Introduction
Glioma is a common malignancy of the brain,
especially in middle-aged and elderly individuals, and the
proportion of elderly patients is on the increase worldwide
(1). Glioblastoma muhiforme patients
are treated by surgery, radiotherapy and chemotherapy because of
the instability of the tumor gene, the heterogeneity of the cells
and extensive infiltration, and the average survival rate is
approximately 12 months (2–5). Conventional radiotherapy and
chemotherapy do not eliminate all tumor cells, such as glial
blastoma cells through the downregulation of p53 gene and
upregulation of DNA repair enzymes including 06 methylguanine DNA
methyl transfer enzyme 1, to avoid the damage of chemotherapy
(6–8).
Thus, more effective combined therapy for patients with malignant
glioma is required at the molecular level. The identification of
neural stem cells (NSCs), NSCs and glioma stem cells, NSCs in the
brain specific targets can gather around the glioma cell clusters
and can even track single glioma cells away from glioma cell
clusters (9–11). Targeting treatment of glioma with NSCs
as a carrier is becoming a hot research topic. However, whether
NSCs have the function to ‘repair’ tumor cells and have the
potential to differentiate tumor is unknown.
The present study aimed to determine the therapeutic
effect of neural progenitor cells (NPCs) on a mouse model of
glioma, and to determine the possible mechanism of action
Materials and methods
Cell culture and drug treatment
The murine glioma cell line GL261 was obtained from
American Type Culture Collection (Manassas, VA, USA). The cells
were cultured in vitro in Iscove's Modified Dulbecco's
Medium (Life Technologies, Grand Island, NY, USA) supplemented with
10% fetal calf serum (Sigma-Aldrich, St. Louis, MO, USA), 100 U/ml
penicillin (P), and 100 g/ml streptomycin (S) (1% P/S; Life
Technologies) and 20 M β-mercaptoethanol (complete medium).
Anti-interleukin (IL)-22 neutralising mAbs were purchased from
Abcam (Cambridge, UK). mIL-22 protein was purchased from Peprotech,
Inc., Rocky Hill, NJ, USA.
Animal model
Six- to 12-week-old female C57BL/6 mice were
obtained from Charles River Laboratories. A brain tumor model was
set up as described previously (11).
A total of 1×104 GL261 glioma cells were washed twice in
phosphate-buffered saline (PBS) and adjusted to 5 µl of PBS in a
26-gauge Hamilton syringe (Yiliaoqixie Ltd., Shanghai, China). The
mice were anesthetized with isoflurane. After shaving the scalp and
making an incision, a burr hole was made in the skull 2 mm lateral
to the midline and 2 mm anterior to the bregma using a dental
drill. GL261 glioma cells and 1×105 NSC cell or PBS were
injected for >1 min at a depth of 2.5 mm below the dura mater
into the right cerebral hemisphere. The animals were observed daily
and sacrificed by cervical dislocation when characteristic symptoms
such as hunched posture, reduced mobility, and significant weight
loss (20%) occurred. The mice were bred under specific
pathogen-free conditions. Experimental protocols were approved by
the Institutional Animal Care and Use Committee. Animals without
such symptoms were regarded as long-term survivors after 90 days.
The study was approved by the ethics committee of Wuhan General
Hospital of Guangzhou Command.
Evaluation of cell proliferation
Cells were analyzed for proliferation using a cell
counting kit 8 assay (CCK-8 kit; Dojindo Molecular Technologies,
Inc., Shanghai, China). The cells were seeded into 96-well plates
at a density of 1×104 cells/well, and incubated in a
humidified atmosphere of 5% CO2 and 95% air overnight.
Normal cell medium containing NSC or vehicle at the concentrations
of 5 mM were added. A total of 10 µl CCK-8 (5 g/l in PBS) was added
after 72 h incubation. The plates were incubated for 4 h and the
blue dye formed was dissolved in 100 µl dimethylsulphoxide (DMSO or
Me2SO). The absorbance at 450 nm was recorded by ELISA
(Tiangen, Beijing, China).
Evaluation of cell apoptosis
Cells were stained with annexin V (fluorescein
isothiocyanate) to evaluate the cell death, according to the
manufacturer's instructions (BD Biosciences, San Jose, CA, USA).
Briefly, the cells were collected, washed with cold PBS and
suspended in binding buffer. After staining with annexin V, the
cells were analyzed using a FACScan flow cytometer (BD
Biosciences).
Quantitative polymerase chain reaction
(qPCR)
The cells were treated with NSC in vitro and
the culture period was 8 h. Total RNA was extracted from the cells
using the RNeasy mini kit (Qiagen, Beijing, China), followed by
complimentary DNA synthesis using the Superscript III first strand
synthesis kit (Invitrogen Life Technologies, Shanghai, China). qPCR
was performed on Bio-Rad amplifier using the Bio-Rad real-time PCR
mix (Bio-Rad, Berkeley, CA, USA). Data were analyzed using the
threshold cycle value normalized to the endogenous reference
gene.
Statistics
Data were presented as mean ± standard deviation.
Statistical differences were determined by a two-tailed paired
Student's t-test. SPSS 17.0 statistical software (SPSS, Inc.,
Chicago, IL, USA) was used for the analyses. P<0.05 was
considered to indicate a statistically significant difference.
Results
Therapeutic effect of NSC on mouse
model of GL261 glioma
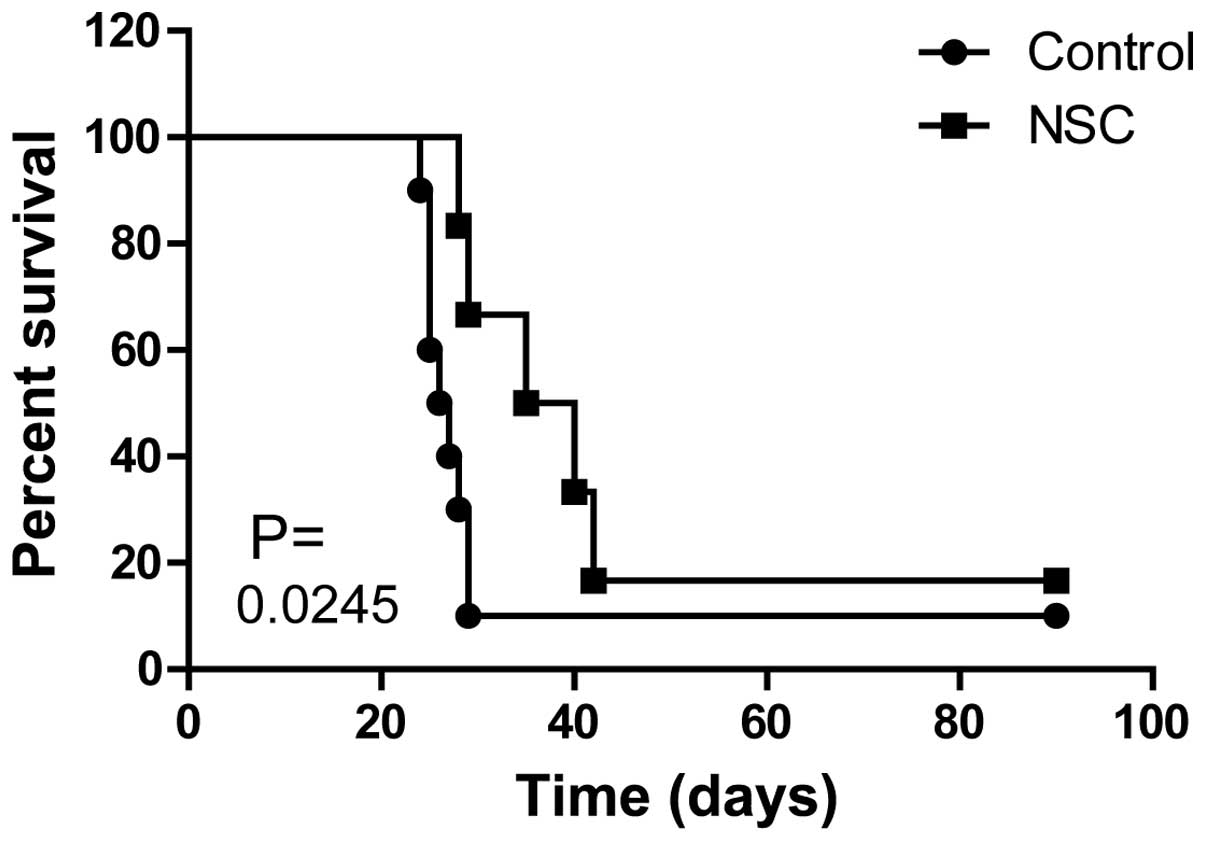
To study the effect of NSCs on the mouse glioma
model, we first injected stem cells or normal saline into the brain
of GL261-transplanted tumor mice. The mice of the stem cell group
showed a reduction of symptoms compared with the control group, and
showed that the survival time of mice was significantly prolonged
and the survival rate was increased (Fig.
1). Further measurement by ELISA (13) showed that the tumor volume of mic/e of
the stem cell group was significantly lower than that of the
control group (Fig. 1), which
supported the data of the survival rate.
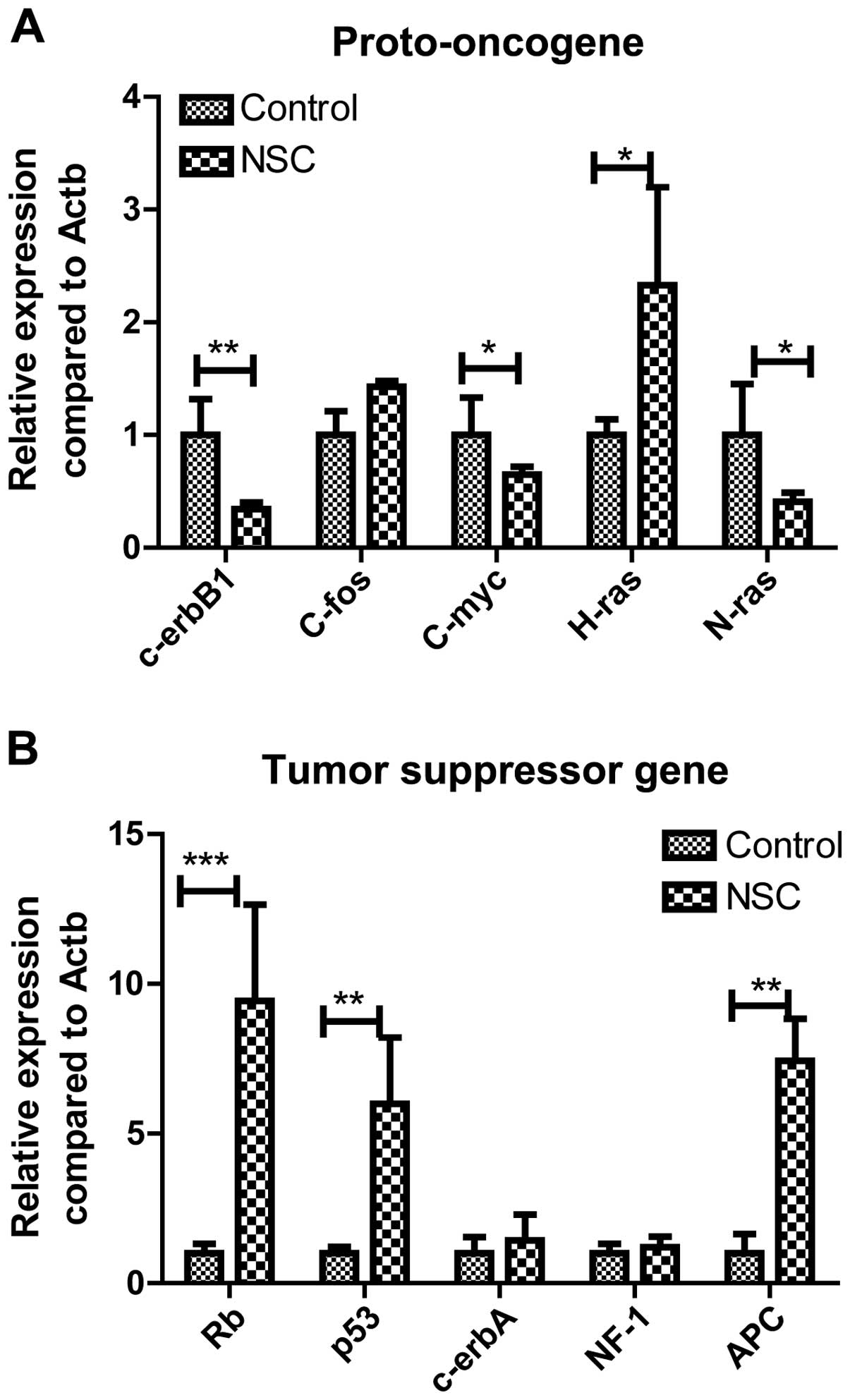
Effect of NSC in vitro on gene
expression of GL261 cells
In order to elucidate the mechanism of NSCs, the
cells were co-cultured with GL261 cells at aratio of 1:1 to detect
the expression of proto-oncogene and tumor suppressor gene. It was
found that NSCs inhibited the expression of c-erbB1/C-myc/N-ras,
but promoted the expression of tumor suppressor genes Rb,
p53, APC. Overall, NSCs can regulate the balance of
the proto-oncogene/tumor suppressor gene of GL261 glioma cells,
thereby effectively improving the symptoms of glioma (Fig. 2A and B).
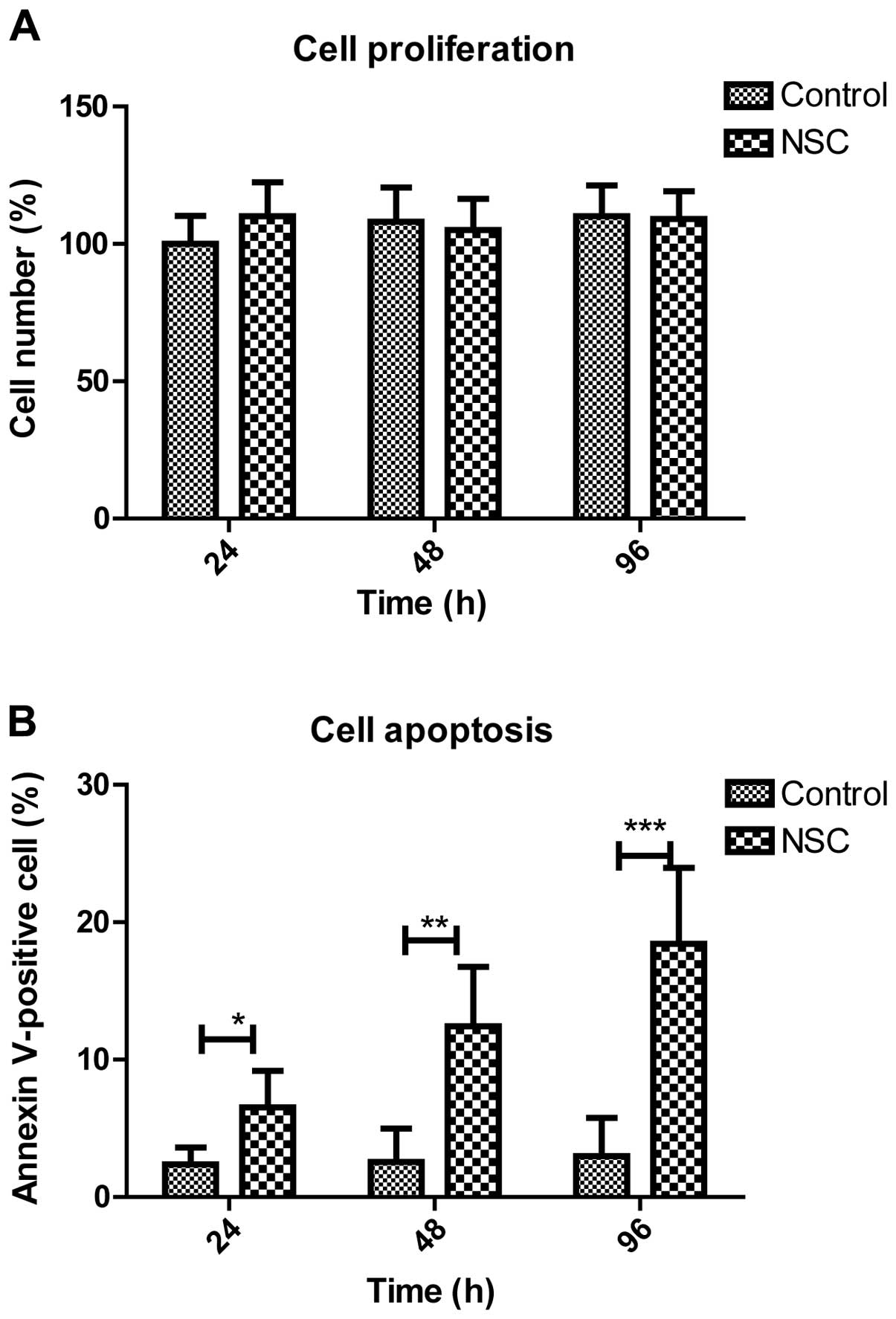
Effect of NSC in vitro on the
phenotype of GL261 cells
The results showed that compared with the control
group, the NSC treatment group inhibited the proliferation of GL261
cells as well inhibition to cell apoptosis, in a time-dependent
manner. The results suggested that NSC cells can mediate the
apoptotic phenotype of GL261 cells by regulating the expression of
oncogenes and tumor suppressor genes, and the mechanism of NSC
in vivo was preliminarily elucidated (Fig. 3A and B).
Discussion
Glioma is a common malignant tumor of the brain that
affects primarily elderly individuals (14). The occurrence and development of many
tumors including gliomas is associated with the imbalance of gene
expression, e.g., proto-oncogene and tumor suppressor gene. In
healthy conditions, the two genes are in a state of equilibrium.
However, once there is imbalance the expression of proto-oncogenes
are upregulated and the tumor suppressor genes are downregulated,
leadimg to development of the tumor eventually (15–17).
In the present study, mouse glioma 261 (Gl261) cells
are used frequently in experimental glioblastoma therapy. However,
no detailed description of the Gl261 tumor model is available. In
the present study, Gl261 cells carried point mutations in the
K-ras and p53 genes. Basal major histocompatibility
complex (MHC) I, but not MHCII, expression was detected in Gl261
cells. The introduction of interferon-γ-encoding genes increased
the expression of both MHCI and MHCII (13). The results of the present study showed
that the cells directly regulated transcription of the tumor genes.
Glioma stem cells and NSCs do not have a dependent relationship,
and there are even mutual constraints. Suzuki et al
(18) found that the cell factor
secreted by the nerve bulb, which is obtained by subculture can
inhibit the proliferation of glioma cells. In vitro, their
results showed that molecules with a weight between 50,000 and
100,000 have a role in inhibiting tumor activity. Additionally,
following implantation of 203G glioma cells and neural progenitor
cells in the cerebellar medulla pool, the survival time of mice in
the combination group was significantly prolonged (18). In addition, transforming growth
factor-β secreted by NSCs has an inhibitory effect on glioma
(19). Proto-oncogene is a gene that
is associated with cell proliferation, and is necessary to maintain
normal life activity with high conservatism in evolution. When the
structure or regulatory region of the proto-oncogene is altered,
the gene products increase or the activity of the gene is
strengthened, causing the cell to proliferate and form a tumor. The
findings of the current study have shown that NSC regulates the
original gene, but does not affect cell proliferation, indicating
that NSC has other regulatory pathways. Tumor occurrence is the
result of the synergistic action of multiple proto-oncogenes and
tumor suppressor genes.
Orian et al (20) detected the co-expression of
e-erbBI, C-mye, Ha/N-ras and C-fos
genes and found that the number of cancer genes was associated with
tumor. There were two types of gene co-expression in the improved
grade of the tumor. However, but there were 3 or 4 types of
co-expression of cancer genes in the variant star tumor and
glioblastoma cell tumor. The co-expression of
c-erbBI/Ha/N-ras/C-fos is the most common in the variant star
tumor, while the co-expression of c-erbBI/C-myc/Ha/N-ras was found
to be the most common in glioblastoma, with the latter being found
to have four co-expressions of oncogenes. Previous findings have
shown that C-erbB1 gene amplification was accompanied with the
amplification of C-myc and/or C-fos gene (21), suggesting that the epidermal growth
factor receptor and C-515 encoding platelet derived growth factor
can lead to growth of the expression level of the C-myc and
C-fos genes. The p53, p6z, pZI'ip and growth factor
receptors often co-regulate the cell proliferation cycle (22). In glioma cells, growth factor receptor
and P53 gene mutation were overexpressed the mutant p53 protein
inhibited the expression of p21c'Pl, while leading to the negative
regulation of p21dPI on the cyclin-dependent kinase being
decreased. Thus, the above factors accelerated the cells from G to
S stage. Previous studies have indicated that the occurrence of
glioma is the result of many cancer and tumor suppressor genes. Of
these, c-erbBI, C-myc, Ha/N-ras, C-fos and p53 genes have cleared
roles, although their interaction mechanism remains to be further
determined (23,24). In conclusion, NSC cell transplantation
can effectively improve MCF-7 breast cancer model in mice through
the dual regulation of the proto-oncogene/suppressor of tumor
cells, and a potentially effective method for treating breast
cancer.
Acknowledgements
The present study was supported by the Medical
Scientific Research Foundation of Hubei Province, China (no.
WJ2015MB118).
References
|
1
|
Goodenberger ML and Jenkins RB: Genetics
of adult glioma. Cancer Genet. 205:613–621. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Buck JR, McKinley ET, Fu A, Abel TW,
Thompson RC, Chambless L, Watchmaker JM, Harty JP, Cooper MK and
Manning HC: Preclinical TSPO Ligand PET to Visualize Human Glioma
Xenotransplants: A Preliminary Study. PLoS One. 10:e01416592015.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Codo P, Weller M, Meister G, Szabo E,
Steinle A, Wolter M, Reifenberger G and Roth P: MicroRNA-mediated
down-regulation of NKG2D ligands contributes to glioma immune
escape. Oncotarget. 5:7651–7662. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Breunig JJ, Levy R, Antonuk CD, Molina J,
Dutra-Clarke M, Park H, Akhtar AA, Kim GB, Hu X, Bannykh SI, et al:
Ets factors regulate neural stem cell depletion and gliogenesis in
Ras Pathway Glioma. Cell Reports. 12:258–271. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Boisselier B, Gállego Pérez-Larraya J,
Rossetto M, Labussière M, Ciccarino P, Marie Y, Delattre JY and
Sanson M: Detection of IDH1 mutation in the plasma of patients with
glioma. Neurology. 79:1693–1698. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Blough MD, Beauchamp DC, Westgate MR,
Kelly JJ and Cairncross JG: Effect of aberrant p53 function on
temozolomide sensitivity of glioma cell lines and brain tumor
initiating cells from glioblastoma. J Neurooncol. 102:1–7. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lalezari S, Chou AP, Tran A, Solis OE,
Khanlou N, Chen W, Li S, Carrillo JA, Chowdhury R, Selfridge J, et
al: Combined analysis of O6-methylguanine-DNA methyltransferase
protein expression and promoter methylation provides optimized
prognostication of glioblastoma outcome. Neuro-oncol. 15:370–381.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Parkinson JF, Wheeler HR, Clarkson A,
McKenzie CA, Biggs MT, Little NS, Cook RJ, Messina M, Robinson BG
and McDonald KL: Variation of O(6)-methylguanine-DNA
methyltransferase (MGMT) promoter methylation in serial samples in
glioblastoma. J Neurooncol. 87:71–78. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Aboody KS, Brown A, Rainov NG, Bower KA,
Liu S, Yang W, Small JE, Herrlinger U, Ourednik V, Black PM, et al:
Neural stem cells display extensive tropism for pathology in adult
brain: Evidence from intracranial gliomas. Proc Natl Acad Sci USA.
97:12846–12851. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Barami K: Relationship of neural stem
cells with their vascular niche: implications in the malignant
progression of gliomas. J Clin Neurosci. 15:1193–1197. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sanai N, Alvarez-Buylla A and Berger MS:
Neural stem cells and the origin of gliomas. N Engl J Med.
353:811–822. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Grauer OM, Molling JW, Bennink E, Toonen
LW, Sutmuller RP, Nierkens S and Adema GJ: TLR ligands in the local
treatment of established intracerebral murine gliomas. J Immunol.
181:6720–6729. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Szatmári T, Lumniczky K, Désaknai S,
Trajcevski S, Hídvégi EJ, Hamada H and Sáfrány G: Detailed
characterization of the mouse glioma 261 tumor model for
experimental glioblastoma therapy. Cancer Sci. 97:546–553. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ostrom QT, Bauchet L, Davis FG, Deltour I,
Fisher JL, Langer CE, Pekmezci M, Schwartzbaum JA, Turner MC, Walsh
KM, Wrensch MR and Barnholtz-Sloan JS: The epidemiology of glioma
in adults: a ‘state of the science’ review. Neuro-Oncol.
16:899–913. 2014. View Article : Google Scholar
|
|
15
|
Lee SG, Kim K, Kegelman TP, Dash R, Das
SK, Choi JK, Emdad L, Howlett EL, Jeon HY, Su ZZ, et al: Oncogene
AEG-1 promotes glioma-induced neurodegeneration by increasing
glutamate excitotoxicity. Cancer Res. 71:6514–6523. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sano M, Genkai N, Yajima N, Tsuchiya N,
Homma J, Tanaka R, Miki T and Yamanaka R: Expression level of ECT2
proto-oncogene correlates with prognosis in glioma patients. Oncol
Rep. 16:1093–1098. 2006.PubMed/NCBI
|
|
17
|
ten Haaf A, Bektas N, von Serenyi S, Losen
I, Arweiler EC, Hartmann A, Knüchel R and Dahl E: Expression of the
glioma-associated oncogene homolog (GLI) 1 in human breast cancer
is associated with unfavourable overall survival. BMC Cancer.
9:2982009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Suzuki T, Izumoto S, Wada K, Fujimoto Y,
Maruno M, Yamasaki M, Kanemura Y, Shimazaki T, Okano H and
Yoshimine T: Inhibition of glioma cell proliferation by neural stem
cell factor. J Neurooncol. 74:233–239. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tran TT, Uhl M, Ma JY, Janssen L, Sriram
V, Aulwurm S, Kerr I, Lam A, Webb HK, Kapoun AM, et al: Inhibiting
TGF-beta signaling restores immune surveillance in the SMA-560
glioma model. Neuro-oncol. 9:259–270. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Orian JM, Vasilopoulos K, Yoshida S, Kaye
AH, Chow CW and Gonzales MF: Overexpression of multiple oncogenes
related to histological grade of astrocytic glioma. Br J Cancer.
66:106–112. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhu R, Kang S and Wu D: Expression of p53,
c-erbB1, c-myc and p16 gene proteins in human glioma. Zhonghua Bing
Li Xue Za Zhi. 26:343–345. 1997.(In Chinese). PubMed/NCBI
|
|
22
|
Zheng X, Yao Y, Xu Q, Tu K and Liu Q:
Evaluation of glioma-associated oncogene 1 expression and its
correlation with the expression of sonic hedgehog, E-cadherin and
S100a4 in human hepatocellular carcinoma. Mol Med Rep. 3:965–970.
2010.PubMed/NCBI
|
|
23
|
Ohta M, Tateishi K, Kanai F, Watabe H,
Kondo S, Guleng B, Tanaka Y, Asaoka Y, Jazag A, Imamura J, et al:
p53-Independent negative regulation of p21/cyclin-dependent
kinase-interacting protein 1 by the sonic
hedgehog-glioma-associated oncogene 1 pathway in gastric carcinoma
cells. Cancer Res. 65:10822–10829. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Godlewski J, Nowicki MO, Bronisz A,
Williams S, Otsuki A, Nuovo G, Raychaudhury A, Newton HB, Chiocca
EA and Lawler S: Targeting of the Bmi-1 oncogene/stem cell renewal
factor by microRNA-128 inhibits glioma proliferation and
self-renewal. Cancer Res. 68:9125–9130. 2008. View Article : Google Scholar : PubMed/NCBI
|