Introduction
Colorectal cancer (CRC) is one of the most common
types of gastrointestinal cancer in China (1). CRC increased from being the fifth most
common cause of mortality in 2000 to the third most common cause in
2015 (2). As colonoscopy is not a
popular screening method in China, a majority of patients are
diagnosed at advanced stages of caner (3). In addition, effective treatment
protocols for patients following surgery or chemotherapy have not
developed successfully (4,5). The patients that are diagnosed at an
early-stage CRC have a better prognosis, and the five-year survival
rate is ~90% following surgery (6).
Radical resection has been the primary treatment for early-stage
CRC (including stage I–II) (7).
However, adjuvant chemotherapy has additionally become an important
treatment (8–10). Regardless of whether patients receive
adjuvant therapy, dynamic monitoring following surgery is
associated with the improved survival rates of patients with
early-stage CRC (11–13). The sensitivity of existing tumor
biomarkers [including carcinoembryonic antigen (CEA), cancer
antigen 125 (CA125) and carbohydrate antigen 19-9] and other tests
[including computed tomography (CT)] used for monitoring CRC is low
(14–16). Therefore, identifying a highly
sensitive clinical biomarker for the detection of CRC is of utmost
importance.
Circulating tumor DNA (ctDNA) is fragmented DNA that
is released from exfoliated tumor cells into the blood (17). ctDNA carries genetic information from
the primary tumor and displays the genetic mutations of the tumor,
which overcomes the problem of tumor heterogeneity (18). CtDNA is additionally used to monitor
responses to treatment and to make decisions on further therapy
during follow-up visits (19–23). Previous studies have revealed that
mutations in plasma ctDNA and tumor biopsies were similar for
patients with early breast cancer and CRC, and the majority of
patients (89%) with driver mutations detected in ctDNA would recur
2–4 weeks postoperatively (24,25).
As a noninvasive detection method, ctDNA as a marker
reveals distinct advantages in patients with advanced cancer over
other detection methods. However, using ctDNA analysis to monitor
therapy is a less sensitive method for patients in the early stages
of cancer, which is likely as early-stage tumors release only a few
DNA fragments into the blood (26).
However, other sequencing methods additionally have problems that
limit early detection (17,27). In the present study, next generation
sequencing (NGS) technology was used to reveal significant
associations between changes in ctDNA and different stages of CRC
progression in order to examine the value of ctDNA in the clinical
diagnosis and treatment of patients with CRC.
Materials and methods
Ethics statement
The present study was approved by the Shenzhen
People's Hospital Ethics Committee, and the protocols were
performed in accordance with the approved guidelines (28). Written informed consent was obtained
from each patient for the use of blood and tumor tissues with the
approval of the Shenzhen People's Hospital Ethics Committee
(Shenzen, China). All samples and clinical data used in the present
study were double-blind.
Patients and collection of clinical
samples
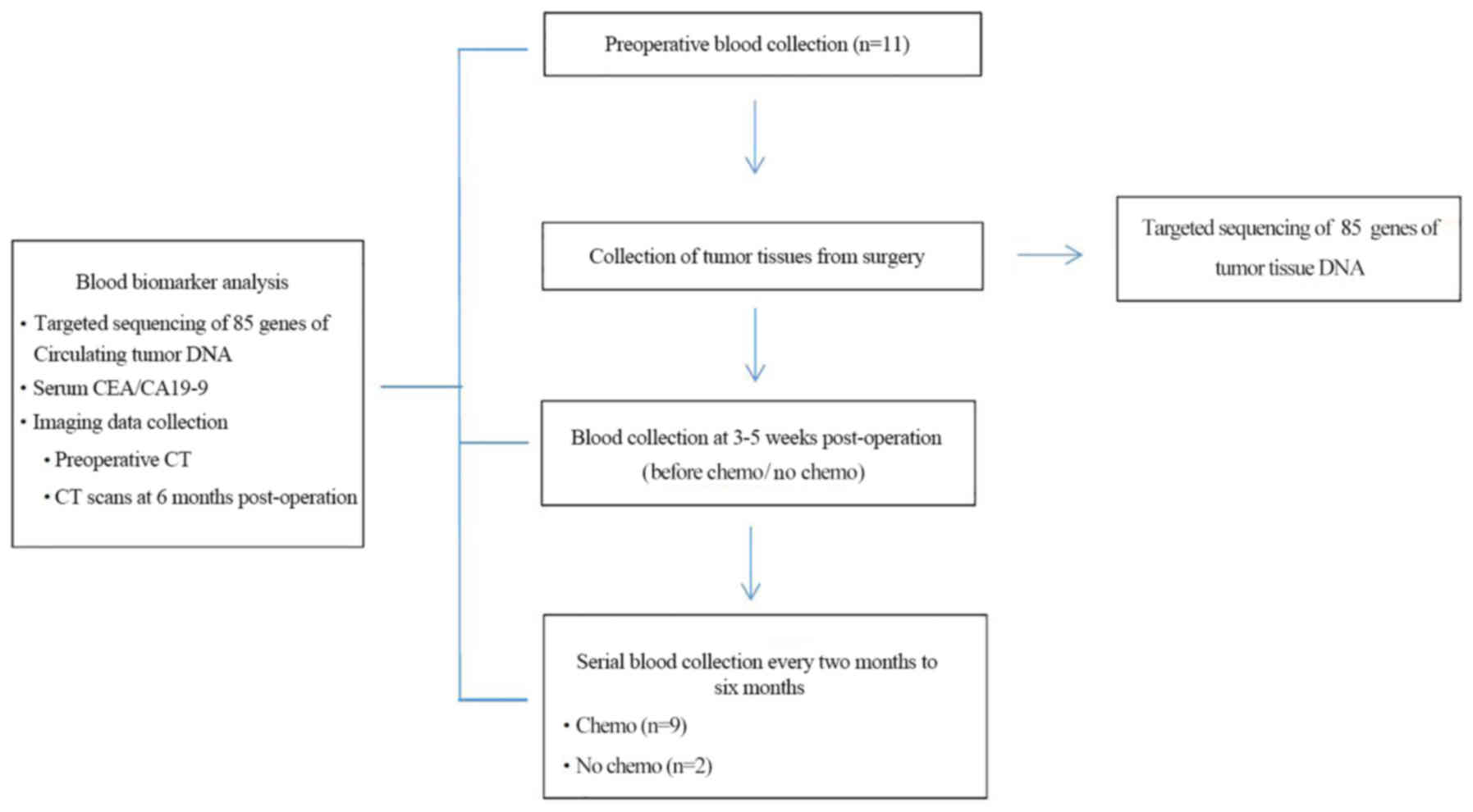
The present study recruited 11 patients with primary
CRC who underwent radical resection in the Department of
Gastrointestinal Surgery at the Shenzhen People's Hospital from
September 2016 to April 2017. Exclusion criteria: i) Pregnant or
lactating women; ii) patients who had received prior treatment with
a MEK inhibitor, and iii) a history of clinically significant
interstitial lung disease or pneumonitis. Tumor tissues were
collected during surgery, quickly frozen in liquid nitrogen and
stored at −80°C. Blood samples (20 ml) for ctDNA and CEA analysis
were collected from 4–10 weeks postoperatively, with serial
twice-monthly blood sample collection, which lasted for up to six
months (Fig. 1). Adjuvant
chemotherapy was used at the discretion of the treating clinician,
who was blinded to the ctDNA results. As per protocol, follow-up
included a twice-monthly clinical review, detection of CEA levels
and CT imaging for six months. Subsequently, follow-up visits were
performed according to the standard of care at participating
institutions, which included three-month and six-month CT visits
for two years.
DNA extraction and sequencing
Tumor DNA was isolated from surgically resected CRC
specimens. Blood samples were collected in tubes containing EDTA
and centrifuged at 1,600 × g for 10 min at 4°C within 2 h of
collection. Peripheral blood lymphocyte (PBL) debris was stored at
−20°C until further use. The supernatants were further centrifuged
at 10,000 × g for 10 min at 4°C, and plasma was harvested and
stored at −80°C until further use. PBL DNA was extracted using the
RelaxGene Blood DNA system (Tiangen Biotech Co., Ltd., Beijing,
China), and ctDNA was extracted from ≥2 ml of plasma using the
QIAamp Circulating Nucleic Acid kit (Qiagen, Inc., Valencia, CA,
USA) according to the manufacturer's protocol. DNA was quantified
using the Qubit 2.0 fluorometer and the Qubit dsDNA HS Assay kit
(Thermo Fisher Scientific, Inc., Waltham, MA, USA) according to the
manufacturer's protocol.
DNA was collected from PBLs and was sheared using
the enzyme dsDNA Fragmentase (New England BioLabs, Inc., Ipswich,
MA, USA). Size selection of the DNA fragments (150–250 bp) was then
performed using Ampure XP beads (Beckman Coulter, Inc., Brea, CA,
USA), which has the additional benefits of higher recovery and
greater speed. DNA fragments and ctDNA were used for library
construction using the KAPA Library Preparation kit (Kapa
Biosystems, Inc., Wilmington, MA, USA) according to the
manufacturer's protocol. Agencourt AMPure XP beads (Beckman
Coulter, Inc., Brea, CA, USA) were used for all the cleanup steps.
End repair and 3′-end A-tailing were performed following DNA
fragmentation. Notably, T-tailed adapters were used and a 3′dA
overhang was added enzymatically onto the fragmented DNA sample.
Ligation was performed for 15 min at 20°C. Single-step size
selection was performed by adding 50 µl (×1) of PEG/NaCl SPRI
Solution buffer (Beckman Coulter, Inc.) to enrich ligated DNA
fragments. The ligated fragments were then amplified using 1× KAPA
HiFi Hot Start Ready mix (Kapa Biosystems, Inc.) and Pre-LM-PCR
Oligos (Kapa Biosystems, Inc.) in 50 µl reactions, and 7–8 PCR
cycles were performed depending on the amount of DNA input. The
thermocycling conditions were as follows: Initial Denaturation,
98°C for 30 sec; 7–8 cycles at 98°C for 10 sec; 60°C for 10 sec;
68°C for 30 sec, and the final extension at 68°C for 60 sec. The
purity and concentration of the DNA fragments were assessed using
the Qubit 2.0 fluorometer and the Qubit dsDNA HS Assay kit.
Fragment length was determined on a 4200 Bioanalyzer (Agilent
Technologies, Inc., Santa Clara, CA, USA) using the DNA 1000 kit
(Agilent Technologies, Inc., Santa Clara, CA, USA). DNA sequencing
was then performed on the Illumina NextSeq 500 system according to
the manufacturer's recommendations at a depth of 7,000x.
Variant calling and analysis
Sequenced genes and their genebank accession numbers
are provided in Table I (Colorectal
cancer gene panel, 85 genes). Sequencing data were de-multiplexed
and aligned to the hg19 genome (GRch37) using Burrows-Wheeler
Aligner (29) version 0.7.15-r1140
using default settings. Pileup files for properly paired reads with
mapping quality ≥60 were generated using Samtools (http://www.htslib.org/) (30). Somatic variants call were created
using VarScan2 (http://varscan.sourceforge.net/) (31) [the minimum read depth at a
position=100; the variant allele frequency (VAF) is the proportion
of reads at a site which contain the variant allele. VAF threshold
≥0.05; somatic-P-value ≤0.01; strand-filter ≥1; otherwise, default
parameters]. Allele frequencies were calculated for all Q30 bases.
Using a custom Python script, previously identified tumor DNA
mutations were intersected with a Samtools mpileup file generated
for each plasma DNA sample; the number and frequency were then
calculated for each mutation. For detection of mutations from
plasma DNA without tumor DNA present, a mutation was identified if
≥4 mutant reads were identified in plasma and ≥1 mutant read was
identified on each strand. No mutant reads were observed in matched
genomic DNA from white blood cells.
 | Table I.Colorectal cancer gene panel (85
genes). |
Table I.
Colorectal cancer gene panel (85
genes).
| Gene name | GenBank accession
number |
|---|
| ABCB1 | NM_000927 |
| AKAP9 | NM_005751 |
| AKT1 | NM_005163 |
| APC | NM_000038 |
| ARID1A | NM_006015 |
| ATIC | NM_004044 |
| ATM | NM_000051 |
| AXIN2 | NM_004655 |
| BARD1 | NM_000465 |
| BMPR1A | NM_004329 |
| BRAF | NM_004333 |
| BRCA1 | NM_007294 |
| BRCA2 | NM_000059 |
| BRIP1 | NM_032043 |
| BUB1 | NM_004336 |
| C8orf34 | NM_052958 |
| CBR3 | NM_001236 |
| CDA | NM_001785 |
| CDH1 | NM_004360 |
| CHEK2 | NM_007194 |
| CREBBP | NM_004380 |
| CTNNB1 | NM_001904 |
| CYP19A1 | NM_000103 |
| CYP2D6 | NM_000106 |
| DPYD | NM_000110 |
| EGFR | NM_005228 |
| EIF3E | NM_001568 |
| EPCAM | NM_002354 |
| ERBB2 | NM_004448 |
| ERBB4 | NM_005235 |
| ERCC1 | NM_001983 |
| FBXW7 | NM_018315 |
| GREM1 | NM_013372 |
| GSTP1 | NM_000852 |
| HRAS | NM_005343 |
| KDR | NM_002253 |
| KMT2C | NM_170606 |
| KMT2D | NM_003482 |
| KRAS | NM_004985 |
| MET | NM_000245 |
| MLH1 | NM_000249 |
| MLH3 | NM_014381 |
| MRE11A | NM_005590 |
| MSH2 | NM_000251 |
| MSH3 | NM_002439 |
| MSH5 | NM_002441 |
| MSH6 | NM_000179 |
| MTHFR | NM_005957 |
| MTRR | NM_002454 |
| MUTYH | NM_012222 |
| NBN | NM_002485 |
| NF1 | NM_000267 |
| NRAS | NM_002524 |
| PALB2 | NM_024675 |
| PIK3CA | NM_006218 |
| PMS1 | NM_000534 |
| PMS2 | NM_000535 |
| POLD1 | NM_002691 |
| POLE | NM_006231 |
| PTCH1 | NM_000264 |
| PTEN | NM_000314 |
| PTPRK | NM_002844 |
| RAD50 | NM_005732 |
| RAD51C | NM_002876 |
| RAD51D | NM_002878 |
| RB1 | NM_000321 |
| RRM1 | NM_001033 |
| RSPO2 | NM_178565 |
| RSPO3 | NM_032784 |
| SLIT1 | NM_003061 |
| SMAD2 | NM_005901 |
| SMAD4 | NM_005359 |
| SOD2 | NM_000636 |
| STK11 | NM_000455 |
| STMN1 | NM_005563 |
| TOP2A | NM_001067 |
| TP53 | NM_000546 |
| TRRAP | NM_003496 |
| TUBB3 | NM_006086 |
| TYMP | NM_001953 |
| TYMS | NM_001071 |
| UGT1A1 | NM_000463 |
| UMPS | NM_000373 |
| XPC | NM_004628 |
| XRCC1 | NM_006297 |
Statistical analysis
A likelihood ratio test was used for comparing the
difference of the number of nonsynonymous mutations between cancer
stages. P<0.05 was considered to indicate a statistically
significant difference. Analyses were performed using the R
(version 3.1.2, https://www.R-project.org) (32).
Results
Patient characteristics
A total of 11 patients with CRC were enrolled,
including 9 patients with colon cancer and 2 patients with rectal
cancer. The cohort included 4 patients with stage I, 3 patients
with stage II, 3 patients with stage III and 1 patient with stage
IV using a pathological staging system (33). CT imaging revealed that 1 patient with
CRC had liver metastasis at surgery. A total of 9 patients received
postoperative chemotherapy following surgery. In total, 11 primary
tumor tissues and 42 plasma samples were collected. Complete
information is presented in Tables
II and III.
 | Table II.Clinical characteristics of 11
patients with CRC. |
Table II.
Clinical characteristics of 11
patients with CRC.
| Patient no. | Sex | Age at surgery
(years) | Histology | TNM | Tumor stage | Size of tumor
tissue (cm) | Maximum diameter
(cm) | Adjuvant
chemotherapy at surgery | Family history of
cancer |
|---|
| H1001 | Male | 69 | Colon | T4bN0M0 | IIC | 4.0×3.0×2.0 | 4.0 | N | N |
| H1002 | Female | 50 | Colon | T2N1aM0 | IIIA | 2.1×1.5×0.6 | 2.1 | Oxaliplatin+5-FU
(20 weeks) | N |
| H1003 | Male | 73 | Rectum | T3N2aM1b | IVB | 5.5×4.0×0.8 | 5.5 | Capecitabine (10
weeks) | Y |
| H1004 | Female | 49 | Rectum | T2N0M0 | I | 2.0×2.0×0.5 | 2.0 | Oxaliplatin+5-FU (2
weeks) | N |
|
|
|
|
|
|
|
|
| Capecitabine (12
weeks) |
|
| H1005 | Female | 60 | Colon | T3N1cM0 | IIIB | 2.2×1.8×1.0 | 2.2 | Oxaliplatin (16
weeks) | NA |
| H1006 | Female | 69 | Colon | T1N0M0 | I | 1.5×1.5×1.0 | 1.5 | N | Y |
| H1007 | Male | 53 | Colon | T2N0M0 | I | 4.3×2.5×1.2 | 4.3 | Oxaliplatin+5-FU (4
weeks) | N |
|
|
|
|
|
|
|
|
| Capecitabine (18
weeks) |
|
| H1008 | Female | 61 | Colon | T1N0M0 | I | 7.5×4.0×3.5 | 7.5 | Capecitabine (18
weeks) | N |
| H1009 | Male | 67 | Colon | T3N0M0 | IIA | 11.0×4.0×1.2 | 11 | Capecitabine (18
weeks) | N |
| H010 | Female | 35 | Colon | T3N1aM0 | IIIB | 3.0×2.5×0.7 | 3 | Oxaliplatin+5-FU
(20 weeks) | N |
| H1011 | Female | 48 | Colon | T3N0M0 | IIA | 2.5×4.5×1.5 | 4.5 | Capecitabine (24
weeks) | N |
 | Table III.Patient and tumor
characteristics. |
Table III.
Patient and tumor
characteristics.
|
Characteristics | Patients
(n=11) |
|---|
| Age at surgery |
|
| Years,
median (range) | 57.6 (35–73) |
| Sex, n (%) |
|
|
Male | 4 (36.4) |
|
Female | 7 (63.6) |
| Tumor stage, n
(%) |
|
| I | 4 (36.3) |
| II | 3 (27.3) |
|
III | 3 (27.3) |
| IV | 1 (9.1) |
| Localization, n
(%) |
|
|
Colon | 9 (81.8) |
|
Rectum | 2 (18.2) |
| Adjuvant
chemotherapy, n (%) |
|
|
Yes | 9 (81.8) |
| No | 2 (18.2) |
| Family history, n
(%) |
|
|
Yes | 2 (18.2) |
| No | 8 (72.7) |
| NA | 1 (9.1) |
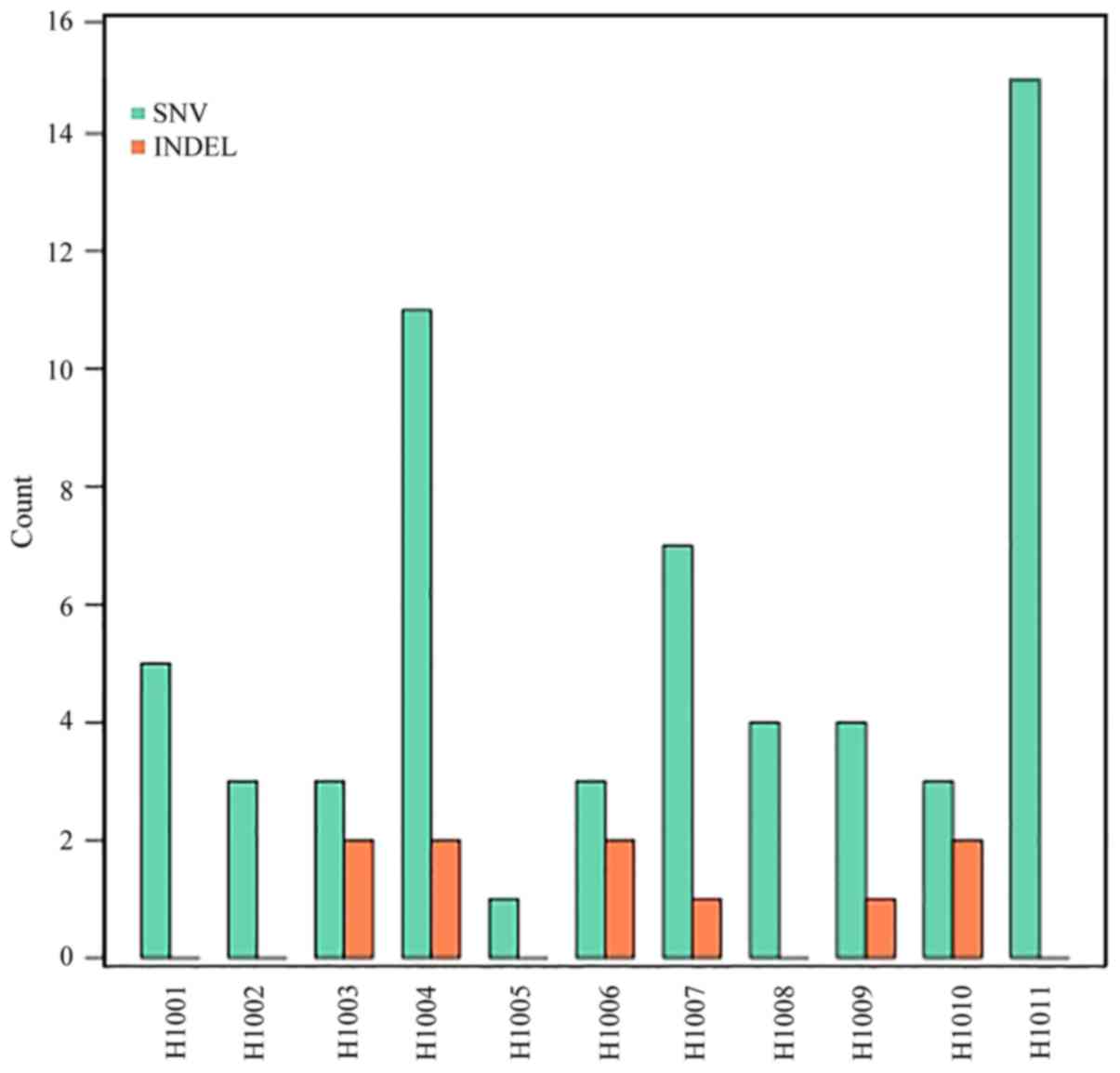
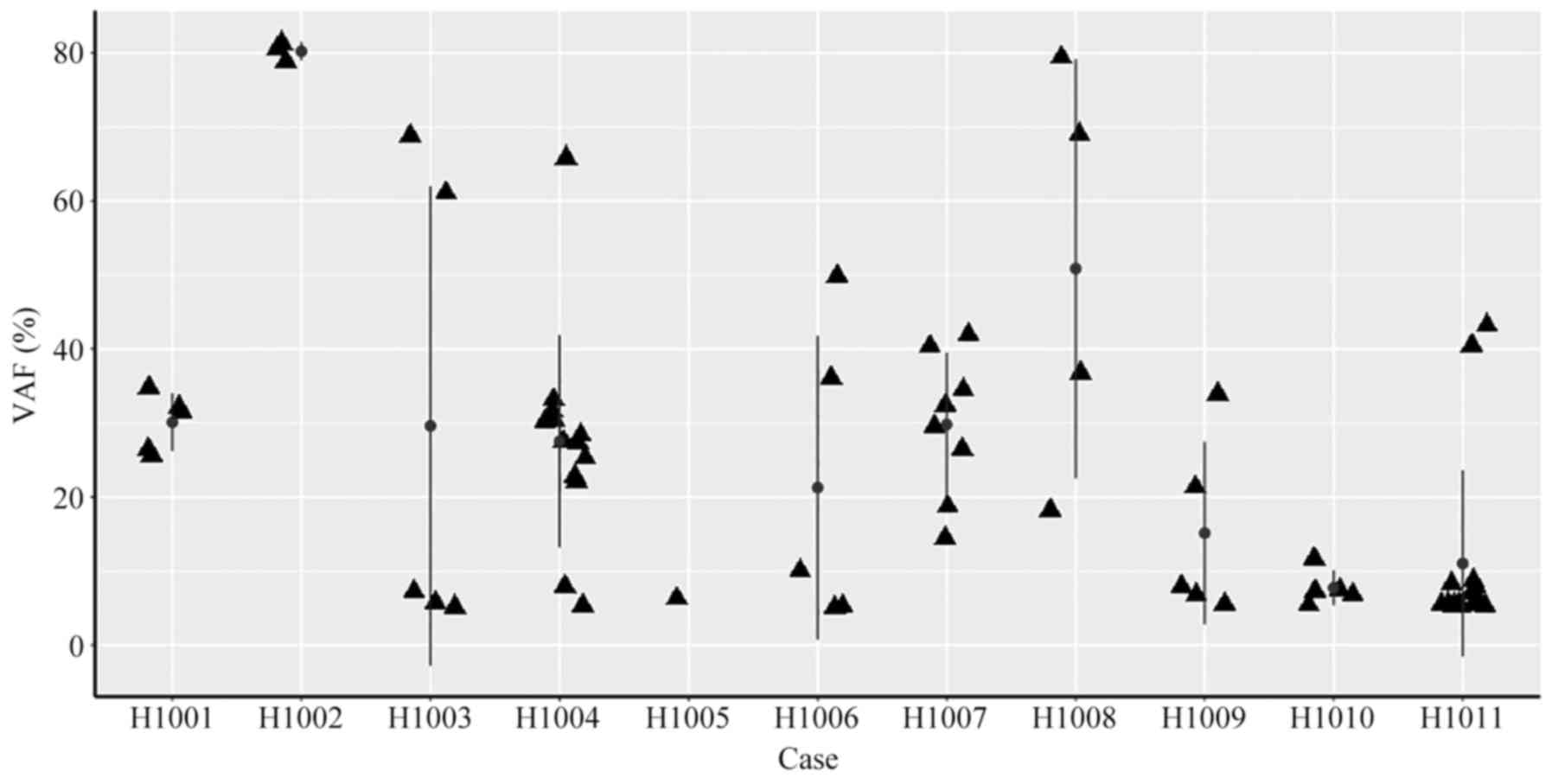
Somatic mutations in tumor tissue samples. Varscan2
(somatic P-value=0.01, minimum variant frequency=5%, strand
filter=true, and otherwise default parameters) was used to detect
tumor somatic single nucleotide variants (SNVs) and short
insertion/deletion events captured by a panel of 85 genes (0.03% of
the human genome; Table I). Within
this small target, the number of nonsynonymous mutations detected
in tumors ranged from 1 to 15, with a median of 5, including SNVs
(59–86%) and small deletions (10–14%; Fig. 2; Table
IV). The number of nonsynonymous mutations was not associated
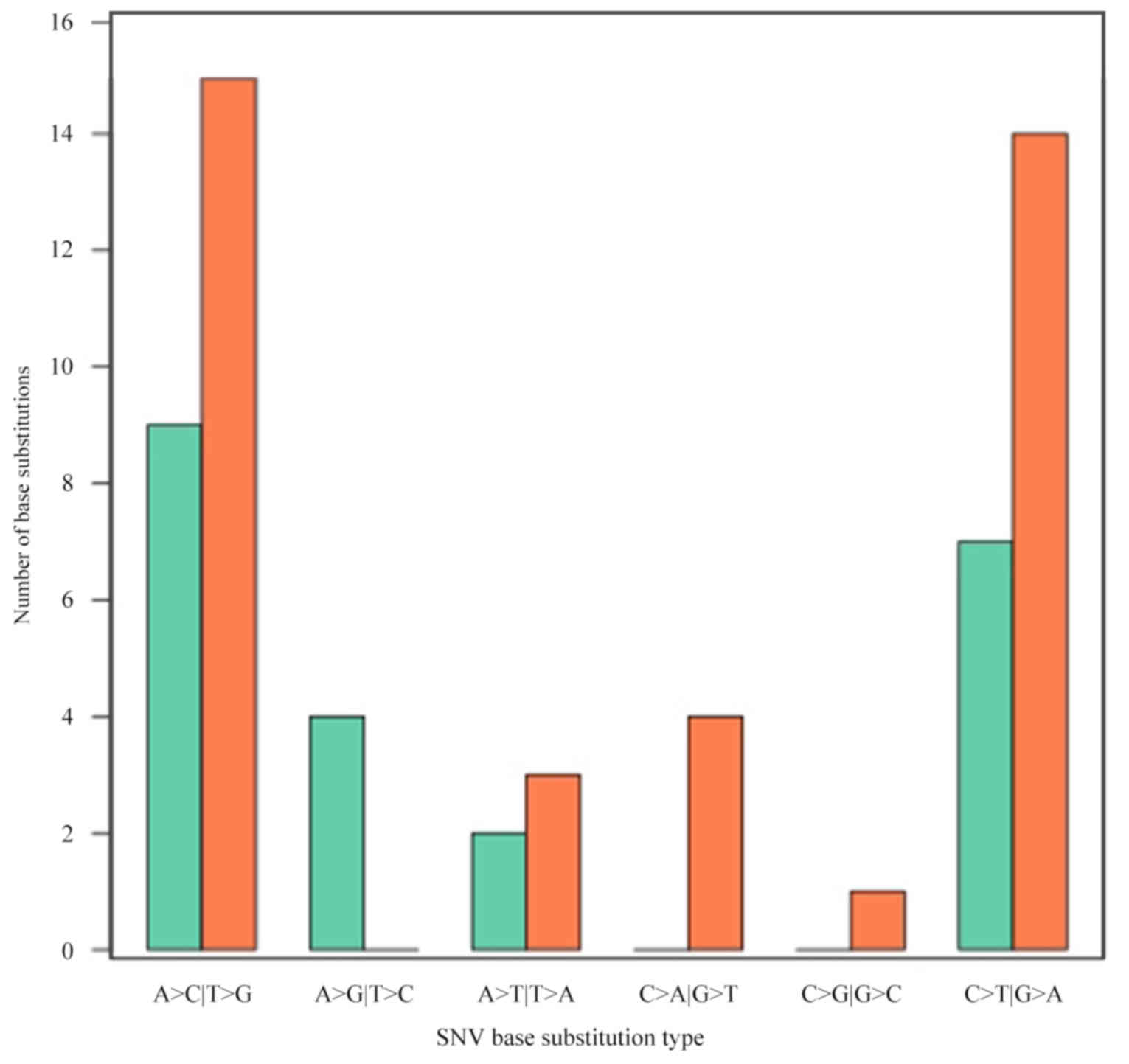
with the CRC stage (P=0.451). Additionally, it was identified that
A>C|T>G transversion occurred most frequently (24 and 41%,
respectively), followed by C>T|G>A transition (Fig. 3).
 | Table IV.Dynamic detection of tumor tissue DNA
and serial plasma circulating tumor DNA driver mutations in 11
patients with colorectal cancer. |
Table IV.
Dynamic detection of tumor tissue DNA
and serial plasma circulating tumor DNA driver mutations in 11
patients with colorectal cancer.
| Case | Gene | Chromosome | Transcript | Exon | Nucleotide
variation | Amino acid
variation | Mutation frequency
of tumor tissue (%) | Cosmid | Mutation frequency
of preoperative plasma (%) | ALT-Depth | Depth | Mutation frequency
of 1 month postoperative plasma (%) | ALT-Depth | Depth | Mutation frequency
of 3 month postoperative plasma(%) | ALT-Depth | Depth | Mutation frequency
of 5 mo postoperative plasma(%) | ALT-Depth | Depth |
|---|
| H1001 | ERBB2 | chr17 | NM_004448 | exon21 | c.G2524A | p.V842I | 34.81 | COSM14065 | 1.26 | 35 | 2,787 | 0 | 0 | 0 | – | – | – | 0 | 0 | 0 |
| H1001 | APC | chr5 | NM_000038 | exon6 | c.G568T | p.E190X | 32.16 | COSM258874 | 0.87 | 23 | 2,653 | 0 | 0 | 0 | – | – | – | 0 | 0 | 0 |
| H1001 | TP53 | chr17 | NM_000546 | exon5 | c.G524A | p.R175H | 31.4 | COSM10648 | 1.02 | 16 | 1,575 | 0.46 | 5 | 1,081 | – | – | – | 0 | 0 | 0 |
| H1001 | ERBB4 | chr2 | NM_005235 | exon28 | c.T3492A | p.N1164K | 26.48 |
| 1.03 | 33 | 3,215 | 0 | 0 | 0 | – | – | – | 0 | 0 | 0 |
| H1001 | KRAS | chr12 | NM_004985 | exon2 | c.G35A | p.G12D | 25.66 | COSM521 | 0.44 | 10 | 2,254 | 0 | 0 | 0 | – | – | – | 0 | 0 | 0 |
| H1002 | TP53 | chr17 | NM_000546 | exon8 | c.G818A | p.R273H | 81.35 | COSM10660 | 0 | 0 | 0 | 0 | 0 | 0 | 0.13 | 3 | 2,352 | 0 | 0 | 0 |
| H1002 | APC | chr5 | NM_000038 | exon16 | c.G4222T | p.E1408X | 80.49 | COSM18822 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1002 | APC | chr5 | NM_000038 | exon16 | c.T2735A | p.L912X | 78.89 | COSM4167198 | 0 | 0 | 0 | 0.26 | 2 | 768 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1003 | TP53 | chr17 | NM_000546 | exon8 | c.A857G | p.E286G | 61.08 | COSM43565 | 8.95 | 233 | 2,602 | 11.32 | 124 | 1,100 | 42.39 | 993 | 2,342 | 71.4 | 1,990 | 2,787 |
| H1003 | MLH3 | chr14 | NM_014381 | exon2 | c.G1163A | p.S388N | 7.23 |
| 0.11 | 3 | 2,790 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1003 | KMT2D | chr12 | NM_003482 | exon31 | c.G7489A | p.A2497T | 5.17 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1003 | PIK3CA | chr3 | NM_006218 | exon9 | c.G1624A | p.E542K | 0 | COSM760 | 0 | 0 | 0 | 0 | 0 | 0 | 12.66 | 195 | 1,542 | 19.96 | 505 | 2,532 |
| H1003 | APC | chr5 | NM_000038 | exon6 | c.617delC | p.T206fs | 68.83 |
| 8.58 | 159 | 1,854 | 15.69 | 96 | 612 | 0 | 0 | 0 | 76.53 | 2,045 | 2,672 |
| H1003 | PTEN | chr10 | NM_001304717 | exon1 | c.19_21del | p.7_7del | 5.76 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1004 | CTNNB1 | chr3 | NM_001904 | exon3 | c.A121G | p.T41A | 65.85 | COSM5664 | 0.27 | 9 | 3,292 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1004 | BRCA2 | chr13 | NM_000059 | exon10 | c.A832G | p.S278G | 33.18 |
| 0.22 | 6 | 2,735 | 0.53 | 6 | 1,125 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1004 | PIK3CA | chr3 | NM_006218 | exon2 | c.A331G | p.K111E | 31.66 | COSM13570 | 0.81 | 25 | 3,076 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1004 | KRAS | chr12 | NM_004985 | exon2 | c.G38A | p.G13D | 30.32 | COSM532 | 1 | 19 | 1,897 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1004 | BRCA2 | chr13 | NM_000059 | exon13 | c.C7006T | p.R2336C | 30.17 | COSM432312 | 0.56 | 15 | 2,702 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1004 | FZR1 | chr19 | NM_016263 | exon7 | c.C542T | p.A181V | 28.37 |
| 0.33 | 9 | 2,733 | 0.21 | 2 | 937 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1004 | SLIT1 | chr10 | NM_003061 | exon11 | c.G1054A | p.A352T | 27.5 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0.27 | 2 | 740 | 0 | 0 | 0 |
| H1004 | CYP19A1 | chr15 | NM_000103 | exon10 | c.C1285T | p.P429S | 27.34 |
| 0.41 | 10 | 2,458 | 0.34 | 4 | 1,178 | 0.52 | 6 | 1,163 | 0 | 0 | 0 |
| H1004 | KMT2D | chr12 | NM_003482 | exon11 | c.C3868T | p.R1290W | 22.82 | COSM4042398 | 0.59 | 12 | 2,031 | 0 | 0 | 0 | 0.22 | 2 | 913 | 0 | 0 | 0 |
| H1004 | KMT2B | chr19 | NM_014727 | exon28 | c.T6737G | p.V2246G | 7.95 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1004 | KMT2B | chr19 | NM_014727 | exon28 | c.T6746G | p.V2249G | 5.39 |
| 0.51 | 3 | 587 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1004 | ARID1A | chr1 | NM_006015 | exon1 | c.433delC | p.P145fs | 25.28 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1004 | AKAP9 | chr7 | NM_005751 | exon19 | c.5010_5013del | p.G1670fs | 22.1 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1005 | EPCAM | chr2 | NM_002354 | exon1 | c.T25G | p.F9V | 6.32 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | – | – | – |
| H1006 | RB1 | chr13 | NM_000321 | exon1 | c.A13C | p.T5P | 10.03 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1006 | KMT2B | chr19 | NM_014727 | exon28 | c.T6746G | p.V2249G | 5.22 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1006 | ARID1A | chr1 | NM_006015 | exon1 | c.A653C | p.N218T | 5.07 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1006 | APC | chr5 | NM_000038 | exon16 | c.4217_4218del | p.Q1406fs | 49.86 | COSM5007948 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1006 | TP53 | chr17 | NM_000546 | exon7 | c.749_756del | p.P250fs | 36.1 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1007 | SMAD4 | chr18 | NM_005359 | exon9 | c.G988C | p.E330Q | 41.96 | COSM14240 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1007 | TP53 | chr17 | NM_000546 | exon5 | c.G473A | p.R158H | 40.38 | COSM10690 | 0.21 | 2 | 972 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1007 | PIK3CA | chr3 | NM_006218 | exon5 | c.A1034T | p.N345I | 34.47 | COSM94978 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.48 | 5 | 1,058 |
| H1007 | FBXW7 | chr4 | NM_018315 | exon8 | c.C1018T | p.H340Y | 32.41 | COSM28798 | 0.28 | 3 | 1,086 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1007 | KRAS | chr12 | NM_004985 | exon2 | c.G35T | p.G12V | 26.5 | COSM520 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1007 | CDH1 | chr16 | NM_004360 | exon9 | c.G1259A | p.G420D | 18.71 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1007 | ATM | chr11 | NM_000051 | exon17 | c.T2570G | p.L857R | 14.45 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1007 | SMAD2 | chr18 | NM_005901 | exon9 | c.1009_1024del | p.R337fs | 29.56 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1008 | KRAS | chr12 | NM_004985 | exon3 | c.A183T | p.Q61H | 79.42 | COSM555 | 3.26 | 52 | 1,593 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1008 | SMAD4 | chr18 | NM_005359 | exon12 | c.T1619G | p.L540R | 68.95 | COSM14165 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1008 | UGT1A1 | chr2 | NM_000463 | exon1 | c.G323A | p.R108H | 36.8 | COSM1018132 | 0.46 | 7 | 1,508 | 0.2 | 4 | 1,992 | 0.17 | 3 | 1,756 | 0 | 0 | 0 |
| H1008 | ATIC | chr2 | NM_004044 | exon6 | c.G515A | p.R172H | 18.27 | COSM1183927 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1009 | KRAS | chr12 | NM_004985 | exon2 | c.G35A | p.G12D | 33.98 | COSM521 | 0.54 | 3 | 553 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1009 | RB1 | chr13 | NM_000321 | exon1 | c.A13C | p.T5P | 7.94 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1009 | ARID1A | chr1 | NM_006015 | exon1 | c.A317C | p.N106T | 6.77 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1009 | MSH6 | chr2 | NM_000179 | exon1 | c.A256C | p.T86P | 5.56 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1009 | ARID1A | chr1 | NM_006015 | exon20 | c.5125_5131del | p.L1709fs | 21.35 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1010 | KMT2B | chr19 | NM_014727 | exon1 | c.T266G | p.V89G | 7.46 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1010 | KMT2B | chr19 | NM_014727 | exon28 | c.T6746G | p.V2249G | 6.78 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1010 | KMT2D | chr12 | NM_003482 | exon11 | c.T2987G | p.V996G | 5.41 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1010 | TP53 | chr17 | NM_000546 | exon7 | c.715_720del | p.239_240del | 11.72 | COSM45055 | 0 | 0 | 0 | 0 | 0 | 0 | 0.19 | 3 | 1,624 | 0 | 0 | 0 |
| H1010 | APC | chr5 | NM_000038 | exon16 | c.2025delA | p.T675fs | 7.35 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | TP53 | chr17 | NM_000546 | exon8 | c.C844T | p.R282W | 43.17 | COSM10704 | 0.39 | 6 | 1,533 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | PIK3CA | chr3 | NM_006218 | exon5 | c.T1035A | p.N345K | 40.51 | COSM754 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | RB1 | chr13 | NM_000321 | exon1 | c.A13C | p.T5P | 8.73 |
| 9.42 | 13 | 138 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | TP53 | chr17 | NM_000546 | exon5 | c.G404A | p.C135Y | 8.28 | COSM10801 | 0.19 | 3 | 1,548 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | KMT2B | chr19 | NM_014727 | exon1 | c.T248G | p.L83R | 7.96 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | KMT2B | chr19 | NM_014727 | exon28 | c.T6746G | p.V2249G | 7.06 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | ERBB2 | chr17 | NM_004448 | exon1 | c.T25G | p.W9G | 6.37 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | KMT2B | chr19 | NM_014727 | exon28 | c.T6737G | p.V2246G | 5.67 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | CREBBP | chr16 | NM_004380 | exon30 | c.A4931C | p.N1644T | 5.61 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | APC | chr5 | NM_000038 | exon16 | c.G4057T | p.E1353X | 5.57 | COSM19048 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | PTCH1 | chr9 | NM_000264 | exon1 | c.A26C | p.E9A | 5.41 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | PTPRK | chr6 | NM_002844 | exon15 | c.C2432T | p.T811I | 5.39 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | TYMP | chr22 | NM_001953 | exon8 | c.T941G | p.L314R | 5.31 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | RB1 | chr13 | NM_000321 | exon1 | c.A24C | p.K8N | 5.29 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| H1011 | TYMP | chr22 | NM_001953 | exon9 | c.T1175G | p.V392G | 5.22 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
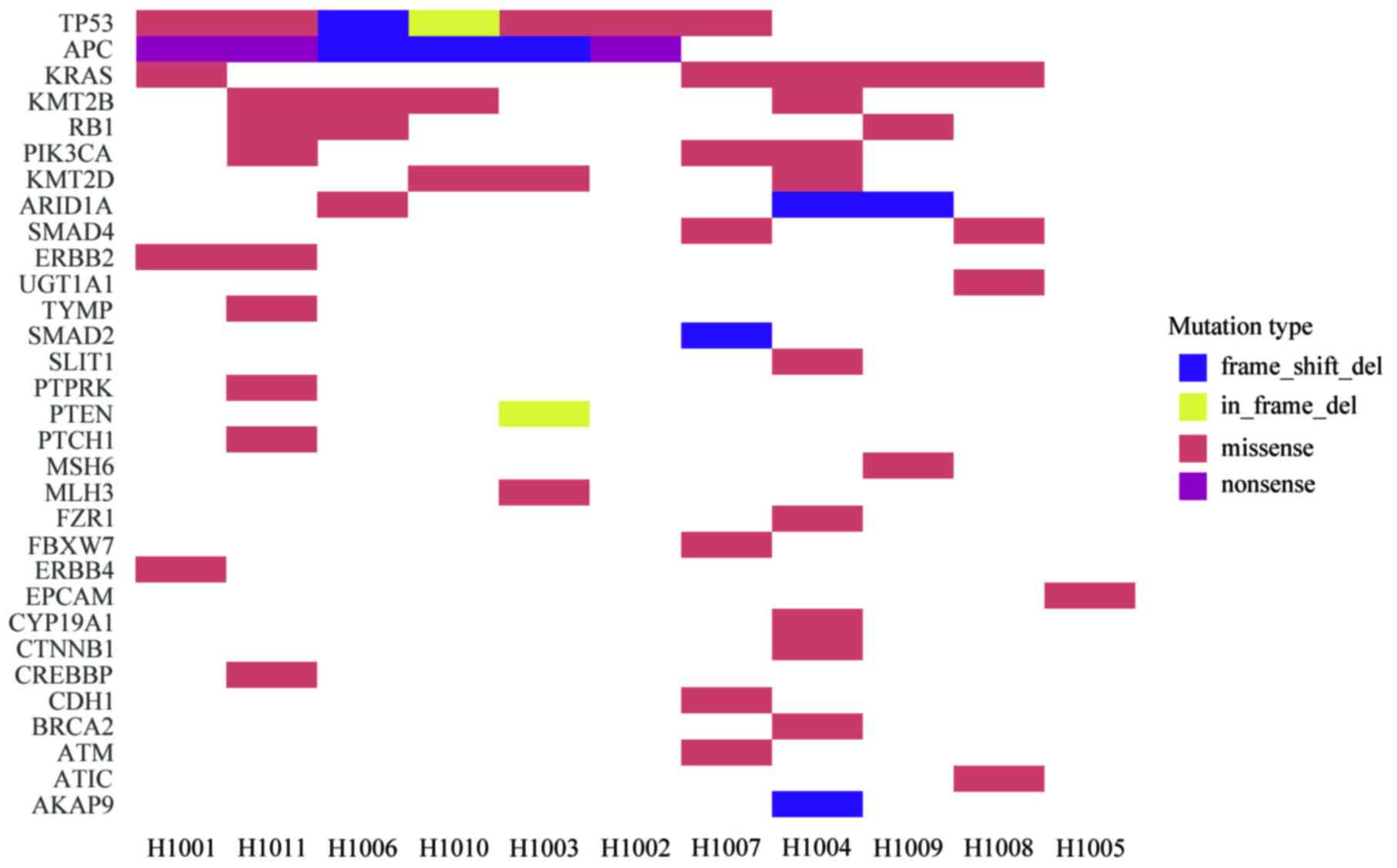
Different driver gene mutations revealed
inter-individual tumor genetic heterogeneity. For example, KRAS
mutations in codons 12 and 13 were observed in 3 patients and 1
patient, respectively. Tumor protein p53 (TP53) mutations in exons
5, 7 and 8 were observed in 3, 2 and 2 patients, respectively. A
heatmap was constructed in order to demonstrate the somatic
mutations occurring in the tumor tissues (Fig. 4). Different mutation loci and their
corresponding mutation rates indicated intratumor genetic
heterogeneity, implying tumor evolution (Fig. 5).
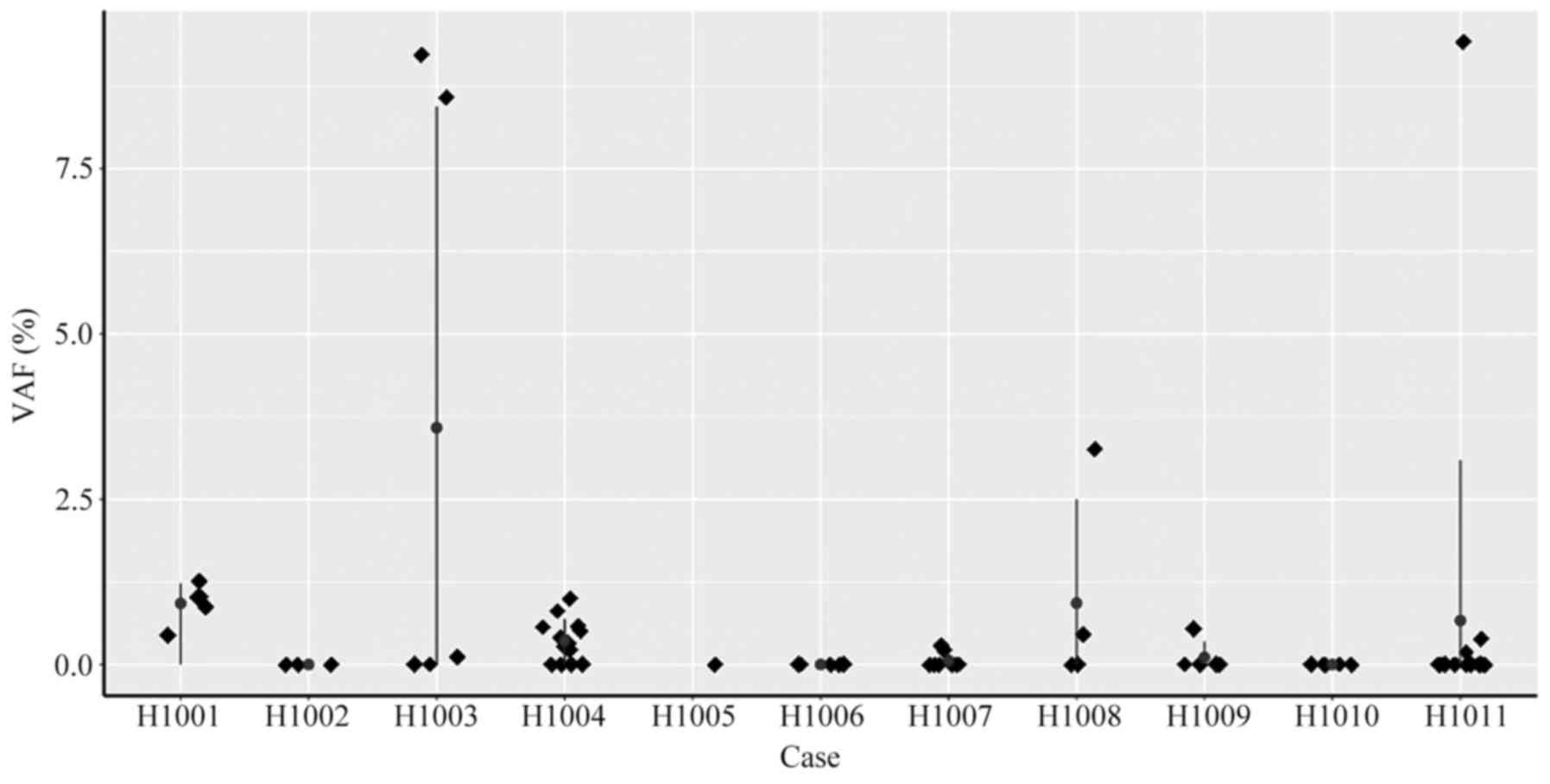
Somatic mutation detection in
preoperative plasma
Next, ctDNA levels in preoperative plasma were
examined. At a mean sequencing depth of x~7,000 (pre-duplication),
a custom Python script was used to identify whether somatic tumor
mutations were present in preoperative plasma. For a total of 7
patients, at least one mutation in tumor tissue was detected in
preoperative plasma, and VAF in plasma ranged from 0.11 to 9.42%
(Fig. 6). For the other 4 patients
(H1002, H1005, H1006 and H1010), who were diagnosed with stage II
or stage III cancer, no mutations that matched the tumor mutations
were detected in the plasma.
Dynamic monitoring of postoperative
patients with CRC using ctDNA
In order to determine prognosis for patients with
CRC and decide whether or not to administer adjuvant chemotherapy,
ctDNA levels were monitored periodically for six months. Driver
gene mutations were detected at low mutation rates in preoperative
plasma ctDNA from 7 patients (7/11; 63.6%), whilst no driver
mutations were detected in the remaining 4 patients (4/11; 36.4%;
patient nos. ID H1002, H1005, H1006 and H1010; see Table IV).
Following surgery, the number of mutations and the
corresponding mutation rate decreased compared with preoperative
plasma in postoperative plasma in the majority of patients. Driver
mutations were not detected in 6 patients (6/11; 54.5%). To
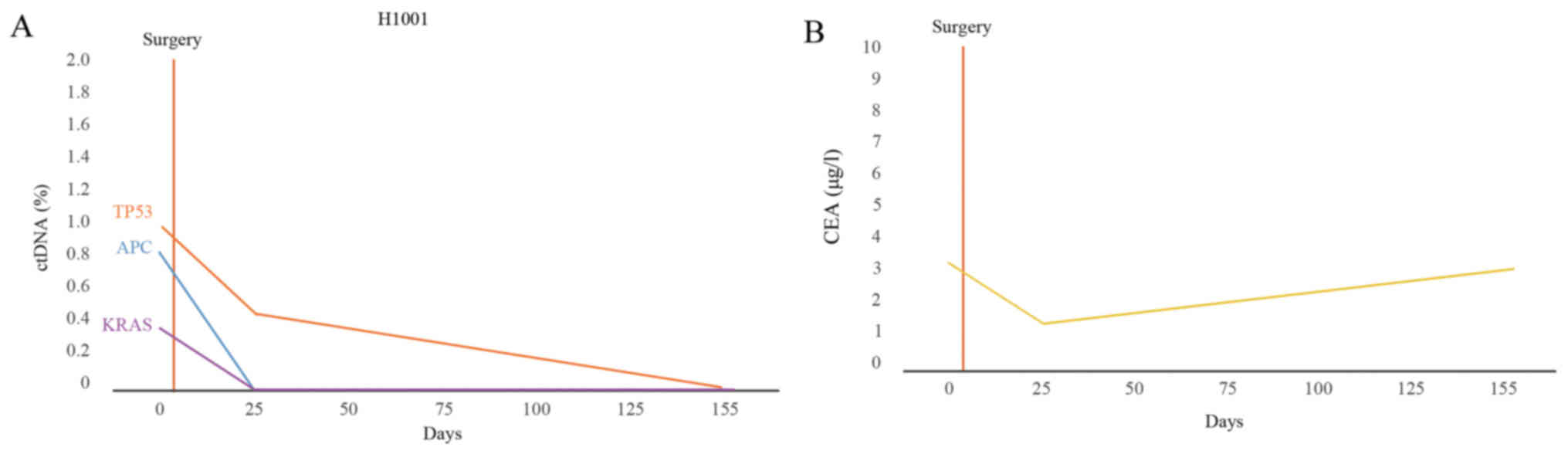
illustrate this, the changes in the percentage of ctDNA and CEA
levels in patient H1001 were plotted (Fig. 7A and B). In patient H1001, the CEA
levels initially declined and then increased slowly, but the levels
remained in the normal range (<5 µg/l; Fig. 7B).
The VAF values for patient H1003 are presented in
Fig. 8A. For patient H1003, the
mutation rate of NM_0,011,26113.2 (TP53):c.857A>G (p.Glu286Gly)
in ctDNA increased from 8.95 (preoperation) to 11.32%
(post-operation) (Fig. 8A). Liver
metastases were detected in patient H1003 at the time of surgery
(Fig. 8B). Following surgery, patient
H1003 received one round of chemotherapy and two rounds of
microwave ablation for liver metastases, but treatments were
subsequently terminated. The periodic monitoring of plasma ctDNA in
patient H1003 revealed an increase in the mutation rate of TP53,
and the development of a new mutation,
phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit α
(PIK3CA). NM_006218.3 (PIK3CA):c.1624G>A (p.Glu542Lys) (Fig. 8A). In the final ctDNA sample from this
patient, the mutation rate of TP53 and PIK3CA reached 71.4 and
19.96%, respectively (Fig. 8A).
Patient H1003 succumbed to mortality six months following surgery.
Furthermore, CEA levels were lower compared to the threshold
(cut-off, 5 µg/l) in the three tests prior to mortality (2.63, 2.02
and 3.45 µg/l), which was not revealed to be associated with the
patient's condition. However, the CEA level increased at the time
of last sampling (11.12 µg/l; Fig.
8B). This finding suggests that ctDNA is more sensitive
compared with CEA as a biomarker for predication of disease
progression.
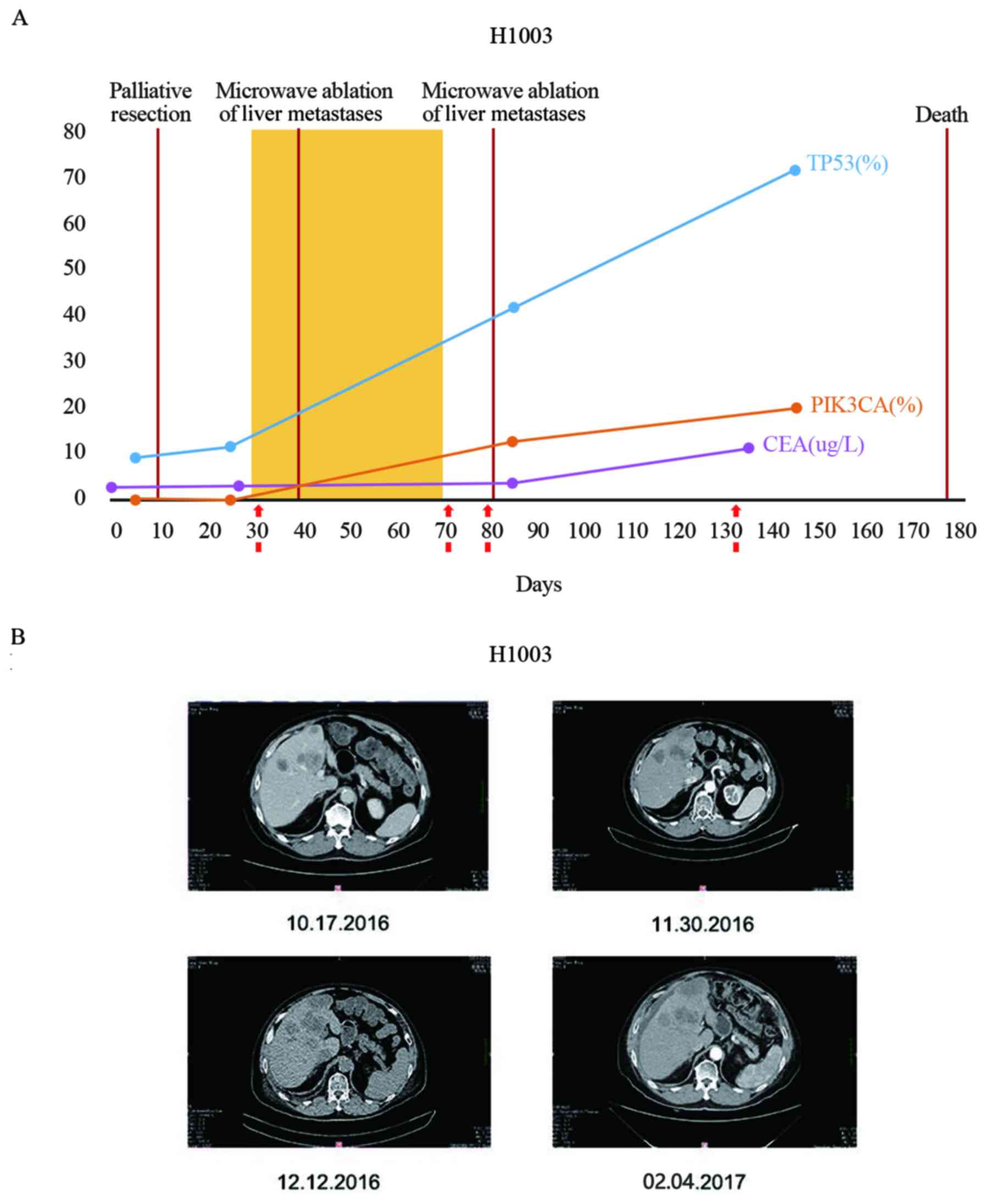 | Figure 8.Dynamic ctDNA monitoring of patient
H1003. (A) ctDNA and CEA levels were assessed pre-operatively and
postoperatively for patient H1003. H1003 presented with recurrence
during colonoscopy and in contrast-enhanced CT. For patient H1003,
the TP53 and PIK3CA mutation rate changes in ctDNA are presented at
the indicated times, and compared with CEA. The number of days
following surgery is indicated on the x-axis is indicated on the
x-axis. The VAF values are indicated on the y-axis. (B) CT images
of liver metastases in patient H1003, in whom the level of ctDNA
was four times compared with other patients in the study without
metastases. Images were captured on October 17th, 2016 (30 days
after surgery), November 30th, 2016 (43 days after surgery),
December 12th, (55 days after surgery) and February 4th, 2017 (98
days after surgery). CT, computed tomography; ctDNA, circulating
tumor DNA; CEA, carcinoembryonic antigen; PIK3CA,
phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit
alpha; TP53, tumor protein P53; VAF, variant allele fraction. |
Discussion
In the present study, somatic mutations in tumor
tissues and preoperative plasma were highly associated as
represented by the status of KRAS gene. In previous studies, ctDNA
detection rates for patients in the early stages of cancer were low
(16,27,34).
Detection was improved in the present study by increasing the depth
of ctDNA sequencing (~7,000X), which allowed for the monitoring of
ctDNA dynamically to follow disease progression. ctDNA analysis
using NGS was demonstrated to be sensitive for monitoring early
therapeutic responses of CRC and metastatic breast cancer (35). Driver mutations in genes such as TP53,
KRAS and adenomatosis polyposis coli tumor suppressor as observed
in the 11 patients in the present study, were previously reported
in patients with CRC (36). These
mutations, thus, appear to be useful biomarkers for postoperative
monitoring of CRC. As the sample size is relatively small,
statistical bias is difficult to avoid.
For patients with early-stage CRC, administration of
adjuvant chemotherapy following surgical resection remains
controversial. Highly sensitive and specific biomarkers to predict
the response to surgery are urgently required. Biomarkers for the
early detection of recurrences would also be clinically valuable.
In addition to previous research (16), the present study indicates that plasma
ctDNA analysis has the potential for the detection of the early
stages of cancer. Tie et al (37) demonstrated that ctDNA detection
subsequent to stage II (33) colon
cancer resection provided evidence of residual disease and
identified patients at a substantially high risk of recurrence.
Similar results were observed in the present study. However, the
previous study by Tie et al (37) made predictions by targeting 15 genes
and one single mutation in the ctDNA was selected for further
analysis. By contrast, the present study examined preoperative and
postoperative ctDNA for a whole spectrum of mutations. By
comparison, the present study not only predicted the risk of
recurrence, but was indicative of the tumor evolution for patients
with cancer recurrence, which increases the current understanding
of mechanism underlying recurrence in cancer.
As tumor-specific DNA is only a small fraction of
cell-free DNA, ctDNA mutations are difficult to detect in patients
with early stage cancer (37). For
the patients in the present study with no preoperative driver gene
mutations, we hypothesized that tumor cells may have invaded the
serous layer of the intestinal wall. During the six months of
monitoring, no changes were observed in the blood of patients with
early-stage cancer. These findings indicate a good surgical outcome
consistent with a greater survival of patients with early-stage CRC
following surgery (37,38).
With the rapid progression of patients with
late-stage cancer, driver mutations may quickly and substantially
change in the blood. A high frequency of TP53 somatic mutation was
detected in preoperative blood and tumor tissue of patient H1003.
One month after surgery, the rate of TP53 mutations increased,
which was associated with the presence of liver metastases observed
on CT imaging subsequent to surgery. Conversely, decreased VAF of
TP53 mutation were observed in all the other patients that did not
have metastases following surgery. These results suggest that ctDNA
ought to be analyzed in plasma at least every six months following
surgery in order to monitor for disease progression in patients
with early-stage cancer. Plasma CEA levels of patients with early
or even advanced CRC were often below the normal threshold,
implying that ctDNA has a higher sensitivity and specificity for
detecting CRC compared with conventional CEA analysis.
Sequencing error rates are reported to be >0.1%
in data analysis (39). In the
present study, the correct mutations were selected by increasing
the sequencing depth and a VAF threshold of <5% was used to
filter the mutations detected. According to the tissue mutation
results, ctDNA mutations were filtered using a VAF threshold of
<0.1%, which may limit the sensitivity of ctDNA and omit a
number of low-frequency mutations. Although the number of patients
in the present study is small, it demonstrates the potential of
using ctDNA to monitor the medical response and disease progression
following surgery. ctDNA analysis will be applied to a greater
number of clinical applications in the future.
Acknowledgements
The authors would like to thank Mr. Liwei Deng and
Mr. Cheng Jin for enhancing the diagram and charts. The present
study was supported by the grants from the Gastrointestinal
Department of Shenzhen People's Hospital (grant no. 201601016), the
Creative Design Program of Nanshan Shenzhen (grant no.
KC2015JSJS0028A) for data collection, and the Special Funds for
Future Industries of Shenzhen (grant no. JSGG20160229123927512) for
sequencers and reagents. Clinicians of the Shenzhen People's
Hospital were involved in study design, sample collection, and
clinical information collection, while researchers and engineers
from HaploX were involved in the study design and sequencing data
analysis.
References
|
1
|
Chen W, Zheng R, Zuo T, Zeng H, Zhang S
and He J: National cancer incidence and mortality in China, 2012.
Chin J Cancer Res. 28:1–11. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ng SC and Wong SH: Colorectal cancer
screening in Asia. Br Med Bull. 105:29–42. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Garborg K: Colorectal cancer screening.
Surg Clin North Am. 95:979–989. 2015. View Article : Google Scholar : PubMed/NCBI
Leung WK, Lau JY, Suen BY, Wong GL, Chow
DK, Lai LH, To KF, Yim CK, Lee ES, Tsoi KK, et al: Repeat-screening
colonoscopy 5 years after normal baseline-screening colonoscopy in
average-risk Chinese: A prospective study. Am J Gastroenterol.
104:2028–2034. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Soon MS, Kozarek RA, Ayub K, Soon A, Lin
TY and Lin OS: Screening colonoscopy in Chinese and Western
patients: A comparative study. Am J Gastroenterol. 100:2749–2755.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
National Cancer Institute's SEER
database.[EB/OL]. http://seer.cancer.gov/August 26–2016
|
|
7
|
Labianca R, Nordlinger B, Beretta GD,
Mosconi S, Mandalà M, Cervantes A and Arnold D; ESMO Guidelines
Working Group, : Early colon cancer: ESMO Clinical Practice
Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 24
Suppl 6:vi64–vi72. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Twelves C, Wong A, Nowacki MP, Abt M,
Burris H III, Carrato A, Cassidy J, Cervantes A, Fagerberg J,
Georgoulias V, et al: Capecitabine as adjuvant treatment for stage
III colon cancer. N Engl J Med. 352:2696–2704. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Murphy CC, Harlan LC, Lund JL, Lynch CF
and Geiger AM: Patterns of colorectal cancer care in the united
states: 1990–2010. J Natl Cancer Inst. 107:pii: djv1982015.
View Article : Google Scholar
|
|
10
|
Jeffery M, Hickey BE, Hider PN and See AM:
Follow-up strategies for patients treated for non-metastatic
colorectal cancer. Cochrane Database Syst Rev.
11:CD0022002016.PubMed/NCBI
|
|
11
|
Reinert T, Schøler LV, Thomsen R, Tobiasen
H, Vang S, Nordentoft I, Lamy P, Kannerup AS, Mortensen FV,
Stribolt K, et al: Analysis of circulating tumour DNA to monitor
disease burden following colorectal cancer surgery. Gut.
65:625–634. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pita-Fernández S, Alhayek-Aí M,
González-Martín C, López-Calviño B, Seoane-Pillado T and
Pértega-Díaz S: Intensive follow-up strategies improve outcomes in
non-metastatic colorectal cancer patients after curative surgery: A
systematic review and meta-analysis. Ann Oncol. 26:644–656. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kraus S, Nabiochtchikov I, Shapira S and
Arber N: Recent advances in personalized colorectal cancer
research. Cancer Lett. 347:15–21. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu Z, Zhang Y, Niu Y, Li K, Liu X, Chen H
and Gao C: A systematic review and meta-analysis of diagnostic and
prognostic serum biomarkers of colorectal cancer. PLoS One.
9:e1039102014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chao M and Gibbs P: Caution is required
before recommending routine carcinoembryonic antigen and imaging
follow-up for patients with early-stage colon cancer. J Clin Oncol.
27:e279–280. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bettegowda C, Sausen M, Leary RJ, Kinde I,
Wang Y, Agrawal N, Bartlett BR, Wang H, Luber B, Alani RM, et al:
Detection of circulating tumor DNA in early- and late-stage human
malignancies. Sci Transl Med. 6:224ra242014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Butler TM, Johnson-camacho K, Peto M, Wang
NJ, Macey TA, Korkola JE, Koppie TM, Corless CL, Gray JW and
Spellman PT: Exome sequencing of cell-free DNA from metastatic
cancer patients identifies clinically actionable mutations distinct
from primary disease. PLoS One. 10:e01364072015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Husain H and Velculescu VE: Cancer DNA in
the circulation: The liquid biopsy. Jama. 318:1272–1274. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Roschewski M, Dunleavy K, Pittaluga S,
Moorhead M, Pepin F, Kong K, Shovlin M, Jaffe ES, Staudt LM, Lai C,
et al: Circulating tumour DNA and CT monitoring in patients with
untreated diffuse large B-cell lymphoma: A correlative biomarker
study. Lancet Oncol. 16:5412015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xu S, Lou F, Wu Y, Sun DQ, Zhang JB, Chen
W, Ye H, Liu JH, Wei S, Zhao MY, et al: Circulating tumor DNA
identified by targeted sequencing in advanced-stage non-small cell
lung cancer patients. Cancer Lett. 370:324–331. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Diehl F, Schmidt K, Choti MA, Romans K,
Goodman S, Li M, Thornton K, Agrawal N, Sokoll L, Szabo SA, et al:
Circulating mutant DNA to assess tumor dynamics. Nat Med.
14:985–990. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tie J, Kinde I, Wang Y, Wong HL, Roebert
J, Christie M, Tacey M, Wong R, Singh M, Karapetis CS, et al:
Circulating tumor DNA as an early marker of therapeutic response in
patients with metastatic colorectal cancer. Ann Oncol.
26:1715–1722. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Stefanie Mortimer: Early, molecular
detection of cancer utilizing circulating cell-free DNA assay with
ultra high accuracy and sensitivity. ASCO. 2016.
|
|
24
|
Zhou J, Chang L, Guan Y, Yang L, Xia X,
Cui L, Yi X and Lin G: Application of circulating tumor DNA as a
non-invasive tool for monitoring the progression of colorectal
cancer. PLoS One. 11:e01597082016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Perdomo S, Anantharaman D, Mckay J and
Brennan P: Abstract 5230: Circulating tumor DNA as a ‘liquid
biopsy’ in head and neck cancer. Cancer Res. 75:5230. 2015.
View Article : Google Scholar
|
|
26
|
Aravanis AM, Lee M and Klausner RD:
Next-generation sequencing of circulating tumor DNA for early
cancer detection. Cell. 168:571–574. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tie J, Wang Y, Kinde I, Steel M, Elsaleh
H, Singh MS, Singh MS, Turner NH, Tran B, Strausberg R, et al:
Circulating tumor DNA (ctDNA) in nonmetastatic colorectal cancer
(CRC): Potential role as a screening tool. J Clin Oncol.
3:5182017.
|
|
28
|
Morgans A and Allen F: Getting ethics
committee approval for research: A beginners guide. Australasian J
Paramedicine. 3:2015.
|
|
29
|
Li H and Durbin R: Fast and accurate
long-read alignment with Burrows-Wheeler transform. Bioinformatics.
26:589–595. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li H, Handsaker B, Wysoker A, Fennell T,
Ruan J, Homer N, Marth G, Abecasis G and Durbin R: 1000 Genome
Project Data Processing Subgroup: The sequence alignment/map (SAM)
format and SAMtools. Bioinformatics. 25:2078–2079. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Koboldt DC, Zhang Q, Larson DE, Shen D,
McLellan MD, Lin L, Miller C, Mardis ER, Ding L and Wilson RK:
VarScan 2: Somatic mutation and copy number alteration discovery in
cancer by exome sequencing. Genome Res. 22:568–576. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
R: A Language and Environment for
Statistical Computing, R Core Team. R Foundation for Statistical
Computing; Vienna, Austria: https://www.R-project.org
|
|
33
|
Hari DM, Leung AM, Lee JH, Sim MS, Vuong
B, Chiu CG and Bilchik AJ: AJCC Cancer Staging Manual 7th edition
criteria for colon cancer: Do the complex modifications improve
prognostic assessment? J Am Coll Surg. 217:181–190. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sausen M, Phallen J, Adleff V, Jones S,
Leary RJ, Barrett MT, Anagnostou V, Parpart-Li S, Murphy D, Kay Li
Q, et al: Clinical implications of genomic alterations in the
tumour and circulation of pancreatic cancer patients. Nat Commun.
6:76862015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Dawson SJ, Tsui DW, Murtaza M, Biggs H,
Rueda OM, Chin SF, Dunning MJ, Gale D, Forshew T, Mahler-Araujo B,
et al: Analysis of circulating tumor DNA to monitor metastatic
breast cancer. N Engl J Med. 368:1199–1209. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Smith G, Carey FA, Beattie J, Wilkie MJ,
Lightfoot TJ, Coxhead J, Garner RC, Steele RJ and Wolf CR:
Mutations in APC, Kirsten-ras and p53-alternative genetic pathways
to colorectal cancer. Proc Natl Acad Sci USA. 99:pp. 9433–9488.
2002; View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tie J, Wang Y, Tomasetti C, Li L, Springer
S, Kinde I, Silliman N, Tacey M, Wong HL, Christie M, et al:
Circulating tumor DNA analysis detects minimal residual disease and
predicts recurrence in patients with stage II colon cancer. Sci
Transl Med. 8:346ra922016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Meyerhardt JA and Mayer RJ: Systemic
therapy for colorectal cancer. N Engl J Med. 352:476–487. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Schmitt MW, Kennedy SR, Salk JJ, Fox EJ,
Hiatt JB and Loeb LA: Detection of ultra-rare mutations by
next-generation sequencing. Proc Natl Acad Sci USA. 109:pp.
14508–14113. 2012; View Article : Google Scholar : PubMed/NCBI
|