Introduction
Cervical cancer is the fourth most frequently
diagnosed type of cancer among females worldwide, with ~527,600 new
cases and 265,700 mortalities reported in 2012 worldwide (1). However, for patients with locally
advanced stage disease and non-operable recurrence, platinum-based
chemoradiotherapy remains the necessary curative therapy (2,3). However,
the chemotherapeutic use of platinum is limited by drug resistance
and severe side effects (4,5). Combinations of chemotherapeutic drugs
with new anti-cancer agents are being investigated to improve
clinical response.
Traditional Chinese medicine serves an important
role in human health to prevent the development of certain diseases
such as cancer. Pedate pinellia rhizome is a traditional Chinese
medicine distributed in the central regions of China, which has
been shown to be effective at treating 81.5% of the 247 cervical
cancer cases treated in the Obstetrics and Gynecology Hospital of
Fudan University in the 1970s (6).
Initial in vitro studies on the active constituents of the
plant demonstrated that the lipid-soluble fraction had the best
inhibitory effect on the proliferation of cancer cells. Alkaloids,
fatty acids and β-sitosterol were the predominant constituents of
the lipid-soluble fraction of the plant (7). The present study investigated a novel
lipid-soluble extract from Pinellia pedatisecta (PE), which
was extracted by the Shanghai Institute of Materia Medica, Chinese
Academy of Sciences (CAS) (8).
Previous studies have evaluated the cytotoxic effect of PE in
cervical cancer cells (8), and it was
observed that PE could enhance the cytotoxicity of CDDP against
human cervical cancer cells in vitro (9). However, little is known about the effect
of PE on the efficacy of chemotherapeutic drugs in animal models.
The present study aimed to assess the synergistic effect of PE when
combined with CDDP on the human cervical cancer cell line CaSki
in vivo. This combined treatment enabled a lower cytotoxic
dose of CDDP without affecting its therapeutic effects. The
detailed signaling pathway involved in the combined action of PE
and CDDP in the human cervical cancer cell line CaSki in
vivo is also discussed.
Materials and methods
Extraction of PE and preparation of PE
solution
Dried rhizomes of PE Schott were obtained from
Jinyao Ruida (Xuchang) Biology Technology Co., Ltd. (Zhengzhou,
China) in June 2013 and were identified by Professor Jin-Gui Shen
of the Shanghai Institute of Materia Medica, CAS (Shanghai, China).
A voucher specimen was deposited at the Herbarium of Shanghai
Institute of Materia Medica, CAS. The extracting technique and PE
preparation process have been described in detail previously
(8). Once prepared, PE was stored in
a freezer at −20°C. Prior to use, PE was dissolved in dimethyl
sulfoxide (DMSO) at a concentration of 20 mg/µl and stored at 4°C.
For animal experiments, a PE solution was freshly prepared by
diluting the stock with 0.9% normal saline to the desired
concentrations. DMSO (final concentration, 1%) was used as a
solvent control.
Cell culture and chemical
reagents
The human cervical cancer cell line CaSki was
obtained from the American Type Culture Collection (Manassas, VA,
USA) and resuscitated by the Cell Bank, CAS. CaSki cells were then
cultured at 37°C in a humidified 5% CO2 atmosphere in
Roswell Park Memorial Institute (RPMI)-1,640 medium (Gibco; Thermo
Fisher Scientific, Inc., Waltham, MA, USA) with 10% fetal calf
serum (Gibco; Thermo Fisher Scientific, Inc.), 100 IU/ml penicillin
G, and 100 mg/ml streptomycin sulfate (Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany). cis-Dichlorodiammineplatinum-II (CDDP) was
purchased from Sigma-Aldrich (Merck KGaA). Rabbit monoclonal
antibodies directed against p53 (2527), p21 Waf1/Cip1 (2947), p27
Kip1(3686s), apoptosis protease activating factor 1 (Apaf-1)
(8969s), B cell lymphoma/leukemia-2 (Bcl-2) (9941), Bcl-2
associated X protein (Bax) (9942s), cleaved-caspase-9 (9501s),
cleaved-caspase-3 (9664s) and GAPDH (2118) were purchased from Cell
Signaling Technology, Inc. (Danvers, MA, USA). Mouse monoclonal
antibody directed against human papilloma virus (HPV) E6 (sc-460)
was provided by Santa Cruz Biotechnology, Inc. (Dallas, TX, USA).
Rabbit monoclonal antibody against Ki-67 (ab16667) was also used
(Abcam, Cambridge, UK).
Animal experiments
Athymic mice (BALB/c nu/nu, female; 16–18 g; 4–6
weeks old) were obtained from the Laboratory Animal Center of the
Shanghai Institutes for Biological Sciences, CAS, and were raised
in cages maintained at a temperature of 22±2°C and 65±5% humidity
in a controlled animal facility with a 12-h light-dark cycle and
ad libitum access to water in the Department of Laboratory
Animal Science, Fudan University (Shanghai, China). All animal
experiments were conducted in accordance with the internationally
accepted principles for laboratory animal use and care, as
described in the European Economic Community (EEC) guidelines (EEC
Directive of 1986; 86/609/EEC) (10),
and with approval from the Ethics Committee for Animal
Experimentation of Fudan University. CaSki cells (2×106
cells in 0.2 ml of RPMI-1,640 without FCS) were subcutaneously
injected into the right flank of the mice for tumor formation. When
established tumors of ~100 mm3 in volume were detected,
the mice were randomly divided into four groups (15 mice/group) and
treated as follows: i) Solvent control; ii) PE at 10 mg/kg/day by
gavage, as determined by a preliminary experiment (data not shown);
iii) CDDP at 3 mg/kg every 3 days by intraperitoneal injection; and
iv) combination of (ii and iii). Tumor volume was assessed every
other day using a caliper, and was calculated as the (length ×
width)2/2, where the width is the smallest measurement
and the length is the longest measurement. After 3 weeks, half of
the mice were killed, and the tumors were excised and weighed. The
tumor inhibitory ratio was calculated using the following formula:
Tumor inhibitory ratio (%)=[(C-T)/C]x100, where C is the mean tumor
weight of the solvent control group and T is the mean tumor weight
of the treated group. Tumor tissues were harvested for tissue
processing. A 4-mm portion of each tumor was used to detect
proteins using immunohistochemistry, and the remaining portion was
frozen in liquid nitrogen for western blot analysis. The rest of
mice were maintained until 70 days post-treatment, and mortality
was recorded to obtain the survival curve of the mice.
Western blot analysis
To evaluate the expression of HPV E6, p53, p21, p27,
Apaf-1, cleaved caspase-3, cleaved caspase-9, Bcl-2 and Bax
proteins, 200 mg of tissue was removed from liquid nitrogen and
grounded into small pieces. The tissue was stirred in 10 volumes of
lysis buffer (50 mM Tris-HCl pH 7.4; 150 mM NaCl; 1% NP40; 0.25%
Na-deoxycholate; 1 mM EDTA; 1 mM phenylmethane sulfonyl fluoride;
and protease inhibitor cocktail) and centrifuged at 288 × g and 4°C
for 20 min. Concentration was determined using the Bio-Rad Protein
Assay kit (Bio-Rad Laboratories, Inc., Hercules, CA, USA). The
proteins were separated by SDS-PAGE with 30% acrylamide in the gel
and then transferred onto polyvinylidene difluoride membranes.
After blocking with 5% non-fat dry milk in PBST (20 mM Tris-HCl,
150 mM NaCl and 0.05% Tween-20) for 1 h at room temperature, the
membranes were incubated with primary antibodies at a 1:1,000
dilution overnight at 4°C. Upon being washed with PBST three times,
the membranes were incubated with horseradish peroxidase-conjugated
goat anti-rabbit secondary antibodies (catalog no., 7170S; Cell
Signaling Technology, Inc.) at a 1:1,000 dilution for 1 h at room
temperature. The membranes were then washed with PBST three times
for 60 min. The specific HPV E6, p53, p21, p27, Apaf-1, cleaved
caspase-3, cleaved caspase-9, Bcl-2 and Bax bands were developed
using an enhanced chemiluminescent reagent (Pierce; Thermo Fisher
Scientific, Inc.) and imaged by a gel scanner. The protein levels
were normalized to GAPDH as reference.
Immunohistochemistry
Ki-67 and p53 expression was detected by
immunohistochemistry. Paraffin-embedded tissue sections were
deparaffinized by xylene, and sequentially rehydrated by 100, 95
and 75% ethanol, followed by PBS. The antigen retrieval step was
performed by heating the slides in citrate buffer for 25 min and
then incubated with endogenous peroxidase blocking solution (30%
H2O2: CH3OH=1:9) for 30 min. The
slides were incubated with primary anti-Ki-67 (1:100) or anti-p53
(1:50) antibodies overnight at 4°C, and PBS was used as a negative
control. Then, the slides were incubated with peroxidase-conjugated
anti-rabbit immunoglobulin G (catalog no., 8114P; Cell Signaling
Technology, Inc.) for 30 min. The staining reaction was performed
with 3,3′-diaminobenzidine. Hematoxylin was used as a counter-stain
to detect the nuclei, which were imaged with light microscopy.
Statistical analysis
Statistical analyses were conducted with SPSS 16.0
(SPSS Inc., Chicago, IL, USA). Data are expressed as the mean ±
standard deviation. Student's t-test was used for comparisons
between groups. Kaplan-Meier analysis with a log-rank test was used
to determine survival and differences between the control and
treatment groups. P<0.05 was considered to indicate a
statistically significant difference.
Results
Synergistic inhibitory effects of PE
and CDDP on subcutaneous xenografts in nude mice
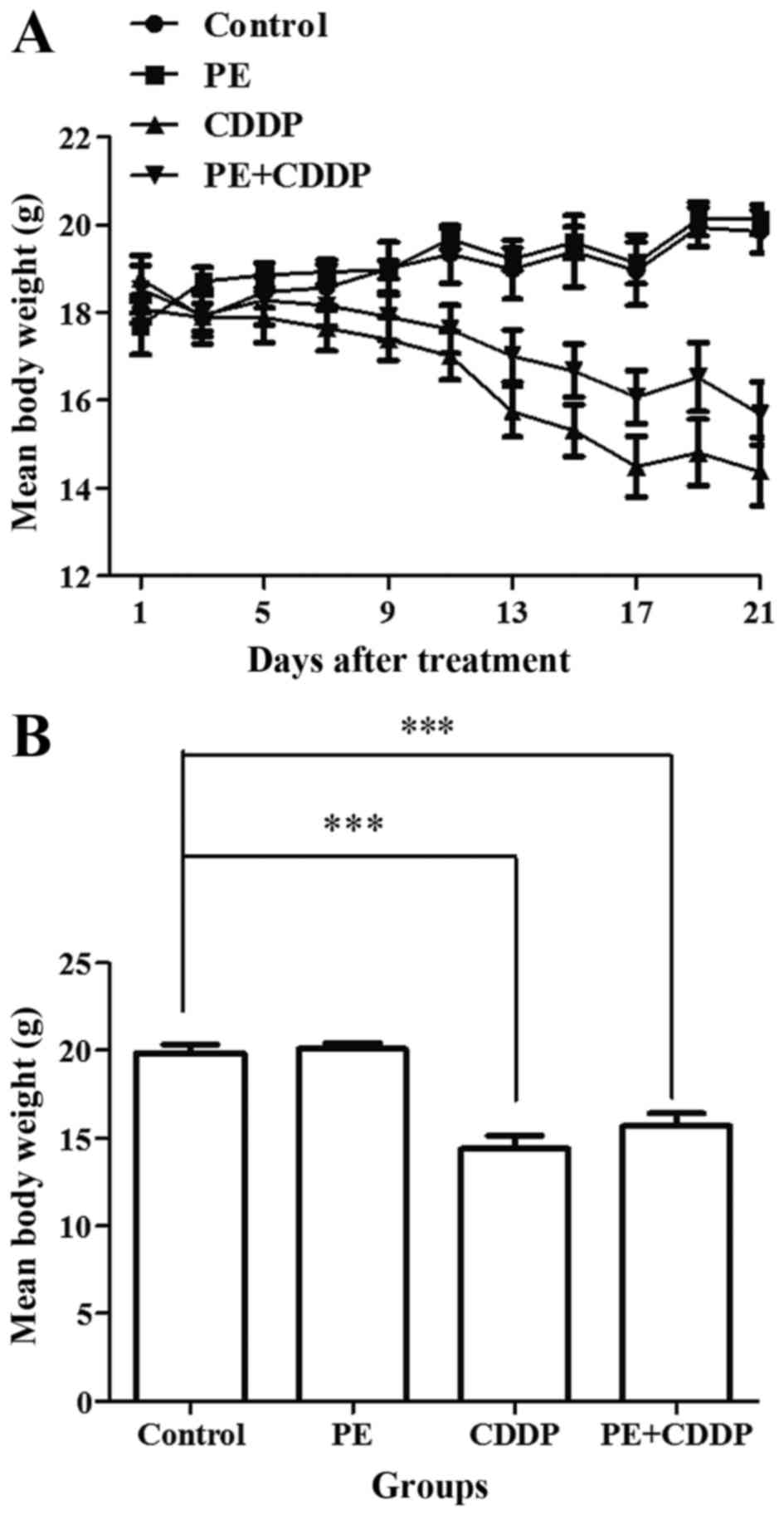
None of the mice exhibited signs of physical
discomfort during the treatment period (21 days). At the start of
the treatment (day 0), the mean body weight of mice in the control
and PE-treated groups was 18.0 and 17.7 g, respectively. Following
treatment, the mean body weight was 19.9 and 20.1 g, respectively
(day 21). Body weight stably increased in both groups (Fig. 1A). However, in the CDDP-treated group,
the mice lost 25% of their body weight, while the mice in the
combined PE and CDDP group lost 15% of their body weight. The
CDDP-treated mice suffered a significant weight loss compared with
that of the PE-treated and control mice (P<0.001). Although not
significantly (P=0.24), the mice treated with CDDP lost more weight
compared with mice undergoing combined treatment (Fig. 1).
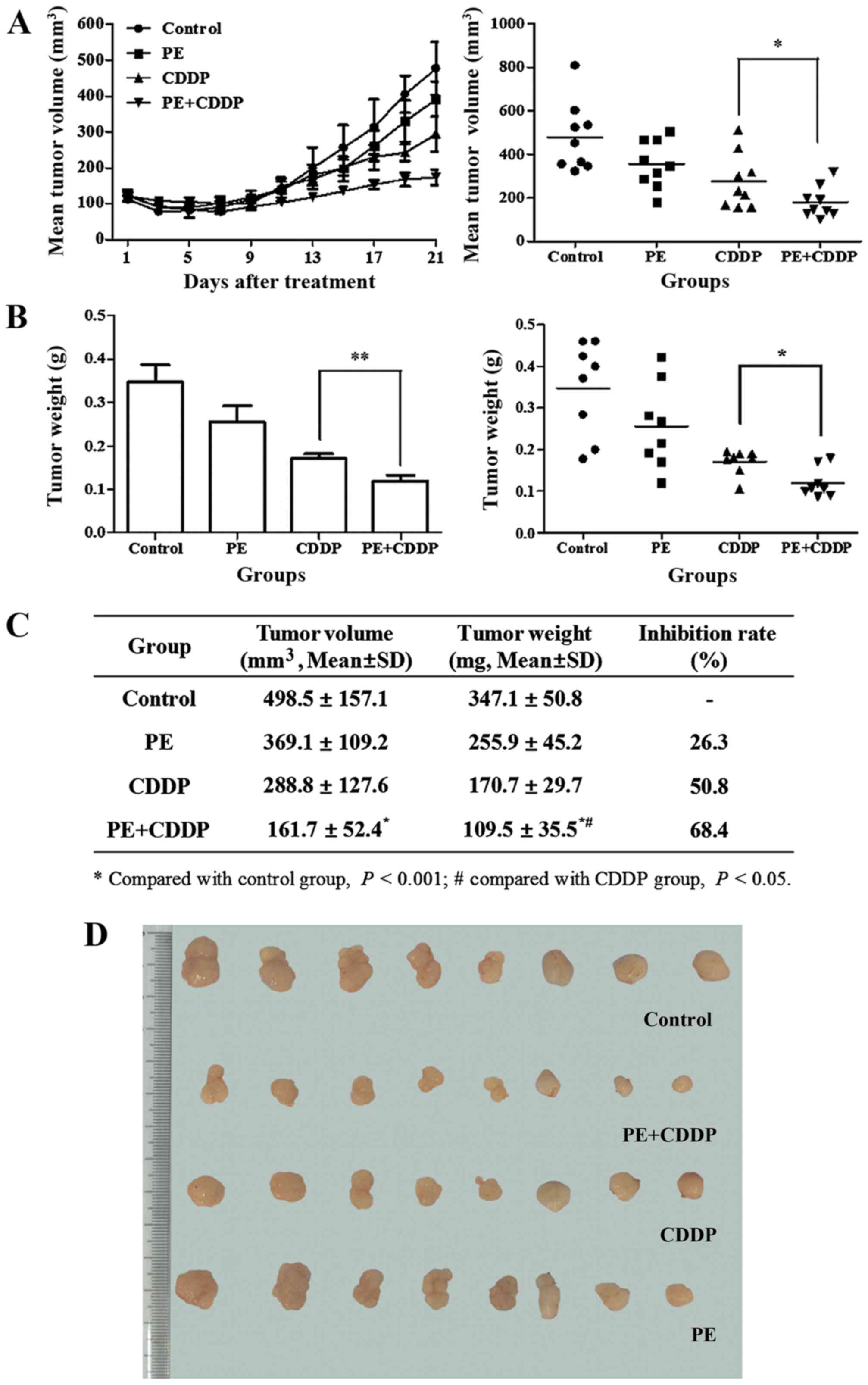
Responses to the treatment regimen were monitored by
assessing the tumor size. The initial xenograft volume in each
group was ~100 mm3. At the end of the treatment, the
volume of the tumor mass was 489.5±157.1 mm3 in the
control group, 369.1±157.1 mm3 in the PE-treated group,
288.8±127.6 mm3 in the CDDP-treated group and
0.161±0.052 mm3 in the combined PE and CDDP-treated
group (Fig. 2A and C). The mean
weight of the resected tumor mass in the combined group was
significantly lower than that of the groups treated with PE or CDDP
alone (mean, 0.110±0.036 g vs. 0.256±0.045 g, P<0.01; and mean,
0.110±0.036 g vs. 0.171±0.030 g, P<0.05, respectively) (Fig. 2B and C). PE slightly decreased the
growth of human cervical cancer cells transplanted subcutaneously
in female athymic mice, and the tumor weight inhibitory ratio was
26.3%. With combined PE and CDDP treatment, the tumor weight
inhibitory ratio increased to 68.4%, which was higher than that of
the CDDP-treated group (50.8%).
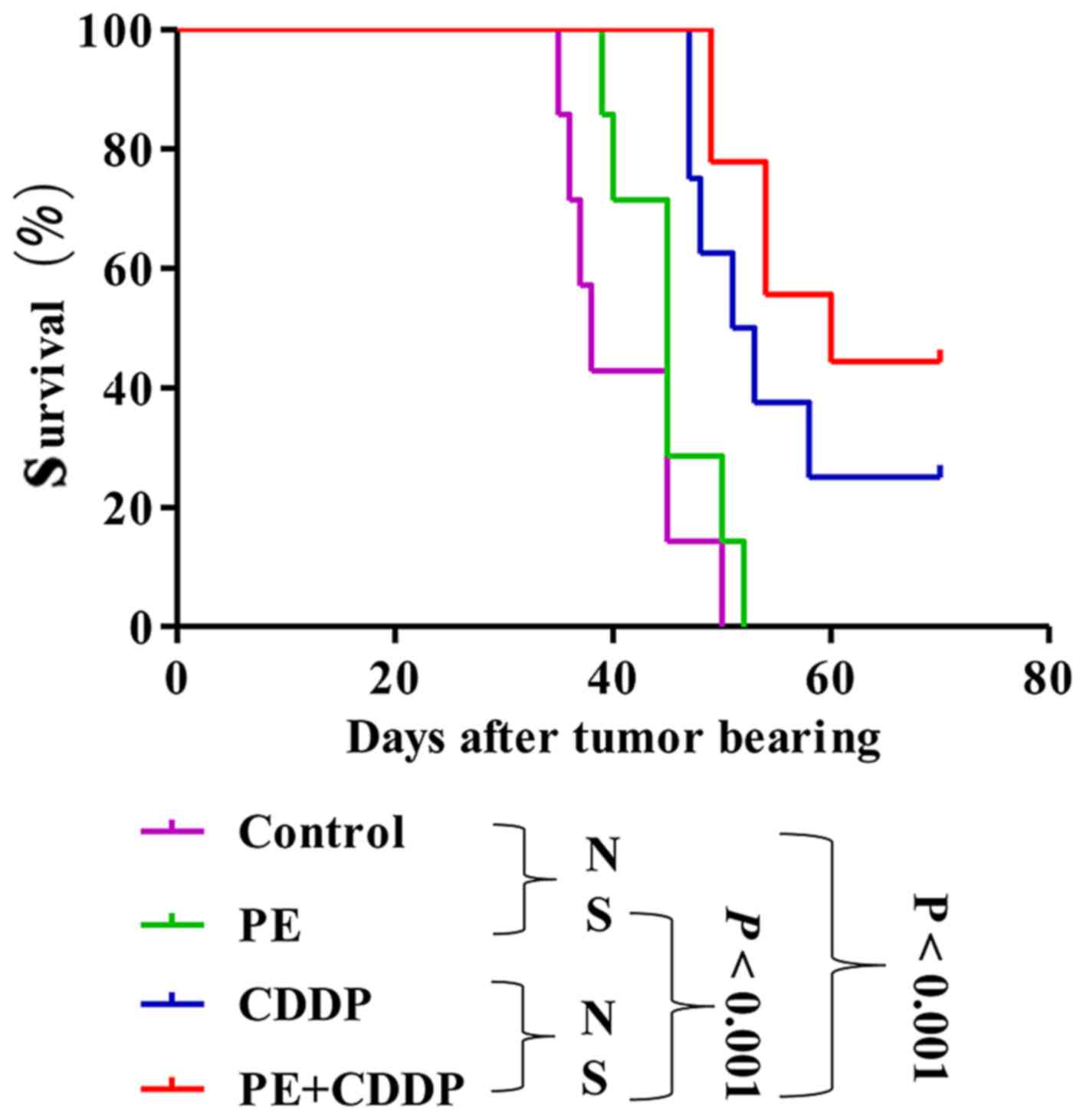
PE does not prolong the survival of
tumor-bearing nude mice
PE did not significantly prolong survival in mice
subcutaneously transplanted with the human cervical cancer CaSki
cell line. Median survival time slightly increased from 38 days in
the untreated control animals to 45 days in mice treated with PE
(P=0.15), 52 days in mice treated with CDDP (P=0.0011), and 60 days
in mice treated with both PE and CDDP (P<0.001) (Fig. 3). The survival of mice in the combined
PE and CDDP treatment group was not longer than that of the
CDDP-treated mice (P=0.22).
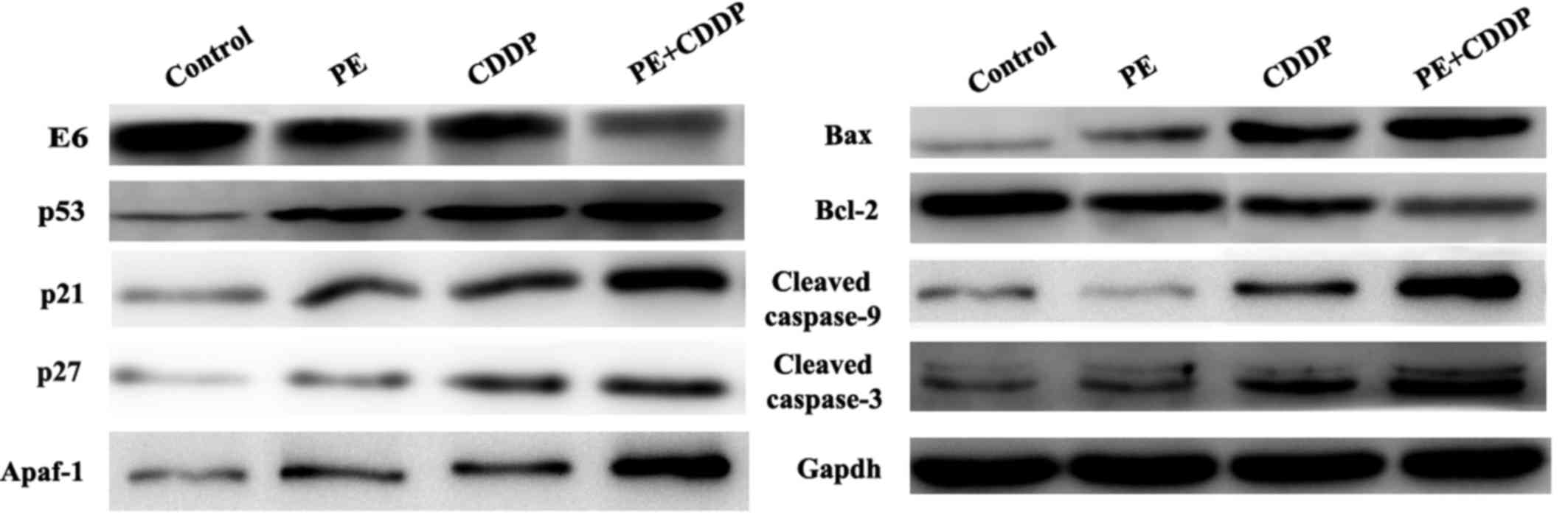
Effects of PE and CDDP on the
expression of E6, p53 and apoptosis-associated proteins by western
blot and immunohistochemistry analyses of tumor tissues
Since the human cervical cancer cell line CaSki
contains the high-risk HPV16 genotype as described in the
introduction of this cell line in ATCC (ATCC®
CRL-1550™), to further understand how PE and CDDP function to
inhibit tumor growth synergistically, it was assessed whether the
HPV E6/p53 signaling pathway was involved. The present study
evaluated the protein expression of E6, p53, p21 and p27 in tumor
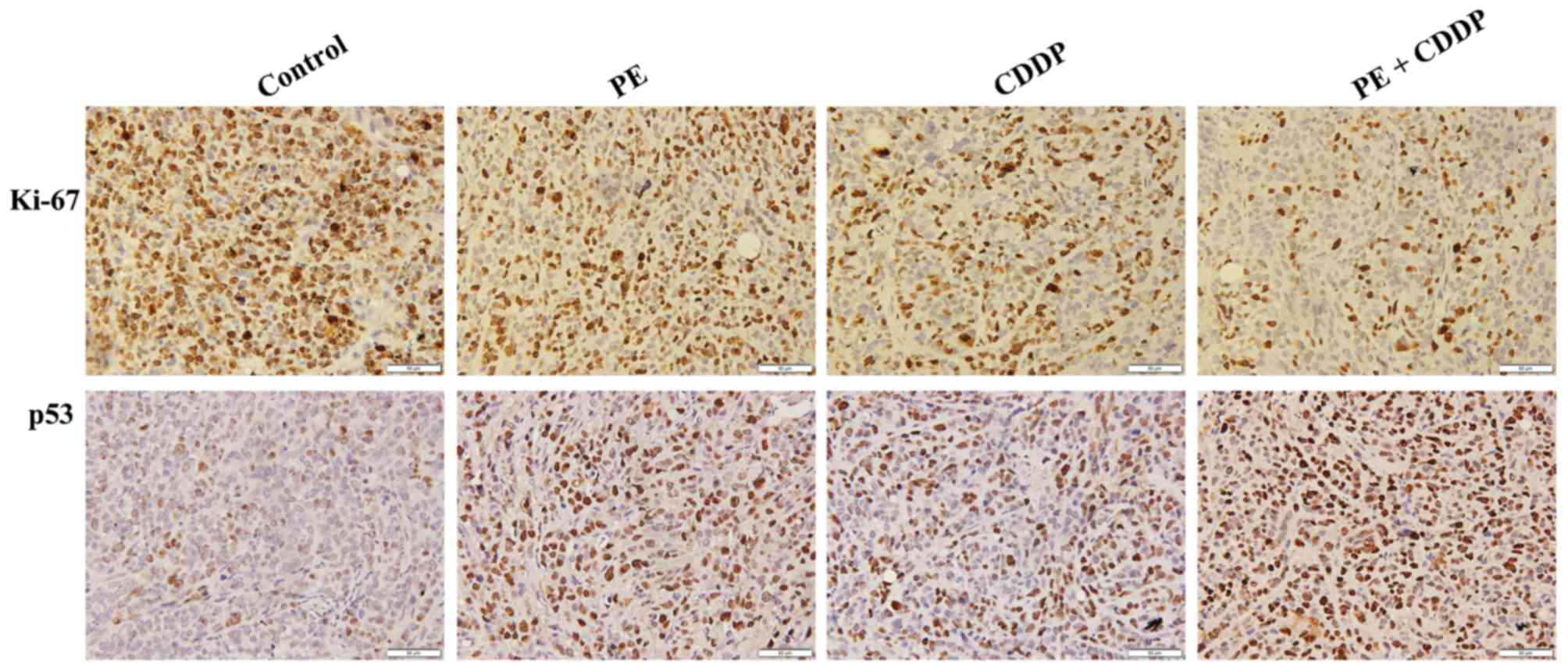
masses resected from mice. As shown in Fig. 4, E6 had remarkably decreased protein
expression levels upon co-treatment by PE and CDDP. By contrast,
p53 expression was increased in the combined PE and CDDP treatment
group, according to western blot and immunohistochemistry analyses.
The Bcl-2 protein family controls apoptosis in mitochondria by
balancing pro-and anti-apoptotic factors (11). The present study assessed the
expression of the pro-apoptotic member Bax and the anti-apoptotic
member Bcl-2. Bcl-2 protein expression was decreased significantly
following combined PE and CDDP treatment. Caspases are the
molecular machinery that directly drives apoptosis (12). Cleaved caspase-9 and cleaved caspase-3
expression was highly increased by treatment with PE or CDDP alone.
The highest upregulation of apoptosis-associated proteins,
including Apaf-1, Bax, and cleaved caspases-9 and −3, was observed
in the combined PE and CDDP treatment group (Fig. 4). As shown in Fig. 5, tumors from the combined PE and CDDP
treatment group exhibited the lowest cellular proliferation by
Ki-67 immunohistochemistry in xenografts.
Discussion
Cervical cancer has decreased to the fourth most
frequently diagnosed type of cancer among females worldwide,
primarily due to the widespread use of cervical screening programs
(1,13). However, 84.2% of new cases were
diagnosed in developing countries, and 86.6% of mortalities
occurred in developing countries (1).
The age-standardized cervical cancer incidence in China was
<5/100,000 from 1998 to 2002 (14), and the age-standardized 5-year (from
2005 to 2009) net survival was 60% (15,16).
However, the incidence of cervical cancer mortality in China is
currently among the highest worldwide (14). It is therefore still a big challenge
for the Chinese government and for other less developed countries
and areas to prevent this disease.
While the first-line treatment for early stages of
cervical cancer is surgical excision, concomitant chemoradiotherapy
based on CDDP is still the standard treatment for local advanced
cervical cancer, particularly for distant control of the disease
(17,18). However, the chemotherapeutic use of
CDDP is limited by severe side effects, including neurotoxicity,
ototoxicity and in particular nephrotoxicity (19,20). Due
to the importance of CDDP chemotherapy for cancer patients,
numerous studies have focused on protective strategies to alleviate
the side effects of CDDP. Herbal and herbal-derived medicine has
been recognized as an attractive approach for cancer therapy with
few side effects (21,22). Furthermore, evidence suggests that
various herbal medicines have synergistic effects when combined
with CDDP by enhancing its toxicity against cancer cells,
prolonging the patients' survival time and preventing the side
effects of chemotherapy (23–25). The purpose of the present study was to
investigate in vivo whether PE had a synergistic cytotoxic
effect when combined with CDDP, and to explore its potential
mechanism.
To demonstrate that PE could increase the
therapeutic activity of CDDP in vivo, a xenograft model with
the human cervical cancer cell line CaSki was established in
athymic female nude mice. Upon treatment, all tumor volumes were
suppressed in the PE, CDDP and combined treatment groups. The
combined treatment group exhibited the highest tumor suppression.
As shown in Fig. 2A, the synergistic
cytotoxic effect of PE and CDDP on tumor volume was first observed
at day 7 after treatment, and achieved the highest effect at the
end of the treatment. The same trend was also observed for tumor
weight. The tumor weight inhibitory ratio in the combined treatment
group reached 68.4%, compared with 50.8% in the CDDP treatment
group. Mice in the CDDP treatment group had the most severe weight
loss. However, body weight in the PE treatment group was stable,
with a slight increasing trend. Furthermore, body weight in the
combined treatment group was higher than that in the CDDP treatment
group. These results indicated that PE was safe and efficient in
enhancing the cytotoxicity of CDDP in vivo, and it may
decrease the effect of CDDP toxicity on weight loss.
As shown in Fig. 3,
the mean survival time of mice in the combined treatment group was
60 days, which is longer than the 52 days observed with CDDP
treatment alone. However, PE alone or combined with CDDP did not
significantly prolong the survival time, likely because 70 days of
tumor bearing was set as the cut-off point of survival time. A
longer observational time or increased number of mice used in each
group could have altered the present results. It is also possible
that an increased dose of CDDP would have increased the mortality
in the CDDP treatment group. Therefore, it cannot be excluded that
combined PE and CDDP may significantly improve survival time.
Western blot assays were used in the present study
to evaluate the expression of primary proteins in the HPV E6/p53
signaling pathway. It is known that~50–60% of cervical cancers
carry HPV16, and E6 (the main viral oncoprotein encoded by HPV16)
serves a vital role in viral replication and oncogenic progression
(26). E6 can regulate tumor growth
signaling by forming specific complexes with tumor-suppressor gene
products such as p53 (27). As a
transcription factor, p53 serves a vital role in integrating
cellular responses to various stimuli, including DNA damage and
hypoxia, and once activated, p53 can stimulate the actions of
various proteins involved in apoptosis and DNA repair (28,29). When
E6 binds to p53 and promotes its degradation through an
ubiquitin-dependent proteasome pathway (30), the normal p53 function is lost
(31). Importantly, reactivation of
p53 can lead to inhibition of cell proliferation and induction of
apoptosis in cervical cancer cells (32). Therefore, restoration of p53 function
through inhibition of the E6/p53 signaling pathway has been
considered an effective therapeutic approach for treating cervical
cancer (33). As shown in Fig. 4, E6 expression was remarkably
decreased when mice were co-treated with PE and CDDP, whilep53
expression was increased. Furthermore, the highest upregulation of
p53-dependent apoptosis-associated proteins such as Bax and cleaved
caspases-9 and −3was observed in the combined treatment group,
whereas Bcl-2 expression was downregulated.
In conclusion, the present data and previous
findings indicate that PE exerts a synergistic cytotoxic effect on
tumor growth in vivo when combined with CDDP. The potential
mechanism is likely associated with the inhibition of the E6/p53
signaling pathway to synergistically induce apoptotic signals in
CaSki cells in vivo. The present study suggests a molecular
basis for the potential application of PE and CDDP combination in
the treatment of cervical cancer as a novel and pharmacologically
safe chemotherapeutic strategy.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China (grant no. 81373867 awarded to
Dr Guiling Li).
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
DiSilvestro PA, Ali S, Craighead PS, Lucci
JA, Lee YC, Cohn DE, Spirtos NM, Tewari KS, Muller C, Gajewski WH,
et al: Phase III randomized trial of weekly cisplatin and
irradiation versus cisplatin and tirapazamine and irradiation in
stages IB2, IIA, IIB, IIIB, and IVA cervical carcinoma limited to
the pelvis: A Gynecologic Oncology Group study. J Clin Oncol.
32:458–464. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Friedlander M and Grogan M: U.S.
Preventative Services Task Force: Guidelines for the treatment of
recurrent and metastatic cervical cancer. Oncologist. 7:342–347.
2002.PubMed/NCBI
|
|
4
|
Travis LB, Fossa SD, Sesso HD, Frisina RD,
Herrmann DN, Beard CJ, Feldman DR, Pagliaro LC, Miller RC, Vaughn
DJ, et al: Chemotherapy-induced peripheral neurotoxicity and
ototoxicity: New paradigms for translational genomics. J Natl
Cancer Inst. 106:pii: dju0442014. View Article : Google Scholar
|
|
5
|
Chen SJ, Kuo CC, Pan HY, Tsou TC, Yeh SC
and Chang JY: Mechanistic basis of a combination D-penicillamine
and platinum drugs synergistically inhibits tumor growth in
oxaliplatin-resistant human cervical cancer cells in vitro and in
vivo. Biochem Pharmaco. l95:28–37. 2015. View Article : Google Scholar
|
|
6
|
Li CJ, Xu YM and Sun YL: Research on
anti-cervical cancer component of Pinellia pedatisecta Schott. J
Shanghai Med Univ. 8:421–423. 1981.(In Chinese).
|
|
7
|
Chemistry Department of Basic Research
Center, Obstetrics and Gynecology Hospital, . Research of Pinellia
pedatisecta Schott on cervical cancer. Shanghai Med J. 1:13–16.
1978.(In Chinese).
|
|
8
|
Li GL, Jiang W, Xia Q, Chen SH, Ge XR, Gui
SQ and Xu CJ: HPV E6 down-regulation and apoptosis induction of
human cervical cancer cells by a novel lipid-soluble extract (PE)
from Pinellia pedatisecta Schott in vitro. J Ethnopharmacol.
132:56–64. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li GL, Gui SQ, Zhu DH, Chen SH and Ye Y:
Effects of pinellia extraction only or combined with cisplatin on
growth of HeLa cells of cervical cancer. Fudan Univ J Med Sci.
34:869–872. 2007.
|
|
10
|
Spielmann H: FRAME Annual Lecture.
International co-operation: An essential requirement for replacing
animal toxicity tests. Altern Lab Anim. 29:637–648. 2001.PubMed/NCBI
|
|
11
|
Hatok J and Racay P: Bcl-2 family
proteins: Master regulators of cell survival. Biomol Concepts.
7:259–270. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Matt S and Hofmann TG: The DNA
damage-induced cell death response: A roadmap to kill cancer cells.
Cell Mol Life Sci. 73:2829–2850. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Harlan LC and Warren JL: Global survival
patterns: Potential for cancer control. Lancet. 385:926–928. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shi JF, Canfell K, Lew JB and Qiao YL: The
burden of cervical cancer in China: Synthesis of the evidence. Int
J Cancer. 130:641–652. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Allemani C, Weir HK, Carreira H, Harewood
R, Spika D, Wang XS, Bannon F, Ahn JV, Johnson CJ, Bonaventure A,
et al: Global surveillance of cancer survival 1995–2009: Analysis
of individual data for 25,676,887 patients from 279
population-based registries in 67 countries (CONCORD-2). Lancet.
385:977–1010. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ferlay J, Forman D, Mathers CD and Bray F:
Breast and cervical cancer in 187 countries between 1980 and 2010.
Lancet. 379:1390–1391. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jelavić TB, Miše BP, Strikic A, Ban M and
Vrdoljak E: Adjuvant chemotherapy in locally advanced cervical
cancer after treatment with concomitant chemoradiotherapy-room for
improvement? Anticancer Res. 35:4161–4165. 2015.PubMed/NCBI
|
|
18
|
Penson RT, Huang HQ, Wenzel LB, Monk BJ,
Stockman S, Long HJ III, Ramondetta LM, Landrum LM, Oaknin A, Reid
TJ, et al: Bevacizumab for advanced cervical cancer:
Patient-reported outcomes of a randomised, phase 3 trial (NRG
Oncology-Gynecologic Oncology Group protocol 240). Lancet Oocol.
16:301–311. 2015. View Article : Google Scholar
|
|
19
|
Sprowl JA, Lancaster CS, Pabla N, Hermann
E, Kosloske AM, Gibson AA, Li L, Zeeh D, Schlatter E, Janke LJ, et
al: Cisplatin-induced renal injury is independently mediated by
OCT2 and p53. Clin Cancer Res. 20:4026–4035. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Karasawa T and Steyger PS: An integrated
view of cisplatin-induced nephrotoxicity and ototoxicity. Toxicol
Lett. 237:219–227. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hirchaud F, Hermetet F, Ablise M,
Fauconnet S, Vuitton DA, Prétet JL and Mougin C: Isoliquiritigenin
induces caspase-dependent apoptosis via downregulation of HPV16 E6
expression in cervical cancer Ca Ski cells. Planta Med.
79:1628–1635. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Peng Y, Guo CS, Li PX, Fu ZZ, Gao LM, Di
Y, Ju YK, Tian R and Xue JJ: Immune and anti-oxidant functions of
ethanol extracts of Scutellaria baicalensis Georgi in mice bearing
U14 cervical cancers. Asian Pac J Cancer Prev. 15:4129–4133. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang LH, Li Y, Yang SN, Wang FY, Hou Y,
Cui W, Chen K, Cao Q, Wang S, Zhang TY, et al: Gambogic acid
synergistically potentiates cisplatin-induced apoptosis in
non-small-cell lung cancer through suppressing NF-κB and MAPK/HO-1
signalling. Br J Cancer. 110:341–352. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li X, Huang T, Jiang G, Gong W, Qian H and
Zou C: Synergistic apoptotic effect of crocin and cisplatin on
osteosarcoma cells via caspase induced apoptosis. Toxicol Lett.
221:197–204. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chen Y, Liu JM, Xiong XX, Qiu XY, Pan F,
Liu D, Lan SJ, Jin S, Yu SB and Chen XQ: Piperlongumine selectively
kills hepatocellular carcinoma cells and preferentially inhibits
their invasion via ROS-ER-MAPKs-CHOP. Oncotarget. 6:6406–6421.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tan S, de Vries EG, van der Zee AG and de
Jong S: Anticancer drugs aimed at E6 and E7 activity in
HPV-positive cervical cancer. Curr Cancer Drug Targets. 12:170–184.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Münger K, Scheffner M, Huibregtse JM and
Howley PM: Interactions of HPV E6 and E7 oncoproteins with tumour
suppressor gene products. Cancer Surv. 12:197–217. 1992.PubMed/NCBI
|
|
28
|
Bradner JE: Cancer: An essential passenger
with p53. Nature. 520:626–627. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Guimarães-Camboa N, Stowe J, Aneas I,
Sakabe N, Cattaneo P, Henderson L, Kilberg MS, Johnson RS, Chen J,
McCulloch AD, et al: HIF1α represses cell stress pathways to allow
proliferation of hypoxic fetal cardiomyocytes. Dev Cell.
33:507–521. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lee D, Kwon JH, Kim EH, Kim ES and Choi
KY: HMGB2 stabilizes p53 by interfering with E6/E6AP-mediated p53
degradation in human papillomavirus-positive HeLa cells. Cancer
Lett. 292:125–132. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang W, Liu Y, Zhao N, Chen H, Qiao L,
Zhao W and Chen JJ: Role of Cdk1 in the p53-independent abrogation
of the postmitotic checkpoint by human papillomavirus E6. J Virol.
89:2553–2562. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kochetkov DV, Il'Inskaia GV, Komarov PG,
Strom E, Agapova LS, Ivanov AV, Budanov AV, Frolova EI and Chumakov
PM: Transcriptional inhibition of human papilloma virus in cervical
carcinoma cells reactivates functions of the tumor suppressor p53.
Mol Biol (Mosk). 41:515–523. 2007.(In Russian). View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Münger K and Howley PM: Human
papillomavirus immortalization and transformation functions. Virus
Res. 89:213–228. 2002. View Article : Google Scholar : PubMed/NCBI
|