Introduction
Research indicates that the annual incidence of
melanoma continues to increase (1,2).
Metastatic melanoma has a poor prognosis with a median survival
time of 6–8 months and a 5-year survival rate of ~6% with
chemotherapy (3,4).
At present, immunotherapy is the fourth most common
treatment approach for solid tumors, following surgery,
chemotherapy and radiotherapy (5,6). Numerous
immunotherapy approaches using various killer cells for solid
tumors have been reported in the literature, including
tumor-infiltrating lymphocytes (TILs), lymphokine-activated killer
cells (LAKs) and anti-CD3 monoclonal antibody-induced killer cells
(7–9).
The anti-tumor functions of TILs are major histocompatibility
complex (MHC)-restricted and a number of patients with solid tumors
are not eligible for TIL-based therapy since their TILs do not
expand sufficiently, their tumors have lost expression of antigens
or MHC molecules or they possess extremely low numbers of TILs
(7). In addition, LAK and
anti-cluster of differentiation (CD)3 monoclonal antibody-induced
killer cells exhibit low anti-tumor activities (10). Therefore, due to their limited
therapeutic efficacy, they are rarely used in the clinic. In 1991,
Schmidt-Wolf et al (11)
observed a novel type of antitumor effector cell, which was termed
a cytokine-induced killer (CIK) cell. CIK cells proliferate rapidly
in vitro, and possess strong antitumor activity against a
broad spectrum of solid tumors (12).
A number of clinical trials have reported that treating patients
with metastatic solid tumors using CIK cells, alone or in
combination with chemotherapy, significantly improves the median
survival time, and in addition, that CIK cell transfusions may
improve the immune function of patients (12–14). There
is limited data on the use of CIK cell therapy for metastatic
malignant melanoma (15). In the
present study, a patient with metastatic malignant melanoma
received CIK cell immunotherapy, which resulted in a relatively
long overall survival time (OS) of 28 months
Case report
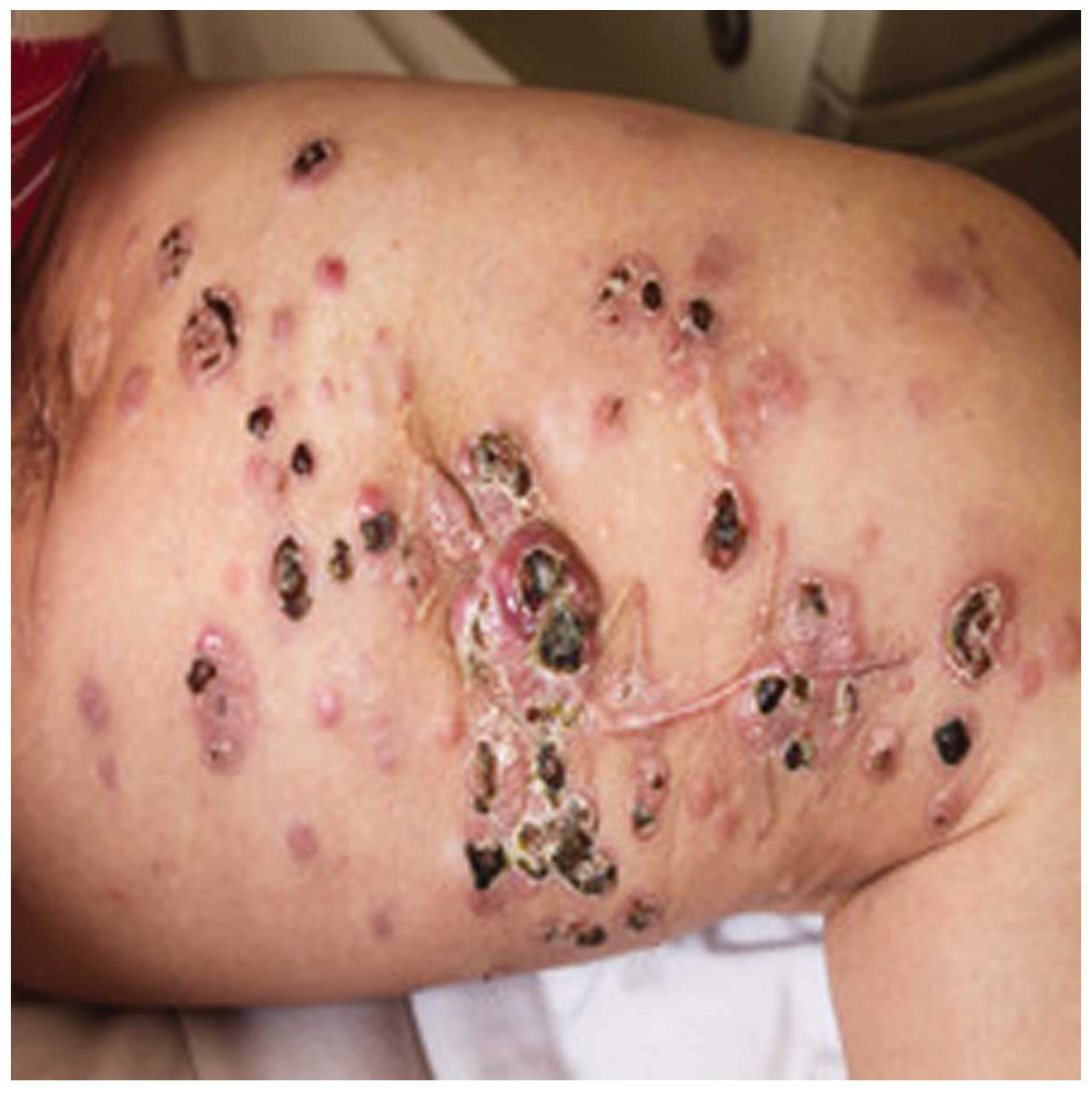
On November 1st, 2010, A 47-year-old female was
admitted to The Affiliated Cancer Hospital of Zhengzhou University
(Zhengzhou, Henan, China) with multiple skin metastatic melanoma
nodes in the left leg (Fig. 1).
Considering that metastatic malignant melanoma is resistant to
chemotherapy and radiotherapy, CIK cell immunotherapy was
administered. Mononuclear cells were collected from 50 ml of the
patient's peripheral blood and cultured in GT-T551 medium (Takara,
Tokyo, Japan) containing anti-CD3 antibody (mouse-anti human
monoclonal IgG2a; catalog no. sc-19590; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA; dilution, 1:300), recombinant
human interleukin-1α (IL-1α; 1,000 U/ml) and interferon-γ (IFN-γ;
1,000 U/ml), at 37°C with 5% CO2 for 24 h. Next,
recombinant human IL-2 (1,000 U/ml) was added to the medium. The
medium was replaced with fresh IL-2 and IFN-γ-containing medium
every 5 days. At day 10, the CIK cells were harvested and their
phenotype was analyzed. All products were free of bacterial,
mycoplasma and fungal contamination. The endotoxin activity was ≤5
EU. Phenotypic analysis of the autologous CIK cells in the patient
prior to culture and following 10 days of culture demonstrated that
the expression of certain antigens in the CIK cells increased
during this time as follows: CD3+, from 44.39±6.21 to
91.36±8.72%; CD3+/CD4+, from 30.08±4.78 to
45.63±7.79%; CD3+/CD8+, from 17.96±6.35 to
36.80±4.45%; CD3+/CD56+, from 3.68±1.47 to
11.98±3.91%; and CD25+, from 17.23±4.45 to 32.67±5.79%,
respectively, with P<0.05 for all differences (t-test;
SPSS software 11.0; SPSS, Inc., Chicago, IL, USA). The results
indicated that the percentages of CD3+,
CD3+/CD4+, CD3+/CD8+
and CD25+ cells were significantly increased following
stimulation and expansion in culture, which is crucial to tumor
immunity. The total number of CIK cells transplanted in 1 cycle of
CIK therapy was ~5×109 cells. Between 10 November 2010
and 27 June 2011, the patient received 8 cycles of CIK cell
immunotherapy and 2×106 units of IL-2 at days 1–5 per
CIK cell infusion. No adverse reactions were observed during the
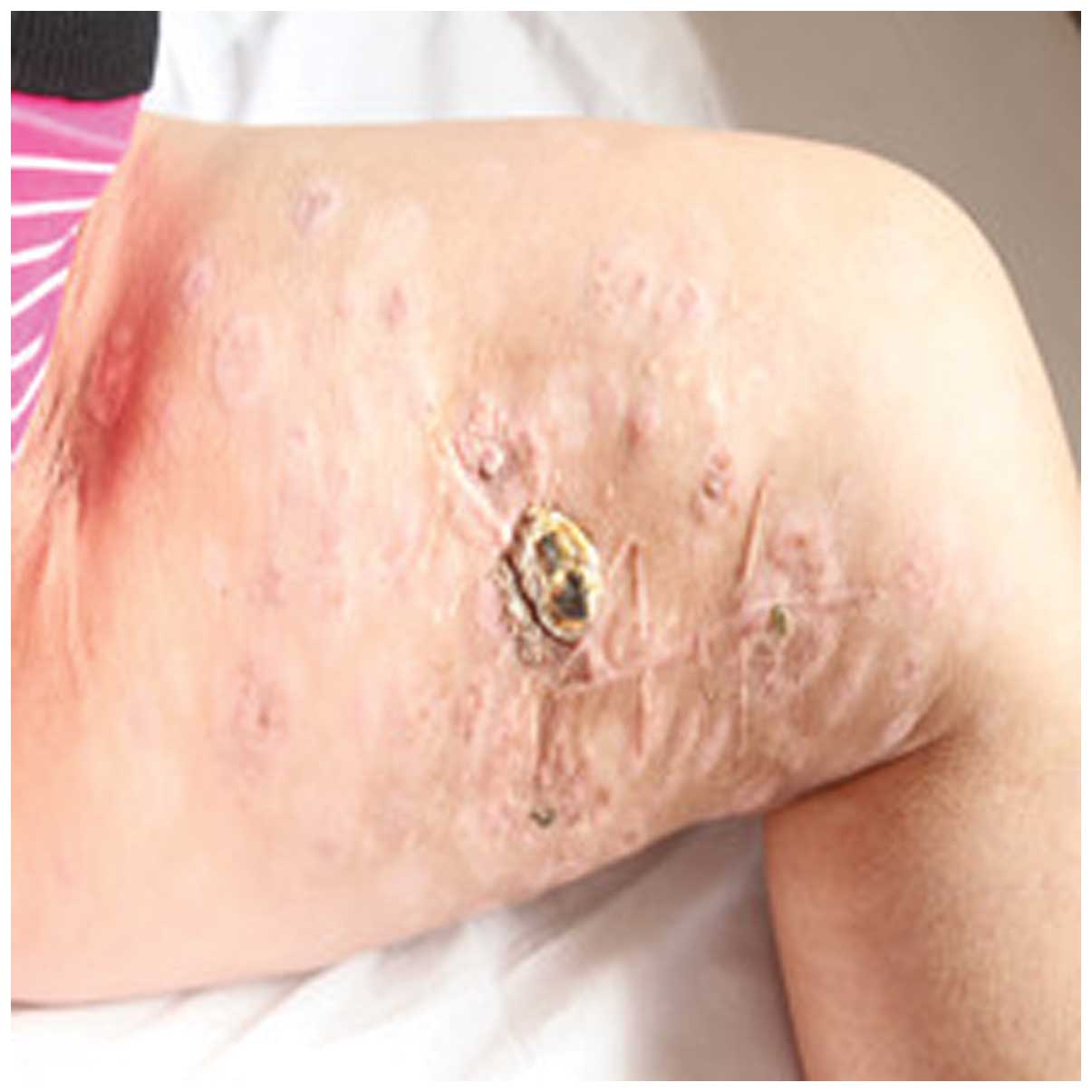
period of CIK cell therapy. Following 8 cycles of CIK cell
immunotherapy, the size and number of metastatic melanoma nodes in
the patient's left leg were notably reduced (Fig. 2). The patient underwent re-examination
every 3 months. At 1 year after the patient's first CIK cell
treatment, re-examination indicated a stable disease state. The
patient then received 4 additional cycles of CIK therapy. However,
2 years later, an abdominal contrast-enhanced CT scan indicated
that multiple nodes were present in the patient's liver and the
treatment was discontinued. The patient succumbed to the disease on
March 10, 2013. The overall survival time was 28 months.
Written informed consent was obtained from the
patient's family for the publication of this study.
Discussion
Patients with metastatic melanoma have a median
survival of 6–8 months and a 5-year survival of 6% (3,4).
FDA-approved treatments for metastatic melanoma including IL-2,
chemotherapy, ipilimumab, vemurafenib, nivolumab and so on, and the
median survival time given these treatments is from 6–24 months
(15,22,23).
Although the survival time of metastatic melanoma patients has
improved, novel regimens remain critical to increase it further. In
1991, Schmidt-Wolf et al (11)
first reported that CIK cells possessed potential antitumor cell
activity in the immunodeficient SCID mouse/human lymphoma model.
Following this finding, additional studies indicated that CIK cells
may be a better choice for patients with solid tumors and
demonstrated that CIKs possess strong antitumor activity in
vitro (24–26). A number of previous studies have also
demonstrated that CIKs may reverse drug resistance in solid tumor
cells (24–26). Therefore CIK cell therapy has been
proposed as an alternative therapeutic strategy to adopt for
patients with solid tumors. Previous studies have demonstrated that
CIK cells are a novel and heterogeneous population of immune
effector cells with a high proliferation rate and potent cytotoxic
activity against a variety of solid tumors, including renal cell
carcinoma, lung cancer and prostate cancer, alone or together with
chemotherapy compared with conventional therapies (Table I) (12,14,20).
 | Table I.Published CIK cell clinical
studies. |
Table I.
Published CIK cell clinical
studies.
| Year | Tumor type | No. of cases | Culture
condition | Therapeutic
approach | Clinical
response |
|---|
| 1999 (16) | RCC, CC,
lymphoma | 10 | IFN-γ, anti-CD3, IL2
plus IL-2 transgene | Auto-CIKs transfected
with IL-2 gene | 1 CR, 3 SD, 6 PD |
| 2006 (17) | GC | 57 | IFN-γ, anti-CD3, IL2
and IL-1α | Arm 1:
Chemotherapy | Arm 1: 4 PR, 8 MR, 6
SD, 7 PD |
|
|
|
| Arm 2: Auto-CIKs
combined with chemotherapy | Arm 2: 7 PR, 11 MR, 8
SD, 6 PD |
| 2008 (18) | NSCLC | 59 | IFN-γ, anti-CD3, IL2
and IL-1α | Arm 1:
Chemotherapy | Arm 1: ORR, 43.3%;
TTP, 4.67; OS, 11 |
|
|
|
|
| Arm 2: Auto-CIKs
combined with chemotherapy | Arm 2: ORR, 44.8%;
TTP, 6.65; OS, 15 |
| 2009 (19) | HCC | 127 | IFN-γ, anti-CD3, IL2
and IL-1α | CIK-I group: 3
courses | The 1-year and
18-month recurrence rates of the CIK-I and CIK-II groups were lower
than that of the control group |
|
|
|
|
| CIK-II group: 6
courses |
|
|
|
|
|
| Control group:
none |
|
| 2012 (14) | RCC | 148 | IFN-γ, anti-CD3, IL2
IL-1β | Arm 1: Auto-CIKs | Arm 1: 13 CR, 26 PR,
25 SD, 10 PD |
|
|
|
|
| Arm 2: IL-2 combined
with IFN-α | Arm 2: 5 CR, 15 PR,
25 SD, 29 PD |
| 2013 (20) | PC | 1 | IFN-γ, anti-CD3, IL2
and IL-1α | Auto-CIKs | CR, PFS ≥19
months. |
| 2013 (21) | HCC | 174 | IFN-γ, anti-CD3, IL2
and IL-1α | Arm 1:
Auto-CIKs+TACE+RFA; | Arm 1: PFS, 17; OS,
56 |
|
|
|
|
| Arm 2: TACE+RFA. | Arm 2: PFS, 10; OS,
31 |
Since CIK cells possess strong antitumor activity
and high amplification efficiency, increasing numbers of studies
have administered CIK cell therapy alone or in combination with
chemotherapy. Previous studies have demonstrated that CIK cells are
a heterogeneous cell population, which express CD3 and CD56 in
addition to the natural killer cell NKG2D activating receptor. CIK
cells possess MHC-unrestricted cytotoxicity towards solid tumors,
but not toward normal targets, and may regulate and enhance the
cellular immune functions in patients with solid tumors by the
secretion of cytokines, such as interferon-γ, and a number of
chemokines, including RANTES, MIP-1α and MIP-1β (27–30).
However, there is currently a lack of studies in which CIK cells
were used to treat metastatic malignant melanoma in clinical
practice (31). The current case
study presents a novel choice of treatment, however, additional
cases are required to verify its efficacy.
In conclusion, the results of the present study
demonstrate that CIK cells may be easily expanded in vitro,
and their transplantation may result in a significant treatment
effect in patients with metastatic malignant melanoma. Furthermore,
CIK cell therapy is a safe technique that exhibits potential
efficacy against metastatic malignant melanoma by significantly
enhancing immune function, by increasing the absolute effector cell
number without notable side-effects. These results may have a
favorable impact on conventional treatments if applied in
large-scale studies.
References
|
1
|
Jemal A, Siegel R, Xu J and Ward E: Cancer
statistics, 2010. CA Cancer J Clin. 60:277–300. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ghosh P and Chin L: Genetics and genomics
of melanoma. Expert Rev Dermatol. 4:1312009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Balch CM, Soong SJ, Gershenwald JE, et al:
Prognostic factors analysis of 17,600 melanoma patients: Validation
of the American Joint Committee on Cancer melanoma staging system.
J Clin Oncol. 19:3622–3634. 2001.PubMed/NCBI
|
|
4
|
Barth A, Wanek LA and Morton DL:
Prognostic factors in 1,521 melanoma patients with distant
metastases. J Am Coll Surg. 181:193–201. 1995.PubMed/NCBI
|
|
5
|
Hontscha C, Borck Y, Zhou H, Messmer D and
Schmidt-Wolf IG: Clinical trials on CIK cells: First report of the
international registry on CIK cells (IRCC). J Cancer Res Clin
Oncol. 137:305–310. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Schwaab T, Schwarzer A, Wolf B, et al:
Clinical and immunologic effects of intranodal autologous tumor
lysate-dendritic cell vaccine with Aldesleukin (Interleukin 2) and
IFN-{alpha}2a therapy in metastatic renal cell carcinoma patients.
Clin Cancer Res. 15:4986–4992. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rosenberg SA, Spiess P and Lafreniere R: A
new approach to the adoptive immunotherapy of cancer with
tumor-infiltrating lymphocytes. Science. 233:1318–1321. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rosenberg S: Lymphokine-activated killer
cells: A new approach to immunotherapy of cancer. J Natl Cancer
Inst. 75:595–603. 1985.PubMed/NCBI
|
|
9
|
Yun YS, Hargrove ME and Ting CC: In vivo
antitumor activity of anti-CD3-induced activated killer cells.
Cancer Res. 49:4770–4774. 1989.PubMed/NCBI
|
|
10
|
Shablak A, Hawkins RE, Rothwell DG and
Elkord E: T cell-based immunotherapy of metastatic renal cell
carcinoma: Modest success and future perspective. Clin Cancer Res.
15:6503–6510. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Schmidt-Wolf IG, Negrin RS, Kiem HP, Blume
KG and Weissman IL: Use of a SCID mouse/human lymphoma model to
evaluate cytokine-induced killer cells with potent antitumor cell
activity. J Exp Med. 174:139–149. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li R, Wang C, Liu L, et al: Autologous
cytokine-induced killer cell immunotherapy in lung cancer: a phase
II clinical study. Cancer Immunol Immunother. 61:2125–2133. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shi L, Zhou Q, Wu J, et al: Efficacy of
adjuvant immunotherapy with cytokine-induced killer cells in
patients with locally advanced gastric cancer. Cancer Immunol
Immunother. 61:2251–2259. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu L, Zhang W, Qi X, et al: Randomized
study of autologous cytokine-induced killer cell immunotherapy in
metastatic renal carcinoma. Clin Cancer Res. 18:1751–1759. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hersey P: Immunotherapy of melanoma. Asia
Pac J Clin Oncol. 6 Suppl:1:S2–S8. 2010. View Article : Google Scholar
|
|
16
|
Schmidt-Wolf IG, Finke S, Trojaneck B, et
al: Phase I clinical study applying autologous immunological
effector cells transfected with the interleukin-2 gene in patients
with metastatic renal cancer, colorectal cancer and lymphoma. Br J
Cancer. 81:1009–1016. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jiang J, Xu N, Wu C, et al: Treatment of
advanced gastric cancer by chemotherapy combined with autologous
cytokine-induced killer cells. Anticancer Res. 26 (3B):2237–2242.
2006.PubMed/NCBI
|
|
18
|
Wu C, Jiang J, Shi L and Xu N: Prospective
study of chemotherapy in combination with cytokine-induced killer
cells in patients suffering from advanced non-small cell lung
cancer. Anticancer Res. 28 (6B):3997–4002. 2008.PubMed/NCBI
|
|
19
|
Hui D, Qiang L, Jian W, Ti Z and Da-Lu K:
A randomized, controlled trial of postoperative adjuvant
cytokine-induced killer cells immunotherapy after radical resection
of hepatocellular carcinoma. Dig Liver Dis. 41:36–41. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li W, Xu LP, DI Zhao L, et al:
Cytokine-induced killer cell therapy for advanced pancreatic
adenocarcinoma: A case report and review of the literature. Oncol
Lett. 5:1427–1429. 2013.PubMed/NCBI
|
|
21
|
Huang ZM, Li W, Li S, et al:
Cytokine-induced killer cells in combination with transcatheter
arterial chemoembolization and radiofrequency ablation for
hepatocellular carcinoma patients. J Immunother. 36:287–293. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Alexandrescu DT, Ichim TE, Riordan NH, et
al: Immunotherapy for melanoma: Current status and perspectives. J
Immunother. 33:570–590. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Robert C, Long GV, Brady B, et al:
Nivolumab in previously untreated melanoma without BRAF mutation. N
Engl J Med. 372:320–330. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhao Q, Zhang H, Li Y, Liu J, Hu X and Fan
L: Anti-tumor effects of CIK combined with oxaliplatin in human
oxaliplatin-resistant gastric cancer cells in vivo and in vitro. J
Exp Clin Cancer Res. 29:1182010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang FS, Liu MX, Zhang B, et al: Antitumor
activities of human autologous cytokine-induced killer (CIK) cells
against hepatocellular carcinoma cells in vitro and in vivo. World
J Gastroenterol. 8:464–468. 2002.PubMed/NCBI
|
|
26
|
Zhang YS, Yuan FJ, Jia GF, et al: CIK
cells from patients with HCC possess strong cytotoxicity to
multidrug-resistant cell line Bel-7402/R. World J Gastroenterol.
11:3339–3345. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Karimi M, Cao TM, Baker JA, Verneris MR,
Soares L and Negrin RS: Silencing human NKG2D, DAP10 and DAP12
reduces cytotoxicity of activated CD8+ T cells and NK cells. J
Immunol. 175:7819–7828. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Verneris MR, Karami M, Baker J, Jayaswal A
and Negrin RS: Role of NKG2D signaling in the cytotoxicity of
activated and expanded CD8+ T cells. Blood. 103:3065–3072. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Schmidt-Wolf IG, Lefterova P, Mehta BA, et
al: Phenotypic characterization and identification of effector
cells involved in tumor cell recognition of cytokine-induced killer
cells. Exp Hematol. 21:1673–1679. 1993.PubMed/NCBI
|
|
30
|
Joshi PS, Liu JQ, Wang Y, et al:
Cytokine-induced killer T cells kill immature dendritic cells by
TCR-independent and perforin-dependent mechanisms. J Leukoc Biol.
80:1345–1353. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gammaitoni L, Giraudo L, Leuci V, et al:
Effective activity of cytokine-induced killer cells against
autologous metastatic melanoma including cells with stemness
features. Clin Cancer Res. 19:4347–4358. 2013. View Article : Google Scholar : PubMed/NCBI
|