Introduction
Gastrointestinal stromal tumors (GISTs) are the most
frequently occurring mesenchymal neoplasms of the gastrointestinal
tract (1). The crude annual incidence
of GISTs varies across the globe, although the incidence is ~130
cases per 1 million individuals in a population (2). In total, 50–70% of the tumors originate
in the stomach, while 20–30% arise in the small bowel, with the
duodenum being the rarest site (1,3).
Contrast-enhanced computed tomography (CT) is the imaging method of
choice to characterize an abdominal mass, to evaluate its extent,
and to determine the presence or absence of metastatic disease
(4). The vast majority of GISTs are
solid tumors, and cystic change is uncommon (5,6). There is
no significant difference between cystic and solid GISTs in terms
of the treatment and prognosis. For peritoneal cystic lesions, the
diagnosis of GIST is not initially considered. The 5-year survival
rate of patients with GIST is ~70% (7).
The most common intraabdominal cystic lesion
originates from the mesentery or retroperitoneal space (8). Other cystic lesion include duplication
cysts, pancreas pseudocysts or liver cysts. The present study
reports the case of a large cystic lesion arising from the stomach,
which was ultimately diagnosed as a GIST.
Case report
A 75-year-old man presented to Kunshan First
People's Hospital Affiliated to Jiangsu University (Kunshan,
Jiangsu, China) on February 25, 2015 with abdominal distension and
anorexia that had persisted for 1 month. There was no history
suggestive of gastric outlet obstruction, and no vomiting, melena
or constitutional symptoms. The patient had undergone no past
surgeries and had no history of pancreatitis. Upon examination, no
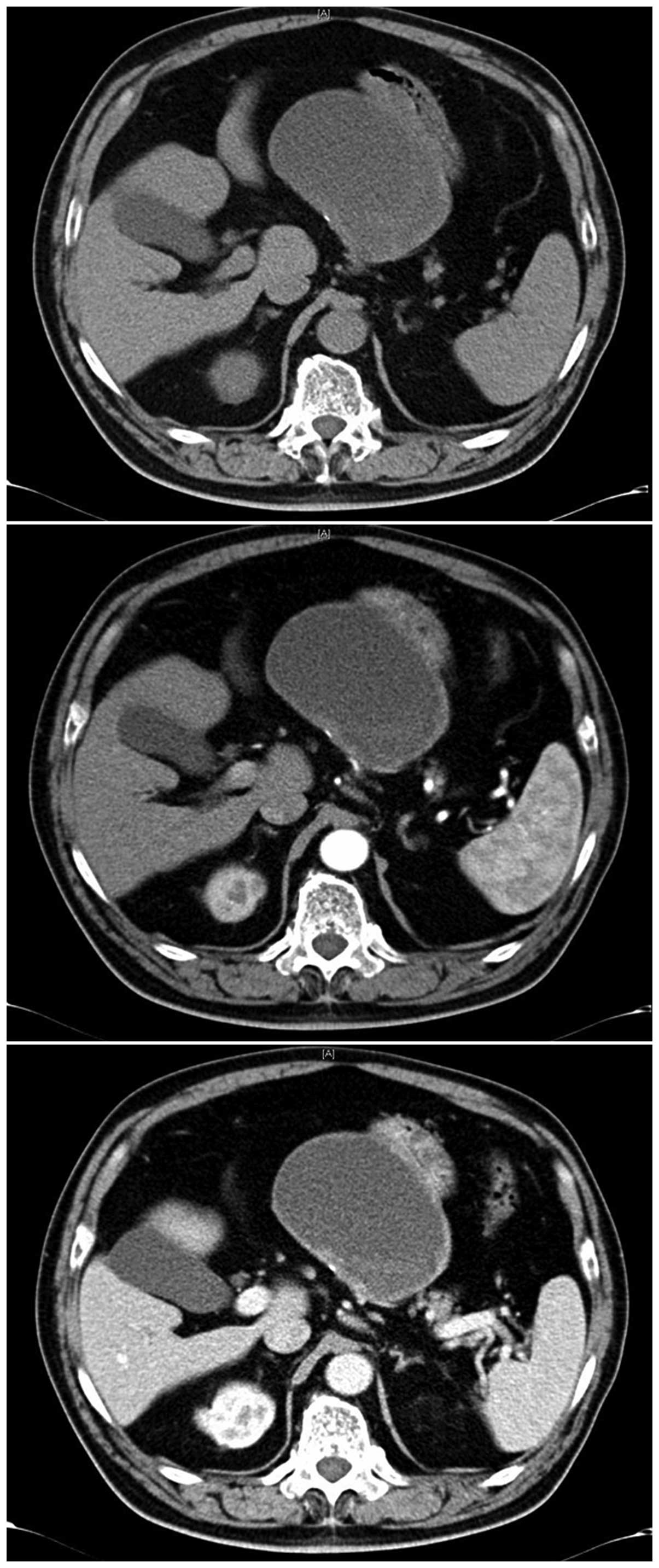
abdominal mass was palpable. A CT scan of the abdomen was
performed, which showed a well-defined exophytic cystic lesion
located between the liver and the stomach (Fig. 1). No metastatic localization was
detectable in the liver or lungs, and neither peritoneal
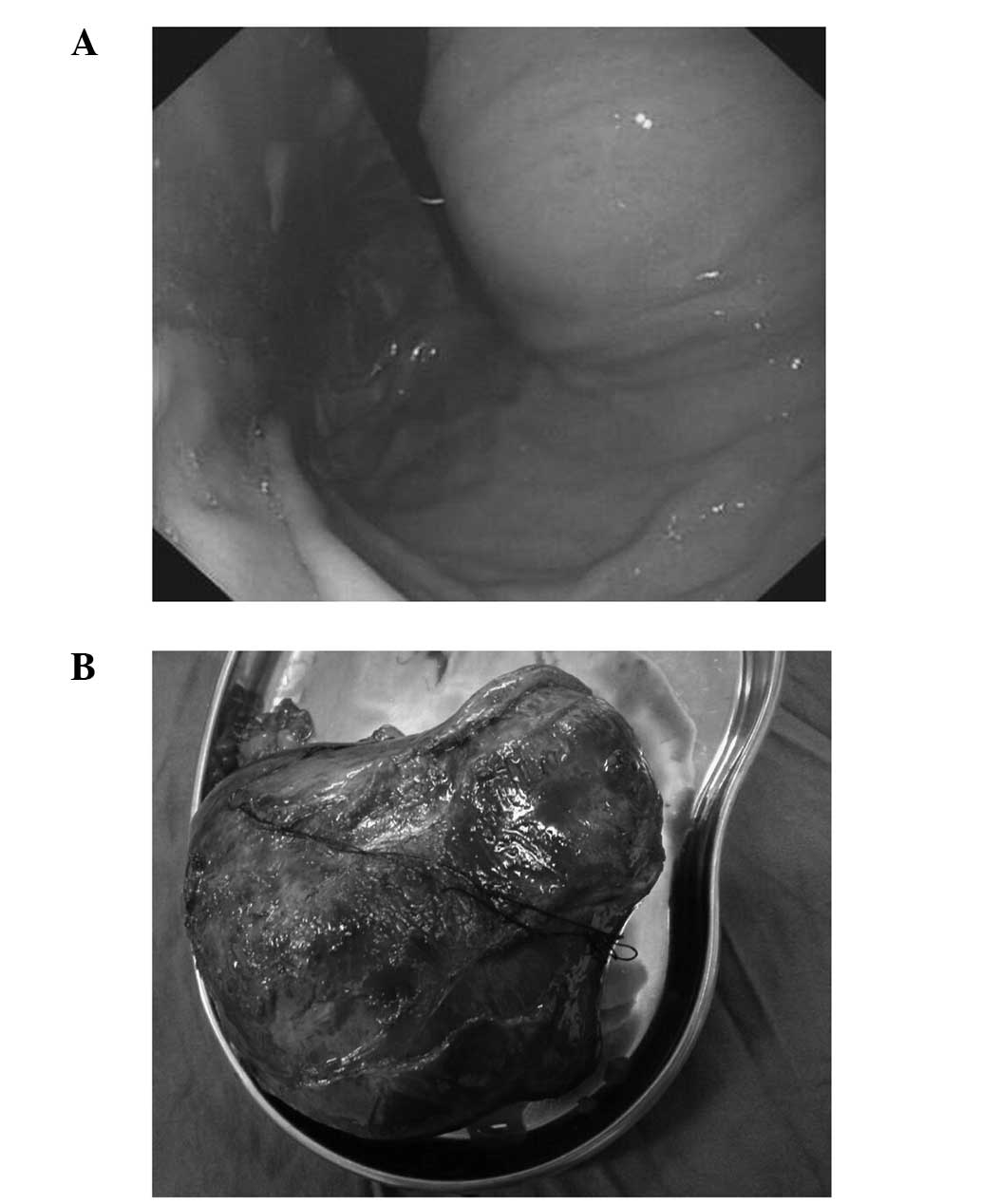
dissemination nor lymph node spreading were revealed. Gastroscopy
observed a large bulge at the lesser curvature of the stomach, with
an unremarkable mucosa (Fig. 2A).
Tumor markers, including carcinoembryonic antigen, α-fetoprotein,
cancer antigen (CA)125 and CA19-9, were all within the normal
ranges. A pre-operative diagnosis of a liver cyst, a pancreas
pseudocyst or a duplication cyst was formed.
Laparotomy revealed a reddish, bulky and
vascularized mass arising from the lesser curvature of the stomach.
A distal gastrectomy with en bloc resection of the mass was
performed (Fig. 2B), and the
continuity of the gastrointestinal tract was restored by a
gastroduodenal anastomosis (Billroth I). For histopathological
analysis, tissue sections (4 mm) were incubated with 4%
paraformaldehyde for 15 min at room temperature, and washed twice
with Tris-buffered saline (TBS) containing 0.1% saponin.
Subsequently, the tissues were incubated with TBS containing 0.3%
H2O2, 0.1% saponin and 0.02% NaN3
for 30 min to bock endogenous peroxidase activity, stained with
hematoxylin and eosin and observed under an optical microscope. The
histopathological analysis showed that the tumor was composed of
spindle cells arranged in bundles and braids. The diagnosis was of
a GIST with microscopically negative margins.
The macroscopically solid tumoral lesion measured
13×10×6 cm. Routine pathology confirmed a spindle-cell GIST
(Fig. 2B) and necrotic cystic
degeneration. Tumor sections were stained with mouse anti-c-kit
[cluster of differentiation (CD)117] polyclonal antibody (cat. no.
sc-393910), mouse anti-CD34 monoclonal antibody (cat. no.
sc-19621), mouse anti-desmin monoclonal antibody (cat. no.
sc-23879) and mouse anti-smooth muscle actin monoclonal antibody
(cat. no. sc-53015; all 1:500; all Santa Cruz Biotechnology, Inc.,
Dallas, TX, USA), followed by horseradish peroxidase-conjugated
goat anti-mouse secondary antibody (cat. no. sc-2005; 1:1,000;
Santa Cruz Biotechnology, Inc.). The tumor lesion showed strong
positive staining for c-kit and CD34, whereas S-100, desmin and
smooth muscle actin were negative. The mitotic count was <5/50
high-power fields. The lymph nodes that were removed were
uninvolved. The patient made an uneventful post-operative recovery,
being discharged from hospital at 10 days post-surgery. The patient
remained under follow-up by the hospital's medical oncology service
and was administered 400 mg imatinib daily for 1 year prior to
abandoning imatinib therapy for economic reasons. The latest CT
scan, performed at 14 months post-surgery, did not show any signs
of recurrence.
Discussion
GISTs are the most common sarcomas of the
gastrointestinal tract, accounting for ~1% of gastric neoplasms
(9). It is difficult to predict the
biological behavior of a GIST, which ranges from benign to
malignant, and the tumor size and mitotic index are believed to be
the most reliable prognostic factors (10). The vast majority of GISTs are solid
tumors and the minority of lesions present with cystic change upon
rapid tumor growth and necrosis (11). GISTs presenting with complete cystic
change and as an extragastric lesion is extremely rare.
Due to the rarity and different modes of
presentation of GISTs, the pre-operative diagnosis is extremely
difficult. A total of 25% of GISTs are found incidentally during
imaging or surgery for other disorders (12). Instrumental examination, including
gastroscopy, CT scan and endoscopic ultrasonography permits
mesenchymal tumors to be distinguished from other tumor types.
Fine-needle tissue acquisition biopsy samples obtained through
endoscopy or endoscopic ultrasonography cannot be used for a
reliable proliferation assessment as they are too small for
immunohistochemical analyses (13).
An accurate diagnosis of a GIST is often formed only after
surgery.
In conclusion, the vast majority of GISTs are solid
tumors, and cystic change is uncommon. Preoperatively, an
intraabdominal cyst may be considered a duplication cyst, a
pancreatic pseudocyst or a liver cyst. To the best of our
knowledge, the present study is the first to report a case of GIST
presenting as a cystic lesion. The patient presented with a cystic
lesion originating from the stomach, which was diagnosed as GIST
via a histological analysis. Therefore, GISTs presenting as cystic
lesions, although rare, should be considered in the differential
diagnosis of cystic lesions of the upper abdomen.
References
|
1
|
Joensuu H, Hohenberger P and Corless CL:
Gastrointestinal stromal tumor. Lancet. 382:973–983. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nilsson B, Bümming P, Meis-Kindblom JM,
Odén A, Dortok A, Gustavsson B, Sablinska K and Kindblom LG:
Gastrointestinal stromal tumors: The incidence, prevalence,
clinical course, and prognostication in the preimatinib mesylate
era - a population-based study in western Sweden. Cancer.
103:821–829. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Samelis GF, Ekmektzoglou KA and Zografos
GC: Gastrointestinal stromal tumours: Clinical overview, surgery
and recent advances in imatinib mesylate therapy. Eur J Surg Oncol.
33:942–950. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Abdel-Monem S, Enaba MM, Hassan TA, et al:
Multislice CT imaging of gastrointestinal stromal tumors (GISTs).
Egypt J Radiol Nuc Med. 42:1–7. 2011. View Article : Google Scholar
|
|
5
|
Kumar A, Jakhmola CK, Chauhan SS and Singh
A: A typical presentation of gastrointestinal stromal tumor
masquerading as a large duodenal cyst: A case report. Int J Surg
Case Rep. 9:123–126. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cavallaro G, Sadighi A, Polistena A, Rossi
V, Cristaldi M, Paparelli C and De Toma G: Pedunculated giant GISTs
of the stomach with exophytic growth: Report of two cases. Int J
Surg. 6:e80–e82. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Joensuu H, Vehtari A, Riihimäki J, Nishida
T, Steigen SE, Brabec P, Plank L, Nilsson B, Cirilli C, Braconi C,
et al: Risk of recurrence of gastrointestinal stromal tumour after
surgery: An analysis of pooled population-based cohorts. Lancet
Oncol. 13:265–274. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Choi BI, Kim YH and Han JK: Hepatobiliary
and pancreatic: Cystic liver lesion in a man with abdominal pain. J
Gastroenterol Hepatol. 14:609–613. 1999.PubMed/NCBI
|
|
9
|
Goettsch WG, Bos SD, Breekveldt-Postma N,
Casparie M, Herings RM and Hogendoorn PC: Incidence of
gastrointestinal stromal tumours is underestimated: Results of a
nation-wide study. Eur J Cancer. 41:2868–2872. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bucher P, Egger JF, Gervaz P, Ris F,
Weintraub D, Villiger P, Buhler LH and Morel P: An audit of
surgical management of gastrointestinal stromal tumours (GIST). Eur
J Surg Oncol. 32:310–314. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shaikh ST, Upwanshi MH, Shetty TS, Ghetla
SR and Gheewala H: A Large Cystic Variant of Gastro-intestinal
Stromal Tumour arising from the Jejunum: A Case Report. J Clin
Diagn Res. 9:11–12. 2015.
|
|
12
|
Al-Thani H, El-Menyar A, Rasul KI,
Al-Sulaiti M, El-Mabrok J, Hajaji K, Elgohary H and Tabeb A:
Clinical presentation, management and outcomes of gastrointestinal
stromal tumors. Int J Surg. 12:1127–1133. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ricci R, Chiarello G, Attili F, Fuccio L,
Alfieri S, Persiani R, Di Pietro S, Martini M, Costamagna G,
Larocca LM and Larghi A: Endoscopic ultrasound-guided fine needle
tissue acquisition biopsy samples do not allow a reliable
proliferation assessment of gastrointestinal stromal tumours. Dig
Liver Dis. 47:291–295. 2015. View Article : Google Scholar : PubMed/NCBI
|