Introduction
Tumor lysis syndrome (TLS) is a metabolic impairment
that arises in cancer patients, and is caused by the release of
cellular components into the bloodstream following the rapid lysis
of cancer cells (1–3). TLS occurs most frequently in patients
with hematological malignancies, such as acute lymphoblastic
leukemia and Burkitt’s lymphoma, following the initiation of
induction chemotherapy. TLS can also occur spontaneously in the
context of any cancer associated with high proliferation and/or a
large tumor burden (1–4). The release of intracellular contents,
including nucleic acids, proteins, phosphorus and potassium, can
lead to hyperuricemia, hyperkalemia, hyperphosphatemia,
hypocalcemia and, subsequently, renal failure, arrhythmias,
seizures and mortality (1–5).
Prevention and prompt treatment of TLS are crucial;
therefore, early recognition of patient condition and evaluation of
the risk of developing TLS is important. Guidelines and
recommendations for the evaluation of risk of and prophylaxis
against TLS have been published (2,3). TLS
is divided into laboratory TLS and clinical TLS (2,3,6), for
which Cairo and Bishop (6)
developed a system based on modifications to the Hande-Garrow
classification (7). Laboratory TLS
is defined as two or more laboratory changes [such as serum uric
acid (S-UA), potassium, phosphate and calcium levels] within 3 days
before or 7 days after cytotoxic therapy (2,3).
Clinical TLS requires the presence of laboratory TLS in addition to
one or more of the following significant clinical complications:
Renal insufficiency, cardiac arrhythmias, seizures or mortality.
Risk of TLS varies with cancer type, tumor burden, renal function
and laboratory findings, and can be categorized into low-,
intermediate- and high-risk groups (2,3).
The primary aims of the prophylaxis against and
direct management of TLS are to control serum concentrations of UA,
potassium, phosphate and calcium, and to avoid the development of
renal failure (8), of which
lowering the S-UA is considered to be the most important.
Hyperuricemia results from a rapid catabolism of purine-containing
nucleic acids from tumor cells, since purine nucleic acids are
converted to hypoxanthine, xanthine and, finally, to UA by xanthine
oxidase. According to published guidelines (2,3),
patients with an intermediate risk of developing TLS may be treated
with allopurinol, while patients with a high risk of developing TLS
may be given the recombinant uricase, rasburicase. Allopurinol is
an hypoxanthine analog and an inhibitor of xanthine oxidase, which
converts hypoxanthine to xanthine, and xanthine to UA (9). The action of allopurinol is relatively
slow, taking several days to produce a reduction in S-UA levels.
Furthermore, allopurinol is associated with various toxicities,
including hypersensitivity reactions and hepatic damage (10). Notably, cancer patients receiving
chemotherapy often exhibit renal dysfunction, which may increase
the toxicity of allopurinol, because allopurinol and its
metabolite, oxypurinol, are excreted by the kidney (11,12).
Febuxostat is a novel nonpurine-structured selective
xanthine oxidase inhibitor, which may be a promising alternative to
allopurinol in patients who are unable to tolerate allopurinol or
in those with renal dysfunction (11,13–15).
The present study retrospectively evaluated the UA-lowering
efficacy of febuxostat in patients with hematological malignancies
who were undergoing chemotherapy and at an intermediate risk of
developing TLS. Evaluation measures focused on the reduction of
S-UA, the management of TLS and the prevention of renal damage.
Patients and methods
Patients
Patients who were admitted to the University of
Fukui Hospital (Fukui, Japan) between September 2011 and May 2012
were retrospectively evaluated in the present study. All the
patients were newly diagnosed as having hematological malignancies
and were at an intermediate risk of developing TLS. These patients
received 10 mg febuxostat daily (the recommended starting dose for
patients with gout and hyperuricemia) during the administration of
induction chemotherapy. Patients did not receive any other
medications that might otherwise affect S-UA levels, such as
losartan, fenofibrate, atorvastatin, pyrazinamide or cyclosporine.
This study was approved by the ethics committee of the University
of Fukui Hospital.
Risk classification
Risk classification for TLS was made based on the
published guidelines (2,3). Diseases associated with an
intermediate risk of TLS included acute myeloid leukemia with a
peripheral white blood cell count between 10,000–50,000/μl; diffuse
large B-cell non-Hodgkin’s lymphoma; and any other diseases
associated with factors that could increase the risk of developing
TLS, including, elevated serum lactate dehydrogenase, extensive
bone marrow involvement, pre-existing renal disease or reduced
urinary output (2,3).
Categorization of hyperuricemia
Hyperuricemia is broadly classified into the
following three types, UA-overproduction type, UA-underexcretion
type and combined type, according to the guidelines for the
Management of Hyperuricemia and Gout, published in Japan in 2010
(16). Urinary UA excretion, UA and
creatinine clearance rate were determined to allow for
categorization of the hyperuricemia type (urinary UA excretion
>0.51 mg/kg/h and UA clearance rate >7.3 ml/min for
overproduction type; urinary UA excretion <0.48 mg/kg/h or UA
clearance rate <7.3 ml/min for underexcretion type; UA excretion
>0.51 mg/kg/h and UA clearance rate <7.3 ml/min for combined
type).
Administration of febuxostat
All patients received 10 mg febuxostat orally after
breakfast, once daily. Induction chemotherapy was initiated within
24 h of administering the first dose of febuxostat. Japanese
insurance coverage approved the following administration schedule
for febuxostat for patients with gout and hyperuricemia: 10 mg for
the initiating dose, and 40–60 mg for maintenance doses. The
administration of febuxostat was continued for ≥7 days.
Assessments
The primary endpoint was the reduction of S-UA.
Febuxostat treatment was considered to be successful, and the
patient considered to be a treatment responder, if S-UA levels
decreased to ≤7.5 mg/dl by day 5 of chemotherapy, according to
previous studies (17,18). S-UA and serum creatinine (S-Cr)
levels were determined in-house using a TBA-c16000 automatic
analyzer (Toshiba Medical Systems, Tochigi, Japan) (19). Secondary endpoints included renal
function and adverse events. Renal function was determined by S-Cr
and estimated glomerular filtration rate (eGFR). Adverse events
were evaluated for 10 days from the initiation of febuxostat
according to the National Cancer Institute Common Terminology
Criteria for Adverse Events 4.0 (May 28, 2009).
Statistical analyses
All statistical analyses were performed using
Microsoft Excel 2007 software (Microsoft Corporation, Redmond, WA,
USA). All graphs were generated using GraphPad Prism software
(Version 5.0; GraphPad Software, Inc., San Diego, CA, USA).
Results
Patient characteristics
Seven patients, who were admitted to the University
of Fukui Hospital between September 2011 and May 2012, were
evaluated retrospectively (Table
I). The median patient age was 70 years (range, 36–79 years),
and the study population consisted of six males and one female. The
diagnoses of the patients included diffuse large B-cell lymphoma
(n=2), acute myeloid leukemia (n=3), chronic myelomonocytic
leukemia (n=1), and chronic myeloid leukemia (n=1). One patient
(patient no. 6) already exhibited clinical TLS, and all other
patients were at an intermediate risk of developing TLS (Table I).
 | Table IPatient’s characteristics. |
Table I
Patient’s characteristics.
| Patient no. | Age (years) | Gender | Diagnosis | WBC (/μl) | LDH (U/l) | S-UA (mg/dl) | S-Cr (mg/dl) | S-Ca (mg/dl) | S-K (mg/dl) | S-P (mg/dl) | eGFR (ml/min/1.73
m2) | LTLS/CTLS |
|---|
| 1 | 53 | M | DLBCL | 11,500 | 159 | 7.6 | 0.89 | 8.8 | 4.1 | 2.8 | 87 | (−)/(−) |
| 2 | 59 | M | AML | 25,400 | 356 | 1.9 | 0.62 | 8.4 | 3.6 | 3.2 | 101 | (−)/(−) |
| 3 | 74 | M | AML | 42,100 | 745 | 5.6 | 0.70 | 8.8 | 3.5 | 3.9 | 83 | (−)/(−) |
| 4 | 36 | M | AML | 3,200 | 195 | 4.2 | 0.88 | 8.3 | 3.5 | 2.9 | 78 | (−)/(−) |
| 5 | 76 | M | DLBCL | 5,800 | 237 | 6.2 | 1.01 | 9.2 | 4.7 | 3.7 | 55 | (−)/(−) |
| 6 | 79 | F | CMML | 60,700 | 302 | 9.6 | 1.38 | 9.5 | 3.8 | 4.6 | 29 | (+)/(+) |
| 7 | 70 | M | CML | 347,000 | 829 | 9.0 | 1.04 | 9.3 | 4.6 | 3.7 | 55 | (−)/(−) |
UA-associated parameters
The parameters associated with UA, which included
the urinary UA excretion and UA clearance, were determined in the
three patients with hyperuricemia (patient nos. 1, 6 and 7)
(Table II). Based on the criteria
for the Management of Hyperuricemia and Gout (16), four cases were classified as UA
overproducers and three cases were classified as UA underexcretors
(Table II). A previous study
revealed that half of the patients with hyperuricemia with
hematological malignancies were of the underexcretion type,
although S-UA should still increase as a consequence of
overproduction through tumor lysis (20).
 | Table IIParameters associated with uric
acid. |
Table II
Parameters associated with uric
acid.
| Patient | U-UA (mg/kg/h) | CUA (ml/min) | Type of
hyperuricemia |
|---|
| 1 | 0.36 | 5.1 | Under-excretion |
| 2 | 0.52 | 35.2 | Overproduction |
| 3 | 0.86 | 15.1 | Overproduction |
| 4 | 0.25 | 11.0 | Under-excretion |
| 5 | 0.24 | 3.8 | Under-excretion |
| 6 | 0.80 | 8.3 | Overproduction |
| 7 | 2.40 | 34.0 | Overproduction |
Therapeutic efficacy
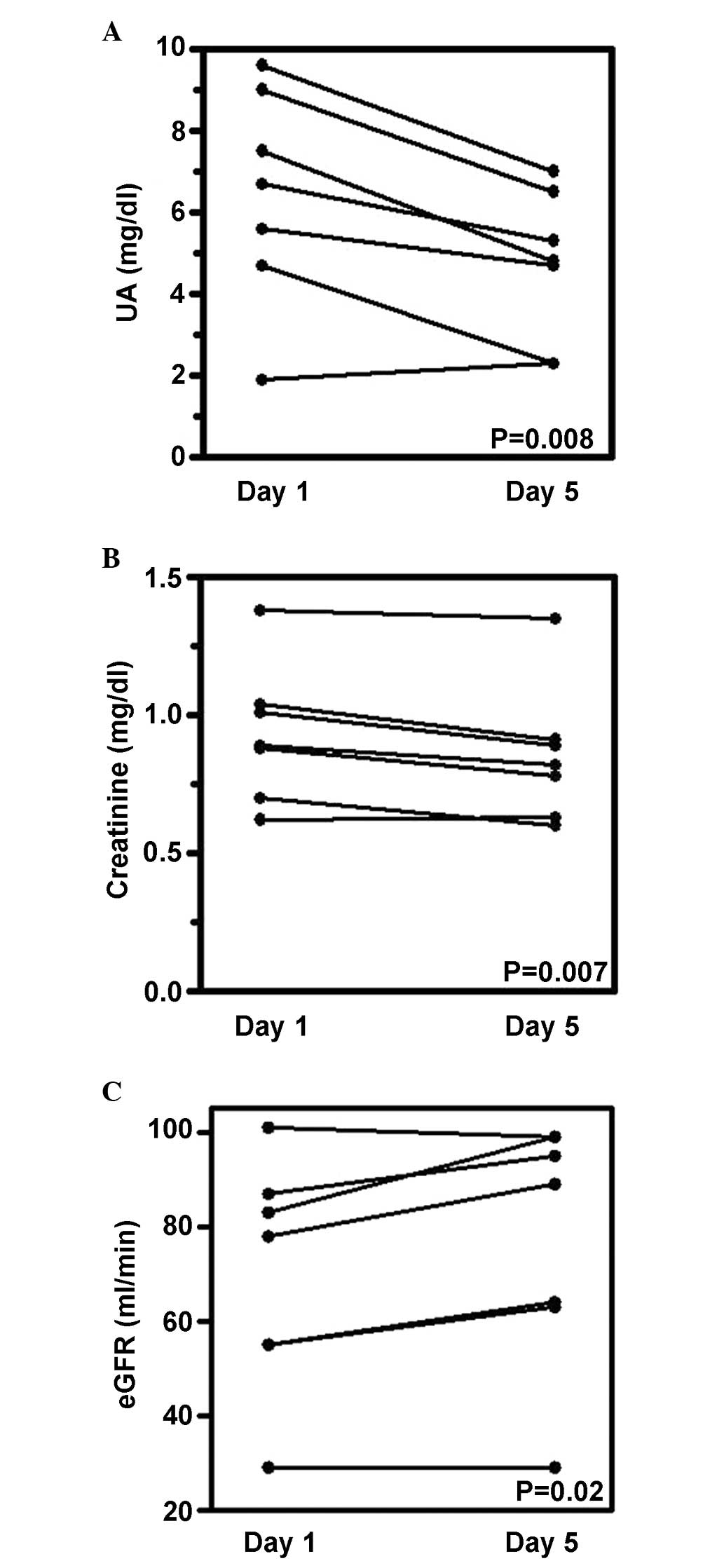
The primary estimate of the present retrospective
study was the S-UA reduction to ≤7.5 mg/dl by day 5 of chemotherapy
treatment. While the S-UA level at base line was 6.4±2.6 mg/dl,
S-UA on day 5 was 4.7±1.8 mg/dl (27% reduction; paired t-test,
P=0.008) (Fig. 1A). All patients
achieved S-UA levels ≤7.5 mg/dl by day 5. One patient (patient no.
2) had very low S-UA levels and the UA level was almost unchanged
(from 1.9 to 2.3 mg/dl) (Fig. 1A).
Assessment of the UA-associated parameters in this patient
indicated the overproduction of UA, which was compensated for by
simultaneous urinary UA overexcretion (Table II). This suggested that the effect
of febuxostat may be masked by UA overexcretion. In terms of the
secondary endpoints, S-Cr levels decreased from 0.93±0.25 to
0.85±0.25 mg/dl (paired t-test, P=0.007) (Fig. 1B). eGFR values increased from
69.7±24.5 to 76.9±26.2 ml/min (paired t-test, P=0.02) (Fig. 1C). No patient exhibited progression
of TLS. These results suggested that febuxostat successfully
controlled S-UA and improved renal function during
chemotherapy.
Adverse events
All adverse events that occurred during the 10 days
of febuxostat administration are summarized in Table III. Grade 3/4 events, including
cytopenia and sepsis, may be attributed to the concomitant
administration of anticancer agents. Nausea and decreased appetite
may also be as a result of the chemotherapy treatment. There were
no indicators of grade 3/4 hepatic or renal dysfunction or attacks
of gout; therefore, no severe adverse events specific to febuxostat
appeared to occur.
 | Table IIIAdverse events following the
administration of febuxostat. |
Table III
Adverse events following the
administration of febuxostat.
| Adverse event | All grades, n | Grade 3/4, n |
|---|
| Constipation | 5 | 0 |
| Hyperglycemia | 4 | 1 |
| White blood cells
decreased | 3 | 3 |
| Neutrophil count
decreased | 3 | 3 |
|
Thrombocytopenia | 3 | 3 |
| Diarrhea | 3 | 0 |
| Decreased
appetite | 2 | 0 |
| Nausea | 2 | 0 |
| Bilirubin
increased | 2 | 0 |
| Hemoglobin
decreased | 1 | 1 |
| Sepsis | 1 | 1 |
| Increased aspartate
aminotransferase | 1 | 0 |
| Increased alanine
aminotransferase | 1 | 0 |
| Fatigue | 1 | 0 |
| Peripheral
edema | 1 | 0 |
| Hyponatremia | 1 | 0 |
| Vomiting | 1 | 0 |
| Stroke | 1 | 0 |
Discussion
Previously published guidelines (2,3) have
stated that allopurinol and the recombinant uricase, rasburicase,
are appropriate treatments for patients at intermediate and high
risk of developing TLS. The hypoxanthine analog, allopurinol, is a
traditional, competitive xanthine oxidase inhibitor that has been
used for >40 years for the treatment of gout and hyperuricemia.
The increase in S-UA observed in patients with TLS usually results
from the catabolism of purine nucleic acid released from cancer
cells upon cell lysis. It is considered to be effective in blocking
xanthine oxidase, which would usually convert purine metabolites
(hypoxanthine and xanthine) to UA, thereby reducing its production
(2,3,8).
Allopurinol, however, is not an ideal therapeutic agent (8), as it has slow onset of action (24–72
h) and is ineffective against already formed UA (21,22).
Side effects occur in 3% of patients receiving allopurinol
(23–25). Although allopurinol is generally
safe and effective, it can induce life-threatening rashes and/or
severe multisystem allopurinol hypersensitivity syndrome (12,26,27).
Furthermore, the prolonged half-life (14–26 h) of the major
allopurinol active oxidation product, oxypurinol, and its further
prolongation in patients with decreased creatinine clearance
necessitates dose reduction to avoid the side effects and severe
toxicities. This dose reduction can attenuate the UA-lowering
efficacy (12).
Febuxostat is a UA-lowering agent used for the
treatment of patients with gout and hyperuricemia. Febuxostat is a
non-purine-structured xanthine oxidase inhibitor that is thought to
be independent of other enzymes in the purine and pyrimidine
metabolic pathways (13–15). Febuxostat is primarily metabolized
in the liver, and is eliminated by both the hepatic and renal
pathways, equally (13–15,28).
Febuxostat has been extensively studied in clinical trials
involving patients with gout (13–15,29).
In the CONFIRMS trial, 2,269 patients were randomized to 40 or 80
mg febuxostat or 300 mg allopurinol. The endpoints included the
proportion of all subjects with S-UA levels <6.0 mg/dl, and the
proportion of subjects with mild/moderate renal impairment and S-UA
levels <6.0 mg/dl. The study revealed that the UA-lowering
efficacy of febuxostat at an 80 mg dose exceeded the efficacy
produced by treatment with allopurinol. Furthermore, in subjects
with mild/moderate renal impairment, both febuxostat doses were
more effective than and were equally safe as allopurinol (29). As a result of the demonstrated
efficacy and safety, febuxostat was used in the present study as an
alternative to allopurinol in cancer patients at risk of TLS.
The present retrospective study revealed that
febuxostat at 10 mg sustained or reduced S-UA to levels ≤7.5 mg/dl
during chemotherapy. In Japan, the maintenance dose of febuxostat
is 40–60 mg/day, and the starting dose is 10 mg. Therefore,
patients in the present study were given a 10-mg dose, which
despite at this low dose level, achieved a 27% reduction in S-UA
within one week. The use of febuxostat at maintenance doses (40–60
mg) has a greater potential for a higher efficacy in the control of
hyperuricemia associated with TLS.
A low starting dose of febuxostat is often
prescribed to reduce the risk of acute flares of gout associated
with a steep decrease in S-UA. Attacks are triggered by urate
crystal mobilization with a lowering of S-UA by febuxostat. A more
abrupt lowering with febuxostat would induce gout more frequently
as compared with allopurinol (12).
Hyperuricemia in cancer patients at a risk for TLS have not been
reported to commonly experience flares of gout (20,30).
This may be attributed to the short duration of a hyperuricemic
state in these patients, which is unlikely to generate urate
crystal deposition.
In conclusion, the present retrospective study has
demonstrated that febuxostat was safe and effective in preventing
or reversing hyperuricemia in patients with hematological
malignancies, who were undergoing chemotherapy and at an
intermediate risk of developing TLS. A prospective study is
undergoing to confirm the efficacy of febuxostat for the prevention
and treatment of hyperuricemia in cancer patients at risk of
TLS.
Abbreviations:
|
TLS
|
tumor lysis syndrome
|
|
UA
|
uric acid
|
|
S-UA
|
serum uric acid
|
|
S-Cr
|
serum creatinine
|
|
eGFR
|
estimated glomerular filtration
rate
|
References
|
1
|
Cohen LF, Balow JE, Magrath IT, Poplack DG
and Ziegler JL: Acute tumor lysis syndrome. A review of 37 patients
with Burkitt’s lymphoma. Am J Med. 68:486–491. 1980.
|
|
2
|
Coiffier B, Altman A, Pui CH, Younes A and
Cairo MS: Guidelines for the management of pediatric and adult
tumor lysis syndrome: an evidence-based review. J Clin Oncol.
26:2767–2778. 2008.
|
|
3
|
Cairo MS, Coiffier B, Reiter A and Younes
A; TLS Expert Panel. Recommendations for the evaluation of risk and
prophylaxis of tumour lysis syndrome (TLS) in adults and children
with malignant diseases: an expert TLS panel consensus. Br J
Haematol. 149:578–586. 2010.
|
|
4
|
Howard SC, Jones DP and Pui CH: The tumor
lysis syndrome. N Engl J Med. 364:1844–1854. 2011.
|
|
5
|
Firwana BM, Hasan R, Hasan N, Alahdab F,
Alnahhas I, Hasan S and Varon J: Tumor lysis syndrome: a systematic
review of case series and case reports. Postgrad Med. 124:92–101.
2012.
|
|
6
|
Cairo MS and Bishop M: Tumour lysis
syndrome: new therapeutic strategies and classification. Br J
Haematol. 127:3–11. 2004.
|
|
7
|
Hande KR and Garrow GC: Acute tumor lysis
syndrome in patients with high-grade non-Hodgkin’s lymphoma. Am J
Med. 94:133–139. 1993.
|
|
8
|
Will A and Tholouli E: The clinical
management of tumour lysis syndrome in haematological malignancies.
Br J Haematol. 154:3–13. 2011.
|
|
9
|
Massey V, Komai H, Palmer G and Elion GB:
On the mechanism of inactivation of xanthine oxidase by allopurinol
and other pyrazolo[3,4-d]pyrimidines. J Biol Chem. 245:2837–2844.
1970.
|
|
10
|
Faruque LI, Ehteshami-Afshar A, Wiebe N,
Tjosvold L, Homik J and Tonelli M: A systematic review and
meta-analysis on the safety and efficacy of febuxostat versus
allopurinol in chronic gout. Semin Arthritis Rheum. 43:367–375.
2013.
|
|
11
|
Bruce SP: Febuxostat: a selective xanthine
oxidase inhibitor for the treatment of hyperuricemia and gout. Ann
Pharmacother. 40:2187–2194. 2006.
|
|
12
|
Schumacher HR Jr, Becker MA, Wortmann RL,
et al: Effects of febuxostat versus allopurinol and placebo in
reducing serum urate in subjects with hyperuricemia and gout: a
28-week, phase III, randomized, double-blind, parallel-group trial.
Arthritis Rheum. 59:1540–1548. 2008.
|
|
13
|
Edwards NL: Febuxostat: a new treatment
for hyperuricaemia in gout. Rheumatology (Oxford). 48:ii15–ii19.
2009.
|
|
14
|
Burns CM and Wortmann RL: Gout
therapeutics: new drugs for an old disease. Lancet. 377:165–177.
2011.
|
|
15
|
Garcia-Valladares I, Khan T and Espinoza
LR: Efficacy and safety of febuxostat in patients with
hyperuricemia and gout. Ther Adv Musculoskelet Dis. 3:245–253.
2011.
|
|
16
|
The guideline revising committee of
Japanese Society of Gout and Nucleic Acid Metabolism. Digest of
Guideline for the management of hyperuricemia and gout. 2nd
edition. Gout Nucleic Acid Metabol. 34. pp. 107–143. 2010, (In
Japanese).
|
|
17
|
Cortes J, Moore JO, Maziarz RT, et al:
Control of plasma uric acid in adults at risk for tumor Lysis
syndrome: efficacy and safety of rasburicase alone and rasburicase
followed by allopurinol compared with allopurinol alone-results of
a multicenter phase III study. J Clin Oncol. 28:4207–4213.
2010.
|
|
18
|
Kikuchi A, Kigasawa H, Tsurusawa M, et al:
A study of rasburicase for the management of hyperuricemia in
pediatric patients with newly diagnosed hematologic malignancies at
high risk for tumor lysis syndrome. Int J Hematol. 90:492–500.
2009.
|
|
19
|
Yamauchi T, Negoro E, Lee S, et al: A high
serum uric acid level is associated with poor prognosis in patients
with acute myeloid leukemia. Anticancer Res. 33:3947–3951.
2013.
|
|
20
|
Inai K, Tsutani H and Ueda T:
Hyperuricemia associated with hematological malignancies. Gout and
Nucleic Acid Metabolism. 23:181–186. 1999.(In Japanese).
|
|
21
|
de Bont JM and Pieters R: Management of
hyperuricemia with rasburicase review. Nucleosides Nucleotides
Nucleic Acids. 23:1431–1440. 2004.
|
|
22
|
Rampello E, Fricia T and Malaguarnera M:
The management of tumor lysis syndrome. Nat Clin Pract Oncol.
3:438–447. 2006.
|
|
23
|
Andreoli SP, Clark JH, McGuire WA and
Bergstein JM: Purine excretion during tumor lysis in children with
acute lymphocytic leukaemia receiving allopurinol: relationship to
acute renal failure. J Pediatr. 109:292–298. 1986.
|
|
24
|
Navolanic PM, Pui CH, Larson RA, et al:
Elitek-rasburicase: an effective means to prevent and treat
hyperuricemia associated with tumor lysis syndrome, a Meeting
Report, Dallas, Texas, January, 2002. Leukaemia. 17:499–514.
2003.
|
|
25
|
Cheson BD and Dutcher BS: Managing
malignancy-associated hyperuricemia with rasburicase. J Support
Oncol. 3:117–124. 2005.
|
|
26
|
Arellano F and Sacristan JA: Allopurinol
hypersensitivity syndrome: a review. Ann Pharmacother. 27:337–343.
1993.
|
|
27
|
Hande KR, Noone RM and Stone WJ: Severe
allopurinol toxicity: Description and guidelines for prevention in
patients with renal insufficiency. Am J Med. 76:47–56. 1984.
|
|
28
|
Khosravan R, Grabowski BA, Mayer MD, Wu
JT, Joseph-Ridge N and Vernillet L: The effect of mild and moderate
hepatic impairment on pharmacokinetics, pharmacodynamics and safety
of febuxostat, a novel non-purine selective inhibitor of xanthine
oxidase. J Clin Pharmacol. 46:88–102. 2006.
|
|
29
|
Becker MA, Schumacher HR, Espinoza LR,
Wells AF, MacDonald P, Lloyd E and Lademacher C: The urate-lowering
efficacy and safety of febuxostat in the treatment of the
hyperuricemia of gout: the CONFIRMS trial. Arthritis Res Ther.
12:R632010.
|
|
30
|
Vadhan-Raj S, Fayad LE, Fanale MA, et al:
A randomized trial of a single-dose rasburicase versus five-daily
doses in patients at risk for tumor lysis syndrome. Ann Oncol.
23:1640–1645. 2012.
|