Introduction
Ovarian cancer is the most serious cause of
gynecologic cancer morbidity and ranks as the fifth highest cause
of cancer-associated mortality in females (1). It was estimated that ~21,290 novel cases
of ovarian cancer and ~14,180 ovarian cancer-associated mortalities
may occur in the US in 2015 (1,2). The
majority of patients with ovarian cancer were diagnosed with stage
III or IV, and <30% of patients with advanced ovarian cancer
survive >5 years following the initial diagnosis (2). Furthermore, 75–80% of females with
advanced ovarian cancer may experience tumor progression or
recurrence following initial therapy (2). The primary type of therapy available for
ovarian cancer has remained surgical resection accompanied by
chemotherapy with platinum, taxane or other chemotherapeutic
agents; however, traditional therapy is typically ineffective if
tumor metastasis or relapse has occurred (2,3).
Therefore, the underlying molecular mechanism of ovarian cancer and
the development of novel targeted drugs, as well as a more
effective strategy for patients with ovarian cancer, requires
investigation.
The Wnt/β-catenin signaling pathway serves vital
functions during embryonic development and the maintenance of
homeostasis (4–6). The 19 members of the Wnt family are
categorized into two classes, on the basis of β-catenin
involvement: The canonical pathway (β-catenin-dependent) and the
non-canonical pathway (β-catenin-independent). In the canonical
pathway, Wnt ligands bind to the frizzled receptor and activate
Disheveled, inhibiting the phosphorylation of β-catenin by glycogen
synthase kinase3-β and resulting in the accumulation of β-catenin
(4–6).
Subsequently, β-catenin undergoes nuclear translocation from the
cytoplasm and activates certain downstream target genes, including
cyclin D1, transcription factor 4 (TCF4), and lymphoid enhancer
binding factor 1 (LEF1) (2,7).
The 19 members of the Wnt family have been
identified in a number of diseases and tumor types, including
ovarian cancer, and have typically been identified to promote
cancer initiation or progression by regulating cell viability,
migration and apoptosis (8–11). In the majority of tumor types, the
mutation of key components in the Wnt signaling pathway, including
β-catenin, Axin-1 and adenomatous polyposis coli, was common
(8). However, in ovarian cancer
(including clear cell, epithelial and mucinous ovarian cancer),
with the exception of endometrioid ovarian cancer, other underlying
molecular mechanisms may serve dominant functions (8,10). For
example, Wnt7a accelerated tumor viability and progression in
advanced-stage ovarian cancer by activating the Wnt/β-catenin
signaling pathway (12). Wnt10b was
identified to be expressed at significantly increased levels in
endometrial cancer tissues, compared with those in healthy tissues
(13). Wnt10a, homologous with
Wnt10b, was also identified to be associated with tooth agenesis
(14); however, it was demonstrated
that Wnt10a was overexpressed in a number of tumor types, including
gastric, colorectal and esophageal cancer, and promyelocytic
leukemia (1,15–19).
Furthermore, Wnt10a was revealed to promote invasion in esophageal
squamous cell carcinoma and function as an oncogene in renal cell
carcinoma, by activating the Wnt/β-catenin signaling pathway
(16,18). However, information regarding the
function of Wnt10a in ovarian cancer, aside from the upregulated
expression of Wnt10a in endometrial cancer, remains limited.
In the present study, the function of Wnt10a in
ovarian cancer was investigated, and it was identified that Wnt10a
may improve the viability and migration of the SKOV-3 cell line
through the activation of Wnt/β-catenin signaling. In addition,
Wnt10a was overexpressed in ovarian cancer tissues and associated
with high tumor grade and late-stage ovarian cancer. The overall
survival time of patients with increased Wnt10a expression was
decreased compared with that of patients with normal Wnt10a
expression. The results of the present study indicated that Wnt10a
serves critical functions in the carcinogenesis and progression of
epithelial ovarian cancer via the Wnt/β-catenin signaling pathway,
which may present a novel therapeutic strategy for female patients
with ovarian cancer.
Materials and methods
Cell lines and ovarian cancer
tissues
The A2780 and SKOV3 human ovarian cancer cell lines
were purchased from the Cell Bank Type Culture Collection of
Chinese Academy of Sciences (Shanghai, China) and maintained in
Dulbecco's modified Eagle's medium (DMEM; Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal
bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.), 100 U/ml
penicillin and 100 µg/ml streptomycin, at 37°C in humidified
incubator containing 5% CO2.
Frozen resected surgical tissues from a total of 86
patients (age range, 32–67 years; mean age, 52.4 years) with
ovarian cancer between July 2006 and December 2010 were collected
from the Department of Obstetrics and Gynecology of Linyi People's
Hospital (Linyi, China). All tumor tissues were diagnosed by a
pathologist prior to use in the present study according to the
World Health Organization criteria and the Federation International
of Gynecology and Obstetrics (FIGO) guidelines (20–22). A
total of 30 patients with early tumor stage (T1/T2) were selected
and underwent primary cytoreductive surgery, the other 56 patients
with late tumor stage (T3/T4) received postoperative chemotherapy
following bilateral oophorectomy. All patients used for further
study had a complete clinicopathological information including
5-year follow-up (three times a year during the first two years and
twice a year during the later years). The development and
pathogenic progression of each patient with ovarian cancer was
diagnosed and classified by histological examination, according to
the World Health Organization criteria (20). Written informed consent was obtained
from individual subjects and the experimental protocols were
approved by the Ethics Committee of Linyi People's Hospital (Linyi,
China). All experiments complied with the current laws in
China.
Reverse transcription-polymerase chain
reaction (RT-PCR) and RT-quantitative PCR (RT-qPCR)
Total RNA from ovarian cancer tissues or ovarian
cancer cells was extracted using TRIzol® (Invitrogen;
Thermo Fisher Scientific, Inc.), followed by treatment with
RQ1RNase-free DNase (Promega Corporation, Madison, WI, USA). cDNA
was prepared from 2 µg total RNA using SuperScript™ III
(Invitrogen; Thermo Fisher Scientific, Inc.), according to the
manufacturer's protocol. A total of 2 µl synthesized cDNA was used
as template for RT-PCR in a 20 µl reaction mixture, containing 10
pmol of each primer (listed in Table
I), 2 U recombinant Taq DNA polymerase (Invitrogen; Thermo
Fisher Scientific, Inc.), 2.0 µl 1X reaction buffer and 200 pmol
dNTPs. The protocol was as follows, for 35 cycles: Denaturation,
95°C for 30 sec; annealing, 58°C for 30 sec; extension, 72°C for 1
min. Subsequently, the products were incubated at 72°C for 10 min.
The PCR products were run on agarose (2% gel) and visualized by
staining with ethidium bromide. RT-qPCR was conducted using the
standard SYBR−Green PCR kit (Takara Bio, Inc., Otsu,
Japan) to evaluate the relative mRNA levels of Wnt10a and GAPDH (as
the internal control) in the aforementioned ovarian cancer cell
lines and ovarian tumor tissues on the ABI 7500 real-time PCR
system (Applied Biosystems, Foster City, CA, USA). The protocol was
as follows: 94°C for 1 min followed by 35 cycles of 94°C for 1 min
and 53°C for 2 min. Data were analyzed using ABI 7500 software
(version 2.0.6; Applied Biosystems) and presented in terms of
relative quantification (RQ) to GAPDH, on the basis of
2−∆Cq, where ∆Cq = Cq (target) - Cq (reference). Fold
change was calculated using the 2−∆∆Cq method (23). All samples were examined in
triplicate. Primers used for RT-PCR and RT-qPCR are listed in
Table I.
 | Table I.Sequences of primers for RT-PCR and
RT-qPCR, and the siRNA targets. |
Table I.
Sequences of primers for RT-PCR and
RT-qPCR, and the siRNA targets.
| Experiment | Gene | Direction/number | Sequence, 5′-3′ |
|---|
| qPCR primers | Wnt10a | Forward |
TCGCAACAAGATCCCCTATGA |
|
|
| Reverse |
CAGGGCACACGCATTGGACA |
|
| GAPDH | Forward |
GGGAGCCAAAAGGGTCATCA |
|
|
| Reverse |
TGATGGCATGGACTGTGGTC |
| RT-PCR primers | Wnt10a | Forward |
TCGCAACAAGATCCCCTATG |
|
|
| Reverse |
GCAGTGCATCCAGTTGTAAG |
|
| GAPDH | Forward |
AGGAGCGAGATCCCTCCA |
|
|
| Reverse |
CCGTTCAGCTCAGGGATGAC |
| siRNA target |
| siRNA1 |
GGTCAGCACCCAATGACAT |
|
|
| siRNA2 |
GCCAACACAGTGTGCCTAA |
|
|
| siRNA3 |
GCGAGAATGAGGCTTCACA |
Transfection of SKOV3 cells with
Wnt10a short interfering (si)RNA
The Wnt10a-siRNA and the negative control siRNA,
summarized in Table I, were designed
and synthesized by Shanghai Gene Pharma (Shanghai, China). SKOV3
cells were transiently transfected with Wnt10a-siRNA fragments
using Lipofectamine®2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol. SKOV3
cells in the exponential growth phase were seeded (1×104
cells/ml) into 12-well plates. After 24 h, cells were transfected
with 100 pmol Wnt10a-siRNA using Lipofectamine 2000. DMEM medium
was replaced with 1 ml DMEM, containing 10% FBS, after 12 h of
incubation at 37°C and subsequently transfected cells were cultured
at 37°C in an atmosphere containing 5% CO2, prior to
further treatment.
MTT assay
Cell viability was evaluated using the MTT assay.
SKOV3 cells, transfected with Wnt10a siRNA, were seeded into
96-well plates (2×103 cells/well) in 100 µl DMEM (Gibco;
Thermo Fisher Scientific, Inc.). A total of 20 µl MTT solution (5
mg/ml; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was added
into each well daily (on days 2, 3, 4 and 5), and the plates were
incubated for 4 h at 37°C prior to the removal of the supernatant.
Subsequently, 100 µl dimethyl sulfoxide (Sigma-Aldrich; Merck KGaA)
was added to dissolve the crystals. Absorbance was determined at a
wavelength of 490 nm with a microplate reader. Each experiment was
performed in triplicate.
Cell migration assay
Following Wnt10a siRNA transfection, a scratch was
created, using a p200 pipette tip, and the cells were gently washed
twice with PBS to remove the suspended cells. Subsequently, the
monolayer cells were cultured in serum-free DMEM for 24 h at 37°C.
At the designated time point 2 and 24 h after a scratch was
created, the width of each scratch at five randomly selected fields
along the scratch were observed under a light microscope (×20
magnification) and recorded. The migration rate was calculated
using the following formula: Migration rate (%) = (S2 h
- S24 h)/S2 h × 100. S2 h
represents the distance of the scratch at 2 h and S24 h
represents the distance at 24 h.
Western blot analysis
Cells were cultured in 6-well plates at
2×106/well and transfected with Wnt10a-siRNA fragments
or treated with 0.4 nM LGK-974, a specific inhibitor for porcupine
homolog (Drosophila; PORCN), in Wnt/β-catenin signaling pathway.
After 48 h of incubation at 37°C, total protein was extracted using
iced lysis buffer (1% Triton X-100, 50 mM Tris-HCl, pH 7.4; 150 mM
NaCl; 0.1% SDS; 1 mM phenylmethanesulfonyl fluoride; 1 mM EDTA).
Subsequently, the total protein concentration was determined using
a bicinchoninic acid assay (BCA Protein Assay kit; Generay Biotech
Co., Ltd., Shanghai, China). A total of 10 µg protein was separated
on an 10% SDS-PAGE gel, transferred to nitrocellulose membranes
followed by blocking with 5% non-fat milk for 2 h at room
temperature, and then incubated overnight with primary mouse
monoclonal antibodies against β-catenin (1:250, sc65480; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA), cyclin D1 (1:400, sc-70899;
Santa Cruz Biotechnology, Inc.), LEF1 (1:800, ab137872), TCF4
(1:500, ab217668) and GAPDH (1:2,000, ab181602) (Abcam, Cambridge,
MA, USA) at 4°C. Membranes were subsequently washed and incubated
with a horseradish peroxidase-conjugated secondary antibody
(1:10,000, ab97051) for 1 h at 25°C. Immunoreactivity was
determined using an enhanced chemiluminescence kit (Merck
KGaA).
Statistical analysis
All data are expressed as the mean ± standard
deviation of three independent experiments and were analyzed using
a two-tailed paired Student's t-test between two groups with SPSS
software (version 16.0; SPSS, Inc., Chicago, IL, USA). Associations
between Wnt10a expression and clinicopathological characteristics
were analyzed using the χ2 test. Survival curves were
plotted using the Kaplan-Meier estimator method and compared using
the log-rank test. P<0.05 was considered to indicate a
statistically significant difference.
Results
Knockdown of Wnt10a in the SKOV3 human
ovarian cancer cell line suppresses cell viability
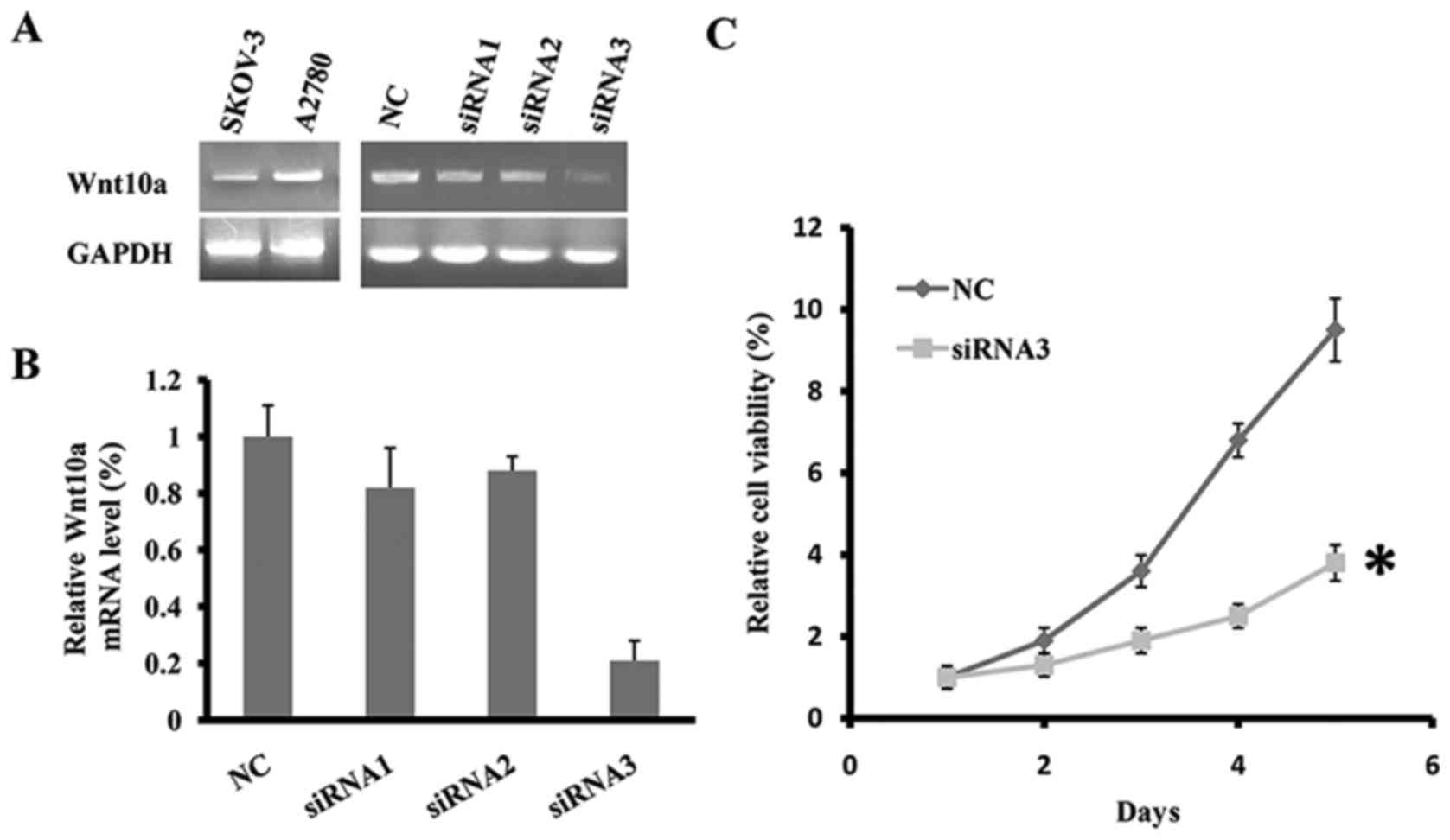
Using the semi-quantitative RT-PCR, the expression
of Wnt10a was determined in two human ovarian cancer cell lines,
SKOV3 and A2780. As presented in Fig.
1A, Wnt10a was expressed at an increased level in the two cell
lines. To determine the function of Wnt10a in cell viability, three
siRNA fragments targeting the coding sequence of Wnt10a were
designed and synthesized. Subsequently, the three Wnt10a-siRNA
fragments or negative control (NC) were transfected into cell line
SKOV3. Following the transfection of Wnt10a-siRNA3 fragment for 48
h, the mRNA level of Wnt10a was significantly decreased, indicating
efficient knockdown of Wnt10a in SKOV3 (Fig. 1A and B). Cell viability of
Wnt10a-siRNA3-SKOV3 was determined using the MTT assay for five
days. The results of the present study identified that the
downregulation of Wnt10a inhibited the viability of SKOV3 cells,
and on day 5 the viability rate of Wnt10a-siRNA3-SKOV3 cells was
~40% that of the control (Fig. 1C).
Therefore, knockdown of Wnt10a inhibited the viability of SKOV3
cells.
Knockdown of Wnt10a suppresses the
migratory ability of SKOV3 cells
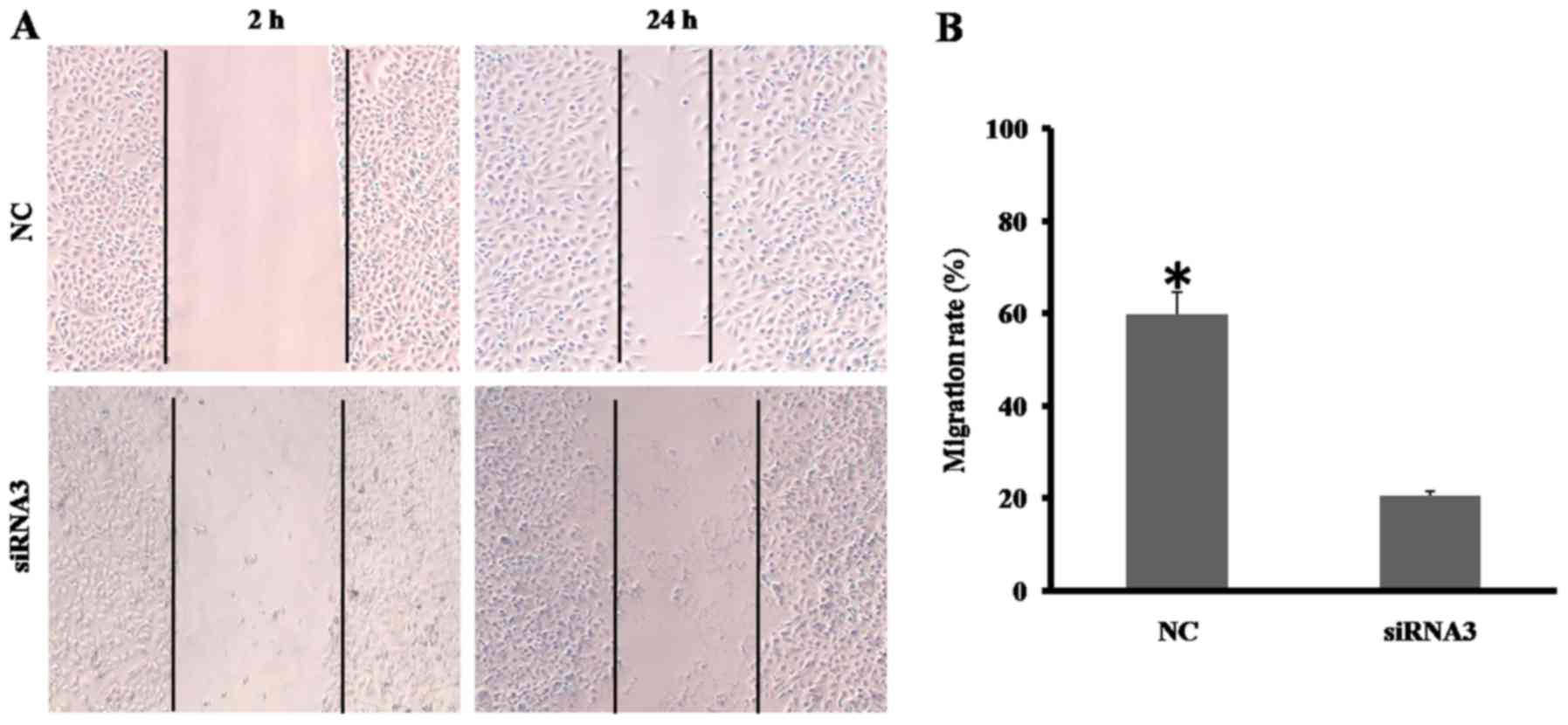
Either migration or invasion is typical for
malignant tumor cells, and Wnt10a has been identified to promote
the migration of A498 and A786-Orenal cancer cells. The effect of
Wnt10a on the motile capability of SKOV3 cells was investigated in
the present study using a wound-healing assay. As presented in
Fig. 2A, the migratory ability of
Wnt10a-siRNA3-SKOV3 cells was significantly decreased, compared
with that of the NC group. The migrated distance of
Wnt10a-siRNA3-SKOV3 cell was ~30% of that of the NC group (Fig. 2B). The results of the present study
demonstrated that decreased Wnt10a expression disrupts the
migratory ability of SKOV3 cells.
Wnt10a activates the Wnt/β-catenin
signaling pathway
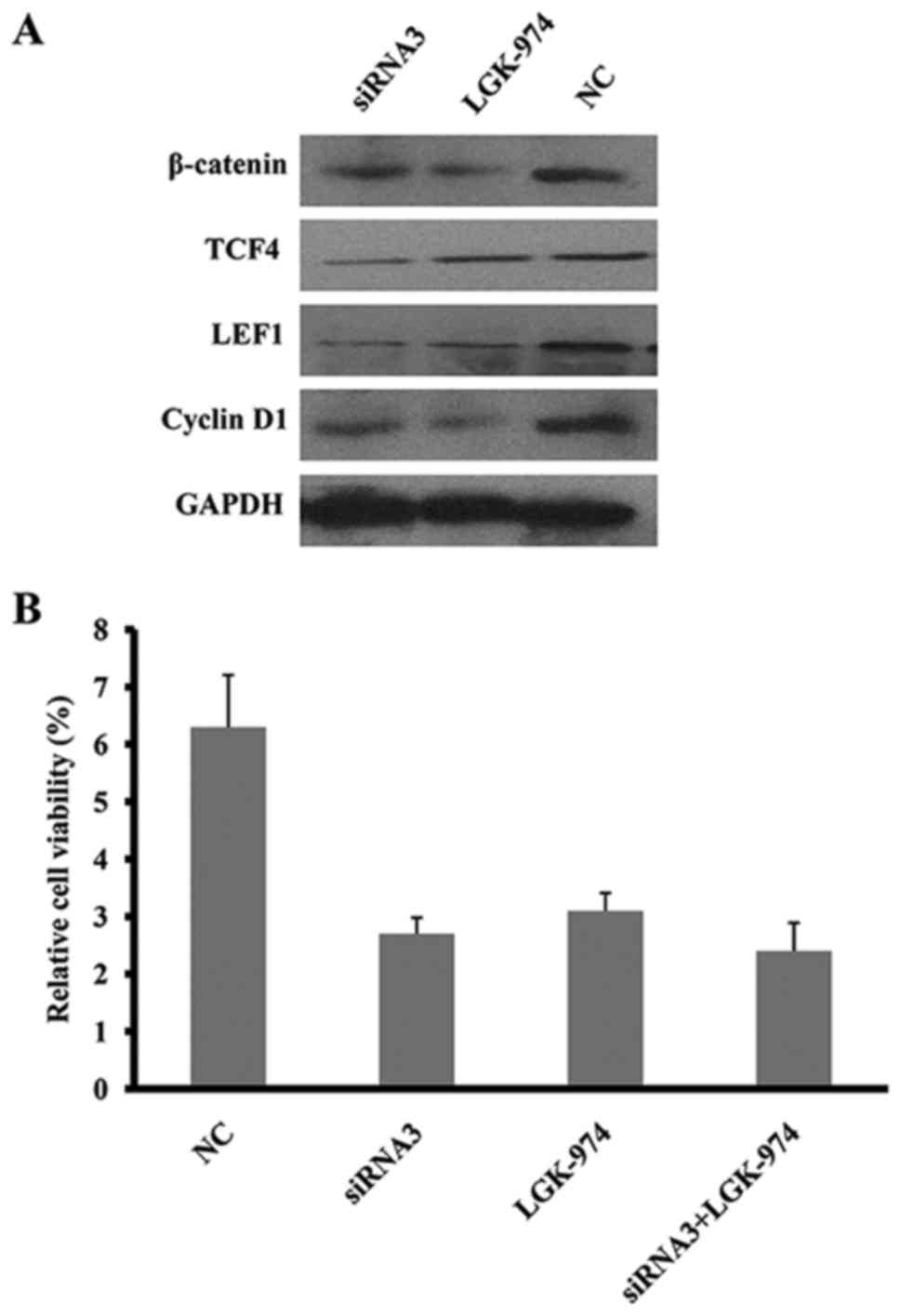
A previous study reported that Wnt10a activates the
Wnt/β-catenin signaling pathway in renal carcinoma cells (18). Therefore, we hypothesized that the
knockdown of Wnt10a in SKOV3 cells may disrupt the Wnt/β-catenin
signaling pathway. As predicted, the expression of molecules in the
Wnt/β-catenin signaling pathway, including β-catenin, TCF4, LEF1
and cyclin D1, was significantly downregulated in
Wnt10a-siRNA3-SKOV3 cells (Fig. 3A),
which was consistent with the effect of LGK-974, a specific
inhibitor for porcupine homolog (Drosophila; PORCN), in
Wnt/β-catenin signaling pathway. In addition, either Wnt10a-siRNA3
or LGK-974 may inhibit the viability of SKOV3; however, there was
no synergic effect exhibited between siRNA3 and LGK-974 (Fig. 3B), suggesting that Wnt/β-catenin
signaling is inhibited by Wnt10a siRNA. Therefore, Wnt10a was
identified to promote the viability and migration of SKOV3 cells
through activation of the Wnt/β-catenin signaling pathway.
Wnt10a overexpression is associated
with carcinogenesis of ovarian cancer and poor survival
The results of the present study identified that
Wnt10a may serve an oncogenic role during ovarian cancer
carcinogenesis; however, the clinical significance of Wnt10a in
ovarian cancer progression remains unknown. To evaluate the
clinical significance of Wnt10a, RT-qPCR was employed to determine
the level of Wnt10a in tumoral and paratumoral tissues from 86
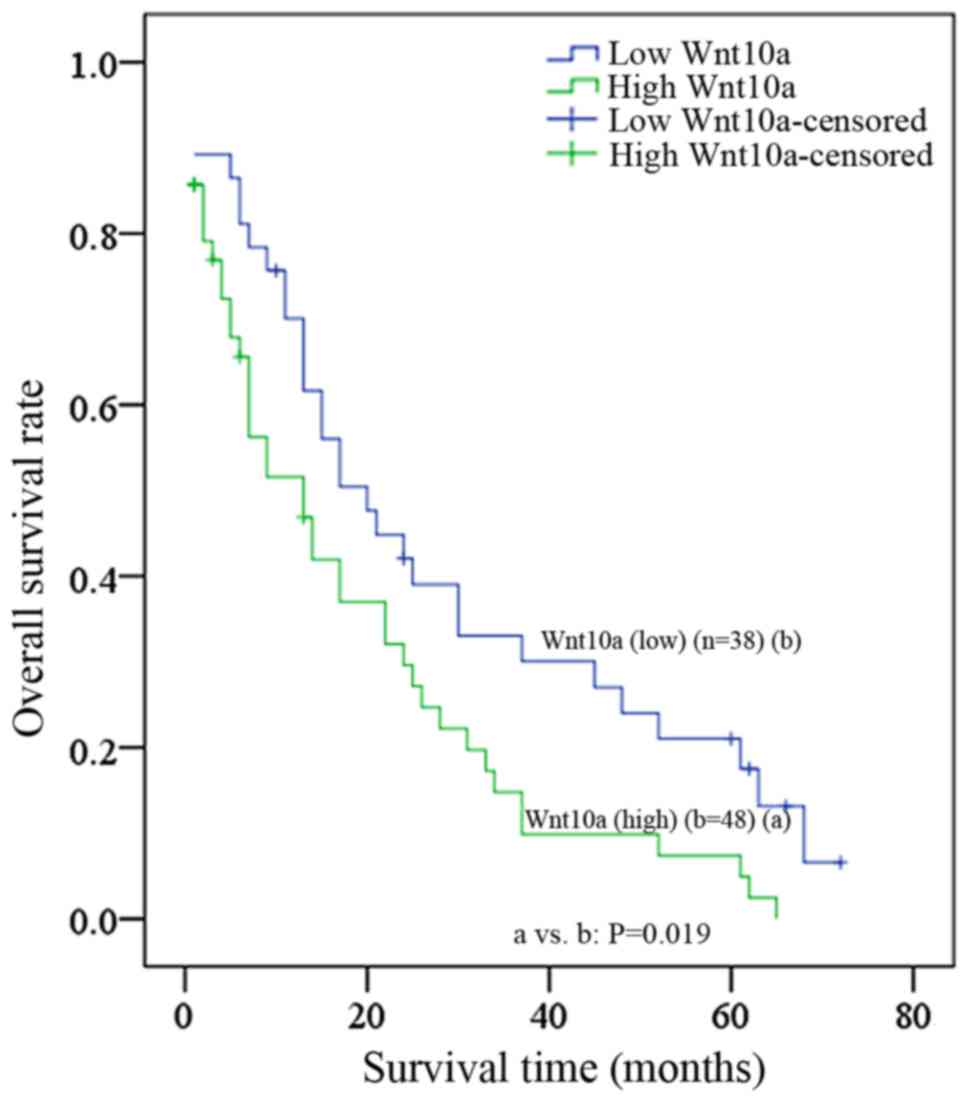
patients with ovarian cancer. As presented in Fig. 4, overexpression of Wnt10a was
significantly associated with high-grade (grade III, P=0.031) and
late-stage (T3, P=0.008); however, overexpression of Wnt10a was not
associated with other clinical characteristics (Table II), including age and histology.
Furthermore, the estimated 5-year survival rate was 18.4% for
patients with decreased Wnt10a expression (n=38), whereas it was
6.3% for patients with increased Wnt10a expression (n=48), and a
significant difference was identified between these two groups
(P=0.019). The median survival time was 20±4.45 months for patients
with low Wnt10a levels, which was elevated when compared with that
for patients with high Wnt10a levels (13±3.67 months).
 | Table II.Association between the expression of
Wnt10a and clinicopathological characteristics in ovarian
cancer. |
Table II.
Association between the expression of
Wnt10a and clinicopathological characteristics in ovarian
cancer.
|
|
| Wnt10a expression
level |
|
|---|
|
|
|
|
|
|---|
| Characteristic | n | ≤4-fold | >4-fold | P-value |
|---|
| Total | 86 | 37 | 49 |
|
| Age, years |
|
|
| 0.829 |
| ≤50 | 48 | 20 | 28 |
|
|
>50 | 38 | 17 | 21 |
|
| TNM stage |
|
|
| 0.008a |
| Early
(T1/T2) | 30 | 22 | 8 |
|
| Late
(T3/T4) | 56 | 15 | 41 |
|
| Grade |
|
|
| 0.031a |
|
Low | 33 | 19 | 14 |
|
|
High | 53 | 18 | 35 |
|
| Histology |
|
|
| 0.733 |
| Clear
cell | 39 | 16 | 23 |
|
|
Other | 47 | 21 | 26 |
|
Discussion
The morbidity and mortality caused by ovarian cancer
is a threat to the health of females. Although efforts have been
made to increase the quantity of novel molecule-targeted drugs,
traditional surgical removal followed by chemotherapy remains the
primary treatment method, despite patients often exhibiting
recurrence following the initial therapy (2). Wnt/β-catenin signaling is known to serve
important functions in fetal development and several types of
cancer (4), and >1 Wnt gene has
been associated with the carcinogenesis of ovarian cancer (8). Wnt10a, the homolog of Wnt10b, was
identified to be overexpressed in various types of tumor cells, and
to function as an oncogene in renal cell carcinoma (18). However, to the best of our knowledge,
the present study was the first to explore the function of Wnt10a
in the carcinogenesis and progression of ovarian cancer.
According to the results of the present study,
Wnt10a was highly expressed in the SKOV3 and A2780 ovarian cancer
cell lines. In the loss of function assay, Wnt10a was knocked-down
by a siRNA designed to target Wnt10a in SKOV3 cells. Subsequently,
the viability rate and the migratory ability of SKOV3 cells were
identified to be markedly disrupted by Wnt10a siRNA. The results of
the present study were consistent with the early report in renal
cell carcinoma and suggested that Wnt10a may serve an oncogenic
role in the carcinogenesis of ovarian cancer (18). Furthermore, in renal cell carcinoma,
Wnt10a was identified to increase intracellular β-catenin
accumulation through nuclear translocation, and to activate
downstream molecules such as TCF4 and LEF1 (18). In the present study, the expression
levels of β-catenin, cyclin D1, LEF1 and TCF4 were significantly
decreased in Wnt10a-siRNA-SKOV3 cells, which was validated by
LGK-974, the specific inhibitor targeting PORCN in Wnt/β-catenin
signaling (24). However, the effect
of Wnt10a was independent of LGK-974 as no synergic effects between
Wnt10a siRNA and LGK-974 were revealed. Therefore, the canonical
Wnt/β-catenin signaling pathway was determined to be activated by
Wnt10a in ovarian cancer.
Tumorigenesis is a complex multistep process;
uncontrolled cell viability and migration are two of the ten
hallmarks of cancer (25). Typically,
cancer cells with potent migratory ability exhibit an increased
likelihood of invading adjacent healthy tissues, forming novel
lesions or leading to metastasis (26). The clinical significance of Wnt10a has
been revealed in renal cell carcinoma, where it was evaluated as an
independent risk factor and identified to increase the incidence
risk of renal carcinogenesis by 2–4-fold (18). Wnt10a is an independent risk factor
for an increased mortality rate in renal cell carcinoma (18). Additionally, the Wnt10a rs7349332
polymorphism has been associated with an increased risk of
developing colorectal cancer via increasing fat intake (27). In the present study, a retrospective
analysis of 86 patients using RT-qPCR revealed that Wnt10a was
expressed at increased levels in the tumoral tissues, as compared
with in the paratumoral tissues. In addition, the overexpression of
Wnt10a was identified to be significantly associated with certain
clinical characteristics of ovarian cancer, including clinical
tumor stage (grade III) and tumor-node-metastasis classification
(T3) (20). Based on Wnt10a
expression, a Kaplan-Meier estimator survival curve was
constructed, which demonstrated that patients with decreased Wnt10a
expression levels exhibited improved overall survival compared with
patients with increased levels of Wnt10a, suggesting that Wnt10a
may be a predictor for the survival of patients with ovarian
cancer. These preliminary results indicated that decreased Wnt10a
expression may be associated with improved prognosis in patients
with ovarian cancer. However, additional studies involving a larger
cohort of patients with ovarian cancer are required to validate
these results.
Wnt10a-Wnt6 are clustered on the human chromosome
2q35 region in a head-to-tail manner and were identified to be
co-expressed in SW480 cells (17).
However, no significant difference was identified between Wnt6 mRNA
expression and Wnt10a in SKOV3 ovarian cells. Wnt10a was identified
to function in an autocrine manner in renal cell carcinoma
(18), and so we hypothesize that
Wnt10a may also activate the Wnt/β-catenin signaling pathway in an
autocrine manner in ovarian cancer cells.
The results of the present study indicated that the
overexpression of Wnt10a serves oncogenic functions in the
promotion of carcinogenesis and progression of human ovarian cancer
by activating the Wnt/β-catenin/TCF/LEF1 signaling pathway. In
addition, the overexpression of Wnt10a was clinically associated
with high tumor grade and with late stage in patients with ovarian
cancer. Wnt10a may be a predictor for the survival of patients with
ovarian cancer, and serve as a novel target for drug development
and as a prognostic marker.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
Statistics, 2015. CA Cancer J Clin. 65:5–29. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bixel K and Hays JL: Olaparib in the
management of ovarian cancer. Pharmacogenomics Pers Med. 8:127–135.
2015. View Article : Google Scholar
|
|
3
|
Luvero D, Milani A and Ledermann JA:
Treatment options in recurrent ovarian cancer: Latest evidence and
clinical potential. Ther Adv Med Oncol. 6:229–239. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Narita T, Sasaoka S, Udagawa K, Ohyama T,
Wada N, Nishimatsu S, Takada S and Nohno T: Wnt10a is involved in
AER formation during chick limb development. Dev Dyn. 233:282–287.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sherwood V: WNT signaling: An emerging
mediator of cancer cell metabolism? Mol Cell Biol. 35:2–10. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lu D, Zhao Y, Tawatao R, Cottam HB, Sen M,
Leoni LM, Kipps TJ, Corr M and Carson DA: Activation of the Wnt
signaling pathway in chronic lymphocytic leukemia. Proc Natl Acad
Sci USA. 101:pp. 3118–3123. 2004; View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hilliard TS, Gaisina IN, Muehlbauer AG,
Gaisin AM, Gallier F and Burdette JE: Glycogen synthase kinase 3β
inhibitors induce apoptosis in ovarian cancer cells and inhibit in
vivo tumor growth. Anticancer Drugs. 22:978–985. 2011.PubMed/NCBI
|
|
8
|
Arend RC, Londoño-Joshi AI, Straughn JM Jr
and Buchsbaum DJ: The Wnt/β-catenin pathway in ovarian cancer: A
review. Gynecol Oncol. 131:772–779. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gatcliffe TA, Monk BJ, Planutis K and
Holcombe RF: Wnt signaling in ovarian tumorigenesis. Int J Gynecol
Cancer. 18:954–962. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wu R, Zhai Y, Fearon ER and Cho KR:
Diverse mechanisms of beta-catenin deregulation in ovarian
endometrioid adenocarcinomas. Cancer Res. 61:8247–8255.
2001.PubMed/NCBI
|
|
11
|
Burkhalter RJ, Symowicz J, Hudson LG,
Gottardi CJ and Stack MS: Integrin regulation of beta-catenin
signaling in ovarian carcinoma. J Biol Chem. 286:23467–23475. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yoshioka S, King ML, Ran S, Okuda H,
MacLean JA II, McAsey ME, Sugino N, Brard L, Watabe K and Hayashi
K: WNT7A regulates tumor growth and progression in ovarian cancer
through the WNT/β-catenin pathway. Mol Cancer Res. 10:469–482.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen H, Wang Y and Xue F: Expression and
the clinical significance of Wnt10a and Wnt10b in endometrial
cancer are associated with the Wnt/β-catenin pathway. Oncol Rep.
29:507–514. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bonds J, Pollan-White S, Xiang L, Mues G
and D'Souza R: Is there a link between ovarian cancer and tooth
agenesis? Eur J Med Genet. 57:235–239. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kirikoshi H, Inoue S, Sekihara H and Katoh
M: Expression of WNT10A in human cancer. Int J Oncol. 19:997–1001.
2001.PubMed/NCBI
|
|
16
|
Long A, Giroux V, Whelan KA, Hamilton KE,
Tétreault MP, Tanaka K, Lee JS, Klein-Szanto AJ, Nakagawa H and
Rustgi AK: WNT10A promotes an invasive and self-renewing phenotype
in esophageal squamous cell carcinoma. Carcinogenesis. 36:598–606.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kirikoshi H, Sekihara H and Katoh M:
WNT10A and WNT6, clustered in human chromosome 2q35 region with
head-to-tail manner, are strongly coexpressed in SW480 cells.
Biochem Biophys Res Commun. 283:798–805. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hsu RJ, Ho JY, Cha TL, Yu DS, Wu CL, Huang
WP, Chu P, Chen YH, Chen JT and Yu CP: WNT10A plays an oncogenic
role in renal cell carcinoma by activating WNT/β-catenin pathway.
PLoS One. 7:e476492012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kirikoshi H, Sekihara H and Katoh M:
Up-regulation of WNT10A by tumor necrosis factor α and Helicobacter
pylori in gastric cancer. Int J Oncol. 19:533–536. 2001.PubMed/NCBI
|
|
20
|
Scully RE: World Health Organization
classification and nomenclature of ovarian cancer. Natl Cancer Inst
Monogr. 42:5–7. 1975.PubMed/NCBI
|
|
21
|
Shepherd JH: Revised FIGO staging for
gynaecological cancer. Br J Obstet Gynaecol. 96:889–892. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Denny L, Quinn M and Hacker N: FIGO cancer
report 2012. Int J Gynaecol Obstet. 119 Suppl 2:S892012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu J, Pan S, Hsieh MH, Ng N, Sun F, Wang
T, Kasibhatla S, Schuller AG, Li AG, Cheng D, et al: Targeting
Wnt-driven cancer through the inhibition of Porcupine by LGK974.
Proc Natl Acad Sci USA. 110:pp. 20224–20229. 2013; View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hanahan D and Weinberg RA: Hallmarks of
Cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Entschladen F, TL IV Drell, Lang K, Joseph
J and Zaenker KS: Tumour-cell migration, invasion, and metastasis:
Navigation by neurotransmitters. Lancet Oncol. 5:254–258. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Galbraith RL, Poole EM, Duggan D, Muehling
J, Hsu L, Makar K, Xiao L, Porter JD and Ulrich CM: Polymorphisms
in WNT6 and WNT10A and colorectal adenoma risk. Nutr Cancer.
63:558–564. 2011. View Article : Google Scholar : PubMed/NCBI
|