Introduction
Lung cancer is a malignant tumor characterized by
high morbidity and mortality rates, accounting for ~1.2 million
mortalities annually worldwide, which usually result from disease
recurrence and metastasis (1).
Although diagnostic methods and treatments have markedly improved
in recent years, the 5- and 10-year survival rates remain at <15
and <7%, respectively (2). At
present, the lack of appropriate molecular diagnostic tools to
predict the potential metastasis of lung cancer represents a major
clinical obstacle. Therefore, identification of biomarkers that
accurately predict future recurrence and metastasis of lung cancer
may improve treatment strategies.
Carboxypeptidase E (CPE) is a metal ion-dependent
exopeptidase that is predominantly expressed in endocrine and
nervous tissues, which converts the prohormones secreted by
endocrine or nerve cells, such as adrenocorticotropin/lipotropin
(ACTH/LPH), proinsulin, opiomelanocortin and enkephalin, into an
active form or into neuropeptides (3–5). Recent
studies have demonstrated that abnormal expression of CPE occurs in
epithelium-derived cancer tissues, including liver cancer, renal
clear cell carcinoma, colorectal cancer, cervical cancer and
melanoma (6–14). In 2011, a novel form of CPE,
N-terminal truncated carboxypeptidase E (CPEΔN), was identified in
hepatocellular carcinoma (HCC) (15,16).
Truncated CPE interacts with histone deacetylase (HDAC) 1 and HDAC2
to form a complex, which regulates the expression of
metastasis-associated proteins. Furthermore, it was reported that
CPEΔN was an independent predictor for recurrence and metastasis of
HCC. HCC patients with high CPEΔN expression levels exhibited
significantly higher recurrence rates in the 2 years following
surgery and lower median survival times than patients with low
CPEΔN expression levels (15).
Similar findings have been observed in primary
pheochromocytomas/paragangliomas (PHEO/PGL) and colorectal cancer
patients (17,18). Previous studies, which have used
quantitative polymerase chain reaction to analyze CPEΔN expression,
revealed that elevated CPEΔN expression is a statistically
significant predictor of poor prognosis (17,18). Since
HCC, PHEO/PGL and colorectal cancer are extremely different tumors
with distinctive tumor origins, these findings suggest that CPEΔN
may be a predictor of metastasis with a broad spectrum.
To evaluate the function of CPEΔN in lung
adenocarcinoma, in the present study, CPEΔN expression was analyzed
in lung adenocarcinoma tumors by western blot analysis and
immunohistochemistry. It has been demonstrated that CPEΔN
expression was associated with lymph node metastasis and distant
metastasis of lung adenocarcinoma, and that the three-year
tumor-free survival rates were significantly lower in patients with
CPEΔN expression than in those without CPEΔN expression. The
present study aimed to evaluate the potential of CPEΔN as a
biomarker for predicting future metastasis of lung
adenocarcinoma.
Materials and methods
Patients
A total of 95 lung adenocarcinoma patients who
underwent radical resection between January 2010 and June 2011 in
Liaoning Cancer Hospital and Institute (Shenyang, Liaoning, China)
were recruited for the current study. The patient cohort included
50 female and 45 male patients, with a mean age of 58.7 years
(range, 35–75 years). Of the 95 patients, 34, 30, 27 and 4 patients
were diagnosed with clinical stage I, II, III and IV disease,
respectively. None of the patients had received any treatment prior
to surgery. Patients at clinical stages II, III and IV received
adjuvant chemotherapy (docetaxel and cisplatin or docetaxel and
nedaplatin regimens) and/or immunotherapy after surgery. Follow-up
interviews were conducted every 3 months. Of the 100 patients
initially included, 3 were lost to follow-up and 2 were without
adequate protein samples for analysis. Therefore, 95 samples were
included in the final analysis. The date of
recurrence/identification of metastasis, data regarding
metastasis-affected organs and the date of patient morality were
recorded. The study was approved by the Ethics Committee of
Liaoning Cancer Hospital and Institute, and informed consent was
obtained from all participants.
Reagents
Monoclonal mouse anti-human CPE antibody was
purchased from BD Biosciences (Franklin Lakes, NJ, USA; cat. no.
610762). Total protein isolation kit (catalog no. WLA019) was
purchased from Wanlei Bio (Shenyang, China) and a lung
adenocarcinoma protein array (150-µm spots) was purchased from
Shanghai Xinchao Biotechnology (Shanghai, China).
Western blot analysis
Adenocarcinoma and pericarcinoma tissues (obtained
>5 cm away from the primary tumor) were maintained in a sterile
cryopreservation tube and stored in liquid nitrogen. All stored
samples were subject to western blot analysis within 1.5 years of
collection. Western blot analysis was performed to analyze the
expression of CPE and CPEΔN in adenocarcinoma and pericarcinoma
tissues, as previously described (19). Expression levels of CPEΔN were
quantified by grayscale scanning (CanoScanLiDE120; Canon, Inc.,
Tokyo, Japan) and analyzed with Gelpro32 (Media Cybernetics,
Rockville, MD, USA). No CPEΔN expression was defined as score ‘0’;
positive expression was defined as score ‘1’ (grayscale ratio of
CPEΔN; and actin, 0.5–1) and strong expression was defined as score
‘2’ (grayscale ratio of CPEΔN and actin, 1.0–1.5).
Immunohistochemistry
The immunohistochemistry assay was performed, as
previously described (20). The
expression levels and intracellular localization of CPE and CPEΔN
were determined by immunohistochemistry using a 150-µm spot lung
adenocarcinoma protein array. Based on immunohistochemical analysis
of patients with positive CPE expression, patients were classified
into two groups, nuclear CPE expression and cytoplasmic CPE
expression, to allow the comparison of overall survival rates
between the groups.
Statistical analysis
Data was analyzed using SPSS 17.0 (SPSS Inc.,
Chicago, IL, USA). The association between CPEΔN expression and T
stage, lymph node metastasis and distant metastasis was analyzed by
χ2 test. Disease-free survival and overall survival
curves were established using the Kaplan-Meier method and
intergroup comparisons were analyzed using the log-rank test.
Multivariate analyses of prognostic factors were performed using
the Cox regression model. P<0.05 was considered to indicate a
statistically significant difference.
Results
CPEΔN expression is higher in lung
adenocarcinoma tumor tissues than non-tumor tissues
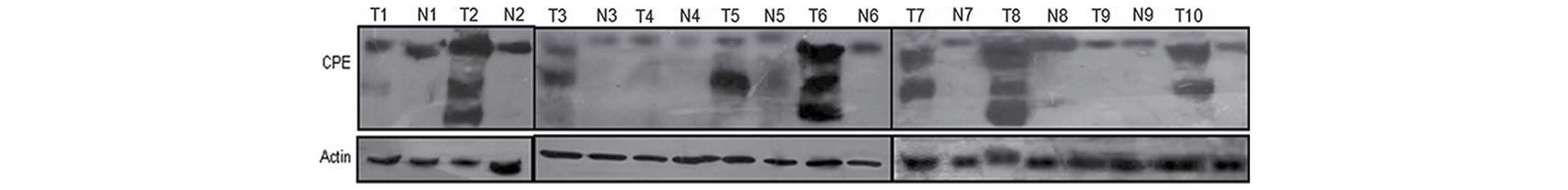
CPEΔN expression in 95 tumor and non-tumor tissues
was analyzed by western blotting. A total of 5 samples were
excluded due to minimal total amount of protein. The results
revealed that the co-expression rate of CPE and CPEΔN in lung
adenocarcinoma tissue was 62.1% (59/95). Full-length CPE was
expressed at similar levels in tumor and pericarcinoma normal
tissues from the same patients, and CPEΔN expression was identified
in only 5.26% (5/95) of non-tumor tissues (Fig. 1). Furthermore, the χ2 test
revealed that CPEΔN expression was closely associated with lymph
node metastasis (P=0.026) and distant metastasis (P=0.002),
however, no significant association was identified between CPEΔN
expression and age (P=0.555), gender (P=0.291) or T stage (P=0.109)
(Table I).
 | Table I.Correlation between CPEΔN protein
expression and clinicopathological features in 95 lung
adenocarcinoma patients. |
Table I.
Correlation between CPEΔN protein
expression and clinicopathological features in 95 lung
adenocarcinoma patients.
|
|
| CPEΔN expression |
|
|---|
|
|
|
|
|
|---|
| Parameter | Patients, n | (−), n | (+), n | P-value |
|---|
| Gender |
|
|
| 0.291 |
|
Female | 50 | 18 | 32 |
|
| Male | 45 | 21 | 24 |
|
| Age at diagnosis,
years |
|
|
| 0.555 |
| ≤55 | 30 | 11 | 19 |
|
|
>55 | 65 | 28 | 37 |
|
| T status |
|
|
| 0.109 |
| T1 | 3 | 1 | 2 |
|
| T2 | 68 | 31 | 37 |
|
| T3 | 20 | 6 | 14 |
|
| T4 | 4 | 1 | 3 |
|
| N status |
|
|
| 0.026 |
| N0 | 45 | 25 | 20 |
|
| N1 | 24 | 6 | 18 |
|
| N2 | 24 | 8 | 16 |
|
| NX | 2 | 0 | 2 |
|
| M status |
|
|
| 0.012 |
| M0 | 91 | 39 | 52 |
|
| M1 | 4 | 1 | 3 |
|
| Clinical stage |
|
|
| 0.061 |
| I | 34 | 19 | 15 |
|
| II | 30 | 11 | 19 |
|
| III | 27 | 9 | 18 |
|
| IV | 4 | 1 | 3 |
|
CPEΔN expression is associated with
lung adenocarcinoma recurrence and metastasis
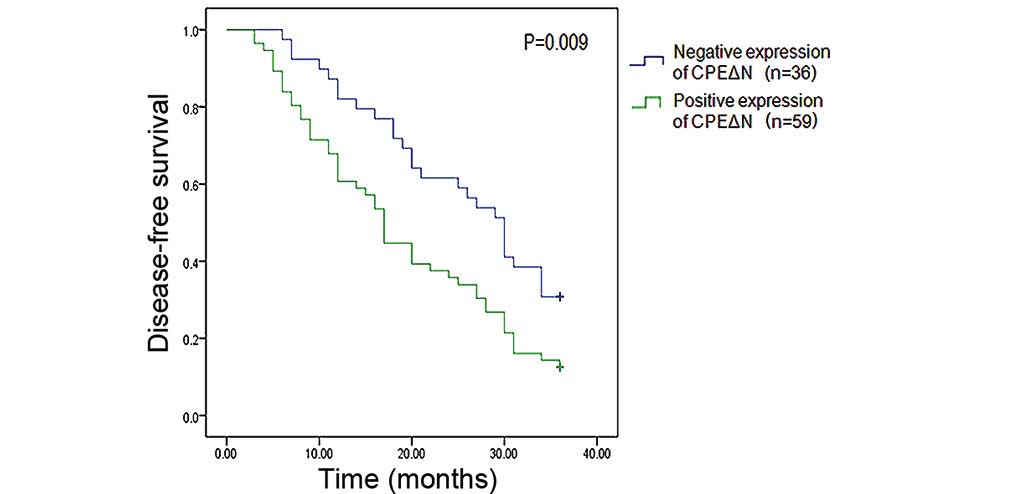
Of the 95 patients included in the study, 36
patients exhibited negative CPEΔN expression and 56 patients
exhibited positive CPEΔN expression. The negative and positive
CPEΔN patient groups were both followed up for 36 months. A
disease-free survival Kaplan-Meier curve was established, which
revealed that patients of the positive CPEΔN expression group
exhibited significantly higher postoperative recurrence and
metastasis rates (87.5%) when compared with patients of the
negative CPEΔN expression group (69.2%) (P=0.009) (Fig. 2). Subsequently, the 59 patients of the
positive CPEΔN expression group were divided into high CPEΔN
expression (immunohistochemistry score, 2) and low CPEΔN expression
immunohistochemistry score, 1) groups. The 2-year disease-free
survival rate of the low CPEΔN expression group (39.3%; 13/33) was
significantly higher than that of the high CPEΔN expression group
(11.5%; 3/26) (P=0.020; Table
II).
 | Table II.Correlation between CPEΔN protein
expression and tumor recurrence 2 years after surgery in 59 lung
adenocarcinoma patients. |
Table II.
Correlation between CPEΔN protein
expression and tumor recurrence 2 years after surgery in 59 lung
adenocarcinoma patients.
| Tumor
recurrence | Patients, n | High CPEΔN
expression, n | Low CPEΔN
expression, n | P-value |
|---|
| Yes | 43 | 23 | 20 | 0.020 |
| No | 16 | 3 | 13 |
|
Patients with predominant nuclear
expression of CPE exhibit a lower overall survival rate than those
with predominant cytoplasmic CPE expression
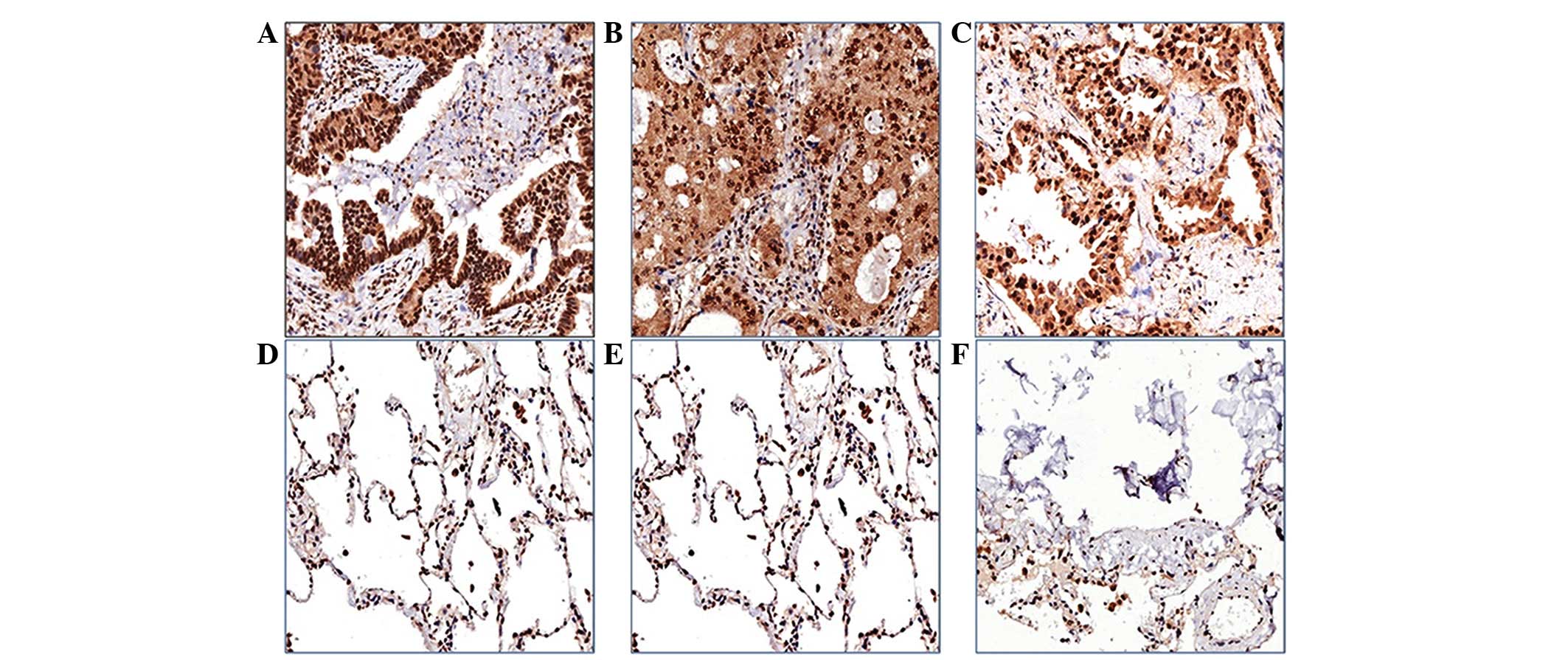
Immunohistochemistry was used to measure CPE
expression using a 150-µm spot protein array of lung
adenocarcinoma. The expression level of CPE in tumor tissue
appeared to be higher than that in the pericarcinoma normal tissues
from the same patients, and CPE was predominantly localized in the
cell nucleus or cytoplasm (Fig. 3).
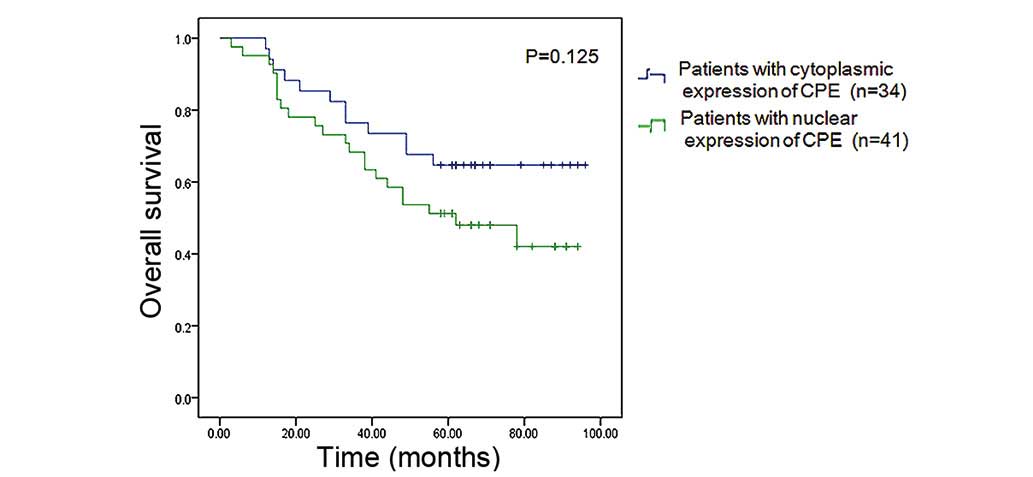
Patients were classified into two groups, a nuclear CPE expression
group (n=41) and a cytoplasmic CPE expression group (n=34), based
on where CPE was predominantly expressed in tumor cells. The
overall survival rate of patients with nuclear predominant nuclear
CPE expression was lower (46.3%) than that of patients with
predominant cytoplasmic CPE expression (64.7%), however, the
difference was not statistically significant (P=0.125) (Fig. 4).
CPEΔN expression is an independent
prognostic factor for recurrence and metastasis of lung
adenocarcinoma
To determine independent prognostic factors for lung
adenocarcinoma, a number of significant variables, including TNM
status (stages according to National Comprehensive Cancer Network
2010 guidelines) (21), tumor
recurrence, distant metastasis and CPEΔN expression were assessed
by multivariate regression analysis. Cox regression analysis
revealed that CPEΔN expression was an independent prognostic
biomarker for recurrence and metastasis of lung adenocarcinoma
(P=0.03; Table III).
 | Table III.Multivariate regression analysis of
prognostic markers in 95 patients with lung adenocarcinoma. |
Table III.
Multivariate regression analysis of
prognostic markers in 95 patients with lung adenocarcinoma.
| Clinicopathological
variable | β | χ2 | P-value | HR | 95% CI |
|---|
| TNM status | 0.424 | 5.195 | 0.023 | 1.527 | 1.061–2.198 |
| Tumor
recurrence | 0.342 | 8.104 | 0.004 | 1.407 | 1.112–1.780 |
| Distant
metastasis | 1.316 | 5.498 | 0.019 | 3.729 | 1.241–11.204 |
| CPEΔN
expression | 0.551 | 4.705 | 0.030 | 1.735 | 1.055–2.854 |
Discussion
CPE is a multifunctional protein that exhibits both
enzymatic and non-enzymatic functions. CPE exists in three
different forms, which are expressed in various subcellular
localizations with distinct functions (18). The first type is released by secretory
granules, which results in the cleavage of basic residues to
generate mature peptide hormones and neuropeptides. Recently,
Skalka et al (22)
demonstrated that this secreted form of CPE forms a complex with
the Wnt3a ligand and frizzled receptor to negatively regulate the
canonical Wnt signaling pathway (22). The second type of CPE exists in the
membrane, anchored at the trans-Golgi network, which functions as a
sorting receptor for prohormones (23). The third type of CPE is located in
both the cytoplasm and nucleus, and is involved in cell signal
transduction and transcription regulation. The function of this
type of CPE is the most diverse, however, further investigation is
required with regard to this protein.
In the present study, the association between CPEΔN
expression and prognosis of lung adenocarcinoma patients was
evaluated. The results revealed that two forms of CPE, full-length
CPE and CPEΔN, are co-expressed in the lung adenocarcinoma tissues.
CPEΔN expression was identified in the primary tumor tissues of
62.1% (59/95) patients and non-tumor tissues of 5.26% (5/95)
patients. Furthermore, multivariate Cox regression analysis
demonstrated that CPEΔN expression was closely associated with the
occurrence of lymph node and distant metastasis. Patients with
positive CPEΔN expression exhibited significantly lower
disease-free survival rates than patients with negative CPEΔN
expression (87.5 vs. 69.2%, respectively; P=0.009). These findings
suggested that CPEΔN expression indicates a higher risk of
recurrence and metastasis in primary lung adenocarcinoma.
Western blot analysis requires a large amount of
sample tissue, which is a major problem in clinical research.
Development of simplified methods for the analysis of CPEΔN or CPE
expression is required. In our previous studies (Sun et al,
unpublished data), the subcellular localization of CPEΔN was
investigated using confocal microscopy, which revealed that CPEΔN
was localized to the nucleus in H1299, A549, 95D, H1395 and Calu3
lung cancer cell lines, whereas full-length CPE was predominantly
localized in the cytoplasm. Therefore, it was hypothesized that
similar findings may be observed in primary lung adenocarcinoma.
The hypothesis was tested with tissue microarray. Since one array
can simultaneously test 75 samples along with corresponding
pericarcinoma tissues, 75 patients with intact overall survival and
progression-free survival profiles were selected. Patients were
divided into two groups, a nuclear CPE expression group and a
cytoplasmic CPE expression group. A difference in overall survival
rate was identified between the nuclear and cytoplasmic CPE
expression groups (46.3 vs. 64.7%, respectively); however, this was
not statistically significant (P=0.125). However, future studies
that include more lung adenocarcinoma patients and samples are
required to validate the results of the present study.
In conclusion, the results of the present study
demonstrated that CPEΔN expression is associated with poor
prognosis in lung adenocarcinoma. These findings may improve
understanding with regard to the underlying molecular mechanisms of
CPE/CPEΔN expression, which promote lung cancer metastasis. Thus,
the evaluation of CPEΔN expression status may aid to identify
primary lung adenocarcinoma patients that require more intensive
treatment. Furthermore, CPEΔN may present a potential target for
therapeutic intervention in the future.
Acknowledgements
This study was supported by the National Natural
Science Foundation of China (grant no. 81372287), Liaoning Province
Science and Technology Key Project (grant no. 2012225016) and
Shenyang City Science and Technology Key Project (grant no.
F12-193-9-15).
References
|
1
|
Hanahan D and Weinberg RA: The hallmarks
of cancer. Cell. 100:57–70. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Crinò L, Weder W, van Meerbeeck J and
Felip E: ESMO Guidelines Working Group: Early stage and locally
advanced (non-metastatic) non-small-cell lung cancer: ESMO clinical
practice guidelines for diagnosis, treatment and follow-up. Ann
Oncol. 21:(Suppl 5). v103–v115. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cool DR, Normant E, Shen F, Chen HC,
Pannell L, Zhang Y and Loh YP: Carboxypeptidase E is a regulated
secretory pathway sorting receptor: Genetic obliteration leads to
endocrine disorders in Cpe(fat) mice. Cell. 88:73–83. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cawley NX, Wetsel WC, Murthy SR, Park JJ,
Pacak K and Loh YP: New roles of carboxypeptidase E in endocrine
and neural function and cancer. Endocr Rev. 33:216–253. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Murthy SR, Pacak K and Loh YP:
Carboxypeptidase E: Elevated expression correlated with tumor
growth and metastasis in pheochromocytomas and other cancers. Cell
Mol Neurobiol. 30:1377–1381. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Coleman DL and Eicher EM: Fat (fat) and
tubby (tub): Two autosomal recessive mutations causing obesity
syndromes in the mouse. J Hered. 81:424–427. 1990.PubMed/NCBI
|
|
7
|
Bachtiary B, Boutros PC, Pintilie M, Shi
W, Bastianutto C, Li JH, Schwock J, Zhang W, Penn LZ, Jurisica I,
et al: Gene expression profiling in cervical cancer: An exploration
of intratumor heterogeneity. Clin Cancer Res. 12:5632–5640. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hong Y, Ho KS, Eu KW and Cheah PY: A
susceptibility gene set for early onset colorectal cancer that
integrates diverse signaling pathways: Implication for
tumorigenesis. Clin Cancer Res. 13:1107–1114. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cool DR and Loh YP: Carboxypeptidase E is
a sorting receptor for prohormones: Binding and kinetic studies.
Mol Cell Endocrinol. 139:7–13. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fricker LD and Snyder SH: Purification and
characterization of enkephalin convertase, an
enkephalin-synthesizing carboxypeptidase. J Biol Chem.
258:10950–10955. 1983.PubMed/NCBI
|
|
11
|
Tang SS, Zhang JH, Liu HX and Li HZ:
PC2/CPE-mediated pro-protein processing in tumor cells and its
differentiated cells or tissues. Mol Cell Endocrinol. 303:43–49.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
He P, Varticovski L, Bowman ED, Fukuoka J,
Welsh JA, Miura K, Jen J, Gabrielson E, Brambilla E, Travis WD and
Harris CC: Identification of carboxypeptidase E and gamma-glutamyl
hydrolase as biomarkers for pulmonary neuroendocrine tumors by cDNA
microarray. Hum Pathol. 35:1196–1209. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cutcliffe C, Kersey D, Huang CC, Zeng Y,
Walterhouse D and Perlman EJ: Renal Tumor Committee of the
Children's Oncology Group: Clear cell sarcoma of the kidney:
Up-regulation of neural markers with activation of the sonic
hedgehog and Akt pathways. Clin Cancer Res. 11:7986–7994. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Walboomers JM, Jacobs MV, Manos MM, Bosch
FX, Kummer JA, Shah KV, Snijders PJ, Peto J, Meijer CJ and Muñoz N:
Human papillomavirus is a necessary cause of invasive cervical
cancer worldwide. J Pathol. 189:12–19. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lee TK, Murthy SR, Cawley NX, Dhanvantari
S, Hewitt SM, Lou H, Lau T, Ma S, Huynh T, Wesley RA, et al: An
N-terminal truncated carboxypeptidase E splice isoform induces
tumor growth and is a biomarker for predicting future metastasis in
human cancers. J Clin Invest. 121:880–892. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mitka M: Researchers discover new
biomarker that may improve cancer care strategies. JAMA.
305:1184–1185. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhou K, Liang H, Liu Y, Yang C, Liu P and
Jiang X: Overexpression of CPE-ΔN predicts poor prognosis in
colorectal cancer patients. Tumour Biol. 34:3691–3699. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liang XH, Li LL, Wu GG, Xie YC, Zhang GX,
Chen W, Yang HF, Liu QL, Li WH, He WG, et al: Upregulation of CPE
promotes cell proliferation and tumorigenicity in colorectal
cancer. BMC Cancer. 13:4122013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sun J, Nie J, Hao B, Li L, Xing G, Wang Z,
Zhou Y, Sun Q, Li G, Zhang L and He F: Ceap/BLOS2 interacts with
BRD7 and selectively inhibits its transcription-suppressing effect
on cellular proliferation-associated genes. Cell Signal.
20:1151–1158. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tong ZT, Cai MY, Wang XG, Kong LL, Mai SJ,
Liu YH, Zhang HB, Liao YJ, Zheng F, Zhu W, et al: EZH2 supports
nasopharyngeal carcinoma cell aggressiveness by forming a
co-repressor complex with HDAC1/HDAC2 and Snail to inhibit
E-cadherin. Oncogene. 31:583–594. 2012.PubMed/NCBI
|
|
21
|
Jazieh AR, Bamefleh H, Demirkazik A,
Gaafar RM, Geara FB, Javaid M, Khader J, Khodadad K, Omar W,
Saadeddin A, et al: MENA Lung Cancer Regional Guidelines Committee:
Modification and implementation of NCCN guidelines on non-small
cell lung cancer in the Middle East and North Africa region. J Natl
Compr Canc Netw. 8:(Suppl 3). S16–S21. 2010.PubMed/NCBI
|
|
22
|
Skalka N, Caspi M, Caspi E, Loh YP and
Rosin-Arbesfeld R: Carboxypeptidase E: A negative regulator of the
canonical Wnt signaling pathway. Oncogene. 32:2836–2847. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen H, Jawahar S, Qian Y, Duong Q, Chan
G, Parker A, Meyer JM, Moore KJ, Chayen S, Gross DJ, et al:
Missense polymorphism in the human carboxypeptidase E gene alters
enzymatic activity. Hum Mutat. 18:120–131. 2001. View Article : Google Scholar : PubMed/NCBI
|