Introduction
A number of primary colorectal carcinoma are often
found by colposcopy or computed tomography (CT) scans. The
detection of these tumors is mainly based on an active complaint of
certain typical digestive symptoms, including abdominal pain,
bright red blood in the stool and weight loss (1). However, tumors occasionally exhibit
atypical presentations with a variety of symptoms or signs at a
distance from the digestive system (2,3). Chest
pain and pleural effusion are particularly uncommon presentations
of digestive malignances that can be easily ignored in the
Outpatient Department compared with other clinical presentations. A
refractory pleural effusion presenting as a primary symptom of
colorectal cancer is rarely reported (<0.9% incidence according
to our own database) and mainly occurs in the elderly. In the
present report, an older patient, who was admitted to The First
Affiliated Hospital of Sun Yat-Sen University (Guangzhou, China)
with a refractory left pleural effusion, was diagnosed with colon
carcinoma and underwent a radical tumor resection. The clinical
features of refractory pleural effusion that result from a colonic
tumor are summarized, with a focus on the differential diagnosis of
gastrointestinal stromal tumors. Written informed consent was
obtained from the patient.
Case report
A 60-year-old male presented to the Outpatient
Department of The First Affiliated Hospital of Sun Yat-Sen
University due to a four-month history of repeated chest pain. The
pain was located mainly on the left side of the chest and had
worsened over the three days prior to admission. The patient had
not developed a cough, excess sputum, hemoptysis or other symptoms.
A chest X-ray examination performed at a local hospital revealed a
left-sided pleural effusion. A closed thoracic drainage procedure
was performed at this time, but the pain recurred in the same
location shortly afterwards. The pleural fluid was exudative and
was found to lack tumor cells by cytological analysis. A
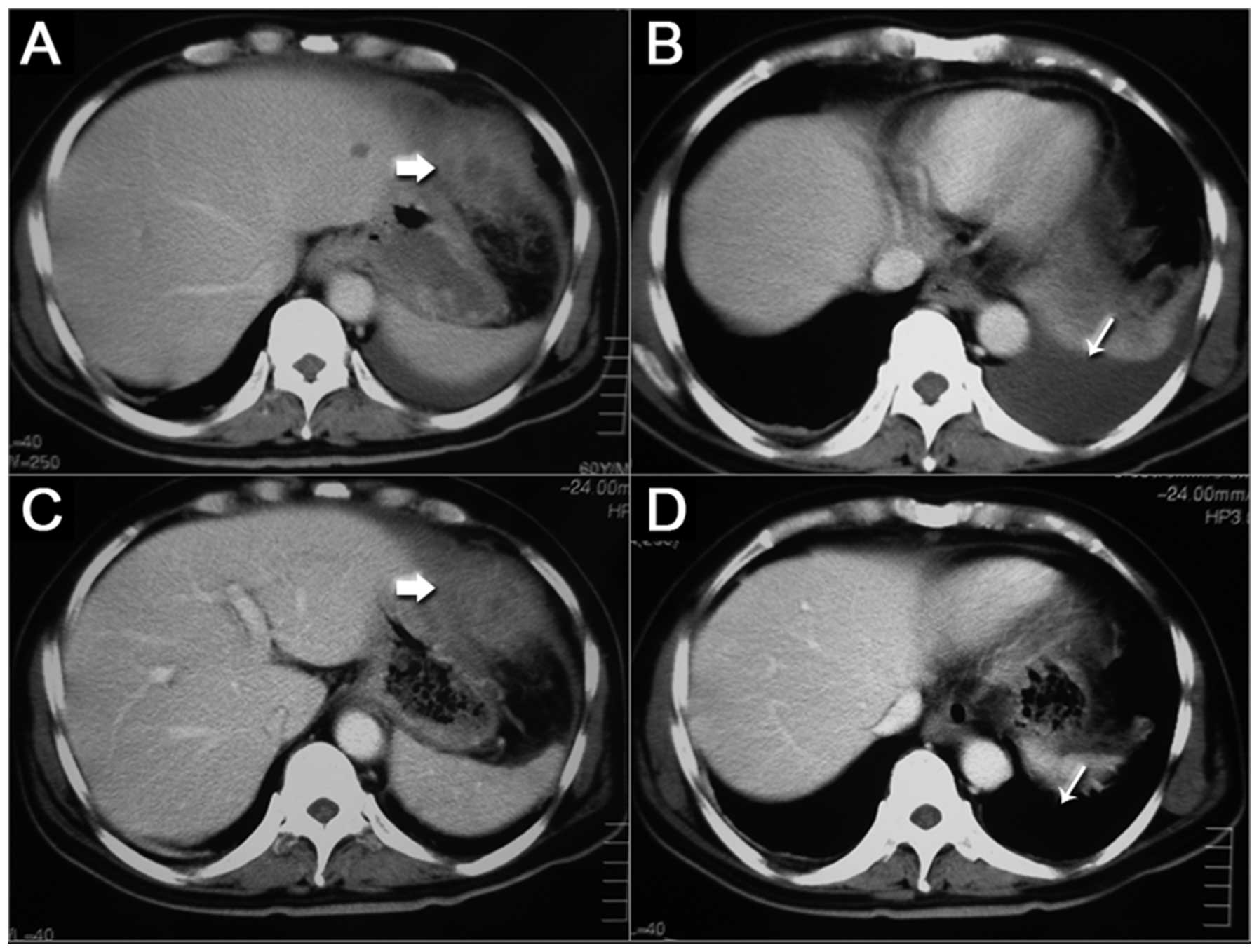
non-enhanced CT scan of the chest and abdomen revealed a marked
soft-tissue prominence in the gastric greater curvature and a mass
posterior to the spleen (Fig. 1).
No intrahepatic lesions were found, and the other organs in the
abdomen were unremarkable. The patient was referred to The First
Affiliated Hospital of Sun Yat-Sen University (Guangzhou, China)
for further diagnosis and treatment. Following admission, an
ensuing examination was performed, consisting of a tumor marker
examination for digestive malignancies, esophagogastroduodenoscopy,
colonoscopy, additional staging by enhanced CT and endoscopic
ultrasonography to identify the characteristics of non-presumed
masses.
An increased carbohydrate antigen (CA)125 level
(67.1 U/ml; normal range, 0–35 U/ml) was recorded, but the other
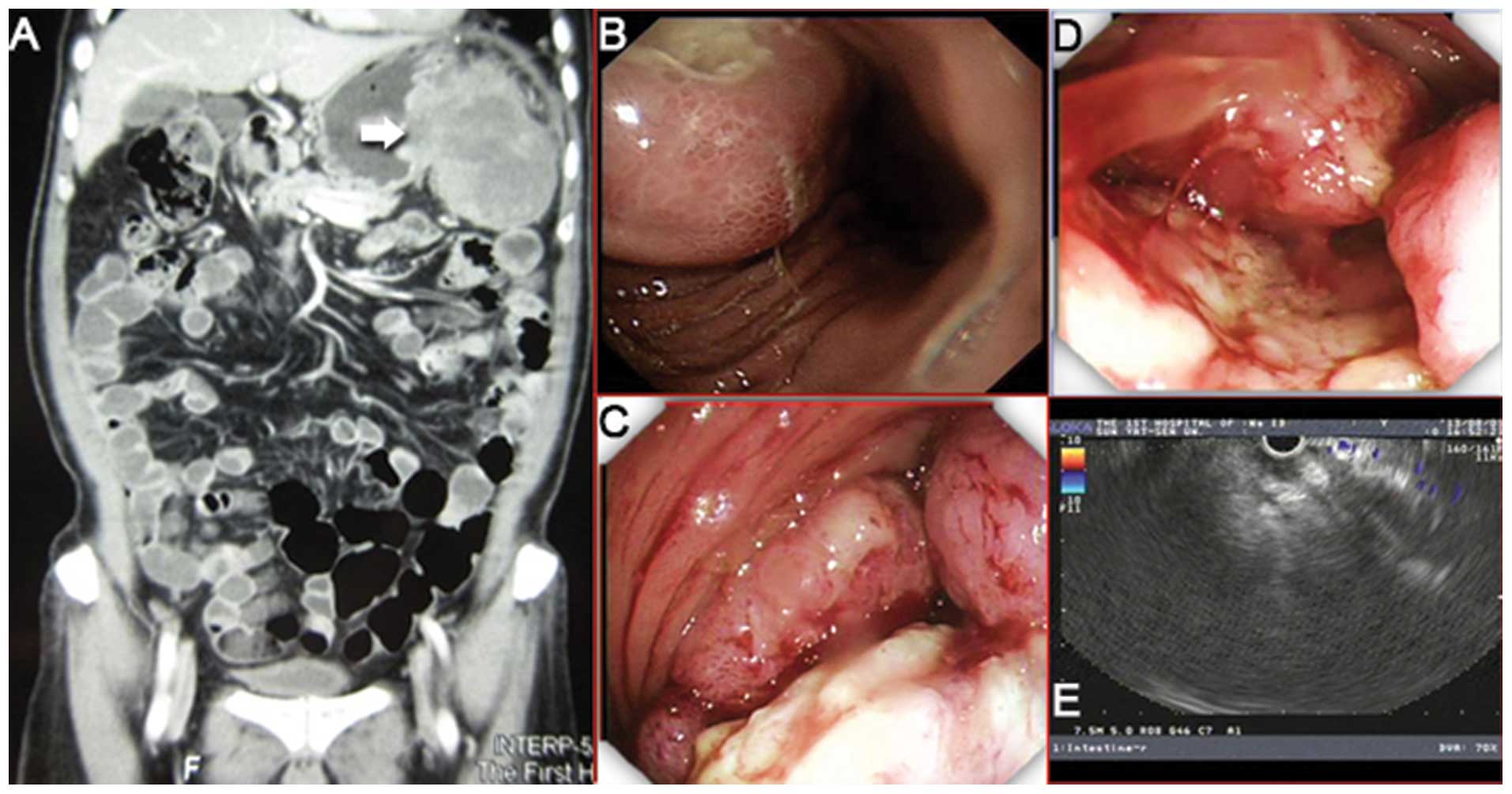
markers were in the normal range. Endoscopic examination revealed a
neoplasm, 6 cm in diameter, in the fundus of the stomach, along the
greater curvature. Another cauliflower-like neoplasm was located at
the splenic curve of the descending colon, which resulted in total
bowel obstruction (Fig. 2). The
biopsy results for the two masses indicated a diagnosis of a
moderate- to poorly-differentiated adenocarcinoma. Taken together
with findings from the endoscopic ultrasonography and abdominal
enhanced CT (Fig. 2), the two
neoplasms were confirmed to be homologous, without any observation
of distal metastasis. Following a multiple-disciplinary team
discussion for the present case, the clinical tumor-node-metastasis
stage was set to cT4bN0M0, stage IIC, and a laparostomy was
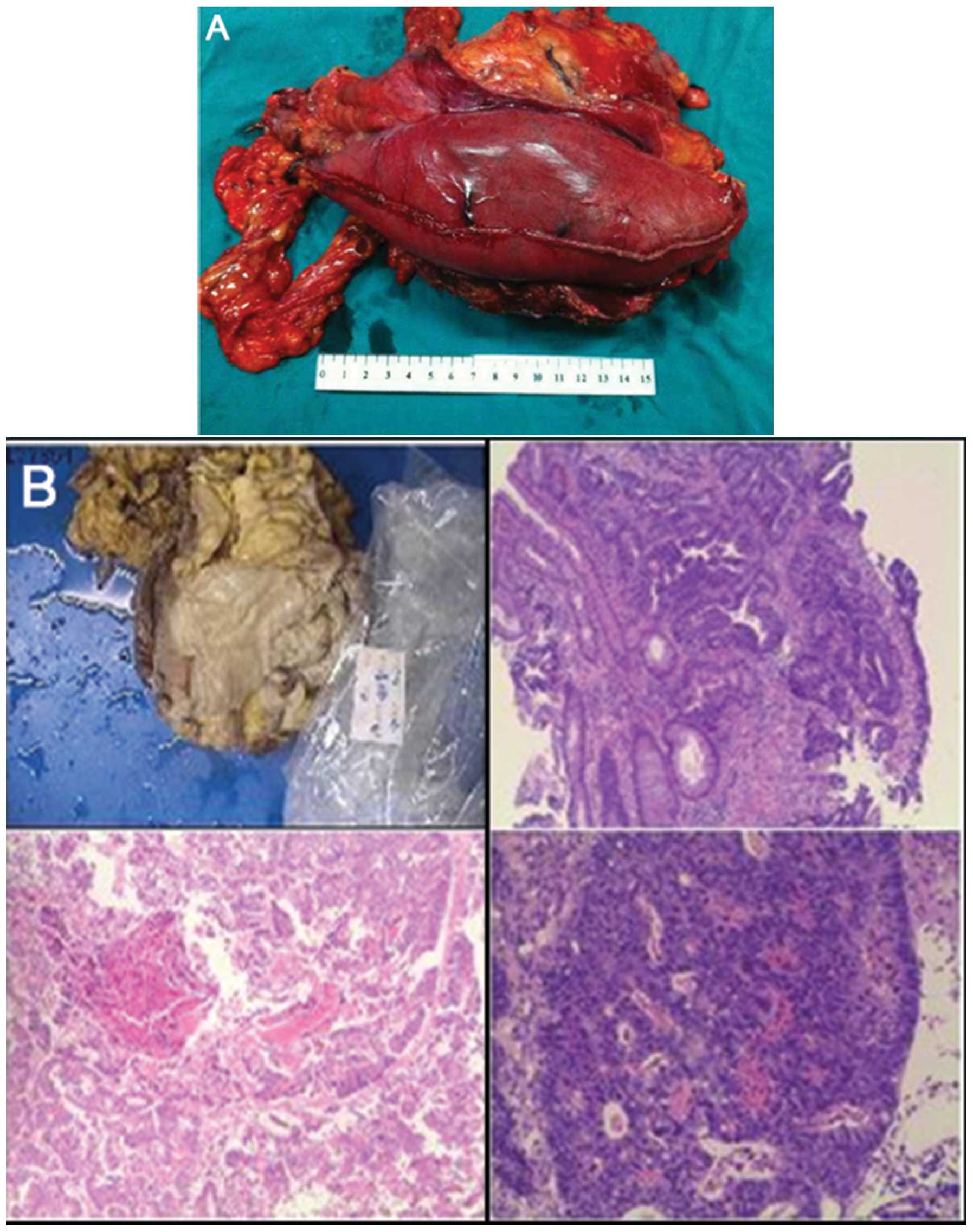
scheduled for a radical resection (R0). During the surgery,
diaphragm and left hepatic lobe involvement was identified, and an
extensive resection, which consisted of a partial greater curvature
gastrectomy, segment 1 hepatic lobectomy, segmental left diagram
resection, partial left abdominal wall resection and left
hemicolectomy, was successfully performed (Fig. 3A).
Six days after the surgery, the patient was
discharged from the department. The final diagnosis was
moderately-differentiated colonic adenocarcinoma, with one out of
25 examined lymph nodes found to be positive for metastasis
(pT4bN1aM0, stage IIIC; Fig. 3B).
The patient was subsequently scheduled for approximately six
courses of oxaliplatin 85 mg/m2 i.v. over 2 hours on day
1, leucovorin 400 mg/m2 i.v. over 2 hours on day 1 and
fluorouracil 400 mg/m2 i.v. bolus on day 1, then 2400
mg/m2 over 48 hours by continuous i.v. infusion
(mFOLFOX6; repeated every two weeks). Within the 12-month follow-up
period, the patient was asymptomatic, without evidence of tumor
recurrence.
Discussion
The present study reported an unusual case of
colorectal adenocarcinoma, with an atypical clinical presentation.
The patient was admitted to hospital with non-specific symptoms of
gastrointestinal malignancies, and a notable amount of time was
spent on finding the etiology of the pleural effusion. Generally,
refractory pleural effusion, which leads to the persistent
complaint of chest pain, can be caused by various diseases,
including bacterial pneumonia, lung cancer, heart failure and
kidney dysfunction. Pleural effusion is often considered to be an
intervention-requiring complication regardless of the primary
disease. However, it is less common that the pleural effusion is
raised by the chronic stimulation of intra-abdominal neoplasm. In
that condition, ascites and peritoneal metastasis often develop
prior to the pleural effusion in the majority of digestive or
gynecological malignancies (4,5).
However, in the present case, no ascites or peritoneal invasion was
observed, with regional invasion analyzed by CT scan and endoscopic
ultrasonography. For the diagnosis of a non-presumed pleural
effusion, CT scans are particularly useful for detecting neoplastic
disease of the upper abdomen prior to certain invasive procedures
(6).
Among the various types of intra-abdominal
neoplasms, digestive tumors, including intestinal lymphoma,
gastrointestinal stromal tumors (GISTs) and colorectal cancer, are
more frequently associated with pleural effusion compared with
gynecological malignancies. In the present case, a GIST was also a
possible diagnosis according to the features of the abdominal CT
scan. Therefore, endoscopic ultrasonography was employed to discern
between the two forms of digestive malignancy, as it is a useful
tool for the accurate diagnosis of a GIST (7). Generally, a GIST is located in the
muscularis propria, whereas colorectal cancer often invades all
layers of the gastrointestinal wall (8). Uncommonly, intra-abdominal
endometriosis can also induce similar symptoms to colon cancer and
caution is therefore necessary when treating young females
(9).
Pleural effusion is an atypical presentation of
colorectal carcinoma, which exhibits its own features (3). According to our clincial experiences,
this complication is always located in the left thoracic cavity and
is frequently found in the elderly by a respiratory physician.
Percutaneous thoracic drainage may be futile and inadequate to
clear the effusion, with a sterile exudate obtained by pleural tap.
Thoracic-abdominal CT scans identify a huge mass in the splenic
curve of the colon, and endoscopic biopsies confirm the diagnosis
of adenocarcinoma. At present, there are no studies to indicate
that GIST is relevant to this specific complication. In the
majority of cases, the diaphragm, stomach, left liver lobe and
partial peritoneum are vulnerable and require excision during a
subtotal colectomy procedure, with several courses of subsequent
adjuvant chemotherapy. If a radical resection is achieved and the
subsequent chemotherapy is successfully completed, the pleural
effusion is quickly resolved and the prognosis of colonic
adenocarcinoma is quite good.
Older patients with left pleural effusion, but no
presumed diagnosis arising from standard clinical examination, as
in the present case, should undergo thoracic-abdominal CT scans to
exclude potential upper abdominal neoplasms. Endoscopic
ultrasonography is useful for the exclusion of GISTs and to guide
the subsequent surgical treatment. A radical tumor resection
combined with adjuvant chemotherapy is indispensable for colorectal
adenocarcinoma elimination, and the prognosis of this specific
cancer is quite favorable.
In conclusion, the successful management of the
present case indicates that refractory pleural effusion is an
atypical presentation of colorectal cancer. Early diagnosis and
radical resection would improve the long-term outcomes of
colorectal cancer patients.
References
|
1
|
Brenner H, Kloor M and Pox CP: Colorectal
cancer. Lancet. 383:1490–1502. 2014. View Article : Google Scholar
|
|
2
|
Bhargava R, Winer-Muram HT, Kauffman WM,
Jennings SG and Pratt CB: Chest radiographic features of thoracic
metastatic disease in adolescents with colon cancer. Pediatr
Radiol. 24:491–493. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fullerton DA, López F, Avendano R,
Aparicio R and Wistuba I: Atypical presentation of a colorectal
carcinoma. Rev Med Chil. 132:985–988. 2004.(In Spanish). PubMed/NCBI
|
|
4
|
Akamatsu M, Tsuji Y, Nakata W, et al: A
patient with recurrent sigmoid colon cancer in whom pleural
effusion and ascites resolved after FOLFOX 4 therapy. Gan To Kagaku
Ryoho. 34:2305–2307. 2007.(In Japanese). PubMed/NCBI
|
|
5
|
Guariglia OO, Scheimberg AB, Feldman L and
Moguilevsky L: Report of a case of ovarian tumor ascites and right
pleural Effusion and Saint’s triad. Prensa Med Argent. 50:947–949.
1963.(In Spanish). PubMed/NCBI
|
|
6
|
Cabriada V, Antoñana JM, Sobradillo V, et
al: Usefulness of computerized tomography in the study of pleural
effusion with no presumed diagnosis. Arch Bronconeumol. 33:503–508.
1997.(In Spanish).
|
|
7
|
Kim MN, Kang SJ, Kim SG, et al: Prediction
of risk of malignancy of gastrointestinal stromal tumors by
endoscopic ultrasonography. Gut Liver. 7:642–647. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lok KH, Lai L, Yiu HL, Szeto ML and Leung
SK: Endosonographic surveillance of small gastrointestinal tumors
originating from muscularis propria. J Gastrointestin Liver Dis.
18:177–180. 2009.PubMed/NCBI
|
|
9
|
Flanagan KL and Barnes NC: Pleural fluid
accumulation due to intra-abdominal endometriosis: a case report
and review of the literature. Thorax. 51:1062–1063. 1996.
View Article : Google Scholar : PubMed/NCBI
|