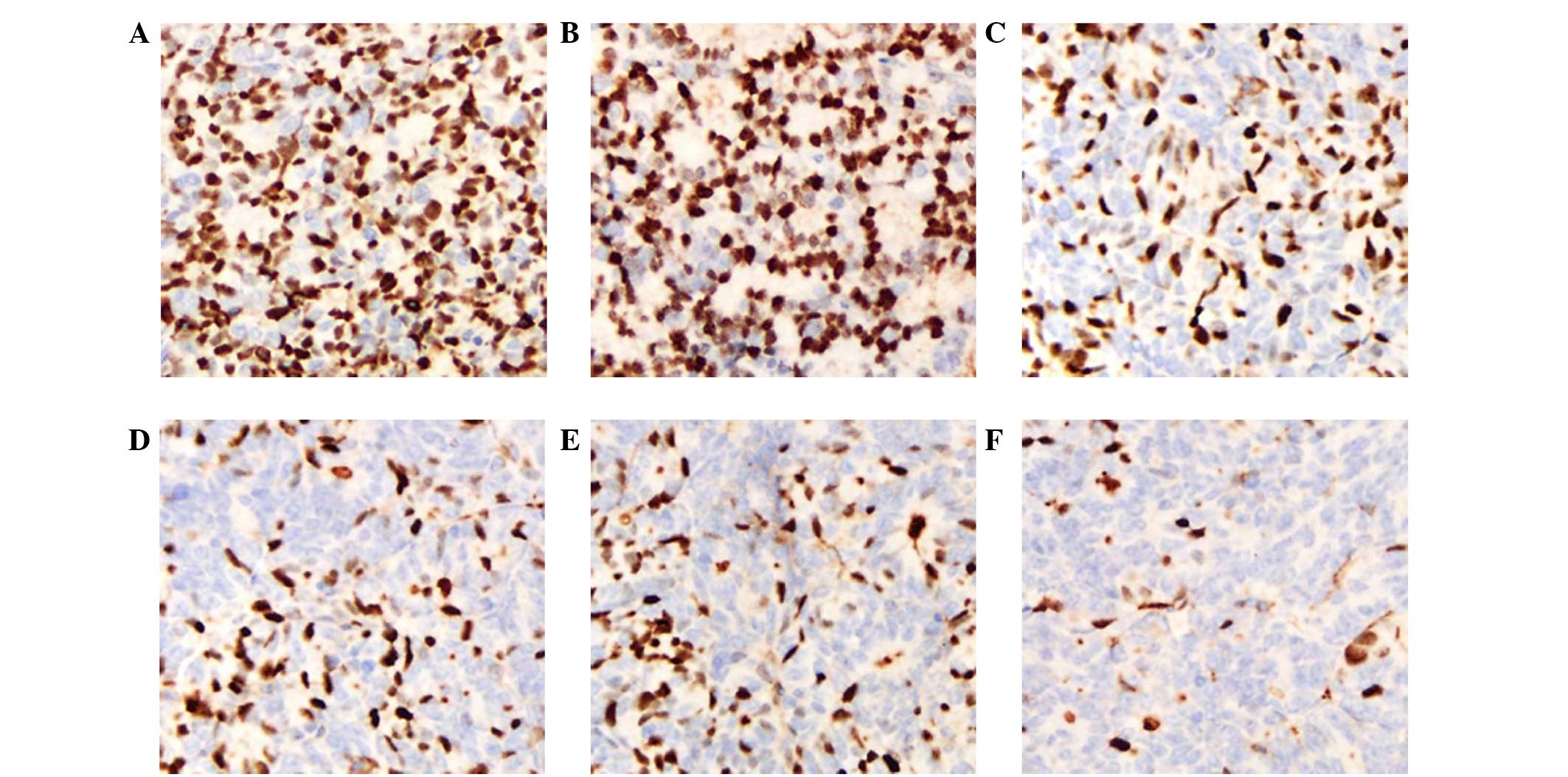
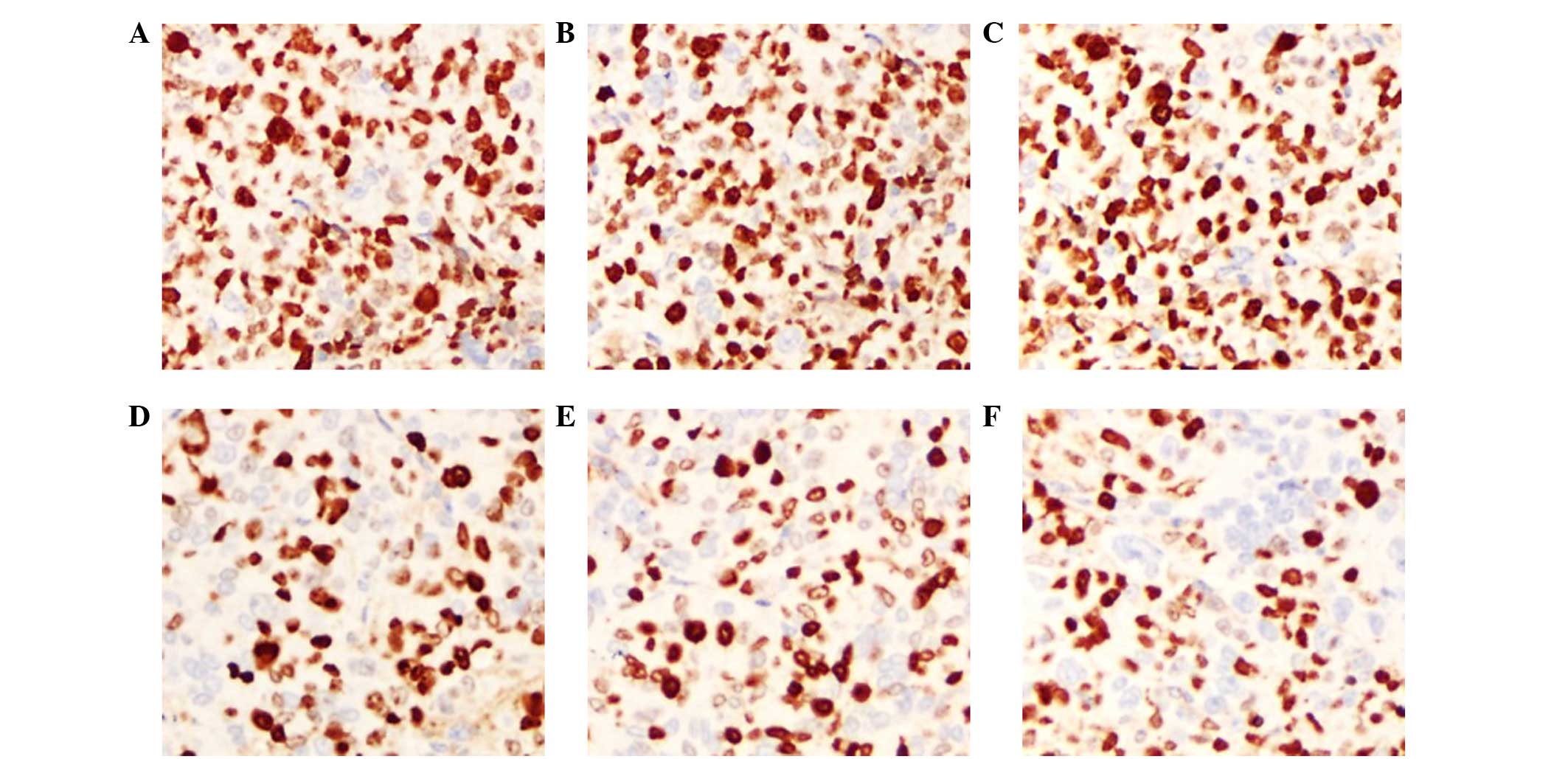
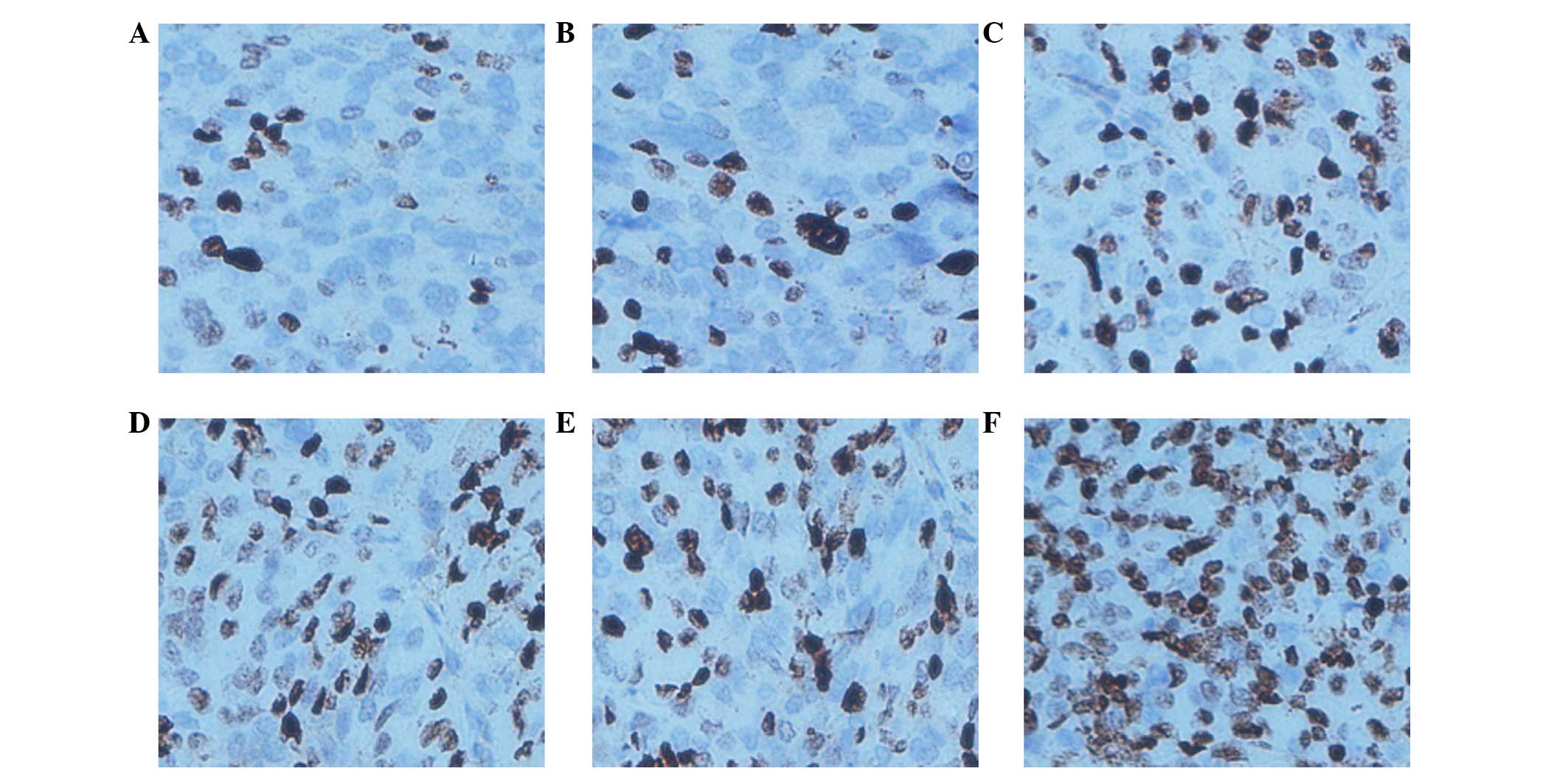
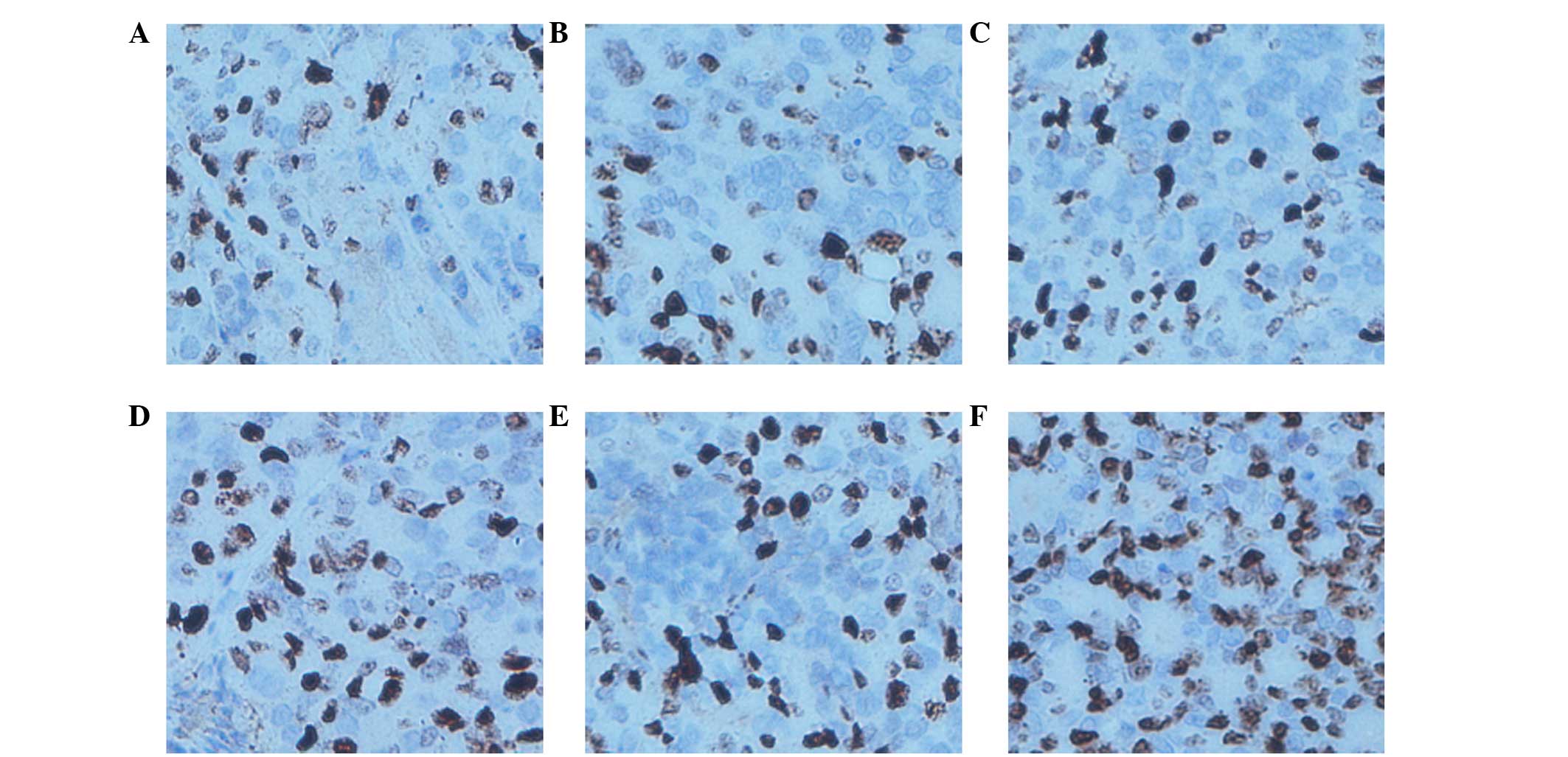
|
1
|
Chen W, Zheng R, Zhang S, Zhao P, Zeng H
and Zou X: Report of cancer incidence and mortality in China, 2010.
Ann Transl Med. 2:612014.PubMed/NCBI
|
|
2
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Noguchi T, Ritter G and Nishikawa H:
Antibody-based therapy in colorectal cancer. Immunotherapy.
5:533–545. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kuppen PJ, Gorter A, Hagenaars M, Jonges
LE, Giezeman-Smits KM, Nagelkerke JF, Fleuren G and van de Velde
CJ: Role of NK cells in adoptive immunotherapy of metastatic
colorectal cancer in a syngeneic rat model. Immunol Rev.
184:236–243. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Barkholt L, Alici E, Conrad R, Sutlu T,
Gilljam M, Stellan B, Christensson B, Guven H, Björkström NK,
Söderdahl G, et al: Safety analysis of ex vivo-expanded NK
and NK-like T cells administered to cancer patients: A phase I
clinical study. Immunotherapy. 1:753–764. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
de Castro-Carpeño J, Belda-Iniesta C,
Casado Sáenz E, Hernández Agudo E, Feliu Batlle J and González
Barón M: EGFR and colon cancer: A clinical view. Clin Transl Oncol.
10:6–13. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Huether A, Höpfner M, Baradari V, Schuppan
D and Scherübl H: EGFR blockade by cetuximab alone or as
combination therapy for growth control of hepatocellular cancer.
Biochem Pharmacol. 70:1568–1578. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Troiani T, Zappavigna S, Martinelli E,
Addeo SR, Stiuso P, Ciardiello F and Caraglia M: Optimizing
treatment of metastatic colorectal cancer patients with anti-EGFR
antibodies: Overcoming the mechanisms of cancer cell resistance.
Expert Opin Biol Ther. 13:241–255. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kurai J, Chikumi H, Hashimoto K, Yamaguchi
K, Yamasaki A, Sako T, Touge H, Makino H, Takata M, Miyata M, et
al: Antibody-dependent cellular cytotoxicity mediated by cetuximab
against lung cancer cell lines. Clin Cancer Res. 13:1552–1561.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sotelo Lezama MJ, Sastre Valera J and
Díaz-Rubio García E: Impact of cetuximab in current treatment of
metastatic colorectal cancer. Expert Opin Biol Ther. 14:387–399.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bachanova V and Miller JS: NK cells in
therapy of cancer. Crit Rev Oncog. 19:133–141. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pahl JH, Ruslan SE, Buddingh EP, Santos
SJ, Szuhai K, Serra M, Gelderblom H, Hogendoorn PC, Egeler RM,
Schilham MW and Lankester AC: Anti-EGFR antibody cetuximab enhances
the cytolytic activity of natural killer cells toward osteosarcoma.
Clin Cancer Res. 18:432–441. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kohrt HE, Colevas AD, Houot R, Weiskopf K,
Goldstein MJ, Lund P, Mueller A, Sagiv-Barfi I, Marabelle A, Lira
R, et al: Targeting CD137 enhances the efficacy of cetuximab. J
Clin Invest. 124:2668–2682. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cheng M, Chen Y, Xiao W, Sun R and Tian Z:
NK cell-based immunotherapy for malignant diseases. Cell Mol
Immunol. 10:230–252. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shiokawa M, Takahashi T, Murakami A, Kita
S, Ito M, Sugamura K and Ishii N: In vivo assay of human
NK-dependent ADCC using NOD/SCID/gamma(null) (NOG) mice. Biochem
Biophys Res Commun. 399:733–737. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lee SC, Srivastava RM, López-Albaitero A,
Ferrone S and Ferris RL: Natural killer (NK): Dendritic cell (DC)
cross talk induced by therapeutic monoclonal antibody triggers
tumor antigen-specific T cell immunity. Immunol Res. 50:248–254.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ogbomo H, Cinatl J Jr, Mody CH and Forsyth
PA: Immunotherapy in gliomas: Limitations and potential of natural
killer (NK) cell therapy. Trends Mol Med. 17:433–441. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ottaiano A, Scala S and Iaffaioli VR:
Cetuximab-dependent ADCC in cancer: Dream or reality? Cancer
Immunol Immunother. 59:1607–1608. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Qu YH and Li Y: Progress of study on
antitumor effects of antibody dependent cell mediated
cytotoxicity-review. Zhongguo Shi Yan Xue Ye Xue Za Zhi.
18:1370–1375. 2010.(In Chinese). PubMed/NCBI
|
|
20
|
Srivastava RM, Lee SC, Andrade Filho PA,
Lord CA, Jie HB, Davidson HC, López-Albaitero A, Gibson SP, Gooding
WE, Ferrone S and Ferris RL: Cetuximab-activated natural killer and
dendritic cells collaborate to trigger tumor antigen–specific
T-cell immunity in head and neck cancer patients. Clin Cancer Res.
19:1858–1872. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Balasa B, Yun R, Belmar NA, Fox M, Chao
DT, Robbins MD, Starling GC and Rice AG: Elotuzumab enhances
natural killer cell activation and myeloma cell killing through
interleukin-2 and TNF-α pathways. Cancer Immunol Immunother.
64:61–73. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Roda JM, Joshi T, Butchar JP, McAlees JW,
Lehman A, Tridandapani S and Carson WE III: The activation of
natural killer cell effector functions by cetuximab-coated,
epidermal growth factor receptor-positive tumor cells is enhanced
by cytokines. Clin Cancer Res. 13:6419–6428. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
JD H, JW Y, RM C, MG Y and QH Z: The ADCC
function of NK cells combined with cetuximab on colon cancer cell.
Fu Jian Yi Ke Da Xue Xue Bao. 46:20–27. 2012.(In Chinese).
|
|
24
|
Yang X, Zhang X, Mortenson ED,
Radkevich-Brown O, Wang Y and Fu YX: Cetuximab-mediated tumor
regression depends on innate and adaptive immune responses. Mol
Ther. 21:91–100. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mandal A and Viswanathan C: Natural killer
cells: In health and disease. Hematol Oncol Stem Cell Ther.
8:47–55. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Vincenzi B, Zoccoli A, Pantano F, Venditti
O and Galluzzo S: Cetuximab: From bench to bedside. Curr Cancer
Drug Targets. 10:80–95. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Baselga J: Why the epidermal growth factor
receptor? The rationale for cancer therapy. Oncologist. 7(Suppl 4):
S2–S8. 2002. View Article : Google Scholar
|
|
28
|
Goldberg RM and Gill S: Recent phase III
trials of fluorouracil, irinotecan, and oxaliplatin as chemotherapy
for metastatic colorectal cancer. Cancer Chemother Pharmacol.
54(Suppl 1): S57–S64. 2004.PubMed/NCBI
|
|
29
|
Holubec L, Liska V, Matejka VM, Fiala O,
Dreslerova J, Mrazkova P, Treska V and Finek J: The role of
cetuximab in the treatment of metastatic colorectal cancer.
Anticancer Res. 32:4007–4011. 2012.PubMed/NCBI
|
|
30
|
Seo Y, Ishii Y, Ochiai H, Fukuda K,
Akimoto S, Hayashida T, Okabayashi K, Tsuruta M, Hasegawa H and
Kitagawa Y: Cetuximab-mediated ADCC activity is correlated with the
cell surface expression level of EGFR but not with the KRAS/BRAF
mutational status in colorectal cancer. Oncol Rep. 31:2115–2122.
2014.PubMed/NCBI
|
|
31
|
Maughan TS, Adams RA, Smith CG, Meade AM,
Seymour MT, Wilson RH, Idziaszczyk S, Harris R, Fisher D, Kenny SL,
et al: MRC COIN Trial Investigators: Addition of cetuximab to
oxaliplatin-based first-line combination chemotherapy for treatment
of advanced colorectal cancer: Results of the randomised phase 3
MRC COIN trial. Lancet. 377:2103–2114. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tveit KM, Guren T, Glimelius B, Pfeiffer
P, Sorbye H, Pyrhonen S, Sigurdsson F, Kure E, Ikdahl T, Skovlund
E, et al: Phase III trial of cetuximab with continuous or
intermittent fluorouracil, leucovorin, and oxaliplatin (Nordic
FLOX) versus FLOX alone in first-line treatment of metastatic
colorectal cancer: The NORDIC-VII study. J Clin Oncol.
30:1755–1762. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li S, Schmitz KR, Jeffrey PD, Wiltzius JJ,
Kussie P and Ferguson KM: Structural basis for inhibition of the
epidermal growth factor receptor by cetuximab. Cancer Cell.
7:301–311. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wild R, Fager K, Flefleh C, Kan D, Inigo
I, Castaneda S, Luo FR, Camuso A, McGlinchey K and Rose WC:
Cetuximab preclinical antitumor activity (monotherapy and
combination based) is not predicted by relative total or activated
epidermal growth factor receptor tumor expression levels. Mol
Cancer Ther. 5:104–113. 2006. View Article : Google Scholar : PubMed/NCBI
|