Introduction
Lung cancer is characterized by the highest
incidence and mortality rates of any malignant tumor (1), and non-small cell lung cancer (NSCLC)
accounts for ~85% of all the diagnostic cases of lung cancer
(2). For decades, numerous
therapeutic strategies, including adjuvant, neoadjuvant and
postoperative chemotherapy, have been used in the treatment of
NSCLC (3). Recently, epidermal growth
factor receptor tyrosine kinase inhibitors, including gefitinib,
erlotinib and afatinib, have been used in the treatment of NSCLC,
resulting in lower toxicity and higher efficiency, compared with
traditional chemotherapeutic drugs (4). However, patients with NSCLC still failed
this novel chemotherapy regime, and exhibited worse overall
survival (OS) due to multidrug resistance and the heterogeneity of
NSCLC tumors (5). It currently
remains difficult to obtain an early diagnosis and execute timely
therapeutic interventions for patients with NSCLC; therefore, novel
effective therapeutic strategies should be investigated (6). Furthermore, novel biological markers for
the early diagnosis and survival prediction of NSCLC are
emerging.
Activating transcription factor 2 (ATF2), a member
of the cAMP response element binding family, is activated by
phosphorylation to c-Jun N-terminal kinases or p38, and
heterodimerizes with those transcription factors in response to
regulatory biological progresses (7,8), such as
stress and cytokines (9–11). ATF2 dimerizes with other members of
the activator protein 1 superfamily to activate the transcription
of genes involved in stress and DNA damage responses, growth,
differentiation, and apoptosis (9–11).
Furthermore, ATF2 can be activated by stress and cytokine stimuli
in response to a variety of cell processes, including DNA damage,
viral infection and cell death (9).
It has previously been demonstrated that ATF2 has an oncogenic role
in melanocyte transformation (12).
Notably, ATF2 executes different roles depending on its
localization. For example, the nuclear localization has been
demonstrated to be associated with poor prognosis in patients with
melanoma, suggesting that ATF2 localization may serve as a
prognostic marker in this disease (13). In addition, protein kinase C ε (PKCε)
may phosphorylate ATF2 on threonine 52 and localize ATF2 to the
nucleus (13). Elevated levels of
PKCε, which are observed in more advanced stage metastatic
melanomas, appear to prevent the nuclear-to-mitochondrial
translocation of ATF2 that enable its tumor-suppressor function
(14), and ATF2 targets include
various pro-survival molecules, such as cell cycle-related proteins
(15). Furthermore, a previous study
indicated that enhancing the nuclear export of ATF2 may be a novel
therapeutic modality for drug-resistant melanomas (5).
In the present study, the expression and
localization of ATF2 was detected in NSCLC cancer tissues and
cells. Subsequently, the association between ATF2 expression and
clinicopathological characteristics were analyzed to determine the
clinical significance of ATF2 in the prognostic prediction of
patients with NSCLC. The results demonstrated that the expression
levels of ATF2 and p-ATF2 were markedly increased in NSCLC tissues.
ATF2 is therefore another promising predictor for prognosis of
NSCLC.
Materials and methods
Cell culture
Normal human bronchial epithelial (HBE) cells (HBE
135-E6E7) and various human NSCLC cells (H1299, H1975, A549, PC9,
H460 and H1650) were purchased from the American Type Culture
Collection (Manassas, VA, USA). The H1299, H1975, H460 and H1650
cells were cultured in RPMI-1640 and the PC9 and A549 cells were
cultured in Dulbecco's modified Eagle's medium (ThermoFisher
Scientific, Inc., Waltham, MA, USA). The culture medium contained
10% heat-inactivated fetal bovine serum (FBS; ThermoFisher
Scientific, Inc.) and antibiotics (100 U/ml penicillin and 100 U/ml
streptomycin; Beyotime Institute of Biotechnology, Inc., Dalian,
China). HBE cells were cultured in RPMI-1640 with 15% FBS. All the
cells were incubated at 37°C and 5% CO2 in a humidified
atmosphere.
Patients and tissue specimens
Written informed consent was obtained from all
patients. Paired cancerous and adjacent normal tissue samples were
obtained from 86 patients with NSCLC who underwent surgical
resection at the First Affiliated Hospital of Nanchang University
(Nanchang, China) between June 2001 and July 2004, according to
protocols approved by the Institute Research Medical Ethics
Committee of the First Affiliated Hospital of Nanchang University.
The 86 patients were aged between 24 and 68 years (median age, 42
years), and the tumors were pathologically graded according to the
International Union against Cancer tumor-node-metastasis
classification of malignant tumors. Following surgical resection,
the 86 tissues specimens were immediately fixed in 10% formalin,
embedded in paraffin and stored at room temperature. In addition,
to investigate RNA levels of AFT2 in fresh tumor tissues, 12 paired
fresh tumor and adjacent normal tissues were obtained from NSCLC
patients who underwent surgical resection at the First Affiliated
Hospital of Nanchang University between November 2011 and February
2012. The 12 patients were aged between 41 and 60 years (median
age, t51.5 years). Following surgical resection, the 12 paired
tumor and adjacent normal tissue specimens were immediately frozen
in RNAlater® (Ambion Life Technologies, Carlsbad, CA, USA) and
stored at −80°C.
Immunohistochemistry (IHC)
The tissues were fixed with 4% paraformaldehyde in
0.1 M phosphate buffer. After 24 h, the tissues were embedded in
paraffin wax and sliced into 14 µM sections and mounted onto glass
slides. The paraffin-embedded tissue sections were then dewaxed in
xylene and alcohol, boiled with EDTA (1 mmol/l; pH 8.0), and
treated with 3% H2O2 for 10 min.
Immunohistochemical analysis was then performed using the EnVision™
two-step staining method (16).
Briefly, the slides were incubated with primary polyclonal rabbit
anti-human ATF2 (1:500 dilution; p-ATF2, 1:1,000 dilution;
Santa-Cruz Biotechnology, Inc.) or mosue anti-human GAPDH (1:1,000
dilution; Zhejiang Kangchen Biotech Co., Hangzhou, China) overnight
at 4°C and then washed in phosphate-buffered saline three times
before the goat anti-rabbit and anti-mouse IgG secondary antibodies
(1:5,000 dilution; Zhejiang Kangchen Biotech Co.) were added. After
1 h at room temperature, the slides were stained with DAB and
counterstained with Mayer's hematoxylin (Beyorime Institute of
Biotechnology, Inc.). Slides that were not incubated with primary
antibody were used as the negative controls. The proportion of
stained cells was scored as previously described (17), and the receptor score was calculated
using the equation: Positively stained cell proportion × staining
intensity. The median value of the IHC scores was 4; therefore, low
and high expression were set at scores of <4 and ≥4,
respectively (18).
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted from the 12 frozen samples
using TRIzol® reagent (Bioo Scientific, Austin, TX, USA), according
to manufacturers instructions. RT-qPCR were performed using SYBR®
Green Real-Time PCR Master Mix and the Stratagene Mx3000P Real-Time
PCR system (Agilent Technologies, Inc., Santa Clara, CA, USA). The
primer sequences were as follows: ATF2, sense
5′-GTAATCACCCAGGCACCATC-3′ and antisense
5′-CTGAGTCCTAACCAATCCCC-3′; GAPDH, sense 5′-GAAGGTGAAGGTCGGAGTC-3′
and antisense 5′-GAAGATGGTGATGGGATTTC-3′. The cycling conditions
were set as follows: 95°C for 10 min followed by 40 amplification
cycles at 95°C for 10 sec and 60°C for 20 sec. The GAPDH gene
served as the control and relative mRNA expression levels were
defined using the 2−ΔΔCt method (19).
Plasmids and short hairpin
(sh)RNAs
shRNA probes targeting the ATF2 gene were purchased
from Genechem, Inc. (Shanghai, China). The ATF2 gene sequence was
5′-GTTGGCGAGTCCATTTGAG-3′ and transfection of H1299 and H1975 cells
was performed using Lipofectamine 2000 (Invitrogen Life
Technologies, Carlsbad, CA, USA), according to the manufacturer's
instructions. The pGIPZ vector was purchased from Genechem, Inc.
(Shanghai, China). Untransfected parental cells H1299 and H1975
were used as control. After 48 h, the expression level of ATF2 was
measured by western blotting and MTT assays.
Western blot analysis
Western blot analysis was performed to assess the
protein expression levels of ATF2 and GAPDH in NSCLC cells and
tissues, according to previously described protocols (20). The GAPDH gene served as the
control. Briefly, The cells and tissues were lysed with lysis
buffer (pH 7.4, containing 1% Triton X-100 and 0.2% SDS) on ice for
30 min and centrifugated at 10,000 rpm for 15 min at 4°C. The
supernatant was separated and used for the experiment. The
concentrations of protein were quantified using a bicinchoninic
acid assay kit (ThermoFisher Scientific). Subsequently, 30 µg
protein from each sample were examined on 10% SDS-PAGE gels
(Sigma-Aldrich). The separated proteins were transferred onto
polyvinylidene fluoride membranes (EMD Millipore, Billerica, MA,
USA) at 80 V for 100 min prior to blocking with 5% non-fat milk for
1 h at room temperature. The membranes were then incubated with
primary antibodies against ATF2 (1:1,000 dilution) or GAPDH
(1:1,000 dilution) overnight at 4°C. After washing with
Tris-buffered saline with Tween 20 three times, the membranes were
incubated with peroxidase-conjugated secondary antibody (1:5,000
dilution) for 1 h at room temperature. The signal was then detected
using an enhanced chemiluminescence detection system (GE
Healthcare, Princeton, NJ, USA). The bands were exposed to the
Kodak medical X-ray processor (Kodak, Rochester, NY, USA).
MTT assay
Cell viability was determined by performing an MTT
assay (Sigma-Aldrich). Briefly, the cells were seeded at a density
of 2 × 103 cells/well in 96-well plates for attachment.
MTT solution (20 µl, 5 mg/ml) was added to each well at a specific
time point (12, 24, 48, 72 and 96 h) and incubated for 4 h at 37°C.
Subsequently, the culture medium was removed and replaced with 150
µl dimethyl sulfoxide to resolve the formazan. Absorbance was
measured at 540 nm on a Model 550 microplate reader (Bio-Rad
Laboratories, Hercules, CA, USA).
Statistical analysis
Statistical differences between groups were analyzed
by performing the Student's t-test. The χ2 test was used
to determine the difference in protein expression between tumor
tissues and adjacent normal tissues. Survival rates were assessed
using Kaplan-Meier survival curves (log-rank test). The Wilcoxon
matched-pairs test was used to determine any significant difference
in ATF2 mRNA expression between the normal and tumor tissues as
well as reproducibility of the measurement of the expression of
ATF2 and p-ATF2 in the 86 paraffin-embedded NSCLC tissues. The SPSS
statistical software package, version 16.0 (SPSS, Inc., Chicago,
IL, USA) was used in data processing and analyzing. Two-tailed
P<0.05 indicated a statistically significant difference.
Results
Expression of ATF2 in NSCLC cell lines
and tissues
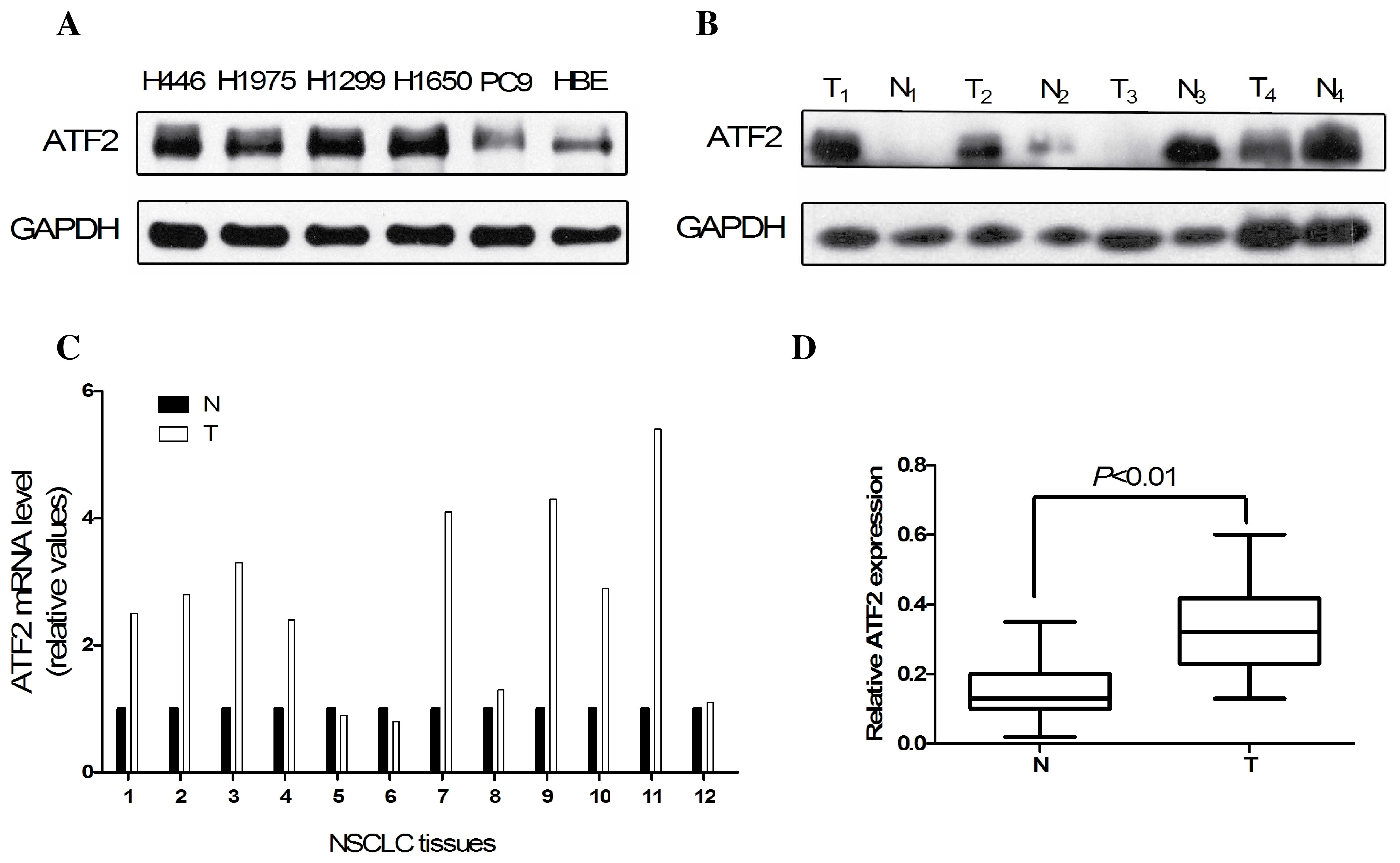
The expression of ATF2 in NSCLC cells was initially
detected by western blotting and RT-qPCR. According to the results,
the protein expression level of ATF2 was markedly higher in the
H446, H1975, H1650, H1299 and PC9 cell lines (Fig. 1A). According to the western blot
results, ATF2 is predominantly expressed in tumor tissues compared
with non-tumor tissues (Fig. 1B).
Furthermore, the mRNA expression level of ATF2 in 8/12 NSCLC tumor
tissues was markedly higher than the adjacent normal tissues
(Fig. 1C). In addition, ATF2
expression levels were significantly higher in tumor tissues
compared to normal tissues according to the IHC results, as
indicated in Fig. 1D (P<0.01).
Association between ATF2/p-ATF2
expression and clinicopathological parameters
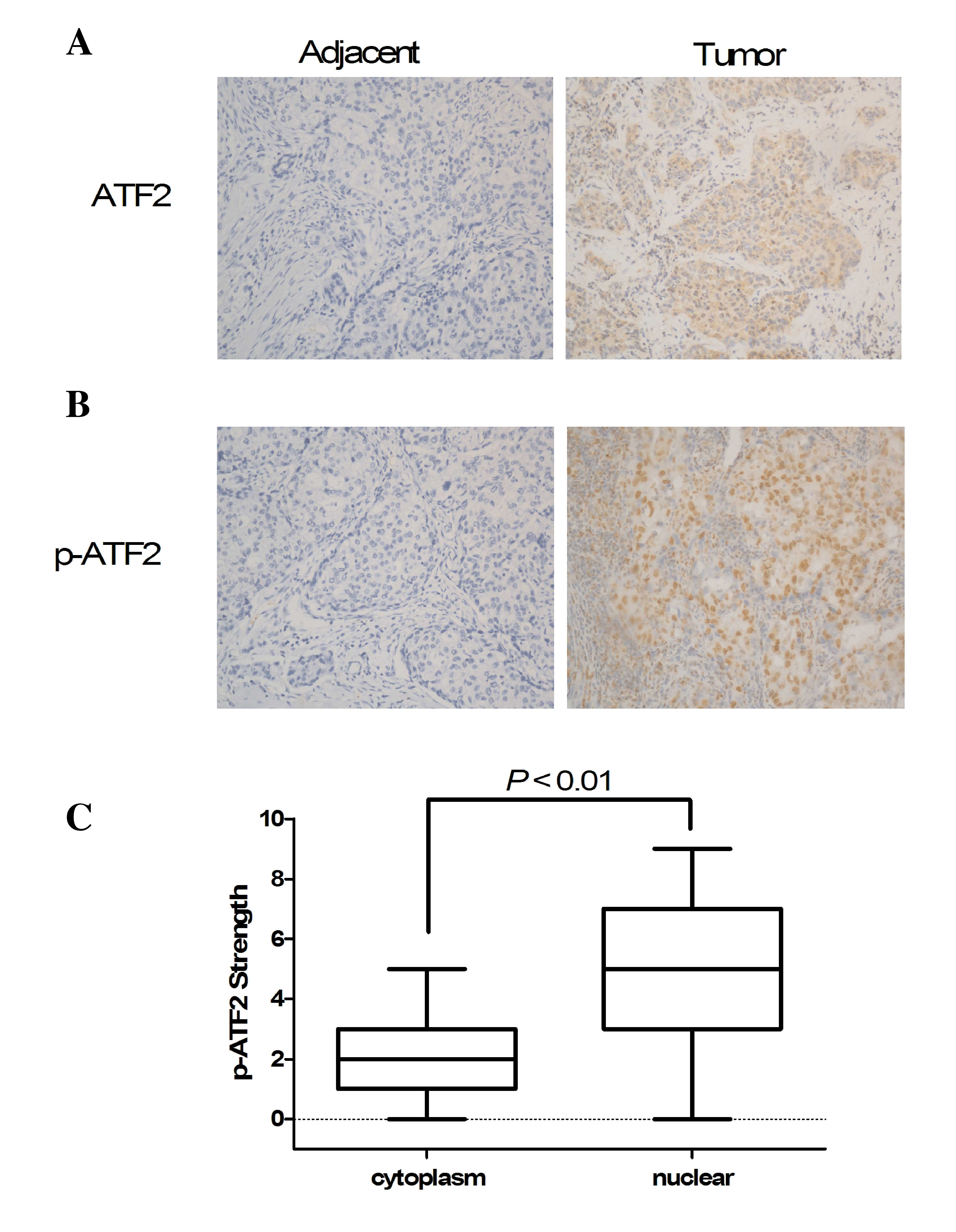
IHC was performed to assess the expression of ATF2
and p-ATF2 in 86 paraffin-embedded NSCLC tissues. The results
showed that the expression of ATF2 was observed in the cytoplasm
and nucleus of cancer cells, however, there was no difference in
expression between the two locations (Fig. 2A). p-ATF2 was predominantly expressed
in the nucleus of cancer cells compared with the cytoplasm
(P<0.01; Fig. 2B and C). In
addition, the expression of p-ATF2 was detected in the cytoplasm.
p-ATF2 was overexpressed in the majority of tumor tissues (65/86
cases, data not shown). The expression of ATF2 and p-ATF2 in
adjacent non-cancerous tissue was also observed in certain cases
(Fig. 2A and B). To determine the
clinical significance by χ2 tests, the samples were
grouped according to a low and high expression of ATF2 or p-ATF2.
Table I shows a significant
difference between ATF2 expression and specific clinicopathological
parameters was observed, including an advanced TNM stage (P=0.002),
large tumor size (P=0.018) and metastasis (P=0.027). However, there
was no significant difference for the remaining clinicopathological
parameters including age, gender or vascular invasion. For p-ATF2,
patients with high p-ATF2 expression had a significant association
with advanced TNM stage (P=0.040) and metastasis (P=0.015; Table II). The remaining clinicopathological
parameters were not significantly associated with p-ATF2 expression
(P>0.05; Table II).
 | Table I.Correlation between ATF2 expression
and clinicopathological parameters in non-small cell lung
cancer. |
Table I.
Correlation between ATF2 expression
and clinicopathological parameters in non-small cell lung
cancer.
|
|
| ATF2 expression,
n |
|
|---|
|
|
|
|
|
|---|
| Variable | Total cases, n | High | Low | P-value |
|---|
| Age, years |
|
|
| 0.245 |
|
<50 | 49 | 30 | 19 |
|
| ≥50 | 37 | 18 | 19 |
|
| Gender |
|
|
| 0.891 |
| Male | 55 | 31 | 24 |
|
|
Female | 31 | 17 | 14 |
|
| Tumor size, cm |
|
|
| 0.018a |
|
<4 | 34 | 16 | 18 |
|
| ≥4 | 52 | 32 | 20 |
|
| TNM stage |
|
|
| 0.002b |
| I | 14 | 5 | 9 |
|
| II | 19 | 11 | 8 |
|
| III | 38 | 32 | 6 |
|
| IV | 15 | 9 | 6 |
|
| Vascular
invasion |
|
|
| 0.813 |
| Yes | 51 | 29 | 22 |
|
| No | 35 | 19 | 16 |
|
| Metastasis |
|
|
| 0.027a |
|
Yes | 52 | 34 | 18 |
|
| No | 34 | 14 | 20 |
|
 | Table II.Correlation between p-ATF2 expression
and clinicopathological parameters in non-small cell lung
cancer. |
Table II.
Correlation between p-ATF2 expression
and clinicopathological parameters in non-small cell lung
cancer.
|
|
| p-ATF2 expression,
n |
|
|---|
|
|
|
|
|
|---|
| Variable | Total cases, n | High | Low | P-value |
|---|
| Age, years |
|
|
| 0.390 |
|
<50 | 49 | 36 | 13 |
|
|
≥50 | 37 | 24 | 13 |
|
| Gender |
|
|
| 0.502 |
|
Male | 55 | 37 | 18 |
|
|
Female | 31 | 23 | 8 |
|
| Tumor size, cm |
|
|
| 0.409 |
|
<4 | 34 | 22 | 12 |
|
| ≥4 | 52 | 38 | 14 |
|
| TNM stage |
|
|
| 0.040a |
| I | 14 | 7 | 7 |
|
| II | 19 | 13 | 6 |
|
|
III | 38 | 32 | 6 |
|
| Vascular
invasion |
|
|
| 0.781 |
|
Yes | 51 | 35 | 16 |
|
| No | 35 | 25 | 10 |
|
| Metastasis |
|
|
| 0.015a |
|
Yes | 52 | 42 | 12 |
|
| No | 34 | 18 | 16 |
|
Association between ATF2/p-ATF2
expression and prognosis in NSCLC
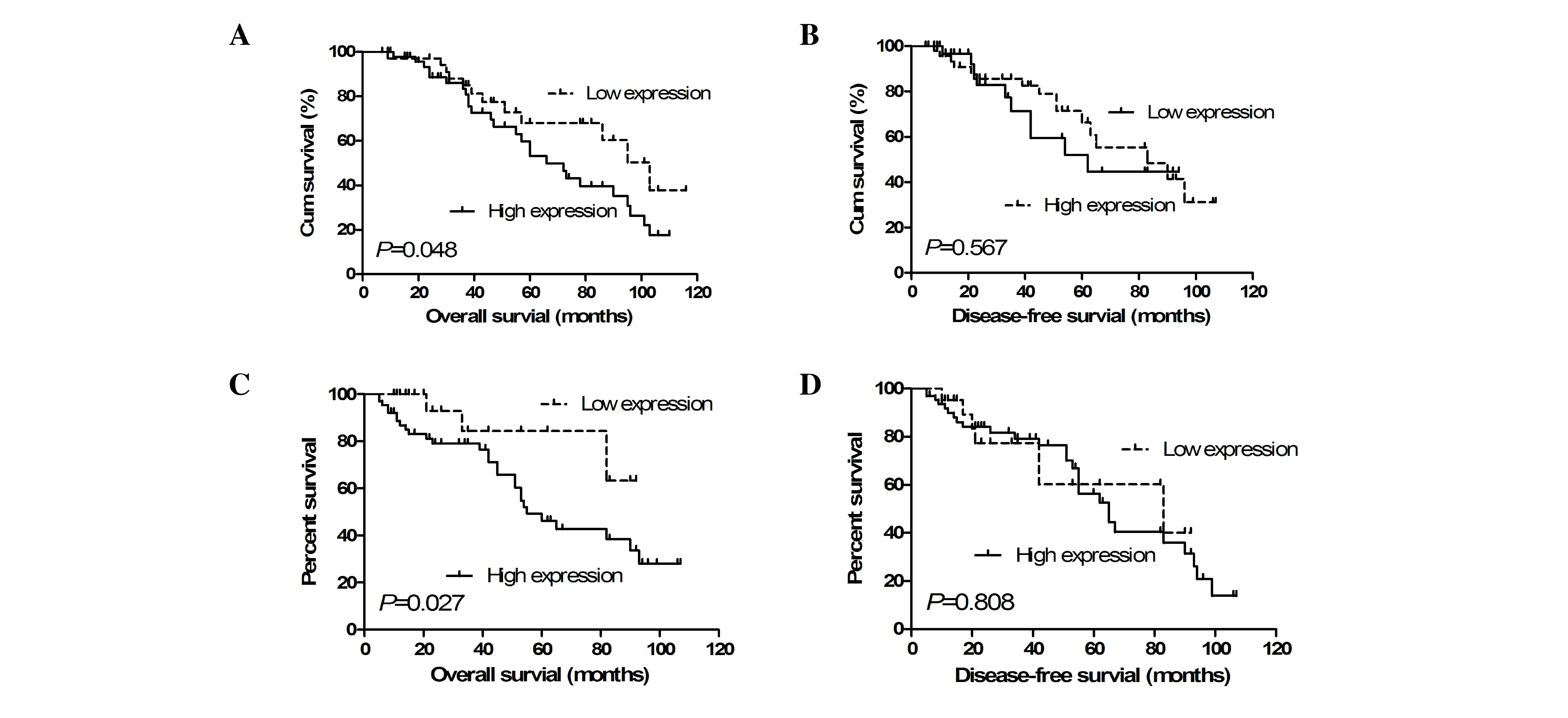
To determine the correlation between ATF2/p-ATF2
expression and survival of patients with NSCLC, the Kaplan-Meier
survival analysis was performed. As shown in Fig. 3A and B, the OS and disease-free
survival (DFS) times in patients with a high level of ATF2
expression were significantly shorter than those of patients with a
low level of ATF2 expression (P<0.01 and P<0.05,
respectively). For p-ATF2, the results revealed that OS was
significantly shorter in patients with a high p-ATF2 expression
compared with patients with a low p-ATF2 expression (P<0.05);
however, there was no significant difference in DFS (P>0.05;
Fig. 3C and D).
Downregulation of ATF2 inhibits cell
proliferation
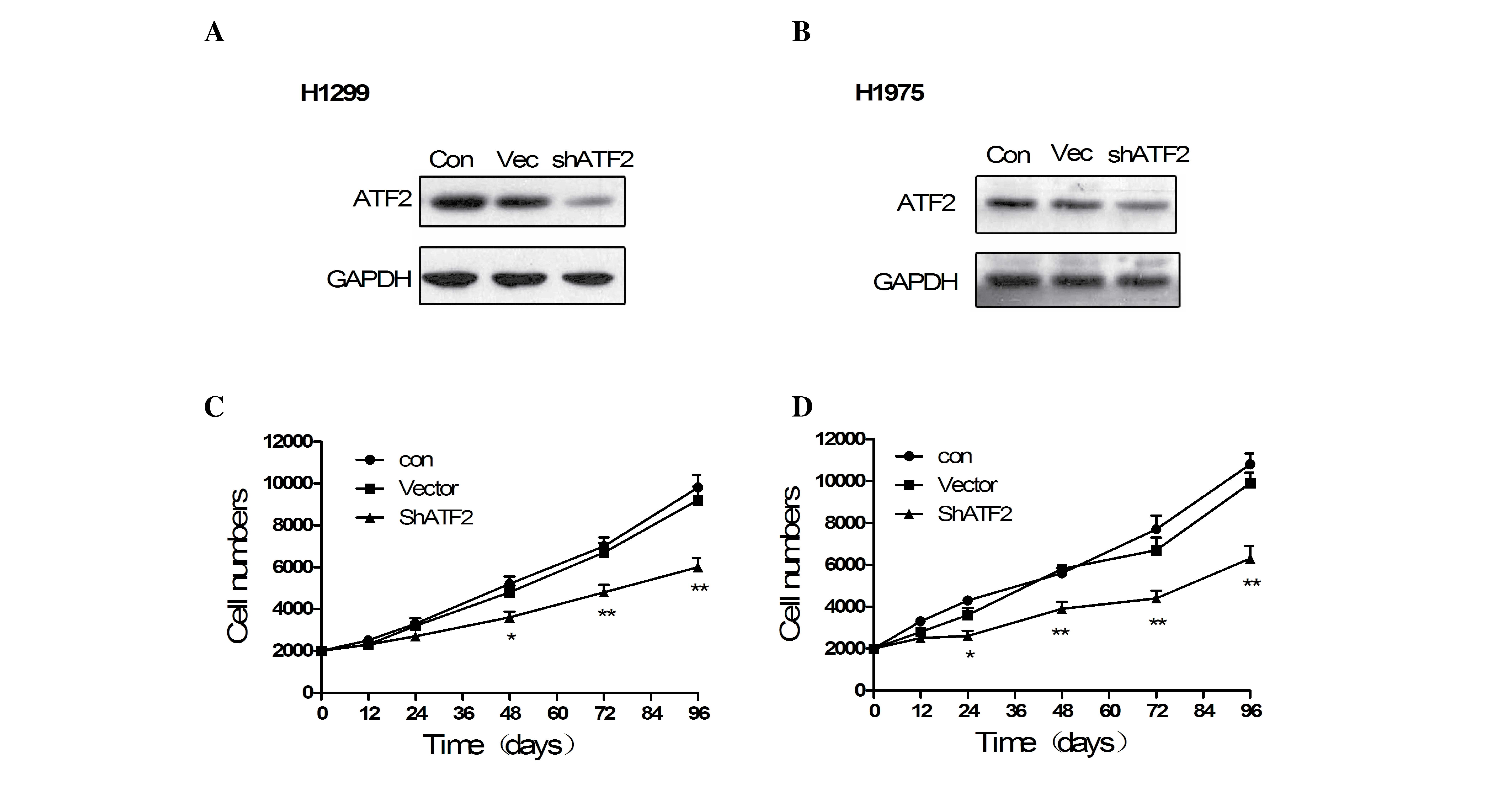
To determine the role of ATF2 in NSCLC cells, ATF2
knockdown cell models were generated in H1299 and H1975 cells. The
ATF2 shRNA was proven to effectively downregulate the expression of
ATF2 by western blotting (Fig. 4A and
B). According to the results of the MTT assay, the knockdown of
ATF2 in H1299 and H1975 cells led to a reduction in cell viability
(Fig. 4C and D; P<0.05). These
data indicated that ATF2 may promote cell proliferation in NSCLC
cells.
Discussion
ATF2, a member of the AP-1 family of transcription
factors, is important in the regulation of cancer cell
proliferation, invasion and survival by modulating various genes,
such as c-Jun and c-Fos (9). A
previous study revealed that ATF2 may play a potential role in
breast cancer invasion and migration by targeting matrix
metalloproteinase-2 (21). However,
the role of ATF2 in NSCLC is poorly understood. To the best of our
knowledge, the present study was the first to investigate the
clinical role of ATF2 in Chinese patients with NSCLC. A marked
increase in ATF2 expression was observed in NSCLC cancer tissues
but not in corresponding normal tissues.
Notably, ATF2 has been demonstrated as a
double-edged sword in different types of cancer. Using a
ATF2-mutant K14-Cre mouse model, Bhoumik et al revealed that
loss of ATF2 in keratinocytes could lead to elevated β-catenin
expression and consequently result in skin tumor formation
(22). Similarly, Maekawa et
al identified that low expression of ATF2 in mammary tissue is
more likely to form a tumor (23). By
contrast, ATF2 gene transcriptional activity was activated in
certain types of tumor. For example, overexpression of ATF2 was
observed in patients with melanoma, and associated with poor
outcome and metastasis (24). The
expression of ATF2 was not proven to be an independent predictor of
prognosis in patients with melanoma, however, the localization of
ATF2 may still serve as a promising prognostic marker. Furthermore,
Duffey et al demonstrated that when head and neck squamous
cell carcinoma (HNSCC) cells were treated with tumor necrosis
factor α and cisplatin, the expression of ATF2 in the nucleus was
enhanced. Thus, enhanced ATF2 expression may predict a poor
prognosis in patients with HNSCC (25). A previous report indicated that the
dual functions of ATF2 may depend on its location: The cytosolic
localization appeared to perform suppressor functions while the
nuclear localization appeared to perform promoter functions
(14). In addition, a previous study
reported a correlation between ATF2 expression and
clinicopathological parameters in breast cancer (26). Through immunohistochemical analysis of
ATF2 and p-ATF2 protein expression levels, the present study
identified important new information in this field. It was
identified that high expression of ATF2 may be an indicator of poor
prognosis for patients with NSCLC, as patients with high ATF2
expression exhibited advanced stage disease, larger tumors and
metastasis. In addition, high expression of p-ATF2 was
significantly associated with tumor stage and metastasis. These
findings are consistent with a previous report, which identified
that high ATF2 expression stimulated cell proliferation in human
gastric cancer cells (27).
The present study investigated the association
between ATF2 and p-ATF2 expression, and outcomes of patients with
NSCLC. By performing Kaplan-Meier survival analysis, it was
identified that high ATF2 expression in tumor tissues predicted
significantly shorter OS and DFS, whereas p-ATF2 expression was
only significantly associated with worse OS. A small number of
previous reports have also demonstrated that ATF2 expression is
associated with higher rates of metastasis and poor prognosis
(28,29). In addition, a previous study
identified that ATF2 is involved in facilitating tumor progression
and cell differentiation (8).
Overall, it can be concluded that elevated ATF2 and p-ATF2
expression may be as a useful biological marker for predicting the
outcomes of patients with NSCLC.
In summary, to the best of our knowledge, the
present study is the first to provide clinical evidence that ATF2
and p-ATF2 may serve as promising predictors for NSCLC prognosis in
a Chinese patient cohort. The results demonstrated that ATF2
expression is elevated in NSCLC tissues and associated with
advanced clinical parameters, such as tumor size, metastasis and
clinical stage. In addition, p-ATF2 overexpression was detected in
tumor tissues and indicated shorter survival of patients with
NSCLC. The results of the current study suggest that ATF2 and
p-ATF2 may be useful prognostic biomarkers for patients with NSCLC.
Thus, the potential mechanisms of ATF2 leading to poor prognosis
should be further analyzed and the current results should be
confirmed in other NSCLC cohorts.
Acknowledgements
The current study was supported by the Educational
Commission of Jiangxi Province of China (no. 20143056).
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
D'Addario G and Felip E: ESMO guidelines
working group: Non-small-cell lung cancer: ESMO clinical
recommendations for diagnosis, treatment and follow-up. Ann Oncol.
4(Suppl 20): S68–S70. 2009.
|
|
3
|
McElnay P and Lim E: Adjuvant or
neoadjuvant chemotherapy for NSCLC. J Thorac Dis. 6(Suppl 2):
S224–S227. 2014.PubMed/NCBI
|
|
4
|
Ellis PM, Coakley N, Feld R, Kuruvilla S
and Ung YC: Use of the epidermal growth factor receptor inhibitors
gefitinib, erlotinib, afatinib, dacomitinib, and icotinib in the
treatment of non-small-cell lung cancer: A systematic review. Curr
Oncol. 22:e183–e215. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
NSCLC Meta-analyses collaborative group.
Arriagada R, Auperin A, et al: Adjuvant chemotherapy, with or
without postoperative radiotherapy, in operable non-small-cell lung
cancer: Two meta-analyses of individual patient data. Lancet.
375:1267–1277. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Spiro SG, Tanner NT, Silvestri GA, et al:
Lung cancer: Progress in diagnosis, staging and therapy.
Respirology. 15:44–50. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lee IT, Lin CC, Cheng SE, Hsiao LD, Hsiao
YC and Yang CM: TNF-α induces cytosolic phospholipase A2 expression
in human lung epithelial cells via JNK1/2- and p38 MAPK-dependent
AP-1 activation. PloS One. 8:e727832013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bengoa-Vergniory N, Gorroño-Etxebarria I,
González Salazar I and Kypta RM: A switch from canonical to
noncanonical Wnt signaling mediates early differentiation of human
neural stem cells. Stem Cells. 32:3196–3208. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lopez-Bergami P, Lau E and Ronai Z:
Emerging roles of ATF2 and the dynamic AP1 network in cancer. Nat
Rev Cancer. 10:65–76. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bhoumik A, Lopez-Bergami P and Ronai Z:
ATF2 on the double-activating transcription factor and DNA damage
response protein. Pigment Cell Res. 20:498–506. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lau E and Ronai ZA: ATF2-at the crossroad
of nuclear and cytosolic functions. J Cell Sci. 125:2815–2824.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shah M, Bhoumik A, Goel V, et al: A role
for ATF2 in regulating MITF and melanoma development. PLoS Genet.
6:e10012582010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lau E, Kluger H, Varsano T, et al: PKCε
promotes oncogenic functions of ATF2 in the nucleus while blocking
its apoptotic function at mitochondria. Cell. 148:543–555. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Varsano T, Lau E, Feng Y, et al:
Inhibition of melanoma growth by small molecules that promote the
mitochondrial localization of ATF2. Clin Cancer Res. 19:2710–2722.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
An JJ, Shi KJ, Wei W, et al: The
ROS/JNK/ATF2 pathway mediates selenite-induced leukemia NB4 cell
cycle arrest and apoptosis in vitro and in vivo. Cell Death Dis.
4:e9732013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Laskin WB and Miettinen M: Epithelioid
sarcoma: New insights based on an extended immunohistochemical
analysis. Arch Pathol Lab Med. 127:1161–1168. 2003.PubMed/NCBI
|
|
17
|
Song H, Ki SH, Kim SG and Moon A:
Activating transcription factor 2 mediates matrix
metalloproteinase-2 transcriptional activation induced by p38 in
breast epithelial cells. Cancer Res. 66:10487–10496. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Knippen S, Löning T, Müller V, et al:
Expression and prognostic value of activating transcription factor
2 (ATF2) and its phosphorylated form in mammary carcinomas.
Anticancer Res. 29:183–189. 2009.PubMed/NCBI
|
|
19
|
Schmittgen TD and Livak KJ: Analyzing
real-time PCR data by the comparative C (T) method. Nat Protoc.
3:1101–1108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yamada H and Sano Y: The biotinylation of
the rabbit serotonin antibody and its application to
immunohistochemical studies using the two-step ABC method.
Histochemistry. 83:285–289. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhao Y, Li Y, Han J, et al: Helicobacter
pylori enhances CIP2A expression and cell proliferation via
JNK2/ATF2 signaling in human gastric cancer cells. Int J Mol Med.
33:703–710. 2014.PubMed/NCBI
|
|
22
|
Bhoumik A, Huang TG, Ivanov V, et al: An
ATF2-derived peptide sensitizes melanomas to apoptosis and inhibits
their growth and metastasis. J Clin Invest. 110:643–650. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Maekawa T, Shinagawa T, Sano Y, et al:
Reduced levels of ATF-2 predispose mice to mammary tumors. Mol Cell
Biol. 27:1730–1744. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Berger AJ, Kluger HM, Li N, et al:
Subcellular localization of activating transcription factor 2 in
melanoma specimens predicts patient survival. Cancer Res.
63:8103–8107. 2003.PubMed/NCBI
|
|
25
|
Duffey D, Dolgilevich S, Razzouk S, Li L,
Green R and Gorti GK: Activating transcription factor-2 in survival
mechanisms in head and neck carcinoma cells. Head Neck.
33:1586–1599. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Knippen S, Löning T, Müller V, et al:
Expression and prognostic value of activating transcription factor
2 (ATF2) and its phosphorylated form in mammary carcinomas.
Anticancer Res. 29:183–189. 2009.PubMed/NCBI
|
|
27
|
Zhao Y, Li Y, Han J, et al:
Helicobacter pylori enhances CIP2A expression and cell
proliferation via JNK2/ATF2 signaling in human gastric cancer
cells. Int J Mol Med. 33:703–710. 2014.PubMed/NCBI
|
|
28
|
Bhoumik A, Huang TG, Ivanov V, et al: An
ATF2-derived peptide sensitizes melanomas to apoptosis and inhibits
their growth and metastasis. J Clin Invest. 110:643–650. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Deng X, Liu H, Huang J, et al: Ionizing
radiation induces prostate cancer neuroendocrine differentiation
through interplay of CREB and ATF2: Implications for disease
progression. Cancer Res. 68:9663–9670. 2008. View Article : Google Scholar : PubMed/NCBI
|