Introduction
Lung cancer is the leading cause of cancer-related
mortality worldwide (1). The
age-standardized (world) lung cancer mortality rate in Greece shows
that one in two cancer-related mortalities in men are caused by
lung cancer (50%); the figure for women is much lower at 6.3%
(2). The majority of patients (70%)
exhibit advanced disease at the time of diagnosis (3), and 5 years after diagnosis, only 16.6%
of patients remain alive (4).
Research in the field of more effective management
of lung cancer is clearly worthwhile, with the goal of increasing
survival while maintaining quality of life, and promising advances
have been made. The current standard of care for advanced non-small
cell lung cancer (NSCLC) is cytotoxic combination chemotherapy with
a platinum compound (carboplatin or cisplatin) and one other agent,
such as pemetrexed, vinorelbine, paclitaxel, gemcitabine or
docetaxel (5,6). Therapy is usually administered for a
maximum of 6 cycles unless the disease progresses or there is no
response (6).
Pemetrexed inhibits thymidylate synthase and other
folate-dependent enzymes involved in the metabolism and synthesis
of DNA precursors. A phase III randomized study showed significant
survival benefits of pemetrexed over gemcitabine in combination
with cisplatin as first-line treatment for non-squamous NSCLC.
Median overall survival (OS) was 11.8 months for the pemetrexed
arm, with a hazard ratio (HR) of 0.81 [95% confidence interval
(CI), 0.70–0.94; P=0.005), compared with 10.4 months for
gemcitabine (7).
Further survival and disease control benefits have
been documented with the addition of bevacizumab to platinum-based
chemotherapy doublets in the first-line treatment of non-squamous
NSCLC (4). Bevacizumab, a humanized
monoclonal antibody against vascular endothelial growth factor, is
a vascular-targeted therapy that may inhibit neovascularization.
The addition of bevacizumab to chemotherapy combinations resulted
in a novel treatment standard based on the results of Eastern
Cooperative Oncology Group (ECOG) study E4599, first presented at
the American Society of Clinical Oncology 2005 meeting and
published in full in 2006. Median OS time was 19% longer when
bevacizumab was added to chemotherapy (paclitaxel plus carboplatin)
(12.3 vs. 10.3 months respectively; HR for mortality, 0.79;
P=0.003) (8). Progression-free
survival (PFS) was also significantly longer in the bevacizumab
group, and survival rates at 1 and 2 years were greater compared
with chemotherapy alone (51 and 23% with bevacizumab, vs. 44 and
15% without bevacizumab, respectively) (8). The addition of bevacizumab to standard
combination chemotherapy appears to improve overall survival
(9).
Currently, the combination of bevacizumab plus
pemetrexed and a platinum agent has emerged as an important
first-line treatment for non-squamous NSCLC. To date, findings with
this combination have been reported in only 7 studies: The first
phase II study in 2009 (10), 4
further phase II studies (11–14), 1
observational study (15) and 1
population-based study (16).
Furthermore, published data for the administration of this
combination to patients with brain metastases (BM) have been
reported for only few cases, and no comparisons of survival between
patients with and without BM are available. More research in this
area is clearly required, as the frequency of tumor metastases to
the central nervous system is increasing, and it is estimated that
20–40% of all patients with systemic cancer develop BM (17).
The present single-center observational study aimed
to contribute to the current knowledge on the efficacy and safety
of combined bevacizumab plus pemetrexed plus carboplatin therapy in
patients with advanced non-squamous NSCLC. This analysis includes
patients with BM.
Patients and methods
The present study was an open-label, observational
study conducted at the St. Savas Anticancer Hospital in Athens,
Greece. Medical records of lung cancer patients treated at the
hospital clinic between September 2011 and October 2014 were
reviewed for eligibility. All patients were treated once written
informed consent was obtained.
Patient eligibility
Patients with inoperable locally advanced stage IIIB
or IV non-squamous NSCLC were eligible for the study. The patients
should have received bevacizumab plus pemetrexed plus carboplatin
as first-line therapy. Further inclusion criteria were an ECOG
performance status (PS) score of ≤2 and adequate hematological,
hepatic and renal function as follows: Hemoglobin, >9.5 g/dl;
absolute neutrophil count, >1,500/dl; platelet count,
>100,000/dl; international normalized ratio, <1.5; total
bilirubin, <1.5 times the upper normal limit; and serum
creatinine, <2.0 mg/dl. Patients were excluded if they had mixed
NSCLC, small cell tumors, mixed adenosquamous carcinoma and if they
were epidermal growth factor receptor- or anaplastic lymphoma
kinase-positive. Patients with currently non-life threatening BM
were included.
Treatment
The treatment schedule consisted of induction
therapy with carboplatin (area under the curve, 5), pemetrexed (500
mg/m2) and bevacizumab (15 mg/kg), every 3 weeks for 6
cycles. The patients also received vitamin B12 and folic acid
supplementation (1 mg) prior to the first chemotherapy
administration. Patients without disease progression at the end of
induction therapy received bevacizumab (15 mg/kg) and pemetrexed
(500 mg/m2) as maintenance therapy until disease
progression or unacceptable toxicity. An evaluation was performed
every 3 cycles.
Response to treatment
Response was assessed using the Response Evaluation
Criteria in Solid Tumors, version 1.1 (18). Computed tomography (CT) scans were
obtained after the first 2 cycles and every 3 cycles thereafter.
The treating physicians assessed the clinical condition of the
patients, and documented improvement or worsening, or stable
disease (SD). Weight loss, appetite changes, pain levels and pain
relief were also documented. The safety profile was monitored and
toxicity grades were assigned according to the National Cancer
Institute Common Terminology Criteria, version 4 (19).
Data collection and statistical
analysis
The primary endpoints were the median PFS and OS
times. Survival estimates were calculated using Kaplan-Meier
analysis and log-rank (Mantel-Cox) testing for between-group
comparisons. PFS time was calculated from the administration of the
first cycle to the time of disease progression. OS was calculated
from the administration of the first cycle to the time of mortality
from any cause. Patients who were alive or remained on treatment at
the data cutoff date for the analysis were censored at that date.
Actuarial PFS and OS percentages were also calculated via the
‘cumulative proportion surviving at end of each month’ Life Tables
analysis (20,21). Secondary endpoints were safety and
response to therapy, namely, a complete response, partial response
(PR), SD and progressive disease. Descriptive statistics were used.
All analyses were performed with the use of the statistical package
SPSS version 13.0 for Windows (SPSS Inc., Chicago, IL, USA).
Results
Patient characteristics
A total of 39 patients with non-squamous NSCLC were
administered induction therapy with bevacizumab plus pemetrexed
plus carboplatin as first-line treatment during the study period.
Half of the patients were >60 years old and the 64.1% were men
(Table I). At diagnosis, 24 patients
(61.5%) presented with stage IV disease and an ECOG PS score of 0
(Table I). The majority of patients
(66.7%) exhibited single site metastasis, and most often this was
lung or bone metastasis (Table I). In
total, 11 patients (28.2%) presented with BM, all symptomatic. Upon
BM progression, as verified by CT scan, radiotherapy was
administered, while study treatment was discontinued and modified
accordingly.
 | Table I.Patient demographics and disease
characteristics at the start of treatment (n=39). |
Table I.
Patient demographics and disease
characteristics at the start of treatment (n=39).
| Characteristic | Value |
|---|
| Age in years |
|
| Mean
(SD) | 60.9 (8.9) |
| Median
(range) | 60.0 (44–78) |
| Male
gender, n (%) | 25 (64.1) |
| Stage, n (%) |
|
|
IIIb | 15 (38.5) |
| IV | 24 (61.5) |
| Histology, n
(%) |
|
|
Adenocarcinoma | 39 (100.0) |
| ECOG PS, n (%) |
|
| 0 | 30 (76.9) |
| 1 | 8 (20.5) |
| 2 | 1 (2.6) |
| Number of organs
with metastasis, n (%) |
|
|
One | 26 (66.7) |
|
Two | 8 (20.5) |
|
Three | 4 (10.3) |
|
Four | 1 (2.6) |
| Site of metastases,
n (%) |
|
|
Lungs | 18 (46.2) |
|
Bones | 13 (33.3) |
|
Brain | 11 (28.2) |
|
Liver | 8 (20.5) |
| Adrenal
glands | 3 (7.7) |
| Pleural
effusion | 1 (2.6) |
|
Pleura | 1 (2.6) |
| Lymph
nodes | 1 (2.6) |
|
Pancreas | 1 (2.6) |
Treatment characteristics
Of the 39 patients, 22 (56.4%) completed 6 cycles of
induction therapy, 5 patients (12.8%) completed 5 cycles, 4
patients (10.3%) completed 4 cycles and another 4 patients (10.3%)
completed 3 cycles. At the end of study follow-up (October 2014), 4
patients remained on treatment. A total of 26 patients received
maintenance therapy. Half of these completed >5 maintenance
therapy cycles (median, 5.5 cycles; range, 1–16 cycles; mean, 6.2
cycles; standard deviation, 3.9 cycles). In total, 13 patients
completed ≥4 cycles, 9 patients competed ≥6 cycles, 5 patients
completed 1–5 cycles, 4 patients completed ≥10 cycles and 8
patients remained on treatment upon completion of follow-up.
Response and survival analysis
A total of 28 patients completed 6 cycles of
induction therapy, and in half of the patient population, disease
was stabilized. From the remaining population, the majority
achieved a PR (Table II). Actuarial
PFS and OS percentages remained high at 6 months and gradually
decreased thereafter (Table
III).
 | Table II.Response to induction therapy
following completion of 6 cycles. |
Table II.
Response to induction therapy
following completion of 6 cycles.
| Clinical response
after completion of 3 cycles (n=28a) | Total, n (%) | Without brain
metastases, n | With brain
metastases, n |
P-valueb |
|---|
| Stable
diseasea | 14 (50.0) | 11 | 3 | 0.482 |
| Partial
response | 9
(32.1) | 8 | 1 | 0.194 |
| Progressive
disease | 5
(17.9) | 3 | 2 | 0.530 |
| Complete
responsea | 1 (3.6) | 0 | 1 | 0.106 |
 | Table III.Selected cumulative survival
percentages for PFS and OS times at 3, 6, 12, 18 and 24 months
calculated via the actuarial life table in monthly interval
analysis. |
Table III.
Selected cumulative survival
percentages for PFS and OS times at 3, 6, 12, 18 and 24 months
calculated via the actuarial life table in monthly interval
analysis.
|
| Actuarial survival
(%) |
|---|
|
|
|
|---|
|
| PFS | OS |
|---|
|
|
|
|
|---|
| Time
post-treatment | Total | With brain
metastases | Without brain
metastases | Total | With brain
metastases |
|---|
| 3 months | 94 | 89 | 96 | 97 | 100 |
| 6 months | 64 | 52 | 68 | 88 | 88 |
| 12 months | 13 | 20 | 10 | 56 | 76 |
| 18 months | 4 | – | 5 | 38 | 15 |
| 24 months | 4 | – | 5 | 32 | 15 |
Median PFS time was ~8 months, and OS ranged between
12 and 16 months, with no statistically significant differences
between men and women (Table
IV).
 | Table IV.Median Kaplan-Meier estimates for PFS
and OS times (months). |
Table IV.
Median Kaplan-Meier estimates for PFS
and OS times (months).
| Parameter | Median, months | 95% confidence
interval |
|---|
| PFS |
|
|
|
Total | 8.2 | 7.05–9.35 |
|
Patients with brain
metastases | 8.2 | 2.83–13.57 |
|
Patients without brain
metastases | 8.0 | 6.77–9.23 |
|
Men | 8.0 | 4.89–11.11 |
|
Women | 8.2 | 7.70–8.70 |
| OS |
|
|
|
Total | 14.0 | 8.46–19.54 |
|
Patients with brain
metastases | 14.0 | 7.52–20.49 |
|
Patients without brain
metastases | 12.0 | 0.00–28.46 |
|
Men | 16.0 | 2.59–29.41 |
|
Women | 12.0 | 6.15–17.85 |
Subpopulation of patients with BM at
diagnosis
The response to treatment for patients with and
without BM did not differ significantly after 3 or 6 months
(Table II), nor were there
significant differences [log rank (Mantel-Cox)] for median PFS
(P=0.748) or OS (P=0.447) time (Table
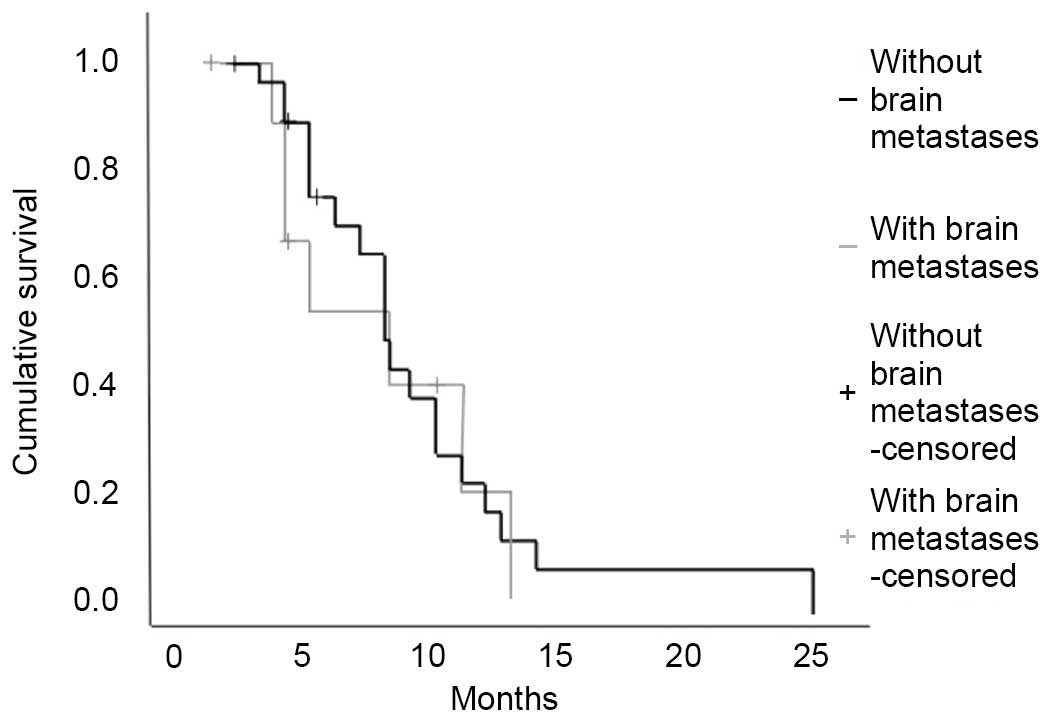
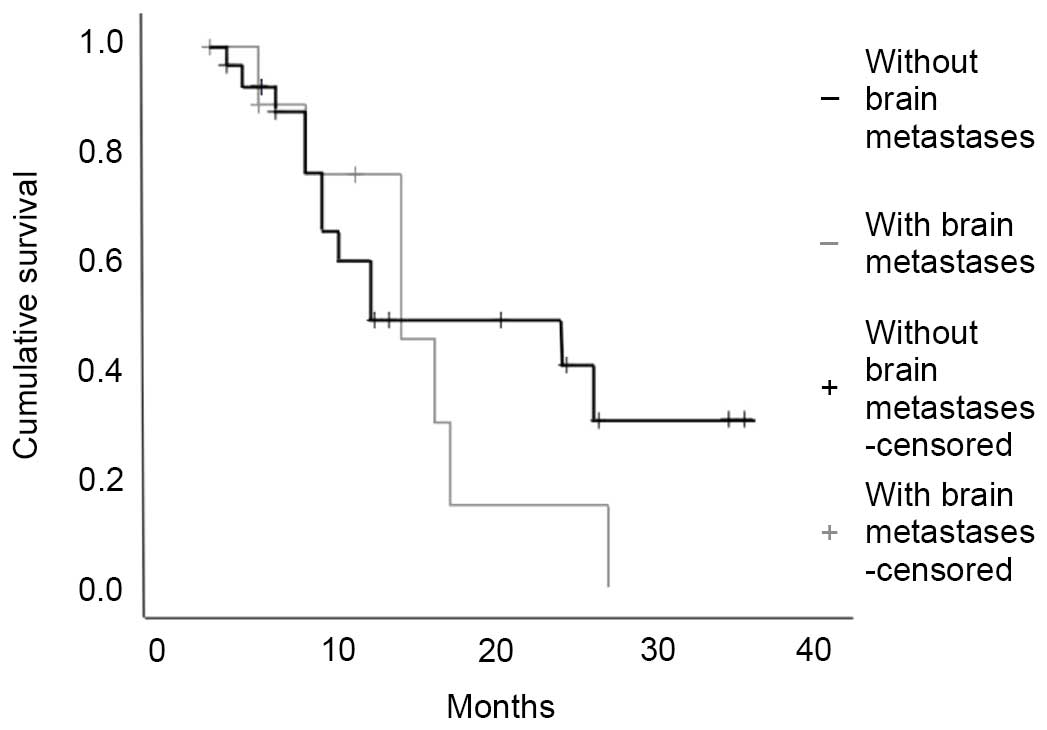
IV; Figs. 1 and 2). However, the OS survival curve for
patients with BM at diagnosis had a much steeper slope, suggesting
worse survival for these patients (Fig.
2).
Toxicity
The majority of patients (30/39; 76.9%) did not
experience adverse events during treatment. Neutropenia occurred in
4 patients, renal function deterioration in 2 patients,
myelotoxicity in 2 patients, hypersensitivity responses to
carboplatin in 2 patients, and anemia and fever in 1 patient,
respectively. All were manageable and none led to treatment
discontinuation or dose modification. There were no
treatment-related mortalities.
Discussion
The present study population consisted of newly
diagnosed, treatment-naïve patients with non-squamous metastatic
NSCLC with or without BM. Patients received induction therapy with
a combination of bevacizumab plus pemetrexed plus carboplatin,
followed by maintenance therapy with bevacizumab plus pemetrexed.
Analysis showed good survival times and a favorable safety profile
in all patients, regardless of the presence of BM.
Findings for the same treatment as in the present
study, i.e., induction and maintenance therapy in treatment-naïve
non-squamous NSCLC patients, were available from only 7 studies
(10–16). The main design characteristics and
treatment outcomes of these studies are summarized in Table V. A total of 5 studies were phase II
studies (10–14), 1 was a population-based study
(16), and only 1 was an
observational study with similar design characteristics to the
present study (15). The present
study results of a median PFS time of 8 months, and a range of OS
times of between 12 and 16 months are similar to those reported in
these previous trials, in which the shortest PFS time was 5.6
months in the population-based study by Nakamura et al
(2012) (16) and the longest was 10.2
months in the phase II study by Spigel et al (2012)
(12). For OS, the shortest median
was 12.6 months in the PointBreak study by Patel et al
(2013) (13) and the longest was 19.3
months in the population-based study by Nakamura et al
(2012) (16). Notably, the
population-based study of Nakamura et al (2012) (16) reported the shortest median PFS time
and the longest median OS time, indicating that a shorter PFS time
does not necessary imply a worse OS time. The only study with a
design similar to the present study, i.e., an observational,
single-center study, was that by Malhotra et al (2010),
which did not report median OS and PFS times, but did report
response rates (RR) and actuarial survival percentages: 52% of
patients exhibited a PR, disease control was documented for 40%,
and the actuarial OS and PFS rates after 12 months were 83 and 63%,
respectively. The corresponding results of the present study were
lower than this, but high overall; in terms of RR, the results were
close to the 34.1% reported in the PointBreak study (13) and the 35% reported in the study by
Spigel et al (12). In terms
of disease control, the present results lie within the range
reported by the remaining studies, with the lowest rate of 30.4%
reported in the study by Yokoi et al (14) and the highest rate of 65.9% in the
PointBreak study (13).
 | Table V.Characteristics and main results of
studies on first-line therapy with bevacizumab plus pemetrexed plus
carboplatin in non-squamous non-small cell lung cancer
patients. |
Table V.
Characteristics and main results of
studies on first-line therapy with bevacizumab plus pemetrexed plus
carboplatin in non-squamous non-small cell lung cancer
patients.
| First author,
year | Study design | Patients,
na | Brain metastases, n
(%) | Median age,
years | Median OS,
months | Median PFS,
months | Response (partial),
% | Disease control
(stable), % | (Ref.) |
|---|
| Patel et al,
2009 | Phase II,
multicenter | 50 | 0 (0.0) | 63.5 | 14.1 |
7.8 | 53.0 | 33.0 | (10) |
| Stevenson et
al, 2012 | Phase II,
two-center | 40 | 0 (0.0) | 65.3 | 17.1 |
7.1 | 42.0 | 38.0 | (11) |
| Spigel et
al, 2012 | Phase II,
randomized, multicenter | 55 | 0 (0.0) | >70.0 | 14.8 | 10.2 | 35.0 | 45.0 | (12) |
| Patel et al,
2013 | (Point break trial)
Phase III, randomized, multicenter | 472 | 52 (11.0) | 60.0 | 12.6 | 6.0 | 34.1 | 65.9 | (13) |
| Yokoi et al,
2014 | Phase II,
prospective single-center | 23 | 0 (0.0) | 64.0 | 18.6 |
8.6 | 56.5 | 30.4 | (14) |
| Malhotra et
al, 2010 | Observational,
retrospective, single-center | 27 | 2 (7.4) | 61.0 | – | – | 52.0 | 40.0 | (15) |
| Nakamura et
al, 2012 | (POSITIVE trial)
Population-based observational | 54 | N/A | 66.0 | 19.3 |
5.6 | 41.6 | 36.9 | (16) |
| Present study | Observational
retrospective-prospective single-center | 39 | 11 (28.2) | 60.0 | 14.0 |
8.2 | 32.1 | 50.0 |
Special attention should be paid to the present
findings with regard to the patients with BM. BM are present in
7–10% of stage IV lung cancer patients at the time of diagnosis,
and in 20–40% following disease progression (22). The incidence of these metastases is
increasing (23). Patients with BM
have a worse prognosis, a worse quality of life and an increased
risk of mortality (22,24), and OS in these patients is extremely
poor (22,25,26).
Patients with BM used to be treated with steroids and whole-brain
radiation therapy. Chemotherapy was not an option due to concerns
over the limited permeability of the blood-brain barrier (27). Patients with BM were therefore
excluded from chemotherapy and were ineligible for clinical trials
(22). Nowadays, it is known that the
blood brain barrier is disrupted when brain metastases develop
(27,28). Certain trials have been performed on
the activity of first-line chemotherapy on brain metastases in
NSCLC and have shown response rates ranging from 23–50% (27). No data, however, have been published
for first-line bevacizumab plus pemetrexed plus carboplatin therapy
in this group of patients. Indeed, except for the PointBreak study
(13), all other studies excluded
patients with BM and the population-based study did not report
outcomes on such patients (16). In
the PointBreak study, the subgroup analysis on the patients with BM
did not compare treatment activity between patients with and
without BM, however, within the subgroup of patients with BM, it
compared the efficacy of the two treatment arms of bevacizumab plus
pemetrexed plus carboplatin and bevacizumab plus paclitaxel plus
carboplatin. The present study compared patients with and without
BM, and did not observe any differences in terms of efficacy and
safety between the two groups. The number of patients was small and
it is therefore not possible to draw firm conclusions, but it is
worth pointing out that despite the worse prognosis of patients
with BM, the survival outcomes with bevacizumab plus pemetrexed
plus carboplatin were similar to those observed in patients without
BM.
To the best of our knowledge, there have been no
further reports on the effects of the combination of bevacizumab
plus pemetrexed plus carboplatin on non-squamous NSCLC patients
with BM, although data with different combinations of the
individual components have been published.
For pemetrexed, data regarding patients with BM have
been published in the first- and second-line settings. A response
rate of 40% for newly diagnosed BM was reported by Bailon et
al (2012) (29) in 30 patients
with non-squamous NSCLC following first-line therapy with
pemetrexed plus carboplatin. The median OS time was 39 weeks
(29). In a phase II study by Barlesi
et al (2011) (30), first-line
therapy with pemetrexed plus cisplatin resulted in an overall RR of
34.9% (95% CI, 21–50.9) and a disease control rate of 72.1%
(overall response plus SD rate). The study enrolled 43 patients
with NSCLC and BM. Median OS time was 7.4 months (95% CI, 5.8–9.6).
Positive effects of pemetrexed in second-line treatment (34
patients) and third-line treatment (5 patients) were reported in an
observational study by Bearz et al (2010) (31) in 39 patients with advanced NSCLC and
BM: 69% of the patients experienced a clinical benefit (15 PR and
12 SD). Radiological evidence for a cerebral benefit that included
PR and stable brain disease was recorded in 82% of the patients (32
patients). The median OS time was 10 months. The results of these
three studies indicate that pemetrexed is a good treatment option
in such patients (29–31).
With regard to carboplatin in patients with BM,
Bernardo et al (2002) (32)
reported an RR of 45% for carboplatin plus vinorelbine plus
gemcitabine in the first-line setting. The study enrolled 22
patients with NSCLC and BM, with a median survival time of 33 weeks
(range, 18–62 weeks) (32). Edelman
et al (2010) (33) evaluated
the activity of the doublets carboplatin plus gemcitabine,
paclitaxel plus gemcitabine and carboplatin plus paclitaxel in
1,135 patients with advanced NSCLC. A median OS time of 7.7 months
(95% CI, 6.7–9.3) and an RR of 28.9% were achieved in the 194
patients (17.1%) with BM (33).
Regarding the use of bevacizumab in NSCLC patients
with BM, it should be mentioned that such patients were initially
excluded from the relative studies. This was due to alleged
concerns over an increased risk of intracranial hemorrhage after
the mortality due to cerebral hemorrhage of a woman with
hepatocellular carcinoma treated in a phase I study (28,34,35).
Hepatocellular carcinoma, however, may itself lead to cerebral
hemorrhage due to impaired liver function and subsequent
coagulopathy (34,36). Furthermore, accumulated safety data do
not show an increased incidence of cerebral hemorrhage in patients
with NSCLC and central nervous system (CNS) metastases. Such
patients should therefore not be denied bevacizumab treatment
(34,37,38). A
retrospective analysis of safety data from 13 randomized controlled
trials with 4,760 patients under bevacizumab therapy (91 with CNS
metastases) did not show an increased risk of developing cerebral
hemorrhage while on treatment with bevacizumab (34). The phase II PASSPORT study was
specifically designed to evaluate the safety of the administration
of bevacizumab in 115 patients with non-squamous NSCLC and BM
(38). Bevacizumab was administered
in the first- (n=76) or second-line (n=39) setting in different
combinations with chemotherapeutic agents. No grade ≥2 cerebral
hemorrhage was observed (38). The
ARIES observational study evaluated the efficacy and safety of
bevacizumab plus chemotherapy in 1,967 treatment-naïve patients
with advanced NSCLC; 8% of the patients exhibited BM (39). Overall, 1.2% of the total population
suffered from pulmonary hemorrhage and 0.2% from grade 3–5 CNS
hemorrhage. The median OS time was 13.0 months (95% CI, 12.2–13.8
months). In general, the efficacy and safety profiles were similar
to the results reported in the large phase III trials, SAIL
(40) and E4599 (8), and the phase IV AVAIL study (41). Although patients with BM are no longer
considered ineligible for bevacizumab administration (4) and the European Medicines Agency (EMA)
removed label restriction on March 25, 2009, to allow patients with
untreated CNS metastases to receive bevacizumab (42,43),
information regarding its activity in non-squamous NSCLC patients
with BM is lacking.
The present findings in everyday clinical practice
with bevacizumab plus pemetrexed plus carboplatin as first-line
therapy for non-squamous NSCLC therefore contribute to the better
understanding of the activity of this regimen in this population,
including patients with BM.
The observational design of the present study is a
limitation, as it is susceptible to systemic bias, for example,
data entry errors, reporting errors, missing data and unrecorded
confounders. However, such a design reflects the everyday clinical
setting and has fewer restrictions than a controlled phase II
study. The small sample size is a further limitation that could be
overcome in future analyses with a multicenter collaborative
design. The major strength of the study is its contribution to the
current understanding of the activity of bevacizumab plus
pemetrexed plus carboplatin in the first-line therapy of advanced
non-squamous NSCLC patients, including those with BM.
Overall, induction treatment with a combination of
bevacizumab plus pemetrexed plus carboplatin, followed by
bevacizumab plus pemetrexed as maintenance therapy in a small
number of patients in the everyday setting was effective and well
tolerated in advanced NSCLC, with similar survival benefits for
patients with and without BM, despite the worse prognosis of the
former.
References
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Atlas of cancer mortality in the European
union and the European Economic Area: 1993–1997. IARC Sci Publ.
1–259. 2008.
|
|
3
|
Besse B, Adjei A, Baas P, Meldgaard P,
Nicolson M, Paz-Ares L, Reck M, Smit EF, Syrigos K, Stahel R, et
al: 2nd ESMO consensus conference on lung cancer: Non-small-cell
lung cancer first-line/second and further lines of treatment in
advanced disease. Ann Oncol. 25:1475–1484. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
NCCN: National Comprehensive Cancer
Network (NCCN) Clinical Practice Guidelines in Oncology, .
Non-Small Cell Lung Cancer. Version I. 2015.https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
|
|
5
|
National Cancer Institute, .
PDQ® Non-Small Cell Lung Cancer Treatment. Bethesda, MD:
National Cancer Institute; http://www.cancer.gov/types/lung/hp/non-small-cell-lung-treatment-pdqAccessed
Nov 26, 2014.
|
|
6
|
Azzoli CG, Baker S Jr, Temin S, Pao W,
Aliff T, Brahmer J, Johnson DH, Laskin JL, Masters G, Milton D, et
al: American society of clinical oncology clinical practice
guideline update on chemotherapy for stage IV non-small-cell lung
cancer. J Clin Oncol. 27:6251–6266. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Scagliotti GV, Parikh P, von Pawel J,
Biesma B, Vansteenkiste J, Manegold C, Serwatowski P, Gatzemeier U,
Digumarti R, Zukin M, et al: Phase III study comparing cisplatin
plus gemcitabine with cisplatin plus pemetrexed in
chemotherapy-naive patients with advanced-stage non-small-cell lung
cancer. J Clin Oncol. 26:3543–3551. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sandler A, Gray R, Perry MC, Brahmer J,
Schiller JH, Dowlati A, Lilenbaum R and Johnson DH:
Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell
lung cancer. N Engl J Med. 355:2542–2550. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schmid-Bindert G: Update on antiangiogenic
treatment of advanced non-small cell lung cancer (NSCLC). Target
Oncol. 8:15–26. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Patel JD, Hensing TA, Rademaker A, Hart
EM, Blum MG, Milton DT and Bonomi PD: Phase II study of pemetrexed
and carboplatin plus bevacizumab with maintenance pemetrexed and
bevacizumab as first-line therapy for nonsquamous non-small-cell
lung cancer. J Clin Oncol. 27:3284–3289. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Stevenson JP, Langer CJ, Somer RA, Evans
TL, Rajagopalan K, Krieger K, Jacobs-Small M, Dyanick N, Milcarek
B, Coakley S, et al: Phase 2 trial of maintenance bevacizumab alone
after bevacizumab plus pemetrexed and carboplatin in advanced,
nonsquamous nonsmall cell lung cancer. Cancer. 118:5580–5587. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Spigel DR, Hainsworth JD, Shipley DL,
Ervin TJ, Kohler PC, Lubiner ET, Peyton JD, Waterhouse DM, Burris
HA III and Greco FA: A randomized phase II trial of
pemetrexed/gemcitabine/bevacizumab or
pemetrexed/carboplatin/bevacizumab in the first-line treatment of
elderly patients with advanced non-small cell lung cancer. J Thorac
Oncol. 7:196–202. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Patel JD, Socinski MA, Garon EB, Reynolds
CH, Spigel DR, Olsen MR, Hermann RC, Jotte RM, Beck T, Richards DA,
et al: PointBreak: A randomized phase III study of pemetrexed plus
carboplatin and bevacizumab followed by maintenance pemetrexed and
bevacizumab versus paclitaxel plus carboplatin and bevacizumab
followed by maintenance bevacizumab in patients with stage IIIB or
IV nonsquamous non-small-cell lung cancer. J Clin Oncol.
31:4349–4357. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yokoi T, Torii Y, Katashiba Y, Sugimoto H,
Tanijiri T, Ogata M, Inagaki N, Kibata K, Hayashi M, Niki M, et al:
Phase II study of pemetrexed and carboplatin plus bevacizumab,
followed by maintenance pemetrexed and bevacizumab in Japanese
patients with non-squamous non-small cell lung cancer. Oncol Lett.
8:2453–2457. 2014.PubMed/NCBI
|
|
15
|
Malhotra B, Evans T, Weiss J, Eaby B,
Stonehouse-Lee S, Sherry V and Langer CJ:
Carboplatin/pemetrexed/bevacizumab in the treatment of patients
with advanced non-small-cell lung cancer: A single-institution
experience. Clin Lung Cancer. 11:192–197. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nakamura H, Satoh H, Kaburagi T, Nishimura
Y, Shinohara Y, Inagaki M, Endo T, Saito T, Hayashihara K, Hizawa
N, et al: Bevacizumab-containing chemotherapy for non-small cell
lung cancer patients: A population-based observational study by the
Ibaraki thoracic integrative (POSITIVE) research group. Med Oncol.
29:3202–3206. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
NCCN: National Comprehensive Cancer
Network (NCCN) Clinical Practice Guidelines in Oncology, . Central
Nervous System Cancers. Version 2. 2014.https://www.nccn.org/professionals/physician_gls/PDF/cns.pdf
|
|
18
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
NIH, . Common Terminology Criteria for
Adverse Events (CTCAE) Version 4.0. simpleevs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf
|
|
20
|
Compton CC, Byrd DR, Garcia-Aguilar J,
Kurtzman SH, Olawaiye A and Washington MK: AJCC Cancer Staging
Atlas: A Companion to the Seventh Editions of the AJCC Cancer
Staging Manual and HandbookAJCC Cancer Staging Atlas: A Companion
to the Seventh Editions of the AJCC Cancer Staging Manual and
Handbook. 2nd. Springer; New York: 2012, View Article : Google Scholar
|
|
21
|
Parkin DM and Hakulinen T: Cancer
registration: Principles and methods. Analysis of survival. IARC
Sci Publ. 159–176. 1991.PubMed/NCBI
|
|
22
|
Ali A, Goffin JR, Arnold A and Ellis PM:
Survival of patients with non-small-cell lung cancer after a
diagnosis of brain metastases. Curr Oncol. 20:e300–e306. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Davis FG, Dolecek TA, McCarthy BJ and
Villano JL: Toward determining the lifetime occurrence of
metastatic brain tumors estimated from 2007 United States cancer
incidence data. Neuro Oncol. 14:1171–1177. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Patchell RA, Tibbs PA, Walsh JW, Dempsey
RJ, Maruyama Y, Kryscio RJ, Markesbery WR, Macdonald JS and Young
B: A randomized trial of surgery in the treatment of single
metastases to the brain. N Engl J Med. 322:494–500. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Louie AV, Rodrigues G, Yaremko B, Yu E,
Dar AR, Dingle B, Vincent M, Sanatani M, Younus J, Malthaner R and
Inculet R: Management and prognosis in synchronous solitary
resected brain metastasis from non-small-cell lung cancer. Clin
Lung Cancer. 10:174–179. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Schuette W: Treatment of brain metastases
from lung cancer: Chemotherapy. Lung Cancer. 45:(Suppl 2).
S253–S257. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zimmermann S, Dziadziuszko R and Peters S:
Indications and limitations of chemotherapy and targeted agents in
non-small cell lung cancer brain metastases. Cancer Treat Rev.
40:716–722. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
D'Antonio C, Passaro A, Gori B, Del
Signore E, Migliorino MR, Ricciardi S, Fulvi A and de Marinis F:
Bone and brain metastasis in lung cancer: Recent advances in
therapeutic strategies. Ther Adv Med Oncol. 6:101–114. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bailon O, Chouahnia K, Augier A, Bouillet
T, Billot S, Coman I, Ursu R, Belin C, Zelek L, Des Guetz G, et al:
Upfront association of carboplatin plus pemetrexed in patients with
brain metastases of lung adenocarcinoma. Neuro Oncol. 14:491–495.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Barlesi F, Gervais R, Lena H, Hureaux J,
Berard H, Paillotin D, Bota S, Monnet I, Chajara A and Robinet G:
Pemetrexed and cisplatin as first-line chemotherapy for advanced
non-small-cell lung cancer (NSCLC) with asymptomatic inoperable
brain metastases: A multicenter phase II trial (GFPC 07–01). Ann
Oncol. 22:2466–2470. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bearz A, Garassino I, Tiseo M, Caffo O,
Soto-Parra H, Boccalon M, Talamini R, Santoro A, Bartolotti M,
Murgia V, et al: Activity of pemetrexed on brain metastases from
non-small cell lung cancer. Lung Cancer. 68:264–268. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bernardo G, Cuzzoni Q, Strada MR, Bernardo
A, Brunetti G, Jedrychowska I, Pozzi U and Palumbo R: First-line
chemotherapy with vinorelbine, gemcitabine, and carboplatin in the
treatment of brain metastases from non-small-cell lung cancer: A
phase II study. Cancer Invest. 20:293–302. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Edelman MJ, Belani CP, Socinski MA, Ansari
RH, Obasaju CK, Chen R, Monberg MJ and Treat J: Alpha Oncology
Research Network: Outcomes associated with brain metastases in a
three-arm phase III trial of gemcitabine-containing regimens versus
paclitaxel plus carboplatin for advanced non-small cell lung
cancer. J Thorac Oncol. 5:110–116. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Besse B, Lasserre SF, Compton P, Huang J,
Augustus S and Rohr UP: Bevacizumab safety in patients with central
nervous system metastases. Clin Cancer Res. 16:269–278. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gordon MS, Margolin K, Talpaz M, Sledge GW
Jr, Holmgren E, Benjamin R, Stalter S, Shak S and Adelman D: Phase
I safety and pharmacokinetic study of recombinant human
anti-vascular endothelial growth factor in patients with advanced
cancer. J Clin Oncol. 19:843–850. 2001.PubMed/NCBI
|
|
36
|
Morgensztern D and Govindan R: Treatment
of patients excluded from Eastern Cooperative Oncology Group 4599
and AVAiL studies: Focus on brain metastasis and squamous
histology. Clin Lung Cancer. 9:(Suppl 2). S57–S61. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Oh Y and Stewart DJ: Systemic therapy for
lung cancer brain metastases: A rationale for clinical trials.
Oncology (Williston Park). 22:168–178; discussion 178, 183, 188
passim. 2008.PubMed/NCBI
|
|
38
|
Socinski MA, Langer CJ, Huang JE, Kolb MM,
Compton P, Wang L and Akerley W: Safety of bevacizumab in patients
with non-small-cell lung cancer and brain metastases. J Clin Oncol.
27:5255–5261. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lynch TJ Jr, Spigel DR, Brahmer J,
Fischbach N, Garst J, Jahanzeb M, Kumar P, Vidaver RM, Wozniak AJ,
Fish S, et al: Safety and effectiveness of bevacizumab-containing
treatment for non-small-cell lung cancer: Final results of the
ARIES observational cohort study. J Thorac Oncol. 9:1332–1339.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Crinò L, Dansin E, Garrido P, Griesinger
F, Laskin J, Pavlakis N, Stroiakovski D, Thatcher N, Tsai CM, Wu YL
and Zhou C: Safety and efficacy of first-line bevacizumab-based
therapy in advanced non-squamous non-small-cell lung cancer (SAiL,
MO19390): A phase 4 study. Lancet Oncol. 11:733–740. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Reck M, von Pawel J, Zatloukal P, Ramlau
R, Gorbounova V, Hirsh V, Leighl N, Mezger J, Archer V, Moore N and
Manegold C: Phase III trial of cisplatin plus gemcitabine with
either placebo or bevacizumab as first-line therapy for nonsquamous
non-small-cell lung cancer: AVAil. J Clin Oncol. 27:1227–1234.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
European Medicines Agency, . Committee for
Medicinal Products for Human Use Post-Authorisation Summary of
Positive Opinion for Avastin. Doc. Ref. No. EMEA/CHMP/121120/2009.
http://www.ema.europa.eu/docs/en_GB/document_library/Summary_of_opinion/human/000582/WC500059420.pdf
|
|
43
|
European Medicines Agency, . Assessment
Report for Avastin. Doc. Ref. No. EMEA/274499/2009. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Assessment_Report_-_Variation/human/000582/WC500029270.pdf
|