Introduction
Cervical cancer is one of the most common cancers
worldwide (1) and the leading cause
of cancer-associated mortality in women in developing countries
(2). The treatment of cervical cancer
mainly consists of surgery, chemotherapy and radiotherapy.
Radiotherapy is an important therapy for cervical cancer, and it is
currently administered to almost 80% of patients with cervical
cancer, at various clinical stages (3,4). However,
radiotherapy is not extremely effective at treating cervical
cancer, particularly in patients at an advanced stage of disease
(5,6).
Therefore, it is necessary to identify novel methods to combine
with radiotherapy to improve the efficacy of treatment.
Chemokines are a superfamily of cytokines that are
important regulators of cell migration, lymphocyte recruitment and
angiogenesis (7). CXC chemokine
ligand 10 (CXCL10), also termed interferon-γ-induced protein 10, is
a small cytokine belonging to the CXC chemokine family that was
initially characterized as a chemoattractant for activated T
lymphocytes. CXCL10 has been identified as a 10-kDa secreted
protein induced by interferon-γ (8)
in a variety of cell types, including endothelial cells,
keratinocytes, fibroblasts, activated monocytes and neutrophils
(9). Over previous years, studies
have increasingly reported that CXCL10 plays an important role in
angiogenesis and tumor growth inhibition (10–12). Our
previous study revealed that CXCL10 may induce apoptosis, inhibit
angiogenesis and human papillomavirus, resulting in effects against
cervical cancer (13). It has also
been found that CXCL10 causes the avoidance of the S phase of the
cell cycle in cervical cancer cells (14), during which time the cells are least
sensitive to radiation.
Therefore, it was assumed that the combination of
CXCL10 gene therapy and radiotherapy may improve the inhibition of
cervical cancer progression through a variety of mechanisms, to
achieve effective coordination in the treatment of cervical
cancer.
In the present study, human HeLa cervical carcinoma
tumors were established in immunodeficient mice, and CXCL10 gene
therapy and radiotherapy were administered to establish the effects
on tumor growth.
Materials and methods
Cell lines
The cervical cancer HeLa cell line was obtained from
the American Type Culture Collection (Manassas, VA, USA). The cells
were cultured in media consisting of Dulbecco's modified Eagle's
medium (DMEM) supplemented with 2 mM L-glutamine (GE Healthcare
Life Sciences, Logan, UT, USA), plus 100 U/ml penicillin and 100
µg/ml streptomycin (Beyotime Institute of Biotechnology, Haimen,
Jiangsu, China), and 10% fetal bovine serum (FBS; GE Healthcare
Life Sciences).
Plasmids
A DNA fragment encoding CXCL10 was cloned into the
pcDNA3.1 vector (Invitrogen, Carlsbad, CA, USA) between the
restriction sites for EcoRI and XhoI. The plasmid was
purified by two rounds of passaging over Endo-free columns (Qiagen,
Valencia, CA, USA), as previously described (15).
Reverse transcription-quantitative
polymerase chain reaction (PCR)
Total RNA was isolated from HeLa cells transfected
with the pcDNA3.1-CXCL10 plasmid using TRIzol reagent (Invitrogen),
according to the manufacturer's instructions. Total RNA was reverse
transcribed to cDNA using the reverse transcription system from
Promega (Madison, WI, USA). PCR was subsequently performed with an
initial denaturation step that was performed at 94°C for 4 min,
followed by 28 cycles at 94°C for 20 s, 60°C for 20 s and 72°C for
30 s, with a final extension of 72°C for 4 min. The primers used
for PCR were as follows: CXCL10 forward, 5′-CCTTATCTTTCTGACTCT
AAGTGGC-3′ and reverse, 5′-ACGTGGACAAAATTG GCTTG-3′; GAPDH forward,
5′-TCATCTCTGCCCCCT CTG-3′ and reverse,
5′-CCTGCTTCACCACCTTCTTG-3′.
Tumor formation in nude mice
To establish the cervical cancer tumors,
eight-week-old female nude mice (Beijing HFK Bioscience Co., Ltd,
Beijing, China) were inoculated with 1×106 HeLa cells
subcutaneously in the right flank. Tumor dimensions were measured
with calipers every 3 days, and tumor volumes were calculated as
follows (16):
Tumor volume (mm3) = 0.5 × length ×
width2
Animal experiments were conducted according to
institutional guidelines concerning animal use and care, and the
study was approved by the ethics committee of Luzhou Medical
College, (Luzhou, China).
CXCL10 gene therapy
The mice were randomly divided into five groups when
the tumors were ~15 mm3 in volume, which occurred ~12
days subsequent to HeLa cell inoculation, and each group consisted
of 10 mice. The recombinant CXCL10 or pcDNA3.1 plasmids were gently
combined with a liposomal transfection agent in a 1:3 ratio (50:150
µg for each mouse), and incubated at room temperature for ~30 min.
The mixture was then administered intravenously 5 times on days 12,
15, 18, 21 and 24. Mice from the phosphate-buffered saline (PBS)
group were injected with PBS as the control agent and acted as the
control group.
Radiotherapy
Radiotherapy was performed when the tumors were ~300
mm3 in volume, which occurred ~27 days subsequent to
inoculation of the mice with HeLa cells. The tumors were radiated
with 6-MV X-rays, and each mouse was administered with a total
radiation dose of 20 Gy, with 4 Gy being administered for each dose
on 5 days, at the rate of 200 cGy/min.
Histological analysis
The mice were sacrificed 39 days subsequent to
inoculation with the HeLa cells and the tumors were removed and
embedded in paraffin. Tissue sections were immunostained with rat
anti-mouse monoclonal antibody against cluster of differentiation
31 (CD31; catalog no. 553369; BD Biosciences, Franklin Lakes, NJ,
USA; dilution, 1:200) or rabbit anti-human polyclonal antibody
against Ki-67 (catalog no. 19972-1-AP; Proteintech Group, Inc.,
Wuhan, Hubei, China; dilution, 1:100). Vessel density was
calculated by counting the number of microvessels per high-power
field (magnification, x200; Leica DM3000, Leica Microsystems GmbH,
Wetzlar, Germany) in the tumor sections, as previously described
(17). The rate of cell proliferation
was calculated for five random fields by dividing the number of
Ki-67-positive cells by the total number of cells. TUNEL was
performed using an in situ cell-death detection kit (Roche,
Mannheim, Germany). In total, five fields were randomly selected and
analyzed. The apoptotic index was calculated as the ratio of the
apoptotic cell number to the total tumor cell number in each
field.
Statistical analysis
The data were assessed by a one-way analysis of
variance and Student's t-test, and a post-hoc test was
conducted according to the Student-Newman-Keuls method. P<0.05
was considered to indicate a statistically significant
difference.
Results
Overexpression of CXCL10 in HeLa
cells
The recombinant plasmid encoding CXCL10 was cloned,
and the transcript of CXCL10 mRNA was confirmed in transfected
cells, as revealed by RT-PCR.
Combination of CXCL10 gene therapy and
radiotherapy markedly inhibits cervical cancer tumor growth
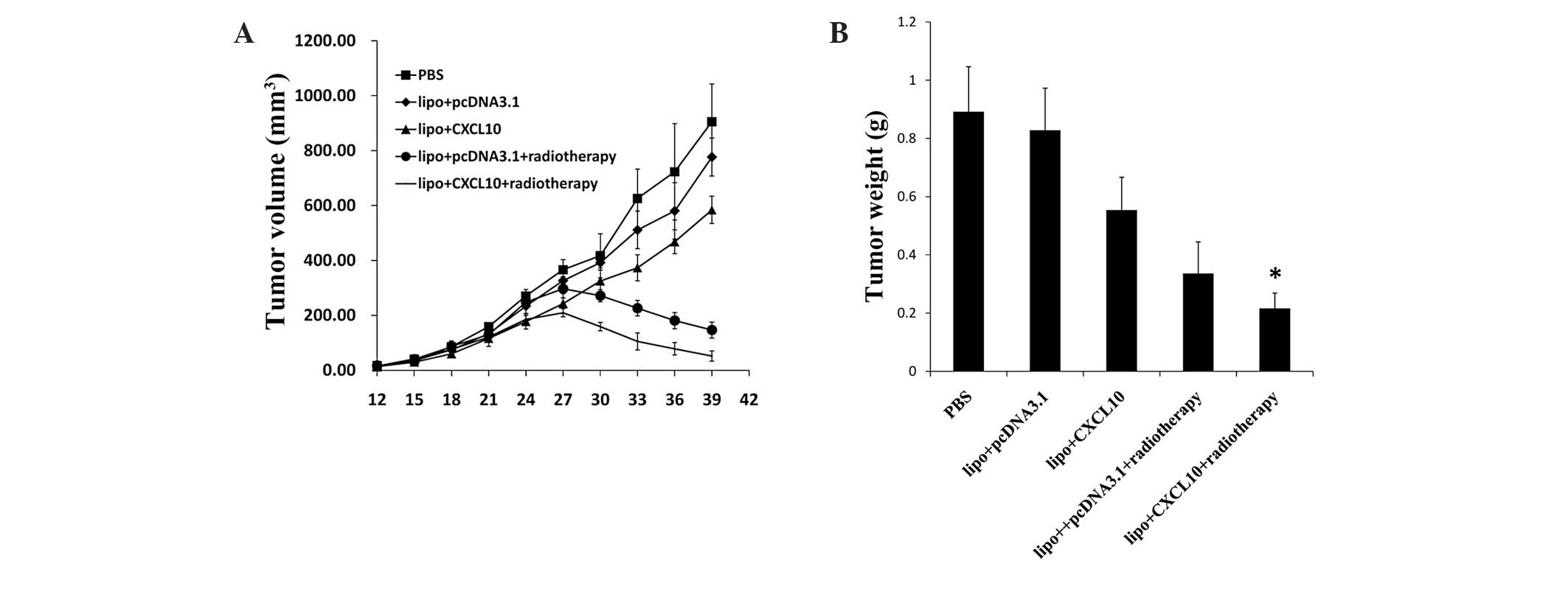
The effects of CXCL10 gene therapy and radiotherapy
alone and in combination on xenograft tumor models of cervical
cancer were assessed in nude mice. The tumor volumes were measured
every 3 days subsequent to inoculation with HeLa cells. The tumor
volume in the PBS and pcDNA3.1 control groups increased rapidly. By
contrast, the tumor growth rate in nude mice treated with CXCL10
gene therapy alone decreased compared with the control group, and
this decline was more evident in the group treated with
radiotherapy. In addition, the combined treatment of CXCL10 gene
therapy and radiotherapy inhibited tumor growth more effectively
compared with all other groups (P<0.05) (Fig. 2A). Similarly, the treatment of tumors
demonstrated corresponding effects on tumor weight (Fig. 2B). Overall, CXCL10 gene therapy in
combination with radiotherapy significantly inhibited cervical
cancer tumor growth.
Effect of CXCL10 gene therapy and
radiotherapy on angiogenesis in cervical cancer
Immunohistochemical labeling of microvessels by CD31
in tumor sections revealed a decrease in tumor vessel density in
the CXCL10 group and radiotherapy group compared with those in the
control group. In combination, treatment with CXCL10 plus
radiotherapy appeared more effective at reducing microvessel
density compared with the other groups (P<0.05; Fig. 3).
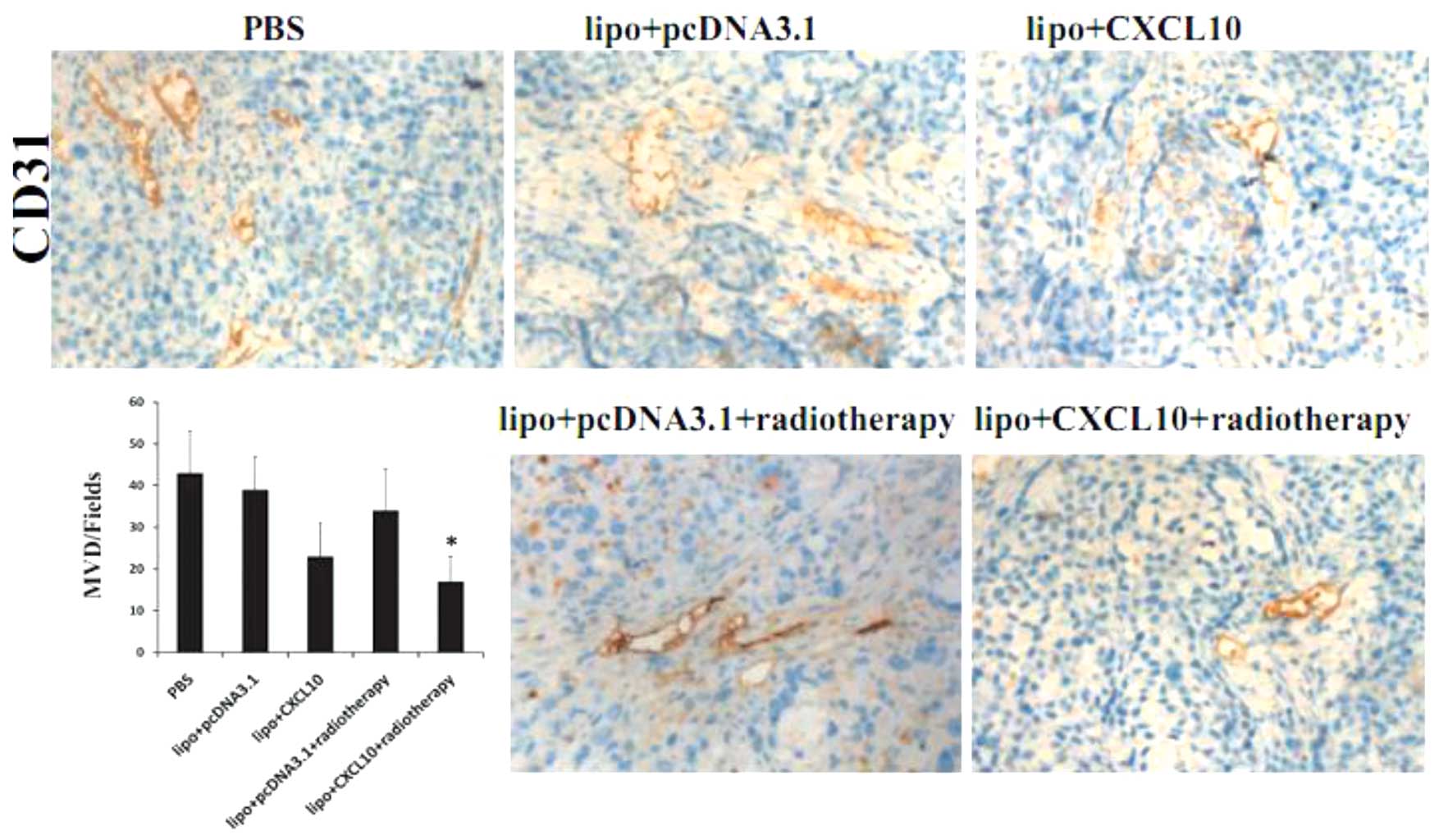 | Figure 3.Effect of CXCL10 gene therapy and
radiotherapy on angiogenesis in cervical cancer. The vascular
structures in tumors treated with PBS, lipo + pcDNA3.1, lipo +
CXCL10, lipo + pcDNA3.1 + radiotherapy or lipo + CXCL10 +
radiotherapy, were detected by immunohistochemical staining for
CD31. The combination group, which was treated with CXCL10 gene
therapy and radiotherapy, exhibited a significantly decreased
vessel density compared with any other groups. Error bars represent
standard error of the entire treatment group. *P<0.05 vs.
controls. CXCL10, CXC chemokine ligand 10; PBS, phosphate buffered
saline; CD31, cluster of differentiation 31; lipo, liposomal
transfection agent. |
Effect of CXCL10 gene therapy and
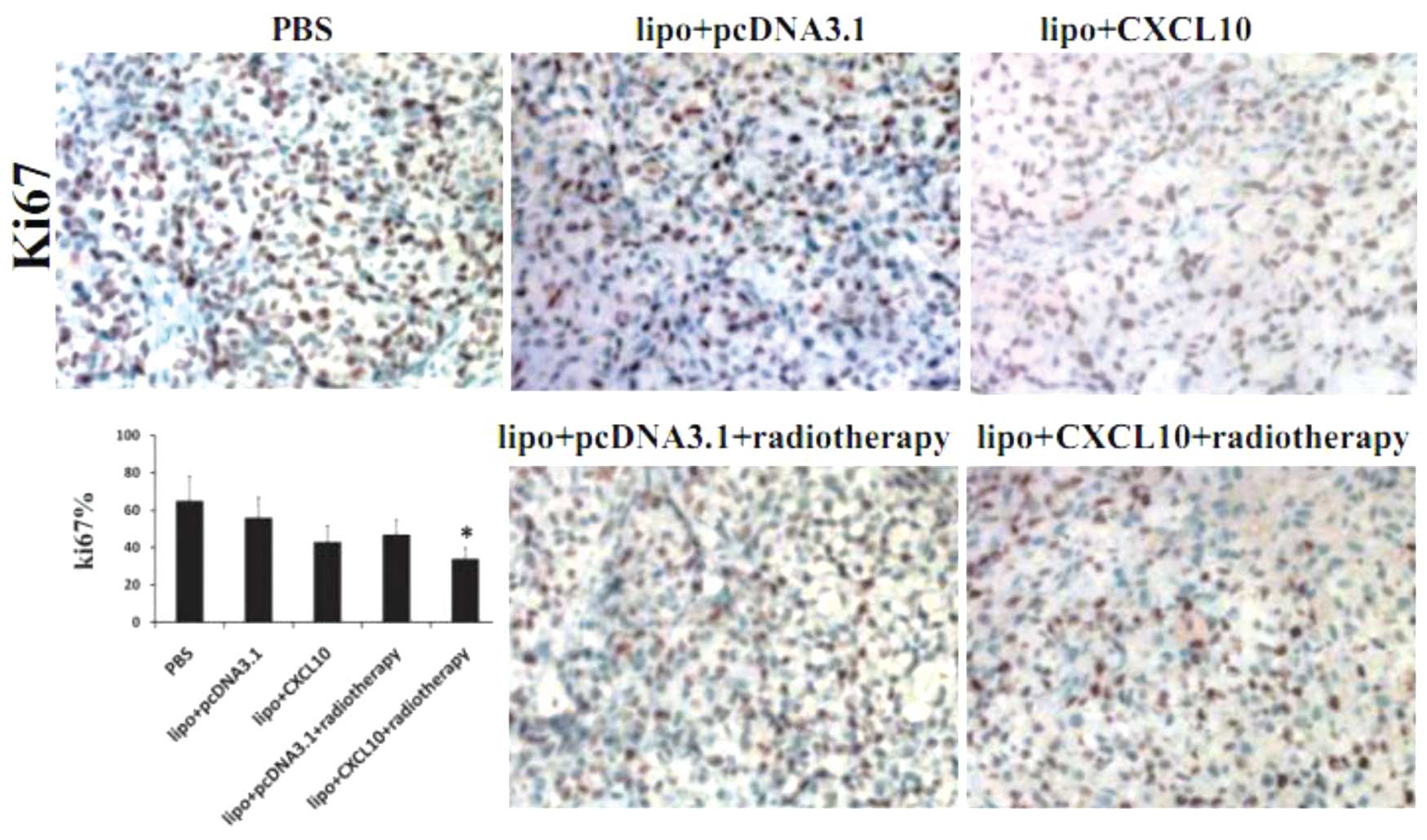
radiotherapy on cell proliferation in cervical cancer
Cell proliferation was analyzed by
immunohistochemical staining of Ki-67 in tumor sections. The number
of Ki-67-positive cells were calculated and divided by the total
number of cells. A slight but significant decrease in the number of
Ki-67-positive cells was observed in the CXCL10 and radiotherapy
groups compared with the control group (P<0.05). Additionally, a
greater reduction in the number of Ki-67-positive cells was
identified in the combination group (P<0.05; Fig. 4).
Effect of CXCL10 gene therapy and
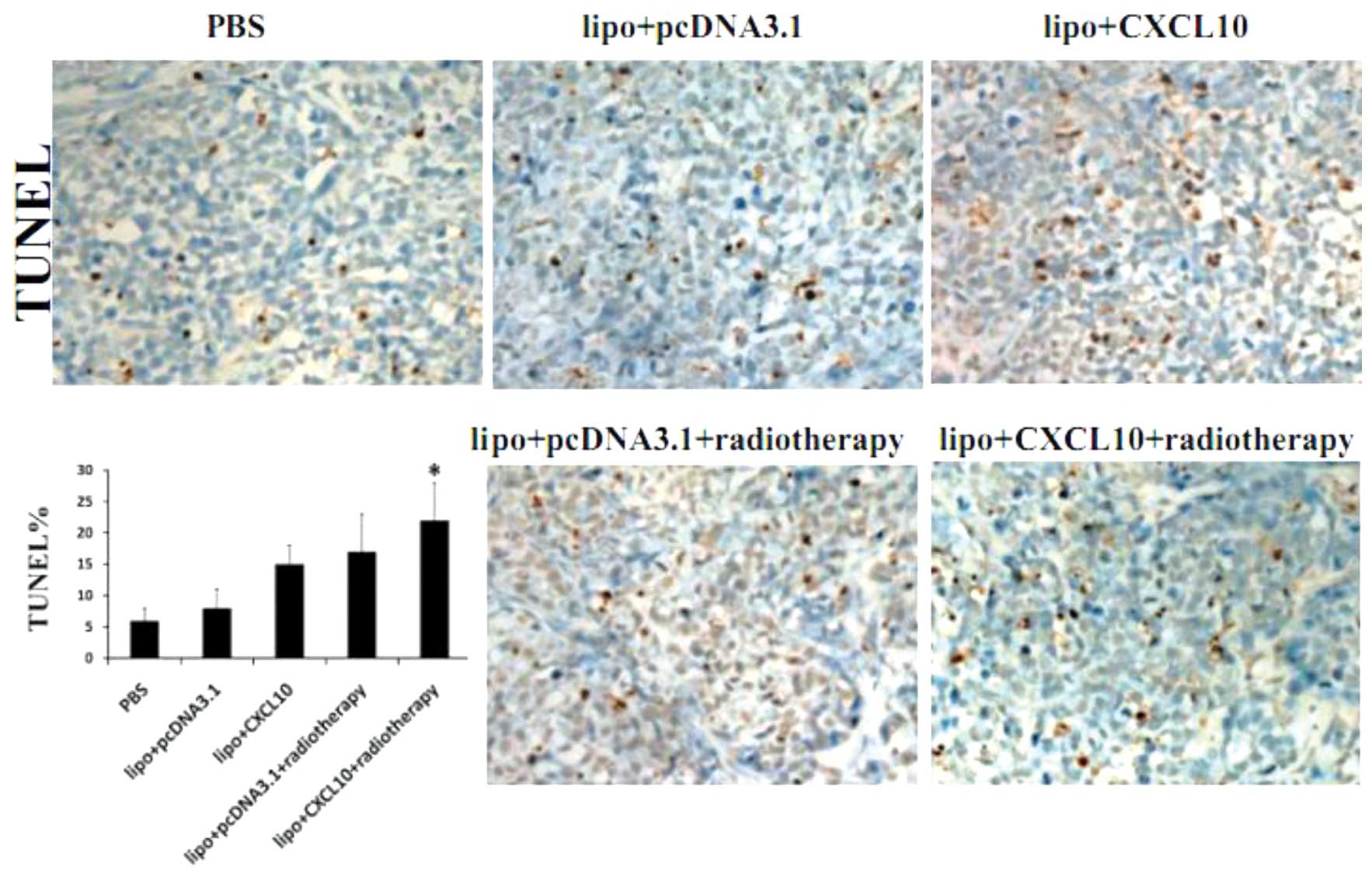
radiotherapy on cell apoptosis in cervical cancer
The apoptotic rate was analyzed by TUNEL assay in
the five groups. As shown in Fig. 5,
the cellular apoptosis rate in the CXCL10 group and radiotherapy
group was found to be ~10% higher compared with the control, while
the rate was even higher in the combination group compared with the
CXCL10 and radiotherapy groups (P<0.05) (Fig. 5).
Discussion
Previously, CXCL10 has been demonstrated to be
involved in cell cycle regulation in a podocyte cell line, with
increased p27Kip1 expression and decreased cyclin E
expression (18). Similar results
were obtained using HeLa cells in previous studies (17). p27Kip1 and cyclin E are
crucial cell cycle regulators that are involved in arresting the
cell cycle at the G1 phase (19) and
promoting cell cycle progression to the S phase (20), respectively. Reduced cyclin E
expression prevents cell cycle transition from the G1 to S phase,
so the cells remain in the G1 phase. Therefore, CXCL10 may inhibit
cell proliferation by influencing cell cycle-associated
proteins.
It has been demonstrated that radiation destructs
genome DNA to induce cell apoptosis (21); however, S phase, during which time
gene synthesis is active in tumor cells, is least sensitive to
radiation (22). Therefore, avoidance
of the S phase in cancer cells is important for the improvement of
the efficacy of radiotherapy. On the basis of our previous analysis
(14), it was assumed that CXCL10 is
a crucial factor that arrests the cell cycle at G1 phase to avoid
the S phase, and therefore promotes the efficacy of
radiotherapy.
In the present study, treatment with
liposome-encapsulated CXCL10 gene therapy and radiotherapy was
combined to assess the resulting inhibition of growth in cervical
cancer tumors. The present results revealed that CXCL10 gene
therapy and radiotherapy each attenuated tumor growth when
administered separately. In addition, the combination of the two
therapies demonstrated a significantly increased inhibitory effect
on tumor growth. To understand the mechanism of the growth
inhibition, tumor sections were analyzed for angiogenesis, cell
proliferation and apoptosis. It was found that CXCL10 gene therapy
combined with radiotherapy enhances the reduction of microvessel
density, decrease in cell proliferation and upregulation of
apoptosis compared with each treatment alone.
Overall, the present study indicated that the
combination of CXCL10 gene therapy and radiotherapy is an efficient
strategy for growth suppression in cervical tumors, which may
result from the attenuation of angiogenesis, decline in cell
proliferation and induction of apoptosis. This approach is novel as
it hypothesizes CXCL10 may enhance the radiosensitivity of cervical
tumors in nude mice. Since the present protocol was not performed
using any other tumor model, whether this approach may be applied
to other tumor types remains unknown, and additional investigations
are required.
Acknowledgements
This study was supported by grants from the National
Natural Science Foundation (grant no. 81201784), Luzhou Medical
College Foundation (grant no. 20130388) and Affliated Hospital of
Luzhou Medical College Foundation (grant no. 201471#).
References
|
1
|
Schiffman M and Castle PE: The promise of
global cervical cancer prevention. N Engl J Med. 353:2101–2104.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mandelblatt JS, Lawrence WF, Womack SM,
Jacobson D, Yi B, Hwang YT, Gold K, Barter J and Shah K: Benefts
and costs of using HPV testing to screen for cervical cancer. JAMA.
287:2372–2381. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Small W Jr, Mell LK, Anderson P, et al:
Consensus guidelines for delineation of clinical target volume for
intensity-modulated pelvic radiotherapy in postoperative treatment
of endometrial and cervical cancer. Int J Radiat Oncol Biol Phys.
71:428–434. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Grigsby PW: Radiotherapy for pelvic
recurrence after radical hysterectomy for cervical cancer. Radiat
Med. 23:327–330. 2005.PubMed/NCBI
|
|
5
|
Klopp AH and Eifel PJ: Chemoradiotherapy
for cervical cancer in 2010. Curr Oncol Rep. 13:77–85. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tanderup K, Georg D, Pötter R, Kirisits C,
Grau C and Lindegaard JC: Adaptive management of cervical cancer
radiotherapy. Semin Radiat Oncol. 20:121–129. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bromley SK, Mempel TR and Luster AD:
Orchestrating the orchestrators: Chemokines in control of T cell
traffic. Nat Immunol. 9:970–980. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Luster AD, Unkeless JC and Ravetch JV:
Gamma-interferon transcriptionally regulates an early-response gene
containing homology to platelet proteins. Nature. 315:672–676.
1985. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Campanella GS, Colvin RA and Luster AD:
CXCL10 can inhibit endothelial cell proliferation independently of
CXCR3. PLoS One. 5:e127002010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Arenberg DA, Kunkel SL, Polverini PJ,
Morris SB, et al: Interferon-gamma-inducible protein 10 (IP-10) is
an angiostatic factor that inhibits human non-small cell lung
cancer (NSCLC) tumorigenesis and spontaneous metastases. J Exp Med.
184:981–992. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Pertl U, Luster AD, Varki NM, Homann D,
Gaedicke G, Reisfeld RA and Lode HN: IFN-gamma-inducible protein-10
is essential for the generation of a protective tumor-specific CD8
T cell response induced by single-chain IL-12 gene therapy. J
Immunol. 166:6944–6951. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Feldman AL, Friedl J, Lans TE, et al:
Retroviral gene transfer of interferon-inducible protein 10
inhibits growth of human melanoma xenografts. Int J Cancer.
99:149–159. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yang LL, Chen P, Luo S, Li J, Liu K, Hu HZ
and Wei YQ: CXC-chemokine-ligand-10 gene therapy efficiently
inhibits the growth of cervical carcinoma on the basis of its
anti-angiogenic and antiviral activity. Biotechnol Appl Biochem.
53:209–216. 2009.PubMed/NCBI
|
|
14
|
Yang LL, Wang BQ, Chen LL, Luo HQ and Wu
JB: CXCL10 enhances radiotherapy effects in HeLa cells through cell
cycle redistribution. Onclogy Lett. 3:383–386. 2012.
|
|
15
|
Xiao F, Wei Y, Yang L, et al: A gene
therapy for cancer based on the angiogenesis inhibitor, vasostatin.
Gene Ther. 9:1207–1213. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Somasundaram K and El-Deiry WS: Inhibition
of p53-mediated transactivation and cell cycle arrest by E1A
through its p300/CBP-interacting region. Oncogene. 14:1047–57.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li G, Tian L, Hou JM, et al: Improved
therapeutic effectiveness by combining recombinant CXC chemokine
ligand 10 with cisplatin in solid tumors. Clin Cancer Res.
11:4217–4224. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Han GD, Suzuki K, Koike H, et al:
IFN-inducible protein-10 plays a pivotal role in maintaining
slit-diaphragm function by regulating podocyte cell-cycle balance.
J Am Soc Nephrol. 17:442–453. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mazumder S, Plesca D and Almasan A: A
jekyll and hyde role of cyclin E in the genotoxic stress response:
switching from cell cycle control to apoptosis regulation. Cell
Cycle. 6:1437–1442. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kawauchi S, Yamamoto Y, Uchida K, Chochi
Y, Kondo T, Oga A and Sasaki K: Signifcance of cyclin E and p27
expression in malignant ovarian germ cell tumors: Correlation with
the cell proliferation activity and clinicopathologic features.
Oncol Rep. 16:1029–1033. 2006.PubMed/NCBI
|
|
21
|
Frosina G: DNA repair and resistance of
gliomas to chemotherapy and radiotherapy. Mol Cancer Res.
7:989–999. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Pawlik TM and Keyomarsi K: Role of cell
cycle in mediating sensitivity to radiotherapy. Int J Radiat Oncol
Biol Phys. 59:928–942. 2004. View Article : Google Scholar : PubMed/NCBI
|