Introduction
Cancer constitutes a global burden and has become a
serious health challenge (1,2). The occurrence of cancer is increasing
due to an increasing aging population and prevalence of established
risk factors (3–6). Although over the last few decades, novel
therapeutic strategies have been developed, the prognosis and
survival rate for patients with cancer is far from satisfactory.
Therefore, investigating the molecular mechanisms underlying
tumorigenesis has become urgent, particularly for the targeted
treatment of human cancer. Investigation of the underlying
molecular mechanisms is required to identify novel therapeutic
targets for cancer.
Aspirin, which is one of the most common therapeutic
drugs worldwide, was primarily introduced as an anti-inflammatory
and analgesic agent (7).
Nevertheless, clinical and epidemiological studies have
demonstrated that aspirin may decrease the risk of several types of
cancer, including colorectal, breast, prostate, lung and skin
cancer (8–12). The anticancer effects of aspirin may
be dependent on its ability to induce apoptosis or inhibit
proliferation in cancer cells. To the best of our knowledge, there
is no evidence to indicate whether aspirin is able to promote
apoptosis and inhibit the proliferation and invasion of Hep-2
cells. The Hep-2 cell line is a mixed neoplastic type of cell type
originally considered to derive from an epidermoid carcinoma of the
larynx, but it has been demonstrated to be contaminated with
cervical adenocarcinoma HeLa cells (13–15).
Therefore, investigating the effect of aspirin on Hep-2 cells in
this general model of cancer may be beneficial to prevent drug
resistance in cancer therapy and develop novel tumor-targeted
drugs.
The aim of the present study was to investigate the
effects of aspirin on apoptosis, proliferation and invasion in
Hep-2 cells. Furthermore, the expression of survivin, a key
anti-apoptotic regulator of the phosphatase and tensin homolog
(PTEN)/protein kinase B (AKT)/nuclear factor (NF)-κB signaling
pathway, was examined. The results demonstrated that aspirin
promoted apoptosis and inhibited proliferation and invasion of
Hep-2 cells. Additionally, aspirin increased the B-cell lymphoma 2
(Bcl-2)-associated X protein (Bax)/Bcl-2 ratio and activated
caspase-3. These results indicate that the proliferation and
metastasis may be partly regulated by aspirin in Hep-2 cells.
Collectively, these results not only contribute to an improved
understanding of the molecular mechanisms of apoptosis underlying
the anticancer effect of aspirin, but also suggest therapeutic
targets for several types of cancer.
Materials and methods
Cell culture
Hep-2 cells were obtained from the American Type
Culture Collection (Manassas, VA, USA). Hep-2 cells were cultured
in Dulbecco's modified Eagle's medium (DMEM; Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal
bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.) and 100
µg/ml penicillin/streptomycin (Beyotime Institute of Biotechnology,
Shanghai, China) at 37°C in a humidified atmosphere containing 5%
CO2.
3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide
(MTT) assay
The drug half-maximal inhibitory concentration
(IC50) values were determined by using MTT. In brief,
1×104 of Hep-2 cells were plated in a 96-well plate and
were treated with aspirin (10, 50, 100 and 200 µg/ml) or 10 µM
bovine papillomavirus (BPV; both Beyotime Institute of
Biotechnology) for different time points. Cells were then incubated
with 10 µl 0.5 mg/ml MTT (Sigma-Aldrich; Merck KGaA, Darmstadt,
Germany) at 37°C for 4 h. The purple formazan crystals that formed
were dissolved in 100 µl dimethyl sulfoxide and added to the cells.
Absorbance was determined at a wavelength of 490 nm using a
multi-plate reader (Synergy 2, BioTek Instruments, Inc., Winooski,
VT, USA) and drug half-maximal inhibitory concentration
(IC50) values were established.
Terminal deoxynucleotidyl transferase
dUTP nick end labeling (TUNEL) assay
Hep-2 cells (1×104) were washed three
times with PBS and fixed with 4% paraformaldehyde at room
temperature for 30 min, permeabilized in 0.1% Triton X-100 in
sodium citrate buffer. Apoptotic cells were labeled using an In
Situ Cell Death Detection kit (Roche Diagnostics, Indianapolis, IN,
USA), according to the manufacturer's protocol at room temperature
for 15 min and the nuclei were stained with 0.3 mmol/l DAPI at room
temperature for 5 min. TUNEL-positive cells were observed in 6
randomly selected fields under the Olympus BX50 (Olympus
Corporation, Tokyo, Japan) fluorescence microscope (magnification,
×200). The apoptotic rate, which was defined as the ratio of
apoptotic cells to total cells, was calculated and analyzed using
Image-Pro Plus 6.0 software (Nikon Corporation, Tokyo, Japan).
Flow cytometric analysis of
apoptosis
Hep-2 cells (1×105) were plated in a
6-well plate for 24 h and then treated with aspirin, aspirin plus
10 µM BPV or saline (control). Cells were subsequently incubated at
room temperature with Annexin V-fluorescein isothiocyanate and
propidium iodine (PI; BD Biosciences, Franklin Lakes, NJ, USA) for
15 min before being analyzed on the Accuri™ C6 flow
cytometer (BD Biosciences) using FlowJo, version 7.6.1 (FlowJo LLC,
Ashland, OR, USA).
Transwell migration and invasion
assay
The migration and invasion assay was carried out
using Transwell plates (EMD Millipore, Billerica, MA, USA). The
filter surfaces (8 µm pores) of the Transwell plates were precoated
with 25 mg Matrigel at 4°C overnight. Culture medium (DMEM)
supplemented with 10% FBS was placed in the lower chambers. Hep-2
cells (1×104) were placed in the upper chambers of the
Transwell plates without FBS. Following incubation for 24 h,
invaded cells were fixed with 4% paraformaldehyde and stained with
Giemsa at room temperature for 5 min. Three replicates were used
for each condition. Stained cells were counted in 15 random fields
using light microscopy (magnification, ×200; CKX41; Olympus
Corporation, Tokyo, Japan). The results are presented as a ratio
relative to the control group.
Western blot analysis
Total protein was extracted from Hep-2 cells using
RIPA buffer (Beyotime Institute of Biotechnology). Total protein
was quantified using bicinchoninic acid protein assay kit (Beyotime
Institute of Biotechnology) and 80 µg protein/lane was separated
using SDS-PAGE (10% gels) and transferred onto nitrocellulose
membranes. The membranes were blocked with 5% non-fat milk
dissolved in PBST for 2 h at room temperature and incubated with
the following primary antibodies: Anti-Bcl-2 (15071; 1:1,000),
anti-Bax (5023; 1:1,000), anti-caspase-3 (9662; 1:1,000), anti-PTEN
(9559; 1:1,000), anti-AKT (9272; 1:1,000), anti-phospho (p)-AKT
(4060; 1:1,000), anti-NF-κB (8242; 1:1,000), anti-survivin (2808;
1:1,000; all Cell Signaling Technology, Inc., Danvers, MA, USA),
anti-β-actin (612657; 1:1,000; BD Biosciences, Franklin Lakes, NJ,
USA) diluted in PBS and incubated at 4°C overnight. Membranes were
then washed with Tris-buffered saline with Tween-20 (TBST) and
incubated with goat anti-rabbit IgG secondary antibody (Alexa
Fluor-conjugated; A32730; Invitrogen; Thermo Fisher Scientific,
Inc.) for 1 h at room temperature. Protein bands were visualized
using the Odyssey Infrared Imaging System (LI-COR Biosciences,
Lincoln, NE, USA) and quantified using Odyssey software version 1.2
(LI-COR Biosciences). GAPDH was used as an internal control.
Statistical analysis
Data were analyzed using GraphPad Prism (version
6.0; GraphPad Software, Inc., La Jolla, CA, USA). Data are
expressed as the mean ± standard error of the mean. Results were
analyzed using an unpaired two-tailed Student's t-test and one-way
analysis of variance followed by Bonferroni's correction for
multiple comparison tests. P<0.05 was considered to indicate a
statistically significant difference.
Results
Aspirin significantly decreases
viability in a dose- and time-dependent manner in Hep-2 cells
In order to examine the effect of aspirin on the
viability of Hep-2 cells, MTT assays were employed. Hep-2 cells
were treated with 0, 10, 50, 100 and 200 µg/ml aspirin for 48 h.
The IC50 value of aspirin was identified to be 91.2
µg/ml (Fig. 1A); therefore, 100 µg/ml
was selected as an optimal concentration for subsequent
experiments. Hep-2 cells were treated with 100 µg/ml aspirin for
12, 24 and 48 h (Fig. 1B). The
results demonstrated that aspirin significantly decreased the
viability of Hep-2 cells in a dose- and time-dependent manner
(Fig. 1A and B; P<0.05). However,
further in vivo and in vitro experiments are required
to confirm these results.
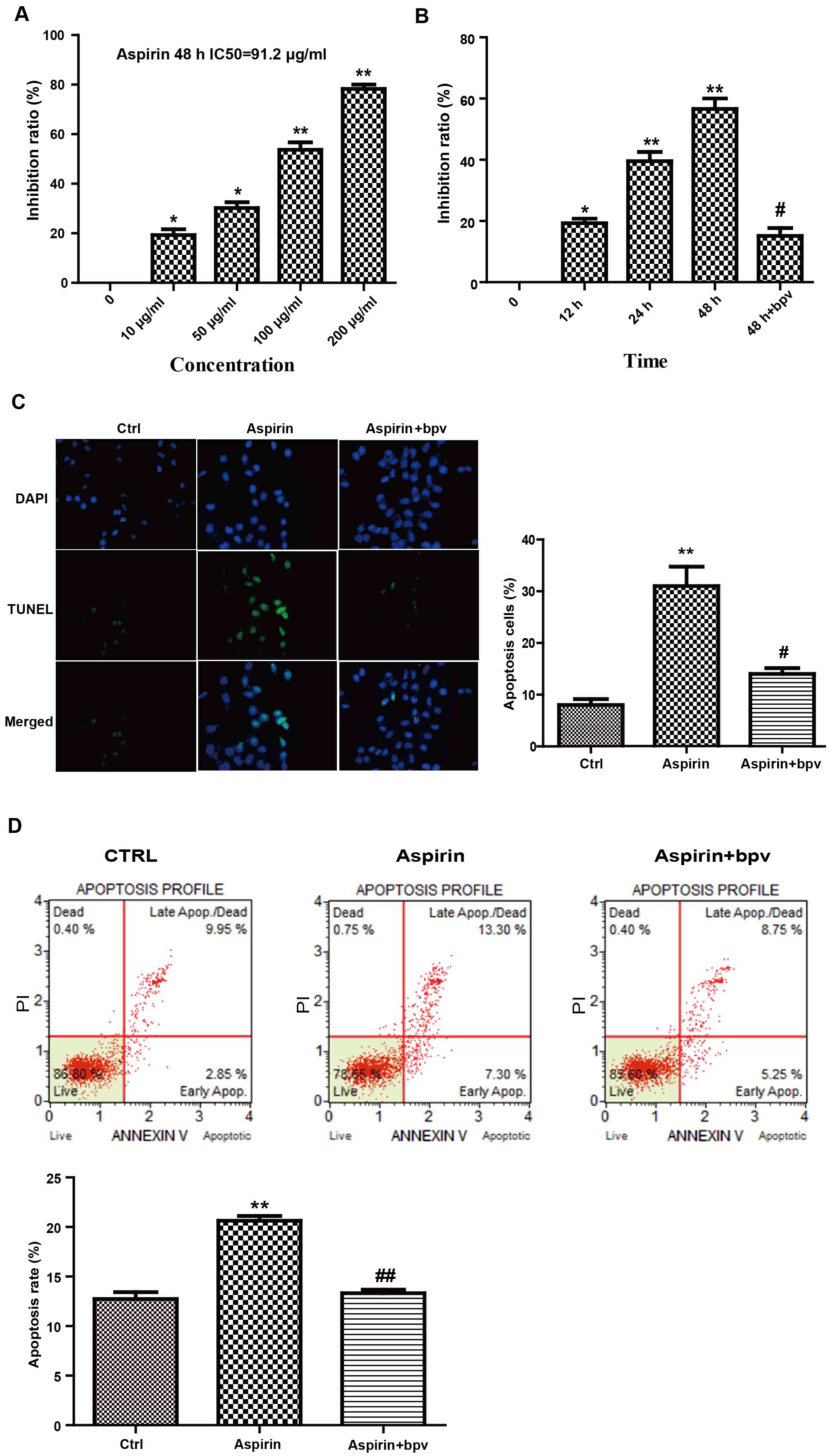 | Figure 1.Aspirin inhibits viability and induces
apoptosis in Hep-2 cells. (A) Cells were treated with 0, 10, 50,
100 and 200 µg/ml aspirin for 48 h, and cell viability was assessed
using an MTT assay. (B) Cells were treated with 100 µg/ml aspirin
for 12, 24 and 48 h, and cell viability was assessed using an MTT
assay. (C) Aspirin promotes cell apoptosis in Hep-2 cells as
demonstrated using a TUNEL assay. (D) Aspirin promotes apoptosis in
Hep-2 cells as demonstrated using flow cytometric analysis of
apoptosis. n=6; *P<0.05 vs. control; **P<0.01 vs. control;
#P<0.05 or ##P<0.01 vs. aspirin. MTT,
3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide;
TUNEL, terminal deoxynucleotidyl-transferase-mediated dUTP nick end
labeling; BPV, bovine papillomavirus; apop., apoptosis; Ctrl,
control. |
Since aspirin is a potential agonist of PTEN
(16), the present study investigated
whether aspirin may decrease cell viability via regulating the PTEN
signaling pathway. Therefore, cells were treated with 100 µg/ml
aspirin and 10 µM BPV, an inhibitor of PTEN, for 48 h, and cell
viability was assessed using an MTT assay. As presented in Fig. 1B, combined treatment with aspirin and
BPV significantly decreased the inhibition in Hep-2 cells,
suggesting that the PTEN signaling pathway may be a molecular
mechanism underlying aspirin-mediated cellular changes in Hep-2
cells.
Aspirin promotes apoptosis in Hep-2
cells
To evaluate the effect of aspirin on apoptosis,
apoptotic cells were stained using a TUNEL assay. The results
demonstrated that treatment with aspirin significantly increased
the numbers of apoptotic cells in Hep-2 cells (Fig. 1C) compared with in the control group
(P<0.05). Additionally, BPV reversed the pro-apoptotic effects
of aspirin in Hep-2 cells (Fig.
1C).
The apoptosis results were confirmed by flow
cytometric analysis. As presented in Fig.
1D, aspirin induced an increase in apoptosis compared with that
in the control group. Additionally, combined treatment with aspirin
and BPV decreased the number of apoptotic cells compared with
aspirin treatment (P<0.05).
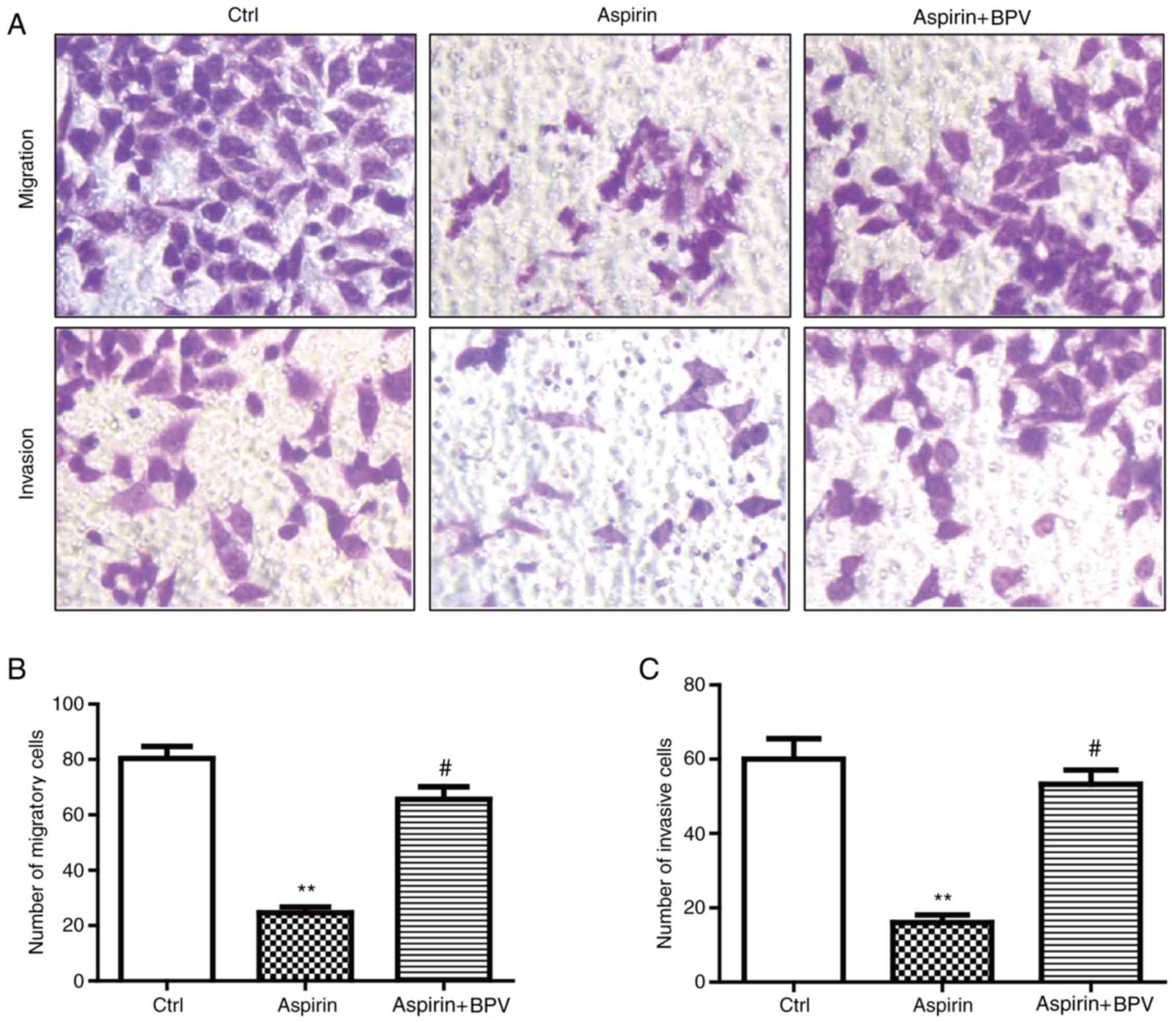
Aspirin inhibits the migratory and
invasive abilities of Hep-2 cells
Transwell assays were employed to investigate the
ability of Hep-2 cells to migrate to and invade the extracellular
matrix. The results demonstrated that the migration rate of Hep-2
cells decreased by >60% compared with that in the control group
(Fig. 2A and B). However, combined
treatment with aspirin and BPV did not affect the migratory ability
of Hep-2 cells compared with those in the control group (Fig. 2B).
The effect of aspirin on cell invasion was
investigated. As presented in Fig.
2C, the number of invasive cells was decreased by ~70% in
response to treatment with aspirin in Hep-2 cells compared with
those in the control group. Taken together, these results indicated
that aspirin significantly inhibited cell invasion and migration
in vitro and these effects were reversed when PTEN
expression was downregulated.
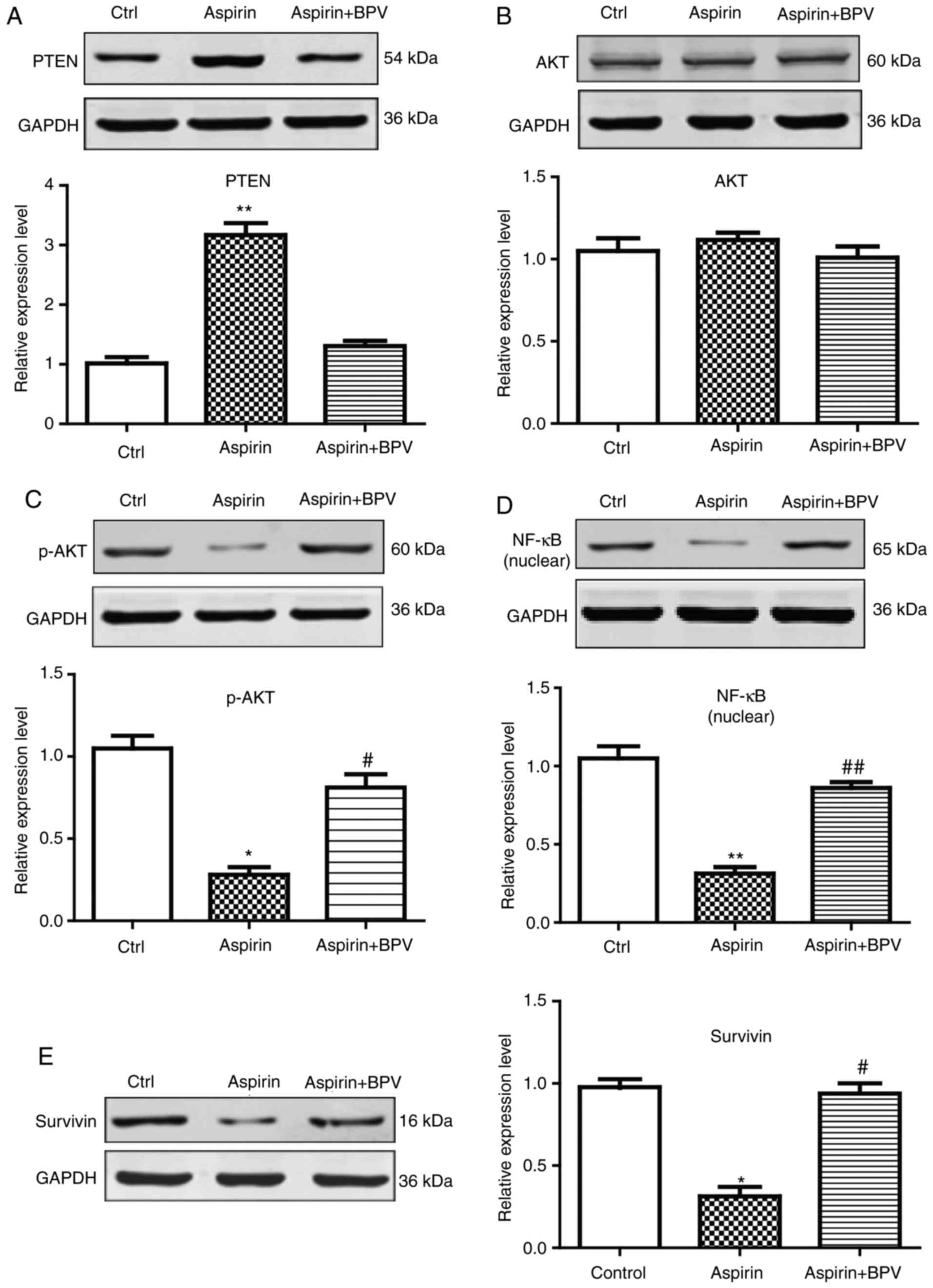
Involvement of the
PTEN/AKT/NF-κB/survivin pathway in aspirin-induced apoptosis in
Hep-2 cells
To determine the molecular mechanism underlying the
pro-apoptotic effects of aspirin, Hep-2 cells were treated with
aspirin alone or with aspirin and BPV, and the expression levels of
proteins involved in the PTEN/AKT/NF-κB/survivin signaling pathway
were determined using western blot analysis. As presented in
Fig. 3A, treatment with aspirin
significantly increased the expression level of PTEN compared with
that in the control group. Additionally, aspirin significantly
decreased the relative expression level of p-AKT without affecting
the expression levels of total AKT in Hep-2 cells (Fig. 3B and C). Furthermore, as presented in
Fig. 3D and E, the expression levels
of NF-κB and survivin were significantly decreased in the aspirin
group compared with that in the control group (P<0.05). However,
combined treatment with aspirin and BPV did not affect the
expression levels of these proteins compared with the control group
(P>0.05). These results indicated that aspirin decreased the
protein expression of NF-κB and survivin by inactivating the
PTEN/AKT signaling pathway in Hep-2 cells.
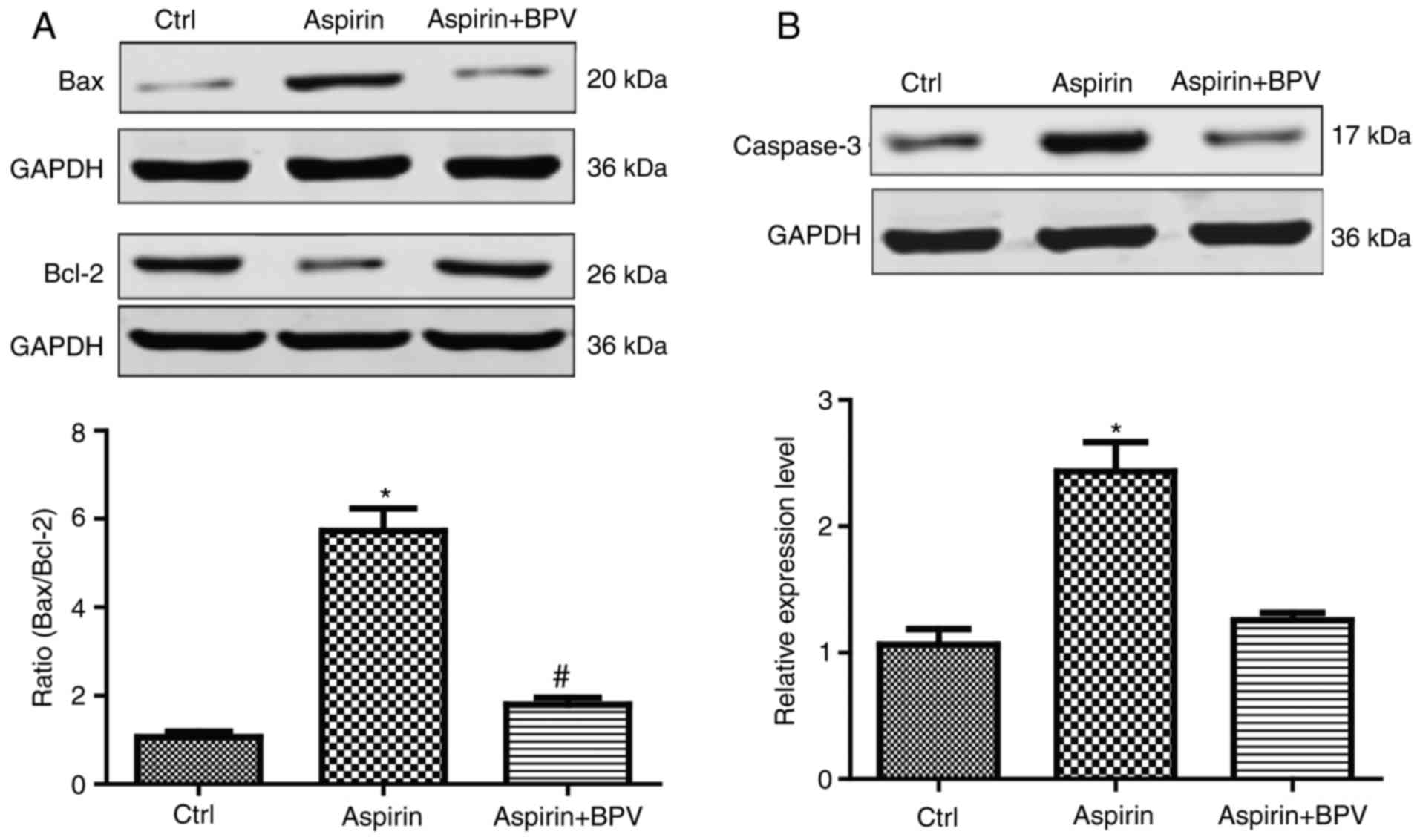
The effects of aspirin on the expression of
downstream apoptotic proteins of the PTEN/AKT/NF-κB/survivin
signaling pathway were investigated in Hep-2 cells. The Bcl-2
family proteins include the pro-apoptotic Bax and anti-apoptotic
Bcl2 proteins (17). Hep-2 cells were
incubated with aspirin or with aspirin and BPV for 24 h. The
results demonstrated that Hep-2 cells treated with aspirin alone
exhibited increased expression levels of Bax and decreased
expression levels of Bcl-2 compared with those in the control group
(Fig. 4A). Additionally, aspirin
treatment induced a >3-fold increase in the Bax/Bcl-2 ratio
compared with that in control group, indicating that aspirin
induces apoptosis in Hep-2 cells.
Caspase-3 is a major caspase and its activation
leads to cell death (18). Therefore,
the expression levels of caspase-3 in response to treatment with
aspirin were evaluated using western blot analysis. The results
revealed that the expression of caspase-3 was significantly
upregulated in response to treatment with aspirin in Hep-2 cells
(Fig. 4B). However, treatment with
BPV attenuated the pro-apoptotic effects of aspirin in Hep-2 cells
(Fig. 4B). Taken together, these
results suggest that aspirin markedly induced apoptosis in Hep-2
cells by inhibiting the PTEN/AKT/NF-κB/survivin signaling
pathway.
Discussion
Malignant tumors are a major health concern. Drug
resistance and relapse are the main reasons for poor prognosis and
low survival rate. Therefore, there is an urgent requirement to
elucidate the underlying molecular mechanisms of drug resistance,
cell proliferation and metastasis in order to identify novel
therapeutic approaches and improve the prognosis for patients with
cancer. In the present study, it was demonstrated that aspirin
induced apoptosis and suppressed the invasive and migratory
abilities of Hep-2 cells. Additionally, aspirin mediated cellular
proliferation, invasion and apoptosis via regulating the expression
of the PTEN/AKT/NF-κB/survivin signaling pathway in vitro.
These results provide insights into the molecular mechanisms of
aspirin to regulate the proliferation, invasion and apoptosis in
Hep-2 cells, but also suggest novel therapeutic targets for the
treatment of various types of cancer.
Aspirin, also known as acetylsalicylic acid, is a
non-steroidal anti-inflammatory drug, which is non-toxic to humans
(19). Previous clinical and
epidemiological studies have demonstrated that long-term treatment
with aspirin may decrease the risk of colorectal, breast, prostate,
lung and skin cancer (8,10,12).
However, whether aspirin may promote cellular apoptosis and inhibit
proliferation and invasion remains unclear. The aim of the present
study was to investigate the effects of aspirin on cell apoptosis,
proliferation and invasion in Hep-2 cells. The results demonstrated
that the molecular mechanism by which aspirin induced apoptosis and
inhibited proliferation and invasion may be associated with changes
in the expression of Bcl-2, Bax and caspase-3. Additionally, the
PTEN/AKT/NF-κB/survivin signaling pathway may have a function in
the molecular mechanism underlying aspirin-induced cellular
changes.
PTEN is a key molecule involved in glucose
metabolism, cellular differentiation, proliferation, apoptosis and
migration and in inflammatory response by targeting AKT and NF-κB.
The PTEN/AKT signaling pathway leads to deregulation of the cell
cycle and to apoptosis, and induces the differentiation of normal
cells into tumor cells (20–22). In the present study, PTEN was selected
as a potential target for aspirin to investigate whether aspirin
may be involved in the PTEN/AKT signaling pathway in Hep-2 cells.
The results demonstrated that aspirin upregulated the expression of
PTEN and inhibited p-AKT, whereas total AKT expression was not
significantly altered. Additionally, aspirin inhibited the
expression of proteins downstream of the PTEN/AKT signaling
pathway, including NF-κB and survivin. Additionally, aspirin
increased the expression of Bcl-2, and decreased the expression of
Bax and caspase-3 in Hep-2 cells. Collectively, these results
demonstrate that aspirin serves a pro-apoptotic function by
activating the PTEN/AKT/NF-κB/survivin signaling pathway in Hep-2
cells. Aspirin-mediated cellular changes were partially inhibited
in response to a PTEN inhibitor, indicating that the
PTEN/AKT/NF-κB/survivin signaling pathway may be an important
target for the development of novel therapeutic strategies for
cancer.
The results of the present study identified that
aspirin was able to suppress proliferation, migration and invasion,
and promote apoptosis of Hep-2 cells via the
PTEN/AKT/NF-κB/survivin signaling pathway. Thus, aspirin has a
critical effect in modulating cell proliferation, migration and
apoptosis, and may serve as a potential therapeutic target for
patients with cancer.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Author's contributions
MJ was responsible for the design and writing of the
article, and performance of experiments. CL performed the TUNEL
assay and flow cytometric analysis of apoptosis. QZ and SX
performed western blotting, and Transwell migration and invasion
assays. XK contributed to data analysis. JW constructed diagrams
and was responsible for study conception and revision of the
article. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kirkegård J, Mortensen FV and
Cronin-Fenton D: Chronic pancreatitis and pancreatic cancer risk: A
systematic review and meta-analysis. Am J Gastroenterol.
112:1366–1372. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Komaki Y, Komaki F, Micic D, Ido A and
Sakuraba A: Risk of colorectal cancer in chronic kidney disease: A
systematic review and meta-analysis. J Clin Gastroenterol.
18–Jul;2017.doi: 10.1097/MCG.0000000000000880. View Article : Google Scholar
|
|
5
|
Bopanna S, Ananthakrishnan AN, Kedia S,
Yajnik V and Ahuja V: Risk of colorectal cancer in Asian patients
with ulcerative colitis: A systematic review and meta-analysis.
Lancet Gastroenterol Hepatol. 2:269–276. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Haugvik SP, Ibrahim Basim I, Hedenstrom P,
Hedenström P, Valente R, Hayes AJ, Siuka D, Gladhaug IP and Capurso
G: Smoking, alcohol and family history of cancer as risk factors
for small intestinal neuroendocrine tumors: A systematic review and
meta-analysis. Scand J Gastroenterol. 52:797–802. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sneader W: The discovery of aspirin: A
reappraisal. BMJ. 321:1591–1594. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sandler RS, Galanko JC, Murray SC, Helm JF
and Woosley JT: Aspirin and nonsteroidal anti-inflammatory agents
and risk for colorectal adenomas. Gastroenterology. 114:441–447.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Harris RE, Namboodiri KK and Farrar WB:
Nonsteroidal antiinflammatory drugs and breast cancer.
Epidemiology. 7:203–205. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dasgupta K, Di Cesar D, Ghosn J, Rajan R,
Mahmud S and Rahme E: Association between nonsteroidal
anti-inflammatory drugs and prostate cancer occurrence. Cancer J.
12:130–135. 2006.PubMed/NCBI
|
|
11
|
Harris RE, Beebe-Donk J and Alshafie GA:
Reduced risk of human lung cancer by selective cyclooxygenase 2
(COX-2) blockade: Results of a case control study. Int J Biol Sci.
3:328–334. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
McCaffrey P: Aspirin use reduces
skin-cancer risk. Lancet Oncol. 7:162006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Nelson-Rees WA, Zhdanov VM, Hawthorne PK
and Flandermeyer RR: HeLa-like marker chromosomes and type-A
variant glucose-6-phosphate dehydrogenase isoenzyme in human cell
cultures producing Mason-Pfizer monkey virus-like particles. J Natl
Cancer Inst. 53:751–757. 1974. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen TR: Re-evaluation of HeLa, HeLa S3,
and HEp-2 karyotypes. Cytogenet Cell Genet. 48:19–24. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Carter KL and Roizman B: The promoter and
transcriptional unit of a novel herpes simplex virus 1 alpha gene
are contained in, and encode a protein in frame with, the open
reading frame of the alpha 22 gene. J Virol. 70:172–178.
1996.PubMed/NCBI
|
|
16
|
Tarnawski AS, Ahluwalia A and Jones MK:
Angiogenesis in gastric mucosa: An important component of gastric
erosion and ulcer healing and its impairment in aging. J
Gastroenterol Hepatol. 4 29 Suppl:S112–S123. 2014. View Article : Google Scholar
|
|
17
|
Suen DF, Norris KL and Youle RJ:
Mitochondrial dynamics and apoptosis. Genes Dev. 22:1577–1590.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Slee EA, Adrain C and Martin SJ:
Executioner caspase-3, −6, and −7 perform distinct, non-redundant
roles during the demolition phase of apoptosis. J Biol Chem.
276:7320–7326. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gilroy DW: The role of aspirin-triggered
lipoxins in the mechanism of action of aspirin. Prostaglandins
Leukot Essent Fatty Acids. 73:203–210. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen X, Yang C, Xu Y, Zhou H, Liu H and
Qian W: The microtubule depolymerizing agent CYT997 effectively
kills acute myeloid leukemia cells via activation of caspases and
inhibition of PI3K/Akt/mTOR pathway proteins. Exp Ther Med.
6:299–304. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ahmad A, Biersack B, Li Y, Kong D, Bao B,
Schobert R, Padhye SB and Sarkar FH: Targeted regulation of
PI3K/Akt/mTOR/NF-κB signaling by indole compounds and their
derivatives: Mechanistic details and biological implications for
cancer therapy. Anticancer Agents Med Chem. 13:1002–1013. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang L, Yao J, Sun H, He K, Tong D, Song T
and Huang C: MicroRNA-101 suppresses progression of lung cancer
through the PTEN/AKT signaling pathway by targeting DNA
methyltransferase 3A. Oncol Lett. 13:329–338. 2017. View Article : Google Scholar : PubMed/NCBI
|