Introduction
Since the late 20th century, a large number of
molecular targeted therapeutic drugs, such as epidermal growth
factor receptor tyrosine kinase inhibitors, have been developed for
cancer chemotherapy. However, platinum-based combination
chemotherapy continues to be important for the treatment of various
cancers, including lung cancer. Cisplatin is a commonly used drug
for combination chemotherapy (1).
Although cisplatin is a potent anticancer drug, various malignant
tumors frequently acquire resistance to cisplatin, limiting the
clinical application of this agent (2,3).
Arsenite is a toxic metalloid that is widely
distributed in the environment (4).
Despite the toxicity, arsenic-containing compounds have been used
in traditional Chinese medicine for >2,000 years (5). In addition, arsenic trioxide (ATO) has
been approved in numerous countries for the treatment of acute
promyelocytic leukemia (APL) (6).
In vitro studies have revealed that a combination of
ATO and cisplatin exerts a synergistic inhibitory effect on the
growth of ovarian cancer and small cell lung cancer cells (7–9).
It is well known that adenosine triphosphate-binding
cassette (ABC) transporters are important molecules for the
resistance of cancer cells to chemotherapy agents and the
detoxification of toxic metals in xenobiotic metabolism (10). Leslie et al previously reported
that ABC subfamily C, member 1 (ABCC1) and ABC, subfamily C, member
2 (ABCC2) each play an important role in multidrug resistance and
arsenic detoxification in cells, through the cellular efflux of the
glutathione (GSH) conjugate (11).
The present study aimed to examine the sensitivity
of the non-small cell lung cancer (NSCLC) PC-9 cell line and
cisplatin-resistant PC-9/CDDP subline to toxic metals. The
cisplatin-resistant PC-9/CDDP cell line demonstrated
hypersensitivity to arsenite and arsenate. Furthermore, the
accumulation of arsenite was decreased in PC-9/CDDP cells relative
to the parental cells. Notably, the sensitivity to arsenite and
accumulation of arsenite were markedly increased compared with the
cisplatin-resistant cell line. Therefore, it was hypothesized that
the GS-X pump systems were downregulated in the cisplatin resistant
NSCLC cell line. In addition, the arsenic-containing
chemotherapeutic agent ATO was applied to the cisplatin-sensitive
and -resistant NSCLC cells, and it was found that the
cisplatin-resistant NSCLC cell lines PC-9/CDDP and PC-14/CDDP were
hypersensitive to ATO.
The results of the present study suggest that
arsenite accumulation depends on the GS-X pump and that treatment
with ATO may overcome certain cases of cisplatin resistance in
NSCLC.
Materials and methods
Cell lines and materials
The NSCLC PC-9 and PC-14 cell lines, which were
derived from untreated patients with pulmonary adenocarcinoma, were
provided by Professor Y. Hayata (Tokyo Medical College, Tokyo,
Japan). The cisplatin-resistant PC-9/CDDP and PC-14/CDDP sublines
were established at the National Cancer Center Research Institute
(Tokyo, Japan) (12).
The mouse anti-human ABCC1 monoclonal antibody
(clone number, QCRL1; ALX-801-010) and mouse anti-human ABCC2
monoclonal antibody (clone number, M2I-4; ALX-801-015) were
obtained from Enzo Life Sciences, Inc. (Farmingdale, NY, USA). The
mouse anti-human GST-π monoclonal antibody (clone number, GST-π/3;
610718) was obtained from BD Biosciences (Franklin Lakes, NJ, USA).
The Vectastain ABC kit and horseradish peroxidase-conjugated
anti-mouse immunoglobulin G secondary antibody were obtained from
Vector Laboratories (Burlingame, CA, USA). Enhanced
chemiluminescence (ECL) reagent and Hyperfilm were obtained from GE
Healthcare Biosciences (Pittsburgh, PA, USA). Sodium serenate,
sodium selenite, sodium arsenite and sodium arsenate were obtained
from Sigma-Aldrich (St. Louis, MO, USA). Antimony and cadmium
chloride were obtained from Nacalai Tesque, Inc. (Kyoto, Japan).
Arsenic trioxide (Trisenox) was obtained from Nippon Shinyaku Co.
Ltd. (Kyoto, Japan). All other chemicals were obtained from Nacalai
Tesque, Inc. or Wako Pure Chemical Industries, Ltd. (Osaka,
Japan).
Cellular cytotoxicity assay to
determine sensitivity to metals
Cell counting kit-8 was obtained from Dojindo
Molecular Technologies, Inc. (Kumamoto, Japan) and, with MTT, was
used for colorimetric assays to determine the cell viability, as
previously described (13). The cells
were plated on 96-well microplates at a density of 2×103
cells per well, incubated overnight, and then treated with various
concentrations of arsenate, arsenite, selenate, selenite, mercury,
cadmium or antimony for 72 h.
Determination of arsenite
accumulation
The cells were plated on 6-cm dishes at a density of
1×106 cells per dish, incubated overnight, and then
treated with 100 µM sodium arsenite for 4 h. Subsequent to washing
with ice-cold PBS (–), the cells were immediately harvested using a
cell scraper. The cells were pelleted by centrifugation, and the
cell pellets were extracted with 20 mM Tris-HCl (pH 7.4) containing
1% Triton-X 100 and 0.1% SDS. The arsenite concentrations in the
cells were determined by atomic absorption spectrometry (AAS;
Z-9000; Hitachi, Ltd., Tokyo, Japan).
Analysis of the GSH concentration and
activity of GSH-associated enzymes
The intracellular GSH concentration was measured
using 5,5-dithiobis(2-nitrobenzoic acid) (DTNB). Cell lysates were
diluted in 100 mM sodium phosphate buffer (pH 7.4) containing 2.5
mM EDTA, 0.25 mM NADPH and 1 unit/ml glutathione reductase
(Oriental Yeast, Co., Ltd., Tokyo, Japan). The reaction was then
initiated by adding 200 µM DTNB, and the absorbance at 415 nm was
subsequently measured. The amount of total GSH was determined by
comparing the absorbance of the cell lysate with a GSH standard
curve.
The activity of γ-glutamyl cysteine synthetase
(γ-GCS) was measured using a previously reported high-performance
liquid chromatography (HPLC) method (14), with slight modification. A 10-µl
aliquot of cell lysate was diluted in 100 µl of assay mixture
containing 75 mM KCl, 10 mM MgCl2, 0.2 mM EDTA, 10 mM
adenosine triphosphate, 10 mM cysteine, 5 mM glutamate and 100 mM
Tris-HCl buffer (pH 7.4). Following 20 min of incubation at 37°C,
the reaction was terminated by the addition of 20 µl of 15%
trichloroacetic acid, and the assay mixture was then chilled on
ice. Subsequent to centrifugation, quantitation of Glu-Cys was
conducted by post-labeled fluorescence detection HPLC using the
ortho-phthalaldehyde reagent (Nacalai Tesqe, Inc., Kyoto,
Japan).
Glutathione-S-transferase (GST) activity was
determined using standard spectrophotometric assays by monitoring
the formation of the conjugate of 1-chloro-2,4-dinitrobenzene
(CDNB) and reduced glutathione (GSH) (15).
GST-π protein expression was determined using
immunoblotting. In total, 20 µg of protein was loaded onto 12.5%
(w/v) SDS-polyacrylamide gels and subjected to electrophoresis.
Subsequent to electrotransfer onto polyvinylidene difluoride (PVDF)
membranes (Bio-Rad Laboratories, Hercules, CA, USA), the membranes
were blocked with Block Ace (Yukijirushi-Nyugyo, Co., Sapporo,
Japan) according to the manufacturer's instructions. Probing was
performed overnight at 4°C with the monoclonal mouse anti-GST-π
antibody diluted with PBS containing 0.1% Tween-20 (1:200; clone
number, 3; BD Biosciences, Franklin Lakes, NJ, USA). Subsequent to
washing, specific signals were detected using horseradish
peroxidase-conjugated anti-mouse immunoglobulin G secondary
antibody diluted with PBS containing 0.1% Tween-20 (1:10,000) and
ECL reagent according to the manufacturer's instructions (GE
Healthcare Biosciences, Pittsburgh, PA, USA).
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR) for
mRNA
Total RNA was isolated using an RNeasy Mini kit
(Qiagen, Hilden, Germany). Reverse transcription was performed on 1
µg of total RNA using High Capacity RNA-to-cDNA Master Mix (Applied
Biosystems, Foster City, CA, USA). RT-qPCR was performed using
converted cDNA and the following reagents: TaqMan Gene Expression
Assays for human ABCC1 (assay ID, Hs00219905_m1), ABCC2 (assay ID,
Hs00166123_m1), ABCC3 (assay ID, Hs00978473_m1), ABCC4 (assay ID,
Hs00988717_m1), ABCC5 (assay ID, Hs00981087_m1) and GAPDH (assay
ID, Hs99999901_s1) genes, and the TaqMan Universal PCR Master Mix,
no AmpErase UNG (Thermo Fisher Scientific Inc., Waltham, MA). An
ABI PRISM 7300 Sequence Detector System (Thermo Fisher Scientific
Inc.) was used to collect the signals according to the
manufacturer's instructions. The relative gene expression was
determined using the comparative cycle threshold (CT) method based
on the normalization of the target gene against the GAPDH reference
gene. ΔCT was calculated by subtracting the CT of GAPDH from the CT
of the target genes.
Immunoblotting of ABCC1 and ABCC2
Plasma membrane fractions were prepared by
ultracentrifugation, as previously described (16). Plasma membrane fractions (50 µg
protein) were loaded onto 7.5% (w/v) SDS-polyacrylamide gels and
resolved by electrophoresis. Following electrotransfer onto PVDF
membranes, probing was performed overnight at 4°C with the
anti-ABCC1 antibody or anti-ABCC2 antibody. Subsequent to washing,
specific signals were detected using the Vectastain ABC system and
ECL chemiluminescence reagent.
Statistical analysis
The results are expressed as the mean ± standard
deviation. Statistical evaluations were performed using Student's
t-tests with Microsoft Excel 2013 (Microsoft Corporation, Redmond,
WA, USA) for the analysis.
Results
Cross-resistance to metals
To elucidate the mechanism underlying cisplatin
resistance, an analysis of the sensitivity of the PC-9 and
PC-9/CDDP cell lines to various metals was performed. The
IC50 values of various metal compounds in the parental
PC-9 and cisplatin-resistant PC-9/CDDP cell lines are reported in
Table I. Only selenate (VI)
demonstrated an increased cross-resistance in PC-9/CDDP cells
compared with parental PC-9 cells, with a 1.90-fold increase. By
contrast, antimony (IV), arsenate (V), arsenite (III) and mercury
(II) were revealed to be more potent inhibitors of PC-9/CDDP cells
compared with the effect on the parental PC-9 cell line. No
difference in sensitivity was observed for the other metals between
the PC-9/CDDP and parental PC-9 cells.
 | Table I.Cross-sensitivity for various metals
in the cisplatin-resistant PC-9/CDDP cell subline. |
Table I.
Cross-sensitivity for various metals
in the cisplatin-resistant PC-9/CDDP cell subline.
|
| IC50,
µM |
|
|---|
|
|
|
|
|---|
| Metal compounds | PC-9 | PC-9/CDDP | Relative
resistance |
|---|
| Antimony (IV) |
97.5±5.0 |
25.0±10.0a |
0.26±0.10 |
| Arsenite (III) |
22.5±5.0 |
3.3±1.3a |
0.14±0.03 |
| Arsenate (Ⅴ) |
240.0±89.4 |
19.0±2.2a |
0.09±0.02 |
| Selenite (IV) |
9.8±0.4 |
9.8±0.4 |
1.00±0.00 |
| Selenate (VI) |
140.0±54.8 |
260.0±134.1 |
1.90±0.55 |
| Mercury (II) |
92.5±9.6 |
22.5±12.6 |
0.25±0.14 |
| Cadmium (II) |
62.5±22.2 |
70.0±21.6 |
1.15±0.24 |
| Cisplatin (II) |
4.1±0.3 |
23.8±4.8 |
5.76±1.20 |
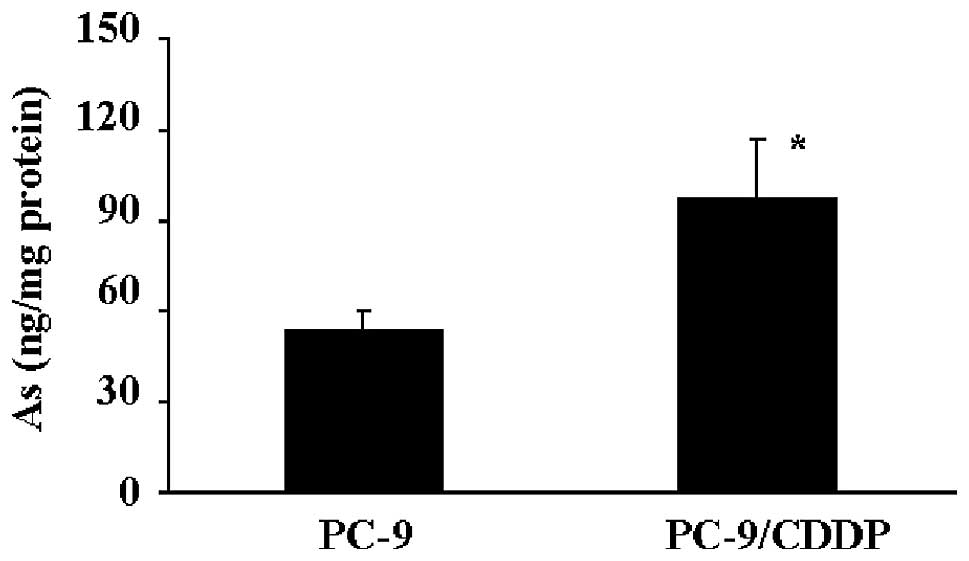
Arsenite accumulation
The PC-9/CDDP cells demonstrated hypersensitivity to
arsenite and arsenate. To elucidate the mechanisms of arsenite
sensitivity in the cisplatin-resistant PC-9/CDDP cell line, the
accumulation of arsenite was measured. When the cells were
incubated in medium containing 100 mM sodium arsenite for 4 h, the
accumulation of arsenite in PC-9/CDDP cells was increased ~1.7-fold
compared with the accumulation demonstrated by PC-9 cells (Fig. 1). When the cisplatin accumulation in
the cells was determined under the same conditions, the cisplatin
concentration in the PC-9/CDDP cells was found to be markedly
decreased, demonstrating an accumulation of ~20% of the cisplatin
found in the parental PC-9 cells (data not shown).
Analysis of the GSH concentration and
activity of GSH-associated enzymes
GS-X pump systems are an important mechanism by
which metals, including cisplatin and arsenite, are transported
(17,18). To investigate the molecular basis for
the decreased accumulation of arsenite, the GS-X pump systems were
analyzed, including cellular GSH levels, activity of the GSH
synthesis rate-limiting enzyme γ-GCS, GST activity and expression
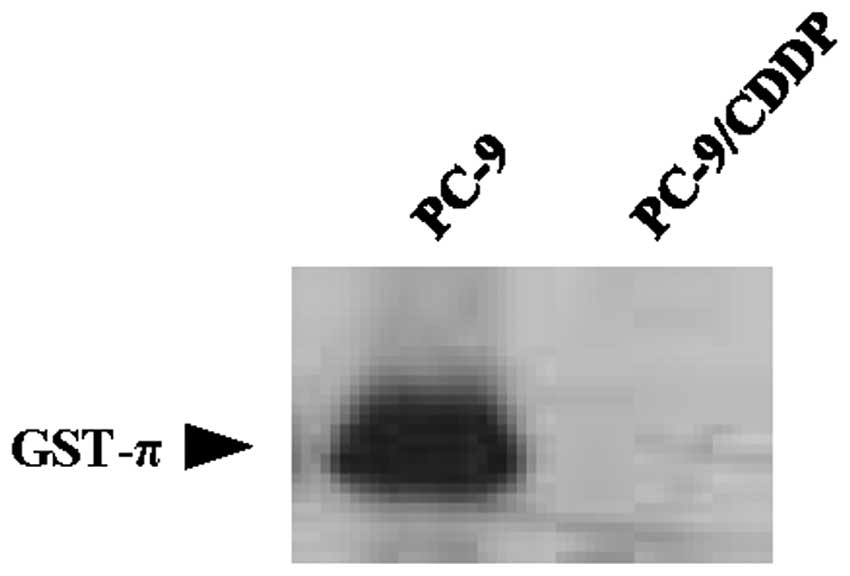
of the ABC transporters. Cellular GSH levels in PC-9/CDDP cells
were 65% of those measured in the parental PC-9 cell line. γ-GCS
activity was also downregulated in PC-9/CDDP cells, where it was
67% of the activity measured in PC-9 cells. GST activity was also
extremely decreased to ~4.0% of the activity observed in the
parental cell line (Table II). The
protein expression of GST-π was also downregulated in PC-9/CDDP
cells compared with the parental cell line (Fig. 2).
 | Table II.Concentration of GSH and activity of
associated enzymes in the cisplatin-resistant PC-9/CDDP cell
subline. |
Table II.
Concentration of GSH and activity of
associated enzymes in the cisplatin-resistant PC-9/CDDP cell
subline.
| Cell line | GSH, nmol/mg | GCS, unit/mg | GST, unit/mg |
|---|
| PC-9 | 144.0 | 1.70 | 0.397 |
| PC-9/CDDP | 95.5 | 1.13 | 0.016 |
| Ratio, % of PC-9 | 66.3 | 66.5 | 4.0 |
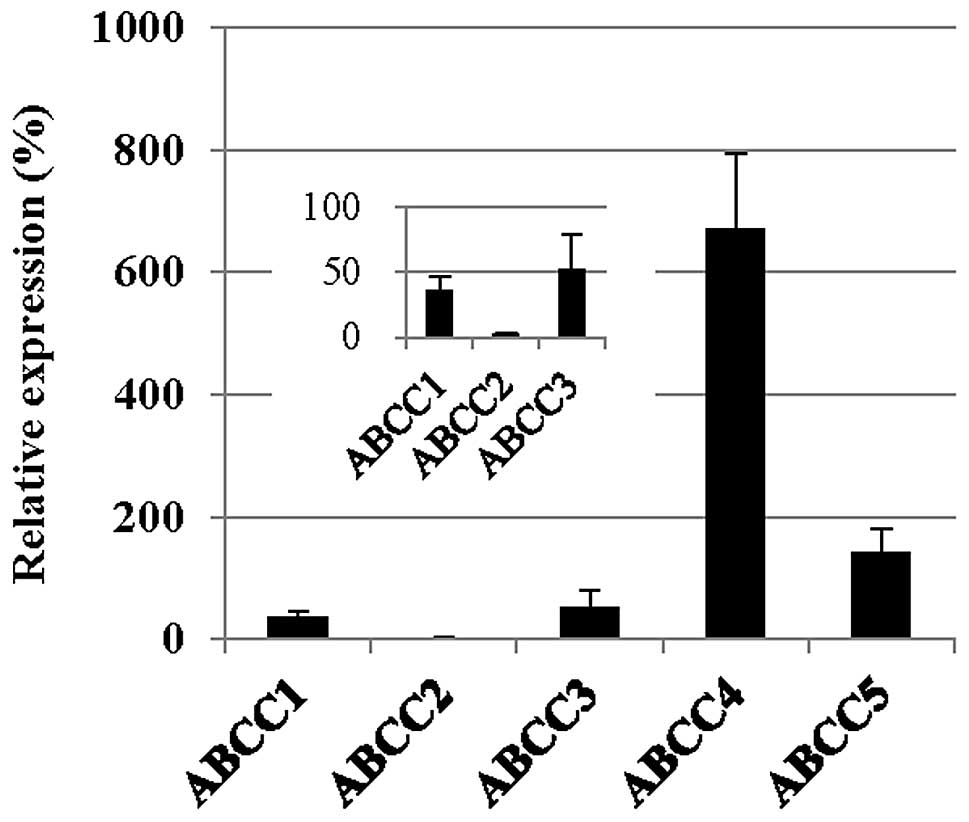
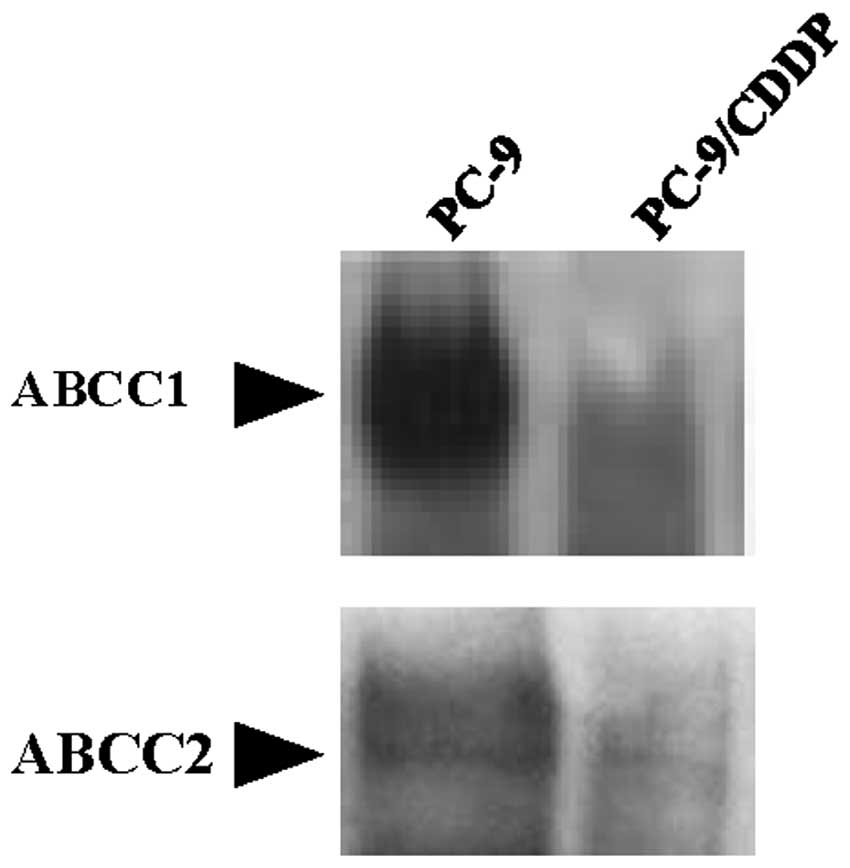
Analysis of the expression of ABCC
transporters
It has previously been indicated that arsenite is a
substrate for the GS-X pump, as a GS-As conjugate (11). As shown in Fig. 3, the expression of ABCC1, 2 and 3 was
downregulated in the PC-9/CDDP cells, with mRNA expression levels
of 36.0, 2.7 and 51.9% of the level measured in the parental PC-9
cell line, respectively. The protein levels of ABCC1 and 2 in the
plasma membrane were also decreased in the PC-9/CDDP cells compared
with the parental cells. In western blot analysis, only faint bands
corresponding to ABCC1 and 2 were obtained from the fractions
derived from the PC-9/CDDP plasma membrane (Fig. 4).
Sensitivity of the cisplatin-resistant
lung cancer PC-9/CDDP and PC-14/CDDP cell lines to ATO
As aforementioned, the cisplatin-resistant PC-9/CDDP
cell line was revealed to demonstrate hypersensitivity to arsenite.
Additionally, the cytotoxic effect of the arsenite-containing
chemotherapeutic agent ATO was examined in the cisplatin-resistant
NSCLC PC-9/CDDP and PC-14/CDDP cell lines.
The findings revealed that the PC-9/CDDP and
PC-14/CDDP cell lines each demonstrated a 3-fold increase in
sensitivity to ATO compared with the PC-9 and PC-14 parental cell
lines (Table III).
 | Table III.Sensitivity to arsenic trioxide in the
parent and cisplatin-resistant non-small cell lung cancer PC-9 and
PC-14 cell lines. |
Table III.
Sensitivity to arsenic trioxide in the
parent and cisplatin-resistant non-small cell lung cancer PC-9 and
PC-14 cell lines.
|
| IC50,
µM |
|
|---|
|
|
|
|
|---|
| Cell line | Parent | Cisplatin
resistant | Relative resistance,
fold of parent |
|---|
| PC-9 |
5.6±1.3 |
1.4±0.2 |
0.3±0.1 |
| PC-14 |
4.5±2.7 |
1.1±0.7 |
0.3±0.1 |
Discussion
Cisplatin is a coordination compound of platinum.
Certain cisplatin-resistant cell lines demonstrate cross-resistance
to metals that include cadmium, antimony and arsenite (19). The identification of cross-resistant
metals aids the evaluation of the mechanism underlying cisplatin
resistance and metal transport systems. Previously,
cisplatin-resistant cell lines were developed from metallothionine
knockout (MT-KO) cells, and these cell lines demonstrated
cross-resistance to arsenite and decreased arsenite accumulation
(20). These results indicate the
presence of a mechanism by which arsenite and cisplatin resistance
in MT-KO cell lines depends on the same transport systems.
By contrast, an in vitro study has revealed
that the combined administration of cisplatin and ATO demonstrates
a synergistic inhibitory effect on the growth of various cancer
cell lines, including squamous carcinoma, ovarian cancer and small
cell lung cancer cells (7–9,21).
In the present study, the cisplatin-resistant
PC-9/CDDP cell line was revealed to be hypersensitive to arsenite.
A marked decrease in the accumulation of arsenite was also observed
in the PC-9/CDDP cells. These results indicate that the transport
systems for cisplatin and arsenite are independent in PC-9 cells.
Consequently, hypersensitivity to arsenite in PC-9/CDDP cells
appears to be dependent on the cellular accumulation of
arsenite.
It has previously been reported that the mechanisms
of arsenite accumulation are mediated by transporter systems,
influx transporters, including the aquaglyceroporins aquaporin
(AQP)7 and AQP9 (22), and efflux
transporters, such as ABCC1 and ABCC2 (11). Although the cisplatin uptake
transporter was not identified in the present cell lines, the
transporter of cisplatin in PC-9/CDDP cells may not contribute to
the uptake of arsenite.
The present results clearly reveal that GSH and GS-X
pump-associated factors, including GST activity and ABC transporter
expression, were downregulated in PC-9/CDDP cells. GSH and GST are
important for the formation of the arsenic-glutathione conjugate,
which is a good substrate for ABCC1 and ABCC2.
In conclusion, ATO, an approved drug, potentially
possesses sensitivity for cisplatin-resistant NSCLC cell lines. The
mechanism of ATO hypersensitivity is dependent on the
downregulation of GS-X pump systems, particularly GSH, GST, ABCC1
and ABCC2.
The aforementioned findings indicate that ATO has
the potential to treat cisplatin resistance in NSCLC and that ABCC1
and ABCCC2 are important biomarkers when considering a
chemotherapeutic regimen containing ATO for the treatment of
cisplatin-resistant NSCLC.
Acknowledgements
This study was supported by the Japan Society for
the Promotion of Science (JSPS) Grants-in-Aid for Scientific
Research KAKENHI (grant nos., 20790147, 22790173 and 24590218).
References
|
1
|
Dasari S and Tchounwou PB: Cisplatin in
cancer therapy: Molecular mechanisms of action. Eur J Pharmacol.
740:364–378. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nishio K, Nakamura T, Koh Y, Suzuki T,
Fukumoto H and Saijo N: Drug resistance in lung cancer. Curr Opin
Oncol. 11:109–115. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Suzuki T, Nishio K and Tanabe S: The MRP
family and anticancer drug metabolism. Curr Drug Metab. 2:367–377.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nordstrom DK: Public health. Worldwide
occurrences of arsenic in ground water. Science. 296:2143–2145.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rao Y, Li R and Zhang D: A drug from
poison: How the therapeutic effect of arsenic trioxide on acute
promyelocytic leukemia was discovered. Sci China Life Sci.
56:495–502. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lengfelder E, Hofmann WK and Nowak D:
Impact of arsenic trioxide in the treatment of acute promyelocytic
leukemia. Leukemia. 26:433–442. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang N, Wu ZM, McGowan E, Shi J, Hong ZB,
Ding CW, Xia P and Di W: Arsenic trioxide and cisplatin synergism
increase cytotoxicity in human ovarian cancer cells: Therapeutic
potential for ovarian cancer. Cancer Sci. 100:2459–2464. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Helm CW and States JC: Enhancing the
efficacy of cisplatin in ovarian cancer treatment-could arsenic
have a role. J Ovarian Res. 2:22009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zheng CY, Lam SK, Li YY, Fong BM, Mak JC
and Ho JC: Combination of arsenic trioxide and chemotherapy in
small cell lung cancer. Lung Cancer. 82:222–230. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Szakács G, Paterson JK, Ludwig JA,
Booth-Genthe C and Gottesman MM: Targeting multidrug resistance in
cancer. Nat Rev Drug Discov. 5:219–234. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Leslie EM: Arsenic-glutathione conjugate
transport by the human multidrug resistance proteins (MRPs/ABCCs).
J Inorg Biochem. 108:141–149. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hong WS, Saijo N, Sasaki Y, Minato K,
Nakano H, Nakagawa K, Fujiwara Y, Nomura K and Twentyman PR:
Establishment and characterization of cisplatin-resistant sublines
of human lung cancer cell lines. Int J Cancer. 41:462–467. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Uemura M, Suzuki T, Nishio K, Chikuma M
and Komeda S: An in vivo highly antitumor-active
tetrazolato-bridged dinuclear platinum (II) complex largely
circumvents in vitro cisplatin resistance: Two linkage isomers
yield the same product upon reaction with 9-ethylguanine but
exhibit different cytotoxic profiles. Metallomics. 4:686–692. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mukai Y, Togawa T, Suzuki T, Ohata K and
Tanabe S: Determination of homocysteine thiolactone and
homocysteine in cell cultures using high-performance liquid
chromatography with fluorescence detection. J Chromatogr B Analyt
Technol Biomed Life Sci. 767:263–268. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Habig WH, Pabst MJ, Fleischner G,
Gatmaitan Z, Arias IM and Jakoby WB: The identity of glutathione
S-transferase B with ligandin, a major binding protein of liver.
Proc Natl Acad Sci USA. 71:3879–3882. 1974. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yoshikawa M, Ikegami Y, Hayasaka S, Ishii
K, Ito A, Sano K, Suzuki T, Togawa T, Yoshida H and Soda H: Novel
camptothecin analogues that circumvent ABCG2-associated drug
resistance in human tumor cells. Int J Cancer. 110:921–927. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kurokawa H, Ishida T, Nishio K, Arioka H,
Sata M, Fukumoto H, Miura M and Saijo N: Gamma-glutamylcysteine
synthetase gene overexpression results in increased activity of the
ATP-dependent glutathione S-conjugate export pump and cisplatin
resistance. Biochem Biophys Res Comm. 216:258–264. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ishikawa T, Bao JJ, Yamane Y, Akimaru K,
Frindrich K, Wright CD and Kuo MT: Coordinated induction of
MRP/GS-X pump and gamma-glutamylcysteine synthetase by heavy metals
in human leukemia cells. The Journal of biological chemistry.
271:14981–14988. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Naredi P, Heath DD, Enns RE and Howell SB:
Cross-resistance between cisplatin, antimony potassium tartrate and
arsenite in human tumor cells. J Clin Invest. 95:1193–1198. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Suzuki T, Ohata S, Togawa T, Himeno S and
Tanabe S: Arsenic accumulation decreased in metallothionein null
Cisplatin-resistant cell lines. J Toxicol Sci. 32:321–328. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nakaoka T, Ota A, Ono T, Karnan S, Konishi
H, Furuhashi A, Ohmura Y, Yamada Y, Hosokawa Y, Kazaoka Y, et al:
Combined arsenic trioxide-cisplatin treatment enhances apoptosis in
oral squamous cell carcinoma cells. Cell Oncol (Dordr). 37:119–129.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Liu Z, Shen J, Carbrey JM, Mukhopadhyay R,
Agre P and Rosen BP: Arsenite transport by mammalian
aquaglyceroporins AQP7 and AQP9. Proc Natl Acad Sci USA.
99:6053–6058. 2002. View Article : Google Scholar : PubMed/NCBI
|