Introduction
Osteosarcoma is the most common bone malignancy
encountered in childhood and adolescence (1). Although conventional chemotherapy and
new agents were employed to overcome osteosarcoma, the clinical
diagnosis and prognosis was always poor (2). Accumulating studies have demonstrated
that miRNAs and their target genes, which perform essential roles
in the formation and progression of osteosarcoma, are a promising
therapeutic strategy (3). However,
the molecular mechanisms of osteosarcoma pathogenesis remain poorly
understood. Therefore, it is critical to identify novel diagnostic
and prognostic biomarkers for improving the clinical outcome of
patients with osteosarcoma.
MicroRNAs (miRNAs), a class of small non-coding RNA
species, regulate or degrade target genes by binding to the 3′
untranslated regions (3′UTR) (4).
miRNAs perform critical roles in the regulation of diverse
biological processes, including embryogenesis, development, cell
maintenance, proliferation and apoptosis. In previous years,
accumulating studies have reported that miRNAs are involved in the
development and progression of various human cancers, including
osteosarcoma (3,5,6). Studies
have revealed that miRNAs are dysregulated and act as prognostic
biomarkers in osteosarcoma (6,7).
Furthermore, identifying the mechanism of miRNAs involved in
osteosarcoma progression may aid the development of strategies for
the future diagnosis, treatment and prognosis of osteosarcoma.
miR-874 has been found to be downregulated and serve
as a potential cancer suppressor in various types of cancer,
including breast cancer (8), gastric
cancer (9), maxillary sinus squamous
cell carcinoma (10), non-small cell
lung cancer (11) and head and neck
squamous cell carcinoma (12). It has
been reported that miR-874 can suppress cell migration and invasion
by targeting its target genes (13).
Previously, miR-874 was reported to be significantly downregulated
in osteosarcoma cell lines and clinical specimens, and the
decreased miR-874 expression was significantly associated with
large tumor size, distant metastasis and advanced clinical stage,
and was an independent predictor of poor survival (14). However, little is known about the
mechanism of miR-874 in osteosarcoma proliferation and
invasion.
In the present study, cyclin-dependent kinase 9
(CDK9) was identified as a direct target of miR-874. CDK9
expression was upregulated in osteosarcoma and inversely correlated
with that of miR-874. In addition, miR-874 suppressed osteosarcoma
proliferation and invasion via the downregulation of CDK9. The
present results demonstrated the mechanism of miR-874 in regulating
the proliferation and invasion of the osteosarcoma cells, and
indicated a potential therapeutic target for the treatment of
osteosarcoma.
Materials and methods
Clinical tissues samples
A total of 30 pairs of human osteosarcoma tissues
and matched adjacent noncancerous bone tissues were obtained from
patients (age, 41–65; 14 female and 16 male) at the Department of
Orthopedics, Yantaishan Hospital (Shandong, China) from January
2013 to December 2014. The present study was approved by the
Research Ethics Committee of Yantaishan Hospital and all patients
provided written informed consent.
Cell culture and transfection
Human osteosarcoma MG-63, U2OS and HOS and human
normal osteoblast NHOst cell lines were obtained from American Type
Culture Collection (Manassas, VA, USA) and cultured in Dulbecco's
modified Eagle's medium (DMEM; Gibco; Thermo Fisher Scientific,
Inc., Waltham, MA, USA) supplemented with 10% (v/v) fetal bovine
serum (HyClone; Thermo Fisher Scientific, Inc.), 100 U/ml
penicillin and 100 mg/ml streptomycin. Cultures were maintained at
37°C in a humidified atmosphere with 5% CO2.
MG-63 cells were seeded on 12-well plates with 40%
confluence prior to transfection and incubated at 37°C in a
humidified atmosphere with 5% CO2 overnight. Cells were
transiently transfected with miR-874 mimics and miR-874 inhibitor
sequences using Lipofectamine® 2000 (Invitrogen; Thermo
Fisher Scientific, Inc.) according to the manufacturer's protocol.
The full-length CDK9 cDNA was amplified by polymerase chain
reaction (PCR) and cloned into the pcDNA3.1 vector to generate the
pcDNA-CDK9 constructs, which were used in the rescue assays. MG-63
cells were co-transfected with miR-874 mimic and the
pcDNA-CDK9.
RNA extraction and reverse
transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted from cells and tissues using
TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. The first strand of cDNA
was synthesized by RT (Takara Biotechnology Co., Ltd., Dalian,
China), and the expression of miRNA was detected by RT-qPCR
analysis using the SYBR-Green detection system (Roche Applied
Science, Penzberg, Germany). U6 and GAPDH mRNA were used as the
internal controls. All tests were run in triplicate. The levels of
mRNA expression were normalized to the level of the β-actin mRNA
expression using the 2−ΔΔCq cycle threshold method
(15). CDK9 primers (forward,
5′-ATGGCAAAGCAGTACGACTCG3′ and reverse, 5′-GCAAGGCTGTAATGGGGAAC-3′;
GAPDH primers (forward, 5′-GGAGCGAGATCCCTCCAAAAT-3′ and reverse,
5′-GGCTGTTGTCATACTTCTCATGG-3′, miR-874 primers (forward,
5′-TGCGGCTGCCCTGGCCCGAGGGAC-3′ and reverse,
5′-CCAGTGCAGGGTCCGAGGT-3′) and U6 primers (forward,
5′-TGCGGGTGCTCGCTTCGGCAGC-3′ and reverse,
5′-CCAGTGCAGGGTCCGAGGT-3′) were purchased from Shanghai Genepharma
Co., Ltd. (Shanghai, China).
Luciferase reporter assay
The 3′UTR of the human CDK9 with the predicted
miR-874 binding site was amplified from a cDNA library of MG-63
cells and cloned to a pGL3-control vector. The binding site mutant
of CDK9/pGL3 was also generated using a Muta-direct™
site-directed mutagenesis kit (SBS Genetech Co., Ltd., Beijing,
China). The cells were seeded on 24-well plates. MG-63 cells were
co-transfected with the CDK9/pGL3 vectors and miR-874 or the miRNA
control. Luciferase activity values were determined using the
Dual-Luciferase reporter assay system (Promega Corporation,
Madison, WI, USA).
Cell proliferation assay
Cell proliferation was measured using the MTT assay
using a Cell Proliferation kit I (Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany). In brief, the cells were plated on 96-well
plates at a density of 5×103 cells per well following
transfection. A MTT assay was conducted. Dimethyl sulfoxide, an
acidified ethanol solution, was added to dissolve the insoluble
purple formazan product into a colored solution. Finally, the
optical density was determined at 570 nm using the ELISA plate
reader (Model 550; Bio-Rad Laboratories, Inc., Hercules, CA, USA).
The MG-63 cells were used as the control group and compared with
transfected MG-63 cells.
Cell invasion assay
Invasion assays were performed in triplicate using
Transwell invasion chambers (Costar 3422; Corning Incorporated,
Corning, NY, USA) coated with Matrigel (50 µl per filter; BD
Biosciences, Franklin Lakes, NJ, USA), according to the
manufacturer's protocol. The MG-63 cells were transferred to the
top of the Matrigel-coated invasion chambers in 1% fetal calf serum
DMEM/F12 (2×104 cells/well). To the wells of the lower
chamber, 1.5 ml of DMEM supplemented with 10% FBS (Gibco; Thermo
Fisher Scientific, Inc.) was added as an attractant. Following an
overnight incubation at 37°C in an atmosphere containing 5%
CO2, the cells in the lower side of the insert membrane
were fixed with 5% glutaraldehyde 37°C for 10 min, followed by
staining with 1% crystal violet in 2% ethanol 37°C for an
additional 20 min. Living cells (with diameters ranging from 7–14
µm) that passed through the membrane were collected from the lower
well and counted using a cell Coulter Counter Channelizer 256
(Beckman Coulter, Inc., Brea, CA, USA). The values for invasion
were obtained by counting 8 fields per membrane and represented the
average of three independent experiments.
Western blot analysis
Total protein was extracted from H1299 and A549
cells with a radioimmunoprecipitation assay buffer with 0.5% SDS
and 3% proteinase inhibitor cocktail (Sigma-Aldrich; Merck KGaA)
for 30 min on ice. The concentration of protein was determined
using the bicinchoninic acid protein assay kit (Santa Cruz
Biotechnology, Inc., Dallas, TX, USA). For western blotting,
proteins were separated by 10% SDS-PAGE and transferred to
polyvinylidene fluoride membranes (Invitrogen; Thermo Fisher
Scientific, Inc.). Subsequent to blocking with 5% bovine serum
albumin (Gibco; Thermo Fisher Scientific, Inc.) at room
temperature for 1 h, membranes were incubated with primary antibody
against CDK9 (cat no. ab76320; dilution, 1:200; Abcam, Shanghai,
China) at 4°C overnight. Membranes were then incubated with
secondary antibody goat anti-rabbit IgG (cat no. sc-2007; dilution,
1:2,500; Santa Cruz Biotechnology, Inc.) for 2 h at room
temperature. The signals were detected using a
RapidStep™ ECL Reagent (EMD Millipore, Billerica, MA,
USA).
Statistical analysis
All experiments were repeated at least three times
with similar results. Representative data are shown. The Student's
t-test was used for comparisons between two groups. One-way
analysis of variance was used for comparisons between multiple
groups followed by Turkey multiple comparison post-hoc analysis.
Pearson's correlation coefficient was used to evaluate the
relationships among the relative expression levels of miR-874 and
CDK9 in osteosarcoma tissues. P<0.05 was considered to indicate
a statistically significant difference.
Results
CDK9 is a direct target of miR-874 in
osteosarcoma
A previous study demonstrated that miR-874 was
downregulated in osteosarcoma and inhibits osteosarcoma cell
proliferation and metastasis (14).
Therefore, it was speculated that the specific genes suppressed by
miR-874 may predominantly function in this process. In the present
study, miRanda (http://www.microrna.org/microrna/home.do) and
Targetscan (http://www.targetscan.org/) were used to predict that
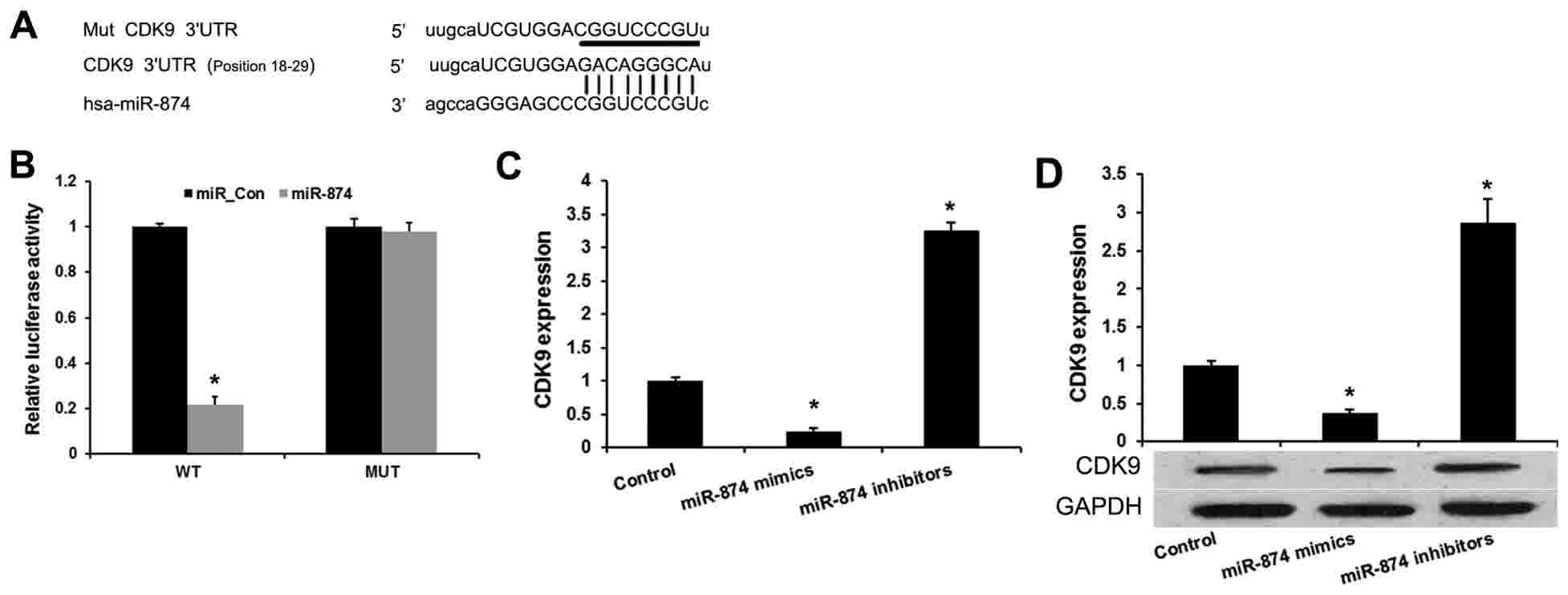
miR-874 directly binds to the 3′UTR of CDK9 mRNA (Fig. 1A). To verify the prediction, wild-type
CDK9 3′UTR and mutated luciferase reporter plasmids were
constructed (Fig. 1B). The luciferase
activity assays indicated that miR-874 mimics evidently inhibited
the reporter activity of the wild type, but not the mutant, CDK9
3′UTR. It was then investigated whether miR-874 affects endogenous
CDK9 expression. Enforced expression of miR-874 induced a
reduction, while silencing of miR-874 induced an upregulation of
endogenous CDK9 mRNA in MG-63 cells (Fig.
1C). miR-874 was also found to inhibit the CDK9 protein level
in MG-63 cells (Fig. 1D). These
results indicated that miR-874 directly targets and modulates the
expression of CDK9 in osteosarcoma cells.
CDK9 expression levels in osteosarcoma
tissues and cell lines
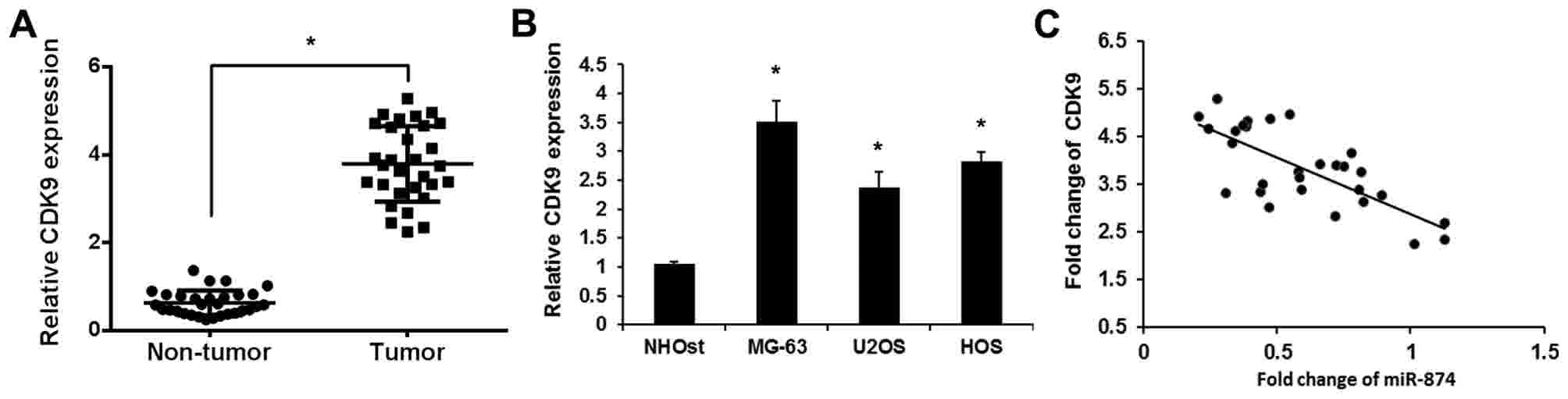
To validate the role of CDK9 in osteosarcoma, the
expression levels of CDK9 were detected in osteosarcoma tissues and
cell lines using RT-qPCR. As shown in Fig. 2A, increased levels of CDK9 were
detected in osteosarcoma tissues compared with adjacent
noncancerous tissues. CDK9 expression was also upregulated in three
osteosarcoma cell lines (MG-63, U2OS and HOS) compared with the
human normal osteoblast cell line (Fig.
2B). The correlation between miR-874 and CDK9 in osteosarcoma
was then assessed using Pearson's correlation coefficient. As
expected, the levels of miR-874 exhibited a significant negative
correlation with the levels of CDK9 mRNA (r=−0.715; P<0.01;
Fig. 2C). Overall, the present
findings indicated that the expression level of CDK9 was
upregulated and negatively associated with those of miR-874 in
clinical osteosarcoma tissues.
miR-874 inhibits osteosarcoma cell
proliferation through CDK9
A previous study reported that miR-874 inhibited
osteosarcoma progression (14).
Therefore, the present study aimed to determine whether miR-874
promotes the proliferation of osteosarcoma cells by targeting CDK9.
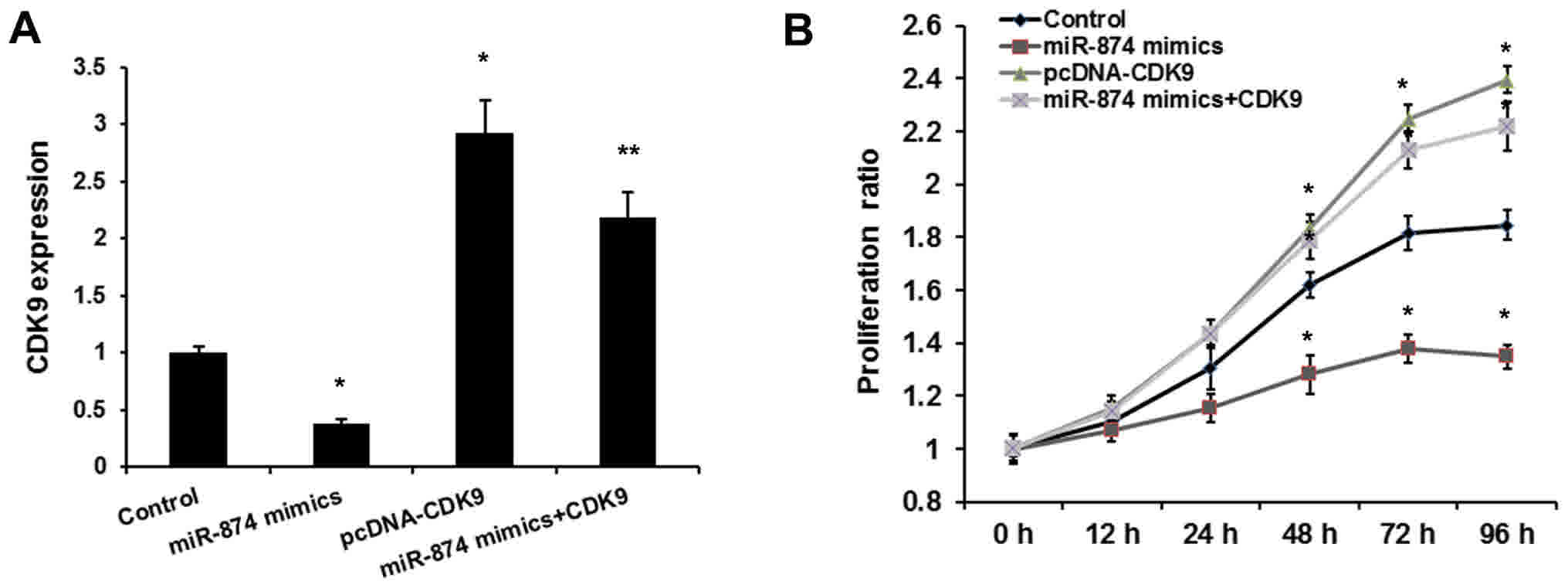
To determine whether miR-874 exerts its function by targeting CDK9,
MG-63 cells were co-transfected with miR-874 mimics and pcDNA-CDK9.
As shown in Fig. 3A, transfection of
the pcDNA-CDK9 plasmid reversed the inhibitory effects of miR-874
on the expression of CDK9. Consistent with a previous study, the
MTT assay revealed that miR-874 decreased osteosarcoma cell
proliferation. Overexpression of CDK9 rescued the oncogenic effects
of miR-874 on osteosarcoma cell proliferation (Fig. 3B). Therefore, it was proposed that
miR-874 promotes the proliferation of osteosarcoma cells by
inhibiting CDK9.
miR-874 inhibits osteosarcoma cell
invasion through CDK9
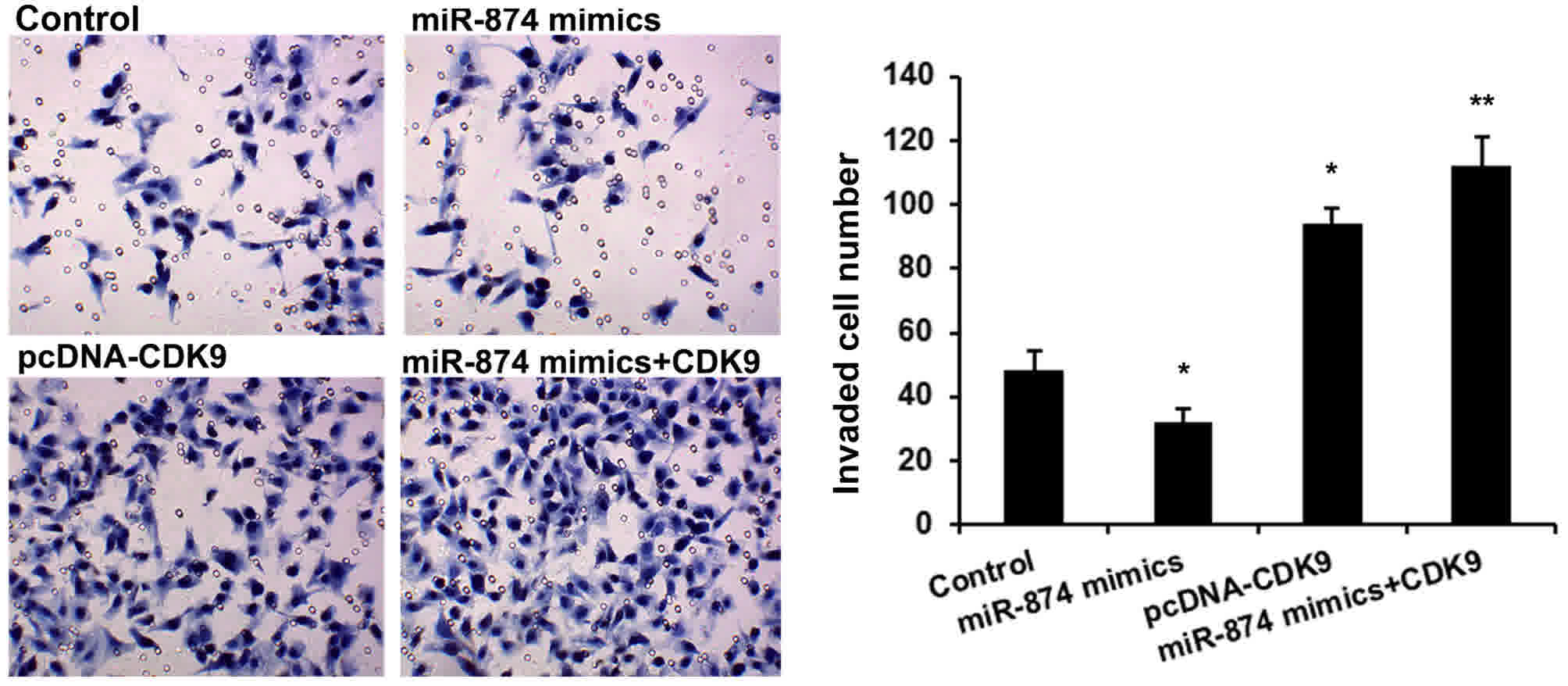
A Transwell invasion assay was then performed to
detect the effects of miR-874 and CDK9 on osteosarcoma cell
invasion. As shown in Fig. 4, MG-63
cells treated with miR-874 mimics had a significantly reduced cell
invasion (P<0.01), and CDK9 upregulation rescued the oncogenic
effects of miR-874 on cell invasion (P<0.01).
Discussion
The underlying mechanism of osteosarcoma
carcinogenesis is critical for predicting prognosis and developing
a therapeutic strategy. Therefore, it is urgent to investigate the
function of deregulated molecules in osteosarcoma progression. In
the present study, miR-874, as a tumor suppressor, was found to
regulate osteosarcoma cell proliferation and metastasis by directly
targeting CDK9.
Downregulated miR-874 has been commonly observed in
various types of cancers (10,13). A
previous study demonstrated that dysregulation of miR-874 is
involved in osteosarcoma carcinogenesis (14). The present study confirmed that
miR-874 is downregulated in osteosarcoma cells, and gain- and
loss-of-function analysis was performed to reveal the effect of
miR-874 on osteosarcoma cells. It was revealed that miR-874
overexpression significantly inhibited tumor cell proliferation
(P<0.01) and invasion (P<0.01), indicating that restoration
of miR-874 expression may be a novel therapeutic strategy in
osteosarcoma treatment.
Subsequently, the present study focused on the
underlying mechanism of how miR-874 exerts its effect on cancer
cells. CDK9 is a CDC2-associated kinase and the catalytic subunit
of positive-transcription elongation factor b and Tat-activating
kinase (16). CDK9 regulates androgen
receptor transcriptional activity and performs an important role in
prostate cancer cell growth (17).
miRanda and Targetscan predicted that miR-874 has putative binding
CDK9 sites in the 3′UTR. The effects of miR-874 on CDK9 were then
validated via a luciferase activity reporter assay and western blot
analysis, which indicated that miR-874 may be a novel negative
regulator for CDK9. In addition, the upregulated expression level
of CDK9 and negative association with the miR-874 expression level
were also observed in clinical osteosarcoma tissues. Studies have
demonstrated that deregulation of CDK9 has been associated with
numerous types of cancer (18–20) and
inhibition of CDK9 may represent potential as a cancer therapeutic
target (21,22). In the present study, it was revealed
that overexpressed CDK9 attenuated the tumor suppressor role of
miR-874, indicating the oncogene function of CDK9 in osteosarcoma
development. Therefore, it was speculated that CDK9 has important
roles in tumorigenesis, and the mechanism of how CDK9 affected
cancer development requires additional study.
In conclusion, the present study provides new
insights into the mechanism of miR-874 in osteosarcoma
proliferation and invasion, and proposes that targeting of the
miR-874/CDK9 axis may provide a potential therapeutic strategy for
osteosarcoma.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
WT and WGW conceived and designed the study. WT, YCZ
and ZYZ performed the experiments. WGW, ZYZ and WT wrote the paper.
All authors read and approved the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Research
Ethics Committee of Yantaishan Hospital and all patients provided
written informed consent.
Consent for publication
The study participants provided consent for the data
to be published
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sun XH, Geg XL, Zhang J and Zhang C:
miRNA-646 suppresses osteosarcoma cell metastasis by downregulating
fibroblast growth factor 2 (FGF2). Tumour Biol. 36:2127–2134. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yang J and Zhang W: New molecular insights
into osteosarcoma targeted therapy. Curr Opin Oncol. 25:398–406.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Han K, Zhao T, Chen X, Bian N, Yang T, Ma
Q, Cai C, Fan Q, Zhou Y and Ma B: microRNA-194 suppresses
osteosarcoma cell proliferation and metastasis in vitro and in vivo
by targeting CDH2 and IGF1R. Int J Oncol. 45:1437–1449. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sun K and Lai EC: Adult-specific functions
of animal microRNAs. Nat Rev Genet. 14:535–548. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li X, Yang H, Tian Q, Liu Y and Weng Y:
Upregulation of microRNA-17-92 cluster associates with tumor
progression and prognosis in osteosarcoma. Neoplasma. 61:453–460.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang G, Shen N, Cheng L, Lin J and Li K:
Downregulation of miR-22 acts as an unfavorable prognostic
biomarker in osteosarcoma. Tumour Biol. 36:7891–7895. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pencheva N and Tavazoie SF: Control of
metastatic progression by microRNA regulatory networks. Nat Cell
Biol. 15:546–554. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang L, Gao W, Hu F, Xu Z and Wang F:
MicroRNA-874 inhibits cell proliferation and induces apoptosis in
human breast cancer by targeting CDK9. FEBS Lett. 588:4527–4535.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang X, Tang J, Zhi X, Xie K, Wang W, Li
Z, Zhu Y, Yang L, Xu H and Xu Z: miR-874 functions as a tumor
suppressor by inhibiting angiogenesis through STAT3/VEGF-A pathway
in gastric cancer. Oncotarget. 6:1605–1617. 2015.PubMed/NCBI
|
|
10
|
Nohata N, Hanazawa T, Kikkawa N, Sakurai
D, Fujimura L, Chiyomaru T, Kawakami K, Yoshino H, Enokida H,
Nakagawa M, et al: Tumour suppressive microRNA-874 regulates novel
cancer networks in maxillary sinus squamous cell carcinoma. Br J
Cancer. 105:833–841. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kesanakurti D, Maddirela DR, Chittivelu S,
Rao JS and Chetty C: Suppression of tumor cell invasiveness and in
vivo tumor growth by microRNA-874 in non-small cell lung cancer.
Biochem Biophys Res Commun. 434:627–633. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nohata N, Hanazawa T, Kinoshita T, Inamine
A, Kikkawa N, Itesako T, Yoshino H, Enokida H, Nakagawa M, Okamoto
Y and Seki N: Tumour-suppressive microRNA-874 contributes to cell
proliferation through targeting of histone deacetylase 1 in head
and neck squamous cell carcinoma. Br J Cancer. 108:1648–1658. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jiang B, Li Z, Zhang W, Wang H, Zhi X,
Feng J, Chen Z, Zhu Y, Yang L, Xu H and Xu Z: miR-874 inhibits cell
proliferation, migration and invasion through targeting aquaporin-3
in gastric cancer. J Gastroenterol. 49:1011–1025. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang LQ, Sun SL, Li WY, Feng Z, Xu XY,
Zhuang QS and Fang J: Decreased expression of tumor suppressive
miR-874 and its clinical significance in human osteosarcoma. Genet
Mol Res. 14:18315–18324. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Garriga J, Bhattacharya S, Calbó J,
Marshall RM, Truongcao M, Haines DS and Graña X: CDK9 is
constitutively expressed throughout the cell cycle, and its
steady-state expression is independent of SKP2. Mol Cell Biol.
23:5165–5173. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gordon V, Bhadel S, Wunderlich W, Zhang J,
Ficarro SB, Mollah SA, Shabanowitz J, Hunt DF, Xenarios I, Hahn WC,
et al: CDK9 regulates AR promoter selectivity and cell growth
through serine 81 phosphorylation. Mol Endocrinol. 24:2267–2280.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Krystof V, Baumli S and Fürst R:
Perspective of cyclin-dependent kinase 9 (CDK9) as a drug target.
Curr Pharm Des. 18:2883–2890. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mitra P, Yang RM, Sutton J, Ramsay RG and
Gonda TJ: CDK9 inhibitors selectively target estrogen
receptor-positive breast cancer cells through combined inhibition
of MYB and MCL-1 expression. Oncotarget. 23:9069–9083. 2016.
|
|
20
|
Morales F and Giordano A: Overview of CDK9
as a target in cancer research. Cell Cycle. 15:519–527. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yin T, Lallena MJ, Kreklau EL, Fales KR,
Carballares S, Torrres R, Wishart GN, Ajamie RT, Cronier DM,
Iversen PW, et al: A novel CDK9 inhibitor shows potent antitumor
efficacy in preclinical hematologic tumor models. Mol Cancer Ther.
13:1442–1456. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Liu X, Shi S, Lam F, Pepper C, Fischer PM
and Wang S: CDKI-71, a novel CDK9 inhibitor, is preferentially
cytotoxic to cancer cells compared to flavopiridol. Int J Cancer.
130:1216–1226. 2012. View Article : Google Scholar : PubMed/NCBI
|