The homeobox (HOX) gene family is a large group of
comparable genes that function during early embryonic development
to direct the formation of various body structures (1,2). In
humans, it is estimated that the HOX gene family contains 65
pseudogenes and 235 functional genes (3). HOX genes are present in every human
chromosome, and are commonly organized in clusters. A number of
subfamilies and classes of HOX genes have been described, although
these categories are used inconsistently.
Various genes of the HOX family are involved in a
broad range of crucial developmental activities (4–7). Tumor
suppressing regions of HOX genes have been demonstrated to inhibit
cell growth in an uncontrolled manner (4). These genes commonly perform important
functions, and mutation or abnormal expression of these genes has
been associated with developmental disorders and several forms of
cancer (5–7).
In the human genome, only 2% of the sequence encodes
proteins that function in biological progression (8), with ~90% consisting of non-coding RNAs
(ncRNAs), which are described as the ‘noise’ of the genome
(9–12). Over the last few years, an increasing
number of long ncRNAs (lncRNAs) have been investigated by
microarray and sequencing technology, and it has been proposed that
they may regulate various major biological processes involved in
metabolism, differentiation and tumor development (13). ncRNAs are divided into two subgroups,
including small ncRNAs (sncRNAs) and lncRNAs. ncRNAs are defined as
sncRNAs when the RNA length is <200 nt, which includes microRNAs
(also named as miRNAs/miRs). Conversely, lncRNAs contain >200 nt
of transcript. Currently, lncRNAs are becoming more popular and
attracting more attention in scientific research (14–16). Two
important platforms have been used for analysis of lncRNAs;
advanced sequencing platforms estimated that there are ~20,000
distinct lncRNAs in humans (17,18). The
other platform is the commercial lncRNAs ChIP, consisting of
~30,000 lncRNA transcripts, and is an ideal tool for making
comparisons between tumorous and non-tumorous samples (19,20). It
has been suggested that lncRNAs have a critical role in cancer
development. However, the biological functions of a large
proportion of lncRNAs remain to be elucidated. Previous studies
reported that lncRNAs are involved in fundamental cellular
processes, including apoptosis, cell cycle and cell proliferation
(21–23), chromatin modification (24,25),
genomic reprogramming (26,27), gene imprinting (28) and RNA processing (29). It was demonstrated that tumor or
non-tumor tissues exhibited different profiles in various forms of
human cancer, and altered lncRNA expression has been functionally
linked to tumorigenesis; thus suggesting that certain lncRNAs
function as tumor promoters or suppressors (17,30,31).
Various studies have determined that certain lncRNAs may be
detected through their altered expression in different forms of
cancer (32–35). The identification of cancer-associated
lncRNAs (CA-lncRNAs), and interactions between the CA-lncRNAs and
target genes, are beneficial to supplement the current knowledge of
signaling pathways and are conducive to a better understanding of
cancer development.
One of the most important and well-studied lncRNA
and HOX transcript gene is HOTAIR, which is located on chromosome
12 within the HOXC gene cluster and is 2,158 nt long (32,36).
HOTAIR is unique in that it is overexpressed in the vast majority
of cancer types and has been recognized as an oncogenic lncRNA
(32). Recently, it was reported that
HOTAIR induced proliferation and metastasis in a variety of tumors
and functioned as a negative prognostic indicator for several forms
of cancer (32,36). In addition, it was observed that the
lncRNA interacted with polycomb repressive complex 2 (PRC2) and
inhibited multiple genes, particularly metastasis-suppressing
genes, in cancer tissues (36,37).
Subsequent studies demonstrated that HOTAIR serves as a molecular
scaffold for at least two distinct histone modification complexes,
coordinating their functions in transcription repression (25). Several transcriptome-wide association
studies have detected extensive alterations in cellular transcript
levels in response to HOTAIR inhibition, indicating that HOTAIR may
regulate hundreds of genes (33–35), thus
providing insight into the underlying mechanisms of this lncRNA in
cancer. Although transcript frequencies are informative, they do
not necessarily reflect the level of cellular protein; this is due
to the fact that protein activity is affected by an array of
post-transcriptional regulatory mechanisms and the association
between mRNA and protein levels is generally modest (34,38).
LncRNAs have been frequently investigated in recent
years and certain lncRNAs associated with HOX genes have been
detected in sequencing and ChIP platform research. It is expected
that an increasing number of HOX gene-associated lncRNAs may be
identified in the coming years, which will further clarify the
association between lncRNAs and HOX in tumorigenesis. The present
review discusses HOX genes and research regarding their association
with lncRNAs, particularly in the context of carcinogenesis.
Accumulating evidence indicates that various tumors
exhibit low levels of HOXA11 expression, which has been described
as a tumor suppressor (63). A study
by Bai et al (64) observed
that the methylation frequencies of HOXA11 in gastric cancer
tissues and adjacent cancer tissues were higher than those in
normal gastric mucosa (P<0.05). Furthermore, HOXA11 expression
was downregulated following hypermethylation of the promoter region
(64); this suggests that HOXA11 may
function as an important repressor in gastric cancer tumorigenesis,
and aberrant promoter methylation may be the primary cause behind
the loss or downregulation of HOXA11 expression, subsequently
resulting in gastric cancer carcinogenesis. The study also observed
that hypermethylation of HOXA11 was significantly associated with
lymph node metastasis and Tumor-Node-Metastasis stage in gastric
cancer, and in vitro experiments demonstrated that HOXA11
may control cell growth with its defect enhancing cell
proliferation (64).
HOXC11 expression has been observed to be
significantly higher in renal cell carcinoma (RCC) compared with
normal kidney tissues (64). It was
identified that different expression levels of HOXC11 represented
different functions, with overexpression promoting proliferation
and downregulation inhibiting proliferation (64). In addition, the study reported that
high immunohistochemical expression of HOXC11 was associated with T
stage, N stage and Ki67 level in RCC (65). The function of HOX genes remains
unknown in oral tumorigenesis (66).
However, HOXB2, HOXB7 and HOXB13 have been reported to be important
in oral tissues, and dysregulation of HOXB7 results in increased
tumor cell proliferation in oral squamous cell carcinoma tissues
(67). Previous studies have
demonstrated that HOXA1 serves an important role in tumorigenesis.
It has been reported that the disorder expression of HOXA1 in
several cancer types (68) is
involved in regulating multiple cellular processes, including
proliferation, apoptosis, etc. (69),
and is therefore closely associated with the shorter overall
survival of patients after resection. In addition, HOXA1 expression
was correlated with shorter overall survival in patients with small
cell lung cancer (70).
HOXA1 overexpression is associated with cell growth
and tumor formation in mice (60). A
previous study reported that elevated HOXA1 expression levels
inhibited differentiation, and resulted in transformation and the
development of acute myeloblastic leukemia in lethally irradiated
mice (71). Increased levels of HOXA1
in the mammary carcinoma MCF-7 cell line have also been
demonstrated to dramatically increase proliferation via the signal
transducers and activators of transcription pathway (72) and transcriptional upregulation of
B-cell lymphoma 2 (Bcl-2) (60).
HOXA1 controls the transcriptional upregulation of Bcl-2, cyclin D1
and c-Myc (73). However, various
studies have reported that HOX1A overexpression does not affect
adhesion, invasion or apoptosis (60,69,71,74).
The gene is dependent on the Meis, Prep or homeothorax protein
cofactors to activate and/or repress transcription (75). Increased HOXA1 expression results in
promoting the proliferation and decreasing the apoptosis of mammary
carcinoma cells (69), thus
contributing to the acceleration of cancer growth.
Previous studies have demonstrated that the
upregulated expression of HOXA1 promotes proliferation, whereas
downregulated HOXA1 expression decreases cell proliferation
(71–73). In addition, HOXA1-positive cells
correlate with N stage, T stage, proliferation and differentiation,
and poor survival (76).
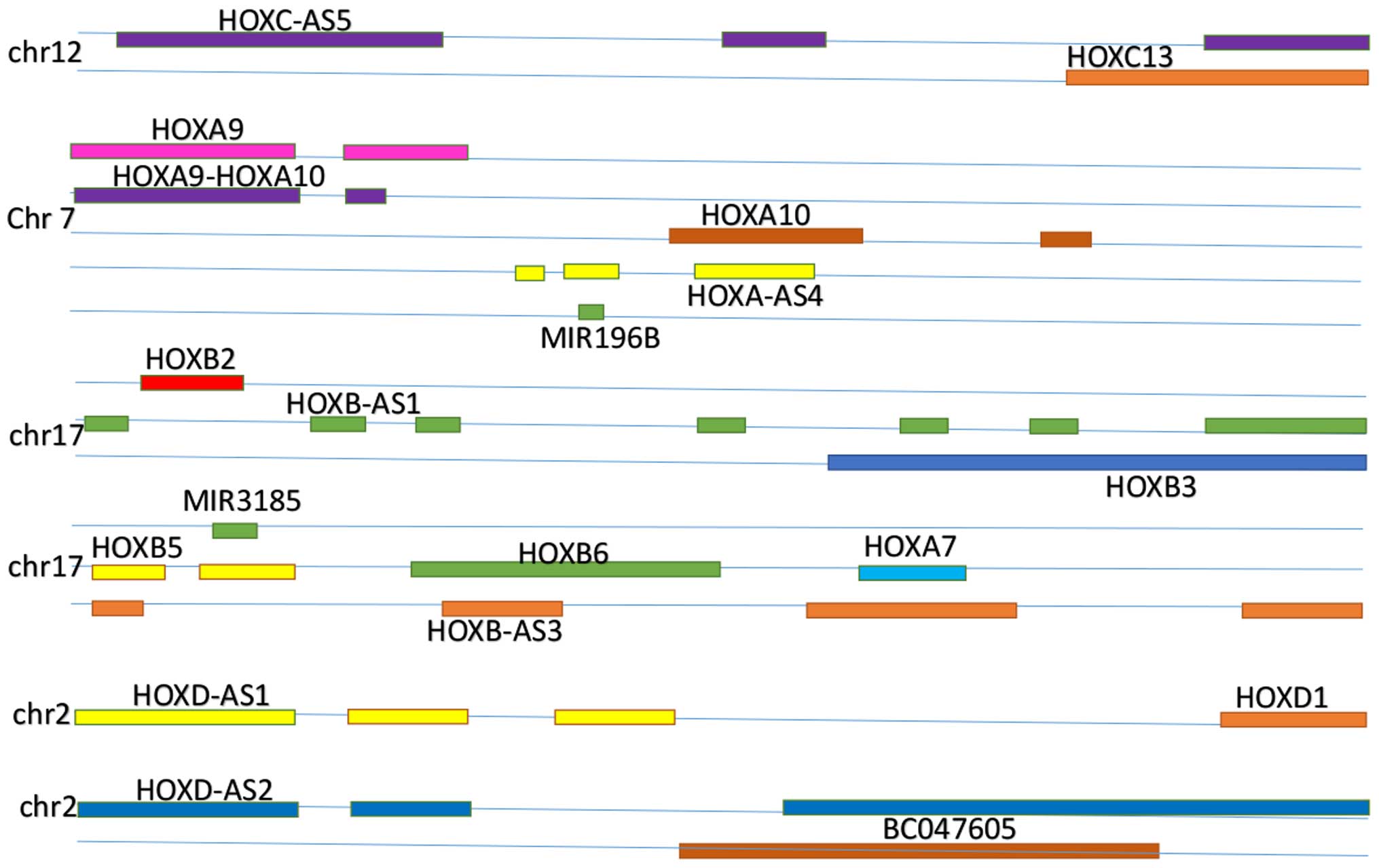
A number of non-coding RNAs are located in and
associated with HOX gene clusters, including HOTAIRM1, HOXC5,
HOXA-AS3, HOXA10, HOXB-AS3, HOXA10-HOXA9, HOXB-AS5, HOXD-AS2,
HOXD-AS1, HOXA-AS3, HOXA-AS4, pre-B-cell leukemia HOX4, HOXA distal
transcript antisense RNA (HOTTIP) and HOTAIR. The associations
between downstream and upstream HOX gene clusters with lncRNAs are
presented in Fig. 1.
DNA-binding proteins target ncRNAs, such as HOTAIR,
to specific sites in chromatin (77).
HOTAIR is a 2.2-kb lncRNA implicated in the suppression of the HOX
loci, which subsequently promotes breast cancer metastasis
(33,78). Whether lncRNAs directly or indirectly
regulate the expression of HOX gene family members has gained
increasing attention in the context of development and
tumorigenesis (24,78,79).
Furthermore, altered HOX gene expression has been observed in
certain forms of cancer (80). In a
recent study, Xu et al used a high-throughput microarray to
analyze the lncRNAs and mRNA expression profiles in lung
adenocarcinoma (81). A total of 564
dysregulated lncRNAs and 83 HOX subtypes (including 35 HOX genes
and 48 HOX ncRNAs) were screened out, which indicated that there is
a close relationship between HOX genes and lncRNAs in lung
adenocarcinoma (81). Taurine
upregulated 1 (TUG1) was able to modulate HOXB7 expression, which
may be partially responsible for TUG1-mediated proliferation
regulation, thus affecting the proliferation of non-small cell lung
cancer (NSCLC) in vitro and in vivo (82). The signals of p53/TUG1/PRC2/HOXB7 may
serve as targets for NSCLC diagnosis and therapy (82). A previous study observed that when
HOXB7 expression was inhibited, proliferation was also subsequently
inhibited (83). HOXB7 primarily
regulates cell growth through the phosphoinositide 3-kinase/Akt and
mitogen-activated protein kinase pathways (83). HOXB7 is able to promote cell
proliferation in different types of tumors through its high levels
of expression (83–85). In addition, HOXB7 has been
demonstrated to be an oncogene (57,83,86,87),
and its involvement is essential in various forms of cancer.
A number of studies have reported that the HOX
family genes serve as classic modification targets of the polycomb
complex during development, with all four gene clusters being
highly enriched in histone 3 lysine 27 trimethylation (88,89).
HOTAIR mediates epigenetic silencing by guiding PRC2 to the HOX
loci (78). Various ncRNAs are
located within the HOX gene clusters, with ~231 ncRNAs estimated to
be present in humans (24). HOTAIR, a
HOX ncRNA, is transcribed from the antisense of canonical HOXC
genes, which interact with PRC2 and silence the subsequent
transcription of the HOXD locus (78).
HOTTIP resides at the 5′ end of the HOXA locus; it
has been has been identified as one of the 231 ncRNAs associated
with the human HOX loci (24) and has
been associated with the activation of multiple 5′ HOXA genes in
vivo (34). HOTTIP was previously
considered as a negative prognostic factor in patients with liver
cancer, and its increased expression was associated with enhanced
liver cancer metastasis (38). It was
observed that inhibition of HOTTIP potentiated the antitumor
effects of gemcitabine in vitro and in vivo.
Furthermore, HOXA13-knockdown by RNA interference (siHOXA13)
demonstrated that HOTTIP promoted pancreatic ductal adenocarcinoma
(PDAC) cell proliferation, invasion and chemoresistance, at least
partially through regulating HOXA13 (91). In addition, immunohistochemical
analysis revealed that high HOXA13 expression correlated with poor
histological differentiation, lymph node metastasis and decreased
overall survival in patients with PDAC (91).
A large number of HOX genes function as
transcriptional regulators, and govern cell proliferation and
differentiation (87). Furthermore,
HOX genes have a distinctive gene network organization (four
chromosomal loci: A, B, C and D) and represent the most repeat-poor
regions within the human genome. The HOXA locus consists of a
cluster of 11 HOX genes with a graded expression pattern along the
body's appendages from proximal (close to the main body) to distal
(appendage tip) (78,88). Among the HOXA genes, HOXA13, which is
a marker of gut primordial posteriorization during development
(78), serves an essential role in
tumorigenesis of the liver and bladder, and in esophageal cancer
(33,78,90). To
examine the association between HOTTIP dysregulation and HOXA
cluster expression, HOTTIP-knockdown studies were performed, and
the expression level of the HOXA cluster (HOXA7, A9, A10, A11, and
A13) was further evaluated by polymerase chain reaction (92). Although HOXA7, A9, A10, A11 and A13
gene expression was lower in the HOTTIP-knockdown group compared
with the control, HOXA13 was the most significantly inhibited gene
within the HOXA locus following depletion of HOTTIP in pancreatic
cancer cells (92). Furthermore, it
was demonstrated that the expression levels of HOTTIP and HOXA13
were significantly positively correlated with 90 PDAC tissues and
paired adjacent non-neoplastic tissues (92). In addition, small interfering
RNA-mediated HOXA13-knockdown inhibited the proliferation, invasion
and EMT of PDAC cells, which was consistent with the functional
changes that occurred after silencing the expression of HOTTIP in
PDAC cells (92). Targeted inhibition
of HOXA13 in two pancreatic cancer cell lines led to a decrease in
the HOXA13 mRNA level and lower HOTTIP expression, which was
consistent with the results of another study (93).
Human HOX gene clusters exhibit a low density of
interspersed repeats, and cis-regulatory elements effect HOX gene
expression (94). The enhancer of the
clusters is able to regulate HOXD temporal co-linearity (95). Previous studies have demonstrated that
ncRNAs are involved in the regulation of HOX gene expression
(96), and certain microRNAs
participate in this regulation (97),
including miR-196 (98) and miR-10
(99). Due to the technical
limitations, earlier lncRNA studies have been performed in only two
species (mouse and human), such as the previous studies of HOTAIR
(33,78) and HOTAIRM1 (100). However, one study performed an
integrated analyses of HOX gene clusters between the kangaroo
family and eutherians (101).
Well-studied miRNAs, including miR-196a/b, miR-10a/b and lncRNAs
(such as HOTAIR, HOTAIRM1 and HOXA11-AS), were observed to
dysregulate gene expression and were associated with tumor
development (24,32,36).
miR-464, miR-10 and miR-414 are located in the tammar HOX clusters
and were identified to have an effect on these, for example
inhibiting lymphangiogenesis (miR-414) and promoting active tumor
cell invasion (miR-10) (102,103).
In addition, certain novel miRNAs are transcribed from elsewhere on
the tammar genome and regulate the expressions of HOXB and HOXD
clusters by specifically interacting with the mRNAs transcribed
from them (104).
A large number of studies have investigated lncRNAs
in recent years, but only a few lncRNAs associated with HOX genes
have been detected with sequencing and ChIP microarray. It is
expected that an increasing number of HOX genes and related lncRNAs
may be identified in the coming years, with further analysis
required to determine their specific functions. Further studying
the associations between lncRNAs and HOX will lead to a deeper
understanding to the mechanism of cancer development.
This study was supported by The Key Clinical
Specialty Discipline Construction Program of Fujian, The
Specialized Science and Technology Key Project of Fujian Province
(grant no. 2013YZ0002-3), The Science and Technology Infrastructure
Construction Program of Fujian Province (grant no. 2014Y2005), The
Natural Science Foundation of Fujian Province (grant no.
2015J05174), The Scientific Research Project of Health and Family
Planning Commission of Fujian Province (grant no. 2015-1-94), The
Scientific Foundation of Fuzhou Health Department (grant no's.
2014-S-139-3, 2014-S-w19 and 2013-S-wp1) and the Postdoctoral
Science Foundation of Fujian Medical University (grant no.
149318).
|
1
|
Pearson JC, Lemons D and McGinnis W:
Modulating Hox gene functions during animal body patterning. Nat
Rev Genet. 6:893–904. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lawrence PA and Morata G: Homeobox genes:
Their function in Drosophila segmentation and pattern formation.
Cell. 78:181–189. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Holland PW, Booth HA and Bruford EA:
Classification and nomenclature of all human homeobox genes. BMC
Biol. 5:472007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Duboule D: Vertebrate Hox genes and
proliferation: An alternative pathway to homeosis? Curr Opin Genet
Dev. 5:525–528. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Peverali FA, D'Esposito M, Acampora D,
Bunone G, Negri M, Faiella A, Stornaiuolo A, Pannese M, Migliaccio
E, Simeone A, et al: Expression of HOX homeogenes in human
neuroblastoma cell culture lines. Differentiation. 45:61–69. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cillo C, Barba P, Freschi G, Bucciarelli
G, Magli MC and Boncinelli E: HOX gene expression in normal and
neoplastic human kidney. Int J Cancer. 51:892–897. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cillo C, Cantile M, Faiella A and
Boncinelli E: Homeobox genes in normal and malignant cells. J Cell
Physiol. 188:161–169. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Esteller M: Non-coding RNAs in human
disease. Nat Rev Genet. 12:861–874. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lee JT: Epigenetic regulation by long
noncoding RNAs. Science. 338:1435–1439. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wapinski O and Chang HY: Long noncoding
RNAs and human disease. Trends Cell Biol. 21:354–361. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hung T and Chang HY: Long noncoding RNA in
genome regulation: Prospects and mechanisms. RNA Biol. 7:582–585.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Guttman M, Amit I, Garber M, French C, Lin
MF, Feldser D, Huarte M, Zuk O, Carey BW, Cassady JP, et al:
Chromatin signature reveals over a thousand highly conserved large
non-coding RNAs in mammals. Nature. 458:223–227. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kugel JF and Goodrich JA: Non-coding RNAs:
Key regulators of mammalian transcription. Trends Biochem Sci.
37:144–151. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Geisler S and Coller J: RNA in unexpected
places: Long non-coding RNA functions in diverse cellular contexts.
Nat Rev Mol Cell Biol. 14:699–712. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ernst C and Morton CC: Identification and
function of long non-coding RNA. Front Cell Neurosci. 7:1682013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ponting CP, Oliver PL and Reik W:
Evolution and functions of long noncoding RNAs. Cell. 136:629–641.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Moran VA, Perera RJ and Khalil AM:
Emerging functional and mechanistic paradigms of mammalian long
non-coding RNAs. Nucleic Acids Res. 40:6391–6400. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jia H, Osak M, Bogu GK, Stanton LW,
Johnson R and Lipovich L: Genome-wide computational identification
and manual annotation of human long noncoding RNA genes. RNA.
16:1478–1487. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Song H, Sun W, Ye G, Ding X, Liu Z, Zhang
S, Xia T, Xiao B, Xi Y and Guo J: Long non-coding RNA expression
profile in human gastric cancer and its clinical significances. J
Transl Med. 11:2252013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ye Y, Chen J, Zhou Y, Fu Z, Zhou Q, Wang
Y, Gao W, Zheng S, Zhao X, Chen T and Chen R: High expression of
AFAP1-AS1 is associated with poor survival and short-term
recurrence in pancreatic ductal adenocarcinoma. J Transl Med.
13:1372015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kino T, Hurt DE, Ichijo T, Nader N and
Chrousos GP: Noncoding RNA gas5 is a growth arrest- and
starvation-associated repressor of the glucocorticoid receptor. Sci
Signal. 3:ra82010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hu W, Yuan B, Flygare J and Lodish HF:
Long noncoding RNA-mediated anti-apoptotic activity in murine
erythroid terminal differentiation. Genes Dev. 25:2573–2578. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Meola N, Pizzo M, Alfano G, Surace EM and
Banfi S: The long noncoding RNA Vax2os1 controls the cell cycle
progression of photoreceptor progenitors in the mouse retina. RNA.
18:111–123. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang KC, Yang YW, Liu B, Sanyal A,
Corces-Zimmerman R, Chen Y, Lajoie BR, Protacio A, Flynn RA, Gupta
RA, et al: A long noncoding RNA maintains active chromatin to
coordinate homeotic gene expression. Nature. 472:120–124. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tsai MC, Manor O, Wan Y, Mosammaparast N,
Wang JK, Lan F, Shi Y, Segal E and Chang HY: Long noncoding RNA as
modular scaffold of histone modification complexes. Science.
329:689–693. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Loewer S, Cabili MN, Guttman M, Loh YH,
Thomas K, Park IH, Garber M, Curran M, Onder T, Agarwal S, et al:
Large intergenic non-coding RNA-RoR modulates reprogramming of
human induced pluripotent stem cells. Nat Genet. 42:1113–1117.
2010. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Guttman M, Donaghey J, Carey BW, Garber M,
Grenier JK, Munson G, Young G, Lucas AB, Ach R, Bruhn L, et al:
lincRNAs act in the circuitry controlling pluripotency and
differentiation. Nature. 477:295–300. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gregg C, Zhang J, Weissbourd B, Luo S,
Schroth GP, Haig D and Dulac C: High-resolution analysis of
parent-of-origin allelic expression in the mouse brain. Science.
329:643–648. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tripathi V, Ellis JD, Shen Z, Song DY, Pan
Q, Watt AT, Freier SM, Bennett CF, Sharma A, Bubulya PA, et al: The
nuclear-retained noncoding RNA MALAT1 regulates alternative
splicing by modulating SR splicing factor phosphorylation. Mol
Cell. 39:925–938. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Spizzo R, Almeida MI, Colombatti A and
Calin GA: Long non-coding RNAs and cancer: A new frontier of
translational research? Oncogene. 31:4577–4587. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gibb EA, Vucic EA, Enfield KS, Stewart GL,
Lonergan KM, Kennett JY, Kennett JY, Becker-Santos DD, MacAulay CE,
Lam S, et al: Human cancer long non-coding RNA transcriptomes. PloS
One. 6:e259152011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang J, Zhang P, Wang L, Piao HL and Ma
L: Long non-coding RNA HOTAIR in carcinogenesis and metastasis.
Acta Biochim Biophys Sin (Shanghai). 46:1–5. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gupta RA, Shah N, Wang KC, Kim J, Horlings
HM, Wong DJ, Tsai MC, Hung T, Argani P, Rinn JL, et al: Long
non-coding RNA HOTAIR reprograms chromatin state to promote cancer
metastasis. Nature. 464:1071–1076. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Schwanhäusser B, Busse D, Li N, Dittmar G,
Schuchhardt J, Wolf J, Chen W and Selbach M: Global quantification
of mammalian gene expression control. Nature. 473:337–342. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kim K, Jutooru I, Chadalapaka G, Johnson
G, Frank J, Burghardt R, Kim S and Safe S: HOTAIR is a negative
prognostic factor and exhibits pro-oncogenic activity in pancreatic
cancer. Oncogene. 32:1616–1625. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wan Y and Chang HY: HOTAIR: Flight of
noncoding RNAs in cancer metastasis. Cell Cycle. 9:3391–3392. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li L, Liu B, Wapinski OL, Tsai MC, Qu K,
Zhang J, Carlson JC, Lin M, Fang F, Gupta RA, et al: Targeted
disruption of Hotair leads to homeotic transformation and gene
derepression. Cell Rep. 5:3–12. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wu L, Candille SI, Choi Y, Xie D, Jiang L,
Li-Pook-Than J, Tang H and Snyder M: Variation and genetic control
of protein abundance in humans. Nature. 499:79–82. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sorge S, Ha N, Polychronidou M, Friedrich
J, Bezdan D, Kaspar P, Schaefer MH, Ossowski S, Henz SR, Mundorf J,
et al: The cis-regulatory code of Hox function in
Drosophila. EMBO J. 31:3323–3333. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ahanger SH, Srinivasan A, Vasanthi D,
Shouche YS and Mishra RK: Conserved boundary elements from the Hox
complex of mosquito, Anopheles gambiae. Nucleic Acids Res.
41:804–816. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Javed S and Langley SE: Importance of HOX
genes in normal prostate gland formation, prostate cancer
development and its early detection. BJU Int. 113:535–540. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
McGrath SE, Michael A, Pandha H and Morgan
R: Engrailed homeobox transcription factors as potential markers
and targets in cancer. FEBS Lett. 587:549–554. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Makiyama K, Hamada J, Takada M, Murakawa
K, Takahashi Y, Tada M, Tamoto E, Shindo G, Matsunaga A, Teramoto
K, et al: Aberrant expression of HOX genes in human invasive breast
carcinoma. Oncol Rep. 13:673–679. 2005.PubMed/NCBI
|
|
44
|
Kanai M, Hamada J, Takada M, Asano T,
Murakawa K, Takahashi Y, Murai T, Tada M, Miyamoto M, Kondo S and
Moriuchi T: Aberrant expressions of HOX genes in colorectal and
hepatocellular carcinomas. Oncol Rep. 23:843–851. 2010.PubMed/NCBI
|
|
45
|
Bodey B, Bodey B Jr, Gröger AM, Siegel SE
and Kaiser HE: Immunocytochemical detection of homeobox B3, B4, and
C6 gene product expression in lung carcinomas. Anticancer Res.
20:2711–2716. 2000.PubMed/NCBI
|
|
46
|
Cheng W, Liu J, Yoshida H, Rosen D and
Naora H: Lineage infidelity of epithelial ovarian cancers is
controlled by HOX genes that specify regional identity in the
reproductive tract. Nat Med. 11:531–537. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Yamatoji M, Kasamatsu A, Yamano Y, Sakuma
K, Ogoshi K, Iyoda M, Shinozuka K, Ogawara K, Takiguchi Y, Shiiba
M, et al: State of homeobox A10 expression as a putative prognostic
marker for oral squamous cell carcinoma. Oncol Rep. 23:61–67.
2010.PubMed/NCBI
|
|
48
|
Ota T, Klausen C, Salamanca MC, Woo HL,
Leung PC and Auersperg N: Expression and function of HOXA genes in
normal and neoplastic ovarian epithelial cells. Differentiation.
77:162–171. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Plowright L, Harrington KJ, Pandha HS and
Morgan R: HOX transcription factors are potential therapeutic
targets in non-small-cell lung cancer (targeting HOX genes in lung
cancer). Br J Cancer. 100:470–475. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Waltregny D, Alami Y, Clausse N, de Leval
J and Castronovo V: Overexpression of the homeobox gene HOXC8 in
human prostate cancer correlates with loss of tumor
differentiation. Prostate. 50:162–169. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Hur H, Lee JY, Yun HJ, Park BW and Kim MH:
Analysis of HOX gene expression patterns in human breast cancer.
Mol Biotechnol. 56:64–71. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Svingen T and Tonissen KF: Altered HOX
gene expression in human skin and breast cancer cells. Cancer Biol
Ther. 2:518–523. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Cantile M, Pettinato G, Procino A,
Feliciello I, Cindolo L and Cillo C: In vivo expression of the
whole HOX gene network in human breast cancer. Eur J Cancer.
39:257–264. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Raman V, Martensen SA, Reisman D, Evron E,
Odenwald WF, Jaffee E, Marks J and Sukumar S: Compromised HOXA5
function can limit p53 expression in human breast tumours. Nature.
405:974–978. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Gilbert PM, Mouw JK, Unger MA, Lakins JN,
Gbegnon MK, Clemmer VB, Benezra M, Licht JD, Boudreau NJ, Tsai KK,
et al: HOXA9 regulates BRCA1 expression to modulate human breast
tumor phenotype. J Clin Invest. 120:1535–1550. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Caré A, Silvani A, Meccia E, Mattia G,
Peschle C and Colombo MP: Transduction of the SkBr3 breast
carcinoma cell line with the HOXB7 gene induces bFGF expression,
increases cell proliferation and reduces growth factor dependence.
Oncogene. 16:3285–3289. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Jin K, Kong X, Shah T, Penet MF, Wildes F,
Sgroi DC, Ma XJ, Huang Y, Kallioniemi A, Landberg G, et al: The
HOXB7 protein renders breast cancer cells resistant to tamoxifen
through activation of the EGFR pathway. Proc Natl Acad Sci USA.
109:2736–2741. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Care A, Felicetti F, Meccia E, Bottero L,
Parenza M, Stoppacciaro A, Peschle C and Colombo MP: HOXB7: A key
factor for tumor-associated angiogenic switch. Cancer Res.
61:6532–6559. 2001.PubMed/NCBI
|
|
59
|
Hayashida T, Takahashi F, Chiba N,
Brachtel E, Takahashi M, Godin-Heymann N, Gross KW, Vivanco Md,
Wijendran V, Shioda T, et al: HOXB9, a gene overexpressed in breast
cancer, promotes tumorigenicity and lung metastasis. Proc Natl Acad
Sci USA. 107:1100–1105. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhang X, Zhu T, Chen Y, Mertani HC, Lee KO
and Lobie PE: Human growth hormone-regulated HOXA1 is a human
mammary epithelial oncogene. J Biol Chem. 278:7580–7590. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Shaoqiang C, Yue Z, Yang L, Hong Z, Lina
Z, Da P and Qingyuan Z: Expression of HOXD3 correlates with shorter
survival in patients with invasive breast cancer. Clin Exp
Metastasis. 30:155–163. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Rodriguez BA, Cheng AS, Yan PS, Potter D,
Agosto-Perez FJ, Shapiro CL and Huang TH: Epigenetic repression of
the estrogen-regulated Homeobox B13 gene in breast cancer.
Carcinogenesis. 29:1459–1465. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Fiegl H, Windbichler G, Mueller-Holzner E,
Goebel G, Lechner M, Jacobs IJ and Widschwendter M: HOXA11 DNA
methylation-a novel prognostic biomarker in ovarian cancer. Int J
Cancer. 123:725–729. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Bai Y, Fang N, Gu T, Kang Y, Wu J, Yang D,
Zhang H, Suo Z and Ji S: HOXA11 gene is hypermethylation and
aberrant expression in gastric cancer. Cancer Cell Int. 14:792014.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Liu YJ, Zhu Y, Yuan HX, Zhang JP, Guo JM
and Lin ZM: Overexpression of HOXC11 homeobox gene in clear cell
renal cell carcinoma induces cellular proliferation and is
associated with poor prognosis. Tumour Biol. 36:2821–2829. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Hassan NM, Hamada J, Murai T, Seino A,
Takahashi Y, Tada M, Zhang X, Kashiwazaki H, Yamazaki Y, Inoue N
and Moriuchi T: Aberrant expression of HOX genes in oral dysplasia
and squamous cell carcinoma tissues. Oncol Res. 16:217–224.
2006.PubMed/NCBI
|
|
67
|
De Souza Setubal Destro MF, Bitu CC,
Zecchin KG, Graner E, Lopes MA, Kowalski LP and Coletta RD:
Overexpression of HOXB7 homeobox gene in oral cancer induces
cellular proliferation and is associated with poor prognosis. Int J
Oncol. 36:141–149. 2010.PubMed/NCBI
|
|
68
|
Abate-Shen C: Deregulated homeobox gene
expression in cancer: Cause or consequence? Nat Rev Cancer.
2:777–785. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
69
|
Zhang X, Emerald BS, Mukhina S, Mohankumar
KM, Kraemer A, Yap AS, Gluckman PD, Lee KO and Lobie PE: HOXA1 is
required for E-cadherin-dependent anchorage-independent survival of
human mammary carcinoma cells. J Biol Chem. 281:6471–6481. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Xiao F, Bai Y, Chen Z, Li Y, Luo L, Huang
J, Yang J, Liao H and Guo L: Downregulation of HOXA1 gene affects
small cell lungcancer cell survival and chemoresistance under the
regulation of miR-100. Eur J Cancer. 50:1541–1554. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Bach C, Buhl S, Mueller D, García-Cuéllar
MP, Maethner E and Slany RK: Leukemogenic transformation by HOXA
cluster genes. Blood. 115:2910–2918. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Mohankumar KM, Xu XQ, Zhu T, Kannan N,
Miller LD, Liu ET, Gluckman PD, Sukumar S, Emerald BS and Lobie PE:
HOXA1-stimulated oncogenicity is mediated by selective upregulation
of components of the p44/42 MAP kinase pathway in human mammary
carcinoma cells. Oncogene. 26:3998–4008. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Zhu T, Starling-Emerald B, Zhang X, Lee
KO, Gluckman PD, Mertani HC and Lobie PE: Oncogenic transformation
of human mammary epithelial cells by autocrine human growth
hormone. Cancer Res. 65:317–324. 2005.PubMed/NCBI
|
|
74
|
Svingen T and Tonissen KF: Hox
transcription factors and their elusive mammalian gene targets.
Heredity (Edinb). 97:88–96. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Gebelein B, Culi J, Ryoo HD, Zhang W and
Mann RS: Specificity of Distalless repression and limb primordia
development by abdominal Hox proteins. Dev Cell. 3:487–498. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Bitu CC, Destro MF, Carrera M, da Silva
SD, Graner E, Kowalski LP, Soares FA nd and Coletta RD: HOXA1 is
overexpressed in oral squamous cell carcinomas and its expression
is correlated with poor prognosis. BMC Cancer. 12:1462012.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Kanhere A and Jenner RG: Noncoding RNA
localisation mechanisms in chromatin regulation. Silence. 3:22012.
View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Rinn JL, Kertesz M, Wang JK, Squazzo SL,
Xu X, Brugmann SA, Goodnough LH, Helms JA, Farnham PJ, Segal E and
Chang HY: Functional demarcation of active and silent chromatin
domains in human HOX loci by noncoding RNAs. Cell. 129:1311–1323.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Maamar H, Cabili MN, Rinn J and Raj A:
linc-HOXA1 is a noncoding RNA that represses Hoxa1 transcription in
cis. Genes Dev. 27:1260–1271. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Shah N and Sukumar S: The Hox genes and
their roles in oncogenesis. Nat Rev Cancer. 10:361–371. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Xu G, Chen J, Pan Q, Huang K, Pan J, Zhang
W, Chen J, Yu F, Zhou T and Wang Y: Long noncoding RNA expression
profiles of lung adenocarcinoma ascertained by microarray analysis.
PloS One. 9:e1040442014. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Zhang EB, Yin DD, Sun M, Kong R, Liu XH,
You LH, Han L, Xia R, Wang KM, Yang JS, et al: P53-regulated long
non-coding RNA TUG1 affects cell proliferation in human non-small
cell lung cancer, partly through epigenetically regulating HOXB7
expression. Cell Death Dis. 5:e12432014. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Liao WT, Jiang D, Yuan J, Cui YM, Shi XW,
Chen CM, Bian XW, Deng YJ and Ding YQ: HOXB7 as a prognostic factor
and mediator of colorectal cancer progression. Bian XW, Deng YJ,
Ding YQ. 17:3569–3578. 2011.
|
|
84
|
Storti P, Donofrio G, Colla S, Airoldi I,
Bolzoni M, Agnelli L, Abeltino M, Todoerti K, Lazzaretti M, Mancini
C, et al: HOXB7 expression by myeloma cells regulates their
pro-angiogenic properties in multiple myeloma patients. Leukemia.
25:527–537. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Yuan W, Zhang X, Xu Y, Li S, Hu Y and Wu
S: Role of HOXB7 in regulation of progression and metastasis of
human lung adenocarcinoma. Mol Carcinog. 53:49–57. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Wu X, Chen H, Parker B, Rubin E, Zhu T,
Lee JS, Argani P and Sukumar S: HOXB7, a homeodomain protein, is
overexpressed in breast cancer and confers epithelial-mesenchymal
transition. Cancer Res. 66:9527–9534. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
di Pietro M, Lao-Sirieix P, Boyle S,
Cassidy A, Castillo D, Saadi A, Eskeland R and Fitzgerald RC:
Evidence for a functional role of epigenetically regulated
midcluster HOXB genes in the development of Barrett esophagus. Proc
Natl Acad Sci USA. 109:9077–9082. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Zhao XD, Han X, Chew JL, Liu J, Chiu KP,
Choo A, Orlov YL, Sung WK, Shahab A, Kuznetsov VA, et al:
Whole-genome mapping of histone H3 Lys4 and 27 trimethylations
reveals distinct genomic compartments in human embryonic stem
cells. Cell Stem cell. 1:286–298. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Ke XS, Qu Y, Rostad K, Li WC, Lin B,
Halvorsen OJ, Haukaas SA, Jonassen I, Petersen K, Goldfinger N, et
al: Genome-wide profiling of histone h3 lysine 4 and lysine 27
trimethylation reveals an epigenetic signature in prostate
carcinogenesis. PLoS One. 4:e46872009. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Yang Z, Zhou L, Wu LM, Lai MC, Xie HY,
Zhang F and Zheng SS: Overexpression of long non-coding RNA HOTAIR
predicts tumor recurrence in hepatocellular carcinoma patients
following liver transplantation. Ann Surg Oncol. 18:1243–1250.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Li Z, Zhao X, Zhou Y, Liu Y, Zhou Q, Ye H,
Wang Y, Zeng J, Song Y, Gao W, et al: The long non-coding RNA
HOTTIP promotes progression and gemcitabine resistance by
regulating HOXA13 in pancreatic cancer. J Transl Med. 13:842015.
View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Cheng Y, Jutooru I, Chadalapaka G, Corton
JC and Safe S: The long non-coding RNA HOTTIP enhances pancreatic
cancer cell proliferation, survival and migration. Oncotarget.
6:10840–10852. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Quagliata L, Matter MS, Piscuoglio S,
Arabi L, Ruiz C, Procino A, Kovac M, Moretti F, Makowska Z,
Boldanova T, et al: Long noncoding RNA HOTTIP/HOXA13 expression is
associated with disease progression and predicts outcome in
hepatocellular carcinoma patients. Hepatology. 59:911–923. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Lander ES, Linton LM, Birren B, Nusbaum C,
Zody MC, Baldwin J, Devon K, Dewar K, Doyle M, FitzHugh W, et al:
Initial sequencing and analysis of the human genome. Nature.
409:860–921. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Duboule D: The rise and fall of Hox gene
clusters. Development. 134:2549–2560. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Petruk S, Sedkov Y, Brock HW and Mazo A: A
model for initiation of mosaic HOX gene expression patterns by
non-coding RNAs in early embryos. RNA Biol. 4:1–6. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Yekta S, Tabin CJ and Bartel DP: MicroRNAs
in the Hox network: An apparent link to posterior prevalence. Nat
Rev Genet. 9:789–796. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Yekta S, Shih IH and Bartel DP:
MicroRNA-directed cleavage of HOXB8 mRNA. Science. 304:594–596.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Woltering JM and Durston AJ: MiR-10
represses HoxB1a and HoxB3a in zebrafish. PloS One. 3:e13962008.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Zhang X, Lian Z, Padden C, Gerstein MB,
Rozowsky J, Snyder M, Gingeras TR, Kapranov P, Weissman SM and
Newburger PE: A myelopoiesis-associated regulatory intergenic
noncoding RNA transcript within the human HOXA cluster. Blood.
113:2526–2534. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Renfree MB, Papenfuss AT, Deakin JE,
Lindsay J, Heider T, Belov K, Rens W, Waters PD, Pharo EA, Sha G,
et al: Genome sequence of an Australian kangaroo, Macropus
eugenii, provides insight into the evolution of mammalian
reproduction and development. Genome Biol. 12:R812011. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Seo M, Choi JS, Rho CR, Joo CK and Lee SK:
MicroRNA miR-466 inhibits Lymphangiogenesis by targeting
prospero-related homeobox 1 in the alkali burn corneal injury
model. J Biomed Sci. 22:32015. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Liu Z, Zhu J, Cao H, Ren H and Fang X:
miR-10b promotes cell invasion through RhoC-AKT signaling pathway
by targeting HOXD10 in gastric cancer. Int J Oncol. 40:1553–1560.
2012.PubMed/NCBI
|
|
104
|
Yu H, Lindsay J, Feng ZP, Frankenberg S,
Hu Y, Carone D, Shaw G, Pask AJ, O'Neill R, Papenfuss AT and
Renfree MB: Evolution of coding and non-coding genes in HOX
clusters of a marsupial. BMC Genomics. 13:2512012. View Article : Google Scholar : PubMed/NCBI
|