Introduction
Glioblastoma multiforme (GBM) is the most prevalent
type of primary human brain tumor, and has low rates of survival
(1–3).
Current therapeutic strategies for GBM, including radiotherapy and
chemotherapy, do not produce satisfactory clinical outcomes
(4,5).
Therefore, a novel and effective approach with which to treat GBM
is required. Mechanistic studies suggest that numerous genetic and
physiological alterations occur in GBM, which affect the survival
and proliferation of glioblastoma cells (6). Receptor tyrosine kinases (RTKs) are
genes/proteins that are frequently modified during the pathogenesis
of GBM (7). Evidence from a number of
studies, supports the hypothesis that platelet-derived growth
factor receptor (PDGFR), a member of the type III RTK family, and
the signaling pathway through PDGFR is strongly associated with the
development of GBM (8–10). PDGF and its receptors have been shown
to be upregulated in one third of surgical glioblastoma samples and
human glioblastoma cell lines (8). In
addition, in vitro and in vivo studies have
demonstrated that PDGF and its receptors are involved in the
proliferation, cell migration and angiogenesis of GBM cells
(11,12). Furthermore, PDGF autocrine loops have
been reported to be involved in the tumorigenesis of GBM (13,14). In
light of these findings, it was hypothesized that inhibition of
PDGF signaling may be a viable therapeutic approach for GBM.
Currently, different types of PDGF antagonists are
being developed and evaluated in animal disease models, in addition
to clinical trials (15). Tyrphostin
AG 1296 is a specific inhibitor of human PDGFR-α and −#x03B2;, as
well as of c-KIT (also termed sCD117), fms-related tyrosine kinase
3 and Bek (a fibroblast growth factor receptor) tyrosine kinases
(16–19). It has been reported that tyrphostin AG
1296 inhibits cell proliferation and promotes apoptosis in small
cell lung cancer, leukemia and anaplastic thyroid cancer (20–22).
Recently, it has been suggested that AG 1296 may suppress the
proliferation and migration of glioblastoma cells (23,24).
Whilst tyrphostin AG 1296 has been shown to affect the
proliferation and migration of glioblastoma cells, its effect on
the rate of apoptosis in GBM remains unclear.
In the present study, the effects of tyrphostin AG
1296 on cell apoptosis in glioblastoma cells were examined in
vitro and in vivo. The in vitro experiments
demonstrated that tyrphostin AG 1296 reduced cell viability,
inhibited cell migration and promoted cell apoptosis. The in
vivo experiments also showed that tyrphostin AG 1296 inhibited
tumor growth and induced tumor cell apoptosis in a mouse xenograft
model of human GBM. Therefore the current data support the
hypothesis that tyrphostin AG 1296 inhibits the growth of
glioblastoma cells by inducing apoptosis.
Materials and methods
U87MG cell culture
U87MG, a representative grade IV glioma cell line,
was obtained from the American Type Culture Collection (Manassas,
VA, USA). U87MG cells were cultured in Dulbecco's modified Eagle's
medium/F12/10% FBS medium (Invitrogen, Grand Island, NY, USA).
Cells were maintained in a humidified incubator at 37°C with 5%
CO2.
Cell viability assay
U87MG Cells were treated with 0.3125, 0.625, 1.25,
2.5, 5, 10 or 20 µM of tyrphostin AG 1296 (ThermoFisher Scientific,
Inc., Waltham, MA USA; Catalog No. 50-230-7882), 72 h after cells
had been seeded in 96-well plates for a period of 24 h. In order to
assess whether the duration of treatment affected cell viability,
cells were treated with 5 mM tyrphostin AG 1296 for 24, 48 or 72 h.
The cell viability assay was performed using a CellTiter-Glo® Assay
kit (Promega Corporation, Madison, WI USA). Experiments were
repeated three times, independently.
Propidium iodide analysis
Cells were treated with tyrphostin AG 1296 at
various concentrations and harvested after 48 h. Propidium iodide
staining was performed using a Cycletest™ plus DNA reagent kit
(Becton Dickinson, San Jose, CA, USA), followed by detection of the
apoptotic cells with DNA content in sub-G1, using a FACSCalibur
flow cytometry. Data were analyzed using CellQuest Pro software,
version 5.1 (Becton Dickinson).
Morphological analysis of U87MG
cells
U87MG cells treated with tyrphostin AG 1296 at
various concentrations, were stained using Hoechst. Cells with
condensed or fragmented nuclei were identified as apoptotic cells
using a Olympus BX53 microscope (Olympus Corporation, Tokyo,
Japan).
Cell invasion assay
For the cell invasion assay, 1×106 cells
were plated into the upper chambers of 24-transwell Boyden chamber
wells (Costar, Bedford, MA, USA), and treated with tyrphostin AG
1296 at various concentrations for 8 hr. Following treatment, cells
were fixed and stained with 0.1% crystal violet for imaging.
Migrated cells were lysed with 10% acetic acid following imaging.
The absorbance of lysate was measured at 595 nm by using a Nanodrop
2000 (Thermo Scientific, Inc., Wilmongton, DE, USA).
Tyrphostin AG 1296 treatment in a
mouse xenograft model in vivo
Approval for the use of mice in the present study,
was obtained from the Animal Ethics Committee and the Institutional
Review Board of Wuhan University (Wuhan, China). C57BL/6J mice were
purchased from Shanghai SLAC laboratory Animal Co., Ltd. (Shanhai,
China). Animals were provided free access to food and water and
were housed in a 13:11 (light:dark) cycle. Female mice aged 6–8
weeks, weighing 20 g were used for experiments. U87MG cells
(2×106 cells/mouse) were injected into the axillary
regions of mice. When tumor volumes had reached 70 mm3, the mice
were randomly divided into a control group and low and high dose
tyrphostin AG 1296 treatment groups (n=6/group). Low-dose (50
mg/kg) or high-dose (100 mg/kg) tyrphostin AG 1296, or vehicle (10%
1-methyl-2-pyrrolidinone and 90% polyethylene glycol 300) was
intravenously injected into the mice each day for 13 days. Tumor
size and body weight were measured daily. The tumor volume was
calculated according to the formula: Volume (mm3) =
(width × width × length)/2.
Terminal deoxynucleotidyl
transferase-mediated dUTP nick end-labeling (TUNEL) assay
Tumor samples from the control and the AG
1296-treatment groups were harvested from mice following 13 days of
treatment. The tumors were cut into 5-mm3 sections and
fixed in 4% paraformaldehyde. Subsequently, samples were embedded
in paraffin blocks and 5-mm sections were produced. Apoptotic cells
were identified using a TUNEL assay. Briefly, tumor tissues were
incubated with TdT enzyme solution (New England Biolabs, Inc.,
Ipswich, MA, USA) for 60 min, washed twice with PBS twice and then
incubated for 30 min at 25°C with DAPI. Images were captured using
an Olympus BX53 fluorescence microscope.
Statistical analysis
All experiments were repeated in triplicates,
independently. The data were analyzed by a one-way or
two-wayanalysis of variance followed by Tukey's post-hoc test.
Graphs were produced using SPSS statistical software, version 17.0
(SPSS, Chicago, IL, USA). Data are expressed as the mean ± standard
deviation. P<0.05 was considered to indicate a statistically
significant difference.
Results
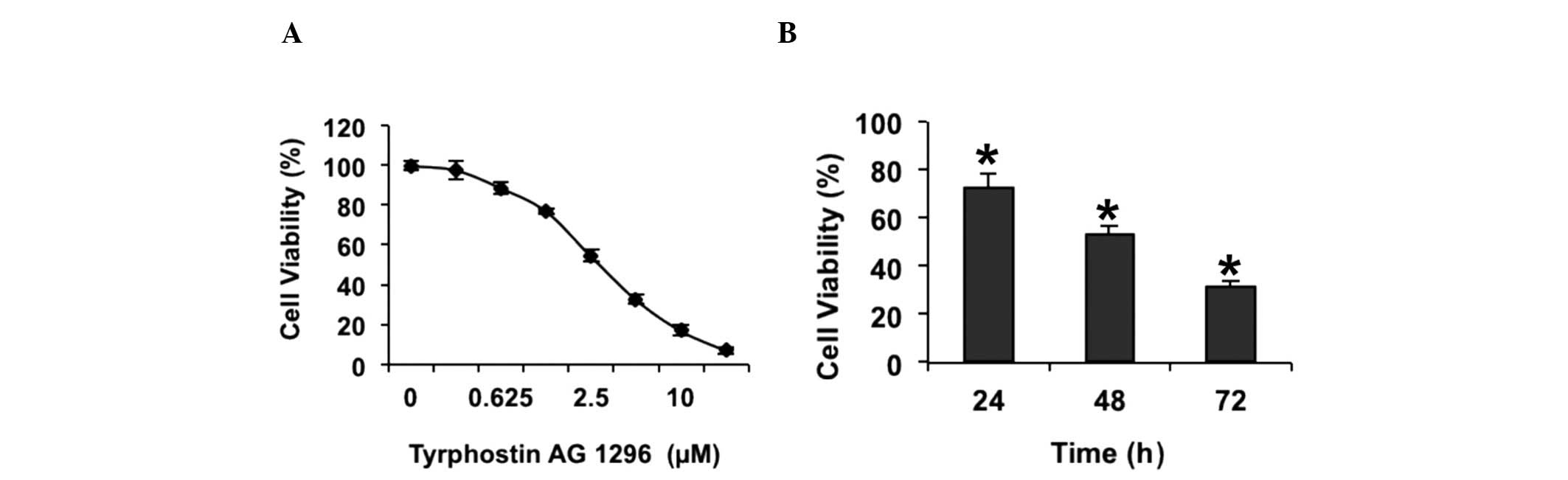
Tyrphostin AG 1296 inhibits U87MG cell
viability
Previous studies have suggested that tyrphostin AG
1296, an inhibitor of PDGFR, reduces glioblastoma cell
proliferation (24). In order to
confirm whether tyrphostin AG 1296 affects glioblastoma cell
growth, a cell viability assay was performed in U87MG cells. The
results showed that tyrphostin AG 1296 inhibited the viability of
U87MG cells, in a dose-dependent manner (Fig. 1A). It was observed that cell viability
was almost fully suppressed when cells were treated with 20 µM
tyrphostin AG 1296 for 72 h (Fig.
1A). In order to investigate whether the effect of tyrphostin
AG 1296 on the viability of U87MG cells is time-dependent, cells
were treated with 5 µM tyrphostin AG 1296 for 24, 48 and 72 h. It
was shown tyrphostin AG 1296 reduced U87MG cell viability in a
time-dependent manner (Fig. 1B).
Therefore, the current data confirm that tyrphostin AG 1296
inhibits glioblastoma cell viability in vitro.
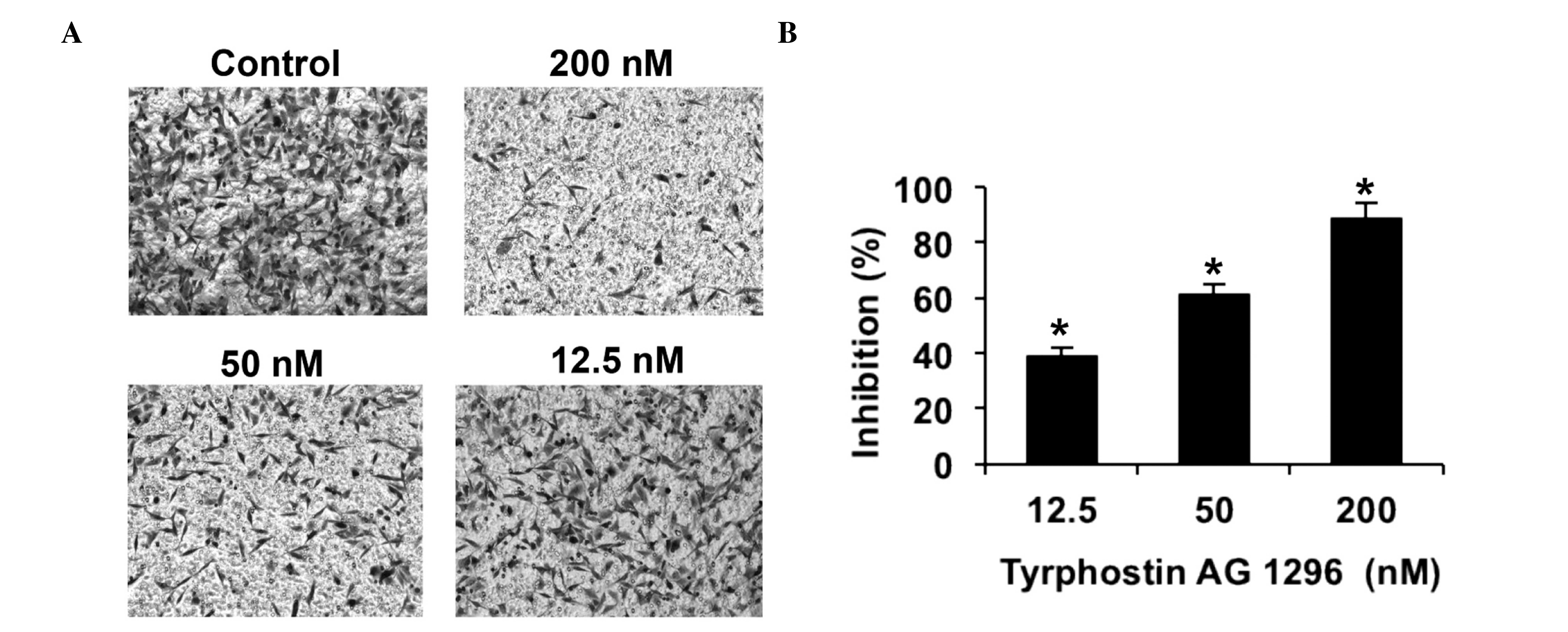
Tyrphostin AG 1296 inhibits U87MG cell
invasion in vitro
Subsequently, the effects of tyrphostin AG 1296 on
U87MG cell invasion were examined. Matrigel invasion assays, with
8.0 µm-pore transwells were used. The results of the invasion
assays showed that low and high concentrations of tyrphostin AG
1296 effectively restricted U87MG cell migration across the filter
(Fig. 2A). Statistical analysis
demonstrated that the anti-migration effect of tyrphostin AG 1296
in U87MG cells, increased in a dose-dependent manner (Fig. 2B). These results are in accordance
with those of a previous study, which suggested that tyrphostin AG
1296 inhibits glioblastoma cell invasion in vitro (23).
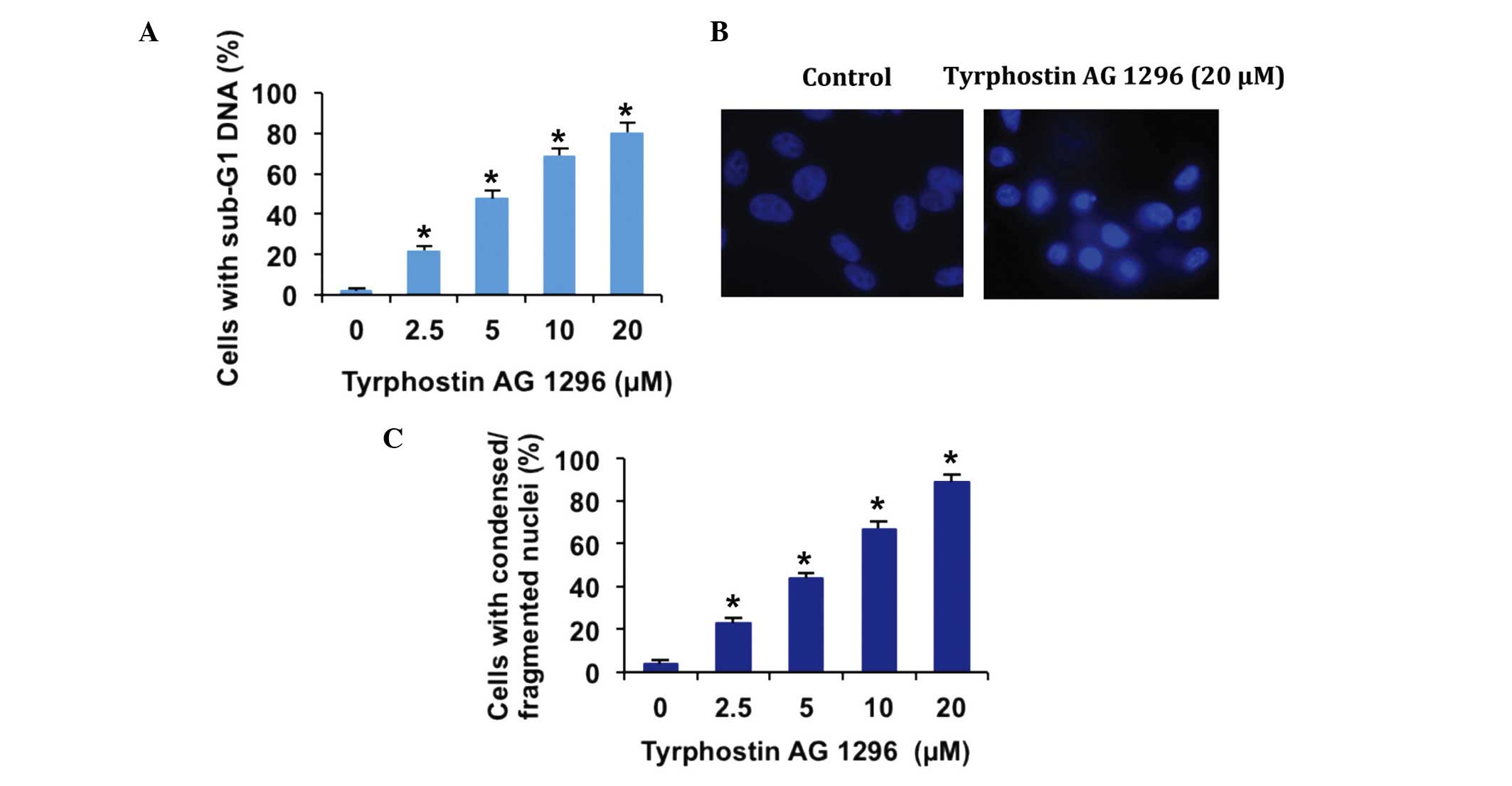
Tyrphostin AG 1296 induces apoptosis
of U87MG cells
Although the effects of tyrphostin AG 1296 on cell
growth and migration have been investigated, its effect on
glioblastoma cell apoptosis has not been fully elucidated. In order
to investigate whether tyrphostin AG 1296 regulates cell apoptosis,
the fraction of apoptotic cells with sub-G1 DNA content, with and
without tyrphostin AG 1296 treatment, was examined. The results
showed that the percentage of cells with sub-G1 DNA content
increased in a dose-dependent manner (Fig. 3A). Furthermore, U87MG cells exposed to
tyrphostin AG 1296 exhibited the condensed and fragmented
morphology (Fig. 3B), which is
characteristic of apoptotic cells. The number of cells with
condensed or fragmented nuclei was markedly increased in U87MG
cells, and this increase occurred in a dose-dependent manner
(Fig. 3C). These results demonstrated
that tyrphostin AG 1296 induces apoptosis in U87MG cells.
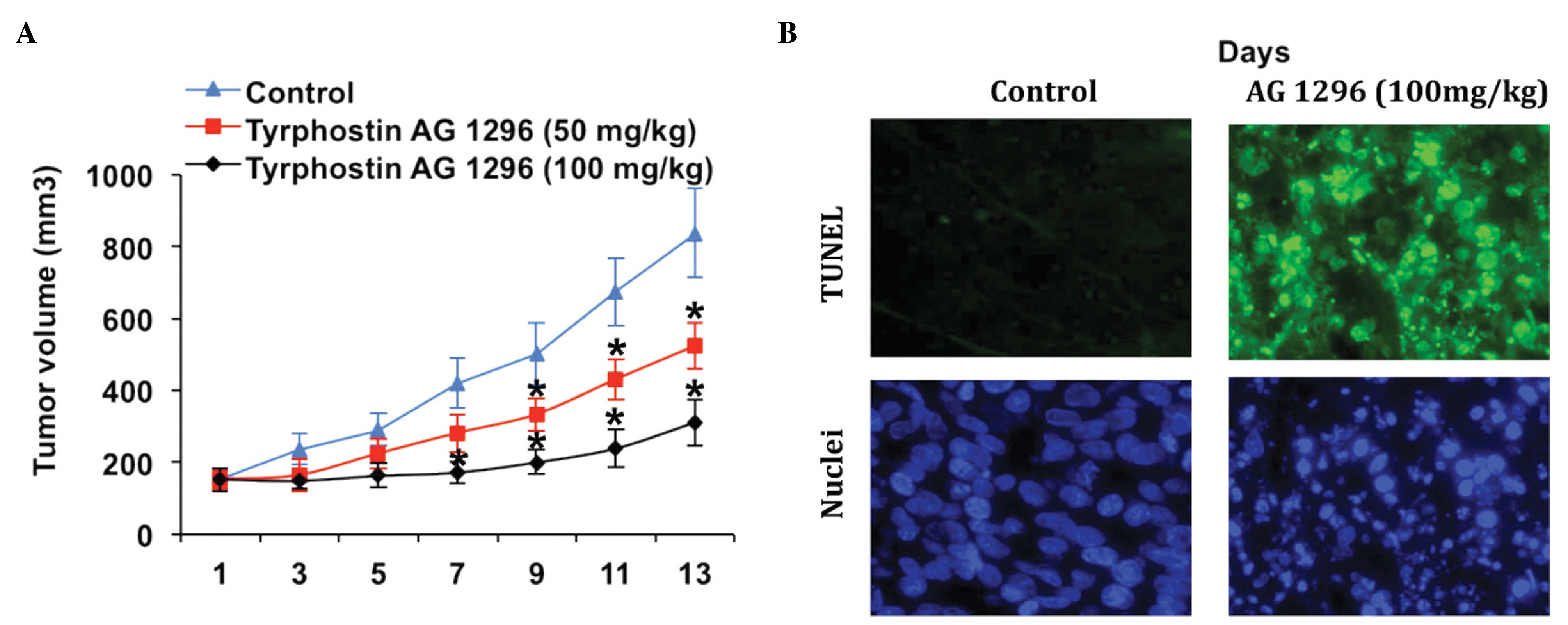
Tyrphostin AG 1296 antagonizes tumor
growth and induces apoptosis of tumor cells in vivo
In order to measure the antitumor activity of
tyrphostin AG 1296 in vivo, a glioblastoma xenograft model
was generated by injecting mice with U87MG cells. Once tumor
volumes reached 70 mm3, various doses of tyrphostin AG
1296 were administered over 13 days. Compared with the control
group, tyrphostin AG 1296 led to reduction of tumor growth at low
(50 mg/kg) and high (100 mg/kg) doses. Furthermore, high-dose
tyrphostin AG 1296 suppressed tumor growth to a greater extent than
low-dose treatment (Fig. 4A). In
accordance with the in vitro data, cell apoptosis was
observed in tumor tissues (Fig. 4B).
Tyrphostin AG 1296 induced changes characteristic of apoptosis, as
measured using a TUNEL assay (Fig.
4B). In addition, mice exhibited no significant signs of
toxicity or weight loss during Tyrphostin AG 1296 treatment (data
not shown). These data indicate that tyrphostin AG 1296 suppresses
GBM tumor growth by inducing cell apoptosis in vivo.
Discussion
GBM is the most common type of primary tumor of the
central nervous system (25–27). The overall median survival period is
~15 months for individuals with GBM (28). The majority of current
chemotherapeutic approaches for GBM involve targeting tyrosine
kinase receptor signaling pathways, due to the central role of
these molecules in the oncogenesis of GBM (29). However, RTK inhibitors have not
produced the level of efficacy in patients with GBM, which had been
anticipated (30,31). Thus, the development of novel
chemotherapeutic agents for GBM is required. The present study
examined the effect of tyrphostin AG 1296 on glioblastoma cell
growth and migration in vitro and in vivo. In
accordance with previous studies (23,24),
tyrphostin AG 1296 exhibited marked efficacy in the inhibition of
glioblastoma cell growth and migration in vitro.
Furthermore, it was demonstrated that tyrphostin AG 1296 induced
cell apoptosis in vitro and in vivo. The inhibitory
effects of tyrphostin AG 1296 on proliferation and migration, and
the induction of apoptosis by this agent, resulted in a reduction
in the growth of GBM tumors in vivo (Fig. 4). The results of previous studies and
of the present study, on tyrphostin AG 1296 improve the current
understanding of GBM treatment and suggest a novel therapeutic
approach to this disease.
In addition to tyrphostin AG 1296, other inhibitors
targeting PDGFR-β, include Imatinib. Imatinib is currently used for
the clinical treatment of certain types of cancer, including
chronic myeloid leukemia and gastrointestinal stromal tumor
(32–34). However, Imatinib has exhibited limited
efficacy in patients with GBM in phase I/II clinical trials
(35), which may be a result of
resistance, due to mutations in the GBM tumors (36,37). The
present study demonstrated a strong antitumor activity of
tyrphostin AG 1296 in a glioblastoma cell line and a GBM mouse
model. Therefore, tyrphostin AG 1296 may be a promising
chemotherapeutic option for malignant GBM. However, its efficacy in
patients with GBM requires further evaluation.
Acknowledgements
The authors would like to thank Professor Xiwen Shi
(Henan Hospital, Zhengzhou, China), Professor Lijun Huang (Xinxiang
Hospital, Xinxiang, China) and Professor Cheng Wang (Shenzhen
hospital, Shenzen, China) for their advice, as well as all members
of the laboratory for their technical support and suggestions.
References
|
1
|
Davis FG, Freels S, Grutsch J, et al:
Survival rates in patients with primary malignant brain tumors
stratified by patient age and tumor histological type: An analysis
based on surveillance, epidemiology and end results (SEER) data,
1973–1991. J Neurosurg. 88:1–10. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Maher EA, Furnari FB, Bachoo RM, et al:
Malignant glioma: Genetics and biology of a grave matter. Genes
Dev. 15:1311–1333. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ricard D, Idbaih A, Ducray F, et al:
Primary brain tumours in adults. Lancet. 379:1984–1996. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Aherne NJ, Benjamin LC, Horsley PJ, et al:
Improved outcomes with intensity modulated radiation therapy
combined with temozolomide for newly diagnosed glioblastoma
multiforme. Neurol Res Int. 2014:9456202014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Aizer AA, Ancukiewicz M, Nguyen PL, et al:
Underutilization of radiation therapy in patients with
glioblastoma: Predictive factors and outcomes. Cancer. 120:238–243.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kanu OO, Hughes B, Di C, et al:
Glioblastoma Multiforme Oncogenomics and Signaling Pathways. Clin
Med Oncol. 3:39–52. 2009.PubMed/NCBI
|
|
7
|
Krause DS and Van Etten RA: Tyrosine
kinases as targets for cancer therapy. N Engl J Med. 353:172–187.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Guha A, Dashner K, Black PM, Wagner JA and
Stiles CD: Expression of PDGF and PDGF receptors in human
astrocytoma operation specimens supports the existence of an
autocrine loop. Int J Cancer. 60:168–173. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lokker NA, Sullivan CM, Hollenbach SJ, et
al: Platelet-derived growth factor (PDGF) autocrine signaling
regulates survival and mitogenic pathways in glioblastoma cells:
Evidence that the novel PDGF-C and PDGF-D ligands may play a role
in the development of brain tumors. Cancer Res. 62:3729–3735.
2002.PubMed/NCBI
|
|
10
|
Nazarenko I, Hede SM, He X, et al: PDGF
and PDGF receptors in glioma. Ups J Med Sci. 117:99–112. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Capdeville R, Buchdunger E, Zimmermann J
and Matter A: Glivec (STI571, imatinib), a rationally developed,
targeted anticancer drug. Nat Rev Drug Discov. 1:493–502. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Guo P, Hu B, Gu W, et al: Platelet-derived
growth factor-B enhances glioma angiogenesis by stimulating
vascular endothelial growth factor expression in tumor endothelia
and by promoting pericyte recruitment. Am J Pathol. 162:1083–1093.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Heldin CH and Westermark B: Mechanism of
action and in vivo role of platelet-derived growth factor. Physiol
Rev. 79:1283–1316. 1999.PubMed/NCBI
|
|
14
|
Nistér M, Claesson-Welsh L, Eriksson A,
Heldin CH and Westermark B: Differential expression of
platelet-derived growth factor receptors in human malignant glioma
cell lines. J Biol Chem. 266:16755–16763. 1991.PubMed/NCBI
|
|
15
|
Ostman A and Heldin CH: Involvement of
platelet-derived growth factor in disease: Development of specific
antagonists. Adv Cancer Res. 80:1–38. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kovalenko M, Rönnstrand L, Heldin CH, et
al: Phosphorylation site-specific inhibition of platelet-derived
growth factor beta-receptor autophosphorylation by the receptor
blocking tyrphostin AG1296. Biochemistry. 36:6260–6269. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kovalenko M, Gazit A, Böhmer A, et al:
Selective platelet-derived growth factor receptor kinase blockers
reverse sis-transformation. Cancer Res. 54:6106–6114.
1994.PubMed/NCBI
|
|
18
|
Strutz F, Zeisberg M, Renziehausen A,
Raschke B, Becker V, van Kooten C and Müller G: TGF-beta 1 induces
proliferation in human renal fibroblasts via induction of basic
fibroblast growth factor (FGF-2). Kidney Int. 59:579–592. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Levitzki A: Protein tyrosine kinase
inhibitors as novel therapeutic agents. Pharmacol Ther. 82:231–239.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Krystal GW, Carlson P and Litz J:
Induction of apoptosis and inhibition of small cell lung cancer
growth by the quinoxaline tyrphostins. Cancer Res. 57:2203–2208.
1997.PubMed/NCBI
|
|
21
|
Minami Y, Yamamoto K, Kiyoi H, Ueda R,
Saito H and Naoe T: Different antiapoptotic pathways between
wild-type and mutated FLT3: Insights into therapeutic targets in
leukemia. Blood. 102:2969–2975. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Che HY, Guo HY, Si XW, You QY and Lou WY:
Additive effect by combination of Akt inhibitor, MK-2206 and PDGFR
inhibitor, tyrphostin AG 1296, in suppressing anaplastic thyroid
carcinoma cell viability and motility. Onco Targets Ther.
7:425–432. 2014.PubMed/NCBI
|
|
23
|
Sciaccaluga M, D'Alessandro G, Pagani F,
et al: Functional cross talk between CXCR4 and PDGFR on
glioblastoma cells is essential for migration. PLoS One.
8:e734262013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Watanabe T, Ohtani T, Aihara M and
Ishiuchi S: Enhanced antitumor effect of YM872 and AG1296
combination treatment on human glioblastoma xenograft models. J
Neurosurg. 118:838–845. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Fuller GN: The WHO classification of
tumours of the central nervous system, 4th edition. Arch Pathol Lab
Med. 132:9062008.PubMed/NCBI
|
|
26
|
Louis DN, Holland EC and Cairncross JG:
Glioma classification: A molecular reappraisal. Am J Pathol.
159:779–786. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Okada H, Kohanbash G, Zhu X, et al:
Immunotherapeutic approaches for glioma. Crit Rev Immunol. 29:1–42.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Anton K, Baehring JM and Mayer T:
Glioblastoma multiforme: Overview of current treatment and future
perspectives. Hematol Oncol Clin North Am. 26:825–853. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Carrasco-García E, Saceda M and
Martinez-Lacaci I: Role of receptor tyrosine kinases and their
ligands in glioblastoma. Cells. 3:199–235. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Taylor TE, Furnari FB and Cavenee WK:
Targeting EGFR for treatment of glioblastoma: Molecular basis to
overcome resistance. Curr Cancer Drug Targets. 12:197–209. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Vivanco I, Robins HI, Rohle D, Nghiemphu
PL, Kubek S, Oldrini B, Chheda MG, Yannuzzi N, et al: Differential
sensitivity of glioma- versus lung cancer-specific EGFR mutations
to EGFR kinase inhibitors. Cancer Discov. 2:458–471. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Johnson JR, Bross P, Cohen M, et al:
Approval summary: Imatinib mesylate capsules for treatment of adult
patients with newly diagnosed philadelphia chromosome-positive
chronic myelogenous leukemia in chronic phase. Clin Cancer Res.
9:1972–1979. 2003.PubMed/NCBI
|
|
33
|
Cohen MH, Williams G, Johnson JR, et al:
Approval summary for imatinib mesylate capsules in the treatment of
chronic myelogenous leukemia. Clin Cancer Res. 8:935–942.
2002.PubMed/NCBI
|
|
34
|
Joensuu H, Roberts PJ, Sarlomo-Rikala M,
et al: Effect of the tyrosine kinase inhibitor STI571 in a patient
with a metastatic gastrointestinal stromal tumor. N Engl J Med.
344:1052–1056. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wen PY, Yung WK, Lamborn KR, et al: Phase
I/II study of imatinib mesylate for recurrent malignant gliomas:
North American brain tumor consortium study 99-08. Clin Cancer Res.
12:4899–4907. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Rubin BP and Duensing A: Mechanisms of
resistance to small molecule kinase inhibition in the treatment of
solid tumors. Lab Invest. 86:981–986. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ma Y, Zeng S, Metcalfe DD, et al: The
c-KIT mutation causing human mastocytosis is resistant to STI571
and other KIT kinase inhibitors; kinases with enzymatic site
mutations show different inhibitor sensitivity profiles than
wild-type kinases and those with regulatory-type mutations. Blood.
99:1741–1744. 2002. View Article : Google Scholar : PubMed/NCBI
|