Introduction
Breast cancer (BC) is the most frequent malignancy
among women worldwide, with an estimated 14 million new cases and 8
million mortalities in 2012, which is projected to rise by at least
70% by 2030 (1). It is most common in
women aged >50 years (>75% of all cases), in contrast to
those aged ≤40 years, who represent only 5–25% of all BC cases.
Notably, BC in these younger women is generally considered to bear
a more unfavorable disease outcome, with a substantially shorter
overall survival as compared with older women (2,3).
Indeed, several multivariate analyses have shown
that young age is an independent predictor of unfavorable disease
outcome (4), and it has been
suggested to be associated with a more aggressive tumor biology
that potentially reduces the survival expectancy (5,6). This
increased aggressiveness of BC among younger women has been
attributed to mutations in the BC susceptibility proteins, breast
cancer (BRCA)1 and 2 (5,6). In addition, BC in younger women is often
diagnosed at more advanced stages than in older women. In addition,
BC tumors in younger women are: i) More frequently negative for
estrogen receptor (ER); ii) show extensive lymphovascular invasion
(LVI); iii) exhibit increased cell proliferation (assessed by the
Ki-67 marker); and v) demonstrate overexpression of p53 oncogene
(6).
Molecular profiling has suggested that race and
ethnicity may be other important factors associated with the
development of BC, particularly with its poor prognosis among
younger women, with those of African American origin being at the
highest risk of increased mortality (7). All of the aforementioned evidence
indicates that BC in younger women bears a worse disease outcome
compared with older women. This difference is only partially
explained by the fact that mammography screening and clinical
breast examinations are targeted to women aged >40 years
(8).
In Brazil, the incidence and mortality rates of BC
are relatively high (9,10). Compared with North America, where the
incidence of BC among younger women has plateaued (11), Brazil continues to present with
relatively high incidence rates among women aged 30–39 years
(12). Considering the increased BC
mortality among young women of African origin (7), it is of note that Brazil is a country
with a relatively high population of African descent, increasing
the importance of assessing the characteristics of BC among young
women in this country.
To the best of our knowledge, the present study
analyzed the largest cohort of patients with BC aged ≤40 years
(n=376) at a single institution to date, by comparing an extensive
set of clinicopathological characteristics and other variables with
a similarly sized cohort of BC patients aged 50–69 years (n=362).
The aim of the present study was to determine whether disease
outcome in younger women is poorer than that in the older women
and, if supported, to estimate the significant determinants of this
differential outcome in multivariate models.
Patients and methods
Study design
The present retrospective analytical study was based
on the hospital records of women with invasive BC (ICD-10: C50) who
were admitted to Barretos Cancer Hospital, Pio XII Foundation
(Barretos, Brazil) with no previous treatment between January 1985
and December 2002. During this 17-year period, a total of 4,134
women were examined. Of those, 1,735 women were eligible and
initially selected into the present study, according to the
inclusion and exclusion criteria. The patients were divided into
two groups: A younger group (≤40 years of age) and an older group
(50–69 years of age). The original cohort included 469 patients
aged ≤40 years and 1,266 patients aged 50–69 years. From this
original cohort, 400 women were selected from each group using a
random sample generator (SPSS software, version 20.0.0.1; IBM SPSS,
Armonk, NY, USA). Clinical treatment and follow-up data were
obtained from the patients' medical records. Patients without
follow-up information for >12 months were contacted by telephone
to update this information.
Inclusion and exclusion criteria
Patients that had undergone cancer treatment prior
to admission (1,290 patients), those aged 41–49 years of age (666
patients) and those aged ≥70 years (443 patients) were excluded
from the cohort. Finally, 24 women were excluded from the ≤40 years
age group and 38 women were excluded from the 50–69 years age group
due to a lack of clinical or treatment information, or due to a
non-invasive disease.
Sample size estimation
To calculate the sample size, the following
assumptions were made: 30–40% mortality (13,14), ±7.5%
estimation error, 5% type I error and 90% power. These conditions
indicated that between 360 and 400 patients in both groups would be
required for an adequately powered study. Accordingly, the final
analysis included 376 women aged ≤40 years and 362 women aged 50–69
years.
Definitions of BC characteristics
BC was defined as multi-centric or multi-focal
disease. Multi-centric was defined as a disease present in >1
quadrant of the breast, whereas multi-focal disease was defined as
a second tumor located at a distance of at least 5 cm from the
primary tumor. The tumors were also classified as synchronous or
metachronous. The criteria recommended by the National Cancer
Institute were used to assess comorbidities (15).
Immunohistochemistry (IHC) was routinely performed
using paraffin sections of the tumors to classify their patterns of
ER, progesterone receptor (PR) and C-erB-2 oncoprotein expression.
Tumors that were negative for all of these markers were considered
‘triple negative’ for IHC expression. Assessment of positive
reactions was performed according to the guidelines in common use
for ER and PR, and the scoring system proposed by Allred et
al was used to determine the proportion of stained nuclei and
the staining intensity (16).
Tumor staging was based on the criteria proposed by
the International Union Against Cancer and the American Joint
Committee on Cancer (17), and the
TNM staging system was used to evaluate the BC in three aspects:
Tumor size and extension (T), regional lymph node involvement (N),
and the presence of distant metastasis (M). Following the
determination of the T, N and M classifications, stage 0, I, II,
III or IV was assigned.
Statistical analysis
Statistical analyses were run using SPSS software
for Windows (version 20.0.0.1; IBM SPSS). Frequency tables used for
univariate analysis of overall survival (OS; the time from
diagnosis until mortality or date last seen alive) were based on
the Kaplan-Meier method, where stratum-specific outcomes were
compared using log-rank (Mantel-Cox) statistics. To adjust for
covariates, a Cox proportional hazards regression model was used,
and covariates (as listed separately) were entered in a stepwise
backwards manner using the default values of entry (P=0.05) and
removal (P=0.10). The variables that were significant in the
univariate analysis were included in the multivariate model. All of
the statistical tests were two-sided and P<0.05 indicated a
statistically significant difference.
Ethics
The present study was approved by the Research
Ethics Committee of Barretos Cancer Hospital, Pio XII
Foundation.
Results
The demographic characteristics of the patients in
the two series are presented in Table
I. The main clinical features are shown in Table II. The histopathological features and
treatment characteristics are shown in Tables III and IV, respectively.
 | Table I.Demographics of patients with breast
cancer stratified by age group. |
Table I.
Demographics of patients with breast
cancer stratified by age group.
|
| ≤40 years
(n=376) | 50-69 years
(n=362) |
|---|
|
|
|
|
|---|
| Variable | n | % | n | % | P-value |
|---|
| Period of
treatment |
|
|
1985–1990 | 59 | 15.7 | 65 | 18.0 | 0.078 |
|
1991–1996 | 108 | 28.7 | 78 | 21.5 |
|
|
1997–2002 | 208 | 55.3 | 217 | 59.9 |
|
|
N/A |
1 |
0.3 |
2 |
0.6 |
|
| Schooling,
years |
|
|
0–8 | 210 | 55.9 | 279 | 77.1 | <0.001 |
|
9–11 | 106 | 28.2 | 32 |
8.8 |
|
|
≥12 | 57 | 15.2 | 34 |
9.4 |
|
|
N/A |
3 |
0.8 | 17 |
4.7 |
|
| Overweight |
|
| No | 284 | 75.5 | 219 | 60.5 | <0.001 |
|
Yes | 44 | 11.7 | 90 | 24.9 |
|
|
N/A | 48 | 12.8 | 53 | 14.6 |
|
| Ethnicity |
|
|
White | 297 | 79.0 | 288 | 79.6 | 0.360 |
|
Non-white | 76 | 20.2 | 70 | 19.3 |
|
|
N/A |
3 |
0.8 |
4 |
1.1 |
|
| Marital status |
|
|
Married | 254 | 67.6 | 214 | 59.1 | <0.001 |
|
Single/divorced | 110 | 29.3 | 134 | 37.0 |
|
|
N/A | 12 |
3.2 | 14 |
3.9 |
|
 | Table II.Main clinical features of patients
with breast cancer stratified by age group. |
Table II.
Main clinical features of patients
with breast cancer stratified by age group.
|
| ≤40 years
(n=376) | 50-69 years
(n=362) |
|
|---|
|
|
|
|
|
|---|
| Clinical
feature | n | % | n | % | P-value |
|---|
| Palpable
nodules |
|
| No | 19 |
5.1 | 20 |
5.5 | 0.761 |
|
Yes | 356 | 94.7 | 338 | 93.4 |
|
|
N/A |
1 |
0.3 |
4 |
1.1 |
|
| Multicentric
cancer |
|
| No | 358 | 95.2 | 344 | 95.0 | 0.158 |
|
Yes | 11 |
2.9 |
5 |
1.4 |
|
|
N/A |
7 |
1.9 | 12 |
3.3 |
|
| Multifocal
cancer |
|
|
|
| 0.017 |
| No | 346 | 92.9 | 341 | 94.2 |
|
|
Yes | 23 |
6.1 |
9 |
2.5 |
|
|
N/A |
1 |
0.3 | 12 |
3.3 |
|
| Second cancer |
|
| No | 371 | 98.7 | 352 | 97.2 | 0.327 |
|
Yes |
4 |
1.1 |
7 |
1.9 |
|
|
N/A |
8 |
2.1 |
3 |
0.8 |
|
| Bilateral breast
cancer |
|
|
|
| 0.037 |
| No | 331 | 88.0 | 336 | 92.8 |
|
|
Yes | 37 |
9.8 | 21 |
5.8 |
|
|
N/A |
8 |
2.1 |
5 |
1.4 |
|
| Familial
history |
|
| No | 211 | 56.1 | 181 | 50.0 | 0.611 |
|
Yes | 49 | 13.0 | 36 |
9.9 |
|
| Other
malignancies | 74 | 19.7 | 53 | 14.6 |
|
|
N/A | 42 | 11.2 | 92 | 25.4 |
|
| Menopause at
diagnosis |
|
| No | 363 | 96.5 | 38 | 10.5 | <0.001 |
|
Yes |
7 |
1.9 | 226 | 62.4 |
|
|
N/A |
6 |
1.6 | 98 | 27.1 |
| Comorbidities |
|
|
|
| <0.001 |
|
Absent | 256 | 68.1 | 145 | 40.1 |
|
| 1 or 2
diseases | 114 | 30.8 | 175 | 48.3 |
|
|
N/A |
6 |
1.6 | 42 | 11.6 |
|
 | Table III.Histological features of patients
with breast cancer stratified by age group. |
Table III.
Histological features of patients
with breast cancer stratified by age group.
|
| ≤40 years
(n=376) | 50–69 years
(n=362) |
|
|---|
|
|
|
|
|
|---|
| Histological
feature | n | % | n | % | P-value |
|---|
| Histological
diagnosis |
|
| Ductal
carcinoma | 301 | 80.1 | 289 | 79.8 | 0.867 |
| Lobular
carcinoma | 59 | 15.7 | 54 | 14.9 |
|
|
Miscellaneous | 15 |
4.0 | 17 |
4.7 |
|
|
N/A |
1 |
0.3 |
2 |
0.6 |
|
| Tumor size
(mastectomy), cm |
|
| ≤2 | 54 | 14.4 | 74 | 20.4 | 0.185 |
|
>2 | 187 | 49.7 | 195 | 53.9 |
|
|
N/A | 135 | 35.9 | 93 | 25.7 |
|
| Ta |
|
|
TI/TII | 196 | 52.1 | 210 | 58.0 | 0.354 |
|
TIII/TIV | 84 | 22.3 | 106 | 29.3 |
|
|
N/A | 94 | 25.0 | 46 | 12.7 |
|
| Na |
|
|
Positive | 147 | 39.1 | 156 | 43.1 | 0.183 |
|
Negative | 197 | 52.4 | 170 | 47.0 |
|
|
N/A | 32 |
8.5 | 36 |
9.9 |
|
| Ma |
|
| No | 311 | 82.7 | 311 | 85.9 | 0.740 |
|
Yes | 38 | 10.1 | 35 |
9.7 |
|
|
N/A | 27 |
7.2 | 16 |
4.4 |
|
| Differentiation
degree |
|
|
I/II | 190 | 50.5 | 201 | 55.5 | 0.035 |
|
III | 87 | 23.1 | 60 | 16.6 |
|
|
N/A | 99 | 26.3 | 101 | 27.9 |
|
| Perineural
invasion |
|
| No | 241 | 64.1 | 84 | 23.2 |
|
|
Yes | 18 |
4.8 | 13 |
3.6 | 0.055 |
|
N/A | 117 | 31.1 | 276 | 76.8 |
|
| Blood vessel
invasion |
|
| No | 248 | 66.0 | 79 | 21.8 |
|
|
Yes |
4 |
1.1 |
7 |
1.9 | 0.007 |
|
N/A | 124 | 33.0 | 276 | 76.2 |
|
| Lymphovascular
invasion |
|
| No | 189 | 50.3 | 53 | 14.6 | <0.001 |
|
Yes | 80 | 21.3 | 81 | 22.4 |
|
|
N/A | 107 | 28.5 | 228 | 63.0 |
|
| Lymph node
invasion |
|
| N1
(1–3) | 78 | 20.7 | 67 | 18.5 | 0.535 |
| N2
(4–9) | 55 | 14.6 | 49 | 13.5 |
|
| N3
(≥10) | 42 | 11.2 | 35 |
9.7 |
|
|
N/A | 201 | 53.5 | 211 | 58.3 |
|
| Estrogen
receptor |
|
|
Negative | 89 | 23.7 | 67 | 18.5 | 0.014 |
|
Positive | 126 | 33.5 | 155 | 42.8 |
|
|
N/A | 161 | 42.8 | 140 | 38.7 |
|
| Progesterone
receptor |
|
|
Negative | 90 | 23.9 | 85 | 23.5 | 0.468 |
|
Positive | 124 | 33.0 | 135 | 37.3 |
|
|
N/A | 162 | 43.1 | 142 | 39.2 |
|
| C-erB-2
oncoprotein |
|
|
Negative | 117 | 31.3 | 125 | 34.5 | 0.032 |
|
Positive | 58 | 15.4 | 53 | 14.6 |
|
|
N/A | 201 | 53.5 | 184 | 50.8 |
|
| Triple
negative |
|
| No | 136 | 36.2 | 155 | 42.8 | 0.027 |
|
Yes | 38 | 10.1 | 23 |
6.4 |
|
|
N/A | 202 | 53.7 | 184 | 50.8 |
|
| Type of breast
surgery |
|
| Without
surgery | 37 |
9.8 | 24 |
6.6 | 0.078 |
|
Lumpectomy | 26 |
6.9 | 36 |
9.9 |
|
|
Quadrantectomy | 87 | 23.1 | 66 | 18.2 |
|
|
Mastectomy | 224 | 59.6 | 234 | 65.6 |
|
|
N/A |
2 |
0.5 |
2 |
0.6 |
|
| Axillary
surgery |
|
| No | 58 | 15.4 | 62 | 17.1 | 0.219 |
|
Yes | 316 | 84.0 | 298 | 82.3 |
|
|
N/A |
2 |
0.5 |
2 |
0.6 |
|
| Number of lymph
nodes dissected |
|
|
1–10 | 61 | 16.2 | 53 | 14.6 | 0.762 |
|
11–15 | 76 | 20.2 | 77 | 21.3 |
|
|
16–20 | 77 | 20.5 | 86 | 23.8 |
|
| ≥2 | 151 | 40.2 | 144 | 39.8 |
|
|
N/A | 11 |
2.9 |
2 |
0.6 |
|
 | Table IV.Treatment of patients with breast
cancer, stratified by age group. |
Table IV.
Treatment of patients with breast
cancer, stratified by age group.
|
| ≤40 years
(n=376) | 50–69 years
(n=362) |
|
|---|
|
|
|
|
|
|---|
| Treatment | n | % | n | % | P-value |
|---|
| Breast
reconstruction |
|
|
Without | 295 | 78.5 | 346 | 95.6 | <0.001 |
|
Immediate | 35 |
9.3 |
6 |
1.7 |
|
|
Late | 43 | 11.4 |
8 |
2.2 |
|
|
N/A |
3 |
0.8 |
2 |
0.6 |
|
| Neoadjuvant
chemotherapy |
|
| No | 312 | 83.0 | 286 | 79.0 | 0.254 |
|
Yes | 62 | 16.5 | 71 | 19.6 |
|
|
N/A |
2 |
0.5 |
5 |
1.4 |
|
| Chemotherapy |
|
| No | 52 | 13.8 | 70 | 19.3 | 0.053 |
| Yes
(adjuvant) | 201 | 53.5 | 196 | 54.1 |
|
| Yes
(palliative) | 120 | 31.9 | 92 | 25.4 |
|
|
N/A |
3 |
0.8 |
4 |
1.1 |
|
| Hormone
therapy |
|
| No | 206 | 54.8 | 176 | 48.6 |
|
| Yes
(adjuvant) | 104 | 27.7 | 131 | 36.2 | 0.056 |
| Yes
(palliative) | 59 | 15.7 | 51 | 14.1 |
|
|
N/A |
7 |
1.9 |
4 |
1.1 |
|
| Radiotherapy |
|
| No | 69 | 18.4 | 72 | 19.9 | 0.592 |
|
Yes | 302 | 80.3 | 284 | 78.5 |
|
|
N/A |
5 |
1.3 |
6 |
1.7 |
|
The two groups displayed significant differences in
various characteristics. The older group showed a higher proportion
of women with low educational level (up to 8 years of school) as
compared with the younger group (77.1 vs. 55.9%, respectively;
P<0.001) (Table I).
Table II shows that
the groups had no differences in the presence of nodules or
multicentric cancer at the first examination. The younger group had
a higher proportion of multi-focal cancer and bilateral cancer as
compared with the older group (6.1 vs. 2.5%, P=0.017 and 9.8 vs.
5.8%, P=0.037, respectively). Comorbidity was less frequent in the
younger group than in the older group; one or two comorbidities
were found in 30.3 vs. 48.3% of patients (P<0.001), respectively
(Table II).
As indicated in Table
III, no significant differences were identified between the
groups in terms of histological types of the cancer or disease
staging. The tumors in the younger group showed a greater frequency
of a low degree of differentiation (23.1 vs. 16.6%; P=0.035),
reduced blood vessel invasion (1.1 vs. 1.9%; P=0.003), reduced LVI
(21.3 vs. 22.4%; P<0.001) and reduced perineural invasion (4.8
vs. 3.6%; P=0.055) as compared with the lesions in the older group
(Table III).
The proportion of tumors that were positive for ER
expression was significantly lower in the younger group than in the
older group (33.5 vs. 42.8%; P=0.014). The two groups had similar
negative rates of PR and C-erB-2 expression (Table III). Tumors classified as triple
negative were more frequent among the younger women (10.1 vs. 6.4%;
P=0.027) (Table III).
The two groups were similar in terms of the number
of dissected lymph nodes. Although the younger group underwent
radical mastectomy less frequently than the older group, the
difference was not statistically significant (59.6 vs. 64.6%,
respectively; P=0.078) (Table III).
There was a higher proportion of breast reconstruction in the
younger group as compared with the older group (20.7 vs. 3.9%;
P<0.001) (Table IV). Furthermore,
the groups had undergone similar neo-adjuvant treatments Table IV); adjuvant hormone therapy was used
less frequently in the younger than in the older group, but this
difference was only of borderline significance (27.7 vs. 36.2%;
P=0.056) (Table IV).
The duration of follow-up ranged between 0 and 22.6
years, with a mean of 6.1 years and 8.5 months when mortality was
excluded. The follow-up data were similar in the two groups
(P=0.816). After 5 and 10 years, 4.9 and 10.5% of the patients,
respectively, were lost to follow-up (data not shown). The OS rates
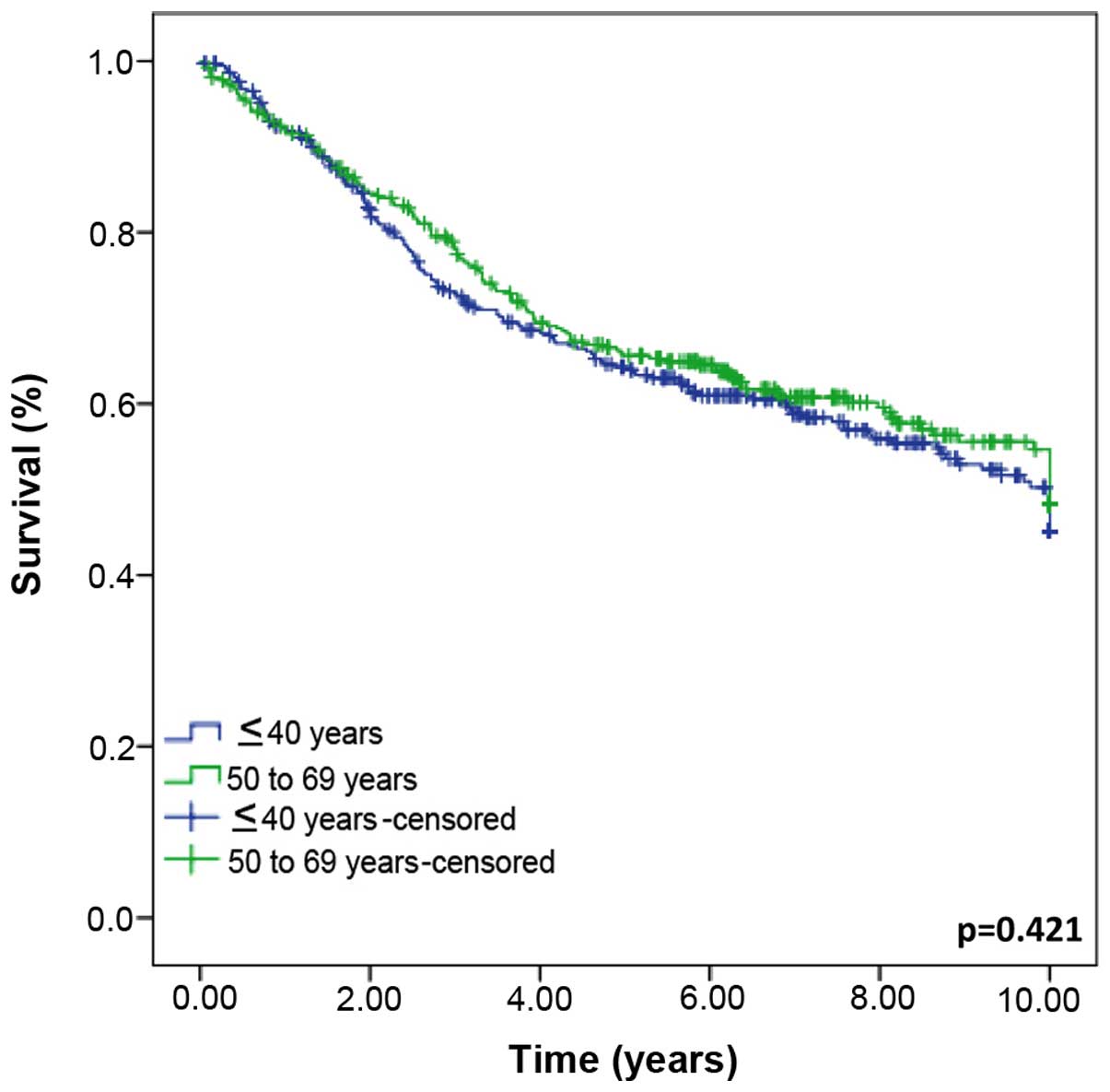
were not significantly different between the groups (P=0.421)
(Fig. 1).
Univariate analysis showed that the following
characteristics were significantly associated with prognosis in the
younger group: Second cancer (P=0.004), bilateral BC (P=0.100),
familial history (P=0.086), menopause at diagnosis (P=0.026),
associated diseases (P=0.077), tumor size (mastectomy) (P=0.075),
T, N and M stage (P<0.001), degree of differentiation (P=0.127),
perineural invasion (P=0.056), LVI (P=0.004), lymph node invasion
(P<0.001), ER status (P=0.002), PR status (P=0.043), type of
breast surgery (P<0.001), axillary surgery (P<0.001), number
of lymph nodes dissected (P<0.001), breast reconstruction
(P<0.001), neoadjuvant chemotherapy (P<0.028), chemotherapy
(P<0.001) and hormone therapy (P<0.001) (Tables V–VIII).
 | Table V.DSS according to the demographics of
patients with breast cancer, stratified by age group. |
Table V.
DSS according to the demographics of
patients with breast cancer, stratified by age group.
|
| ≤40 years
(n=376) | 50–69 years
(n=362) |
|---|
|
|
|
|
|---|
| Variable | n | 10-year DSS, % | P-value | n | 10-year DSS, % | P-value |
|---|
| Period of
treatment |
|
|
1985–1990 | 59 | 46.1 | 0.656 | 65 | 33.3 | 0.002 |
|
1991–1996 | 108 | 46.4 |
| 78 | 42.0 |
|
|
1997–2002 | 208 | 40.7 |
| 217 | 60.0 |
|
|
N/A |
1 |
|
|
2 |
|
|
| Schooling,
years |
|
|
0–8 | 210 | 42.5 | 0.751 | 279 | 50.0 | 0.551 |
|
9–11 | 106 | 48.5 |
| 32 | 44.6 |
|
|
≥12 | 57 | 47.5 |
| 34 | 38.5 |
|
|
N/A |
3 |
|
| 17 |
|
|
| Overweight |
|
| No | 284 | 48.1 | 0.624 | 219 | 47.5 | 0.978 |
|
Yes | 44 | 34.1 |
| 90 | 44.9 |
|
|
N/A | 48 |
|
| 53 |
|
|
| Ethnicity |
|
|
White | 297 | 46.0 | 0.534 | 288 | 50.1 | 0.129 |
|
Non-white | 76 | 42.7 |
| 70 | 41.0 |
|
|
N/A |
3 |
|
|
4 |
|
|
| Marital status |
|
|
Married | 254 | 44.2 | 0.538 | 214 | 52.2 | 0.170 |
|
Single/divorced | 110 | 49.5 |
| 134 | 42.6 |
|
|
N/A | 12 |
|
| 14 |
|
|
 | Table VIII.DSS according to the treatment of
patients with breast cancer, stratified by age group. |
Table VIII.
DSS according to the treatment of
patients with breast cancer, stratified by age group.
|
| ≤40 years
(n=376) | 50–69 years
(n=362) |
|---|
|
|
|
|
|---|
| Treatment | n | 10-year DSS, % | P-value | n | 10-year DSS, % | P-value |
|---|
| Breast
reconstruction |
|
|
Without | 295 | 41.0 | <0.001 | 346 | 47.7 | 0.263 |
|
Immediate | 35 | 43.3 |
|
6 | – |
|
|
Late | 43 | 75.7 |
|
8 | 83.3 |
|
|
N/A |
3 |
|
|
2 |
|
|
| Neoadjuvant
chemotherapy |
|
| No | 312 | 48.4 | 0.028 | 286 | 51.0 | 0.010 |
|
Yes | 62 | 28.2 |
| 71 | 37.3 |
|
|
N/A |
2 |
|
|
5 |
|
|
| Chemotherapy |
|
| No | 52 | 65.5 | <0.001 | 70 | 49.6 | <0.001 |
|
Yes-adjuvant | 201 | 60.9 |
| 196 | 62.5 |
|
|
Yes-palliative | 120 | 14.0 |
| 92 | 19.0 |
|
|
N/A |
3 |
|
|
4 |
|
|
| Hormone
therapy |
|
| No | 206 | 51.8 | 0.001 | 176 | 52.5 | <0.001 |
|
Yes-adjuvant | 104 | 50.3 |
| 131 | 53.2 |
|
|
Yes-palliative | 59 | 10.8 |
| 51 | 11.2 |
|
|
N/A |
7 |
|
|
4 |
|
|
| Radiotherapy |
|
| No | 69 | 50.5 | 0.935 | 72 | 36.7 | 0.016 |
|
Yes | 302 | 43.2 |
| 284 | 52.9 |
|
|
N/A |
5 |
|
|
6 |
|
|
In the older group, the following variables were
significantly associated with prognosis: Period of treatment
(P=0.002), ethnicity (P=0.129), marital status (P=0.170), palpable
nodules (P=0.062), tumor size (mastectomy) (P=0.042), T, N and M
stage (P<0.001), degree of differentiation (P<0.001),
peri-neural invasion (P=0.095), blood vessel invasion(P<0.001),
LVI (P=0.003), lymph node invasion (P<0.001), ER status
(P=0.001), PR status (P=0.015), C-erB-2 expression (P=0.006), type
of breast surgery (P<0.001), axillary surgery (P<0.001),
number of lymph nodes dissected (P=0.005), neo-adjuvant
chemotherapy (P=0.010), chemotherapy (P<0.001), hormone therapy
(P<0.001), and radiotherapy (P=0.016). None of the other
variables were statistically significant predictors of survival
(Tables V–VIII).
When stratified by clinical stage, no significant
differences in the stage-specific survival rates between the two
age groups were found (P=0.421) (Fig.
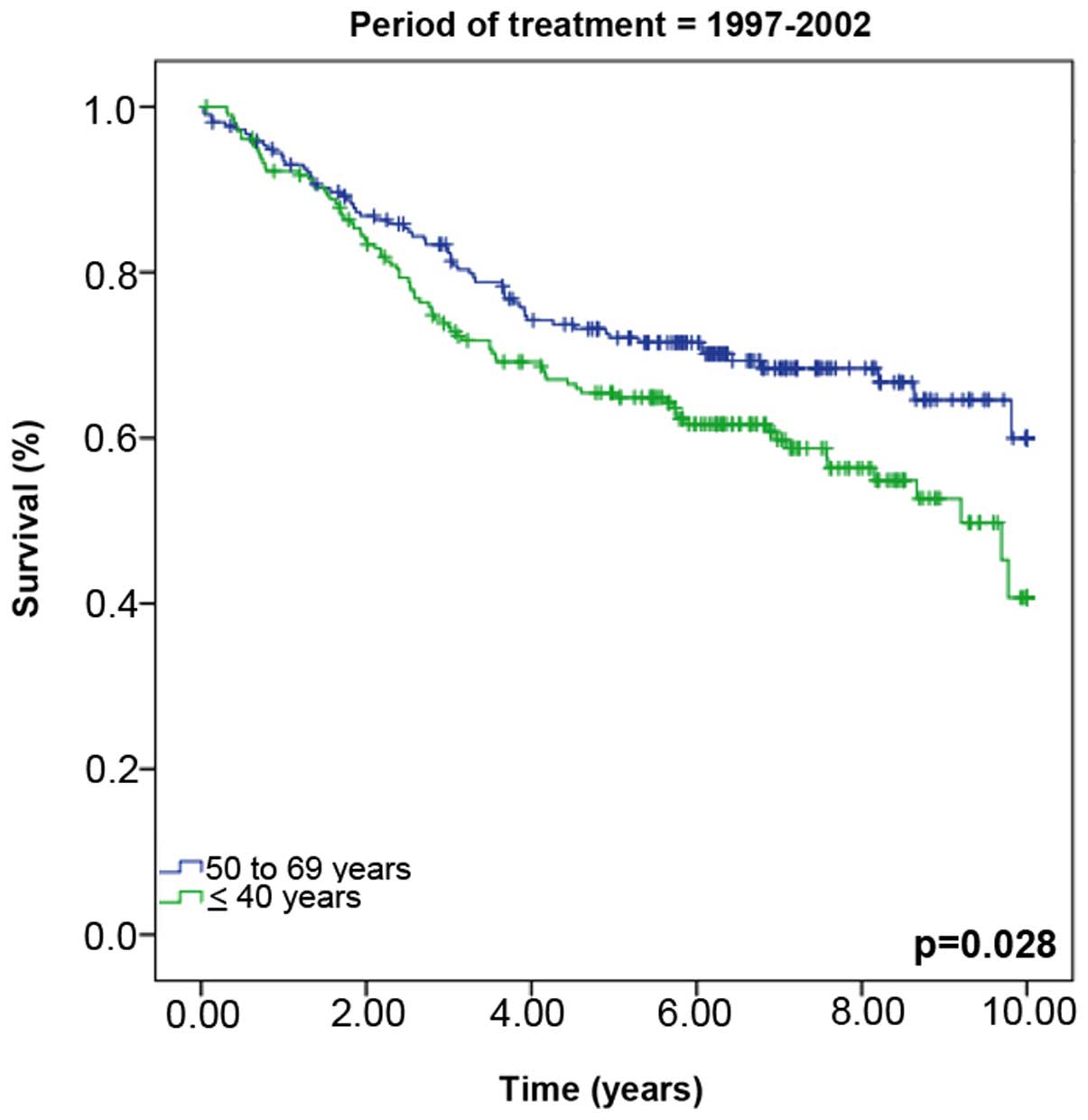
1). However, when the groups were stratified by the period of
treatment, significant differences in survival rates were observed
between the two groups during the treatment period between 1997 and
2002 (young vs. old group, 40.7 vs. 60.0% survival, P=0.028)
(Fig. 2).
In the multivariate analysis, second cancer [hazard
ratio (HR), 2.20; 95% confidence interval (CI), 1.500–29.044;
P=0.013] and bilateral BC (HR, 6.60; 95% CI, 1.228–3.930; P=0.008)
were independent predictors of a poor outcome, with an increased
risk of mortality in the younger group. C-erB-2 (HR, 1.97; 95%; CI,
1.003–3.869; P=0.049) was a positive predictor of a poor outcome in
the older group, showing an increased risk of mortality. The ER
status and TNM stage were significantly associated with disease
prognosis in both groups (Tables IX
and X).
 | Table IX.Multivariate survival analysis of
patients aged ≤40 years (n=178). |
Table IX.
Multivariate survival analysis of
patients aged ≤40 years (n=178).
| Variable | n | Hazard ratio | 95% confidence
interval | P-value |
|---|
| Bilateral breast
cancer |
|
| No | 158 | – | – | – |
|
Yes | 20 | 2.20 | 1.228–3.930 | 0.008 |
| Second cancer |
|
| No | 176 | – | – | – |
|
Yes |
2 | 6.60 | 1.500–29.044 | 0.013 |
| Estrogen
receptor |
|
|
Positive | 106 | – | – | – |
|
Negative | 72 | 2.29 | 1.389–3.777 | 0.001 |
| T |
|
|
I/II | 133 | – | – | – |
|
III/IV | 45 | 2.14 | 1.258–3.640 | 0.005 |
| N |
|
| No | 80 | – | – | – |
|
Yes | 98 | 2.66 | 1.531–4.621 | 0.001 |
| M |
|
| No | 173 | – | – | – |
|
Yes |
5 | 5.92 | 2.172–16.151 | 0.001 |
 | Table X.Multivariate survival analysis of
patients aged 50–69 years (n=151). |
Table X.
Multivariate survival analysis of
patients aged 50–69 years (n=151).
| Variable | n | Hazard ratio | 95% confidence
interval | P-value |
|---|
| Estrogen
receptor |
|
|
Positive | 44 | – | – | – |
|
Negative | 107 | 2.34 | 1.175–4.663 | 0.016 |
| C-erB-2
oncoprotein |
|
|
Negative | 108 | – | – | – |
|
Positive | 43 | 1.97 | 1.003–3.869 | 0.049 |
| Ta |
|
|
I/II | 110 | – | – | – |
|
III/IV | 41 | 2.12 | 1.073–4.180 | 0.030 |
| Na |
|
| No | 71 | – | – | – |
|
Yes | 80 | 2.93 | 1.394–6.149 | 0.005 |
| Ma |
|
| No | 150 | – | – | – |
|
Yes |
1 | 9.99 | 1.049–95.105 | 0.045 |
Discussion
The results described herein reveal important
differences between the younger and older patients with BC. This is
an almost unanimously accepted fact among BC investigators.
Notably, however, the present study was unable to confirm the most
controversial of these issues, the suggested worse survival rate of
young patients with BC, and failed to identify any difference in
disease outcome between the two study groups. This finding is of
major importance, asthe OS (Kaplan-Meier) did not reveal
significant differences between the two groups, despite several
parameters indicating that a more aggressive tumor biology is
confined to the younger patients. Only when calculated for the
treatment period 1997–2002 were the OS rates significantly worse
among young women. We propose that there are several explanations
for this finding, with insights discussed herein.
Whether there are clinicopathological differences in
BC between younger and older women remains a controversial issue.
Typically, the young BC age group ranges from 35 (13,18,19) to 40
(2,14,20,21),
however, there is no clear consensus supporting these strict age
limits for young and old. However, BC is more frequent in women
aged >50 years old. In a population-derived cohort from Brazil,
the prevalence of BC was found to be 14% (12), 4.5% of these patients were aged ≤35
years (3) and 26.4% were aged ≤40
years (2).
Thus, in the present study, a 40-year cut-off was
used to stratify the women as younger or older. This criterion was
partially supported by epidemiological data demonstrating different
tumor biology in different age groups (14,22). It is
also important to emphasize that in Europe, regular mammography
screening is a standard practice for women aged 50–69 years. This
group is considered to be a biologically distinct group at high
risk of developing BC with particular radiological characteristics
(23). For this reason, women aged
50–69 years old were selected for comparison in the present study.
As BC studies have different definitions regarding age groups, it
is challenging to provide a reasonable comparison between different
studies.
A number of reports also disagree regarding the
importance of including different types of BC, for example,
carcinoma in situ (CIS) (13,21) or
metastatic disease (2,24). Other studies disagree regarding the
pertinence of including patients with clinical stage I and II
(25,26) or clinical stages I–III (2,20,24) cancer. The present study included TNM
stage, and excluded cases of CIS due to their different
histological features (27) and
relatively favorable disease outcome.
The present study is based on robust parameters that
were selected to reflect the true significance of the behavior of
BC in young women. To the best of our knowledge, the current study
includes the largest cohort of patients with BC aged ≤40 years
(n=376) reported from a single institution to date. Worldwide, few
institutions have studied larger samples (24,28) and
the majority of large studies have been based on cancer registries
(7,14,21).
The present study included a large number of
patients with invasive BC presenting at advanced clinical stages.
These features are not unusual and are inherent to the existing
healthcare system in Brazil. Brazilian women have limited access to
clinics that provide mammography examinations and organized
population-based screening programs do not exist; consequently, the
majority of the BC cases in Brazil are detected at advanced stages.
This is in contrast to the situation in developed countries, where
regular mammography screening enables the detection of the majority
of cases of BC at an early stage (14,19,21).
Indeed, prior to 2002, there was no formal recommendation from the
Brazilian Health Authorities to provide mammography examinations in
asymptomatic women. Therefore, as the presence of palpable and/or
ulcerated lesions were a requirement for medical care, a
substantial proportion of women exhibited these signs at their
first examination. These characteristics are an inherent bias of
the present study design resulting in a cohort enriched by BC
patients with aggressive tumors at baseline. However, the overall
relative survival rate of the older women (50–69 years old)
improved between 1997 and 2002, attributable to improvements in
treatment, such as chemotherapy and hormone therapy (29).
Mammography is undoubtedly an important diagnostic
tool for non-palpable lesions, despite sensitivity limitations
among women aged ≤40 years. It is speculated that regular programs
for BC prevention could minimize the number of cases of advanced
stage disease in Brazil (30).
Hereditary BC associated with BRCA1/2 mutations have a prevalence
of ~2.3% (31) in Brazil.
Furthermore, studies of Latin American women have demonstrated that
a family history of BC is less prevalent in Latin America than in
the North American population (32).
In the present study, mutations were not investigated, however, the
familial history of BC was similar between the two study groups.
However, the presence of second cancer was similar in the younger
and older groups; this is consistent with the results of a previous
study, where it was attributed to longer exposures to pathogenic
factors (33).
The prevalence of bilateral disease was higher in
the younger age group and is one of the indicators of more
aggressive tumors, for example tumors with T stage II/III, grade
III differentiation, negative ER/PR status and positive lymph node
invasion are more aggressive (33).
Conversely, the older women more frequently presented with other
diseases associated with their BC, which can have an adverse
influence on clinical outcome and contribute to a poor prognosis
(34). However, the present results
identified indicators of worse prognosis to be more common among
the younger women.
Accordingly, younger women presented with a higher
proportion of tumors with grade III differentiation (poorly
differentiated), as reported in a previous study (33). Similarly, the present study observed
significant differences between the two groups in terms of
perivascular blood vessel invasion and LVI, with older women
showing higher proportions of both types of invasion. Notably, this
result conflicts with the previously reported data (35,36).
The present study also observed that a higher
proportion of older women were positive for ER compared with
younger women. This finding is consistent with the results of a
previous study, in which advanced age was correlated with positive
ER status and vice versa (36). This
observation also agrees with the disease outcome data of the
current cohort, as a negative ER status is typically associated
with a more unfavorable prognosis. By contrast, PR expression did
not significantly differ between the two age groups, in agreement
with a previous report (26).
Conversely, a previous study observed that a negative PR status
occurs more frequently in the BC of younger women (35). However, the prognostic value of PR was
visible only as a part of an algorithm, but not on its own
(37). The present findings also
revealed that triple negative BC, which are considered to predict a
poor prognosis, were more common in younger women, as previously
reported (13).
As anticipated, more important differences in
factors that usually influence the efficacy of treatment were
observed between the two groups, including tumor stage at the time
of diagnosis, tumor characteristics and comorbidities. Of note, the
present study included patients with follow-up times of up to 22
years (mean, 6.75 years). This is another important feature of the
present study, as only a small number of studies have reported
long-term follow-up data of 10 (20,25,36,38)
or 15 years (2,14,24,28). In
addition, BC treatment strategies have undergone rapid developments
in the past two decades, and this development may have contributed
to the prognostic differences between the two groups included in
the current study. In the younger group, fewer patients received
adjuvant hormone therapy and more women received palliative
chemotherapy when compared with the older group. This is a clear
limitation inherent to all retrospective studies where active
intervention is no longer possible. However, the current
observations clearly support proposals to also initiate monitoring
programs for long-term BC patients.
Finally, the present results did not show
significant differences in survival rates between the two study
groups (Fig. 1). By contrast, a
previous study conducted in central Brazil demonstrated a
significant difference in survival rates between patients aged ≤40
years and those aged 40–50 years (P=0.0002) or those aged >50
years (P=0.03). However, this study excluded clinical stage IV
disease and included far fewer patients than the present study
(20). A previous study reported that
BC mortality remains high among young patients, with an OS of ~60%
over 5 years, compared with 85% among women aged >50 years
(20). The equal survival rates
between young and old women observed in the present study have been
reported in previous studies (38,39),
however, in general, the results have been highly variable among
the published literature (40–43).
In conclusion, BC among young women is generally
considered to be more aggressive and associated with a worse
prognosis compared with BC in older women, whereas the prognostic
value of age itself remains more controversial. The results of the
present study suggest that BC in young women is associated with
numerous pathological features of aggressiveness, including tumor
grade, hormone receptor status and degree of differentiation,
having been implicated as independent determinants of BC prognosis.
However, the OS rates of the two age groups proved to be very
similar, except when the data were stratified by the treatment
period. For women treated during 1997–2002, the 10-year survival
rate of the older women was ~60% compared with 40.7% in the younger
cohort. The current study concludes that this increase in 10-year
survival is due largely to improvements in treatment, increased
early diagnosis and certain tumor characteristics, with the
patient's age itself not being an independent prognostic factor.
The present study indicates that medical advances associated with
prevention of breast cancer may improve screening programs, which
may therefore increase early diagnosis and subsequently lower
mortality rates.
Acknowledgements
The authors thank the Public Ministry of Labor
(Research, Prevention and Education of Occupational Cancer) in
Campinas, Brazil, and the Lions Club of Brazil for partial
financial support of the present study and Dr. Vinicius de Lima
Vazquez (Department of Skin cancer and Melanoma, Barretos Cancer
Hospital, Pio XII Foundation, Barretos, Brazil) for assistance with
the statistical analysis. The abstract was previously published in
The Breast 23 (Suppl): S11, 2014.
References
|
1
|
Antoni S, Soerjomataram I, Møller B, Bray
F and Ferlay J: An assessment of GLOBOCAN methods for deriving
national estimates of cancer incidence. Bull World Health Organ.
94:174–184. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Elkum N, Dermime S, Ajarim D, Al-Zahrani
A, Alsayed A, Tulbah A, Al Malik O, Alshabanah M, Ezzat A and
Al-Tweigeri T: Being 40 or younger is an independent risk factor
for relapse in operable breast cancer patients: The Saudi Arabia
experience. BMC Cancer. 7:2222007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fredholm H, Eaker S, Frisell J, Holmberg
L, Fredriksson I and Lindman H: Breast cancer in young women: Poor
survival despite intensive treatment. PLoS One. 4:e76952009.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Anders CK, Johnson R, Litton J, Phillips M
and Bleyer A: Breast cancer before age 40 years. Semin Oncol.
36:237–249. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Musolino A, Bella MA, Bortesi B, Michiara
M, Naldi N, Zanelli P, Capelletti M, Pezzuolo D, Camisa R, Savi M,
et al: BRCA mutations, molecular markers, and clinical variables in
early-onset breast cancer: A population-based study. Breast.
16:280–292. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Klauber-DeMore N: Tumor biology of breast
cancer in young women. Breast Dis. 23:9–15. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Smigal C, Jemal A, Ward E, Cokkinides V,
Smith R, Howe HL and Thun M: Trends in breast cancer by race and
ethnicity: Update 2006. CA Cancer J Clin. 56:168–183. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Samphao S, Wheeler AJ, Rafferty E,
Michaelson JS, Specht MC, Gadd MA, Hughes KS and Smith BL:
Diagnosis of breast cancer in women age 40 and younger: Delays in
diagnosis result from underuse of genetic testing and breast
imaging. Am J Surg. 198:538–543. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mauad EC, Nicolau SM, Moreira LF, Haikel
RL Jr, Longatto-Filho A and Baracat EC: Adherence to cervical and
breast cancer programs is crucial to improving screening
performance. Rural Remote Health. 9:12412009.PubMed/NCBI
|
|
10
|
Milani V, Goldman SM, Finguerman F,
Pinotti M, Ribeiro CS, Abdalla N and Szejnfeld J: Presumed
prevalence analysis on suspected and highly suspected breast cancer
lesions in São Paulo using BIRADS criteria. Sao Paulo Med J.
125:210–214. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tarone RE: Breast cancer trends among
young women in the United States. Epidemiology. 17:588–590. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Freitas-Junior R, Freitas NM, Curado MP,
Martins E, Moreira MA and e Silva CM: Variations in breast cancer
incidence per decade of life (Goiânia, GO, Brazil): 16-year
analysis. Cancer Causes Control. 19:681–687. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Dutra MC, Rezende MA, de Andrade VP,
Soares FA, Ribeiro MV, de Paula EC and Gobbi H: Immunophenotype and
evolution of breast carcinomas: A comparison between very young and
postmenopausal women. Rev Bras Ginecol Obstet. 31:54–60. 2009.(In
Portuguese). PubMed/NCBI
|
|
14
|
Gnerlich JL, Deshpande AD, Jeffe DB, Sweet
A, White N and Margenthaler JA: Elevated breast cancer mortality in
women younger than age 40 years compared with older women is
attributed to poorer survival in early-stage disease. J Am Coll
Surg. 208:341–347. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yancik R, Havlik RJ, Wesley MN, Ries L,
Long S, Rossi WK and Edwards BK: Cancer and comorbidity in older
patients: A descriptive profile. Ann Epidemiol. 6:399–412. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Allred DC, Harvey JM, Berardo M and Clark
GM: Prognostic and predictive factors in breast cancer by
immunohistochemical analysis. Mod Pathol. 11:155–168.
1998.PubMed/NCBI
|
|
17
|
Greene FL, Page DL, Fleming ID, Fritz AG,
Balch CM, Haller DG and Morrow M: AJCC Cancer Staging Manual. 6th.
Springer-Verlag; New York: pp. 227–229. 2002
|
|
18
|
Rapiti E, Fioretta G, Verkooijen HM,
Vlastos G, Schäfer P, Sappino AP, Kurtz J, Neyroud-Caspar I and
Bouchardy C: Survival of young and older breast cancer patients in
Geneva from 1990 to 2001. Eur J Cancer. 41:1446–1452. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Varga D, Koenig J, Kuhr K, Strunz K, Geyer
V, Kurzeder C, Atassi Z, Blettner M, Kreienberg R and Woeckel A:
Comparison of early onset breast cancer patients to older
premenopausal breast cancer patients. Arch Gynecol Obstet.
282:427–432. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Clagnan WS, Andrade JM, Carrara HH, Tiezzi
DG, Reis FJ, Marana HR and Abrão RA: Age as an independent
prognostic factor in breast cancer. Rev Bras Ginecol Obstet.
30:67–74. 2008.(In Portuguese). View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Maggard MA, O'Connell JB, Lane KE, Liu JH,
Etzioni DA and Ko CY: Do young breast cancer patients have worse
outcomes? J Surg Res. 113:109–113. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Coleman MP, Quaresma M, Berrino F, Lutz
JM, De Angelis R, Capocaccia R, Baili P, Rachet B, Gatta G,
Hakulinen T, et al: Cancer survival in five continents: A worldwide
population-based study (CONCORD). Lancet Oncol. 9:730–756. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Perry N, Broeders M, de Wolf C, Törnberg
S, Holland R and von Karsa L: European guidelines for quality
assurance in breast cancer screening and diagnosis. Fourth
edition-summary document. Ann Oncol. 19:614–622. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chan A, Pintilie M, Vallis K, Girourd C
and Goss P: Breast cancer in women <or=35 years: Review of 1002
cases from a single institution. Ann Oncol. 11:1255–1262. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Fowble BL, Schultz DJ, Overmoyer B, Solin
LJ, Fox K, Jardines L, Orel S and Glick JH: The influence of young
age on outcome in early stage breast cancer. Int J Radiat Oncol
Biol Phys. 30:23–33. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yildirim E, Dalgic T and Berberoğlu U:
Prognostic significance of young age in breast cancer. J Surg
Oncol. 74:267–272. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Goldstein NS, Vicini FA, Kestin LL and
Thomas M: Differences in the pathologic features of ductal
carcinoma in situ of the breast based on patient age. Cancer.
88:2553–2560. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zabicki K, Colbert JA, Dominguez FJ, Gadd
MA, Hughes KS, Jones JL, Specht MC, Michaelson JS and Smith BL:
Breast cancer diagnosis in women < or=40 versus 50 to 60 years:
Increasing size and stage disparity compared with older women over
time. Ann Surg Oncol. 13:1072–1077. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Berry DA, Cronin KA, Plevritis SK, Fryback
DG, Clarke L, Zelen M, Mandelblatt JS, Yakovlev AY, Habbema JD and
Feuer EJ: Cancer Intervention and Surveillance Modeling Network
(CISNET) Collaborators: Effect of screening and adjuvant therapy on
mortality from breast cancer. N Engl J Med. 353:1784–1792. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tabar L, Yen MF, Vitak B, Chen HH, Smith
RA and Duffy SW: Mammography service screening and mortality in
breast cancer patients: 20-year follow-up before and after
introduction of screening. Lancet. 361:1405–1410. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gomes MC, Costa MM, Borojevic R, Monteiro
AN, Vieira R, Koifman S, Koifman RJ, Li S, Royer R, Zhang S and
Narod SA: Prevalence of BRCA1 and BRCA2 mutations in breast cancer
patients from Brazil. Breast Cancer Res Treat. 103:349–353. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Risendal B, Hines LM, Sweeney C, Slattery
ML, Giuliano AR, Baumgartner KB, Curtin K and Byers TE: Family
history and age at onset of breast cancer in Hispanic and
non-Hispanic white women. Cancer Causes Control. 19:1349–1355.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bharat A, Aft RL, Gao F and Margenthaler
JA: Patient and tumor characteristics associated with increased
mortality in young women (<or=40 years) with breast cancer. J
Surg Oncol. 100:248–251. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Siegelmann-Danieli N, Khandelwal V, Wood
GC, Mainali R, Prichard J, Murphy TJ, Evans JF, Yumen O and Bernath
AM: Breast cancer in elderly women: Outcome as affected by age,
tumor features, comorbidities, and treatment approach. Clin Breast
Cancer. 7:59–66. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Colleoni M, Rotmensz N, Robertson C,
Orlando L, Viale G, Renne G, Luini A, Veronesi P, Intra M, Orecchia
R, et al: Very young women (<35 years) with operable breast
cancer: Features of disease at presentation. Ann Oncol. 13:273–279.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
El Saghir NS, Seoud M, Khalil MK,
Charafeddine M, Salem ZK, Geara FB and Shamseddine AI: Effects of
young age at presentation on survival in breast cancer. BMC Cancer.
6:1942006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Parise CA, Bauer KR, Brown MM and Caggiano
V: Breast cancer subtypes as defined by the estrogen receptor (ER),
progesterone receptor (PR), and the human epidermal growth factor
receptor 2 (HER2) among women with invasive breast cancer in
California, 1999–2004. Breast J. 15:593–602. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Han W, Kim SW, Park IA, Kang D, Youn YK,
Oh SK, Choe KJ and Noh DY: Young age: An independent risk factor
for disease-free survival in women with operable breast cancer. BMC
Cancer. 4:822004. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Jmor S, Al-Sayer H, Heys SD, Payne S,
Miller I, Ah-See A, Hutcheon A and Eremin O: Breast cancer in women
aged 35 and under: Prognosis and survival. J R Coll Surg Edinb.
47:693–699. 2002.PubMed/NCBI
|
|
40
|
Aebi S, Gelber S, Castiglione-Gertsch M,
Gelber RD, Collins J, Thürlimann B, Rudenstam CM, Lindtner J,
Crivellari D, Cortes-Funes H, et al: Is chemotherapy alone adequate
for young women with oestrogen-receptor-positive breast cancer?
Lancet. 355:1869–1874. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Fernandopulle SM, Cher-Siangang P and Tan
PH: Breast carcinoma in women 35 years and younger: A pathological
study. Pathology. 38:219–222. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gajdos C, Tartter PI, Bleiweiss IJ, Bodian
C and Brower ST: Stage 0 to stage III breast cancer in young women.
J Am Coll Surg. 190:523–529. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Sidoni A, Cavaliere A, Bellezza G,
Scheibel M and Bucciarelli E: Breast cancer in young women:
Clinicopathological features and biological specificity. Breast.
12:247–250. 2003. View Article : Google Scholar : PubMed/NCBI
|