Introduction
Lung cancer is the most prevalent type of cancer in
the world. According to the World Health Organization (WHO), about
1.59 million cases of lung cancer-related deaths were recorded
worldwide in 2012; and among those, 597 thousand cases took place
in China. As a populated country with lots of risk factors such as
smoking and air pollution, medical care departments in China is
facing with a growing burden of cancer treatment. Therefore, it is
crucial to uncover the mechanism of lung cancer formation and thus
develop new biomarkers for the detection and treatment of the
disease. DEK is an ubiquitous protein in multicellular organisms as
well as in some unicellular organisms (1). It was originally discovered as a fusion
protein with CAN nucleoprotein in a subtype of acute myeloid
leukemia in 1992 (2). Recent findings
showed that DEK may act as an oncoprotein during tumor formation,
because of its active role in regulating cell proliferation,
differentiation, migration, apoptosis, senescence, self-renewal and
DNA repairing (3). Meanwhile, it has
been found that DEK is over-expressed in various malignant tumor
cells, such as neuroendocrine prostate cancer cells, gastric
adenocarcinoma cells, colorectal cancer cells, breast cancer cells,
bladder cancer cells, human hepatocellular carcinoma cells and so
on, while it remains in a low level or is even undetectable in
quiescent and terminally differentiated cells (4–16).
Over-expression of DEK boosts proliferation and transformation of
epithelial cells, promotes tumor growth and metastasis of breast
cancer cells, while it inhibits normal cell differentiation,
suppresses p53 pathway-induced apoptosis and acts as a senescence
inhibitor with E6 and E7 oncogene (17,18). DEK
depletion, on the other hand, may trigger cell apoptosis or
senescence (19–23), and also sensitize cells to
chemotherapy or genotoxic agents (9,23–26). These facts demonstrate the potential
applications of DEK as a tumor diagnostic biomarker and also as a
therapeutic target.
This study focused on the poorly understood function
of DEK in lung cancer cells, and discovered the clinical
pathological significance of DEK in lung cancer detection and
treatment.
Materials and methods
Clinical samples and ethics
statement
In this study, 81 specimens of surgically resected
lung tumors as well as corresponding adjacent precancerous tissue
(with 30 cm distance from the tumor's edge) and/or normal tissues
(with 50 cm distance from the tumor's edge) were collected at the
Fourth Military Medical University Affiliated Tangdu Hospital and
the General Hospital of People's Liberation Army from December 2010
to November 2014 after receiving the patients' informed consent and
the approval of hospital authorities. None of the patients received
radiotherapy or chemotherapy prior to the pulmonary surgery. After
surgical removal, all of the samples were immediately snap-frozen
in liquid nitrogen and stored at −80°C for RNA (29 cases) and
protein analyses. All cancerous tissues were primary tumors
according to the International Union against Cancer guidelines. The
Beijing Jiaotong University Institutional Review Board approved the
present study.
Cell cultures, siRNAs and
plasmids
A549 cell line was obtained from American Type
Culture Collection (Manassas, VA, USA). Cells were cultured in
Dulbecco's Modified Eagle Medium (DMEM; Invitrogen; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) with 10% fetal bovine serum
(FBS) and were passaged every 3 days using 0.25% trypsin (Merck
KGaA, Darmstadt, Germany). The siRNA targeting DEK CDS region was
synthesized by Invitrogen, and it was transfected into A549 cells
using Lipofectamine 3000 (Invitrogen; Thermo Fisher Scientific,
Inc.) according to the manufacturer's protocol. Sequences of the
siRNAs are listed (Table I). The
pcDNA3.1-hDEK plasmid was constructed using the pcDNA3.1 (−)
plasmid and the DEK CDS sequence with a 6xCAT Histidine Tag at its
N-terminal. A corresponding negative control plasmid was
constructed with an EGFP CDS sequence replacing the His-DEK, and it
acted as a positive control for the transfection.
 | Table I.List of siRNA sequences. |
Table I.
List of siRNA sequences.
| Name | Sense-strand sequence
(5′ to 3′) | Antisense-stand
sequence (5′ to 3′) |
|---|
| siRNA-DEK |
GGAUAGUUCAGAUGAUGAACCdTdT | GGUUCAUCAUCUGAACUAUCC
dTdT |
| siRNA-NC | UUCUCCGAACGUGUCACGU
dTdT |
ACGUGACACGUUCGGAGAAdTdT |
RNA extraction and reverse
transcription-PCR
Total RNA was extracted from the lung tumor and
matched non-tumorous tissues using Trizol reagent (Invitrogen;
Thermo Fisher Scientific, Inc.). A total of 1.5 µg mRNA aliquot
from each sample were reverse transcribed to double-stranded cDNAs
using Go Script™ Reverse Transcription System (Promega
Corporation, Madison, WI, USA) with the final volume of 20 µl.
Quantitative reverse transcription-PCR (qRT-PCR) was performed
using Super Real PreMix Plus (SYBR Green; Tiangen Biotech Co.,
Ltd., Beijing, China) and Light Cycler 480 (Roche Diagnostics,
Indianapolis, IN, USA) according to the manufacturer's protocol.
cDNAs were diluted into quarters and 1 µl of diluted cDNAs were
added into the reaction mix with the final volume of 20 µl.
Reaction conditions were as followed: 95°C 15 min; followed by 40
cycles of 95°C 10 sec, 60°C 20 sec (signal acquisition). Experiment
was performed in triplicates and the fold change of each gene was
calculated using the 2−ΔΔCt method. Primers used in this
experiment are listed (Table
II).
 | Table II.Primers used in the experiment. |
Table II.
Primers used in the experiment.
| Name of primer | Sequence (5′ to
3′) | Product size
(bp) |
|---|
| DEK-Fw |
TGTTAAGAAAGCAGATAGCAGCACC | 96 |
| DEK-Rv |
ATTAAAGGTTCATCATCTGAACTATCCTC |
|
| B2M-Fw |
GGCTATCCAGCGTACTCC | 247 |
| B2M-Rv |
ACGGCAGGCATACTCATC |
|
| GAPDH-Fw |
TGACATCAAGAAGGTGGTGAAGCAGG | 123 |
| GAPDH-Rv |
GCGTCAAAGGTGGAGGAGTGGGT |
|
| p53-Fw |
GCGAGCACTGCCCAACAACA | 83 |
| p53-Rv |
GGATCTGAAGGGTGAAATATTCT |
|
| p65-Fw |
ATGTGGAGATCATTGAGCAGC | 152 |
| p65-Rv |
CCTGGTCCTGTGTAGCCATT |
|
| ATM-Fw |
TTGATCTTGTGCCTTGGCTAC | 142 |
| ATM-Rv |
TATGGTGTACGTTCCCCATGT |
|
| DEK-BSP-Fw |
GGGATTGTTTATTATTTTTTTTAGGAAG | 388 |
| DEK-BSP-Rv |
CGACTCCCCAAAATCAACAAAAT |
|
Immunohistochemistry
Immunohistochemistry (IHC) was performed on
formalin-fixed paraffin-embedded tumor tissues, which were
sectioned (4 mm) and mounted on polylysine-coated slides. The
sections were deparaffinized in xylene and then rehydrated in
decreasing concentrations of ethanol (100, 95, 80 and 70%,
respectively). Antigen was retrieved by incubating sections with
sodium citrate buffer (pH 6.0) for 10 min in the microwave. Slides
were incubated with 3% (v/v) H2O2 for 15 min
to inhibit endogenous peroxidase activity. To further reduce
non-specific background staining, sections were incubated in 20%
(v/v) normal goat serum for 20 min and were then incubated with the
DEK-specific primary rabbit antibody (dilution 1:300; ProteinTech
Group, Inc., Chicago, IL, USA) at 37°C for 2 h. The Goat anti
Rabbit IgG/HRP polymer conjugate (OriGene Technologies, Inc.,
Rockville, MD, USA) were added to the sections and incubated at
37°C for 15 min according to the manufacturer's protocol. Between
each aforementioned steps, the sections were washed with
phosphate-buffered saline (PBS) for 6 min. Immunostaining was
visualized by developing the slides in diaminobenzidin (DAB). The
sections were counterstained with hematoxylin for 4 sec, dehydrated
and mounted. Each slide was observed under the microscope at
magnification, ×400. Semi-quantitative percentage score was given
according to the nuclear staining of cells from five views.
Percentage scores were assigned as follows: 0–5%: negative (−),
6–25%: weak positive (+), 26–50%: positive (++), 51–100%: strong
positive (+++).
Western blotting analysis
Cells were trypsinized after transfection for 48 h
using 0.25% trypsin (Merck KGaA), then lysed using 1X SDS-PAGE
loading buffer [0.045 mol/l Tris-HCl (pH 6.8); 10% glycerin, 1%
SDS; 0.004% bromophenol blue; 0.5 mol/l DTT; with a final
concentration of 200 µl loading buffer every 1×106
cells]. 30 µl of each sample was loaded and separated in a 12%
SDS-polyacrylamide gel by electrophoresis. Proteins were
transferred onto nitrocellulose filter membrane (NC membrane; Pall
Life Sciences, Port Washington, NY, USA) and incubated in the
following reagents: 5% non-fat milk (4°C, over-night); primary
antibodies (DEK rabbit polyclonal antibody: 1:1,000, ProteinTech
Group, Inc.; β-actin mouse monoclonal antibody: 1:1,000, OriGene
Technologies, Inc.; His mouse monoclonal antibody: 1:500, OriGene
Technologies, Inc.); secondary antibodies (Peroxidase-Conjugated
AffiniPure Goat anti-mouse IgG: 1:2,000, OriGene Technologies,
Inc.; or Peroxidase-Conjugated AffiniPure Goat anti-rabbit IgG:
1:2,000, OriGene Technologies, Inc.; 37°C, 1 h). After each
aforementioned incubation step, the membrane was washed in TBST
buffer for 3 times, 5 min each time. So were the reagent and
antibodies diluted in TBST buffer. After the final washing step,
bound proteins were visualized using ECL (Thermo Fisher Scientific,
Inc.) and X-ray film (Fujifilm, Shanghai, China). The relative
expression level of each gene was analyzed based on β-actin as the
loading control.
MTT assay
A549 cells were planted on 96-well plate before
transfection, with 100 µl of medium, 1,000 cells per well. 10 µl
MTT reagent was added to wells 24, 48, 72 and 96 h after
transfection, and the plate was incubated in 37°C for 4 h. After
discarding the media, 100 µl of DMSO was added to dissolve the MTT
formazan in each well. The plate was incubated in 37°C for another
4 h and the optical density was read using a wavelength of 570 nm.
Each test was performed in triplicates.
Colony formation assay
Cells were replanted in 6-well cell culture dishes
48 h after transfection with 1,000 cells per well. Medium was
renewed every two days. 7 days after transfection, colonies were
stained by 0.1% crystal violet (Beyotime Institute of
Biotechnology, Haimen, China), and visible colonies were counted.
Each test was performed in triplicates.
Matrigel cell invasion assay
48 h after transfection, 10,000 cells were replanted
onto the upper membrane of transwell chamber (EMD Millipore,
Billerica, MA, USA) covered with Matrigel (BD Biosciences, Franklin
Lakes, NJ, USA) according to the manufacturer's protocol. The upper
chamber contained 200 µl of DMEM, and the lower chamber contained
600 µl of DMEM with 10% FBS as chemoattractant. 24 h after
incubation, the chamber was washed with PBS and cells on the upper
membrane were gently removed. Then the membrane was stained with
crystal violet (Beyotime Institute of Biotechnology, Haimen,
China). Number of migrated cells were counted under the microscope
with magnification, ×100, 5 randomly selected areas on each
membrane were counted.
Statistical analysis
All data were analyzed using SPSS version 20.
Immunohistochemistry results were analyzed using the chi-squared
tests, and all other results were analyzed using the unpaired,
two-tailed Student's t-tests. All statistical data were presented
as means ± standard deviation. A test with a P<0.05 was
considered to indicate a statistically significant difference.
Results
Over-expression of DEK mRNA detected
in lung cancerous tissues
Real-time quantitative RT-PCR (qRT-PCR) was
performed in 29 cases of lung cancerous tissues paired with
non-cancerous counterparts. DEK-over expression means that in tumor
samples the DEK is more expressed than in the adjacent normal
tissue samples. As shown in Table
III, DEK mRNA was over-expressed in 15 out of 29 (51.7%) cases
of lung cancerous tissues paired with normal tissues. Among those,
14 cases (48.3%) showed over two-fold up-regulation of DEK mRNA
level, 9 cases (31.0%) showed over five-fold up-regulation of DEK
mRNA level. In adenocarcinoma tissues, 7 out of 8 cases (87.5%)
showed over-expression of DEK mRNA, which was significantly higher
than those of other histopathological types. However, no
correlation between the over-expression of DEK mRNA and patient's
age, gender, pleural invasion status as well as TMN stage was found
(Table IV).
 | Table III.Association between DEK mRNA
expression and tissue position. |
Table III.
Association between DEK mRNA
expression and tissue position.
| Groups compared | Cases | DEK over-expressed
(%) | DEK not
over-expressed (%) |
|---|
| Cancerous/normal
tissue | 29 | 15 (51.7) | 14 (48.3) |
|
Cancerous/precancerous tissue | 30 | 11 (36.7) | 19 (63.3) |
| Precancerous/normal
tissue | 24 | 10 (41.7) | 14 (58.3) |
 | Table IV.Association between DEK mRNA
expression and clinicopathological characteristics. |
Table IV.
Association between DEK mRNA
expression and clinicopathological characteristics.
| Clinicopathological
characteristic | Cases | DEK over-expressed
(%) | DEK not
over-expressed (%) | χ2 | P-value |
|---|
| Total | 29 | 15 (51.7) | 14 (48.3) |
|
|
| Age |
|
|
| 0.181 | 0.671 |
|
≤60 | 12 | 7 (58.3) | 5 (41.7) |
|
|
|
>60 | 14 | 7 (50.0) | 7 (50.0) |
|
|
|
Unknown | 3 | 1 (33.3) | 2 (66.7) |
|
|
| Gender |
|
|
| 0.379 | 0.678 |
|
Male | 19 | 11 (57.9) | 8 (42.1) |
|
|
|
Female | 7 | 3 (42.9) | 4 (57.1) |
|
|
|
Unknown | 3 | 1 (33.3) | 2 (66.7) |
|
|
| Pleural
invasion |
|
|
| 0.422 | 0.516 |
|
Positive | 10 | 6 (60.0) | 4 (40.0) |
|
|
|
Negative | 17 | 8 (47.1) | 9 (52.9) |
|
|
|
Unknown | 2 | 1 (50.0) | 1 (50.0) |
|
|
| Histopathological
type |
|
|
| 8.471 | 0.132 |
|
NSCLC | 22 | 12 (54.5) | 10 (45.5) | 0.049 | >0.999 |
|
Adenocarcinoma | 8 | 7 (87.5) | 1 (12.5) | 4.698 | 0.043a |
|
Squamous carcinoma | 9 | 3 (33.3) | 6 (66.7) |
|
|
|
Adenosquamous carcinoma | 2 | 1 (50.0) | 1 (50.0) |
|
|
|
Sarcomatoid carcinoma | 2 | 0 (0.0) | 2 (100.0) |
|
|
| Large
cell carcinoma | 1 | 1 (100.0) | 0 (0.0) |
|
|
| Small
cell carcinoma | 5 | 3 (60.0) | 2 (40.0) |
|
|
|
Unknown | 2 | 0 (0.0) | 2 (100.0) |
|
|
| TMN |
|
|
| 2.912 | 0.573 |
| Benign
tumor | 1 | 0 (0.0) | 1 (100.0) |
|
|
| I | 10 | 6 (60.0) | 4 (40.0) |
|
|
| II | 7 | 4 (57.1) | 3 (42.9) |
|
|
|
III | 10 | 4 (40.0) | 6 (60.0) |
|
|
| IV | 1 | 1 (100.0) | 0 (0.0) |
|
|
|
Differentiation |
|
|
|
|
|
| Well to
moderately-well | 4 | 4 (100.0) | 0 (0.0) |
|
|
|
Moderate to poor | 3 | 3 (100.0) | 0 (0.0) |
|
|
|
Unknown | 1 | 0 (0.0) | 1 (100.0) |
|
|
Over-expression of DEK protein
detected in lung cancerous tissues
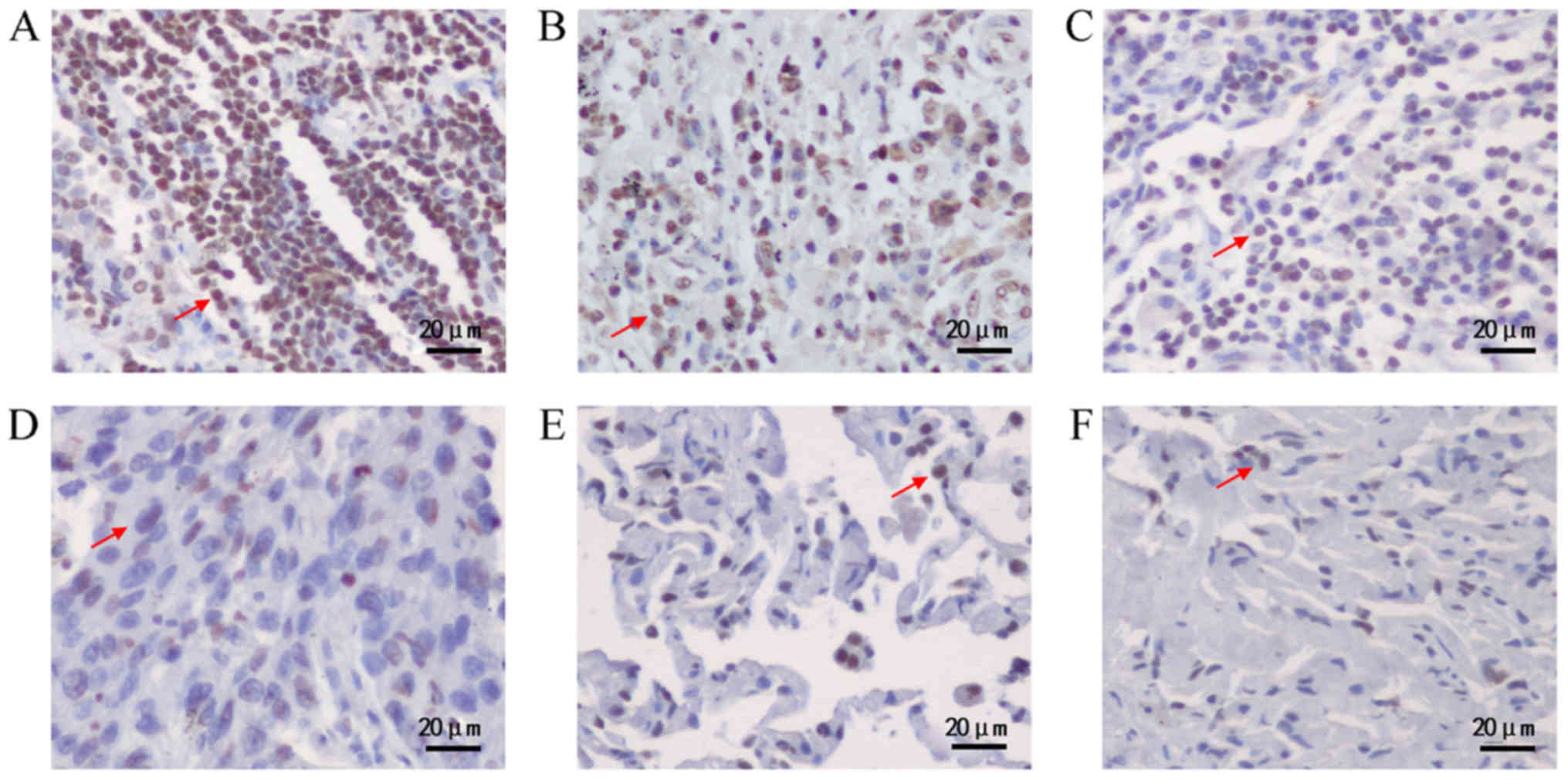
The expression level of DEK protein in lung
cancerous tissues and non-cancerous counterparts were detected by
IHC (Fig. 1). Among 81 cases of lung
cancerous tissues, 66 cases (81.5%) showed positive staining of DEK
protein, while 32 cases (39.5%) showed strong positive staining of
DEK protein, which was significantly higher than those in normal
tissues (Table V). However, there was
no significant correlation between strong positive staining of DEK
protein and clinicopathological characteristics of patients
(Table VI).
 | Table V.Association between DEK protein
expression and tissue position according to immunohistochemistry
results. |
Table V.
Association between DEK protein
expression and tissue position according to immunohistochemistry
results.
| Sample | Cases | − | + | ++ | +++ | Positive rate
(%) | Strong positive
rate (%) |
|---|
| C | 81 | 15 | 34 | 21 | 11 | 81.5 | 39.5a |
| P | 81 | 13 | 45 | 21 | 2 | 84.0 | 28.4 |
| N | 73 | 23 | 35 | 13 | 2 | 68.5 | 20.5 |
 | Table VI.Association between DEK protein
expression and clinicopathological characteristics. |
Table VI.
Association between DEK protein
expression and clinicopathological characteristics.
| Clinicopathological
characteristic | Cases | Strong positive
staining of DEK in cancerous tissues, +++ or ++ (%) | Weak or negative
staining of DEK in cancerous tissues, + or - (%) | χ2 | P-value |
|---|
| Total | 81 | 32 (39.5) | 49 (60.5) |
|
|
| Age |
|
|
| 2.339 | 0.126 |
|
≤60 | 40 | 14 (35.0) | 26 (65.0) |
|
|
|
>60 | 30 | 16 (53.3) | 14 (46.7) |
|
|
|
Unknown | 11 | 2 (18.2) | 9 (81.8) |
|
|
| Gender |
|
|
| 2.500 | 0.114 |
|
Male | 49 | 24 (49.0) | 25 (51.0) |
|
|
|
Female | 21 | 6 (28.6) | 15 (71.4) |
|
|
|
Unknown | 11 | 2 (18.2) | 9 (81.8) |
|
|
| Pleural
invasion |
|
|
| 0.002 | 0.968 |
|
Positive | 16 | 10 (62.5) | 6 (37.5) |
|
|
|
Negative | 19 | 12 (63.2) | 7 (36.8) |
|
|
|
Unknown | 46 | 10 (21.7) | 36 (78.3) |
|
|
| Histopathological
type |
|
|
| 5.344 | 0.375 |
|
NSCLC | 61 | 25 (41.0) | 36 (59.0) | 0.039 | >0.999 |
|
Adenocarcinoma | 24 | 7 (29.2) | 17 (70.8) |
|
|
|
Squamous carcinoma | 24 | 11 (45.8) | 13 (54.2) |
|
|
|
Adenosquamous carcinoma | 4 | 2 (50.0) | 2 (50.0) |
|
|
|
Sarcomatoid carcinoma | 5 | 4 (80.0) | 1 (20.0) |
|
|
| Large
cell carcinoma | 4 | 1 (25.0) | 3 (75.0) |
|
|
| Small
cell carcinoma | 9 | 4 (44.4) | 5 (55.6) |
|
|
|
Unknown | 11 | 3 (27.3) | 8 (72.7) |
|
|
| TMN |
|
|
| 2.167 | 0.705 |
| Benign
tumor | 1 | 1 (100.0) | 0 (0.0) |
|
|
| I | 19 | 8 (42.1) | 11 (57.9) |
|
|
| II | 12 | 6 (50.0) | 6 (50.0) |
|
|
|
III | 25 | 14 (56.0) | 11 (44.0) |
|
|
| IV | 3 | 1 (33.3) | 2 (66.7) |
|
|
|
Unknown | 21 | 2 (9.5) | 19 (90.5) |
|
|
|
Differentiation |
|
|
| 0.198 | 0.673 |
| Well to
moderately-well | 9 | 3 (33.3) | 6 (66.7) |
|
|
|
Moderate to poor | 16 | 4 (25.0) | 12 (75.0) |
|
|
|
Unknown | 56 | 25 (44.6) | 31 (55.4) |
|
|
DEK depletion and over-expression in
A549 lung cancer cells
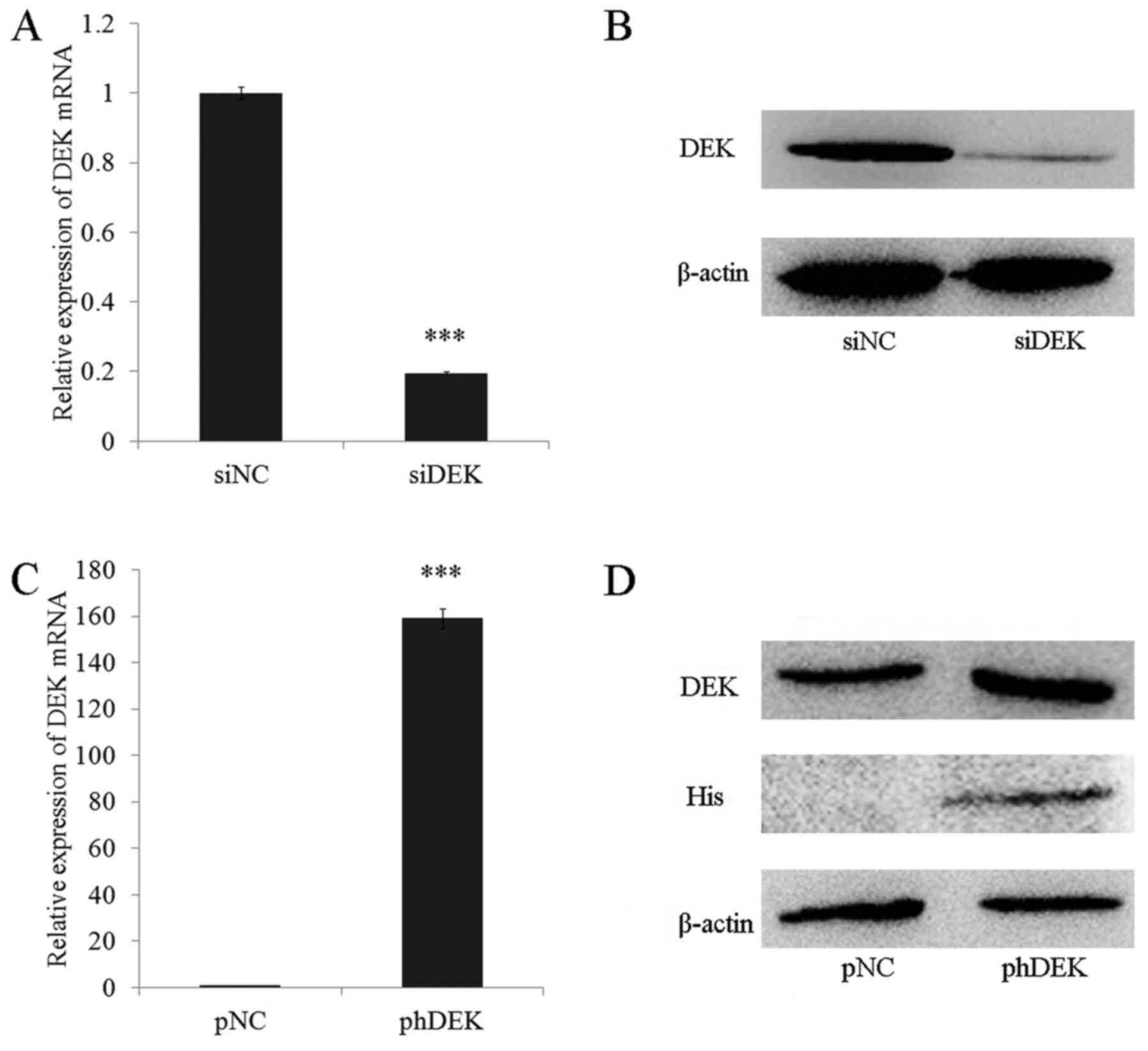
To further investigate the function of DEK in lung
cancer cells, siRNA as well as plasmid encoding His-tagged DEK CDS
were employed respectively to down-regulate and up-regulate the
expression level of DEK in A549 lung cancer cell line. The
expression level of DEK mRNA and protein were analyzed by qRT-PCR
and Western Blotting (WB). Results showed the expression level of
DEK mRNA and protein were down-regulated by siRNA transfection;
while the transfection of plasmid encoding His-tagged DEK caused
DEK over-expression in A549 cells (Fig.
2).
DEK positively regulates proliferation
in A549 cells
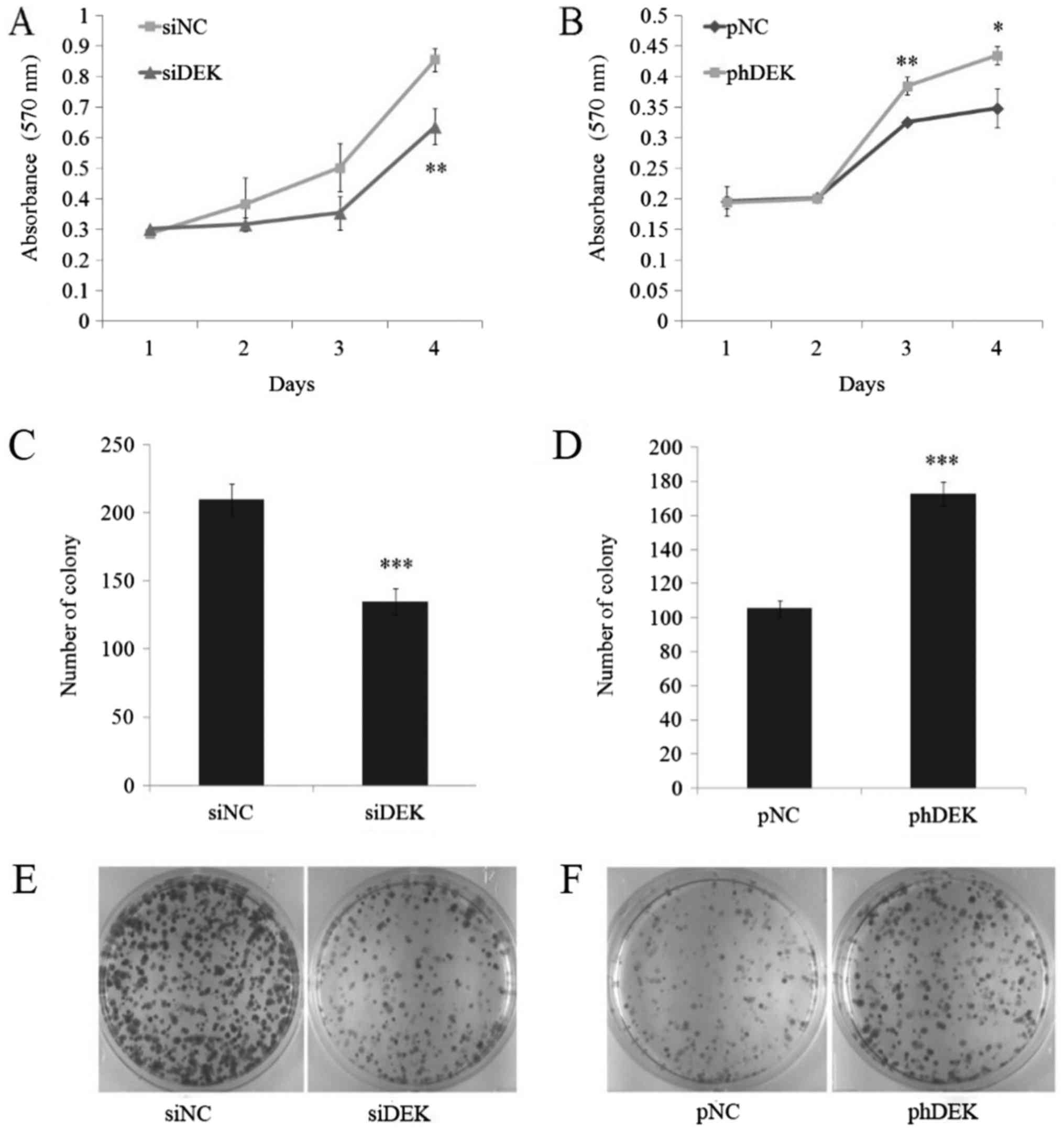
To detect whether DEK could regulate cell
proliferation in lung cancer cell lines, MTT assay was performed in
DEK depleted and over-expressed A549 cells. Results showed that
cell proliferation was significantly restricted after day 4 in DEK
depleted cells, while being significantly promoted after day 3 and
day 4 in DEK over-expressed cells (Fig.
3A and B). A supplementary experiment of colony formation assay
was performed and the results were in accordance with the former
one. A significant decrease of colony numbers in DEK depleted cells
and a significant increase of colony numbers in DEK over-expressed
cells were detected (Fig. 3C-F). Both
experiments manifested the positive correlation between DEK
expressional level and cell proliferation, showing DEK may act as a
pro-proliferation factor in A549 cells. As a chromatin associated
architectural protein, DEK may play an important role in change of
chromatin structure and thereby the expression of genes related to
cell proliferation.
DEK positively regulates invasion of
A549 cells
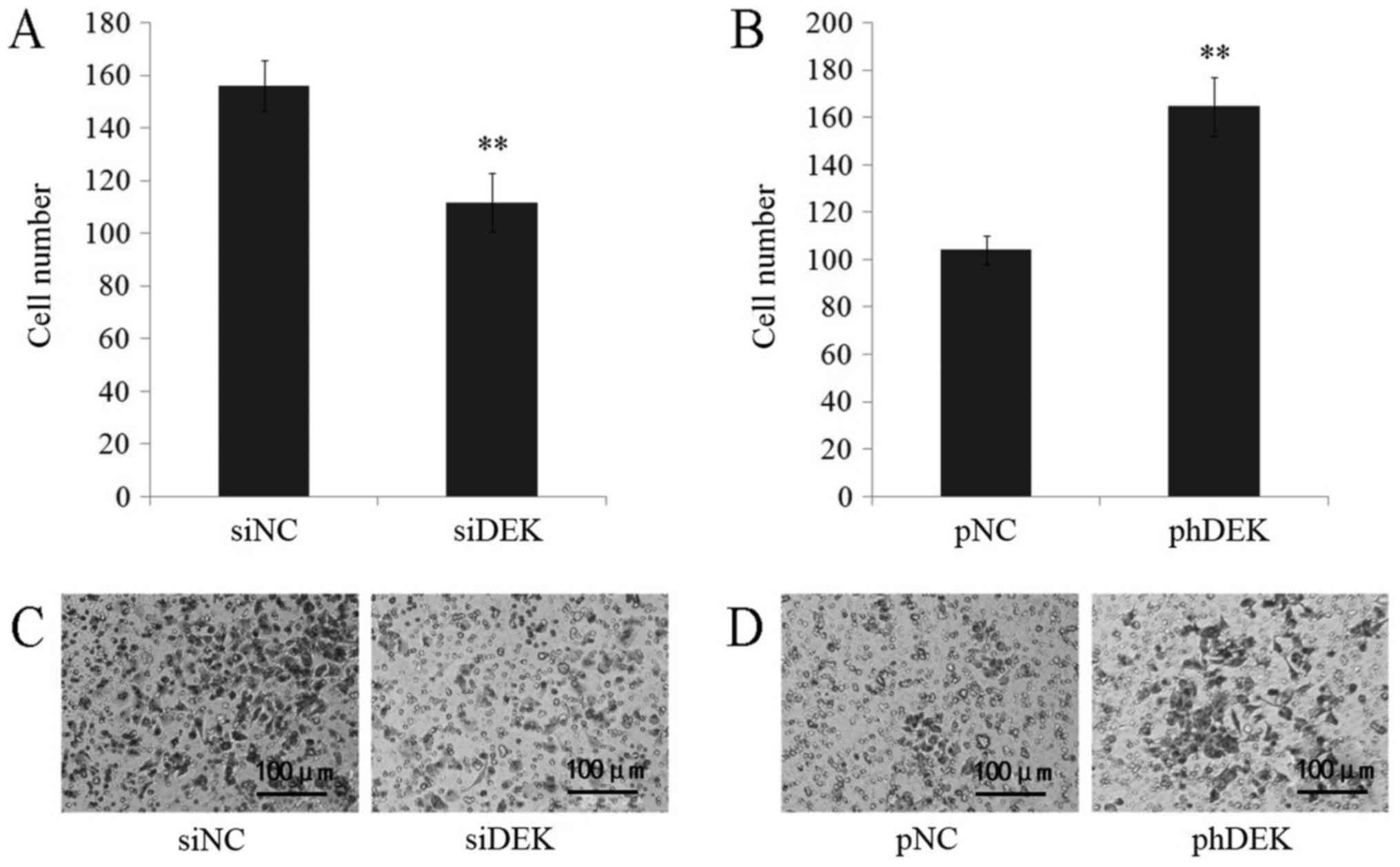
Matrigel invasion assay was performed to detect
alterations DEK made to cell invasion. Decrease of invaded cell
numbers was shown in DEK depleted A549 cells, while DEK
over-expressed cells resulted in an increase of invaded cell
numbers (Fig. 4). According to the
results, the invasion of A549 cells was positively correlated with
DEK expression.
Discussion
DEK is over-expressed in various type of malignant
carcinomas, such as acute myeloid leukemia, retinoblastoma, brain
malignant glioma, hepatocellular carcinoma, breast cancer, uterine
cervical cancer, bladder cancer, colon cancer, colorectal cancer,
gastric adenocarcinoma, oral squamous cell carcinoma,
neuroendocrine prostate cancer (5–8,10–17,27). It
was first discovered that DEK was over-expressed in high-grade
neuroendocrine carcinoma of the lung by Shibata et al
(9), yet it was only limited on the
type of neuroendocrine carcinoma. Here we examined the expression
level of DEK in various subtypes of lung cancer, and discovered
that DEK mRNA is over-expressed in 51.7% of lung cancerous tissues.
Especially, in adenocarcinoma tissues of the lung, DEK showed a
significantly high over-expression rate of 87.5%. The IHC
experiment showed similar results, 39.5% of lung cancerous tissues
showed strong positive expression of DEK protein, while the number
was only 20.5% in normal tissues. These findings gave valuable
hints to the biomarker development of DEK, showing that DEK might
have the potential to be applied as a diagnostic biomarker for lung
cancers, especially for adenocarcinoma of the lung. However,
neither of these studies demonstrated the relationship between DEK
expression and patient's age, gender, or TMN stage, differentiation
status of the carcinoma.
We have analyzed the expression of DEK in mRNA and
protein level in lung cancer samples, not only the NSCLC samples
reported in the study by Liu et al (28), which explored the role of DEK
expression for the prognostic evaluation of non-small cell lung
carcinoma (NSCLC). Besides, based on the previous findings, the
downstream effects of aberrant DEK expressional level were analyzed
using the A549 lung cancer cell line. Through silence and
overexpression of DEK in A549 cells, we found that DEK could
influence the proliferation and invasion of A549 tumor cells. In
our study, it was discovered that the over-expression of DEK
promotes cell proliferation and invasion, which was in accordance
with former studies in other types of cancers (3,8,17,19). The
findings support the carcinogenic role of DEK in lung cancer
formation, and it could be the reason for the aberrant expressional
level of DEK in lung cancerous tissues we detected earlier. On the
other hand, depletion of DEK in A549 cells gave us the exact
opposite story. Our study showed that DEK depletion could repress
proliferation and invasion of lung tumor cells. Therefore, DEK
could act as a target for lung tumor therapy applied in clinical
studies. In our study, the depletion of the gene expression was
accomplished by siRNA transfection, which made the therapy easier
to be applied clinically.
To sum up, the present study discovered that DEK was
over-expressed in lung cancerous tissues. The aberrant expression
of DEK may promote cell survival and tumorigenesis. The more
detailed discussion of DEK's mechanism in lung cancer formation
should be done in the future studies, and clinical studies using
larger groups of samples should be performed to discover the
further application value of DEK as a novel biomarker.
Acknowledgements
Not applicable.
Funding
This study has been funded by the National Natural
Science Foundation of China (grant no. 81201762).
Availability of data and material
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Author's contributions
QCZ, JY, MXQ, HHL and ZQ performed the experiments
and analyzed the data. XFD contributed to the collection of human
lung tissue samples and RNA extraction. LLH, HJ and HGH conceived
and designed the study. QCZ and JY drafting the manuscript, which
was critically revised by LLH and HGH.
Ethics approval and consent to
participate
The present study was approved by the Beijing
Jiaotong University Institutional Review Board. Written informed
consent was obtained from all patients prior to their
inclusion.
Consent for publication
Informed consent was obtained from all patients' for
the publication of their data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Waldmann T, Scholten I, Kappes F, Hu HG
and Knippers R: The DEK protein-an abundant and ubiquitous
constituent of mammalian chromatin. Gene. 343:1–9. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
von Lindern M, Breems D, van Baal S,
Adriaansen H and Grosveld G: Characterization of the translocation
breakpoint sequences of two DEK-CAN fusion genes present in t(6;9)
acute myeloid leukemia and a SET-CAN fusion gene found in a case of
acute undifferentiated leukemia. Genes Chromosomes Cancer.
5:227–234. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Vinnedge Privette LM, Kappes F, Nassar N
and Wells SI: Stacking the DEK: From chromatin topology to cancer
stem cells. Cell Cycle. 12:51–66. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Grottke C, Mantwill K, Dietel M,
Schadendorf D and Lage H: Identification of differentially
expressed genes in human melanoma cells with acquired resistance to
various antineoplastic drugs. Int J Cancer. 88:535–546. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang X, Huang SH, Yu LQ, Liu J, Diao Y and
Sun CF: Relationship between DEK oncogene expression and oral
squamous cell carcinoma. Shanghai Kou Qiang Yi Xue. 23:75–79.
2014.(In Chinese). PubMed/NCBI
|
|
6
|
Piao J, Shang Y, Liu S, Piao Y, Cui X, Li
Y and Lin Z: High expression of DEK predicts poor prognosis of
gastric adenocarcinoma. Diagn Pathol. 9:672014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lin L, Piao J, Gao W, Piao Y, Jin G, Ma Y,
Li J and Lin Z: DEK over expression as an independent biomarker for
poor prognosis in colorectal cancer. BMC Cancer. 13:3662013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Vinnedge Privette LM, McClaine R, Wagh PK,
Wikenheiser-Brokamp KA, Waltz SE and Wells SI: The human DEK
oncogene stimulates β-catenin signaling, invasion and mammosphere
formation in breast cancer. Oncogene. 30:2741–2752. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shibata T, Kokubu A, Miyamoto M, Hosoda F,
Gotoh M, Tsuta K, Asamura H, Matsuno Y, Kondo T, Imoto I, et al:
DEK oncoprotein regulates transcriptional modifiers and sustains
tumor initiation activity in high-grade neuroendocrine carcinoma of
the lung. Oncogene. 29:4671–4681. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wu Q, Li Z, Lin H, Han L, Liu S and Lin Z:
DEK overexpression in uterine cervical cancers. Pathol Int.
58:378–382. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Orlic M, Spencer CE, Wang L and Gallie BL:
Expression analysis of 6p22 genomic gain in retinoblastoma. Genes
Chromosomes Cancer. 45:72–82. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Carro MS, Spiga FM, Quarto M, Di Ninni V,
Volorio S, Alcalay M and Müller H: DEK Expression is controlled by
E2F and deregulated in diverse tumor types. Cell Cycle.
5:1202–1207. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sanchez-Carbayo M, Socci ND, Lozano JJ, Li
W, Charytonowicz E, Belbin TJ, Prystowsky MB, Ortiz AR, Childs G
and Cordon-Cardo C: Gene discovery in bladder cancer progression
using cDNA microarrays. Am J Pathol. 163:505–516. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Larramendy ML, Niini T, Elonen E, Nagy B,
Ollila J, Vihinen M and Knuutila S: Overexpression of
translocation-associated fusion genes of FGFRI, MYC, NPMI, and DEK,
but absence of the translocations in acute myeloid leukemia. A
microarray analysis. Haematologica. 87:569–577. 2002.PubMed/NCBI
|
|
15
|
Kroes RA, Jastrow A, McLone MG, Yamamoto
H, Colley P, Kersey DS, Yong VW, Mkrdichian E, Cerullo L, Leestma J
and Moskal JR: The identification of novel therapeutic targets for
the treatment of malignant brain tumors. Cancer Lett. 156:191–198.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kondoh N, Wakatsuki T, Ryo A, Hada A,
Aihara T, Horiuchi S, Goseki N, Matsubara O, Takenaka K, Shichita
M, et al: Identification and characterization of genes associated
with human hepatocellular carcinogenesis. Cancer Res. 59:4990–4996.
1999.PubMed/NCBI
|
|
17
|
Wise-Draper TM, Mintz-Cole RA, Morris TA,
Simpson DS, Wikenheiser-Brokamp KA, Currier MA, Cripe TP, Grosveld
GC and Wells SI: Overexpression of the cellular DEK protein
promotes epithelial transformation in vitro and in vivo. Cancer
Res. 69:1792–1799. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wise-Draper TM, Allen HV, Thobe MN, Jones
EE, Habash KB, Münger K and Wells SI: The human DEK proto-oncogene
is a senescence inhibitor and an upregulated target of high-risk
human papillomavirus E7. J Virol. 79:14309–14317. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wise-Draper TM, Allen HV, Jones EE, Habash
KB, Matsuo H and Wells SI: Apoptosis inhibition by the human DEK
oncoprotein involves interference with p53 functions. Mol Cell
Biol. 26:7506–7519. 2016. View Article : Google Scholar
|
|
20
|
Liu K, Feng T, Liu J, Zhong M and Zhang S:
Silencing of the DEK gene induces apoptosis and senescence in CaSki
cervical carcinoma cells via the up-regulation of NF-κB p65. Biosci
Rep. 32:323–332. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kim DW, Chae JI, Kim JY, Pak JH, Koo DB,
Bahk YY and Seo SB: Proteomic analysis of apoptosis related
proteins regulated by proto-oncogene protein DEK. J Cell Biochem.
106:1048–1059. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Waldmann T, Eckerich C, Baack M and Gruss
C: The ubiquitous chromatin protein DEK alters the structure of DNA
by introducing positive supercoils. J Biol Chem. 277:24988–24994.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kavanaugh GM, Wise-Draper TM, Morreale RJ,
Morrison MA, Gole B, Schwemberger S, Tichy ED, Lu L, Babcock GF,
Wells JM, et al: The human DEK oncogene regulates DNA damage
response signaling and repair. Nucleic Acids Res. 39:7465–7476.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kappes F, Fahrer J, Khodadoust MS, Tabbert
A, Strasser C, Mor-Vaknin N, Moreno-Villanueva M, Bürkle A,
Markovitz DM and Ferrando-May E: DEK is a poly(ADP-ribose) acceptor
in apoptosis and mediates resistance to genotoxic stress. Mol Cell
Biol. 28:3245–3257. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Khodadoust MS, Verhaegen M, Kappes F,
Riveiro-Falkenbach E, Cigudosa JC, Kim DS, Chinnaiyan AM, Markovitz
DM and Soengas MS: Melanoma proliferation and chemoresistance
controlled by the DEK oncogene. Cancer Res. 69:6405–6413. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Martinez-Useros J, Rodriguez-Remirez M,
Borrero-Palacios A, Moreno I, Cebrian A, del Pulgar Gomez T, del
Puerto-Nevado L, Vega-Bravo R, Puime-Otin A, Perez N, et al: DEK is
a potential marker for aggressive phenotype and irinotecan-based
therapy response in metastatic colorectal cancer. BMC Cancer.
14:9652014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lin D, Dong X, Wang K, Wyatt AW, Crea F,
Xue H, Wang Y, Wu R, Bell RH, Haegert A, et al: Identification of
DEK as a potential therapeutic target for neuroendocrine prostate
cancer. Oncotarget. 6:1806–1820. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu X, Qi D, Qi J, Mao Z, Li X, Zhang J,
Li J and Gao W: Significance of DEK overexpression for the
prognostic evaluation of non-small cell lung carcinoma. Oncol Rep.
35:155–162. 2016. View Article : Google Scholar : PubMed/NCBI
|