Introduction
Hamartomas are benign tumors formed from the
disordered growth of normal cells and tissues at the affected site
(1). Pancreatic hamartoma is
extremely rare and accounts for <1% of hamartoma; the etiology
is unknown and the disease has a low morbidity (2). With the increased utilization of imaging
technology, such as endoscopic ultrasound, there has been an
increased detection of asymptomatic lesions of the pancreas;
however, the differentiation between hamartomas and other benign
tumors or malignancies is extremely difficult (3). The final diagnosis is usually made
according to histopathological and immunohistochemical results
(4). Due to the dangers of biliary
obstruction or hemorrhage, a pancreatectomy is necessary (5). No recurrence usually occurs following
radical excision. Pancreatic hamartoma can be divided into two
subgroups: Solid and cystic, or solid (6). The majority of cases reported in the
literature are cystic hamartomas (1–3,7–13). The
present study reports the case of a solid pancreatic hamartoma that
was diagnosed after surgery.
Case report
A 53-year old female was admitted to the Department
of Abdominal Surgery, Chinese Academy of Medical Sciences, Cancer
Hospital (Beijing, China) in October 2014, due to a 2-month history
of abdominal pain, accompanied by slight anorexia and 2 kg of
weight loss. Physical examination did not detect any abnormalities.
The history of the patient did not disclose any systemic diseases
or previous surgeries. The patient reported no alcohol consumption
or cigarette use, and there was no family history of
tumor-associated disease. Blood tests indicated that tumor marker
levels [carcinoembryonic antigen, carbohydrate antigen (CA)19-9,
CA-125 and CA72-4] were within the normal ranges, and pancreatic
exocrine and endocrine function was sufficient. Other standard
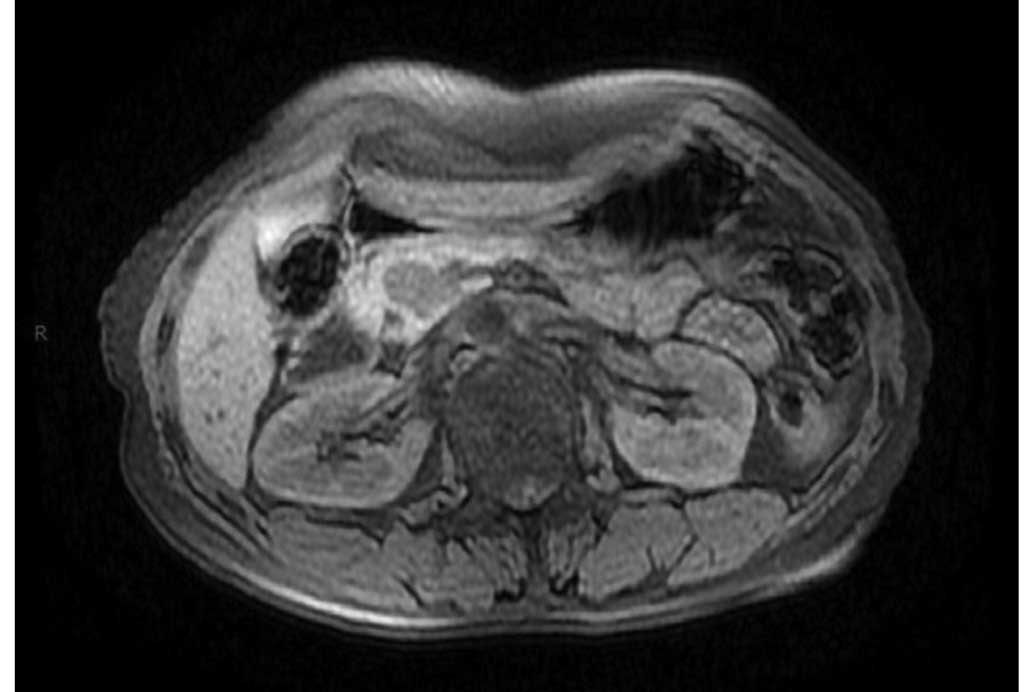
blood tests were unremarkable. Abdominal magnetic resonance imaging
(MRI; MAGNETOM Skyra, Siemens Healthcare, Erlangen, Germany) was
performed and revealed a 22×14-mm, predominantly well-circumscribed
lesion in the pancreatic tuberculosis. The lesion exhibited a low
signal on T1-weighted images (T1WI) and T2WI/fat suppression
images, accompanied by certain hyperintense areas (Fig. 1). Endosonography (TGF-UC260J; Olympus
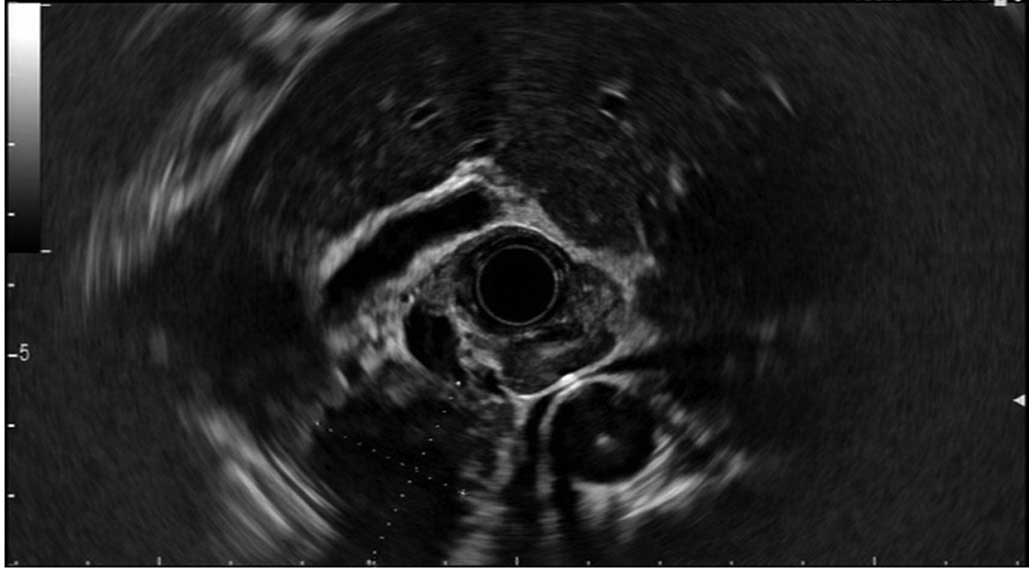
Corporation, Tokyo, Japan) was also performed and revealed a
2.83×2.27-cm, hypoechogenic lesion located in the head of the
pancreas. Endoscopic ultrasound (EUS) fine-needle aspiration was
not performed due to the problematic position of the lesion, which
was particularly close to the superior mesenteric vein (Fig. 2). The combined imaging results were
not enough to form a reliable diagnosis; therefore, a formal
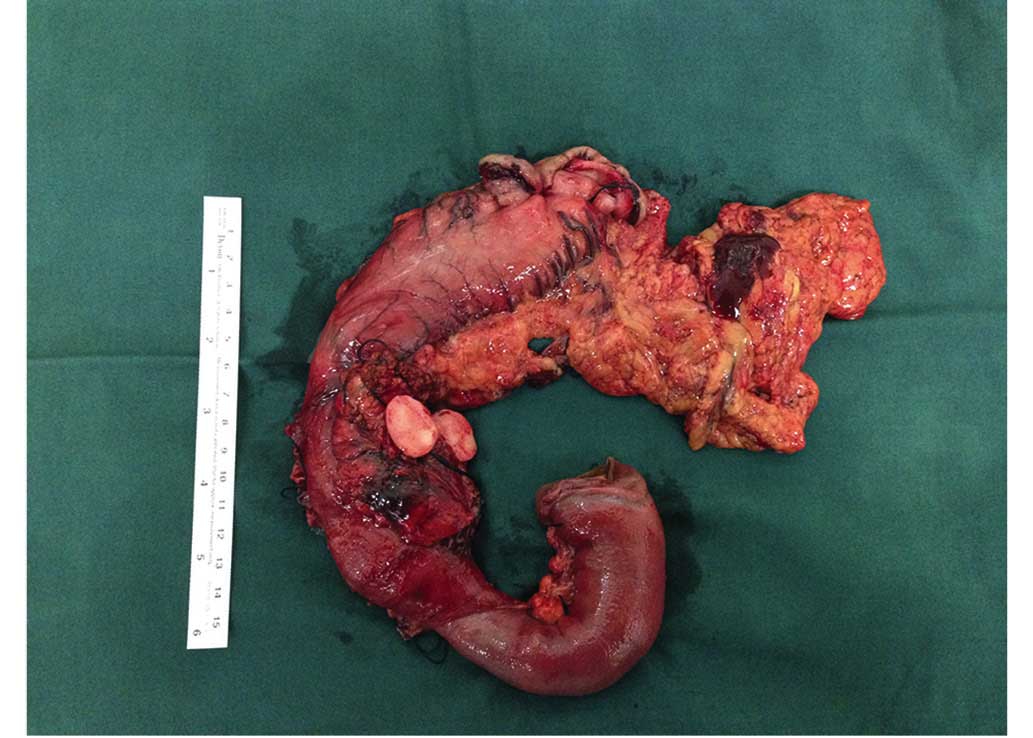
pancreaticoduodenectomy was performed. During the surgery, a
2.3×1.5×1.5-cm, firm mass with an intact capsule was identified,
which appeared to be embedded in the parenchyma of the head of the
pancreas (Fig. 3). On the resected
surface of the tumor, a well-demarcated, solid, homogeneously
white-colored nodule was identified. The resected tissue was
formalin (Beijing Saichi Biological Technology Co., Ltd., Beijing,
China)-fixed, paraffin (Beijing Saichi Biological Technology Co.,
Ltd., Beijing, China)-embedded and cut into 4-µm sections prior to
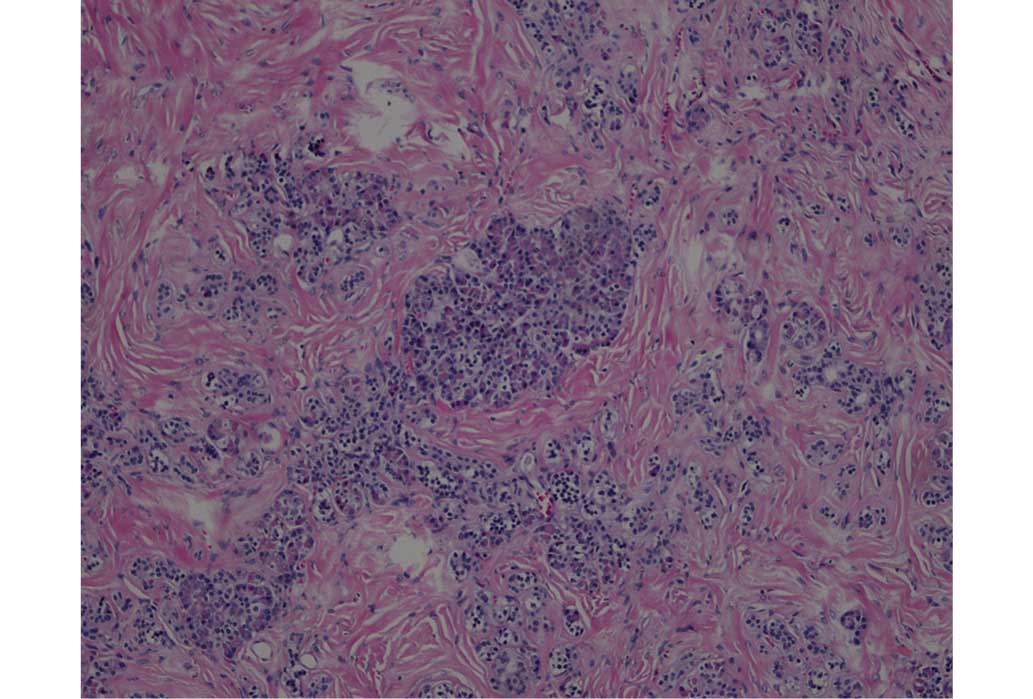
microscopic examination (CX23; Olympus Corporation), which
demonstrated that the lesion was composed of disarranged ductal and
acinar cells, embedded in a markedly fibrous stroma (hematoxylin
and eosin stain; Beijing Saichi Biological Technology Co., Ltd.)
(Fig. 4). Immunohistochemical
staining indicated that the lesion was positive for
α-1-antitrypsin, α-1-antichymotrypsin and s100, with a Ki-67 score
of <1%. Small amounts of disordered acinar cells and ductal
epithelium were immunohistochemically positive for cluster of
differentiation (CD)117, CD56, chromogranin A, progesterone
receptor and synaptophysin, and negative for cytokeratin 19, p63
and vimentin. Thus, the histological diagnosis was confirmed as
pancreatic hamartoma. The post-operative clinical course was stable
without complications, and the patient was discharged following 15
days of hospitalization. At 55 months of follow-up, no recurrence
was observed.
Discussion
A hamartoma is a focal, benign malformation that
resembles a neoplasm in the tissue of its origin (14). This lesion is not a real tumor, and it
grows at the same rate as its adjacent tissues. Hamartomas may
develop in various areas of the body, and are typically observed in
the lungs, heart, kidneys, spleen or other vascular organs. These
lesions are commonly asymptomatic and may remain undetected unless
identified incidentally during imaging analyses (14).
Pancreatic hamartoma is extremely rare. Only 23
cases, including the present case, have been reported in the
literature (Table I), with the first
case being described by Anthony et al in 1977 (7). The development of this disease may occur
at any age, even in young children, however, the average age of
occurrence is 40–60 years (median, 42.23 years) (4,5,8–10,15–18).
Morbidity is not equal between men and women, with a male to female
ratio of 1.5:1. In contrast to hamartoma in other organs, the
majority of pancreatic hamartoma cases are accompanied by pain,
abdominal discomfort or certain vague symptoms, including dyspepsia
and weight loss (11,19). Tumors are often located in the head of
the pancreas, but signs of jaundice are rare.
 | Table I.Clinicopathological features of
pancreatic tumors reported as pancreatic hamartoma in the
literature (n=23). |
Table I.
Clinicopathological features of
pancreatic tumors reported as pancreatic hamartoma in the
literature (n=23).
| First author/s,
year | Age, years | Gender | Size, cm | Surgery | Pancreatitis | Solid and
cystic/solid | Refs. |
|---|
| Anthony et al,
1977 | 46 | M | 1.6 | LD | No | Solid and cystic | (7) |
|
| 35 | M | Multiple | LR | Yes | NR |
|
|
| 58 | M | 1.0 | Autopsy | No | NR |
|
| Noltenius and
Colmant, 1977 | 52 | F | Multiple | Autopsy | Yes | NR | (15) |
| Burt et al,
1983 | 0.65 | F | 11.5 | TP | No | Solid and cystic | (8) |
| Flaherty and
Benjamin, 1992 | 1.67 | F | 9.0 | LR | No | Solid and cystic | (9) |
| Izbicki et al,
1994 | 25 | M | 10.6 | PD | No | Solid and cystic | (10) |
| Wu et al,
1998 | 39 | M | 8.0 | PD | Yes | NR | (16) |
| McFaul et al,
2004 | 29 | M | 1.0 | PD | Yes | NR | (19) |
|
| 62 | M | 3.5 | PD | Yes | NR |
|
| Pauser et al,
2005 | 36 | F | 7.0 | PD | No | Solid and cystic | (12) |
|
| 55 | F | 3.0 | DP | No | Solid and cystic |
|
| Pauser et al,
2005 | 51 | M | 3.0 | LR | No | Solid | (13) |
|
| 54 | F | 2.0 | DP | No | Solid |
|
| Nagata et al,
2007 | 58 | F | 1.9 | DP | No | Solid | (1) |
| Thrall et al,
2008 | 3 | M | 3.0 | PD | No | Solid and cystic | (11) |
| Sampelean et
al, 2009 | 46 | M | 8.0 | PD | NR | Solid | (17) |
| Durczynski et
al, 2011 | 69 | M | 3.0 | LR | No | Solid | (3) |
| Kawakami et
al, 2012 | 78 | F | 1.8 | PD | No | Solid | (6) |
| Kim et al,
2012 | 52 | M | 2.2 | PD | No | Solid and cystic | (2) |
| Sueyoshi et
al, 2013 | 3 | M | 3.0 | PD | No | Solid and cystic | (18) |
| Addeo et al,
2014 | 61 | M |
2.4 | DP | No | Solid | (4) |
| Present study | 48 | F |
2.2 | PD | No | Solid |
|
Pancreatic hamartoma is typically composed of
disorganized, well-differentiated exocrine and endocrine pancreatic
tissue. Acinar, islet and ductal cells are the three main
components that form the lesions (1).
A specific cell type, termed spindle-shaped cells, exhibit
immunoreactivity for CD34 and CD117 in a number of pancreatic
hamartoma cases (1,12). Pancreatic hamartoma is classified as
solid or solid and cystic by macroscopic findings (13). Although the pathogenesis and origin of
this disease remains unclear, it should only be diagnosed in
patients without evidence of chronic pancreatitis, as chronic
pancreatitis often presents with a depletion of acinar cells in the
fibrous stroma, thus mimicking hamartoma with a lack of such cells
(12).
To date, the existing imaging methods employed to
characterize pancreatic hamartoma lack the accuracy to successfully
distinguish it from other diseases. The majority of lesions that
develop in the pancreas are malignant or have malignant potential,
therefore, surgery is the optimal treatment in such instances, and
a complete resection with negative margins is imperative (3). The treatment of choice for hamartoma is
a conventional pancreatectomy or a pancreas-preserving surgery
(central pancreatectomy or enucleation). A conventional
pancreatectomy, including pancreatoduodenectomy and distal
pancreatectomy, may sacrifice the normal pancreatic parenchyma and
be associated with the risk of post-operative diabetes mellitus or
endocrine and exocrine pancreatic insufficiency (2). Considering the benign behavior of this
tumor, pancreas-preserving surgery is recommended, which has the
advantages of preserving the integrity of the gastrointestinal
tract and splenic function, and sparing the maximal pancreatic
endocrine and exocrine function by avoiding an extended resection
of the pancreas (20). However, there
is debate regarding the use of this treatment. Since the majority
of pancreatic hamartoma cases have a good prognosis and
pancreatectomies are prone to the development of fistulas or other
complications, the ‘wait and see’ policy is an alternative
treatment for asymptomatic patients in order to exclude the
diagnosis of a malignant tumor (20).
In conclusion, pancreatic hamartoma may be detected
incidentally, without the patient presenting with any signs or
symptoms of disease. Clinical symptoms are dependent on tumor size
and location, and the diagnosis of this disease primarily depends
on imaging techniques, including computed tomography, MRI and EUS.
However, pancreatic hamartoma is difficult to differentiate from
other benign pancreatic lesions. For symptomatic patients or those
with an indefinite diagnosis, a complete surgical excision is
recommended. Following a successful pancreaticoduodenectomy, the
patient in the present study was diagnosed with a pancreatic
hamartoma on histological examination, and no recurrence has since
been observed.
References
|
1
|
Nagata S, Yamaguchi K, Inoue T, Yamaguchi
H, Ito T, Gibo J, Tanaka M and Tsuneyoshi M: Solid pancreatic
hamartoma. Pathol Int. 57:276–280. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kim HH, Cho CK, Hur YH, Koh YS, Kim JC,
Kim HJ, Kim JW, Kim Y and Lee JH: Pancreatic hamartoma diagnosed
after surgical resection. J Korean Surg Soc. 83:330–334. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Durczynski A, Wiszniewski M, Olejniczak W,
Polkowski M, Sporny S and Strzelczyk J: Asymptomatic solid
pancreatic hamartoma. Arch Med Sci. 7:1082–1084. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Addeo P, Tudor G, Oussoultzoglou E,
Averous G and Bachellier P: Pancreatic hamartoma. Surgery.
156:1284–1285. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Inoue H, Tameda M, Yamada R, Tano S,
Kasturahara M, Hamada Y, Tanaka K, Horiki N and Takei Y: Pancreatic
hamartoma: A rare cause of obstructive jaundice. Endoscopy.
46(Suppl 1): 157–158. 2014.
|
|
6
|
Kawakami F, Shimizu M, Yamaguchi H, Hara
S, Matsumoto I, Ku Y and Itoh T: Multiple solid pancreatic
hamartomas: A case report and review of the literature. World J
Gastrointest Oncol. 4:202–206. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Anthony PP, Faber RG and Russell RC:
Pseudotumours of the pancreas. BMJ. 1:8141977. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Burt TB, Condon VR and Matlak ME: Fetal
pancreatic hamartoma. Pediatr Radiol. 13:287–289. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Flaherty MJ and Benjamin DR: Multicystic
pancreatic hamartoma: A distinctive lesion with immunohistochemical
and ultrastructural study. Hum Pathol. 23:1309–1312. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Izbicki JR, Knoefel WT, Müller-Höcker J
and Mandelkow HK: Pancreatic hamartoma: A benign tumor of the
pancreas. Am J Gastroenterol. 89:1261–1262. 1994.PubMed/NCBI
|
|
11
|
Thrall M, Jessurun J, Stelow EB, Adsay NV,
Vickers SM, Whitson AK, Saltzman DA and Pambuccian SE: Multicystic
adenomatoid hamartoma of the pancreas: A hitherto undescribed
pancreatic tumor occurring in a 3-year-old boy. Pediatr Dev Pathol.
11:314–320. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pauser U, da Silva MT, Placke J, Klimstra
DS and Klöppel G: Cellular hamartoma resembling gastrointestinal
stromal tumor: A solid tumor of the pancreas expressing c-kit
(CD117). Mod Pathol. 18:1211–1216. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pauser U, Kosmahl M, Kruslin B, Klimstra
DS and Klöppel G: Pancreatic solid and cystic hamartoma in adults:
Characterization of a new tumorous lesion. Am J Surg Pathol.
29:797–800. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Heald B, Burke CA, Kalady M and Eng C: ACG
guidelines on management of PTEN-hamartoma tumor Ssyndrome: Does
the evidence support so much so young? Am J Gastroenterol.
110:1733–1734. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Noltenius H and Colmant HJ: Excessive
hyperplasia of the exocrine pancreatic tissue and Wernicke's
encephalopathy (author's transl). Med Klin. 72:2155–2158. 1977.(In
German). PubMed/NCBI
|
|
16
|
Wu SS, Vargas HI and French SW: Pancreatic
hamartoma with Langerhans cell histiocytosis in a draining lymph
node. Histopathology. 33:485–487. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sampelean D, Adam M, Muntean V, Hanescu B
and Domsa I: Pancreatic hamartoma and SAPHO syndrome: A case
report. J Gastrointestin Liver Dis. 18:483–486. 2009.PubMed/NCBI
|
|
18
|
Sueyoshi R, Okazaki T, Lane GJ, Arakawa A,
Yao T and Yamataka A: Multicystic adenomatoid pancreatic hamartoma
in a child: Case report and literature review. Int J Surg Case Rep.
4:98–100. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
McFaul CD, Vitone LJ, Campbell F, Azadeh
B, Hughes ML, Garvey CJ, Ghaneh P, Neoptolemos JP and Longnecker
DS: Pancreatic hamartoma. Pancreatology. 4:533–538. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Beger HG, Siech M, Poch B, Mayer B and
Schoenberg MH: Limited surgery for benign tumours of the pancreas:
A systematic review. World J Surg. 39:1557–1566. 2015. View Article : Google Scholar : PubMed/NCBI
|