Introduction
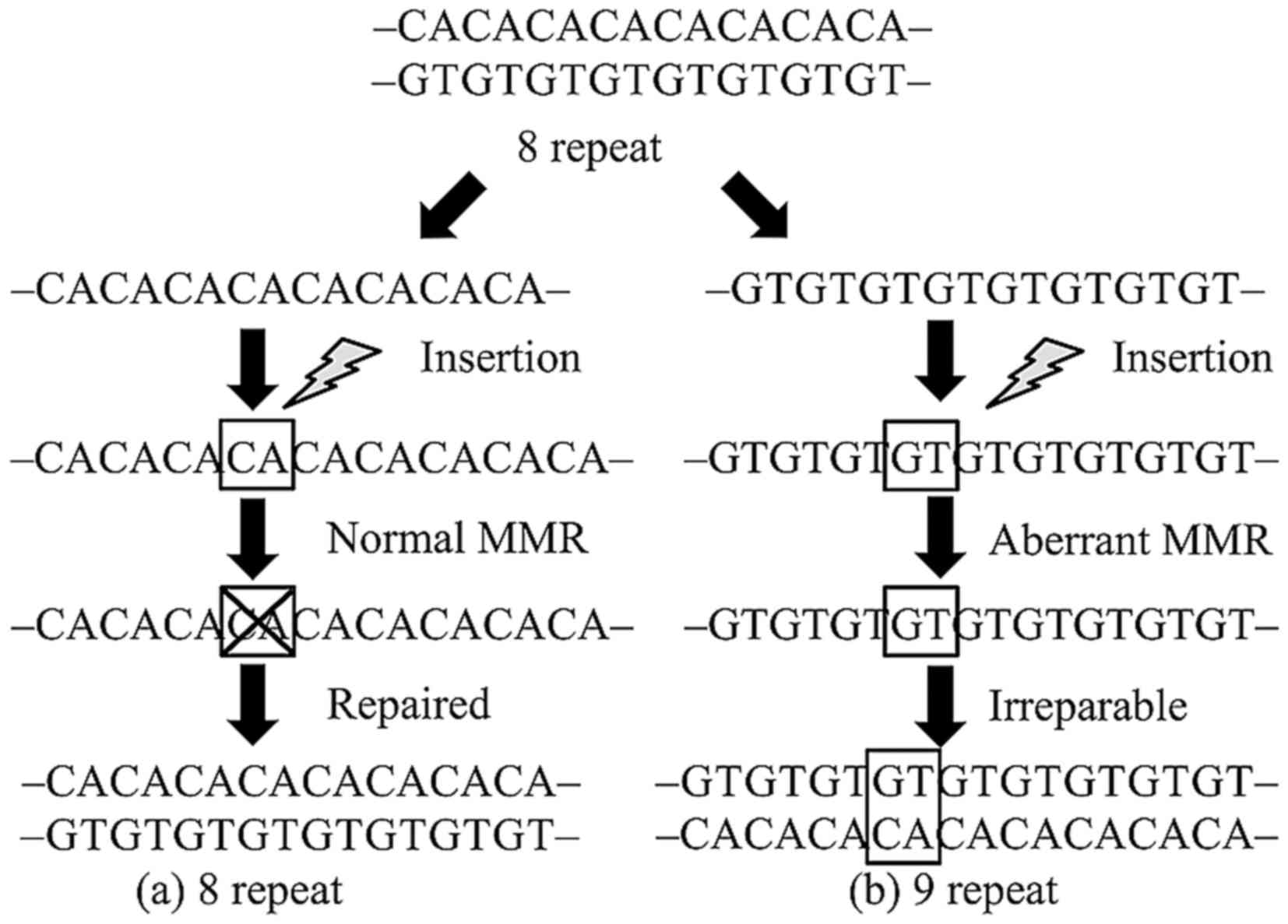
Microsatellites are repeat sequences of one to
several DNA bases. These sequences are used for forensic
identification and paternity testing because they are polymorphic,
occurring widely in both coding and non-coding regions. Repeat
errors during DNA replication are likely to occur in these regions
and are usually repaired by DNA mismatch repair (MMR) genes. In
neoplastic lesions that develop due to aberration of this
mechanism, the microsatellite repeat number in tumor tissues
differs from that in normal tissues (1). This phenomenon is called microsatellite
instability (MSI) and is closely related to carcinogenicity of
hereditary tumors, including Lynch syndrome and others (Fig. 1). MSI analysis is currently performed
as secondary screening for patients suspected for Lynch
syndrome.
MMR function is lost in 20–30% of patients with
endometrial cancer (2,3). Lynch syndrome accounts for approximately
25% of these cases, and the majority involve hypermethylation of
MLH1 promoter or somatic mutations of MMR genes (4). A recent study showed that MSI analysis
is effective as a predictive biomarker for the effect of immune
checkpoint inhibitors, which are new anticancer drugs, including
anti-PD-1 antibody and anti-PD-L1 antibody (5). This suggests that MSI analysis may be
useful as a biomarker for the effect of immunotherapy for
endometrial cancer. In this article, the utility of MSI analysis in
patients with endometrial cancer and new testing procedures are
discussed.
Classification of endometrial cancer by
genetic alterations and MSI
Bokhman classified endometrial cancer into type 1
and 2 (6). Type 1 is characterized by
relatively young onset, well-differentiated tumor with high
expression of estrogen receptor (ER), and good prognosis. Type 2 is
typically elderly-onset, ER-negative poorly differentiated cancer
with a poor prognosis. Histologically, endometrioid adenocarcinoma
has the highest incidence, followed by serous adenocarcinoma and
clear cell adenocarcinoma. Type 1 cases are mostly
well-differentiated endometroid adenocarcinoma, and Type 2 often
involves other histological types (7,8).
PTEN, KRAS, CTNNB1 and PI3KCA mutations
are frequently found in type 1 cases, whereas HER2 and
TP53 mutations occur in type 2 (7–10).
Although there are certain tendencies for mutated genes (11–13), the
Bokhman classification is limited by its difficulty in
classification of endometrial cancer associated with MSI and Lynch
syndrome (2,3).
Using exome sequencing, The Cancer Genome Atlas
Research Network categorized endometrial cancer into 4 types based
on gene mutation pattern and frequency, copy number variation, and
MSI status (13). These four types
are referred to as POLE ultramutated, MSI hypermutated, copy-number
low and copy-number high (Table I),
and the incidences are 7.3, 28.0, 38.8 and 25.9%, respectively. All
tumors categorized in the POLE ultramutated group carry mutations
in the exonuclease domain of POLE, and possessed the highest
incidence of other gene mutations such as PTEN,
PIK3R1 and PIK3CA. The copy-number low and high
groups both have the lowest gene mutation rate and are categorized
into two groups based on the existence of somatic copy number
alterations. Distinct from these other types, the MSI type showed
hypermethylation, which were mostly found in the MLH1
promoter region, and has the second highest incidence of gene
mutation following the POLE ultramutation type. MSI-type
endometrial cancer is histologically characterized by lymphocyte
invasion and immunogenicity (2).
Because MLH1 promoter methylation is a somatic event which
leads to sporadic endometrial cancer (14), the effectiveness of immunotherapy
should be determined not only in Lynch syndrome-related endometrial
cancers, but also in sporadic cases classified in the MSI
hypermethylated group.
 | Table I.Classification and characteristics of
endometrial cancer [modified from (13)]. |
Table I.
Classification and characteristics of
endometrial cancer [modified from (13)].
|
| POLE
(ultramutated) | MSI
(hypermutated) | Copy number
low | Copy number
high |
|---|
| Frequency | 7.3% | 28.0% | 38.8% | 25.9% |
| Copy number
aberrations | Low | Low | Low | High |
| MSI status | Mixed | High | Stable | Stable |
| Mutation rate | Very high
232×106 | High 18×106 | Low
2.9×106 | Low 2.3×106 |
|
| mutations/Mb | mutations/Mb | mutations/Mb | mutations/Mb |
| Genes commonly
mutated | POLE
(100%) | PTEN
(88%) | PTEN
(77%) | TP53
(92%) |
|
| PTEN
(94%) | RPL22
(37%) | CTNNB1
(52%) | PPP2R1A
(22%) |
|
| PIK3CA
(71%) | KRAS
(35%) | PIK3CA
(53%) | PIK3CA
(47%) |
|
| PIK3R1
(65%) | PI3CA
(54%) | PIK3R1
(33%) |
|
|
| FBXW7
(82%) | PIK3R1
(40%) | ARID1A
(42%) |
|
|
| ARID1A
(76%) | ARID1A
(37%) |
|
|
|
| KRAS
(53%) |
|
|
|
|
| ARID5B
(47%) |
|
|
|
| Histological
type | Endometrioid | Endometrioid | Endometrioid | Endometrioid,
Serous, mixed |
| Tumor grade | Mixed (grade
1–3) | Mixed (grade
1–3) | Grade 1 and 2 | Grade 3 |
| Progression-free
survival | Good | Intermediate | Intermediate | Poor |
MSI analysis as a predictive biomarker for
the efficacy of immune checkpoint inhibitors
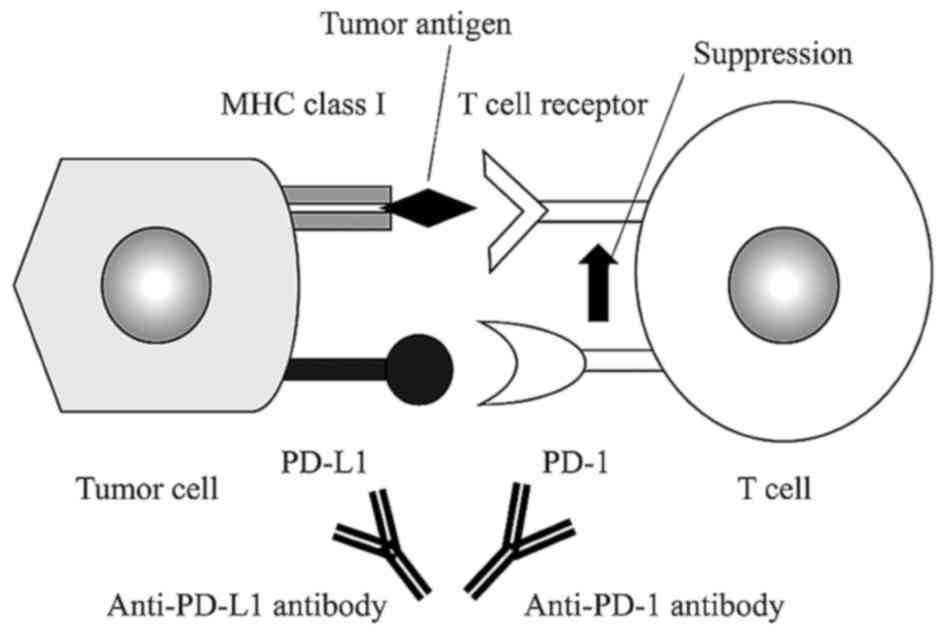
Cancer cells have two mechanisms to avoid the host
immune response; the first involving the cytotoxic
T-lymphocyte-associated protein 4 (CTLA-4) pathway, and the second
linked with programmed cell death-1 (PD-1) and PD ligand (PD-L1)
(15). Activated T cells express
PD-1, and its interaction with PD-L1 decreases T cell activity
(16,17). Physiologically, PD-L1 is expressed in
organs related to immune tolerance, including the tonsils, lungs
and placental syncytiotrophoblasts (18,19).
Expression of PD-L1 on the surface of tumor cells causes the tumor
to avoid host T-cell activity (20).
Therefore, blocking of the PD-1 interaction with PD-L1 in such
cancers is likely to enhance the host immune response and have an
antitumor effect (Fig. 2). This has
been shown in malignant melanoma and non-small-cell cancer, and an
effect on ovarian cancer has been found in gynecological diseases
(20,21).
Le et al conducted a phase 2 study using an
anti-PD-1 antibody, pembrolizumab, given every two weeks at 10
mg/kg in 11 patients with colon cancer associated with MMR
deficiency (group A), 21 patients with colon cancer without MMR
aberration (group B), and 9 non-colorectal cancer patients with MMR
deficiency (group C) (5). The
objective response rate (ORR) and 20-week progression-free survival
(PFS) were 40 and 78% in group A, 0 and 11% in group B, and 71 and
67% in group C. Median PFS and overall survival (OS) could not be
examined in group A, but were 2.2 and 5.0 months, respectively, in
group B. Compared to group B, the patients in group A had
significantly lower hazard ratios of 0.10 (P<0.001) for disease
progression and 0.22 (P=0.05) for death. Exome sequencing showed
that group B (wild-type MMR function) had significantly fewer
somatic mutations than groups A and C (MMR deficient) (73 vs. 1778,
P=0.007).
Although confirmation by phase 3 trials needs to be
awaited, the results above suggest that anti-PD-1 antibody may be a
new therapeutic candidate in cancer patients with aberrant MMR
genes. Howitt et al found that MSI-type endometrial cancer
had 7-fold higher neoepitope levels in comparison with
microsatellite-stable (MSS) cancer (22). In POLE- and MSI-type cancers, the
number of CD3- and CD8-positive cells invading cancer tissues was
significantly higher than that in MSS-type cancer (P=0.001 and
P<0.001, respectively), with no significant differences between
POLE- and MSI-type cancers (P=0.86, P=0.29) (22). Since the incidence of somatic mutation
is high in tumors associated with MSI, it is suggested that
proteins with new immunogenicity are produced in these tumors,
leading to excessive T cell infiltration (23–25).
Expression of PD-L1 in tumors is not necessarily a
precise marker to estimate the therapeutic effect of PD-1/PD-L1
checkpoint blockade (26), and the
creation of a new strategy is imperative. MSI analysis may be a
candidate predictive biomarker for the effect of immunotherapy,
including immune checkpoint inhibitors.
New modalities of MSI analysis and
perspectives
The Bethesda panel is the conventional approach for
MSI analysis, which is optimized for the secondary screening of
Lynch syndrome (27). This method
uses a PCR assay at 5 microsatellites in total, consisting 3
dinucleotide repeats (D2S123, D5S346, D17S250) and 2 mononucleotide
repeats (BAT26, BAT25), and determines differences in repeat number
between tumor and non-tumor regions. Cases with ≥2, 1 and 0
positive markers are classified as MSI-high (MSI-H), MSI-low
(MSI-L), and microsatellite stable (MSS), respectively. In the
Bethesda panel, dinucleotide repeats have been shown to have less
sensitivity and specificity than mononucleotide repeats (28), with particularly low sensitivity in
patients with non-colorectal cancer, or tumors related to
MSH6 mutation (29–33). Consequently, the pentaplex panel was
developed as a procedure with higher sensitivity and specificity,
and has been proposed as a replacement for the Bethesda panel
(28,33–38). This
panel uses 5 mononucleotide repeats (NR-21, NR-22, NR-24, BAT-25,
BAT-26) as markers. A modified pentaplex panel with replacement of
NR-22 with NR-27 is also used (39).
Pagin et al developed a hexaplex panel method using 6
mononucleotide repeats (NR-21, NR-22, NR-27, BAT-25, BAT-26,
BAT-40) as markers and showed that this approach had higher
sensitivity and specificity than the pentaplex panel in patients
with MSH6 mutation and those with non-colorectal cancer
(40).
The type of microsatellite marker that is most
appropriate for MSI analysis remains uncertain. Upon consideration
of the use of MSI analysis as a predictive biomarker for the effect
of anticancer drugs in endometrial cancer, the development of an
optimal method for MSI detection in endometrial cancer, due to both
somatic mutation and Lynch syndrome, is required. Hause et
al developed the MOSAIC method for cross-sectional MSI analysis
in 18 cancer types using the cancer exomes from the Cancer Genome
Atlas (TCGA) database (41). In this
model, a total of 223,082 microsatellites from exome sequencing
were investigated to estimate mean mutation numbers in tumor and
normal tissues obtained from cancer cases in the database. 17,564
microsatellites were identified as loci especially unstable in
MSI-H tumors, which were located frequently in known oncogenes,
suggesting that the other loci may also be located in so far
unknown oncogenes. Characteristic microsatellite regions were
involved among specific types of cancer, which distinguished four
cancer-specific signatures based on MSI patterns. The MOSAIC method
had a high sensitivity and specificity in identifying MSI-H tumors,
with a possibly higher diagnostic accuracy in endometrial cancer
compared to conventional MSI panels. The incidence of MSI-H tumor
was highest in endometrial cancer among 18 types of tumors.
There is an ongoing debate about the methods for MSI
analysis that can include results for unknown MMR genes in
endometrial cancer. Therefore, the method proposed by Hause et
al (41) may be an effective new
approach with wider application compared to current MSI analysis
optimized for Lynch syndrome.
Conclusion
MSI is found in approximately 30% of cases of
endometrial cancer. Immunotherapy is a promising therapeutic
strategy for MSI-type endometrial cancer; however, this therapy is
very expensive and there is a need to select patients who will
benefit from the therapy. The current MSI assay is optimized for
Lynch syndrome, whereas many cases of MSI-type endometrial cancer
are caused by MLH1 promoter methylation or somatic mutation,
and a new method of MSI analysis focused on these cancers is
needed. MSI analysis for advanced endometrial cancer may contribute
to establishment of new therapeutic strategies, including
neoadjuvant therapy, for patients with this cancer.
Acknowledgments
We thank Dr E. Sou (Keio University School of
Medicine) for helpful assistance. The authors gratefully
acknowledge support from the Keio Gijyuku Academic Development
Fund. The funders had no role in data collection and analysis,
decision to publish, or preparation of the manuscript.
References
|
1
|
Thibodeau SN, Bren G and Schaid D:
Microsatellite instability in cancer of the proximal colon.
Science. 260:816–819. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Karamurzin Y and Rutgers JK: DNA mismatch
repair deficiency in endometrial carcinoma. Int J Gynecol Pathol.
28:239–255. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Murali R, Soslow RA and Weigelt B:
Classification of endometrial carcinoma: More than two types.
Lancet Oncol. 15:e268–e278. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Garg K and Soslow RA: Lynch syndrome
(hereditary non-polyposis colorectal cancer) and endometrial
carcinoma. J Clin Pathol. 62:679–684. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Le DT, Uram JN, Wang H, Bartlett BR,
Kemberling H, Eyring AD, Skora AD, Luber BS, Azad NS, Laheru D, et
al: PD-1 blockade in tumors with mismatch-repair deficiency. N Engl
J Med. 372:2509–2520. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bokhman JV: Two pathogenetic types of
endometrial carcinoma. Gynecol Oncol. 15:10–17. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Dedes KJ, Wetterskog D, Ashworth A, Kaye
SB and Reis-Filho JS: Emerging therapeutic targets in endometrial
cancer. Nat Rev Clin Oncol. 8:261–271. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Matias-Guiu X and Prat J: Molecular
pathology of endometrial carcinoma. Histopathology. 62:111–123.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Salvesen HB, Haldorsen IS and Trovik J:
Markers for individualised therapy in endometrial carcinoma. Lancet
Oncol. 13:e353–e361. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Weigelt B and Banerjee S: Molecular
targets and targeted therapeutics in endometrial cancer. Curr Opin
Oncol. 24:554–563. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
McConechy MK, Ding J, Cheang MC, Wiegand
KC, Senz J, Tone AA, Yang W, Prentice LM, Tse K, Zeng T, et al: Use
of mutation profiles to refine the classification of endometrial
carcinomas. J Pathol. 228:20–30. 2012.PubMed/NCBI
|
|
12
|
Urick ME, Rudd ML, Godwin AK, Sgroi D,
Merino M and Bell DW: PIK3R1 (p85α) is somatically mutated at high
frequency in primary endometrial cancer. Cancer Res. 71:4061–4067.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cancer Genome Atlas Research Network, .
Kandoth C, Schultz N, Cherniack AD, Akbani R, Liu Y, Shen H,
Robertson AG, Pashtan I, Shen R, et al: Integrated genomic
characterization of endometrial carcinoma. Nature. 497:67–73. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Le Gallo M and Bell DW: The emerging
genomic landscape of endometrial cancer. Clin Chem. 60:98–110.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mellman I, Coukos G and Dranoff G: Cancer
immunotherapy comes of age. Nature. 480:480–489. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Freeman GJ, Long AJ, Iwai Y, Bourque K,
Chernova T, Nishimura H, Fitz LJ, Malenkovich N, Okazaki T, Byrne
MC, et al: Engagement of the PD-1 immunoinhibitory receptor by a
novel B7 family member leads to negative regulation of lymphocyte
activation. J Exp Med. 192:1027–1034. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Okazaki T and Honjo T: PD-1 and PD-1
ligands: From discovery to clinical application. Int Immunol.
19:813–824. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dong H, Strome SE, Salomao DR, Tamura H,
Hirano F, Flies DB, Roche PC, Lu J, Zhu G, Tamada K, et al:
Tumor-associated B7-H1 promotes T-cell apoptosis: A potential
mechanism of immune evasion. Nat Med. 8:793–800. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Keir ME, Butte MJ, Freeman GJ and Sharpe
AH: PD-1 and its ligands in tolerance and immunity. Annu Rev
Immunol. 26:677–704. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hamanishi J, Mandai M, Matsumura N, Abiko
K, Baba T and Konishi I: PD-1/PD-L1 blockade in cancer treatment:
Perspectives and issues. Int J Clin Oncol. 21:462–473. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hamanishi J, Mandai M, Ikeda T, Minami M,
Kawaguchi A, Murayama T, Kanai M, Mori Y, Matsumoto S, Chikuma S,
et al: Safety and antitumor activity of anti-PD-1 antibody,
nivolumab, in patients with platinum-resistant ovarian cancer. J
Clin Oncol. 33:4015–4022. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Howitt BE, Shukla SA, Sholl LM,
Ritterhouse LL, Watkins JC, Rodig S, Stover E, Strickland KC,
D'Andrea AD, Wu CJ, et al: Association of polymerase e-mutated and
microsatellite-instable endometrial cancers with neoantigen load,
number of tumor-infiltrating lymphocytes, and expression of PD-1
and PD-L1. JAMA Oncol. 1:1319–1323. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dudley JC, Lin MT, Le DT and Eshleman JR:
Microsatellite instability as a biomarker for PD-1 blockade. Clin
Cancer Res. 22:813–820. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mills AM, Liou S, Ford JM, Berek JS, Pai
RK and Longacre TA: Lynch syndrome screening should be considered
for all patients with newly diagnosed endometrial cancer. Am J Surg
Pathol. 38:1501–1509. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mlecnik B, Bindea G, Angell HK, Maby P,
Angelova M, Tougeron D, Church SE, Lafontaine L, Fischer M,
Fredriksen T, et al: Integrative analyses of colorectal cancer show
immunoscore is a stronger predictor of patient survival than
microsatellite instability. Immunity. 44:698–711. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Meng X, Huang Z, Teng F, Xing L and Yu J:
Predictive biomarkers in PD-1/PD-L1 checkpoint blockade
immunotherapy. Cancer Treat Rev. 41:868–876. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
de la Chapelle A and Hampel H: Clinical
relevance of microsatellite instability in colorectal cancer. J
Clin Oncol. 28:3380–3387. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Buhard O, Suraweera N, Lectard A, Duval A
and Hamelin R: Quasimonomorphic mononucleotide repeats for
high-level microsatellite instability analysis. Dis Markers.
20:251–257. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wu Y, Berends MJ, Mensink RG, Kempinga C,
Sijmons RH, van Der Zee AG, Hollema H, Kleibeuker JH, Buys CH and
Hofstra RM: Association of hereditary nonpolyposis colorectal
cancer-related tumors displaying low microsatellite instability
with MSH6 germline mutations. Am J Hum Genet. 65:1291–1298. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hartmann A, Zanardo L, Bocker-Edmonston T,
Blaszyk H, Dietmaier W, Stoehr R, Cheville JC, Junker K, Wieland W,
Knuechel R, et al: Frequent microsatellite instability in sporadic
tumors of the upper urinary tract. Cancer Res. 62:6796–6802.
2002.PubMed/NCBI
|
|
31
|
Kuismanen SA, Moisio AL, Schweizer P,
Truninger K, Salovaara R, Arola J, Butzow R, Jiricny J,
Nyström-Lahti M and Peltomäki P: Endometrial and colorectal tumors
from patients with hereditary nonpolyposis colon cancer display
different patterns of microsatellite instability. Am J Pathol.
160:1953–1958. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hendriks YM, Wagner A, Morreau H, Menko F,
Stormorken A, Quehenberger F, Sandkuijl L, Møller P, Genuardi M,
Van Houwelingen H, et al: Cancer risk in hereditary nonpolyposis
colorectal cancer due to MSH6 mutations: Impact on counseling and
surveillance. Gastroenterology. 127:17–25. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
You JF, Buhard O, Ligtenberg MJ, Kets CM,
Niessen RC, Hofstra RM, Wagner A, Dinjens WN, Colas C, Lascols O,
et al: Tumours with loss of MSH6 expression are MSI-H when screened
with a pentaplex of five mononucleotide repeats. Br J Cancer.
103:1840–1845. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Suraweera N, Duval A, Reperant M, Vaury C,
Furlan D, Leroy K, Seruca R, Iacopetta B and Hamelin R: Evaluation
of tumor microsatellite instability using five quasimonomorphic
mononucleotide repeats and pentaplex PCR. Gastroenterology.
123:1804–1811. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Umar A, Boland CR, Terdiman JP, Syngal S,
de la Chapelle A, Rüschoff J, Fishel R, Lindor NM, Burgart LJ,
Hamelin R, et al: Revised Bethesda Guidelines for hereditary
nonpolyposis colorectal cancer (Lynch syndrome) and microsatellite
instability. J Natl Cancer Inst. 96:261–268. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wong YF, Cheung TH, Lo KW, Yim SF, Chan
LK, Buhard O, Duval A, Chung TK and Hamelin R: Detection of
microsatellite instability in endometrial cancer: Advantages of a
panel of five mononucleotide repeats over the National Cancer
Institute panel of markers. Carcinogenesis. 27:951–955. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Xicola RM, Llor X, Pons E, Castells A,
Alenda C, Piñol V, Andreu M, Castellví-Bel S, Payá A, Jover R, et
al: Performance of different microsatellite marker panels for
detection of mismatch repair-deficient colorectal tumors. J Natl
Cancer Inst. 99:244–252. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Goel A, Nagasaka T, Hamelin R and Boland
CR: An optimized pentaplex PCR for detecting DNA mismatch
repair-deficient colorectal cancers. PLoS One. 5:e93932010.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Buhard O, Cattaneo F, Wong YF, Yim SF,
Friedman E, Flejou JF, Duval A and Hamelin R: Multipopulation
analysis of polymorphisms in five mononucleotide repeats used to
determine the microsatellite instability status of human tumors. J
Clin Oncol. 24:241–251. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Pagin A, Zerimech F, Leclerc J, Wacrenier
A, Lejeune S, Descarpentries C, Escande F, Porchet N and Buisine
MP: Evaluation of a new panel of six mononucleotide repeat markers
for the detection of DNA mismatch repair-deficient tumours. Br J
Cancer. 108:2079–2087. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hause RJ, Pritchard CC, Shendure J and
Salipante SJ: Classification and characterization of microsatellite
instability across 18 cancer types. Nat Med. 22:1342–1350. 2016.
View Article : Google Scholar : PubMed/NCBI
|