Introduction
Gastric cancer (GC) is a common malignancy
worldwide, characterized by high invasiveness and aggressiveness
(1). It is estimated that there are
~990,000 new cases of GC and that ~738,000 succumb from this type
of cancer per year (2). Although a
number of conventional therapeutic methods, including surgical
excision and chemotherapy achieve satisfactory therapeutic effects
for patients with early GC, a greater number of patients with GC at
an advanced stage have poor prognosis (3). Therefore, despite increasing knowledge
of the genetic and biochemical basis of GC, it is also essential to
search for novel therapeutic targets and to develop more effective
diagnostic and treatment methods.
A series of studies have reported that nonsteroidal
anti-inflammatory drugs (NSAIDs) have an anti-cancer effect in
various types of cancer, including breast cancer (4), esophageal cancer (5), colorectal cancer (6), prostate cancer (7) and GC (8).
In addition to aspirin, celecoxib, a cyclooxygenase-2 (COX-2)
inhibitor, has been demonstrated to be involved in uncontrolled
cell proliferation, apoptosis, angiogenesis and metastasis
(9,10). Previous studies have suggested that
celecoxib is able to induce cell apoptosis through the
phosphoinositide 3 kinase/Akt signaling pathway (11) and to inhibit invasion through the
adenine nucleotide translocator-dependent signaling pathway in GC
cells (12). In addition, celecoxib
has been reported to be able to prevent the development of GC in
rats (13). Increasing evidence
suggests that long non-coding RNAs (lncRNAs) are able to regulate
tumor-suppressing or oncogenic effects, and lncRNAs may be
considered as novel biomarkers and therapeutic targets for cancer
(14,15). However, an understanding of the
function of lncRNAs in the treatment of GC with celecoxib remains
limited.
A previous bioinformatics study has demonstrated
that DVL1 and/or ITGA3, as well as their co-expressed lncRNAs,
including lnc-SCD-1:13 and lnc-PTMS-1:3, were abnormally expressed
in patients with GC following treatment with celecoxib. In the
present study, the effects of a low dose of celecoxib on the
viability, cell cycle and apoptosis of human GC NCI-N87 cells was
investigated. In addition, the mRNA levels of lnc-SCD-1:13,
lnc-PTMS-1:3, COX-2, ITGA3 and DVL1 were
analyzed in order to investigate further whether lncRNAs are
involved in the treatment of NCI-N87 cells with celecoxib.
Materials and methods
Cell culture
NCI-N87 cells were purchased from the Type Culture
Collection of the Chinese Academy of Sciences (Shanghai, China).
The cells were maintained in Dulbecco's modified Eagle medium
(DMEM; Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA)
supplemented with 10% fetal bovine serum (FBS; Gibco; Thermo Fisher
Scientific, Inc.) in a 37°C incubator with a humidified atmosphere
of 5% CO2 (Thermo Fisher Scientific, Inc.).
MTT assay
The cells (5×103) were seeded into each
well of a 96-well plate and cultured in DMEM supplemented with 10%
FBS. Following 24 h of culture at 37°C and 5% CO2 with
saturated humidity, the cells were treated with 0 (control group),
15, 30 and 60 µM celecoxib (Sigma-Aldrich; Merck KGaA, Darmstadt,
Germany), and subsequently incubated for 72 h (37°C, 5%
CO2 with saturated humidity) (11). MTT (10 µl, 5 mg/ml; Shanghai Sangon
Biotech Co., Ltd, Shanghai, China) was added to each well at the
same time, and the cells were subsequently incubated for 4 h at
37°C. Following the removal of the medium, dimethyl sulfoxide
(DMSO; 100 µl; Shanghai Sangon Biotech Co., Ltd) was added into
each well for 10 min to solubilize the formazan crystals at room
temperature. The zero control (medium, MTT, DMSO) and blank control
(no reagent) were set up. The absorbance was read at 570 nm using a
microplate reader (BioTek Instruments, Inc., Winooski, VT, USA).
All experiments were performed in triplicate.
Flow cytometry
The NCI-N87 cells were treated with a low dose of
celecoxib (15 µM) for 72 h at 37°C. For cell cycle detection, the
treated cells were collected by centrifugation (250 × g for 6 min)
at 25°C and subsequently fixed with ice-cold 70% ethanol overnight.
Next, the cells were centrifuged (111 × g for 5 min) at 25°C and
re-suspended in 500 µl phosphate buffered saline (PBS). The cells
were subsequently treated with 50 µg/ml RNase A (Shanghai Sangon
Biotech Co., Ltd.) for 30 min at 37°C. Finally, the cells were
stained with propidium iodide (PI; BD Pharmingen, San Diego, CA,
USA) in the dark for 15 min at 4°C and detected using a FACSCalibur
flow cytometer (BD Pharmingen). An annexin V-fluorescein
isothiocyanate (FITC) apoptosis detection kit (BD Pharmingen) was
used for the detection of apoptotic cells. The treated cells were
digested with 0.25% trypsin-EDTA and collected by centrifugation at
250 × g for 6 min at 25°C. The cells were then washed once with
PBS. Subsequently, the cells were resuspended with 1X binding
buffer (from the annexin V-FITC apaoptosis detection kit) and
stained with FITC-annexin V and PI in the dark for 15 min at 25°C.
Following this, 1X binding buffer (400 µl) was added to the cells,
and apoptosis was detected using a FACSCalibur flow cytometer (BD
Pharmingen).
Reverse transcription quantitative
polymerase chain reaction (RT-qPCR)
The cells were treated with a low dose of celecoxib
(15 µM) for 72 h at 37°C. Total RNA was extracted using 1 ml TRIzol
reagent (Takara Biotechnology Co., Ltd., Dalian, China), and cDNA
was obtained once using a reverse transcription kit (Takara
Biotechnology Co., Ltd), according to the manufacturer's protocol.
Primer sequences for ITGA3, DVL1, COX-2,
lnc-SCD-1:13, lnc-PTMS-1:3 and
glyceraldehyde-3-phosphate dehydrogenase are listed in Table I. SYBR Premix Ex Taq (Applied
Biosystems; Thermo Fisher Scientific, Inc.) was used to perform
qPCR. The thermocycler conditions used were as follows: 50°C for 3
min, 95°C for 3 min, 40 cycles of 95°C for 10 sec and 60°C for 30
sec. Relative quantification and calculations were performed using
the comparative threshold (Cq) cycle method (2−ΔΔCq)
(16).
 | Table I.Primer sequences for real-time
quantitative polymerase chain reaction. |
Table I.
Primer sequences for real-time
quantitative polymerase chain reaction.
| Gene | Primer sequences
(5′-3′) |
|---|
| GAPDH-hf |
GAAGGTGAAGGTCGGAGTC |
| GAPDH-hr |
GAAGATGGTGATGGGATTTC |
| ITGA3-hf |
GGACCTTACAACGCCGAGTG |
| ITGA3-hr |
GGAGGCTCTTTGGCTTGTTTT |
| DVL1-hf |
AGCACCTCATCCAGACTCATCC |
| DVL1-hr |
GATGCTGATGCCCAGAAAGTGAT |
| COX-2-hf |
CCCTGAGCATCTACGGTTTG |
| COX-2-hr |
CAGTATTAGCCTGCTTGTCT |
|
lnc-SCD-1:13-hf |
AAGTCTTGAAGTTGGGTGTT |
|
lnc-SCD-1:13-hr |
GAAGATGGCAGAGCAGAAAG |
|
lnc-LRR1-1:2-hf |
GTGTCCGCACTAAGTTCGGCATCA |
|
lnc-LRR1-1:2-hr |
GTGTCCGCACTAAGTTCGGCATCA |
|
lnc-PTMS-1:3-hf |
ATCCCAAACGGCAGAAGACA |
|
lnc-PTMS-1:3-hr |
CAGAGCAGGGAGCCAGGTGA |
Statistical analysis
SPSS statistical analysis software (version 12.0;
SPSS Inc., Chicago, IL, USA) was used to perform statistical
analysis. Data are expressed as the mean ± standard error and were
analyzed by one-way analysis of variance followed by least
significance difference test. P<0.05 was considered to indicate
a statistically significant difference.
Results
Effect of celecoxib on the viability
of NCI-N87 cells
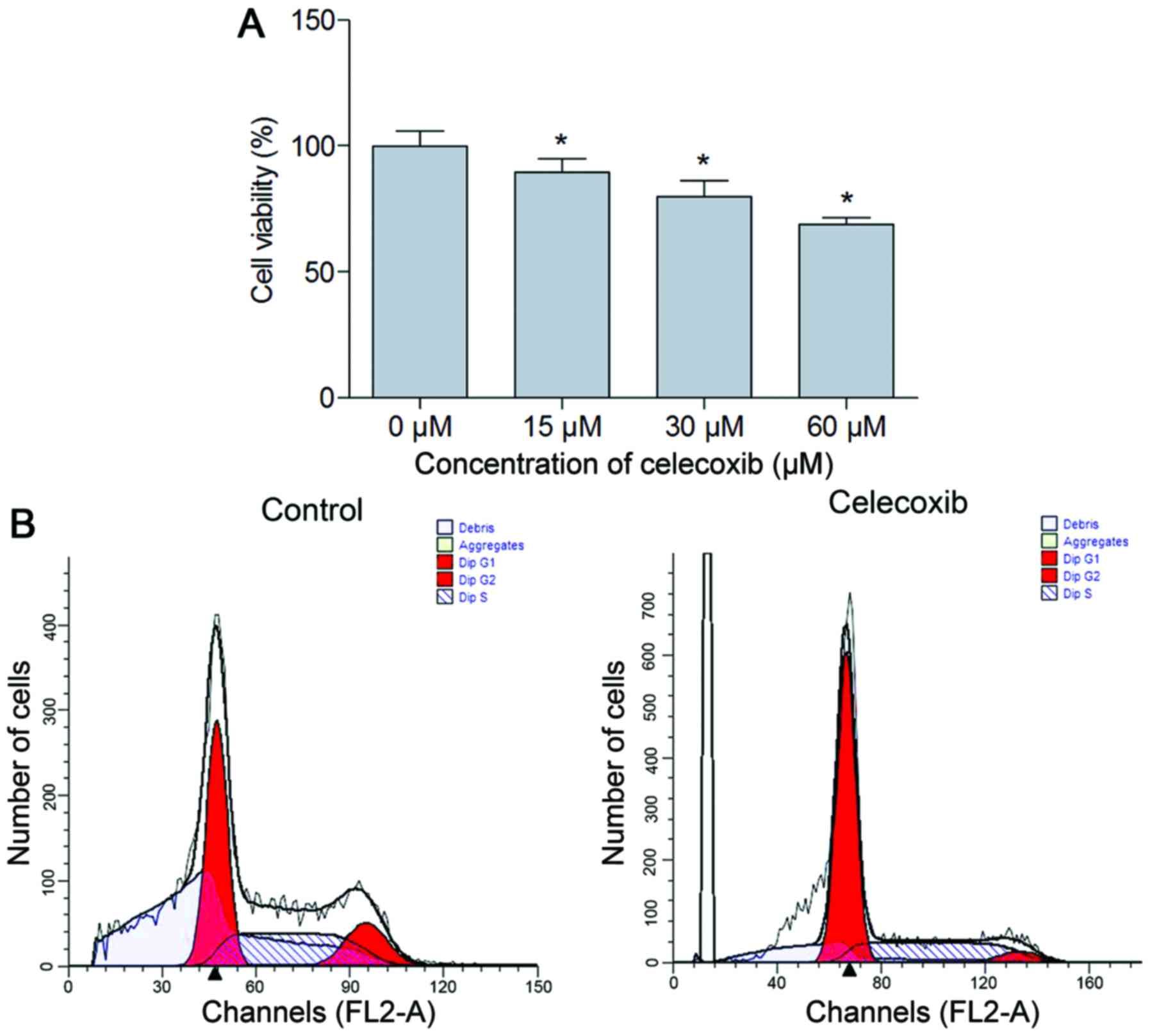
MTT analysis was performed to observe the effect of
celecoxib treatment on NCI-N87 cell viability. The results
demonstrated that cell viability was significantly inhibited in the
celecoxib treatment group compared with the untreated cells
(P<0.05; Fig. 1A). To further
understand the nature of the decrease in cell viability associated
with the addition of celecoxib, cell cycle analysis was performed.
The results suggested that compared with the control, there was an
increased percentage of cells in the G0/G1
phase (P<0.05) and a decreased percentage of cells in the S and
G2 phase in the celecoxib (15 µM) treatment group
(P<0.05; Fig. 1B, Table II).
 | Table II.Cell cycle analysis of celecoxib
treatment and control groups. |
Table II.
Cell cycle analysis of celecoxib
treatment and control groups.
| Group |
G0/G1 phase (%) | S phase (%) | G2 phase
(%) |
|---|
| Control | 47.07±0.70 | 39.05±4.34 | 13.88±3.65 |
| Celecoxib |
65.56±0.16a |
29.19±0.98a |
5.25±1.16a |
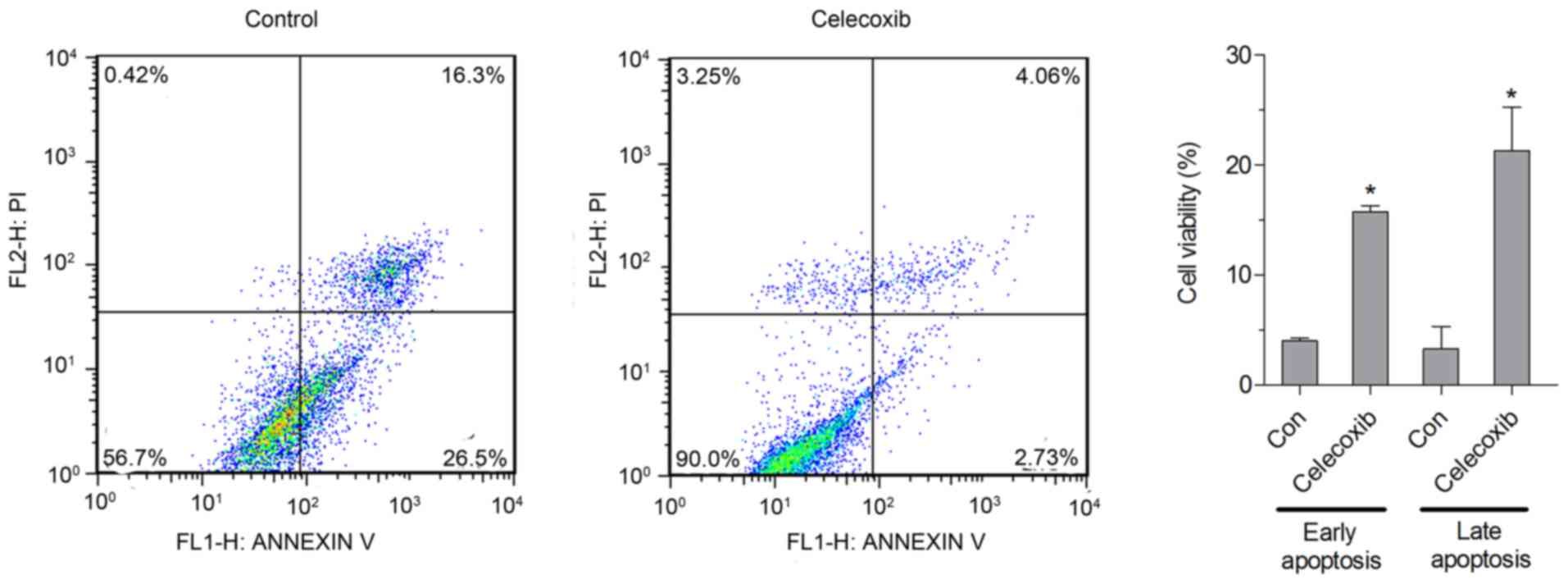
Effect of celecoxib on apoptosis in
NCI-N87 cells
The apoptotic rate was analyzed by flow cytometry,
and the results indicated that the percentage of early and late
apoptotic cells was significantly increased in cells treated with
celecoxib (15 µM) compared with untreated cells (P<0.05;
Fig. 2).
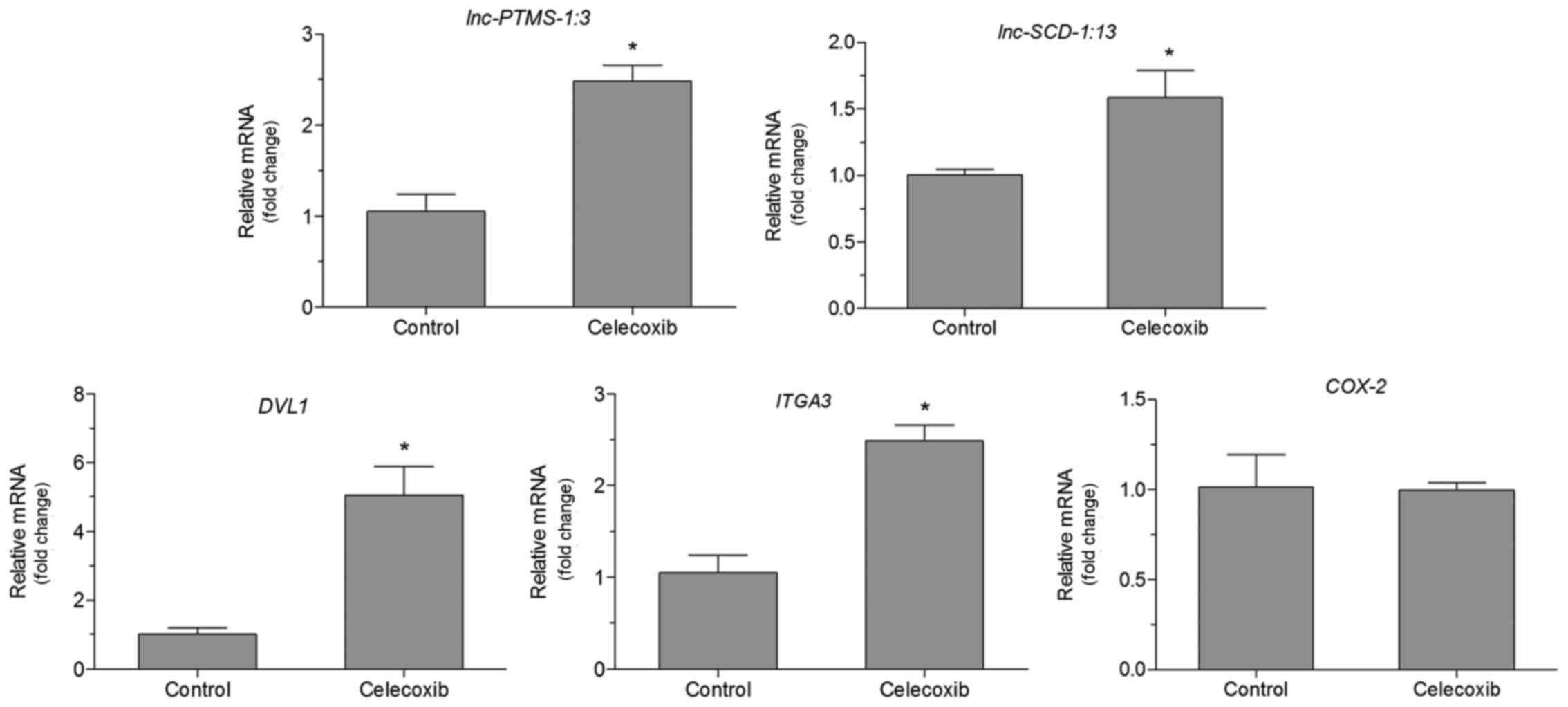
Effect of a low dose of celecoxib on
the mRNA levels of lnc-SCD-1:13, lnc-PTMS-1:3, COX2, ITGA3 and DVL1
in NCI-N87 cells
There was no significant difference in the
expression of COX2 in cells treated with celecoxib (15 µM)
and untreated cells (Fig. 3). The
mRNA levels of lnc-SCD-1:13, lnc-PTMS-1:3,
ITGA3 and DVL1 were also detected. The expression of
these genes was indicated to be aberrant in GC by our previous
bioinformatics analysis (17).
RT-qPCR analysis demonstrated that there was an increase in mRNA
expression of lnc-SCD-1:13, lnc-PTMS-1:3 ITGA3 and
DVL1 in cells treated with celecoxib (15 µM) compared with
untreated cells (P<0.05, Fig. 3).
Expression of COX2 was not significantly altered in cells
treated with celecoxib (15 µM) compared with untreated cells
(Fig. 3).
Discussion
Celecoxib, a novel NSAID that is able to inhibit
COX-2 activity, is considered to be an agent for the
chemoprevention of GC (18). A
previous study demonstrated that increased COX-2 expression is an
independent prognostic factor for poor prognosis and is associated
with reduced survival in patients with GC (19). Therefore, increasing attention had
been directed to the mechanisms of celecoxib action on GC. In the
present study, it was demonstrated that a low dose of celecoxib (15
µM) was able to significantly inhibit viability of NCI-N87 cells by
arresting the cell cycle at the the G0/G1
phase and promoting apoptosis. Similarly, Cho et al
(20) demonstrated the anti-cancer
effect of celecoxib on GC cells (AGS and MKN-45) by regulating
cell-cycle arrest and apoptosis. In addition, Liu et al
(11) suggested that celecoxib
induced apoptosis through the mitochondrial and death receptor
pathways in GC cells. These results indicated that a low dose of
celecoxib may have a chemopreventive function in the development of
GC by regulating cell cycle and apoptosis.
The association of COX2 with tumor development by
promoting cell proliferation and invasion or metastasis has been
well established (21,22). Previous studies have demonstrated that
COX-2 is overexpressed in human GC (23,24), and
in vitro downregulation of COX-2 is able to induce growth
inhibition (25). However, the
present study indicated that 15 µM celecoxib was not able inhibit
COX2 mRNA expression, but was able to suppress cell
viability. Consistent with the findings of the present study, Kim
et al (26) also demonstrated
that COX2 expression is not inhibited by 10 µM celecoxib, but
expression is suppressed by 25 µM celecoxib. Taken together, these
results indicated that the anti-cancer effect of celecoxib may not
be fully dependent on COX-2 suppression. Previous studies have also
demonstrated that chemopreventive effect of celecoxib on cancer may
be associated with a COX-2-independent mechanism (13,27–29).
Combined with the results of the present study, it is possible to
hypothesize that a low dose of celecoxib may inhibit cell growth
independent of COX-2. However, the exact mechanism requires further
elucidation.
To further investigate the molecular mechanisms
underlying the inhibition of cell viability induced by a low dose
of celecoxib, the mRNA levels of differentially expressed genes
(ITGA3 and DVL1) and lncRNAs (lnc-SCD-1:13 and
lnc-PTMS-1:3) were detected in NCI-N87 cells. ITGA3 has been
previously been demonstrated to be involved in the development of
GC (30). Integrins, a family of
adhesion receptors, are associated with cell adhesion and migration
as well as signal transduction (31,32).
Several previous studies have also demonstrated that decreased
expression of ITGA3 is associated with cancer growth and
development (33,34). In addition, overexpression of DVL1 was
observed in the metastasis of colorectal cancer (35). Dishevelled homologs (DVL1, DVL2, DVL2)
are core signaling molecules of the WNT/planar cell polarity (PCP)
signaling pathways (36).
Accumulating evidence has demonstrated that PCP signaling is
associated with tumorigenesis (31).
Tang et al (37) reported that
microRNA (miR)-200b and miR-22 were able to synergistically inhibit
the growth of GC through the Wnt-1 signaling pathway. Notably, a
previous study by our group demonstrated that ITGA3 and/or DVL1
were co-expressed with lnc-SCD-1:13 and lnc-PTMS-1:3 in GC cells.
Further studies have reported that lncRNAs are involved in the
pathogenesis of GC (38–40). These results suggested that genes
co-expressed with lnc-SCD-1:13 and lnc-PTMS-1:3 may be involved in
the effects of a low dose of celecoxib on GC. However, the
associations of ITGA3 and DVL1 with lnc-SCD-1:13 and lnc-PTMS-1:3
remain unclear, and should be investigated in further studies.
In summary, the present study demonstrated that a
low dose of celecoxib may exert an anti-cancer effect by regulating
the cell cycle and apoptosis independent of COX-2 in GC cells.
Furthermore, ITGA3 and/or DVL1 co-expressed with
lnc-SCD-1:13 and lnc-PTMS-1:3 may be involved in the effects of a
low dose of celecoxib on GC.
Acknowledgements
The present study was supported by the Special Fund
for Medical Service of Jilin Finance Department project (grant no.
SCZSY201507).
References
|
1
|
Bray F, Ren JS, Masuyer E and Ferlay J:
Global estimates of cancer prevalence for 27 sites in the adult
population in 2008. Int J Cancer. 132:1133–1145. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ferlay J, Shin HR, Bray F, Forman D,
Mathers C and Parkin DM: Estimates of worldwide burden of cancer in
2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Akagi H, Higuchi H, Sumimoto H, Igarashi
T, Kabashima A, Mizuguchi H, Izumiya M, Sakai G, Adachi M,
Funakoshi S, et al: Suppression of myeloid cell leukemia-1 (Mcl-1)
enhances chemotherapy-associated apoptosis in gastric cancer cells.
Gastric Cancer. 16:100–110. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Harris RE, Namboodiri KK and Farrar WB:
Nonsteroidal antiinflammatory drugs and breast cancer.
Epidemiology. 7:203–205. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Funkhouser EM and Sharp GB: Aspirin and
reduced risk of esophageal carcinoma. Cancer. 76:1116–1119. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Levy GN: Prostaglandin H synthases,
nonsteroidal anti-inflammatory drugs and colon cancer. FASEB J.
11:234–247. 1997.PubMed/NCBI
|
|
7
|
Hsu AL, Ching TT, Wang DS, Song X,
Rangnekar VM and Chen CS: The cyclooxygenase-2 inhibitor celecoxib
induces apoptosis by blocking Akt activation in human prostate
cancer cells independently of Bcl-2. J Biol Chem. 275:11397–11403.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang WH, Huang JQ, Zheng GF, Lam SK,
Karlberg J and Wong BC: Non-steroidal anti-inflammatory drug use
and the risk of gastric cancer: A systematic review and
meta-analysis. J Natl Cancer Inst. 95:1784–1791. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pairet M and Engelhardt G: Distinct
isoforms (COX-1 and COX-2) of cyclooxygenase: Possible
physiological and therapeutic implications. Fundam Clin Pharmacol.
10:1–17. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Williams CS, Mann M and Dubois RN: The
role of cyclooxygenases in inflammation, cancer, and development.
Oncogene. 18:7908–7916. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu M, Li CM, Chen ZF, Ji R, Guo QH, Li Q,
Zhang HL and Zhou YN: Celecoxib regulates apoptosis and autophagy
via the PI3K/Akt signaling pathways in SGC-7901 gastric cancer
cells. Int J Mol Med. 33:1451–1458. 2014.PubMed/NCBI
|
|
12
|
Lan C, Yang L, Fan L, Zhang Y, Wang J, Guo
GJ, Wan S, Yang S, Wang R and Fang D: Celecoxib inhibits
helicobacter pylori-induced invasion of gastric cancer cells
through an adenine nucleotide translocator-dependent mechanism.
Anticancer Agents Med Chem. 13:1267–1272. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hu P, Yu J, Zeng Z, Leung WK, Lin HL, Tang
BD, Bai AH and Sung JJ: Chemoprevention of gastric cancer by
celecoxib in rats. Gut. 53:195–200. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Du Z, Fei T, Verhaak RG, Su Z, Zhang Y,
Brown M, Chen Y and Liu XS: Integrative genomic analyses reveal
clinically relevant long noncoding RNAs in human cancer. Nat Struct
Mol Biol. 20:908–913. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Passon DM, Lee M, Rackham O, Stanley WA,
Sadowska A, Filipovska A, Fox AH and Bond CS: Structure of the
heterodimer of human NONO and paraspeckle protein component 1 and
analysis of its role in subnuclear body formation. Proc Natl Acad
Sci USA. 109:pp. 4846–4850. 2012; View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Song B, Du J, Feng Y, Gao YJ and Zhao JS:
Co-expressed differentially expressed genes and long non-coding
RNAs involved in the celecoxib treatment of gastric cancer: An RNA
sequencing analysis. Exp Ther Med. 12:2455–2468. 2016.PubMed/NCBI
|
|
18
|
Yeh TS, Wu CW, Hsu KW, Liao WJ, Yang MC,
Li AF, Wang AM, Kuo ML and Chi CW: The activated Notch1 signal
pathways is associated with gastric cancer progression through
cyclooxygenase-2. Cancer Res. 69:5039–5048. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Thiel A, Mrena J and Ristimäki A:
Cyclooxygenase-2 and gastric cancer. Cancer Metastasis Rev.
30:387–395. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cho SJ, Kim N, Kim JS, Jung HC and Song
IS: The anti-cancer effect of COX-2 inhibitors on gastric cancer
cells. Dig Dis Sci. 52:1713–1721. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fu SL, Wu YL, Zhang YP, Qiao MM and Chen
Y: Anti-cancer effects of COX-2 inhibitors and they correlation
with angiogenesis and invasion in gastric cancer. World J
Gastroenterol. 10:1971–1974. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Greenhough A, Smartt HJ, Moore AE, Roberts
HR, Williams AC, Paraskeva C and Kaidi A: The COX-2/PGE2 pathways:
Key roles in the hallmarks of cancer and adaptation to the tumour
microenvironment. Carcinogenesis. 30:377–386. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ristimäki A, Honkanen N, Jänkälä H,
Sipponen P and Härkönen M: Expressions of cyclooxygenase-2 in human
gastric carcinoma. Cancer Res. 57:1276–1280. 1997.PubMed/NCBI
|
|
24
|
Uefuji K, Ichikura T and Mochizuki H:
Cyclooxygenase-2 expressions is relate to prostaglandin
biosynthesis and angiogenesis in human gastric cancer. Clin Cancer
Res. 6:135–138. 2000.PubMed/NCBI
|
|
25
|
Tsuji S, Kawano S, Sawaoka H, Takei Y,
Kobayashi I, Nagano K, Fusamoto H and Kamada T: Evidences for
involvement of cyclooxygenase-2 in proliferation of two
gastrointestinal cancer cell lines. Prostaglandins Leukot Essent
Fatty Acids. 55:179–183. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kim N, Kim CH, Ahn DW, Lee KS, Cho SJ,
Park JH, Lee MK, Kim JS, Jung HC and Song IS: Anti-gastric cancer
effects of celecoxib, a selective COX-2 inhibitor, through
inhibition of Akt signaling. J Gastroenterol Hepatol. 24:480–487.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Charalambous D and O'brien PE: Inhibition
of colon cancer precursors in the rat by sulindac sulphone is not
dependent on inhibition of prostaglandin synthesis. J Gastroenterol
Hepatol. 11:307–310. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Elder D, Halton DE, Hague A and Paraskeva
C: Induction of apoptotic cell death in human colorectal carcinoma
cell lines by a cyclooxygenase-2 (COX-2)-selective nonsteroidal
anti-inflammatory drug: Independence from COX-2 protein
expressions. Clin Cancer Res. 3:1679–1683. 1997.PubMed/NCBI
|
|
29
|
Hanif R, Pittas A, Feng Y, Koutsos MI,
Qiao L, Staiano-Coico L, Shiff SI and Rigas B: Effects of
nonsteroidal anti-inflammatory drugs on proliferation and on
induction of apoptosis in colon cancer cells by a
prostaglandin-independent pathways. Biochem Pharmacol. 52:237–245.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Song X, Zhong H, Zhou J, Hu X, Zhou Y, Ye
Y, Lu X, Wang J, Ying B and Wang L: Association between
polymorphisms of microRNA-binding sites in integrin genes and
gastric cancer in Chinese Han population. Tumor Biol. 36:2785–2792.
2015. View Article : Google Scholar
|
|
31
|
Wang Y: Wnt/Planar cell polarity
signaling: A new paradigm for cancer therapy. Mol Cancer Ther.
8:2103–2109. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hemler ME: Integrin associated proteins.
Curr Opin Cell Biol. 10:578–585. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dedhar S, Saulnier R, Nagle R and Overall
CM: Specific alterations in the expressions of α3β1 and α6β4
integrins in highly invasive and metastatic variants of human
prostate carcinoma cells selected by in vitro invasion through
reconstituted basement membrane. Clin Exper Met. 11:391–400. 1993.
View Article : Google Scholar
|
|
34
|
Sampson-Johannes A, Wang W and Shtivelman
E: Colonization of human lung grafts in SCID-hu mice by human colon
carcinoma cells. Int J Cancer. 65:864–869. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Huang MY, Yen LC, Liu HC, Liu PP, Chung
FY, Wang TN, Wang JY and Lin SR: Significant overexpression of DVL1
in taiwanese colorectal cancer patients with liver metastasis. Int
J Mol Sci. 14:20492–20507. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Katoh M: WNT/PCP signaling pathways and
human cancer (review). Oncol Rep. 14:1583–1588. 2005.PubMed/NCBI
|
|
37
|
Tang H, Kong Y, Guo J, Tang Y and Xie X,
Yang L, Su Q and Xie X: Diallyl disulfide suppresses proliferation
and induces apoptosis in human gastric cancer through Wnt-1
signaling pathways by up-regulation of miR-200b and miR-22. Cancer
Lett. 340:72–81. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lin XC, Zhu Y, Chen WB, Lin LW, Chen DH,
Huang JR, Pan K, Lin Y, Wu BT, Dai Y and Tu ZG: Integrated analysis
of long non-coding RNAs and mRNA expressions profiles reveals the
potential role of lncRNAs in gastric cancer pathogenesis. Int J
Oncol. 45:619–628. 2014.PubMed/NCBI
|
|
39
|
Chen S, Li P, Xiao B and Guo J: Long
noncoding RNA HMlincRNA717 and AC130710 have been officially name
as gastric cancer associated transcript 2 (GACAT2) and GACAT3,
respectively. Tumor Biol. 35:8351–8352. 2014. View Article : Google Scholar
|
|
40
|
Okugawa Y, Toiyama Y, Hur K, Toden S,
Saigusa S, Tanaka K, Inoue Y, Mohri Y, Kusunoki M, Boland CR and
Goel A: The expressions of metastasis-associated long non-coding
RNA, HOTAIR, is involved in cancer development and peritoneal
metastasis in gastric cancer. Cancer Res. 74:3553. 2014. View Article : Google Scholar
|