Introduction
Adenoid cystic carcinoma (ACC) is the second most
frequent salivary malignancy, accounting for 10–18% of all salivary
malignancies (1,2). Clinical characteristics for ACC
include invasive local growth, frequent local recurrence and
distant metastasis. The conventional treatment for ACC is radical
surgery with postoperative radiotherapy. Despite a relatively high
short-term survival rate, the likelihood of recurrence and
metastasis is high. The 5- and 10-year overall survival rates for
patients without distant metastasis are 85.4 and 67.4%,
respectively. The 5- and 10-year overall survival rates for
patients with distant metastasis are 69.1 and 45.7%, respectively
(3). Further investigation of the
possible biomarkers involved in the progression and metastasis of
salivary ACC (SACC) may help identify novel therapeutic strategies
and improve the long-term outcome of the disease.
The tumor-associated calcium signal transducer
(TACSTD) gene family consists of two highly related genes, TACSTD2
and TACSTD1. TACSTD1 (also known as Trop1 or EpCAM) was originally
identified as a marker for epithelial carcinomas due to aberrant
expression in various tumors (4).
TACSTD2 (also known as Trop2, GA733-1, M1S1 or EGP-1) is a 36-kDa
type-I transmembrane glycoprotein that was originally identified in
human placental trophoblasts, and may function to transduce an
intracellular calcium signal (5,6).
TACSTD1 and 2 share ~50% sequence identity and both contain a
thyroglobulin repeat domain region (7). The present study speculated that
TACSTD2 may also function in tumor formation and development. It
has been previously shown that the cytoplasmic tail of TACSTD2 is
essential for signaling, and phosphorylation of serine 303
regulated by protein kinase C can control the tumor growth
stimulatory capacity of TACSTD2 (8,9).
TACSTD2 overexpression has been reported in numerous
human cancers, such as colorectal, ovarian, pancreatic, cervical,
gastric, bile duct cancer and squamous cell carcinoma of the oral
cavity, as compared with the corresponding normal tissue, and the
expression level has been shown to correlate with the poor patient
prognosis (10–16). The expression and function of
TACSTD2 in human SACC has not been previously reported.
The present study aimed to retrospectively
investigate TACSTD2 antigen expression by immunohistochemistry, in
paraffin-embedded primary tumor tissue samples from a series of
consecutive SACC patients (n=81). The correlation of TACSTD2
expression with clinicopathological variables and survival analysis
was evaluated.
Materials and methods
Patient material and tumor samples
The present study was approved by the Institutional
Ethics Committee of Sichuan University (Chengdu, China) and consent
was obtained from the patients. Paraffin-embedded tissue specimens,
including 81 SACCs, were randomly selected from the West China
College of Stomatology, Sichuan University between January 1991 and
December 1996. All of these recruited patients underwent radical
surgery without preoperative chemotherapy or radiotherapy, and the
collected samples were reconfirmed by two pathologists. Data from
the patient follow-up and clinicopathological characteristics were
collected from the database of West China College of Stomatology
and from telephone interviews. The recurrence and metastasis during
the follow-up period was confirmed by radiography and pathology
biopsy. Of the 81 patients, there were 37 males and 44 females, and
the median age was 47 (range, 20–81 years). The overall survival
(OS) was calculated from the first radical surgery until death due
to any cause. The patients who had survived at the time of the last
follow-up visit were lost to further follow-up. The details of the
patient data are listed in Table I.
Five normal salivary gland tissue samples were selected to be used
as controls. The clinical staging of the patients was determined
according to the International Union against Cancer TNM
Classification of Malignant Tumors (17). The histologic subtypes of the
samples were decided on the basis of the World Health
Organization’s histologic classification of salivary gland tumors
(18).
 | Table IExpression of TACSTD2 in salivary
adenoid cystic carcinoma and its association with
clinicopathological variables. |
Table I
Expression of TACSTD2 in salivary
adenoid cystic carcinoma and its association with
clinicopathological variables.
| | TACSTD2
expression | |
|---|
| |
| |
|---|
| n | N, n (%) | W, n (%) | M, n (%) | S, n (%) | P-value |
|---|
| Gender | 81 | 18 (22.2) | 27 (33.3) | 20 (24.7) | 16 (19.7) | 0.582a |
| Male | 37 | 10 (27.0) | 11 (29.7) | 9 (24.3) | 7 (18.9) | |
| Female | 44 | 8 (18.2) | 16 (36.4) | 11 (25.0) | 9 (20.5) | |
| Age (years) | | | | | | 0.328a |
| ≤50 | 46 | 12 (26.1) | 16 (34.8) | 9 (19.6) | 9 (19.6) | |
| >50 | 35 | 6 (17.1) | 11 (31.4) | 11 (31.4) | 7 (19.8) | |
| Histological
subtype | | | | | | 0.289b |
| Tubular | 32 | 6 (18.8) | 14 (43.8) | 7 (21.9) | 5 (15.6) | |
| Cribriform | 28 | 9 (32.1) | 8 (28.6) | 5 (17.9) | 6 (21.4) | |
| Solid | 21 | 3 (14.3) | 5 (23.8) | 8 (38.1) | 5 (23.8) | |
| TNM Stage | | | | | | 0.020a |
| I+II | 32 | 13 (40.6) | 8 (25.0) | 6 (18.8) | 5 (15.6) | |
| III+IV | 49 | 5 (10.2) | 19 (38.8) | 14 (28.6) | 11 (22.4) | |
| Perineural
invasion | | | | | | 0.053a |
| Negative | 41 | 13 (34.2) | 11 (28.9) | 8 (21.1) | 6 (15.8) | |
| Positive | 40 | 5 (11.6) | 16 (37.2) | 12 (27.9) | 10 (23.3) | |
| Local recurrence | | | | | | 0.002a |
| Negative | 35 | 12 (34.3) | 14 (40.0) | 6 (17.1) | 3 (8.6) | |
| Positive | 46 | 6 (13.0) | 13 (28.3) | 14 (30.4) | 13 (28.3) | |
| Distant
metastasis | | | | | | 0.001a |
| Negative | 52 | 15 (28.8) | 20 (38.5) | 12 (23.1) | 5 (9.6) | |
| Positive | 29 | 3 (10.3) | 7 (24.1) | 8 (27.6) | 11 (37.9) | |
Immunohistochemical staining
Formalin-fixed, paraffin-embedded tissues were
consecutively cut into 4-μm sections and transferred onto the
silanized glass slides. The slides were dewaxed in xylene and
rehydrated through a graded alcohol series. Endogenous peroxidase
activity was blocked with 3% hydrogen peroxide solution for 20 min
at room temperature. The slides were heated to high-temperature in
0.1 M citrate buffer (pH 6.0) for 4 min to repair the antigen and
then blocked with 1% bovine serum albumen Tris-HCl buffer. The
slides were incubated overnight at 4°C with a polyclonal goat
antibody against the human TACSTD2 extracellular domain (R&D
Systems, Inc., Minneapolis, MN, USA) at a 1:100 dilution. A
biotinylated polyclonal rabbit anti-goat antibody (Vector Labs,
Burlingame, CA, USA) at a 1:200 dilution was applied to reveal the
primary antibody. Horseradish peroxidase-streptavidin (Dako,
Glostrup, Denmark) and diaminobenzidine were used as chromogens.
All the sections were counter-stained with hematoxylin and then
dehydrated and mounted. Negative controls were sections that had
been incubated with phosphate-buffered saline instead of the
primary antibody.
Immunohistochemical evaluation
The level of immunoreactivity was separately
evaluated by two independent pathologists without any prior
information of the clinical data. Samples with disagreement in the
immunohistochemical score were rescored under the discussion of the
two observers. A scoring method according to the percentage of
positive tumor cells and the intensity of the staining was applied.
For intensity: 0, negative; 1, weak; 2, moderate; 3, strong; for
the percentage: scored 0, <1%; 1, ≥1% to <10%; 2, ≥10% to
<50%; 3, ≥50%. The final score was obtained by multiplying the
two scores: score 0, negative; scores 1 and 2, weakly positive;
scores 3 and 4, moderately positive; scores 6 and 9, strongly
positive.
Statistical analysis
The correlation of TACSTD2 expression with
clinicopathological variables was evaluated by Pearson’s
χ2 test or Fisher’s exact test. The survival curves were
plotted using the Kaplan-Meier method, and the significance of
differences in the OS rate was tested by the log-rank test. The
parameters of prognostic significance found by univariate analyses
were verified in a multivariate Cox proportional-hazards regression
model. P<0.05 was considered to indicate a statistically
significant difference. Statistical analysis was performed using
SPSS statistical software, version 18.0 (SPSS, Inc., Chicago,
IL).
Results
TACSTD2 expression in SACC specimens and
association with clinicopathological parameters
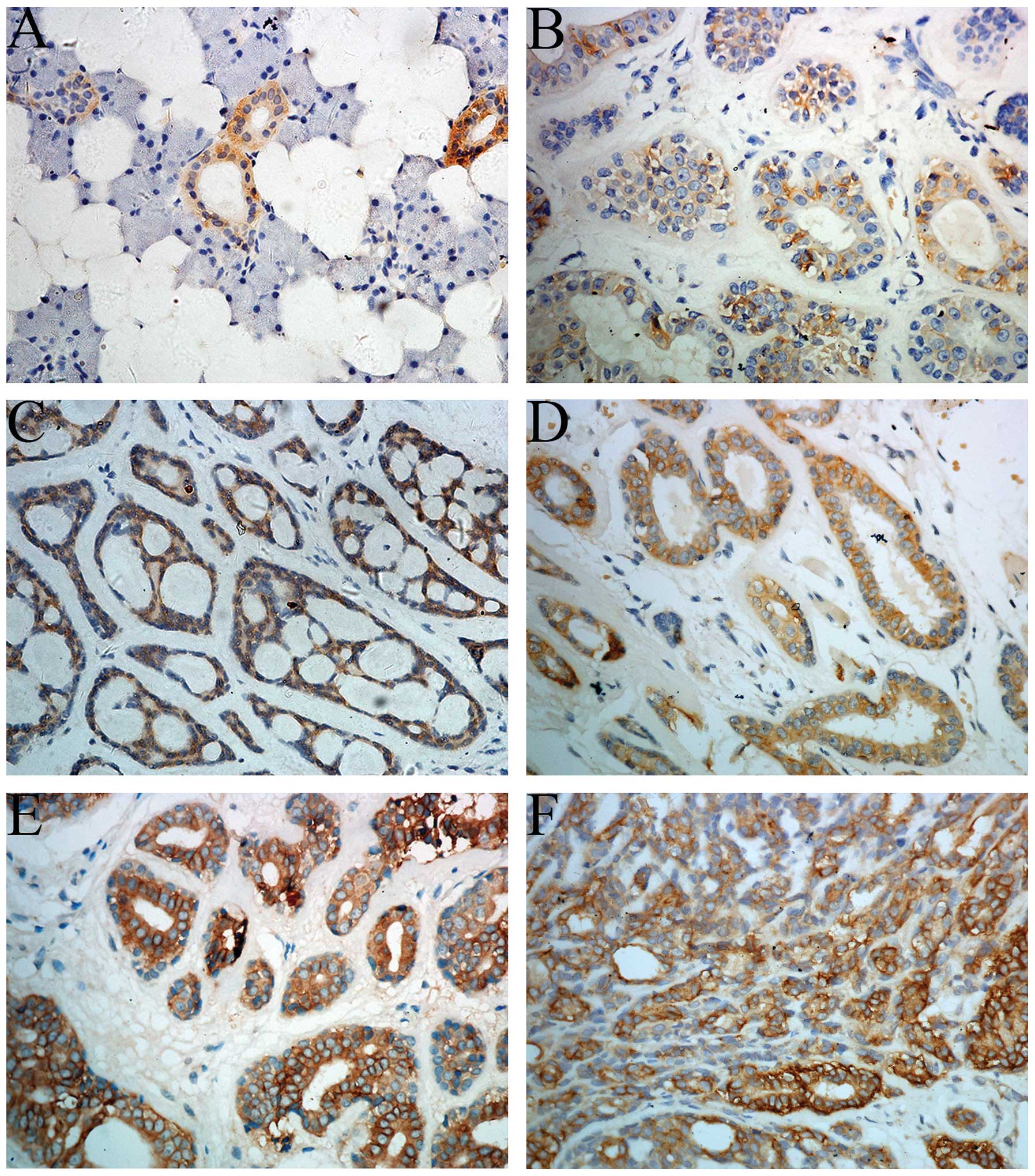
Expression of TACSTD2 in normal salivary gland
tissue was not detected except in some ductal epithelial cells
(Fig. 1A). The positive staining of
TACSTD2 primarily occurred on the cell membrane. Positive
expression of TACSTD2 was noted in 63/81 cases (Fig. 1B–F). By subgroup analysis, negative
expression was observed in 18 (22.2%) cases, weak expression in 27
(33.3%) cases, moderate expression in 20 (24.7%) cases and strong
expression in 16 (19.7%) cases. The correlations between TACSTD2
expression and clinicopathological variables are listed in Table I. The statistical analysis
identified that no significant difference was observed between the
TACSTD2 expression level and clinical factors of age, gender,
histological subtype and perineural invasion; however, TNM stage
(P=0.020), local recurrence (P=0.002) and distant metastasis
(P=0.001) were noted to be associated with a higher expression of
TACSTD2.
Survival analysis
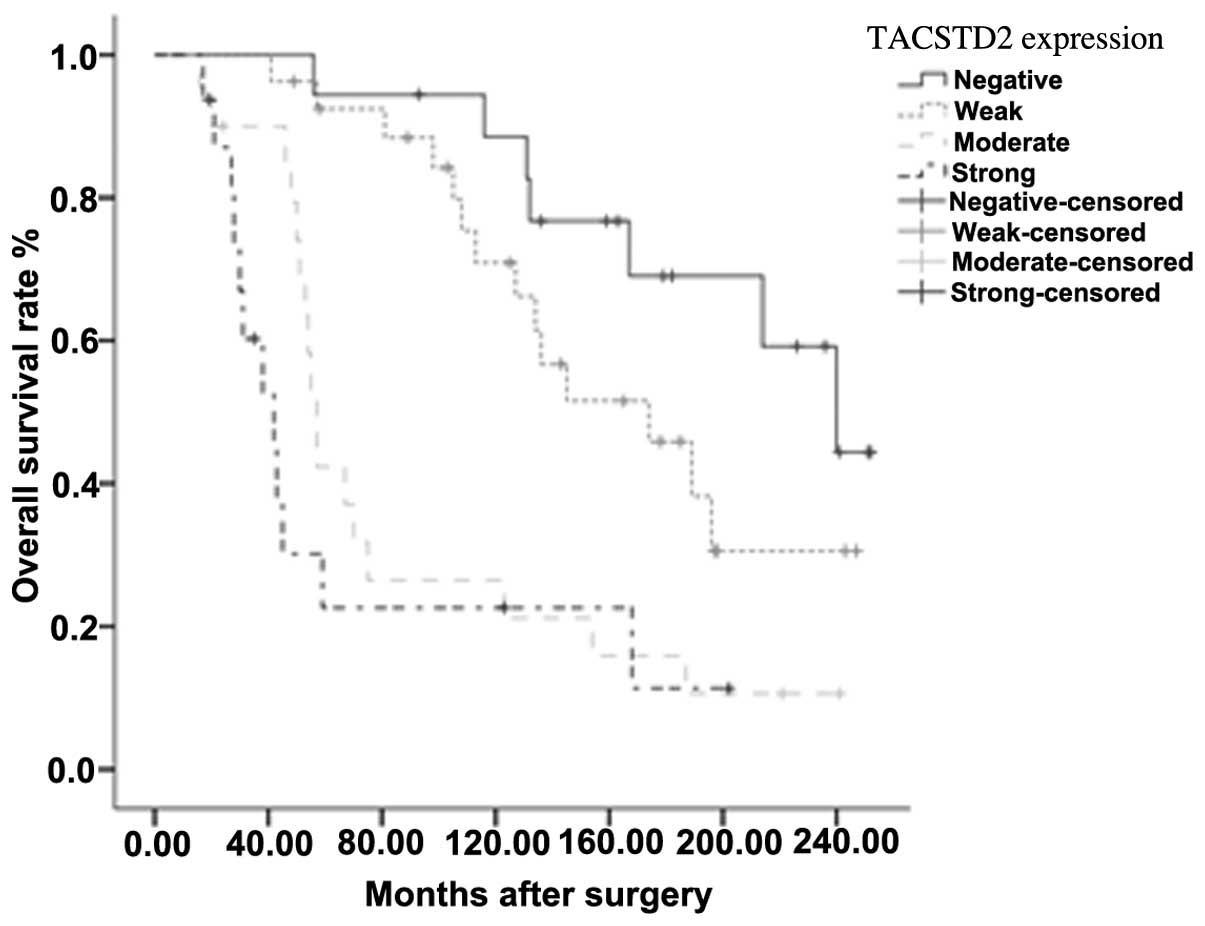
The Kaplan-Meier method was used to perform a
univariate analysis. A survival curve, according to the TACSTD2
expression level, was plotted. The OS rates decreased with
increasing TACSTD2 expression level (Fig. 2). The variables with a significant
difference, as tested by log-rank analysis, were histological
subtype (P<0.001), TNM stage (P=0.003), local recurrence
(P<0.001), distant metastasis (P<0.001) and TACSTD2
expression (P<0.001). The statistical data are detailed in
Table II. To further assess the
impact of TACSTD2 expression on survival, all the statistically
significant variables, including TACSTD2 expression, were subjected
to the Cox proportional-hazards regression model. TACSTD2,
histological subtype and distant metastasis were identified to be
independent prognostic indicators for overall survival (Table II).
 | Table IIUnivariate and multivariate analysis
of clinicopathological variables and Trop2 expression in relation
to overall survival in patients with salivary adenoid cystic
carcinoma. |
Table II
Univariate and multivariate analysis
of clinicopathological variables and Trop2 expression in relation
to overall survival in patients with salivary adenoid cystic
carcinoma.
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|---|
| Risk factors | P-value | HR | 95% CI | P-value |
|---|
| Overall survival |
| Age
(≤50/>50) | 0.134 | Not included in
model | |
| Gender
(Male/Female) | 0.549 | Not included in
model | |
| Histological subtype
(C, S/T) | <0.001 | 2.610 | 1.159–5.876 | 0.020 |
| TNM Stage
(I+II/III+IV) | 0.003 | 1.802 | 0.871–3.731 | 0.112 |
| Perineural invasion
(N/P) | 0.180 | Not included in
model | |
| Local recurrence
(N/P) | <0.001 | 1.427 | 0.726–2.807 | 0.302 |
| Distant metastasis
(N/P) | <0.001 | 3.163 | 1.379–7.258 | 0.007 |
| TACSTD2 Expression
(N/W/M/S) | <0.001 |
<0.001b | | |
| TACSTD2 (W/N) | | 1.521 | 0.584–3.960 | 0.390 |
| TACSTD2 (M/N) | | 3.791 | 1.436–10.010 | 0.007 |
| TACSTD2 (S/N) | | 11.193 | 3.953–31.690 | <0.001 |
Discussion
Despite numerous advances in the diagnosis and
treatment of SACC over the past 20 years, a high frequency of local
recurrence and distant metastasis has remained. Treatments based on
conventional clinicopathological parameters are valuable but are
not satisfactory in improving the OS. Specific gene-targeting
therapy may be an alternative option to consider for patients with
SACC. Investigations of cell surface markers expressed in tumor
cells could be a possible method to identify novel gene
targets.
TACSTD2, formed by the retropositioning of EpCAM via
an mRNA intermediate, is located on chromosome 1p32 and encodes an
intronless gene product (19).
TACSTD2 protein is a type I membrane protein conducting calcium
signaling (5). Retrospective
studies have found that overexpression of TACSTD2 predicts poor
prognosis in the majority of human cancers. Serological
identification of TACSTD2 in patients with esophageal squamous cell
carcinoma suggested that the TACSTD2 antigen may be a valuable
serum tumor marker (20). The
bicistronic cyclin D1-TACSTD2 mRNA was isolated and was shown to be
a potent oncogene both in vitro and in vivo 21). The
expression of the chimeric mRNA was observed to improve the
stability of cyclin D1 and enhance the proliferation of the
expressing cells (21). The
signaling mechanism of TACSTD2 involved in tumor pathogenesis was
first explained by activating the extracellular signal regulated
kinase (ERK)/mitogen-activated protein kinase pathway, and cyclin
D1 was activated as an important downstream factor of the pathway
(22). A detailed TACSTD2 signaling
network in cancer growth was further elucidated by Guerra et
al (21); TACSTD2 upregulation
was shown to subsequently drive the expression and activation of
CREB1, Jun, NF-kB, Rb, STAT1 and STAT3 through induction of the
cyclin D1 and ERK/MEK pathways (8).
Chemoresistance has been shown in cell lines overexpressing cyclin
D1, which suggested that cyclin D1 may contribute to
chemoresistance (23). Recent
studies have shown that TACSTD2 has a critical role in the
metastasis of prostate cancers by modulating β1 integrin function,
and activation of PAK4 induced by TACSTD2 was observed in the
experiment (24,25).
Immunotherapeutic agents against EpCAM, the analog
of TACSTD2, have produced promising results (26,27).
Furthermore, hRS7, a human monoclonal anti-TACSTD2 antibody has
been used in endometrial endometrioid carcinoma (EEC). Cases of EEC
overexpressing TACSTD2 were shown to be highly sensitive to
hRS7-mediated cytotoxicity in vitro (28). Similar results were achieved in
uterine and ovarian carcinosarcomas (29,30).
In addition, hRS7 also showed a significant therapeutic advantage
in an in vivo breast cancer model (31,32).
To the best of our knowledge, the present study is
the first to analyze the expression, prognostic value and clinical
significance of TACSTD2 in 81 patients with SACC. The results were
consistent with the previously known functions of TACSTD2 in tumor
development and metastasis. The expression of TACSTD2 was
significantly associated with tumor TNM stage (P=0.020), local
recurrence (P=0.002) and distant metastasis (P=0.001). By
multivariate analysis, TACSTD2 was shown to be an independent
prognostic indicator of SACC; however, for the inherent
retrospective analyses limitations, overexpression of TACSTD2 as
the prognostic indicator needs to be validated in larger
prospective studies. In conclusion, TACSTD2 is a surface antigen
that is overexpressed in various epithelial cancers and may be a
novel therapeutic target.
References
|
1
|
Li LJ, Li Y, Wen YM, et al: Clinical
analysis of salivary gland tumor cases in West China in past 50
years. Oral Oncol. 44:187–192. 2008.
|
|
2
|
Spiro RH: Salivary neoplasms: overview of
a 35-year experience with 2,807 patients. Head Neck Surg.
8:177–184. 1986.
|
|
3
|
Gao M, Hao Y, Huang MX, et al:
Clinicopathological study of distant metastases of salivary adenoid
cystic carcinoma. Int J Oral Maxillofac Surg. 42:923–928. 2013.
|
|
4
|
Trzpis M, McLaughlin PM, de Leij LM and
Harmsen MC: Epithelial cell adhesion molecule: more than a
carcinoma marker and adhesion molecule. Am J Pathol. 171:386–395.
2007.
|
|
5
|
Ripani E, Sacchetti A, Corda D and Alberti
S: Human Trop-2 is a tumor-associated calcium signal transducer.
Int J Cancer. 76:671–676. 1998.
|
|
6
|
Lipinski M, Parks DR, Rouse RV and
Herzenberg LA: Human trophoblast cell-surface antigens defined by
monoclonal antibodies. Proc Natl Acad Sci USA. 78:5147–5150.
1981.
|
|
7
|
Linnenbach AJ, Wojcierowski J, Wu SA, et
al: Sequence investigation of the major gastrointestinal
tumor-associated antigen gene family, GA733. Proc Natl Acad Sci
USA. 86:27–31. 1989.
|
|
8
|
Guerra E, Trerotola M, Aloisi AL, et al:
The Trop-2 signalling network in cancer growth. Oncogene.
32:1594–1600. 2013.
|
|
9
|
El Sewedy T, Fornaro M and Alberti S:
Cloning of the murine TROP2 gene: conservation of a PIP2-binding
sequence in the cytoplasmic domain of TROP-2. Int J Cancer.
75:324–330. 1998.
|
|
10
|
Wang J, Day R, Dong Y, et al:
Identification of Trop-2 as an oncogene and an attractive
therapeutic target in colon cancers. Mol Cancer Ther. 7:280–285.
2008.
|
|
11
|
Fong D, Spizzo G, Gostner JM, et al:
TROP2: a novel prognostic marker in squamous cell carcinoma of the
oral cavity. Mod Pathol. 21:186–191. 2008.
|
|
12
|
Fong D, Moser P, Krammel C, et al: High
expression of TROP2 correlates with poor prognosis in pancreatic
cancer. Br J Cancer. 99:1290–1295. 2008.
|
|
13
|
Liu T, Liu Y, Bao X, et al: Overexpression
of TROP2 predicts poor prognosis of patients with cervical cancer
and promotes the proliferation and invasion of cervical cancer
cells by regulating ERK signaling pathway. PloS One.
8:e758642013.
|
|
14
|
Ning S, Guo S, Xie J, Xu Y, Lu X and Chen
Y: TROP2 correlates with microvessel density and poor prognosis in
hilar cholangiocarcinoma. J Gastrointest Surg. 17:360–368.
2013.
|
|
15
|
Mühlmann G, Spizzo G, Gostner J, et al:
TROP2 expression as prognostic marker for gastric carcinoma. J Clin
Pathol. 62:152–158. 2009.
|
|
16
|
Bignotti E, Todeschini P, Calza S, et al:
Trop-2 overexpression as an independent marker for poor overall
survival in ovarian carcinoma patients. Eur J Cancer. 46:944–953.
2010.
|
|
17
|
da Cruz Perez DE, de Abreu Alves F, Nobuko
Nishimoto I, et al: Prognostic factors in head and neck adenoid
cystic carcinoma. Oral Oncol. 42:139–146. 2006.
|
|
18
|
Seifert G and Sobin LH: The World Health
Organization’s Histological Classification of Salivary Gland
Tumors. A commentary on the second edition. Cancer. 70:379–385.
1992.
|
|
19
|
Linnenbach AJ, Seng BA, Wu S, et al:
Retroposition in a family of carcinoma-associated antigen genes.
Mol Cell Biol. 13:1507–1515. 1993.
|
|
20
|
Nakashima K, Shimada H, Ochiai T, et al:
Serological identification of TROP2 by recombinant cDNA expression
cloning using sera of patients with esophageal squamous cell
carcinoma. Int J Cancer. 112:1029–1035. 2004.
|
|
21
|
Guerra E, Trerotola M, Dell’ Arciprete R,
et al: A bicistronic CYCLIN D1-TROP2 mRNA chimera demonstrates a
novel oncogenic mechanism in human cancer. Cancer Res.
68:8113–8121. 2008.
|
|
22
|
Cubas R, Zhang S, Li M, Chen C and Yao Q:
Trop2 expression contributes to tumor pathogenesis by activating
the ERK MAPK pathway. Mol Cancer. 9:2532010.
|
|
23
|
Biliran H Jr, Wang Y, Banerjee S, et al:
Overexpression of cyclin D1 promotes tumor cell growth and confers
resistance to cisplatin-mediated apoptosis in an elastase-myc
transgene-expressing pancreatic tumor cell line. Clin Cancer Res.
11:6075–6086. 2005.
|
|
24
|
Trerotola M, Li J, Alberti S and Languino
LR: Trop-2 inhibits prostate cancer cell adhesion to fibronectin
through the β1 integrin-RACK1 axis. J Cell Physiol. 227:3670–3677.
2012.
|
|
25
|
Trerotola M, Jernigan DL, Liu Q, et al:
Trop-2 promotes prostate cancer metastasis by modulating β(1)
integrin functions. Cancer Res. 73:3155–3167. 2013.
|
|
26
|
de Bono JS, Tolcher AW, Forero A, et al:
ING-1, a monoclonal antibody targeting Ep-CAM in patients with
advanced adenocarcinomas. Clin Cancer Res. 10:7555–7565. 2004.
|
|
27
|
Mosolits S, Markovic K, Frodin JE, et al:
Vaccination with Ep-CAM protein or anti-idiotypic antibody induces
Th1-biased response against MHC class I- and II-restricted Ep-CAM
epitopes in colorectal carcinoma patients. Clin Cancer Res.
10:5391–5402. 2004.
|
|
28
|
Bignotti E, Ravaggi A, Romani C, et al:
Trop-2 overexpression in poorly differentiated endometrial
endometrioid carcinoma: implications for immunotherapy with hRS7, a
humanized anti-trop-2 monoclonal antibody. Int J Gynecol Cancer.
21:1613–1621. 2011.
|
|
29
|
Raji R, Guzzo F, Carrara L, et al: Uterine
and ovarian carcinosarcomas overexpressing Trop-2 are sensitive to
hRS7, a humanized anti-Trop-2 antibody. J Exp Clin Cancer Res.
30:1062011.
|
|
30
|
Varughese J, Cocco E, Bellone S, et al:
Uterine serous papillary carcinomas overexpress human
trophoblast-cell-surface marker (Trop-2) and are highly sensitive
to immunotherapy with hRS7, a humanized anti-Trop-2 monoclonal
antibody. Cancer. 117:3163–3172. 2011.
|
|
31
|
Govindan SV, Stein R, Qu Z, et al:
Preclinical therapy of breast cancer with a radioiodinated
humanized anti-EGP-1 monoclonal antibody: advantage of a
residualizing iodine radiolabel. Breast Cancer Res Treat.
84:173–182. 2004.
|
|
32
|
Lin H, Zhang H, Wang J, et al: A novel
human Fab antibody for Trop2 inhibits breast cancer growth in vitro
and in vivo. Int J Cancer. 34:1239–1249. 2014.
|