Introduction
At present, esophageal cancer is the sixth most
common cause of cancer-associated mortality worldwide (1). Based on the recent data collected from
the National Central Cancer Registry in 2016, esophageal squamous
cell carcinoma (ESCC) represents ~88% of esophageal cancer cases in
China (2). Despite a myriad of
improvement in therapeutic techniques, including chemotherapeutic,
radiotherapeutic and surgical treatment during the previous 30
years, the prognosis of ESCC remains poor with a 5-year survival
rate of 10–15% (3). Therefore,
improved treatment strategies are urgently required. In recent
years, molecular targeted therapy against key somatic alterations
has become an important endeavor towards pathway-driven treatment.
ESCC is frequently associated with the overexpression of the
epidermal growth factor receptor (EGFR) gene (4). The dysregulation of EGFR has been
implicated in the development of resistance to the conventional
chemotherapy and poor clinical outcome (5). Therefore, inhibition of EGFR may be a
promising approach for the management of ESCC. Previously, numerous
novel compounds, including small molecule tyrosine kinase
inhibitors, gefitinib and erlotinib, and monoclonal antibodies
(cetuximab) targeting various proteins in the EGFR signaling
cascade have been developed, and their therapeutic effects have
been extensively evaluated in vitro and in clinical studies
(6–10). Although EGFR inhibitors have higher
efficacies and lower toxicities compared with conventional
chemotherapeutic agents, patients have demonstrated highly variable
responses to these inhibitors (11).
Therefore, there is an urgent requirement to develop clinically
useful agents with optimal therapeutic efficacies.
MicroRNAs (miRNAs) are a type of small non-coding
RNAs that bind to the 3′ untranslated region of target mRNAs.
miRNAs control gene expression by degrading the target mRNAs or
inhibiting their translation into functional proteins (12). Emerging evidence has indicated that
miRNAs may serve diverse roles in the regulation of cancer
initiation and progression (13,14).
miRNAs function as oncogenes or tumor suppressor genes depending on
their specific target mRNAs (15–19).
miR-1, which was first identified as a muscle-specific miRNA and is
known to be abundantly expressed in cardiac and skeletal muscles,
was previously identified as a tumor suppressor in various types of
human cancer, including rhabdomyosarcoma, lung, thyroid, prostatic,
bladder, colorectal and hepatocellular carcinomas (20–27).
Additionally, a lower expression level of miR-1 in lung cancer is
an indicator of poor prognosis (28).
Previous studies have revealed that miR-1 is downregulated in human
ESCC tissues and cell lines (29,30).
However, the functional significance of miR-1 in ESCC has not yet
been clarified. Insights into the association between miR-1 and its
target oncogenes may further the understanding of the molecular
mechanism underlying ESCC oncogenes and subsequently enable the
development of improved therapies.
The phosphatiditylinositide-3-kinase (PI3K)
signaling pathway is central to growth and survival of numerous
types of cancer, and PI3K signaling can be directly activated by
genetic alterations. Phosphatidylinositol-4,5-bisphosphate 3-kinase
catalytic subunit α (PIK3CA), the p110α subunit of PI3K, functions
as an oncogene and serves an important role in numerous types of
cancer, including ESCC (31,32). A previous study found that higher
expression of PIK3CA is associated with a poor prognosis in
non-small cell lung cancer (28).
Previous studies also revealed that PIK3CA was amplified in ESCC
(33) and that the expression level
of PIK3CA mRNA and protein was associated with lymph node
metastasis (32,34). A previous study by the present authors
has demonstrated that miR-1 inhibited tumorigenic properties of
lung cancer cells by targeting PIK3CA (35). The present study aimed to investigate
the levels of miR-1 and PIK3CA expression in resected esophageal
tumor tissue samples and determined the association between their
expression levels and clinicopathological features of patients with
ESCC.
Materials and methods
Clinical samples
A total of 74 patients (61 men and 13 women) with
ESCC were included in this study, ranging in age between 38 and 76
years (mean, 62 years). All patients were clinically staged
according to the seventh edition of the American Joint Committee on
Cancer (AJCC) system for esophageal cancer (36). Human ESCC tissues and matched adjacent
normal tissues (2 cm between tumor and normal tissue) were
collected directly following surgical resection at the First
Affiliated Hospital of Nanjing Medical University (Nanjing, China)
between January 2011 and December 2012. None of the patients had
received chemotherapy or radiotherapy prior to surgery. All samples
were immediately frozen in liquid nitrogen and stored at −80°C.
ESCC diagnosis was confirmed following histological evaluation,
which was performed by a pathologist who was blinded to the aim of
the study. The present study was approved by the Ethical Review
Committee in the First Affiliated Hospital of Nanjing Medical
University. Written informed consent was obtained from all patients
prior to enrollment in the present study.
Cell culture
The TE-1 human ESCC cell line was obtained from the
Cell Bank of Type Culture Collection of the Chinese Academy of
Sciences (Shanghai, China). The cells were incubated in RPMI-1640
medium (HyClone, Logan, UT, USA) supplemented with 10% fetal bovine
serum (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA),
100 U/ml penicillin and 100 µg/ml streptomycin at 37°C in a
humidified incubator containing 5% CO2. The cells in the
exponential phase were used for all subsequent assays.
Drug
Gefitinib was provided by AstraZeneca
Pharmaceuticals (Macclesfield, UK). Gefitinib was dissolved in DMSO
to obtain a stock concentration of 10 mM and stored at −20°C. The
10 mM stocks were diluted in fresh medium prior to each experiment.
The control cells were treated with the medium supplemented with an
equal concentration of DMSO (<0.1%).
Transfection
The cells were transiently transfected with 50 nM
miR-1 mimics or the 50 nM negative control (Genepharma, Inc.,
Sunnyvale, CA, USA) using Lipofectamine™ 2000 (Invitrogen; Thermo
Fisher Scientific, Inc.) diluted in Opti-MEM (Invitrogen; Thermo
Fisher Scientific, Inc.), according to the manufacturer's protocol.
The negative control was a scrambled oligonucleotide not encoding
any known miRNA. The sequences of miR-1 mimics and the negative
control are presented in Table I.
Transfection efficiency was confirmed by analyzing miR-1 expression
level using the TaqMan real-time polymerase chain reaction (PCR)
system (Applied Biosystems; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA). Quantitative real-time PCR was run on ABI PRISM
7900HT (Applied Biosystems; Thermo Fisher Scientific, Inc.).
Subsequent experimentation was performed 24–48 h after
transfection.
 | Table I.Sequences of miR-1 mimics and the
negative control. |
Table I.
Sequences of miR-1 mimics and the
negative control.
| Name of the
primers | Sequences
(5′-3′) |
|---|
| miR-1 mimics |
|
|
Forward |
UGGAAUGUAAAGAAGUAUGUAU |
|
Reverse |
ACAUACUUCUUUACAUUCCAUU |
| Negative
control |
|
|
Forward |
UUCUCCGAACGUGUCACGUTT |
|
Reverse |
ACGUGACACGUUCGGAGAATT |
Quantitative RT-PCR
Quantitative real time polymerase chain reaction
(qRT-PCR) was performed to determine the expression of miR-1 and
all related genes. Total RNA was extracted from tissues and cells
using TRIzol® (Invitrogen; Thermo Fisher Scientific,
Inc.), according to the manufacturer's protocol. Total RNA (500 ng)
was quantitated at 260 nm and reverse-transcribed into cDNA using
the PrimeScript RT reagent kit (Takara Biotechnology, Co., Ltd.,
Dalian, China) at 37°C for 15 min and 85°C for 30 sec. qPCR was
performed using the SYBR Premix Ex Taq™ kit (Takara Biotechnology,
Co., Ltd.) in the ABI PRISM 7900HT (Applied Biosystems; Thermo
Fisher Scientific, Inc.) system. The thermocycling conditions were:
50°C for 2 min, 95°C for 10 min followed by 40 cycles with each
cycle consisting of 30s at 95°C, and 1 min at 60°C. Cycle threshold
(Ct) values were determined using the SDS version 2.4 software
(Applied Biosystems; Thermo Fisher Scientific, Inc.). PIK3CA
expression levels were normalized to β-actin expression using the
2−ΔΔCt method (37). All
primers (Invitrogen; Thermo Fisher Scientific, Inc.) are presented
in Table II. These experiments were
performed in triplicate.
 | Table II.Sequences of primers for reverse
transcription-quantitative polymerase chain reaction. |
Table II.
Sequences of primers for reverse
transcription-quantitative polymerase chain reaction.
| Name of gene | Sequences
(5′-3′) |
|---|
| PIK3CA |
|
|
Forward |
CCACGACCATCATCAGGTGAA |
|
Reverse |
CCTCACGGAGGCATTCTAAAGT |
| Akt |
|
|
Forward |
GCGGCATCCACGAAACTAC |
|
Reverse |
TGATCTCCTTCTGCATCCTGTC |
| Survivin |
|
|
Forward |
GGCTCTTTCTCTGTCCAGTT |
|
Reverse |
ACCACCGCATCTCTACATTC |
| β-actin |
|
|
Forward |
CCAACCGCGAGAAGATGA |
|
Reverse |
CCAGAGGCGTACAGGGATAG |
For miR-1 detection, 1 µg total RNA extracted from
clinical samples was converted to cDNA using the TaqMan MicroRNA
Reverse Transcription kit (Applied Biosystems; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol. The
resulting cDNA was diluted in the ratio 1:40 and mixed with 1 µl
miR-1 or U6 TaqMan primers in triplicate wells using TaqMan
Universal Master Mix II without Uracil DNA glycosylase (Applied
Biosystems; Thermo Fisher Scientific, Inc.). The reaction condition
was as follows: Denaturation at 95°C for 30 sec, and followed by 40
cycles at 95°C for 5 sec and 60°C for 30 sec, and extension at 95°C
for 15 sec. The plates were read using the ABI PRISM 7900HT system
(Applied Biosystems; Thermo Fisher Scientific, Inc.). Ct values
were calculated using the SDS version 2.4 software (Applied
Biosystems; Thermo Fisher Scientific, Inc.). miR-1 expression level
was normalized to that of U6 using the 2−ΔΔCt method.
The TaqMan probes for miR-1 (assay ID, 002222) and U6 (assay ID,
001973) were purchased from Applied Biosystems (Thermo Fisher
Scientific, Inc.). The assay was performed in triplicate.
Western blotting
A total of 48 h after transfection, the cells were
lysed in lysis buffer [50 mM Tris (pH 7.4), 1% Triton X-100, 1%
sodium deoxycholate, 0.1% SDS, 150 mM NaCl, 1.0 mM EDTA, 1.0 mM
Na3VO4 and 1 µg/ml freshly added leupeptin].
The protein concentration of the lysates was determined using a BCA
protein assay (Bio-Rad, Laboratories Inc., Hercules, CA, USA).
Equivalent amounts of protein lysates (30 µg per lane) and loading
buffer were loaded onto 7.5 or 12.5% polyacrylamide gels, separated
by SDS-PAGE and electrophoretically transferred to polyvinylidene
membranes. The membranes were blocked for 1 h with 5% non-fat milk
in TBST buffer [20 mM Tris (pH 8.0), 150 nM NaCl and 0.05%
Tween-20] at room temperature and incubated overnight at 4°C with
one of the following primary antibodies: Anti-PIK3CA (cat. no.
AP80166; Abgent, Inc., San Diego, CA, USA) at 1:200, anti-Akt (cat.
no. 9272; Cell Signaling Technology, Inc., Danvers, MA, USA) at
1:1,000, rabbit monoclonal anti-phosphorylated (p)-Akt (cat. no.
4056; Thr308; 244F9; Cell Signaling Technology, Inc.) at 1:1,000,
biotinylated anti-human survivin (cat. no., BAF6471, R&D
Systems, Inc., Minneapolis, MN, USA) at 1:2,000 or anti-GAPDH (cat.
no., 5014; Cell Signaling Technology Inc.). The membranes were
incubated with the corresponding horseradish peroxidase-conjugated
rabbit anti-goat or goat anti-rat secondary antibodies (cat. nos.
MR-G100 and 2B-2305; Beyotime Institute of Biotechnology) at
1:5,000 for 1 h at room temperature, and proteins were visualized
using an ECL Chemiluminescence kit (EMD Millipore, Billerica, MA,
USA), detected and analyzed using the Bio-Rad Gel Doc XR system
(Bio-Rad, Laboratories Inc.) and ImageJ 2.1.4.7 software (National
Institutes of Health, Bethesda, MD, USA).
Cell proliferation assay
Cell proliferation was assessed using Cell Counting
kit-8 (Dojindo Molecular Technologies, Inc., Kumamoto, Japan).
Briefly, the transfected cells (5,000 cells/well) were seeded in
96-well plates and incubated overnight at 37°C. The cells were then
treated with various concentrations of gefitinib (0, 0.01, 0.1, 1
or 10 µM) for 48 h at 37°C. The water-soluble tetrazolium salt
WST-8 (10 µl) was added to each well and incubated for 1 h at 37°C.
The optical density was evaluated at 450 nm using a microplate
reader to determine the number of viable cells. Each experiment was
performed in triplicate.
Cell cycle arrest and apoptosis
assays
Transfected cells were treated with 3.0 µM gefitinib
for 24 h at 37°C. The cells were harvested and washed with PBS and
fixed with 70% ice-cold ethanol at −20°C for 10 min. Fixed cells
were rehydrated in PBS, stained with propidium iodide (PI)/RNase
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) for 30 min at 37°C
and analyzed by fluorescence-activated cell sorter (BD Biosciences,
San Jose, CA, USA). The percentage of cells in the
G0/G1, S and G2/M phases were
counted and compared. The experiments were performed in
triplicate.
Flow cytometry was used to determine the percentage
of apoptotic cells using the Annexin V/fluorescein isothiocyanate
and PI Apoptosis Detection kit (BD Biosciences), according to the
manufacturer's instructions. The cells were classified as viable,
dead, early apoptotic or late apoptotic. Flow cytometry was
performed using FACSCalibur flow cytometer and analyzed using the
automated CellQuest Pro 5.1 software (BD Biosciences). The
experiments were performed in triplicate.
Statistical analysis
Statistical analysis was performed using the
GraphPad Prism 5.0 software (GraphPad Software, Inc., La Jolla, CA,
USA). Values are presented as the mean ± standard deviation.
Differences between two groups were calculated using the Student's
t-test or the Mann-Whitney U test. The correlation between the
expression levels of miR-1 and PIK3CA were analyzed using Pearson's
correlation analysis. ESCC tissues expressing miR-1 and PIK3CA at
levels less than the median expression level were assigned to the
low expression group, and those with expression above the median
value were assigned to the high expression group. Associations
between clinicopathological features and the levels of miR-1 and
PIK3CA expression were analyzed using the χ2 test.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Levels of miR-1 and PIK3CA expression
in human ESCC samples
RT-qPCR was performed to analyze the levels of miR-l
and PIK3CA expression in 74 ESCC tissues and corresponding
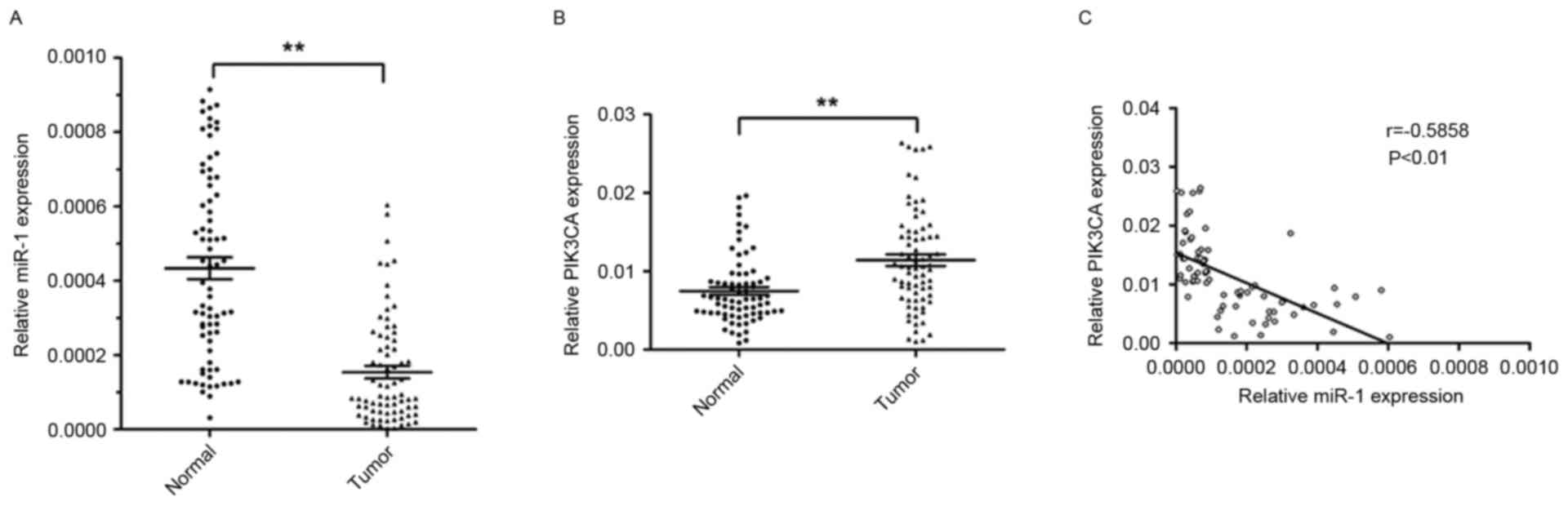
non-tumor tissues. The relative expression level of miR-1 in ESCC
tissues was significantly lower (~64.5%) compared with the
corresponding non-tumor tissues (P<0.01; Fig. 1A). However, the level of PIK3CA
expression was significantly higher in ESCC tissues (1.5 fold)
compared with corresponding non-tumor tissues (P<0.01; Fig. 1B).
The present study further investigated the
association between the level of miR-1 and PIK3CA expression. The
expression of miR-1 was inversely correlated with PIK3CA mRNA
expression (r=−0.5858; P<0.01; Fig.
1C). These results suggested that miR-1 may serve an important
role in suppressing the expression of PIK3CA in patients with
ESCC.
Association between the expression of
miR-1 and PIK3CA and clinicopathological characteristics of
ESCC
All patients were divided into four groups (miR-1
high, miR-1 low, PIK3CA high and PIK3CA low) on the basis of the
mean levels of miR-1 and PIK3CA expression in 74 ESCC samples. The
clinicopathological characteristics of these groups are summarized
in Table III. Low levels of miR-1
(P<0.001) and high levels of PIK3CA (P=0.006) expression were
strongly correlated with lymph node metastasis. Of the 41 tissue
samples without lymph node metastasis, 25 samples (60.98%)
exhibited high miR-1 expression levels and 13 samples (31.71%)
exhibited high PIK3CA expression. Of the 33 tissue samples with
lymph node metastasis, 5 samples (15.15%) exhibited high miR-1
expression and 21 samples (63.64%) exhibited high PIK3CA
expression. Furthermore, low miR-1 expression was associated with
high TNM stage (P<0.001). However, miR-1 and PIK3CA expression
levels were not associated with other patient characteristics,
including sex, age, pathological grading and invasion depth. These
results suggested that low miR-1 expression and high PIK3CA
expression may be associated with the pathophysiology of ESCC.
 | Table III.Association between the levels of
miR-1 and PIK3CA expression, and clinicopathological
characteristics in patients with esophageal squamous cell
carcinoma. |
Table III.
Association between the levels of
miR-1 and PIK3CA expression, and clinicopathological
characteristics in patients with esophageal squamous cell
carcinoma.
|
| miR-1
expression | PIK3CA
expression |
|---|
|
|
|
|
|---|
| Factors | High | Low | P-value | High | Low | P-value |
|---|
| Sex |
|
| 0.650 |
|
| 0.551 |
|
Male | 24 | 37 |
| 29 | 32 |
|
|
Female | 6 | 7 |
| 5 | 8 |
|
| Age, years |
|
| 0.255 |
|
| 0.801 |
|
<60 | 17 | 19 |
| 16 | 20 |
|
|
≥60 | 13 | 25 |
| 18 | 20 |
|
| Pathological
grading |
|
| 0.450 |
|
| 0.830 |
|
Well-moderately | 17 | 21 |
| 17 | 21 |
|
|
Poorly | 13 | 23 |
| 17 | 19 |
|
| Invasion depth |
|
| 0.080 |
|
| 0.653 |
|
T1/T2 | 10 | 7 |
| 7 | 10 |
|
|
T3/T4 | 20 | 37 |
| 27 | 30 |
|
| Lymph node
metastasis |
|
| <0.001 |
|
| 0.006 |
|
Positive | 5 | 28 |
| 21 | 12 |
|
|
Negative | 25 | 16 |
| 13 | 28 |
|
| TNM stage |
|
| 0.001 |
|
| 0.641 |
|
I/II | 22 | 15 |
| 16 | 21 |
|
|
III/IV | 8 | 29 |
| 18 | 19 |
|
Levels of miR-1 and PIK3CA expression
in transfected cells
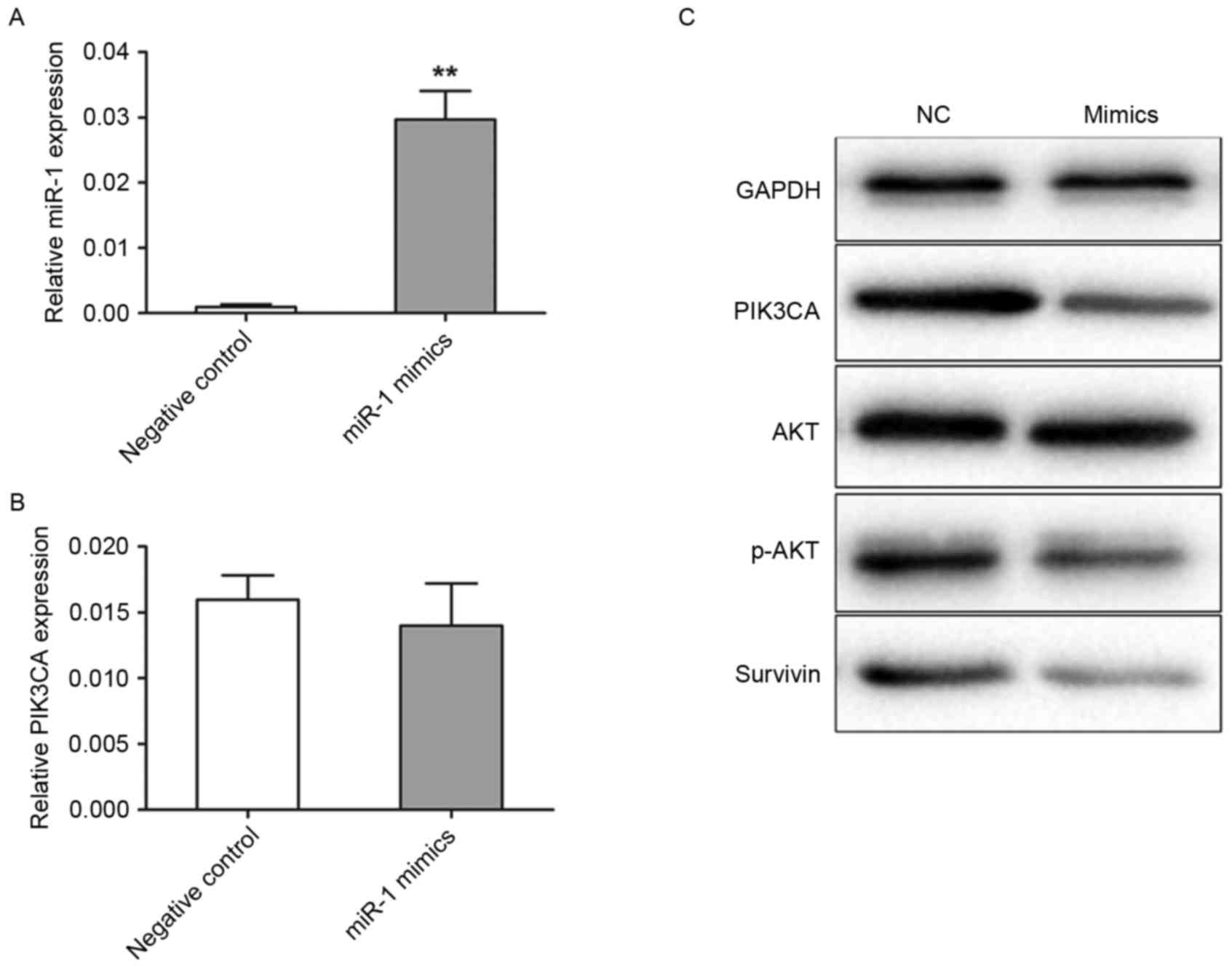
Transfection efficiency was detected using TaqMan
RT-qPCR. In the miR-1 mimics group, miR-1 expression level was
20.8-fold (P<0.001) higher compared with the negative control
group (Fig. 2A). The present study
further analyzed the levels of PIK3CA protein and mRNA expression
by western blotting and RT-qPCR in transfected TE-1 cells,
respectively. The levels of PIK3CA mRNA expression were not
significantly different between the miR-1 mimic and negative
control groups (Fig. 2B). However,
the level of PIK3CA protein expression was revealed to be markedly
decreased in the cells transfected with miR-1 mimics compared with
the cells transfected with control miRNA (Fig. 2C). Akt and survivin are important
downstream targets of PIK3CA, therefore in the present study the
activation of Akt and survivin following PIK3CA regulation were
analyzed. PIK3CA downregulation by miR-1 mimics induced a marked
reduction in the levels of p-Akt and survivin expression, but not
in the levels of total Akt expression, when compared with the
negative control (Fig. 2C).
Exogenous expression level of miR-1
inhibited growth of TE-1 cells
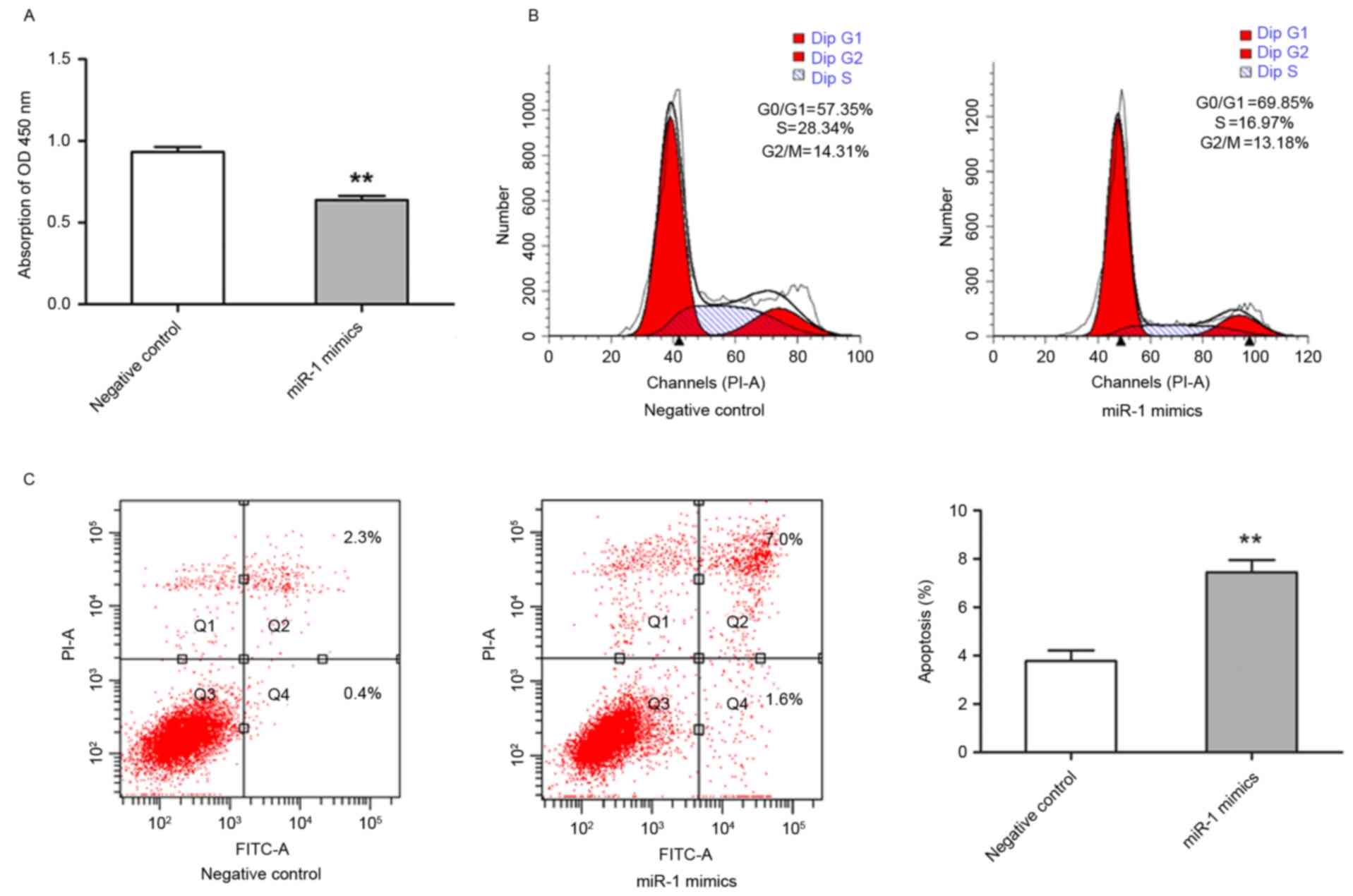
It was demonstrated that miR-1 expression was
downregulated in ESCC, therefore, subsequent study aimed to
establish the biological effect of this change in expression on
cell growth. TE-1 cells were transfected with miR-1 mimics, and the
effects on cell proliferation were analyzed. TE-1 cell growth was
significantly inhibited in miR-1-transfected cells compared with
control miRNA-transfected cells (P<0.01; Fig. 3A). To further characterize
miR-1-mediated inhibition of cell proliferation, cell cycle
distribution and apoptotic rate were evaluated by flow cytometry.
The cells transfected with miR-1 had an increased percentage of
G0/G1 phase cells and a decreased percentage
of G2/M phase cells in comparison with the cells
transfected with control miRNA. The percentage of S phase cells was
also markedly decreased in miR-1-expressing cells (Fig. 3B). In addition, the cells transfected
with miR-1 exhibited significantly increased apoptosis, including
early and late apoptosis, compared with the control group
(P<0.01; Fig. 3C). These results
suggested that miR-1 was able to inhibit growth of ESCC cells by
modulating apoptosis and cell cycle progression.
miR-1 increased sensitivity to
gefitinib in TE-1 cells
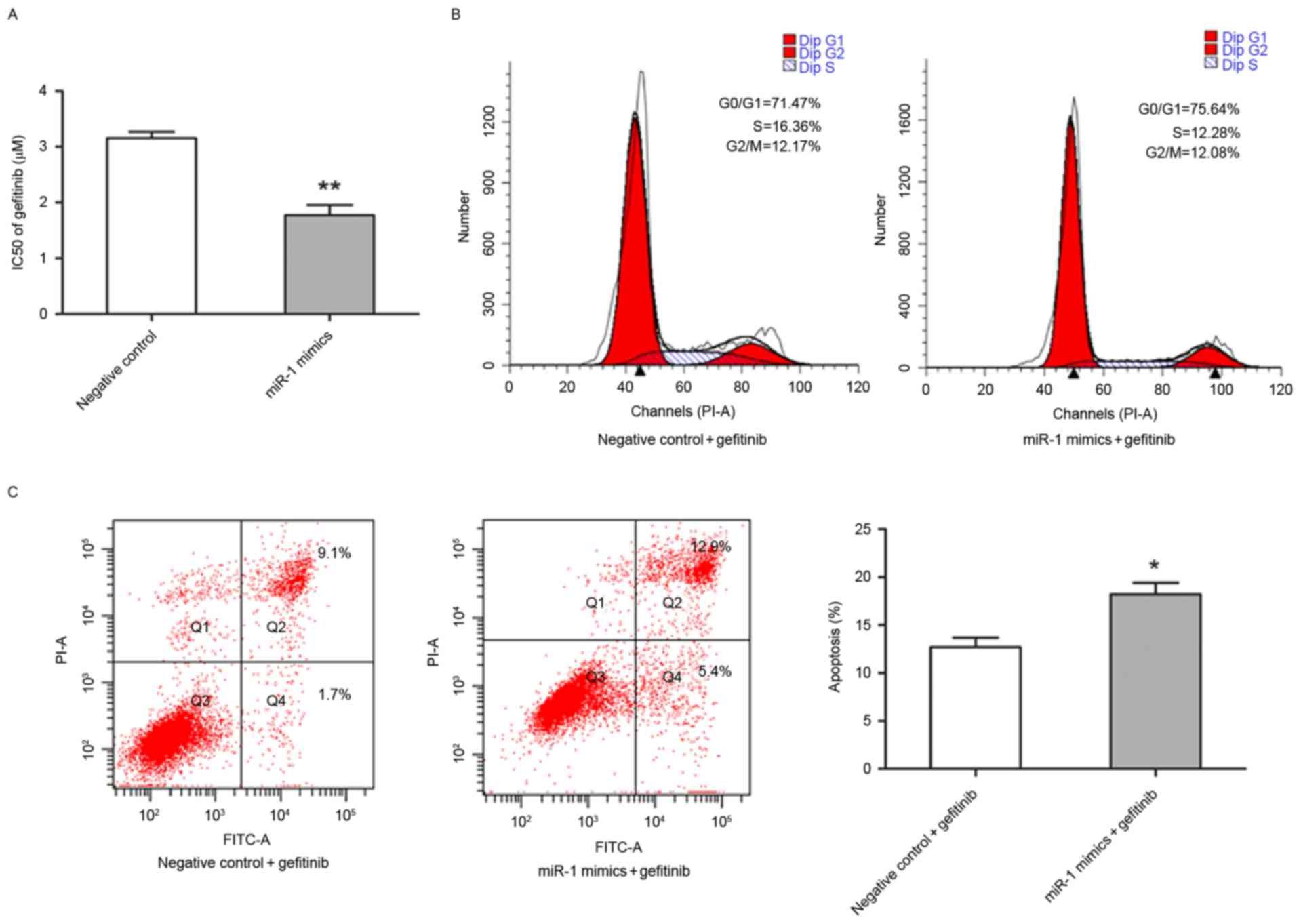
The present study also investigated whether miR-1 is
able to affect the sensitivity of TE-1 cells to gefitinib. The
half-maximal inhibitory concentration value of gefitinib was
significantly lower in miR-1-expressing TE-1 cells (1.77±0.18 µM)
compared with the control TE-1 cells (3.16±0.11 µM; P<0.01;
Fig. 4A). Flow cytometry was
performed to determine whether the increased sensitivity to
gefitibnib was due to alternation of cell cycle progression and
apoptotic rate. Notably, that the percentage of TE-1 cells in
different stages of the cell cycle were distinct in
miR-1-expressing cells compared with control cells following
exposure to 3.0 µM gefitinib for 24 h (Fig. 4B). In the presence of gefitinib, the
apoptotic rate of TE-1 cells transfected with miR-1 mimics was
significantly higher compared with TE-1 cells transfected with the
control miRNA (P<0.05; Fig.
4C).
Discussion
The carcinogenesis of ESCC is a multi-stage process
involving a variety of changes in gene expression and physiological
structure. miRNA expression is aberrant in ESCC, suggesting that
miRNAs serve an important role in ESCC progression (33). Recently, it was confirmed that miR-1
functions as a tumor suppressor in carcinogenesis (21–29,34,35).
The present study revealed that miR-1 was significantly
downregulated in ESCC tissues compared with normal esophageal
tissues. Therefore, miR-1 may act as a tumor suppressor in
ESCC.
Previous studies by the present authors revealed
that miR-1 expression was significantly lower in patients with lung
cancer compared with normal tissues, and the low-miR-1 expression
group exhibited a significantly higher recurrence rate compared
with those in the moderate-miR1 expression level group (28,35).
Therefore, detection of miR-1 expression may be a valuable tool to
evaluate invasion and metastasis of human NSCLC. Similar to the
results obtained for patients with NSCLC, the present study
demonstrated that the level of miR-1 expression was associated with
clinical stage and lymph node metastasis in patients with ESCC,
suggesting that low miR-1 expression may be associated with ESCC
progression. Functional studies in the TE-1 ESCC cell line
confirmed that upregulation of miR-1 may inhibit growth via
increased apoptosis and/or cell cycle arrest in the
G0/G1 phase.
The PI3K/Akt signaling pathway serves a critical
role in esophageal cancer pathogenesis (32). The involvement of the PIK3CA
gene, which encodes the PI3K protein p110α catalytic subunit, is
activated by a series of cell surface tyrosine kinase receptors,
including platelet-derived growth factor receptor and insulin
growth factor receptor (38). Upon
activation of these receptors, PIK3CA binds to its heterodimer p85
and promotes Akt phosphorylation at Thr308 and/or Ser473. P-Akt
then activates a series of processes that drive tumor progression,
including cell growth, proliferation, survival and motility
(39,40). Previously, PIK3CA was identified as a
target of miR-1 in the NSCLC A549 cell line by the present authors
(35). miR-1 overexpression in A549
cells suppressed tumorigenic properties via PIK3CA repression and
constitutive suppression of the PI3K/Akt/survivin signaling pathway
(28,35). The present study revealed that the
level of PIK3CA expression was higher in ESCC tissues compared with
normal tissues and was inversely correlated with the level of miR-1
expression. Upregulation of miR-1 may also inhibit growth of ESCC
cells via downregulation of the PI3K/Akt/survivin signaling
pathway. On the basis of these findings, the present study
suggested that decreased miR-1 expression modulates PIK3CA
signaling in the PI3K/Akt/survivin pathway and promotes cell
growth. Therefore, increasing miR-1 expression may be a novel
approach for the treatment of ESCC.
High expression of EGFR may correlate with a poor
response to therapy, development of cytotoxic drug resistance,
disease progression and poor survival in various types of human
cancer (41–43). Therefore, blockade of EGFR signal
transduction appears to be a promising strategy for cancer therapy.
Gefitinib is an orally active EGFR tyrosine kinase inhibitor. The
antitumor activity of gefitinib has been confirmed in vitro
and in vivo in various types of human cancer, including
NSCLC, and colorectal, breast and head and neck cancer (41–44).
Approximately 40–70% of patients with ESCC demonstrated high
expression levels of EGFR (45,46).
Numerous previous studies revealed that gefitinib had strong
antitumor activity against ESCC in vitro and in vivo
(7–9).
However, a large recent clinical trial involving patients with
esophageal cancer revealed that the use of gefitinib as a
second-line treatment in unselected patients does not improve
overall survival compared with the placebo (47). Another phase II study demonstrated
that gefitinib was well tolerated by patients with recurrent or
metastatic adenocarcinoma or squamous cell carcinoma of the
esophagus or gastroesophageal junction, but had limited efficacy
(48). It has been revealed that the
PI3K/Akt signaling pathway confers gefitinib resistance independent
of EGFR (49). Gefitinib in
combination with specific inhibitors of the PI3K/Akt signaling
pathway may cause additional cytotoxic effects in ESCC cell lines.
Previous studies indicated that certain miRNAs may alter the
sensitivity of cancer cells to therapeutic agents (50,51). The
present study investigated whether exogenous miR-1 is able to alter
sensitivity of ESCC cells to gefitinib. It was revealed that miR-1
blocked the activation of the PI3K/Akt signaling pathway and
increased the sensitivity of ESCC cells to gefitinib. Therefore,
the results of the present study suggested a role for microRNAs in
chemosensitivity of ESCC cells. The present study suggested that a
combination of miR-1 and gefitinib may be a successful therapeutic
strategy for ESCC. It would be of interest to elucidate the
underlying molecular mechanisms for miR-1-mediated
gefitinib-induced antitumor activity. Although efficacy and
tolerability of gefitinib and miR-1 needs to be extensively tested
in preclinical models, the results of the present study provided a
novel promising approach to improving chemotherapeutic
efficacy.
To conclude, the present study demonstrated that
miR-1 was downregulated in ESCC and is able to function as a tumor
suppressor in this type of cancer. Overexpression of miR-1
inhibited growth, increased apoptosis and induced cell cycle arrest
in the G0/G1 cell cycle phase, possibly by
suppressing the PI3K/Akt/survivin signaling pathway. Additionally,
miR-1 may increase the sensitivity of ESCC cells to gefitinib.
Therefore, miR-1 appears to be a promising therapeutic target for
ESCC treatment.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China (grant no. 81172217), the
Medical Important Talents of Jiangsu Province (grant no. RC201157)
and the Project of Oncology Translational Medicine Central of
Jiangsu Province (grant no. BL2012008).
References
|
1
|
Smyth EC, Lagergren J, Fitzgerald RC,
Lordick F, Shah MA, Lagergren P and Cunningham D: Oesophageal
cancer. Nat Rev Dis Primers. 3:170482017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zeng H, Zheng R, Zhang S, Zuo T, Xia C,
Zou X and Chen W: Esophageal cancer statistics in China, 2011:
Estimates based on 177 cancer registries. Thorac Cancer. 7:232–237.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Office for National Statistics, . Cancer
survival in England: Patients diagnosed 2006–2010 and followed up
to 2011. Newport: Office for National Statistics; 2012
|
|
4
|
Hanawa M, Suzuki S, Dobashi Y, Yamane T,
Kono K, Enomoto N and Ooi A: EGFR protein overexpression and gene
amplification in squamous cell carcinomas of the esophagus. Int J
Cancer. 118:1173–1180. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mendelsohn J and Baselga J: Status of
epidermal growth factor receptor antagonists in the biology and
treatment of cancer. J Clin Oncol. 21:2787–2799. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Teraishi F, Kagawa S, Watanabe T, Tango Y,
Kawashima T, Umeoka T, Nisizaki M, Tanaka N and Fujiwara T: ZD1839
(Gefitinib, ‘Iressa’), an epidermal growth factor receptor-tyrosine
kinase inhibitor, enhances the anti-cancer effects of TRAIL in
human esophageal squamous cell carcinoma. FEBS Lett. 579:4069–4075.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hara F, Aoe M, Doihara H, Taira N, Shien
T, Takahashi H, Yoshitomi S, Tsukuda K, Toyooka S, Ohta T and
Shimizu N: Antitumor effect of gefitinib (‘Iressa’) on esophageal
squamous cell carcinoma cell lines in vitro and in vivo. Cancer
Lett. 226:37–47. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wei Z, Ma W, Qi X, Zhu X, Wang Y, Xu Z,
Luo J, Wang D, Guo W, Li X, et al: Pinin facilitated proliferation
and metastasis of colorectal cancer through activating EGFR/ERK
signaling pathway. Oncotarget. 7:29429–29439. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Baselga J and Arteaga CL: Critical update
and emerging trends in epidermal growth factor receptor targeting
in cancer. J Clin Oncol. 23:2445–2459. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sutter AP, Höpfner M, Huether A, Maaser K
and Scherübl H: Targeting the epidermal growth factor receptor by
erlotinib (Tarceva) for the treatment of esophageal cancer. Int J
Cancer. 118:1814–1822. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Carey KD, Garton AJ, Romero MS, Kahler J,
Thomson S, Ross S, Park F, Haley JD, Gibson N and Sliwkowski MX:
Kinetic analysis of epidermal growth factor receptor somatic mutant
proteins shows increased sensitivity to the epidermal growth factor
receptor tyrosine kinase inhibitor, erlotinib. Cancer Res.
66:8163–8171. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Saxena S, Jonsson ZO and Dutta A: Small
RNAs with imperfect match to endogenous mRNA repress translation.
Implications for off-target activity of small inhibitory RNA in
mammalian cells. J Biol Chem. 278:44312–44319. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yanaihara N, Caplen N, Bowman E, Seike M,
Kumamoto K, Yi M, Stephens RM, Okamoto A, Yokota J, Tanaka T, et
al: Unique microRNA molecular profiles in lung cancer diagnosis and
prognosis. Cancer Cell. 9:189–198. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Iorio MV, Ferracin M, Liu CG, Veronese A,
Spizzo R, Sabbioni S, Magri E, Pedriali M, Fabbri M, Campiglio M,
et al: MicroRNA gene expression deregulation in human breast
cancer. Cancer Res. 65:7065–7070. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Voorhoeve PM, le Sage C, Schrier M, Gillis
AJ, Stoop H, Nagel R, Liu YP, van Duijse J, Drost J, Griekspoor A,
et al: A genetic screen implicates miRNA-372 and miRNA-373 as
oncogenes in testicular germ cell tumors. Cell. 124:1169–1181.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kent OA and Mendell JT: A small piece in
the cancer puzzle: microRNAs as tumor suppressors and oncogenes.
Oncogene. 25:6188–6196. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ma W, Yu J, Qi X, Liang L, Zhang Y and
Ding Y, Lin X, Li G and Ding Y: Radiation-induced MicroRNA-622
inhibits radiosensitivity in colorectal cancer cells by targeting
Rb. Oncotarget. 6:15984–15994. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Meng F, Henson R, Wehbe-Janek H, Ghoshal
K, Jacob ST and Patel T: MicroRNA-21 regulates expression of the
PTEN tumor suppressor gene in human hepatocellular cancer.
Gastroenterology. 133:647–658. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lu XJ, Qi X, Zheng DH and Ji LJ: Modelling
cancer processes with CRISPR-Cas9. Trends Biotechnol. 33:317–319.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yan D, Dong Xda E, Chen X, Wang L, Lu C,
Wang J, Qu J and Tu L: MicroRNA-1/206 targets c-Met and inhibits
rhabdomyosarcoma development. J Biol Chem. 284:29596–29604. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nasser MW, Datta J, Nuovo G, Kutay H,
Motiwala T, Majumder S, Wang B, Suster S, Jacob ST and Ghoshal K:
Down-regulation of micro-RNA-1 (miR-1) in lung cancer. Suppression
of tumorigenic property of lung cancer cells and their
sensitization to doxorubicin-induced apoptosis by miR-1. J Biol
Chem. 283:33394–33405. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Leone V, D'Angelo D, Rubio I, de Freitas
PM, Federico A, Colamaio M, Pallante P, Medeiros-Neto G and Fusco
A: MiR-1 is a tumor suppressor in thyroid carcinogenesis targeting
CCND2, CXCR4, and SDF-1alpha. J Clin Endocrinol Metab.
96:E1388–E1398. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kojima S, Chiyomaru T, Kawakami K, Yoshino
H, Enokida H, Nohata N, Fuse M, Ichikawa T, Naya Y, Nakagawa M and
Seki N: Tumour suppressors miR-1 and miR-133a target the oncogenic
function of purine nucleoside phosphorylase (PNP) in prostate
cancer. Br J Cancer. 106:405–413. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hudson RS, Yi M, Esposito D, Watkins SK,
Hurwitz AA, Yfantis HG, Lee DH, Borin JF, Naslund MJ, Alexander RB,
et al: MicroRNA-1 is a candidate tumor suppressor and prognostic
marker in human prostate cancer. Nucleic Acids Res. 40:3689–3703.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yoshino H, Enokida H, Chiyomaru T,
Tatarano S, Hidaka H, Yamasaki T, Gotannda T, Tachiwada T, Nohata
N, Yamane T, et al: Tumor suppressive microRNA-1 mediated novel
apoptosis pathways through direct inhibition of splicing factor
serine/arginine-rich 9 (SRSF9/SRp30c) in bladder cancer. Biochem
Biophys Res Commun. 417:588–593. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Migliore C, Martin V, Leoni VP, Restivo A,
Atzori L, Petrelli A, Isella C, Zorcolo L, Sarotto I, Casula G, et
al: MiR-1 downregulation cooperates with MACC1 in promoting MET
overexpression in human colon cancer. Clin Cancer Res. 18:737–747.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Datta J, Kutay H, Nasser MW, Nuovo GJ,
Wang B, Majumder S, Liu CG, Volinia S, Croce CM, Schmittgen TD, et
al: Methylation mediated silencing of MicroRNA-1 gene and its role
in hepatocellular carcinogenesis. Cancer Res. 68:5049–5058. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhao Q, Zhang B, Shao Y, Chen L, Wang X,
Zhang Z, Shu Y and Guo R: Correlation between the expression levels
of microRNA-1 and PIK3CA in non-small-cell lung cancer and their
relationship with clinical characteristics and prognosis. Future
Oncol. 10:49–57. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Nohata N, Sone Y, Hanazawa T, Fuse M,
Kikkawa N, Yoshino H, Chiyomaru T, Kawakami K, Enokida H, Nakagawa
M, et al: miR-1 as a tumor suppressive microRNA targeting TAGLN2 in
head and neck squamous cell carcinoma. Oncotarget. 2:29–42. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Guo Y, Chen Z, Zhang L, Zhou F, Shi S,
Feng X, Li B, Meng X, Ma X, Luo M, et al: Distinctive microRNA
profiles relating to patient survival in esophageal squamous cell
carcinoma. Cancer Res. 68:26–33. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen H, Guan R, Lei Y, Chen J, Ge Q, Zhang
X, Dou R, Chen H, Liu H, Qi X, et al: Lymphangiogenesis in gastric
cancer regulated through Akt/mTOR-VEGF-C/VEGF-D axis. BMC Cancer.
15:1032015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Akagi I, Miyashita M, Makino H, Nomura T,
Hagiwara N, Takahashi K, Cho K, Mishima T, Ishibashi O, Ushijima T,
et al: Overexpression of PIK3CA is associated with lymph node
metastasis in esophageal squamous cell carcinoma. Int J Oncol.
34:767–775. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yang YL, Chu JY, Luo ML, Wu YP, Zhang Y,
Feng YB, Shi ZZ, Xu X, Han YL, Cai Y, et al: Amplification of
PRKCI, located in 3q26, is associated with lymph node metastasis in
esophageal squamous cell carcinoma. Genes Chromosomes Cancer.
47:127–136. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wada S, Noguchi T, Takeno S and Kawahara
K: PIK3CA and TFRC located in 3q are new prognostic factors in
esophageal squamous cell carcinoma. Ann Surg Oncol. 13:961–966.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yu QQ, Wu H, Huang X, Shen H, Shu YQ,
Zhang B, Xiang CC, Yu SM, Guo RH and Chen L: miR-1 targets PIK3CA
and inhibits tumorigenic properties of A549 cells. Biomed
Pharmacother. 68:155–161. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Mönig SP and Hölscher AH: Clinical
classification systems of adenocarcinoma of the esophagogastric
junction. Recent Results Cancer Res. 182:19–28. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Vanhaesebroeck B, Stein RC and Waterfield
MD: The study of phosphoinositide 3-kinase function. Cancer Surv.
27:249–270. 1996.PubMed/NCBI
|
|
39
|
Andjelković M, Alessi DR, Meier R,
Fernandez A, Lamb NJ, Frech M, Cron P, Cohen P, Lucocq JM and
Hemmings BA: Role of translocation in the activation and function
of protein kinase B. J Biol Chem. 272:31515–31524. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Vivanco I and Sawyers CL: The
phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nat Rev
Cancer. 2:489–501. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
41
|
Bianco C, Tortora G, Bianco R, Caputo R,
Veneziani BM, Caputo R, Damiano V, Troiani T, Fontanini G, Raben D,
et al: Enhancement of antitumor activity of ionizing radiation by
combined treatment with the selective epidermal growth factor
receptor tyrosine kinase inhibitor ZD1839 (Iressa). Clin Cancer
Res. 8:3250–3258. 2002.PubMed/NCBI
|
|
42
|
Qi X, Zhang L and Lu X: New insights into
epithelial-to-mesenchymal transition in cancer. Trends Pharmacol
Sci. 37:246–248. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Sirotnak FM, Zakowski MF, Miller VA, Scher
HI and Kris MG: Efficacy of cytotoxic agents against human tumor
xenografts is markedly enhanced by coadministration of ZD1839
(Iressa), an inhibitor of EGFR tyrosine kinase. Clin Cancer Res.
6:4885–4892. 2000.PubMed/NCBI
|
|
44
|
Baselga J, Rischin D, Ranson M, Calvert H,
Raymond E, Kieback DG, Kaye SB, Gianni L, Harris A, Bjork T, et al:
Phase I safety, pharmacokinetic, and pharmacodynamic trial of
ZD1839, a selective oral epidermal growth factor receptor tyrosine
kinase inhibitor, in patients with five selected solid tumor types.
J Clin Oncol. 20:4292–4302. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Iihara K, Shiozaki H, Tahara H, Kobayashi
K, Inoue M, Tamura S, Miyata M, Oka H, Doki Y and Mori T:
Prognostic significance of transforming growth factor-alpha in
human esophageal carcinoma. Implication for the autocrine
proliferation. Cancer. 71:2902–2909. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ozawa S, Ueda M, Ando N, Abe O and Shimizu
N: High incidence of EGF receptor hyperproduction in esophageal
squamous-cell carcinomas. Int J Cancer. 39:333–337. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Dutton SJ, Ferry DR, Blazeby JM, Abbas H,
Dahle-Smith A, Mansoor W, Thompson J, Harrison M, Chatterjee A,
Falk S, et al: Gefitinib for oesophageal cancer progressing after
chemotherapy (COG): A phase 3, multicentre, double-blind,
placebo-controlled randomised trial. Lancet Oncol. 15:894–904.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Adelstein DJ, Rodriguez CP, Rybicki LA,
Ives DI and Rice TW: A phase II trial of gefitinib for recurrent or
metastatic cancer of the esophagus or gastroesophageal junction.
Invest New Drugs. 30:1684–1689. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Janmaat ML, Kruyt FA, Rodriguez JA and
Giaccone G: Response to epidermal growth factor receptor inhibitors
in non-small cell lung cancer cells: Limited antiproliferative
effects and absence of apoptosis associated with persistent
activity of extracellular signal-regulated kinase or Akt kinase
pathways. Clin Cancer Res. 9:2316–2326. 2003.PubMed/NCBI
|
|
50
|
Meng F, Henson R, Lang M, Wehbe H,
Maheshwari S, Mendell JT, Jiang J, Schmittgen TD and Patel T:
Involvement of human micro-RNA in growth and response to
chemotherapy in human cholangiocarcinoma cell lines.
Gastroenterology. 130:2113–2129. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Weidhaas JB, Babar I, Nallur SM, Trang P,
Roush S, Boehm M, Gillespie E and Slack FJ: MicroRNAs as potential
agents to alter resistance to cytotoxic anticancer therapy. Cancer
Res. 67:11111–11116. 2007. View Article : Google Scholar : PubMed/NCBI
|