prospective investigation on the correlation between AEG-1/MTDH
mRNA or protein in the blood and circulating tumor cells, and in
urine and biopsy samples compared with clinical characteristics are
urgently required.
Acknowledgements
The authors would like to thank Dr Yong Zhao for the
critical reading of the manuscript.
References
|
1
|
Hanahan D and Weinberg RA: The hallmarks
of cancer. Cell. 100:57–70. 2000.
|
|
2
|
Gupta GP and Massague J: Cancer
metastasis: building a framework. Cell. 127:679–695. 2006.
|
|
3
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: the next generation. Cell. 144:646–674. 2011.
|
|
4
|
Weinstein IB and Joe AK: Mechanisms of
disease: Oncogene addiction-a rationale for molecular targeting in
cancer therapy. Nat Clin Pract Oncol. 3:448–457. 2006.
|
|
5
|
Cancer Genome Atlas Research Network.
Weinstein JN, Collisson EA, Mills GB, Shaw KR, Ozenberger BA,
Ellrott K, Shmulevich I, Sander C and Stuart JM: The Cancer Genome
Atlas Pan-Cancer analysis project. Nat Genet. 1113–1120. 2013.
|
|
6
|
Emdad L, Sarkar D, Su ZZ, et al: Astrocyte
elevated gene-1: recent insights into a novel gene involved in
tumor progression, metastasis and neurodegeneration. Pharmacol
Ther. 114:155–170. 2007.
|
|
7
|
Kwong LN and Chin L: The metastasis
problem gets stickier. Cancer Cell. 15:1–2. 2009.
|
|
8
|
Su ZZ, Kang DC, Chen Y, et al:
Identification and cloning of human astrocyte genes displaying
elevated expression after infection with HIV-1 or exposure to HIV-1
envelope glycoprotein by rapid subtraction hybridization, RaSH.
Oncogene. 21:3592–3602. 2002.
|
|
9
|
Kang DC, Su ZZ, Sarkar D, et al: Cloning
and characterization of HIV-1-inducible astrocyte elevated gene-1,
AEG-1. Gene. 353:8–15. 2005.
|
|
10
|
Brown DM and Ruoslahti E: Metadherin, a
cell surface protein in breast tumors that mediates lung
metastasis. Cancer Cell. 5:365–374. 2004.
|
|
11
|
Britt DE, Yang DF, Yang DQ, et al:
Identification of a novel protein, LYRIC, localized to tight
junctions of polarized epithelial cells. Exp Cell Res. 300:134–148.
2004.
|
|
12
|
Sutherland HG, Lam YW, Briers S, Lamond AI
and Bickmore WA: 3D3/lyric: a novel transmembrane protein of the
endoplasmic reticulum and nuclear envelope, which is also present
in the nucleolus. Exp Cell Res. 294:94–105. 2004.
|
|
13
|
Hu G, Wei Y and Kang Y: The multifaceted
role of MTDH/AEG-1 in cancer progression. Clin Cancer Res.
15:5615–5620. 2009.
|
|
14
|
Warr T, Ward S, Burrows J, et al:
Identification of extensive genomic loss and gain by comparative
genomic hybridisation in malignant astrocytoma in children and
young adults. Genes Chromosomes Cancer. 31:15–22. 2001.
|
|
15
|
Yoo BK, Emdad L, Su ZZ, et al: Astrocyte
elevated gene-1 regulates hepatocellular carcinoma development and
progression. J Clin Invest. 119:465–577. 2009.
|
|
16
|
Hu G, Chong RA, Yang Q, et al: MTDH
activation by 8q22 genomic gain promotes chemoresistance and
metastasis of poor-prognosis breast cancer. Cancer Cell. 15:9–20.
2009.
|
|
17
|
Zhou Z, Deng H, Yan W, et al: Expression
of metadherin/AEG-1 gene is positively related to orientation
chemotaxis and adhesion of human hepatocellular carcinoma cell
lines of different metastatic potentials. J Huazhong Univ Sci
Technolog Med Sci. 3:353–357. 2012.
|
|
18
|
Jiang T, Zhu A, Zhu Y and Piao D: Clinical
implications of AEG-1 in liver metastasis of colorectal cancer. Med
Oncol. 29:2858–2863. 2012.
|
|
19
|
Lee SG, Kang DC, Desalle R, Sarkar D and
Fisher PB: AEG-1/MTDH/LYRIC, the beginning: initial cloning,
structure, expression profile, and regulation of expression. Adv
Cancer Res. 120:1–38. 2013.
|
|
20
|
Sarkar D and Fisher PB: AEG-1/MTDH/LYRIC:
clinical significance. Adv Cancer Res. 120:39–74. 2013.
|
|
21
|
Lee SG, Su ZZ, Emdad L, Sarkar D and
Fisher PB: Astrocyte elevated gene-1 (AEG-1) is a target gene of
oncogenic Ha-ras requiring phosphatidylinositol 3-kinase and c-Myc.
Proc Natl Acad Sci USA. 103:17390–17395. 2006.
|
|
22
|
Lee SG, Su ZZ, Emdad L, et al: Astrocyte
elevated gene-1 activates cell survival pathways through PI3K-Akt
signaling. Oncogene. 27:1114–1121. 2008.
|
|
23
|
Ke ZF, Mao X, Zeng C, et al: AEG-1
expression characteristics in human non-small cell lung cancer and
its relationship with apoptosis. Med Oncol. 30:3832013.
|
|
24
|
Li J, Yang L, Song L, et al: Astrocyte
elevated gene-1 is a proliferation promoter in breast cancer via
suppressing transcriptional factor FOXO1. Oncogene. 28:3188–3196.
2009.
|
|
25
|
Song L, Li W, Zhang H, et al:
Over-expression of AEG-1 significantly associates with tumour
aggressiveness and poor prognosis in human non-small cell lung
cancer. J Pathol. 219:317–326. 2009.
|
|
26
|
Emdad L, Sarkar D, Su ZZ, et al:
Activation of the nuclear factor kappaB pathway by astrocyte
elevatedgene-1: implications for tumor progression and metastasis.
Cancer Res. 66:1509–1516. 2006.
|
|
27
|
Gnosa S, Shen YM, Wang CJ, et al:
Expression of AEG-1 mRNA and protein in colorectal cancer patients
and colon cancer cell lines. J Transl Med. 10:1092012.
|
|
28
|
Zhang J, Zhang Y, Liu S, et al: Metadherin
confers chemoresistance of cervical cancer cells by inducing
autophagy and activating ERK/NF-κB pathway. Tumor Biol.
34:2433–2440. 2013.
|
|
29
|
Ash SC, Yang DQ and Britt DE: LYRIC/AEG-1
overexpression modulates BCCIPalpha protein levels in prostate
tumor cells. Biochem Biophys Res Commun. 371:333–338. 2008.
|
|
30
|
Sarkar D, Park ES, Emdad L, et al:
Molecular basis of nuclear factor-kappaB activation by astrocyte
elevated gene-1. Cancer Res. 68:1478–1484. 2008.
|
|
31
|
Khuda II, Koide N, Noman AS, et al:
Astrocyte elevated gene-1 (AEG-1) is induced by lipopolysaccharide
as toll-like receptor 4 (TLR4) ligand and regulates TLR4
signalling. Immunology. 128(Suppl 1): e700–e706. 2009.
|
|
32
|
Zhao Y, Kong X, Li X, et al: Metadherin
mediates lipopolysaccharide-induced migration and invasion of
breast cancer cells. PLoS One. 6:e293632011.
|
|
33
|
Wei J, Li Z, Chen W, et al: AEG-1
participates in TGF-beta1-induced EMT through p38 MAPK activation.
Cell Biol Int. 37:1016–1021. 2013.
|
|
34
|
Ge X, Lv X, Feng L, et al: Metadherin
contributes to the pathogenesis of diffuse large B-cell lymphoma.
PLoS One. 7:e394492012.
|
|
35
|
Long M, Hao M, Dong K, et al: AEG-1
overexpression is essential for maintenance of malignant state in
human AML cells via up-regulation of Akt1 mediated by AURKA
activation. Cell Signal. 25:1438–1446. 2013.
|
|
36
|
Zhang X, Zhang N and Zhang MX: Astrocyte
elevated gene-1 induces breast cancer proliferation and invasion
through upregulating HER2/neu expression. Chin Med J (Engl).
124:3546–3550. 2011.
|
|
37
|
Emdad L, Lee SG, Su ZZ, et al: Astrocyte
elevated gene-1 (AEG-1) functions as an oncogene and regulates
angiogenesis. Proc Natl Acad Sci USA. 106:21300–21305. 2009.
|
|
38
|
Noch E, Bookland M and Khalili K:
Astrocyte-elevated gene-1 (AEG-1) induction by hypoxia and glucose
deprivation in glioblastoma. Cancer Biol Ther. 11:32–9. 2011.
|
|
39
|
Li C, Li R, Song H, et al: Significance of
AEG-1 expression in correlation with VEGF, microvessel density and
clinicopathological characteristics in triple-negative breast
cancer. J Surg Oncol. 103:184–192. 2011.
|
|
40
|
Chen D, Yoo BK, Santhekadur PK, et al:
Insulin-like growth factor-binding protein-7 functions as a
potential tumor suppressor in hepatocellular carcinoma. Clin Cancer
Res. 17:6693–6701. 2011.
|
|
41
|
Long M, Dong K, Gao P, et al:
Overexpression of astrocyte-elevated gene-1 is associated with
cervical carcinoma progression and angiogenesis. Oncol Rep.
30:1414–1422. 2013.
|
|
42
|
Li C, Liu J, Lu R, et al: AEG -1
overexpression: a novel indicator for peritoneal dissemination and
lymph node metastasis in epithelial ovarian cancers. Int J Gynecol
Cancer. 21:602–608. 2011.
|
|
43
|
Liu Y, Su Z, Li G, et al: Increased
expression of metadherin protein predicts worse disease-free and
overall survival in laryngeal squamous cell carcinoma. Int J
Cancer. 133:671–679. 2013.
|
|
44
|
Deng N and Feng Y: Expression of EphA7 and
MTDH and clinicopathological significance in the squamous cell
cancer of the tongue. Zhong Nan Da Xue Xue Bao Yi Xue Ban.
12:1195–1198. 2011.(In Chinese).
|
|
45
|
Wang F, Ke ZF, Sun SJ, et al: Oncogenic
roles of astrocyte elevated gene-1 (AEG-1) in osteosarcoma
progression and prognosis. Cancer Biol Ther. 12:539–548. 2011.
|
|
46
|
Yoo BK, Gredler R, Vozhilla N, et al:
Identification of genes conferring resistance to 5-fluorouracil.
Proc Natl Acad Sci USA. 106:12938–12943. 2009.
|
|
47
|
Yoo BK, Chen D, Su ZZ, et al: Molecular
mechanism of chemoresistance by astrocyte elevated gene-1. Cancer
Res. 70:3249–3258. 2010.
|
|
48
|
Liu H, Song X, Liu C, et al: Knockdown of
astrocyte elevated gene-1 inhibits proliferation and enhancing
chemo-sensitivity to cisplatin or doxorubicin in neuroblastoma
cells. J Exp Clin Cancer Res. 28:192009.
|
|
49
|
Bhutia SK, Kegelman TP, Das SK, et al:
Astrocyte elevated gene-1 induces protective autophagy. Proc Natl
Acad Sci USA. 107:22243–22248. 2010.
|
|
50
|
Meng X, Thiel KW and Leslie KK: Drug
Resistance Mediated by AEG-1/MTDH/LYRIC. Adv Cancer Res.
120:135–157. 2013.
|
|
51
|
Zhao Y, Moran MS, Yang Q, et al:
Metadherin regulates radioresistance in cervical cancer cells.
Oncol Rep. 27:1520–1526. 2012.
|
|
52
|
Emdad L, Das SK, Dasgupta S, et al:
AEG-1/MTDH/LYRIC: signaling pathways, downstream genes, interacting
proteins, and regulation of tumor angiogenesis. Adv Cancer Res.
120:75–111. 2013.
|
|
53
|
Srivastava J, Siddiq A, Emdad L, et al:
Astrocyte elevated gene-1 promotes hepatocarcinogenesis: novel
insights from a mouse model. Hepatology. 56:1782–1791. 2012.
|
|
54
|
Gong Z, Liu W, You N, et al: Prognostic
significance of metadherin overexpression in hepatitis B
virus-related hepatocellular carcinoma. Oncol Rep. 27:2073–2079.
2012.
|
|
55
|
Zhu K, Dai Z, Pan Q, et al: Metadherin
promotes hepatocellular carcinoma metastasis through induction of
epithelial-mesenchymal transition. Clin Cancer Res. 17:7294–7302.
2011.
|
|
56
|
Yoo BK, Santhekadur PK, Gredler R, et al:
Increased RNA-induced silencing complex (RISC) activity contributes
to hepatocellular carcinoma. Hepatology. 53:1538–1548. 2011.
|
|
57
|
Ahn S, Hyeon J and Park CK: Metadherin is
a prognostic predictor of hepatocellular carcinoma after curative
hepatectomy. Gut Liver. 7:206–212. 2013.
|
|
58
|
Sarkar D: AEG-1/MTDH/LYRIC in Liver
Cancer. Adv Cancer Res. 120:193–221. 2013.
|
|
59
|
Tokunaga E, Nakashima Y, Yamashita N, et
al: Overexpression of metadherin/MTDH is associated with an
aggressive phenotype and a poor prognosis in invasive breast
cancer. Breast Cancer. 21:341–349. 2012.
|
|
60
|
Li X, Kong X, Huo Q, et al: Metadherin
enhances the invasiveness of breast cancer cells by inducing
epithelial to mesenchymal transition. Cancer Sci. 102:1151–1157.
2011.
|
|
61
|
Liu X, Zhang N, Li X, et al:
Identification of novel variants of metadherin in breast cancer.
PLoS One. 6:e175822011.
|
|
62
|
Wan L and Kang Y: Pleiotropic Roles of
AEG-1/MTDH/LYRIC in Breast Cancer. Adv Cancer Res. 120:113–134.
2013.
|
|
63
|
Kong X, Moran MS, Zhao Y and Yang Q:
Inhibition of metadherin sensitizes breast cancer cells to AZD6244.
Cancer Biol Ther. 13:43–49. 2012.
|
|
64
|
Zhang N, Wang X, Huo Q, et al:
MicroRNA-30a suppresses breast tumor growth and metastasis by
targeting metadherin. Oncogene. Jul 15–2013.(Epub ahead of
print).
|
|
65
|
Thirkettle HJ, Girling J, Warren AY, et
al: LYRIC/AEG-1 is targeted to different subcellular compartments
by ubiquitinylation and intrinsic nuclear localization signals.
Clin Cancer Res. 15:3003–3013. 2009.
|
|
66
|
Kikuno N, Shiina H, Urakami S, et al:
Knockdown of astrocyte-elevated gene-1 inhibits prostate cancer
progression through upregulation of FOXO3a activity. Oncogene.
26:7647–7655. 2007.
|
|
67
|
Lee HJ, Jung DB, Sohn EJ, et al:
Inhibition of hypoxia inducible factor alpha and astrocyte-elevated
gene-1 mediates cryptotanshinone exerted antitumor activity in
hypoxic PC-3 cells. Evid Based Complement Alternat Med.
2012:3909572012.
|
|
68
|
Lee SG, Kim K, Kegelman TP, et al:
Oncogene AEG-1 promotes glioma-induced neurodegeneration by
increasing glutamate excitotoxicity. Cancer Res. 71:6514–6523.
2011.
|
|
69
|
Liu L, Wu J, Ying Z, et al: Astrocyte
elevated gene-1 upregulates matrix metalloproteinase-9 and induces
human glioma invasion. Cancer Res. 70:3750–3759. 2010.
|
|
70
|
Emdad L, Sarkar D, Lee SG, et al:
Astrocyte elevated gene-1: a novel target for human glioma therapy.
Mol Cancer Ther. 9:79–88. 2010.
|
|
71
|
Yu C, Chen K, Zheng H, et al:
Overexpression of astrocyte elevated gene-1 (AEG-1) is associated
with esophageal squamous cell carcinoma (ESCC) progression and
pathogenesis. Carcinogenesis. 30:894–901. 2009.
|
|
72
|
Chun YH, Kim SU, Park JY, et al:
Prognostic value of the 7th edition of the AJCC staging system as a
clinical staging system in patients with hepatocellular carcinoma.
Eur J Cancer. 47:2568–2575. 2011.
|
|
73
|
Ahn HS, Lee HJ, Hahn S, et al: Evaluation
of the seventh American Joint Committee on Cancer/International
Union Against Cancer Classification of gastric adenocarcinoma in
comparison with the sixth classification. Cancer. 116:5592–5598.
2010.
|
|
74
|
Jian-bo X, Hui W, Yu-long H, et al:
Astrocyte-elevated gene-1 overexpression is associated with poor
prognosis in gastric cancer. Med Oncol. 28:455–462. 2011.
|
|
75
|
Zhang CF, Xia YH, Zheng QF, et al: Effect
of silencing AEG-1 with small interfering RNA on the proliferation
and cell cycle of gastric carcinoma SGC-7901 cells. Zhonghua Zhong
Liu Za Zhi. 35:22–27. 2013.(In Chinese).
|
|
76
|
Baygi ME and Nikpour P: Deregulation of
MTDH gene expression in gastric cancer. Asian Pac J Cancer Prev.
13:2833–2836. 2012.
|
|
77
|
Blanco MA, Alečković M, Hua Y, et al:
Identification of staphylococcal nuclease domain-containing 1
(SND1) as a Metadherin-interacting protein with
metastasis-promoting functions. J Biol Chem. 286:19982–19992.
2011.
|
|
78
|
Wang N, Du X, Zang L, et al: Prognostic
impact of Metadherin-SND1 interaction in colon cancer. Mol Biol
Rep. 39:10497–10504. 2012.
|
|
79
|
Zhang F, Yang Q, Meng F, et al: Astrocyte
elevated gene-1 interacts with β-catenin and increases migration
and invasion of colorectal carcinoma. Mol Carcinog. 52:603–610.
2013.
|
|
80
|
Fong Y, Wagman L, Gonen M, et al:
Evidence-based gallbladder cancer staging: changing cancer staging
by analysis of data from the National Cancer Database. Ann Surg.
243:767–774. 2006.
|
|
81
|
Sun W, Fan YZ, Xi H, et al: Astrocyte
elevated gene-1 overexpression in human primary gallbladder
carcinomas: an unfavorable and independent prognostic factor. Oncol
Rep. 26:1133–1142. 2011.
|
|
82
|
Liu DC and Yang ZL: MTDH and EphA7 are
markers for metastasis and poor prognosis of gallbladder
adenocarcinoma. Diagn Cytopathol. 41:199–205. 2013.
|
|
83
|
Su P, Zhang Q and Yang Q:
Immunohistochemical analysis of Metadherin in proliferative and
cancerous breast tissue. Diagn Pathol. 5:382010.
|
|
84
|
Sun S, Ke Z, Wang F, et al: Overexpression
of astrocyte-elevated gene-1 is closely correlated with poor
prognosis in human non-small cell lung cancer and mediates its
metastasis through up-regulation of matrix metalloproteinase-9
expression. Hum Pathol. 43:1051–1060. 2012.
|
|
85
|
Chen W, Ke Z, Shi H, Yang S and Wang L:
Overexpression of AEG-1 in renal cell carcinoma and its correlation
with tumor nuclear grade and progression. Neoplasma. 57:522–529.
2010.
|
|
86
|
Erdem H, Oktay M, Yildirim U, Uzunlar AK
and Kayikci MA: Expression of AEG-1 and p53 and their
clinicopathological significance in malignant lesions of renal cell
carcinomas: a microarray study. Pol J Pathol. 64:28–32. 2013.
|
|
87
|
Liu B, Wu Y and Peng D: Astrocyte elevated
gene-1 regulates osteosarcoma cell invasion and chemoresistance via
endothelin-1/endothelin A receptor signaling. Oncol Lett.
5:505–510. 2013.
|
|
88
|
Li C, Li Y, Wang X, et al: Elevated
expression of astrocyte elevated gene-1 (AEG-1) is correlated with
cisplatin-based chemoresistance and shortened outcome in patients
with stages III-IV serous ovarian carcinoma. Histopathology.
60:953–963. 2012.
|
|
89
|
Yuan C, Li X, Yan S, et al: The MTDH
(−470G>A) polymorphism is associated with ovarian cancer
susceptibility. PLoS One. 7:e515612012.
|
|
90
|
Wang S, Shu JZ, Cai Y, Bao Z and Liang QM:
Establishment and characterization of MTDH knockdown by artificial
MicroRNA interference - functions as a potential tumor suppressor
in breast cancer. Asian Pac J Cancer Prev. 13:2813–2818. 2012.
|
|
91
|
Ward A, Balwierz A, Zhang JD, et al:
Re-expression of microRNA-375 reverses both tamoxifen resistance
and accompanying EMT-like properties in breast cancer. Oncogene.
32:1173–1182. 2013.
|
|
92
|
Luparello C, Longo A and Vetrano M:
Exposure to cadmium chloride influences astrocyte-elevated gene-1
(AEG-1) expression in MDA-MB231 human breast cancer cells.
Biochimie. 94:207–213. 2012.
|
|
93
|
Vartak-Sharma N and Ghorpade A: Astrocyte
elevated gene-1 regulates astrocyte responses to neural injury:
implications for reactive astrogliosis and neurodegeneration. J
Neuroinflammation. 9:1952012.
|
|
94
|
Zhang N, Wang X, Huo Q, et al: The
oncogene metadherin modulates the apoptotic pathway based on the
tumor necrosis factor superfamily member TRAIL (Tumor Necrosis
Factor-related Apoptosis-inducing Ligand) in breast cancer. J Biol
Chem. 288:9396–9407. 2013.
|
|
95
|
Liu K, Guo L, Miao L, et al: Ursolic acid
inhibits epithelial-mesenchymal transition by suppressing the
expression of astrocyte-elevated gene-1 in human nonsmall cell lung
cancer A549 cells. Anticancer Drugs. 24:494–503. 2013.
|
|
96
|
Wu C, Jin B, Chen L, et al: MiR-30d
induces apoptosis and is regulated by the Akt/FOXO pathway in renal
cell carcinoma. Cell Signal. 25:1212–1221. 2013.
|
|
97
|
Yoo BK, Emdad L, Lee SG, et al: Astrocyte
elevated gene-1 (AEG-1): A multifunctional regulator of normal and
abnormal physiology. Pharmacol Ther. 130:1–8. 2011.
|
|
98
|
Sarkar D, Emdad L, Lee SG, et al:
Astrocyte elevated gene-1: far more than just a gene regulated in
astrocytes. Cancer Res. 69:8529–8535. 2009.
|
|
99
|
Chen X, Dong K, Long M, et al: Serum
anti-AEG-1 auto-antibody is a potential novel biomarker for
malignant tumors. Oncol Lett. 4:319–323. 2012.
|
|
100
|
Qian BJ, Yan F, Li N, et al:
MTDH/AEG-1-based DNA vaccine suppresses lung metastasis and
enhances chemosensitivity to doxorubicin in breast cancer. Cancer
Immunol Immunother. 60:883–893. 2011.
|















