Introduction
Tumors of the ovaries account for 30% of all cancers
of the female genital system; they are a heterogeneous group of
neoplasms, divided into a number of different subgroups, depending
largely on histological and cytological features (1).
The majority (90%) of ovarian tumors are epithelial
in nature. Malignant epithelial ovarian tumors are currently
divided into high-grade serous, low-grade serous, endometrioid,
mucinous and clear cell carcinomas (2).
Borderline tumors of the ovary are neoplasms of low
malignant potential. These tumors present with cellular atypia, but
are not invasive. A few of these tumors share genomic and molecular
features with carcinomas (3), but it
remains unclear as to whether this is a feature of only a subset or
of borderline tumors in general.
Sex-cord stromal tumors account for 8% of all
ovarian tumors and are further classified based on their
predominant cell content; for example, granulosa cell tumors
contain ≥10% granulosa cells. The thecoma-fibroma group of tumors
is dominated by theca cells (thecoma), stromal fibroblasts
(fibroma) or the two cell types in different proportions
(thecofibroma) (4).
Heterogeneity among ovarian tumors biologically and
clinically represents a considerable challenge. The molecular and
genetic profiles of the tumors could aid in predicting their
inherent aggressiveness and could also provide keys to more
specific treatments. As a contribution toward this goal, the
present study analyzed 187 samples of different types of ovarian
tumors, namely, granulosa cell tumors, thecofibromas, fibromas,
teratomas, borderline tumors and infiltrating carcinomas of
different histologies, for their expression of the high-mobility
group AT-hook 2 (HMGA2) gene, for mutations of the
isocitrate dehydrogenase (NADP(+)) 1, cytosolic (IDH1),
isocitrate dehydrogenase (NADP(+)) 2, mitochondrial (IDH2)
and telomerase reverse transcriptase (TERT) genes, and for
methylation of the promoter of the O6-methylguanine-DNA
methyltransferase (MGMT) gene.
Materials and methods
Tumor material
The material consisted of fresh frozen samples from
187 ovarian tumors surgically resected at The Norwegian Radium
Hospital (Oslo, Norway) between December 1999 and January 2010. The
tumors have been previously characterized for chromosomal
aberrations and genomic imbalances (3,5,6). The present series consisted of 39
sex-cord stromal tumors (19 thecofibromas, 16 fibromas, 2 granulosa
cell tumors and 2 teratomas), 22 borderline tumors and 126
carcinomas (56 serous high-grade carcinomas, 30 endometrioid
carcinomas, 18 mucinous carcinomas, 12 clear cell carcinomas, 4
serous low-grade carcinomas, 1 undifferentiated carcinoma, and 5
mixed-type carcinomas). This study was approved by the regional
ethics committee (Regional Komité For Medisinsk Forskning-Setikk
Sør-Øst, Norge; http://helseforskning.etikkom.no) and written informed
consent was obtained from the patients.
DNA and RNA extraction, and cDNA
synthesis
DNA extraction was performed using the Maxwell 16
Extractor (Promega, Madison, WI, USA) and the Maxwell 16 Tissue DNA
Purification kit (Promega) according to the manufacturer's
instructions. RNA was extracted using the miRNeasy kit (Qiagen
GmbH, Hilden, Germany) and QIAcube (Qiagen GmbH). The concentration
and purity of the DNA and RNA were measured with the Nanovue
Spectrophotometer (GE Healthcare, Pittsburgh, PA, USA). Extracted
RNA (1 µg) was reverse-transcribed in a 20-µl reaction volume using
the iScript Advanced cDNA Synthesis kit according to the
manufacturer's instructions (Bio-Rad Laboratories, Oslo,
Norway).
Molecular analyses
Molecular analyses of IDH1, IDH2, TERT and
MGMT were performed according to previously described
protocols (7).
HMGA2
Molecular analyses of HMGA2 were performed
using a previously described protocol (7), but with minor variations. In cases 7, 8,
9, 10 and 11, the nested polymerase chain reaction (PCR) of the 3′
rapid amplification of cDNA ends (3′RACE) was performed using the
Touchdown-PCR conditions described by Korbie et al (8) to increase the specificity of the PCR and
improve the quality of the products. The 3′RACE results for cases
7, 8, 9 and 11 were validated through additional PCRs using primers
obtained by the sequence of the 3′RACE products (Table I). More specifically, HMGA2-928-F1 and
HMGA2R169 were used in case 7, HMGA2F233 and HMGA2R233 were used in
case 8, HMGA2-928F and HMGA2R263 were used in case 9, and
HMGA2-928F and HMGA2R175 were used in case 11 (Table II). The products of these PCRs were
sequenced and analyzed by BLAT (http://genome.ucsc.edu/cgi-bin/hgBlat).
 | Table I.Specific primers for the 3′ rapid
amplification of cDNA ends product amplification. |
Table I.
Specific primers for the 3′ rapid
amplification of cDNA ends product amplification.
| Primer name | Primer
sequence |
|---|
| HMGA2-982-F1 |
5′-CAAGAGTCCCTCTAAAGCAGCTCA-3′ |
| HMGA2R169 |
5′-TGGGATGCAGACTTCAGTTGGAA-3′ |
| HMGA2F323 |
5′-CCCTATCACCTCATCTCCCG-3′ |
| HMGA2R323 |
5′-TTGTCCACTCATTCAGCAGATC-3′ |
| HMGA2R363 |
5′-CAGGCATGGCTCTGCATGTG-3′ |
| HMGA2R175 |
5′-TGACCACTGAATTCTGGCCTCA-3′ |
 | Table II.Truncated forms of HMGA2 found
in ovarian tumors. |
Table II.
Truncated forms of HMGA2 found
in ovarian tumors.
| Case/lab no. | Diagnosis | RT-PCR | Sequence | Karyotype |
|---|
| 1/05–1270 | Mucinous
carcinoma | Truncated
transcript | U29117 | Culture
failure |
| 2/07–1630 | Mucinous
carcinoma | Truncated
transcript | Not available | 46,XX[69] |
| 3/08–1650 | Mucinous
carcinoma | Truncated
transcript | U29117 | 46, XX,
del(1)(q21)[2] / 46, XX[88] |
| 4/01–196 | Endometrioid
carcinoma | Truncated
transcript | U29117 | 46,XX[16] |
| 5/07–0449 | Endometrioid
carcinoma | Truncated
transcript | U29117 | 46,XX[12] |
| 6/03–481 | Borderline | Truncated
transcript | AK31139 | 46,XX[32] |
| 7/01–169 | Serous
high-grade | Truncated
transcript | Novel | 46,XX[48] |
| 8/02–333 | Serous
high-grade | Truncated
transcript | Novel |
47~49,XX,+8,+9[2]/49,idem,+5,-6,+7[4]/54,
idem,+3,+5,+6,+7,+14,+17,+19[5] |
| 9/02–363 | Serous
high-grade | Truncated
transcript | Not available |
65~68,XX,-X,+1,+2,del(3)(p13),+4, add(4)(p12) ×2, add(5)(p15),-6,+7, add(7)(p15),-8,-9,-12,-13,-14,-15,-17,-18,der(19)add(19)(p13)add(19)(q13)+20,+20,-21,-22,+2mar[9] |
| 10/04–499 | Serous
high-grade | Truncated
transcript | Not available | 46,XX[5] |
| 11/04–715 | Serous
high-grade | Truncated
transcript | Novel | Culture
failure |
Methylation-specific
quantitative-polymerase chain reaction (MSP-qPCR)
Unmethylated cytosine residues were converted to
uracil by bisulfite treatment of 1.3 µg DNA using the EpiTect
Bisulfite Kit (Qiagen GmbH) according to the manufacturers'
protocol. Following conversion, DNA was eluted in buffer (Qiagen
GmbH) to a final concentration of 30 ng/µl. Bisulfite-treated DNA
(2 µl) was amplified with a qPCR in a 20-µl reaction mixture
containing 10 µl of Precision Mix (Bio-Rad Laboratories, Oslo,
Norway), 0.4 µl of forward and reverse primers, and 7.2 µl of
H2O. Two different mixes were used, one containing the
primer sequences of MGMT for the unmethylated reaction
[5′-TTTGTGTTTTGATGTTTGTAGGTTTTTGT-3′ (forward) and
5′-AACTCCACACTCTTCCAAAAACAAAACA-3′ (reverse)] and one mix
containing the primers for the methylated reaction
[5′-TTTCGACGTTCGTAGGTTTTCGC-3′ (forward) and
5′-GCACTCTTCCGAAAACGAAACG-3′ (reverse)]. All qPCR analyses were
performed in triplicate with the CFX96 Touch Real-Time PCR
detection system (Bio-Rad Laboratories) and methylated and
unmethylated standards (Epitect PCR Control DNA Set; Qiagen GmbH)
were used as controls. The thermal cycling included 2 min at 90°C,
followed by 40 cycles of 50 sec at 95°C, 50 sec at 60°C, 50 sec at
72°C, and 10 sec at 95°C, and a final process in which the
temperature increased by 0.5°C in steps, starting from 65°C and
ending at 95°C. Data were analyzed with the CFX Manager software
(Bio-Rad Laboratories), and all the melting curves of the samples
were referenced to the methylated and unmethylated controls
(9).
Results
HMGA2
Informative results for HMGA2 expression were
obtained for all 161 tumors from which RNA was available. The gene
was found to be expressed in 74.5% of the samples (120/161 tumors).
The frequency varied among the different tumor types and
histological subgroups, with the thecofibromas showing the highest
frequency (100%; 16/16 tumors), followed by the high-grade serous
carcinomas (90.2%; 37/41 tumors), the fibromas (73.3%; 11/15), the
borderline tumors (66.7%; 14/21 tumors), the mucinous carcinomas
(61.1%; 11/18 tumors), the endometrioid carcinomas (60.0%; 18/30
tumors) and the clear cell carcinomas with only 18.2% (2/11
tumors).
The samples were run for two parallel PCRs, which
amplified exons 1–3 and exons 1–5 of HMGA2, respectively.
Out of the 161 samples analyzed, 109 showed expression of
full-length HMGA2, whereas 41 tumors showed no HMGA2
expression. The remaining 11 tumors showed expression of a
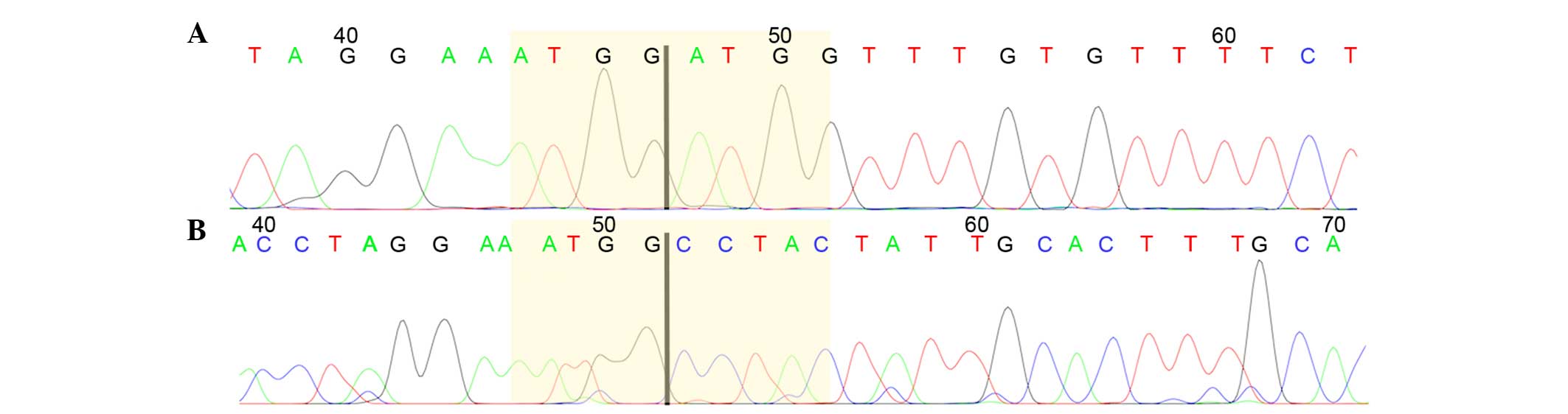
truncated HMGA2, i.e., exons 1–3 (Table II). In these cases, 3′RACE PCR was
performed in search of possible fusion transcripts. In 9 cases, the
HMGA2 transcript could be identified by Sanger sequencing,
whereas in cases 2 and 10, sequencing was not informative. In 4 out
of the 9 tumors showing a truncated form of HMGA2 (cases 1,
3, 4 and 5), an ectopic sequence was found fused with HMGA2.
This sequence has been previously found in human lipomas (accession
number U29117). A single tumor (case 6) displayed a truncated
transcript of HMGA2 with a sequence similar to the
previously reported Homo sapiens cDNA FLJ18441 (accession
number AK311399). Another 4 serous high-grade tumors (cases 7, 8, 9
and 11) showed a truncated form of HGMA2 not reported
previously. 3′RACE results for these cases were validated through
an additional PCR using the primers obtained by the sequence of the
3′RACE products. This was informative for 3 out of 4 cases, but
gave no informative result for case 9. The transcripts contained
the normal mRNA sequence until exon 3, with different regions of
the third intron varying in length from case to case (Fig. 1).
TERT
All 185 cases from which DNA was available were
analyzed for mutations in the promoter region of TERT. The
most frequent mutations, i.e., C228T and C250T, were focused on
since they have been observed in a large number of tumors of
different types (10). Only 1
borderline tumor and 3 fibromas showed the C228T mutation. All
other cases showed no mutation.
IDH1 and IDH2 mutations
A total of 184 samples were analyzed for mutations
in IDH1 and IDH2. The following mutation sites were
investigated: IDH1R100, IDH1R109, and IDH1R132 of IDH1, and
IDH2R140, IDH2R149 and IDH2R172 of IDH2. All analyses gave
informative results. No mutations were found in these tumors. In 8
tumors, the single nucleotide polymorphism (SNP) IDH1G105 was
identified.
MGMT
The MGMT promoter methylation status of the
185 samples from which DNA was extracted was assessed using
MSP-qPCR. All analyses gave informative results. Only 2 borderline
tumors were found to have MGMT promoter methylation.
Discussion
The high-mobility group AT-hook proteins, HMGA1 and
HMGA2, are non-histone proteins that are involved in a range of
nuclear processes from chromatin dynamics to gene regulation.
HMGA gene family expression is observed during embryonic
development (11), while expression
is largely absent in adult normal tissues (11). However, high HMGA2 expression
levels have been found in a variety of benign and malignant tumors
(12,13). HMGA2 is involved in a number of
different processes, from cellular proliferation to
epithelial-mesenchymal transition (14). Recent studies have highlighted a
pivotal role for HMGA2 in tumor metastasis (15). HMGA2 expression, in its full
and/or truncated length, has not been investigated extensively in
ovarian tumors, although a study by Hetland et al (16) showed the expression of HGMA2 by
immunohistochemistry in primary solid tumors (96%), metastases
(90%) and effusions (94%) of serous carcinomas. The present results
showed that HMGA2 is expressed in a number of ovarian tumors
(74.5%; 120/161 tumors). High-level expression was noted throughout
the entire spectrum of malignancy, i.e., from sex-cord stromal
neoplasms to borderline tumors to aggressive carcinomas. The only
quantitative exception was the clear cell carcinomas, among which
only 18.2% showed HMGA2 expression (2/11 tumors). Whether
neoplasms of this subtype really express HMGA2 more rarely
is a conclusion that must await examination of more tumors.
A truncated gene was found in 11 ovarian tumors of
the present cohort. HMGA2 has previously been found
disrupted, due to rearrangement of chromosomal band 12q15, in
different benign connective tissue tumors, including lipomas
(17), pleomorphic salivary gland
adenomas (18), uterine leiomyomas
(19) and lung hamartomas (20). The alterations involve exon 3 and
cause the deletion of downstream regions, resulting in a truncated
transcript that is able to evade miRNA-dependent gene silencing.
Another alteration involves the chromosomal rearrangement of
12q13-15 to form a fusion gene. In the present study, karyotypic
information was available for 9 out of the 11 tumors with a
truncated HMGA2 (Table II).
The karyotype was normal in 6 cases, whereas case 3 showed a
del(1)(q21) as a sole rearrangement,
case 8 exhibited a karyotype described as
47~49,XX,+8,+9[2]/49,idem,+5,-6,+7[4]/54,idem,+3,+5,+6,+7,+14,+17,+19[5],
and the karyotype of case 9 was interpreted as 65~68,XX,-X,+1,+2,
del(3)(p13),+4, add(4) (p12)x2,add(5)(p15),-6,+7,add(7) (p15),-8,-9,-12,-13,-14,-15,-17, −18,
der(19) add (19)(p13) add(19)(q13), +20,+20,-21,-22,+2mar[9]. Notably,
none of these cases showed a structural rearrangement involving
12q15 despite the fact that a truncated form of the HMGA2
gene was found in all of them. Possibly, this gene-level change may
be due to a small deletion not visible at the chromosomal
level.
The HMGA2 truncated transcripts were further
characterized by 3′RACE-PCR, searching for possible fusion
partners. However, it emerged that in 9 tumors (2 cases were not
informative), the HMGA2 transcript was disrupted in the
third exon. Notably, in 4 of them (cases 1, 3, 4 and 5), a sequence
previously found in human lipomas was identified. Moreover, a novel
truncated transcript was detected for HMGA2 in 4 high-grade
serous carcinomas. The sequence of these transcripts contains the
normal mRNA sequence until exon 3, followed by different regions of
the third intron, which vary in length from case to case. It
therefore appears that HMGA2 breakage leads to a truncated
transcript that possesses exonic and intronic sequences. He et
al (21) suggested that
HMGA2 transcript shortening in serous ovarian cancer is the
result of alternative polyadenylation that leads to a novel 3′UTR
formation. In the present study, the finding of HMGA2
expression in different ovarian tumors, even the less aggressive
types such as fibroma, thecofibroma and borderline tumors,
highlights once more the importance of HMGA2 for the
development of ovarian tumors.
TERT encodes the telomerase reverse
transcriptase. It is well known that the gene is involved in
cancer, and numerous studies have shown that mutations in the
promoter region of the gene can increase telomerase expression
(22–24). The present study focused on the most
frequently occurring TERT mutations, i.e., C228T and C250T.
These mutations introduce a novel binding site (TTCCGG) for members
of the E-twenty-six/ternary complex factor transcription factor
family (10). The C228T mutation was
found in 4 out of the 184 tumors analyzed, 3 of which were
fibromas, whereas the fourth was a borderline tumor. The low
percentage of TERT mutations found in the various ovarian
tumor samples analyzed leads us to hypothesize that it is neither a
primary event nor even an important step in the majority of types
of ovarian tumors. Notably, TERT C228T appears to be
recurrent in ovarian fibromas, but further studies of a larger
cohort are necessary to more reliably evaluate the frequency of
this mutation. The present study did not find any TERT
mutations in the clear cell samples, however, Huang et al
(25) found the mutation in 16% of
tumors (9/56 tumors) of this carcinoma subtype. The discrepancy
between the present data and previous data may be due to the low
number of clear cell carcinomas analyzed in the present cohort
(n=12).
The IDH1 and IDH2 genes encode two
types of isocitrate dehydrogenase. Mutations in either of the genes
can result in an enzyme that produces 2-hydroxyglutarate. This
metabolite is an inhibitor of α-ketoglutarate-dependent oxygenases,
which can cause genome-wide methylations that exhibit an effect on
gene expression when impaired. IDH1 and/or IDH2
mutations have been found in gliomas (26) and hematological malignancies (27). SNP IDH1G105, which is an adverse
prognostic factor in cytogenetically normal acute myeloid leukemia
(28), was identified in 8 out of the
184 tumors analyzed in the present series. This indicates that
IDH1 and IDH2 are only rarely involved in ovarian
tumorigenesis.
MGMT encodes O6-methylguanine DNA
methyltransferase, a DNA repair enzyme that removes alkyl adducts
from the O6-position of guanine. Expression of
MGMT can result in resistance to alkylating cytostatics.
MGMT promoter methylation increases the sensitivity of cells
to alkylating drugs, as has been shown in a number of cancer types,
particularly gliomas (29). Due to
the efficacy of MGMT promoter methylation status as a
prognostic and predictive tumor marker, this assessment has become
one of the most commonly requested analyses for gliomas (30). The present study found this gene to be
altered in only 2 borderline tumors out of 184 analyzed tumors of
different types, indicating that MGMT promoter methylation
is not a common event in ovarian tumorigenesis. In conclusion, the
present study contributes to elucidating the genetic features of
the HMGA2 gene in ovarian neoplasms in that it has been
found expressed in benign and malignant tumors. Furthermore, a
novel truncated form of HMGA2 has been identified.
Acknowledgements
This study was supported by grants from the
Norwegian Radium Hospital Foundation, the Norwegian Cancer Society,
the Inger and John Fredriksen Foundation for Ovarian Cancer
Research, and the Research Council of Norway through its Centers of
Excellence funding scheme, project number 179571.
References
|
1
|
Tavassoli FA and Devilee P: Pathology and
Genetics. Tumours of the Breast and Female Genital organs. IARC
Press. (Lyon). 2003.
|
|
2
|
Prat J: Ovarian carcinomas: Five distinct
diseases with different origins, genetic alterations, and
clinicopathological features. Virchows Arch. 460:237–249. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Micci F, Haugom L, Ahlquist T, Andersen
HK, Abeler VM, Davidson B, Trope CG, Lothe RA and Heim S: Genomic
aberrations in borderline ovarian tumors. J Transl Med. 8:212010.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Buy JN and Ghossain M: Sex cord-stromal
tumors. Gynecological Imaging: A Reference Guide to Diagnosis.
Springer-Verlag. (Berlin, Heidelberg). 329–375. 2013. View Article : Google Scholar
|
|
5
|
Micci F, Haugom L, Abeler VM, Tropé CG,
Danielsen HE and Heim S: Consistent numerical chromosome
aberrations in thecofibromas of the ovary. Virchows Arch.
452:269–276. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Micci F, Haugom L, Abeler VM, Davidson B,
Tropé CG and Heim S: Genomic profile of ovarian carcinomas. BMC
Cancer. 14:3152014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Agostini A, Panagopoulos I, Andersen HK,
Johannesen LE, Davidson B, Tropé CG, Heim S and Micci F: HMGA2
expression pattern and TERT mutations in tumors of the vulva. Oncol
Rep. 33:2675–2680. 2015.PubMed/NCBI
|
|
8
|
Korbie DJ and Mattick JS: Touchdown PCR
for increased specificity and sensitivity in PCR amplification. Nat
Protoc. 3:1452–1456. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Smith E, Jones ME and Drew PA:
Quantitation of DNA methylation by melt curve analysis. BMC Cancer.
9:1–12. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Heidenreich B, Rachakonda PS, Hemminki K
and Kumar R: TERT promoter mutations in cancer development. Curr
Opin Genet Dev. 24:30–37. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chiappetta G, Avantaggiato V, Visconti R,
Fedele M, Battista S, Trapasso F, Merciai BM, Fidanza V, Giancotti
V, Santoro M, et al: High level expression of the HMGI (Y) gene
during embryonic development. Oncogene. 13:2439–2446.
1996.PubMed/NCBI
|
|
12
|
Rogalla P, Drechsler K, Frey G, Hennig Y,
Helmke B, Bonk U and Bullerdiek J: HMGI-C expression patterns in
human tissues. Implications for the genesis of frequent mesenchymal
tumors. Am J Pathol. 149:775–779. 1996.PubMed/NCBI
|
|
13
|
Pallante P, Sepe R, Puca F and Fusco A:
High mobility group A (HMGA) proteins as tumor markers. Front Med
(Lausanne). 2:152015.PubMed/NCBI
|
|
14
|
Wu J and Wei JJ: HMGA2 and high-grade
serous ovarian carcinoma. J Mol Med (Berl). 91:1155–1165. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Morishita A, Zaidi MR, Mitoro A,
Sankarasharma D, Szabolcs M, Okada Y, D'Armiento J and Chada K:
HMGA2 is a driver of tumor metastasis. Cancer Res. 73:4289–4299.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hetland TE, Holth A, Kærn J, Flørenes VA,
Tropé CG and Davidson B: HMGA2 protein expression in ovarian serous
carcinoma effusions, primary tumors, and solid metastases. Virchows
Arch. 460:505–513. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Schoenmakers EF, Wanschura S, Mols R,
Bullerdiek J, Van den Berghe H and Van de Ven WJ: Recurrent
rearrangements in the high mobility group protein gene, HMGI-C, in
benign mesenchymal tumours. Nat Genet. 10:436–444. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Geurts JM, Schoenmakers EF and Van de Ven
WJ: Molecular characterization of a complex chromosomal
rearrangement in a pleomorphic salivary gland adenoma involving the
3′-UTR of HMGIC. Cancer Genet Cytogenet. 95:198–205. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mine N, Kurose K, Nagai H, Doi D, Ota Y,
Yoneyama K, Konishi H, Araki T and Emi M: Gene fusion involving
HMGIC is a frequent aberration in uterine leiomyomas. J Hum Genet.
46:408–412. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kazmierczak B, Meyer-Bolte K, Tran KH,
Wöckel W, Breightman I, Rosigkeit J, Bartnitzke S and Bullerdiek J:
A high frequency of tumors with rearrangements of genes of the
HMGI(Y) family in a series of 191 pulmonary chondroid hamartomas.
Genes Chromosomes Cancer. 26:125–133. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
He X, Yang J, Zhang Q, Cui H and Zhang Y:
Shortening of the 3′ untranslated region: An important mechanism
leading to overexpression of HMGA2 in serous ovarian cancer. Chin
Med J (Engl). 127:494–499. 2014.PubMed/NCBI
|
|
22
|
Killela PJ, Reitman ZJ, Jiao Y, Bettegowda
C, Agrawal N, Diaz LA Jr, Friedman AH, Friedman H, Gallia GL,
Giovanella BC, et al: TERT promoter mutations occur frequently in
gliomas and a subset of tumors derived from cells with low rates of
self-renewal. Proc Natl Acad Sci USA. 110:6021–6026. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Huang DS, Wang Z, He XJ, Diplas BH, Yang
R, Killela PJ, Meng Q, Ye ZY, Wang W, Jiang XT, et al: Recurrent
TERT promoter mutations identified in a large-scale study of
multiple tumour types are associated with increased TERT expression
and telomerase activation. Eur J Cancer. 51:969–976. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Vinagre J, Almeida A, Pópulo H, Batista R,
Lyra J, Pinto V, Coelho R, Celestino R, Prazeres H, Lima L, et al:
Frequency of TERT promoter mutations in human cancers. Nat Commun.
4:21852013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Huang HN, Chiang YC, Cheng WF, Chen CA,
Lin MC and Kuo KT: Molecular alterations in endometrial and ovarian
clear cell carcinomas: Clinical impacts of telomerase reverse
transcriptase promoter mutation. Mod Pathol. 28:303–311. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Havik AB, Lind GE, Honne H, Meling TR,
Scheie D, Hall KS, van den Berg E, Mertens F, Picci P, Lothe RA, et
al: Sequencing IDH1/2 glioma mutation hotspots in gliomas and
malignant peripheral nerve sheath tumors. Neuro Oncol. 16:320–322.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Patel KP, Barkoh BA, Chen Z, Ma D, Reddy
N, Medeiros LJ and Luthra R: Diagnostic testing for IDH1 and IDH2
variants in acute myeloid leukemia an algorithmic approach using
high-resolution melting curve analysis. J Mol Diagn. 13:678–686.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wagner K, Damm F, Göhring G, Görlich K,
Heuser M, Schäfer I, Ottmann O, Lübbert M, Heit W, Kanz L, et al:
Impact of IDH1 R132 mutations and an IDH1 single nucleotide
polymorphism in cytogenetically normal acute myeloid leukemia: SNP
rs11554137 is an adverse prognostic factor. J Clin Oncol.
28:2356–2364. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Margison GP and Santibáñez-Koref MF:
O6-alkylguanine-DNA alkyltransferase: Role in carcinogenesis and
chemotherapy. Bioessays. 24:255–266. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Håvik AB, Brandal P, Honne H, Dahlback HS,
Scheie D, Hektoen M, Meling TR, Helseth E, Heim S, Lothe RA and
Lind GE: MGMT promoter methylation in gliomas-assessment by
pyrosequencing and quantitative methylation-specific PCR. J Transl
Med. 10:362012. View Article : Google Scholar : PubMed/NCBI
|