Introduction
Pancreatic cancer an aggressive malignancy that is
one of the leading causes of cancer-associated mortality worldwide
(1). Due to the lack of effective
treatment for pancreatic adenocarcinoma, the prognosis of this
cancer type is very poor (1,2). At present, the molecular mechanisms
underlying pancreatic cancer remain unclear. Understanding how
cancer-associated signaling molecules cooperate to regulate the
pathogenesis of pancreatic cancer may aid in the development of
novel treatments.
Curcumin is the primary active ingredient of the
traditional Chinese medicinal plant Curcuma longa. Curcumin
has been demonstrated to exert pleotropic beneficial functions,
including antioxidative and anticancer effects (3–5). In
addition, several previous studies have demonstrated that curcumin
promotes apoptosis and inhibits the proliferation of pancreatic
cancer cells (6–8). Previous studies have suggested that
curcumin sensitizes drug-resistant cancer cells to chemotherapy,
which indicates that curcumin may be effective in treating
drug-resistant pancreatic cancer (9–11). Several
intracellular targets of curcumin have been identified (12,13);
however, the molecular mechanism by which curcumin inhibits tumor
growth remains unclear.
MicroRNAs (miRNAs/miRs) are a class of small
non-coding RNAs that regulate gene expression and result in altered
phenotypic changes in cancer cells. miRNAs have emerged as
promising drug targets, and miRNA-based therapies have been
proposed in various cancer models (14,15).
Several miRNAs have been revealed to serve key roles in the
development and progression of pancreatic cancer (16–18). In
addition, previous studies have demonstrated that miRNA signatures
change following chemotherapy (19,20).
Curcumin, a natural compound that exhibits low toxicity and yields
strong antitumor effects, has been reported to significantly alter
the miRNA expression profile in pancreatic cancers (21,22). This
indicates that miRNAs may mediate the antitumor effects of
curcumin.
In the present study, miR-340, a tumor-suppressive
miRNA (23), was investigated as a
potential miRNA associated with the antitumor effects of curcumin
on pancreatic cancer cells. This investigation revealed that
miR-340 expression was markedly induced by curcumin treatment. In
addition, an antiapoptotic protein named X-linked inhibitor of
apoptosis (XIAP) was functionally targeted by miR-340, which likely
confers the proapoptotic effect of curcumin. Therefore, the present
study demonstrates that the miR-340/XIAP signaling pathway is
essential for curcumin-induced pancreatic cancer cell apoptosis and
may provide clinical insights for treating pancreatic cancer using
curcumin.
Materials and methods
Cell culture, drug treatment and
transfection
The pancreatic adenocarcinoma cell line PANC-1 and
human embryonic kidney HEK293 cell line were purchased from the
American Type Culture Collection (Manassas, VA, USA). Cells were
cultured in Dulbecco's modified Eagle's medium (HyClone; GE
Healthcare Life Sciences, Logan, UT, USA) supplemented with 10%
fetal bovine serum (HyClone; GE Healthcare Life Sciences) in an
incubator with 5% CO2 at 37°C. Negative controls for
miR-340 and miR-340 inhibitor (NC; scrambled miRNA, supplied in
each miRNA set), miR-340 (catalog no. miR10004692-1-5) or an
miR-340 inhibitor (catalog no. miR20004692-1-5; Guangzhou RiboBio,
Co., Ltd., Guangzhou, China) were transfected into cells using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) at a final concentration of 100
nM according to the manufacturer's protocol. To avoid bacterial
contamination, 100 U/ml penicillin and 100 µg/ml streptomycin were
added to the culture media following transfection. Curcumin was
purchased from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany) and
applied to cells at 2.5, 5, 10 or 20 µM for 72 h. Cells were
subjected to curcumin treatment 48 h following transfection.
Cell viability assay
Cell viability was measured using an MTT assay.
PANC-1 cells were seeded into a 96-well plate at a concentration of
2.5×104 cells/ml/well. Following cell attachment for 24
h, cells were treated with 2.5, 5, 10 or 20 µmol/l curcumin for 72
h, followed by incubation with 20 µl MTT (5 mg/ml) for 4 h.
Dimethyl sulfoxide (200 µl) was then added to each well to
visualize the living cells. The raw data were obtained by measuring
the absorbance value of the wells at 490 nm with a
spectrophotometer.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Following curcumin treatment, the cells were
homogenized with TRIzol reagent (Invitrogen; Thermo Fisher
Scientific, Inc.). A total of 0.2 ml chloroform was then used to
separate the upper phase that contained total RNA. The RNA samples
were precipitated using isopropyl alcohol and washed with 70%
ethanol. All the RNA samples were resolved in nuclease free water
(Promega Corporation, Madison, WI, USA). To quantify the level of
miR-340, the RNA samples were reverse-transcribed using a stem-loop
primer set for miR-340 (catalog no. miRQ0004692-1-1 Guangzhou
RiboBio, Co., Ltd.). The internal control gene used was U6
(provided in the miR-340 set, catalog no. miRQ0004692-1-1). Each
qPCR volume was 25 µl in total and the components were as follows:
12.5 µl 2X SYBR Green mastermix (Promega Corporation, Madison, WI,
USA), 7.5 µl nuclease-free water and 5 µl cDNA template. The
thermocycling conditions for qPCR were as follows: 94°C for 30 sec,
58°C for 20 sec and 72°C for 20 sec, for a total of 40 cycles. The
fold change in miR-340 was calculated using the 2−ΔΔCq
method (24).
Western blot analysis
Curcumin-treated cells were lysed using SDS lysis
buffer supplemented with a protease inhibitor cocktail (Roche
Diagnostics GmbH, Mannheim, Germany) and untreated cells were used
as the control. The lysates were centrifuged at 12,000 × g for 15
min at 4°C. The supernatants were collected and mixed with SDS-PAGE
buffer prior to protein denaturation at 100°C for 5 min prior to
electrophoresis. The proteins (30 µg/lane) were separated via
SDS-PAGE (12% gel) and then transferred onto polyvinylidene
difluoride membranes. Proteins of interest were probed with their
respective primary antibodies overnight at 4°C. Rabbit polyclonal
antibodies for cleaved caspase-3 (dilution, 1:1,000; catalog no.
9661), poly(ADP-ribose) polymerase (PARP) (dilution, 1:500; catalog
no. 9542) and XIAP (dilution, 1:1,000; catalog no. 2042) were
obtained from Cell Signaling Technology, Inc. (Danvers, MA, USA),
and a mouse monoclonal antibody for β-actin (dilution, 1:2,000;
catalog no. A5441) was purchased from Sigma-Aldrich (Merck KGaA).
Horseradish peroxidase-conjugated goat anti-rabbit (dilution,
1:5,000; catalog no. sc-2004) and goat anti-mouse (dilution,
1:5,000; catalog no. sc-2005) secondary antibodies (Santa Cruz
Biotechnology, Inc., Dallas, TX, USA) were subsequently incubated
at room temperature for 1 h and used according to the host species
of the primary antibodies. Protein bands were detected using the
BeyoECL Plus P0018 kit (Beyotime Institute of Biotechnology,
Haimen, China), and band intensities were determined using ImageJ
software (version 2.1.4.7; National Institutes of Health, Bethesda,
MD, USA).
Luciferase activity assay
To construct the luciferase reporter, a 600 bp
fragment of the XIAP 3′-untranslated region (UTR) that was
extracted from the PANC1 cell line was amplified by PCR. Human
cDNA, extracted from the PANC1 cell line, was used as a template
and Pfu DNA polymerase (Promega Corporation) was used. The primers
were as follows: Forward primer, 5′-TTCCTCAGCATAACAGAGTT-3′;
reverse primer, 5′-CAGGCTGGCATCATATACT-3′. Subsequently, this was
subcloned into the pmirGLO plasmid. The plasmid containing the XIAP
3′-UTR and a pRL-TK plasmid, in addition to miR-340, anti-miR-340
or the NC was transfected into HEK293 cells (2.5×104
cells/ml) that were seeded 1 day before transfection. Luciferase
activity was tested 48 h after transfection. Cells were lysed,
using lysis buffer (Dual Luciferase kit; Promega Corporation), and
20 µl supernatant was used for each reaction. A total of 75 µl
firefly luciferase substrate was added to each mixture, mixed for
10 sec and the luciferase activity was determined. Subsequently, 75
µl Renilla luciferase substrate was added to each well and the
activity was determined. Relative activity was determined as the
ratio of firefly luciferase to Renilla luciferase using a
GloMax®-Multi Detection System (Promega Corporation)
according to the manufacturer's protocol. Experiments were
performed in triplicate. All the reagents use in the luciferase
activity were obtained from Promega Corporation (Madison, WI,
USA).
Identification of downstream targets
of miR-340
To investigate the mechanism of the proapoptotic
effect of miR-340, the online miRanda database (www.microrna.org) was searched to identify downstream
targets of miR-340. The miR-mRNA interaction was searched, using
the criteria of mirSVR score <-0.2 and PhastCons score
>0.5.
Statistical analysis
SPSS software (version 19.0; IBM Corp., Armonk, NY,
USA) was used to perform statistical tests. All data are presented
as the mean ± standard deviation. A one-way analysis of the
variance and post hoc Student-Newman-Keuls test was used to test
for statistically significant differences. P<0.05 was considered
to indicate a statistically significant significance.
Results
Curcumin induces the apoptosis of
PANC-1 cells
As curcumin has previously been reported to exert
anti-proliferative actions on pancreatic cancer cells, the effect
of various doses of curcumin on the pancreatic cancer cell line
PANC-1 was examined. Cells were treated with curcumin for 72 h and
the effect on cell viability was investigated using an MTT assay.
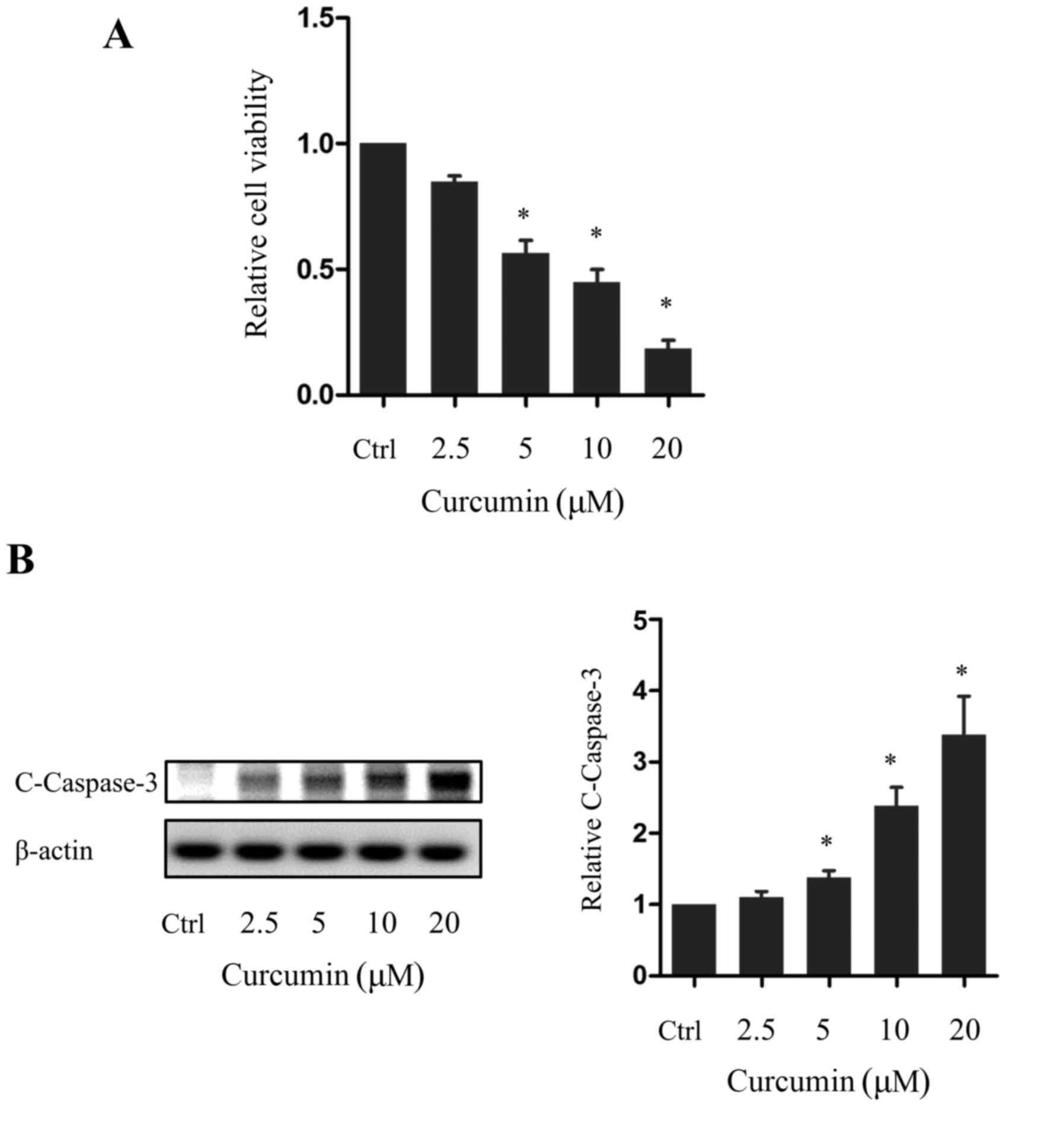
PANC-1 cell viability was decreased in a dose-dependent manner by
curcumin, with the decrease being significant in cells treated with
5, 10 or 20 µm curcumin compared with the control group (P<0.05;
Fig. 1A). In addition, western blot
analysis revealed that the expression of cleaved caspase-3, an
apoptotic marker, was increased in a dose-dependent manner by
curcumin, with this difference again being significant in cells
treated with 5, 10 or 20 µm curcumin compared with the control
group (P<0.05; Fig. 1B).
Consistent with previous reports, these results indicated that
curcumin induces the apoptosis of pancreatic cancer cells.
miR-340 confers the proapoptotic
effect of curcumin
To explore the potential molecular mechanism
underlying the antitumor effect of curcumin, the effects of
curcumin treatment on the level of miR-340, a previously
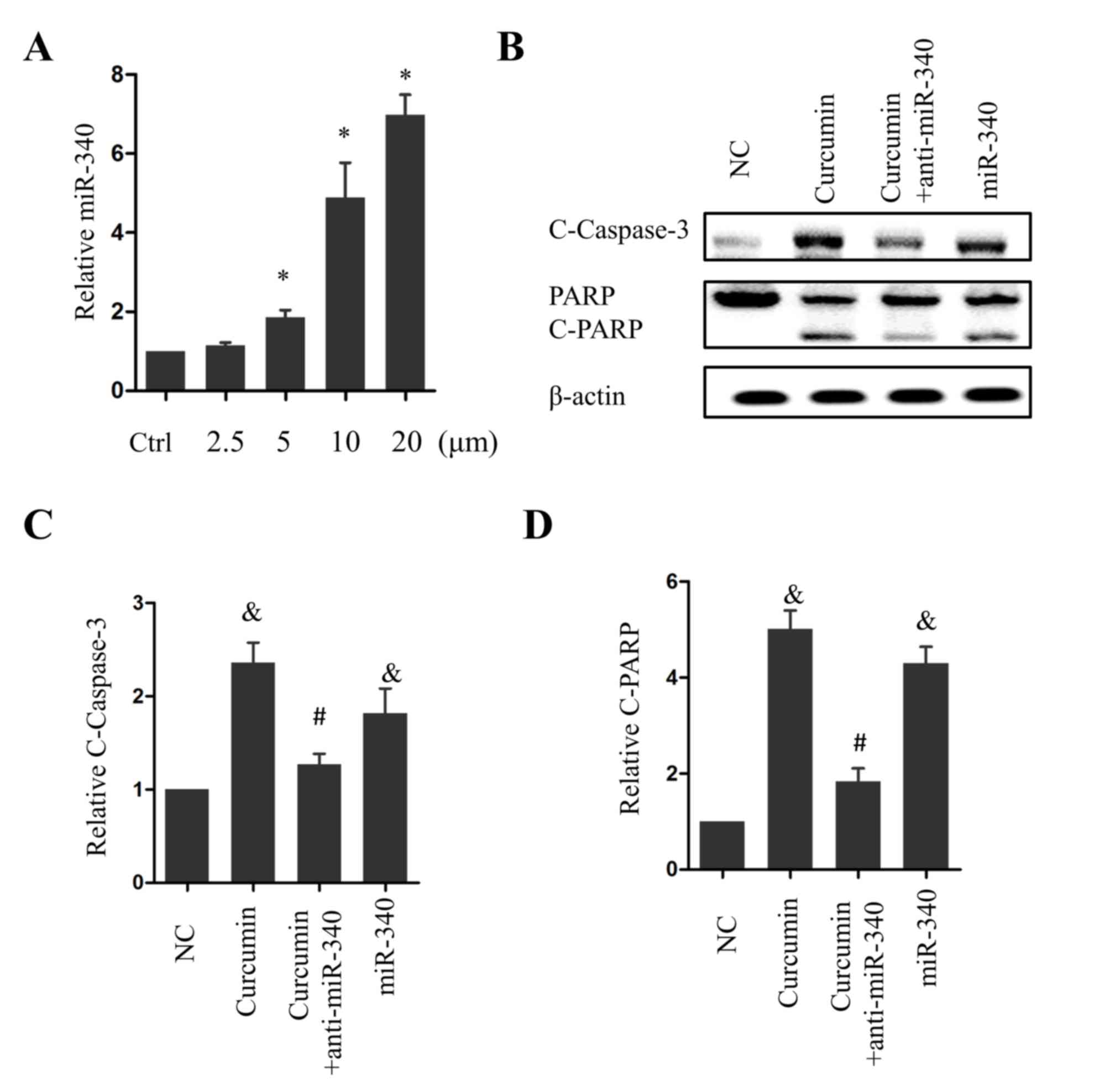
characterized tumor-suppressive miRNA, was investigated. Notably,
miR-340 levels were significantly elevated following treatment with
5, 10 or 20 µM curcumin (P<0.05 vs. the control group; Fig. 2A). Overexpression of miR-360
significantly increased caspase-3 cleavage compared with the NC
group (P<0.05; Fig. 2B and C), and
PARP, a substrate of caspase-3, was cleaved as a result (Fig. 2B and D). The cotreatment of PANC-1
cells with 10 µM curcumin and anti-miR-340 significantly inhibited
caspase-3 and PARP cleavage (P<0.05 vs. curcumin treatment
alone; Fig. 2B-D). These results
suggest that miR-340 is a positive regulator of apoptosis and acts
as a downstream effector of curcumin in pancreatic cancer
cells.
XIAP is a target of miR-340
To investigate the mechanism of the proapoptotic
effect of miR-340, the online miRanda database was searched to
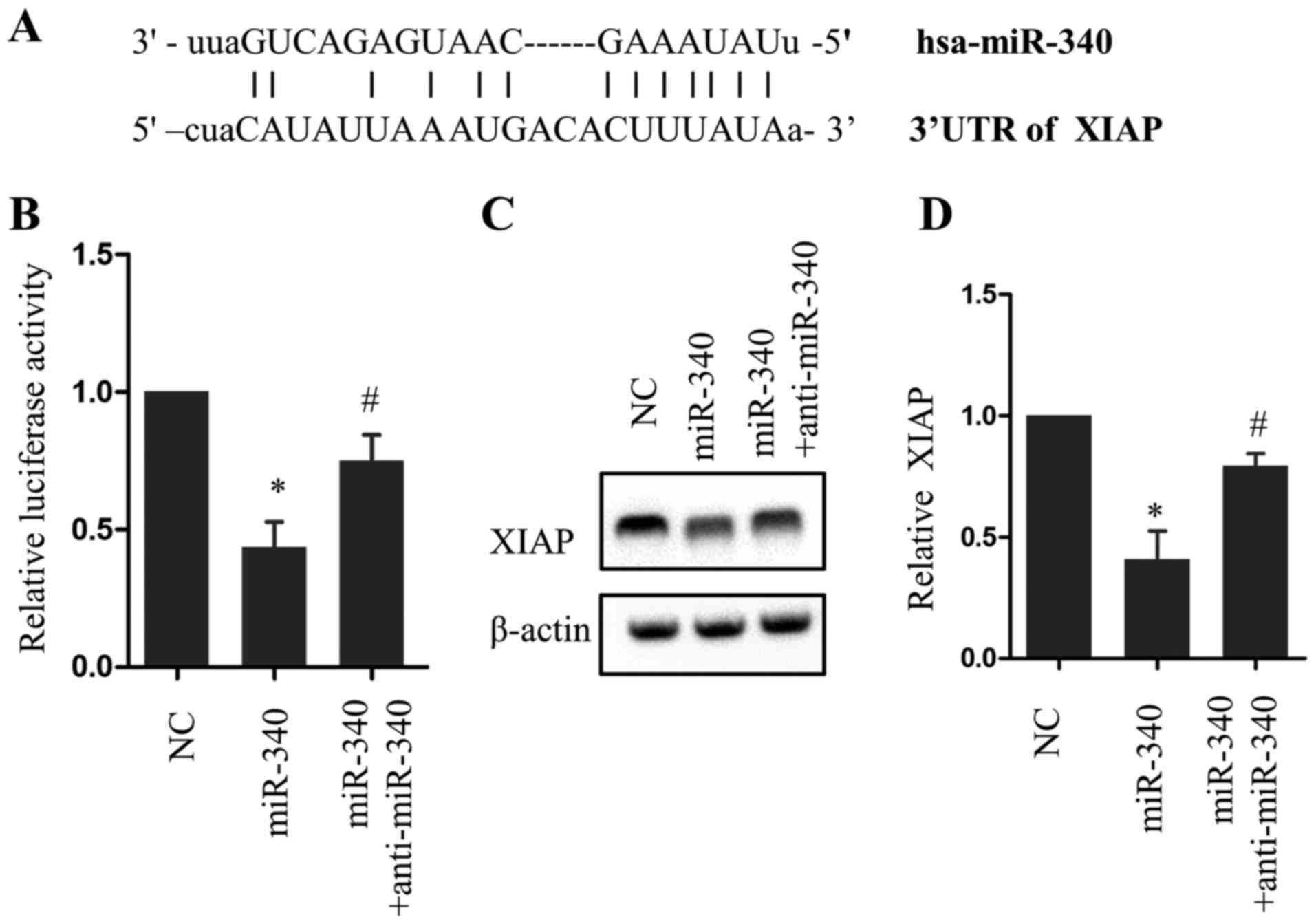
identify downstream targets of miR-340. The 3′-UTR of XIAP was
analyzed and a potential incomplete base pair match with miR-340
was identified (Fig. 3A). A
luciferase reporter assay revealed that overexpression of miR-340
significantly reduced luciferase activity compared with the NC
group (P<0.05), an effect that was partially rescued with
anti-miR-340 (Fig. 3B). Consistent
with this result, miR-340 and anti-miR-340 exerted opposing effects
on XIAP protein expression (Fig. 3C and
D). These results indicate a direct association between miR-340
and XIAP.
Curcumin activates the miR-340/XIAP
signaling pathway
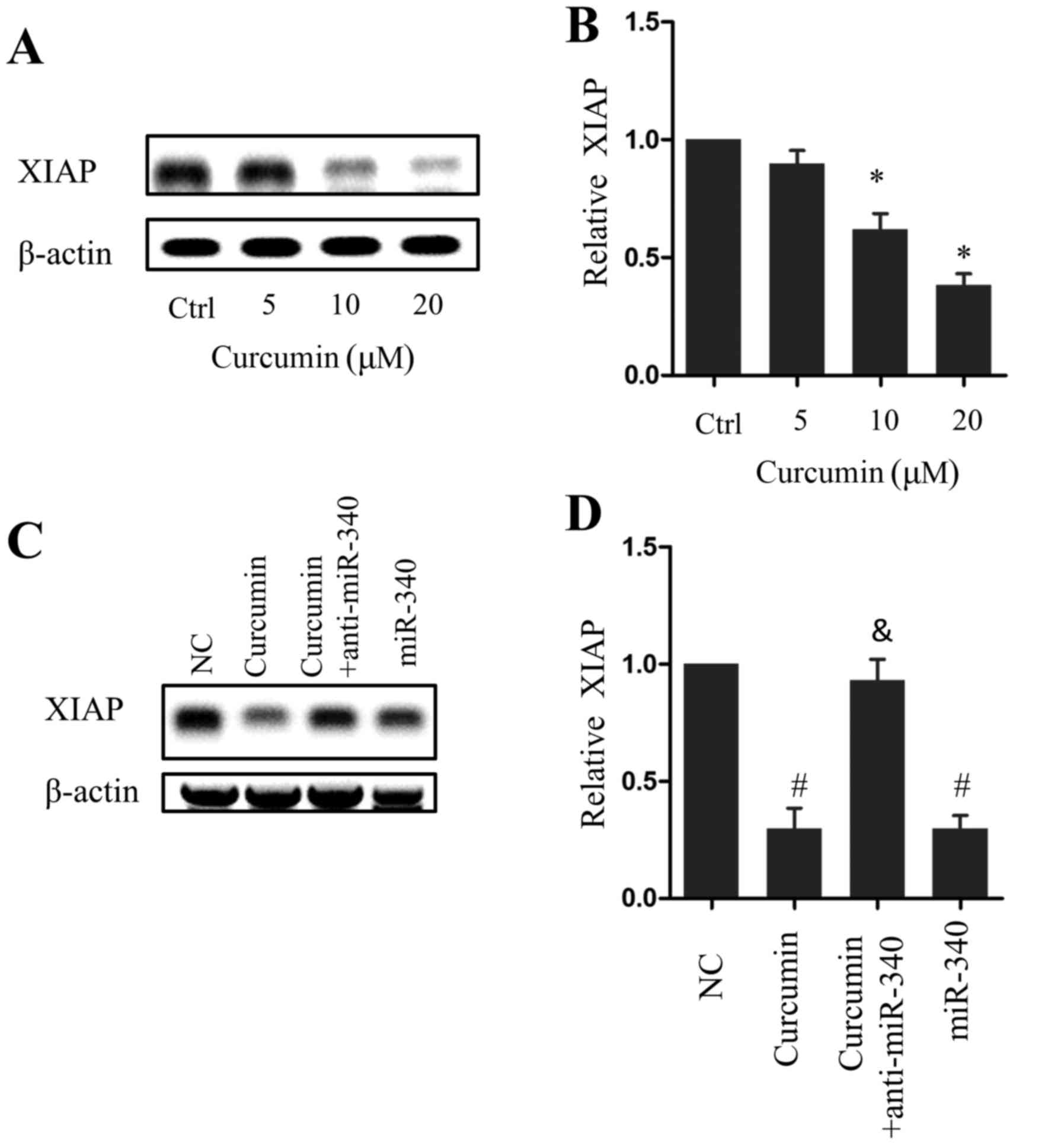
XIAP expression decreased in a dose-dependent manner
following curcumin treatment, which was significant in cells
treated with 10 or 20 µM curcumin compared with the control group
(P<0.05; Fig. 4A and B).
Similarly, miR-340 overexpression led to a significant decrease in
XIAP expression compared with the NC group (P<0.05), whereas
anti-miR-340 treatment rescued XIAP expression in the presence of
10 µM curcumin (Fig. 4C and D). These
results indicate that the miR-340/XIAP signaling pathway serves a
role in the curcumin-induced apoptosis of pancreatic cancer
cells.
Discussion
Developing an effective treatment strategy for
pancreatic cancer remains a significant challenge worldwide. It has
been suggested that miRNAs could form the basis for novel cancer
treatments; however, there are limited successful delivery methods
for miRNAs and they exhibit relatively low bioavailability.
Curcumin has been demonstrated to alter the expression profile of a
number of miRNAs in pancreatic cancer (22). Data from the present study confirmed
that curcumin exerts an antitumor effect on PANC-1 cells by
inducing apoptotic cell death. Notably, miR-340 was significantly
upregulated following curcumin treatment. Curcumin treatment
significantly decreased XIAP expression; however, silencing
endogenous miR-340 abrogated the proapoptotic effect of curcumin
and increased XIAP expression, suggesting that the miR-340/XIAP
signaling pathway is responsible for the proapoptotic effect of
curcumin. To the best of our knowledge, the present study clarifies
for the first time that the miR-340/XIAP pathway is associated with
the antitumor action of curcumin. These findings may also aid in
the development of targeted treatments for patients with pancreatic
cancer.
Previous studies have demonstrated that miR-340
serves an antitumor role in several types of cancers (25–27). A
number of mechanisms have been proposed to explain this effect. For
example, a set of negative regulators of p27 have been demonstrated
to be targeted by miR-340 in non-small cell lung cancer (NSCLC)
(27). Notably, clinical data
supports an inverse association between miR-340 and disease
progression in patients with NSCLC (27). In addition, several oncogenic
signaling pathways and associated proteins, including the Ras
GTPase/RAF-proto-oncogene serine/threonine-protein
kinase/mitogen-activated protein kinase 1 signaling pathway,
Rho-associated protein kinase 1 and proto-oncogene c-Met are
modulated by miR-340 (23,28,29), which
suggests that miR-340 may function upstream of a series of positive
regulators of cancer cell survival. The results from the present
study demonstrated a tumor suppressive role for miR-340 in
pancreatic cancer, identified XIAP as a target of miR-340 and
identified a mechanism by which miR-340 induces apoptosis.
Curcumin is a natural compound extracted from the
plant C. longa. Despite the previously demonstrated
antitumor activity of curcumin, the mechanism underlying this
effect remains elusive. Epigenetic changes induced by curcumin may
mediate its effects (30). Previous
studies have revealed that the expression of a number of miRNAs is
altered by curcumin. Ye et al (31) demonstrated that cellular tumor antigen
p53 (p53) is upregulated following curcumin treatment, and that the
miR-192/XIAP signaling pathway is a downstream target of p53 that
mediates the effects of curcumin treatment in NSCLC. Ma et
al (21) demonstrated that miR-7
mediates the tumor suppressive effects of curcumin in pancreatic
cancer cells. Similarly, the present study demonstrated that
curcumin increases miR-340 expression in pancreatic cancer cells.
The aforementioned studies by Ma et al (21) and Ye et al (31) demonstrated increased p53 expression
following curcumin treatment. The expression of a number of
antitumor miRNAs is regulated by p53 (32). However, whether miR-340 is regulated
by p53 remains unclear and requires further investigation.
Data from the present study indicated that XIAP is a
target of miR-340. XIAP inhibits caspase activity and subsequently
inhibits apoptosis (33); however,
following curcumin treatment, XIAP expression was identified to
decrease as a result of increased miR-340 expression, resulting in
the proapoptotic effect of curcumin. XIAP expression has previously
been associated with cancer development and progression (34). In addition, previous clinical studies
have revealed that XIAP may function as a biomarker for various
types of cancer (33,35–37). Data
from the present study demonstrated that the miR-340/XIAP signaling
pathway serves a role in curcumin-induced apoptosis, which suggests
that modulating this pathway may synergistically enhance the effect
of curcumin. Notably, the results of the present study do not
exclude the possibility of the involvement of other signaling
pathways downstream of curcumin and miR-340. Due to the pleiotropic
effects of natural compounds, other mechanisms may contribute to
the pro-apoptotic effect of curcumin.
In conclusion, the data from the present study
demonstrated that the miR-340/XIAP signaling pathway serves a role
in the curcumin-induced apoptosis of pancreatic cancer cells. This
suggests that administering the natural compound curcumin or
modulating endogenous miR-340 expression may be effective treatment
strategies for patients with pancreatic cancer.
References
|
1
|
Bond-Smith G, Banga N, Hammond TM and
Imber CJ: Pancreatic adenocarcinoma. BMJ. 344:e24762012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ryan DP, Hong TS and Bardeesy N:
Pancreatic adenocarcinoma. N Engl J Med. 371:1039–1049. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Casas-Grajales S and Muriel P:
Antioxidants in liver health. World J Gastrointest Pharmacol Ther.
6:59–72. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
He Y, Yue Y, Zheng X, Zhang K, Chen S and
Du Z: Curcumin, inflammation, and chronic diseases: How are they
linked? Molecules. 20:9183–9213. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shanmugam MK, Rane G, Kanchi MM, Arfuso F,
Chinnathambi A, Zayed ME, Alharbi SA, Tan BK, Kumar AP and Sethi G:
The multifaceted role of curcumin in cancer prevention and
treatment. Molecules. 20:2728–2769. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhao Z, Li C, Xi H, Gao Y and Xu D:
Curcumin induces apoptosis in pancreatic cancer cells through the
induction of forkhead box O1 and inhibition of the PI3K/Akt
pathway. Mol Med Rep. 12:5415–5422. 2015.PubMed/NCBI
|
|
7
|
Bimonte S, Barbieri A, Palma G, Luciano A,
Rea D and Arra C: Curcumin inhibits tumor growth and angiogenesis
in an orthotopic mouse model of human pancreatic cancer. Biomed Res
Int. 2013:8104232013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Youns M and Fathy GM: Upregulation of
extrinsic apoptotic pathway in curcumin-mediated antiproliferative
effect on human pancreatic carcinogenesis. J Cell Biochem.
114:2654–2665. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shakibaei M, Mobasheri A, Lueders C, Busch
F, Shayan P and Goel A: Curcumin enhances the effect of
chemotherapy against colorectal cancer cells by inhibition of NF-κB
and Src protein kinase signaling pathways. PLoS One. 8:e572182013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
James MI, Iwuji C, Irving G, Karmokar A,
Higgins JA, Griffin-Teal N, Thomas A, Greaves P, Cai H, Patel SR,
et al: Curcumin inhibits cancer stem cell phenotypes in ex vivo
models of colorectal liver metastases, and is clinically safe and
tolerable in combination with FOLFOX chemotherapy. Cancer Lett.
364:135–141. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang BL, Shen YM, Zhang QW, Li YL, Luo M,
Liu Z, Li Y, Qian ZY, Gao X and Shi HS: Codelivery of curcumin and
doxorubicin by MPEG-PCL results in improved efficacy of
systemically administered chemotherapy in mice with lung cancer.
Int J Nanomedicine. 8:3521–3531. 2013.PubMed/NCBI
|
|
12
|
Kasi PD, Tamilselvam R, Skalicka-Woźniak
K, Nabavi SF, Daglia M, Bishayee A, Pazoki-Toroudi H and Nabavi SM:
Molecular targets of curcumin for cancer therapy: An updated
review. Tumour Biol. 37:13017–13028. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shehzad A and Lee YS: Molecular mechanisms
of curcumin action: Signal transduction. Biofactors. 39:27–36.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sharma P and Sharma R: miRNA-mRNA
crosstalk in esophageal cancer: From diagnosis to therapy. Crit Rev
Oncol Hematol. 96:449–462. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Orellana EA and Kasinski AL: MicroRNAs in
cancer: A historical perspective on the path from discovery to
therapy. Cancers (Basel). 7:1388–1405. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chitkara D, Mittal A and Mahato RI: miRNAs
in pancreatic cancer: Therapeutic potential, delivery challenges
and strategies. Adv Drug Deliv Rev. 81:34–52. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gayral M, Jo S, Hanoun N, Vignolle-Vidoni
A, Lulka H, Delpu Y, Meulle A, Dufresne M, Humeau M, du Rieu M
Chalret, et al: MicroRNAs as emerging biomarkers and therapeutic
targets for pancreatic cancer. World J Gastroenterol.
20:11199–11209. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Khan S, Ansarullah, Kumar D, Jaggi M and
Chauhan SC: Targeting microRNAs in pancreatic cancer: Microplayers
in the big game. Cancer Res. 73:6541–6547. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tormo E, Pineda B, Serna E, Guijarro A,
Ribas G, Fores J, Chirivella E, Climent J, Lluch A and Eroles P:
MicroRNA profile in response to Doxorubicin treatment in breast
cancer. J Cell Biochem. 116:2061–2073. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hou N, Han J, Li J, Liu Y, Qin Y, Ni L,
Song T and Huang C: MicroRNA profiling in human colon cancer cells
during 5-fluorouracil-induced autophagy. PLoS One. 9:e1147792014.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ma J, Fang B, Zeng F, Pang H, Zhang J, Shi
Y, Wu X, Cheng L, Ma C, Xia J and Wang Z: Curcumin inhibits cell
growth and invasion through up-regulation of miR-7 in pancreatic
cancer cells. Toxicol Lett. 231:82–91. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sun M, Estrov Z, Ji Y, Coombes KR, Harris
DH and Kurzrock R: Curcumin (diferuloylmethane) alters the
expression profiles of microRNAs in human pancreatic cancer cells.
Mol Cancer Ther. 7:464–473. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhou X, Wei M and Wang W: MicroRNA-340
suppresses osteosarcoma tumor growth and metastasis by directly
targeting ROCK1. Biochem Biophys Res Commun. 437:653–658. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Huang D, Qiu S, Ge R, He L, Li M, Li Y and
Peng Y: miR-340 suppresses glioblastoma multiforme. Oncotarget.
6:9257–9270. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li X, Gong X, Chen J, Zhang J, Sun J and
Guo M: miR-340 inhibits glioblastoma cell proliferation by
suppressing CDK6, cyclin-D1 and cyclin-D2. Biochem Biophys Res
Commun. 460:670–677. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Fernandez S, Risolino M, Mandia N, Talotta
F, Soini Y, Incoronato M, Condorelli G, Banfi S and Verde P:
miR-340 inhibits tumor cell proliferation and induces apoptosis by
targeting multiple negative regulators of p27 in non-small cell
lung cancer. Oncogene. 34:3240–3250. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Strong AM Poenitzsch, Setaluri V and
Spiegelman VS: MicroRNA-340 as a modulator of RAS-RAF-MAPK
signaling in melanoma. Arch Biochem Biophys. 563:118–124. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wu ZS, Wu Q, Wang CQ, Wang XN, Huang J,
Zhao JJ, Mao SS, Zhang GH, Xu XC and Zhang N: miR-340 inhibition of
breast cancer cell migration and invasion through targeting of
oncoprotein c-Met. Cancer. 117:2842–2852. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Teiten MH, Dicato M and Diederich M:
Curcumin as a regulator of epigenetic events. Mol Nutr Food Res.
57:1619–1629. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ye M and Zhang J and Zhang J, Miao Q, Yao
L and Zhang J: Curcumin promotes apoptosis by activating the
p53-miR-192-5p/215-XIAP pathway in non-small cell lung cancer.
Cancer Lett. 357:196–205. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hünten S, Kaller M, Drepper F, Oeljeklaus
S, Bonfert T, Erhard F, Dueck A, Eichner N, Friedel CC, Meister G,
et al: p53-regulated networks of protein, mRNA, miRNA, and lncRNA
expression revealed by integrated pulsed stable isotope labeling
with amino acids in cell culture (pSILAC) and next generation
sequencing (NGS) analyses. Mol Cell Proteomics. 14:2609–2629. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Deveraux QL and Reed JC: IAP family
proteins-suppressors of apoptosis. Genes Dev. 13:239–252. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wilkinson JC, Cepero E, Boise LH and
Duckett CS: Upstream regulatory role for XIAP in receptor-mediated
apoptosis. Mol Cell Biol. 24:7003–7014. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Srivastava AK, Singh PK, Singh D, Dalela
D, Rath SK, Goel MM and Bhatt ML: Evaluation of urinary XIAP as a
diagnostic biomarker of carcinoma of urinary bladder. Tumour Biol.
35:8243–8248. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Xu YC, Liu Q, Dai JQ, Yin ZQ, Tang L, Ma
Y, Lin XL and Wang HX: Tissue microarray analysis of X-linked
inhibitor of apoptosis (XIAP) expression in breast cancer patients.
Med Oncol. 31:7642014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Baykara M, Yaman M, Buyukberber S, Tufan
G, Demirci U, Benekli M, Coskun U, Ozet A and Bagriacik E Umit:
Clinical and prognostic importance of XIAP and USP8 in advanced
stages of non-small cell lung cancer. J BUON. 18:921–927.
2013.PubMed/NCBI
|