Introduction
Polymyositis is a type of inflammatory myopathy that
mostly involves the striated muscles. Typical features of
polymyositis are sub-acute onset, proximal, symmetric muscle
weakness, elevated serum creatine kinase levels and mononuclear
cell infiltrates in the muscle biopsy. The current treatment for
polymyositis includes first-line high-dose steroids and various
conventional second-line treatments, such as azathioprine,
cyclosporine, methotrexate, cyclophosphamide, tacrolimus,
intravenous immunoglobulins, tumor necrosis factor inhibitors,
rituximab and sifalimumab (1). It
has been confirmed that polymyositis is highly associated with
certain malignancies, particularly non-Hodgkin’s lymphomas
(2). Peripheral T-cell lymphoma is
a relatively rare type of non-Hodgkin’s lymphoma, and the majority
of patients have a poor prognosis with frequent relapse and
unfavorable outcome (3).
Hypercalcemia is a common metabolic complication of these
malignancies, which can present as a hypercalcemic crisis in
certain patients with serum calcium levels of >3.5 mmol/l and
requires emergency management (4).
Common symptoms of hypercalcemia include osteoclasia, renal
calculus, ectopic calcification, abdominal groans, psychiatric
moans and electrocardiogram changes. Emergency management is
required to restore the serum calcium level to the normal range
(2.08–2.60 mmol/l) (5). The present
study reports a case of peripheral T-cell lymphoma with
hypercalcemic crisis as a primary symptom accompanied by
polymyositis, in order to improve our present understanding of the
diagnosis and treatment of such diseases.
Case report
A 61-year-old male was admitted to The Second
Affiliated Hospital of Zhejiang University Medical College
(Hangzhou, Zhejiang, China) due to pain and weakness of the lower
limbs that had been present for one year and a recurrent fever that
had persisted for one month. One year previously, the patient had
experienced pain and bilateral weakness of the muscles of the lower
legs, and walking became limited. Several days later, a recurrent
low to moderate fever developed and scattered red spots were
visible on the extensor skin of the lower legs, with no pruritus or
pain upon compression. The skin biopsy revealed erythema nodosum.
The patient was administered 10 mg prednisone three times a day and
the symptoms were attenuated. At 11 months prior to the present
admittance, the pain and weakness of the lower limbs got worse,
with involvement of the thighs and lower legs. Magnetic resonance
imaging, electromyography and a biopsy of the muscle of the right
thigh were performed, which diagnosed polymyositis. The condition
was treated with 80 mg intravenous methylprednisolone per day and
10 mg oral methotrexate per week. Following attenuation of the
symptoms, the dosage of methylprednisolone was gradually
reduced.
Four months prior to admittance, the patient
experienced exacerbation of the weakness in the lower limbs. A
positron emission tomography/computed tomography (PET-CT) scan was
performed two months later, which showed multiple regions of
elevated fluorodeoxyglucose metabolism in the lymph nodes, lungs,
liver, spleen and bones (Fig. 1).
One month after the scan, the patient developed a high fever of
39.2°C, with no chill or cough. This raised temperature did not
decrease subsequent to treatment with antibiotics (intravenous
cefperazone-sulbactam, 3 g every 8 h) and, therefore, 10 mg
methylprednisolone per day was administered to control the
polymyositis and diclofenac potassium (50 mg, every 12 h) was
administered to control the fever. No abnormal cells were found in
bone marrow smears. Two lymph node puncture biopsies were
performed, but the pathology showed granulomatous inflammation in
each sample. Upon admission, the symptoms of nonchalance and
confusion were identified, along with poor orientation and
calculation abilities, and a bad memory. An enlarged lymph node
could be palpated in the right lingual area. The muscles of the
lower limbs were atrophied. The myodynamia of the lower limb was
grade IV and the myodynamia of the upper limbs was grade V,
according to Lovett’s myodynamic grading criterion (6). No other positive signs were found. A
routine blood test showed a white blood cell count of
7.0×109/l (normal range, 4.0–10.0 ×109/l), a
hemoglobin level of 94 g/l (normal range, 110–160 g/l), a platelet
level of 285×109/l (normal range,
100–300×109/l) and a neutrophil count of 80.2% (normal
range, 50–70%). The blood biochemistry results were as follows:
Albumin, 23.4 g/l (normal range, 35.0–52.0 g/l); serum globulin,
45.8 g/l (normal range, 15.0–30.0 g/l); alkaline phosphatase, 1,169
U/l (normal range, 30–120 U/l); γ-glutamyl transpeptidase, 293 U/l
(normal range, 9–64 U/l); aspartate aminotransferase, 59 U/l
(normal level, <35 U/l); lactate dehydrogenase, 280 U/l (normal
range, 140–271 U/l); calcium, 3.87 mmol/l (normal range, 2.08–2.60
mmol/l); blood urea nitrogen (BUN), 13.20 mmol/l (normal range,
2.80–7.20 mmol/l); creatine, 152 μmol/l (normal range, 53–133
μmol/l); uric acid, 749 μmol/l (normal range, 208–428 μmol/l);
β2-microglobulin, 16.55 mg/l (normal range, 1.00–3.00 mg/l); and
C-reactive protein, 100.9 mg/l (normal level, <6.0 mg/l). The
erythrocyte sedimentation rate was 70.00 mm/h (normal level, <20
mm/h), the rheumatoid factor level was 15.8I U/ml (normal level,
<15.0I U/ml), and the tests for antinuclear antibodies (ANA),
anti-neutrophil cytoplasmic antibodies (ANCA), anticardiolipin
antibodies and cyclic citrullinated peptide antibodies were all
negative. The parathyroid hormone (PTH) level was 4.96 pg/ml
(normal range, 15.00–60.00 pg/ml). A hypercalcemic crisis was
diagnosed and a fluid infusion of 20 mg intravenous furosemide, 100
IU intramuscular salmon calcitonin and 10 mg intravenous
dexamethasone per day was administered.
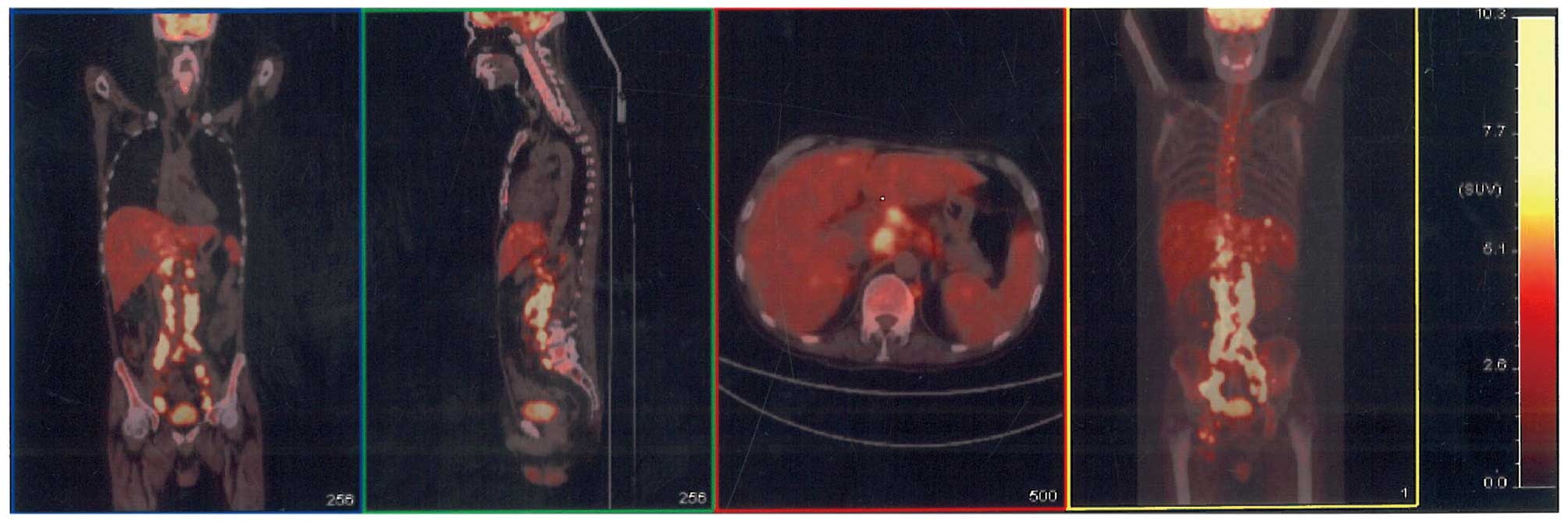 | Figure 1Positron emission tomography-computed
tomography scan of the patient revealing multiple enlarged lymph
nodes in the root of the left side of neck, mediastinum, bilateral
hilus of the lungs, right cardiophrenic angle, hepatic portal area,
peripancreatic area, diaphragm angle, posterior peritoneum,
inter-mesangial area and anterior sacral area, and along the iliac
blood vessels, bilateral iliac fossa, pelvic wall and bilateral
lingual areas. The scans reveal multiple nodules in the bilateral
lungs, an enlarged liver with nodular appearance of the parenchyma,
an enlarged spleen with multiple low-density shadows in the
parenchyma, and multiple regions of elevated fluorodeoxyglucose
metabolism in the bones, such as the sternum, multiple vertebrae,
and the bilateral ilia, pubis and ischia. A diagnosis of lymphoma
was therefore considered. |
Following treatment, the patient’s memory and
orientation improved, and the calcium level decreased to 2.86
mmol/l the next day. A chest CT scan showed bilateral pulmonary
exudation and pneumonia was indicated, therefore, antibiotics (400
mg/day moxifloxacin and 100 mg/day fluconazole) were administered.
Subsequent to the attenuation of the symptoms, a biopsy of the
right lingual lymph node was performed, which showed peripheral
T-cell lymphoma, not otherwise specified (NOS). The disease stage
was IVB according to the Ann Arbor staging system (7), and chemotherapy consisting of 1.3 g
cyclophosphamide, 60 mg liposomal doxorubicin and 40 mg vinorelbine
on day 1, and 15 mg dexamethasone on days 1–5 was administered.
Following two courses of chemotherapy, the result of a B-mode
ultrasound and CT scan showed that the patient achieved partial
remission.
Discussion
Certain studies based on population have confirmed
the association between inflammatory myopathies and malignancies.
Hill et al (8) performed a
pooled analysis of the populations in Sweden, Denmark and Finland,
and revealed a strong association between dermatomyositis and
malignancies [standardized incidence ratio (SIR), 3.0; 95%
confidence interval (CI), 2.5–3.6], particularly ovarian (SIR,
10.5; 95% CI, 6.1–18.1), lung (SIR, 5.9; 95% CI, 3.7–9.2),
pancreatic (SIR, 3.8; 95% CI, 1.6–9.0), stomach (SIR, 3.5; 95% CI,
1.7–7.3), and colorectal (SIR, 2.5; 95% CI, 1.4–4.4) cancer, and
non-Hodgkin’s lymphoma (SIR, 3.6; 95% CI, 1.2–11.1). Polymyositis
was associated with a higher risk of non-Hodgkin’s lymphoma (SIR,
3.7; 95% CI, 1.7–8.2) and lung (SIR, 2.8; 95% CI, 1.8–4.4) and
bladder (SIR, 2.4; 95% CI, 1.3–4.7) cancer. The majority of
malignancies associated with dermatomyositis are adenocarcinomas,
while polymyositis is mainly associated with lymphomas (9). The majority of malignancies are
discovered within one year of a confirmed diagnosis of
polymyositis/dermatomyositis. It has been revealed that
inflammatory myopathies are essentially a type of para-neoplastic
syndrome. Additionally, the prolonged utility of immune suppressive
medicine is also associated with an increased incidence of
malignancies (10). The majority of
patients with cancer-associated myositis (CAM) are negative for
auto-antibodies and antisynthetase antibodies. In the present case,
the patient developed myositis of the lower limbs one year
previously, and no signs of malignancies were found at that time.
Four months prior to admission, the weakness of the lower limbs was
aggravated and a PET-CT scan revealed multiple lesions with
enhanced metabolism around the body. Multiple biopsies were
performed, which led to the final diagnosis of peripheral T-cell
lymphoma, NOS. Throughout the course of the disease, the tests for
auto-antibodies, such as ANA and ANCA, and antisynthetase
antibodies, were negative. The clinical manifestation and disease
progression matched the characteristics of CAM.
A PET/CT scan upon aggravation of the disease
revealed signs of malignancy and aided in the determination of the
biopsy site. The value of PET/CT and traditional examinations in
polymyositis/dermatomyositis patients have been previously compared
(10). Traditional examinations
include chest and abdominal CT, breast molybdenum photography,
gynecological examination and tests for neoplasm biomarkers. For
patients of inflammatory myopathies, the positive and negative
predictive values of PET/CT are 85.7 and 93.8%, respectively,
whereas the positive and negative predictive values of traditional
examinations are 77.8 and 95.7%, respectively. The total predictive
value of PET/CT and traditional examinations is 92.7% (11). Traditional examinations expend a
high amount of time and energy, while in comparison, PET/CT is
effective and convenient. However, in China, PET/CT is extremely
expensive and is not covered by medical insurance, therefore,
consideration of economic conditions and the clinical situation is
required when choosing examinations.
The primary symptom of the present patient was
hypercalcemia. Common reasons for hypercalcemia are primary
hyperparathyroidism and chronic renal insufficiency (during
treatment with calcium tablets and vitamin D, or accompanied by
tertiary hyperparathyroidism). Relatively rare reasons include
vitamin D-related diseases (granulomatous diseases or vitamin D
poisoning), other endocrine diseases (such as hyperthyroidism),
metabolic factors (such as milk-alkali syndrome, diuretics and the
utility of lithium salt) and other various reasons, including
breaking limbs and familial low urinary calcium hypercalcemia
(12). In the present study, the
patient’s PTH level was not high and hyperparathyroidism could be
ruled out. The BUN and creatine levels were moderately high,
suggesting renal insufficiency, however, moderate renal
insufficiency would not cause such serious hypercalcemia.
Consequently, malignancy-associated hypercalcemia was diagnosed.
Malignancies associated with hypercalcemia in adults include
cancers such as lung cancer, head and neck neoplasms, urinary tract
neoplasms and breast cancer, and also hematological malignancies
such as multiple myeloma (incidence rate, 13–30%), adult T-cell
leukemia/lymphoma (ATLL; 50–70%), Hodgkin’s lymphoma (5%),
non-Hodgkin’s lymphoma (0.8–13%) and acute myeloid leukemia
(extremely rare) (4). Among the
hematological malignancies, hypercalcemia is common in multiple
myeloma and ATLL, but relatively rare in non-Hodgkin’s lymphoma
(13). The final diagnosis in the
present study was of peripheral T-cell lymphoma with hypercalcemic
crisis as a primary symptom accompanied by polymyositis; to the
best of our knowledge, such a case has not previously been
reported.
Hypercalcemia is caused by factors such as enhanced
bone absorption, elevated calcium re-absorption by the renal
tubules and elevated calcium absorption by the intestine (14). Enhanced bone absorption is the most
significant cause and is mediated by the bone metastasis of tumors
or by cytokines secreted by tumor cells. Common detectable
cytokines include parathyroid hormone-related peptide (PTHrP),
interleukin-1 (IL-1), IL-6, transforming growth factor-α, tumor
necrosis factor-α, macrophage inflammatory protein-1α and
calcitriol [1,25-(OH)2D3]. Occasionally,
ectopic PTH secretion can be detected. However, the division of
these causes into these categories is possibly too simplified and
in the clinic, numerous factors could exist simultaneously to cause
hypercalcemia (14). Hypercalcemia
often occurs in stage III/IV cases of B-cell non-Hodgkin’s lymphoma
and indicates a poor prognosis (15). Calcitriol is the most important
mediator in almost all Hodgkin’s lymphomas and in 30–40% of
non-Hodgkin’s lymphomas. A previous immunohistochemical study
revealed that lymphoma-associated macrophages are possibly a main
source of ectopic PTH (16). In the
present study, the patient was stage IVB, according to the Ann
Arbor staging system (5), and
presented with a broad area of tumor cell infiltration. The
International Prognostic Index score (17) was 5 (high-risk group), which
indicated a poor prognosis. The PTHrP and calcitriol levels were
not detected due to the limited time and laboratory devices in the
hospital.
The main treatments for hypercalcemia include saline
rehydration and dialysis, and administration of loop diuretics,
bisphosphonates, calcitonin, mithramycin, gallium and
corticosteroids (18). In the
present case, the patient was treated with saline rehydration,
furosemide, salmon calcitonin and dexamethasone, and the calcium
level decreased rapidly as a consequence. The calcium concentration
decreased by 1 mmol/l on the first day and was restored to within
the normal range on the third day. Chemotherapy was prescribed,
following which, no occurrences of hypercalcemia or renal
insufficiency were noted.
In conclusion, polymyositis/dermatomyositis are
strongly associated with malignancies, therefore, physicians should
screen for tumors in patients with inflammatory myopathies to avoid
missing the diagnosis, particularly within five years of the onset
of the disease. Hypercalcemia is a common metabolic complication of
malignancies, and should be considered in order to adopt
appropriate treatment measures immediately. Treating the primary
disease and achieving remission are fundamental methods to treat
hypercalcemia. Progression has been made in the analysis of the
pathogenesis of CAM, however, it is not simple or unique to a
specific malignancy. Further investigations are required to
elucidate the mechanisms involved.
References
|
1
|
Hak AE, de Paepe B, de Bleecker JL, Tak PP
and de Visser M: Dermatomyositis and polymyositis: new treatment
targets on the horizon. Neth J Med. 69:410–421. 2011.PubMed/NCBI
|
|
2
|
Buchbinder R and Hill CL: Malignancy in
patients with inflammatory myopathy. Curr Rheumatol Rep. 4:415–426.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Savage KJ, Chhanabhai M, Gascoyne RD and
Connors JM: Characterization of peripheral T-cell lymphomas in a
single North American institution by the WHO classification. Ann
Oncol. 15:1467–1475. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sargent JT and Smith OP: Haematological
emergencies managing hypercalcaemia in adults and children with
haematological disorders. Br J Haematol. 149:465–477. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shepard MM and Smith JW III:
Hypercalcemia. Am J Med Sci. 334:381–385. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dyck PJ, Boes CJ, Mulder D, Millikan C,
Windebank AJ, Dyck PJ and Espinosa R: History of standard scoring,
notation, and summation of neuromuscular signs. A current survey
and recommendation. J Peripher Nerv Syst. 10:158–173. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lister TA, Crowther D, Sutcliffe SB, et
al: Report of a committee convened to discuss the evaluation and
staging of patients with Hodgkin’s disease: Cotswolds meeting. J
Clin Oncol. 7:1630–1636. 1989.PubMed/NCBI
|
|
8
|
Hill CL, Zhang Y, Sigurgeirsson B, et al:
Frequency of specific cancer types in dermatomyositis and
polymyositis: a population-based study. Lancet. 357:96–100. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Aggarwal R and Oddis CV: Paraneoplastic
myalgias and myositis. Rheum Dis Clin North Am. 37:607–621. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen YJ, Wu CY, Huang YL, Wang CB, Shen JL
and Chang YT: Cancer risks of dermatomyositis and polymyositis: a
nationwide cohort study in Taiwan. Arthritis Res Ther. 12:R702010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Selva-O’Callaghan A, Grau JM,
Gámez-Cenzano C, et al: Conventional cancer screening versus PET/CT
in dermatomyositis/polymyositis. Am J Med. 123:558–562. 2010.
View Article : Google Scholar
|
|
12
|
Endres DB: Investigation of hypercalcemia.
Clin Biochem. 45:954–963. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Vassilopoulou-Sellin R, Newman BM, Taylor
SH and Guinee VF: Incidence of hypercalcemia in patients with
malignancy referred to a comprehensive cancer center. Cancer.
71:1309–1312. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Clines GA and Guise TA: Hypercalcaemia of
malignancy and basic research on mechanisms responsible for
osteolytic and osteoblastic metastasis to bone. Endocr Relat
Cancer. 12:549–583. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Majumdar G: Incidence and prognostic
significance of hypercalcaemia in B-cell non-Hodgkin’s lymphoma. J
Clin Pathol. 55:637–638. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hewison M, Kantorovich V, Liker HR, et al:
Vitamin D-mediated hypercalcemia in lymphoma: evidence for hormone
production by tumor-adjacent macrophages. J Bone Miner Res.
18:579–582. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
No authors listed. A predictive model for
aggressive non-Hodgkin’s lymphoma. The International Non-Hodgkin’s
Lymphoma Prognostic Factors Project. N Engl J Med. 329:987–994.
1993. View Article : Google Scholar
|
|
18
|
Koh LK: The diagnosis and management of
hypercalcaemia. Ann Acad Med Singapore. 32:129–139. 2003.PubMed/NCBI
|















